Fracture Resistance of Zirconia Abutments with or without a Titanium Base: An In Vitro Study for Tapered Conical Connection Implants
Abstract
1. Introduction
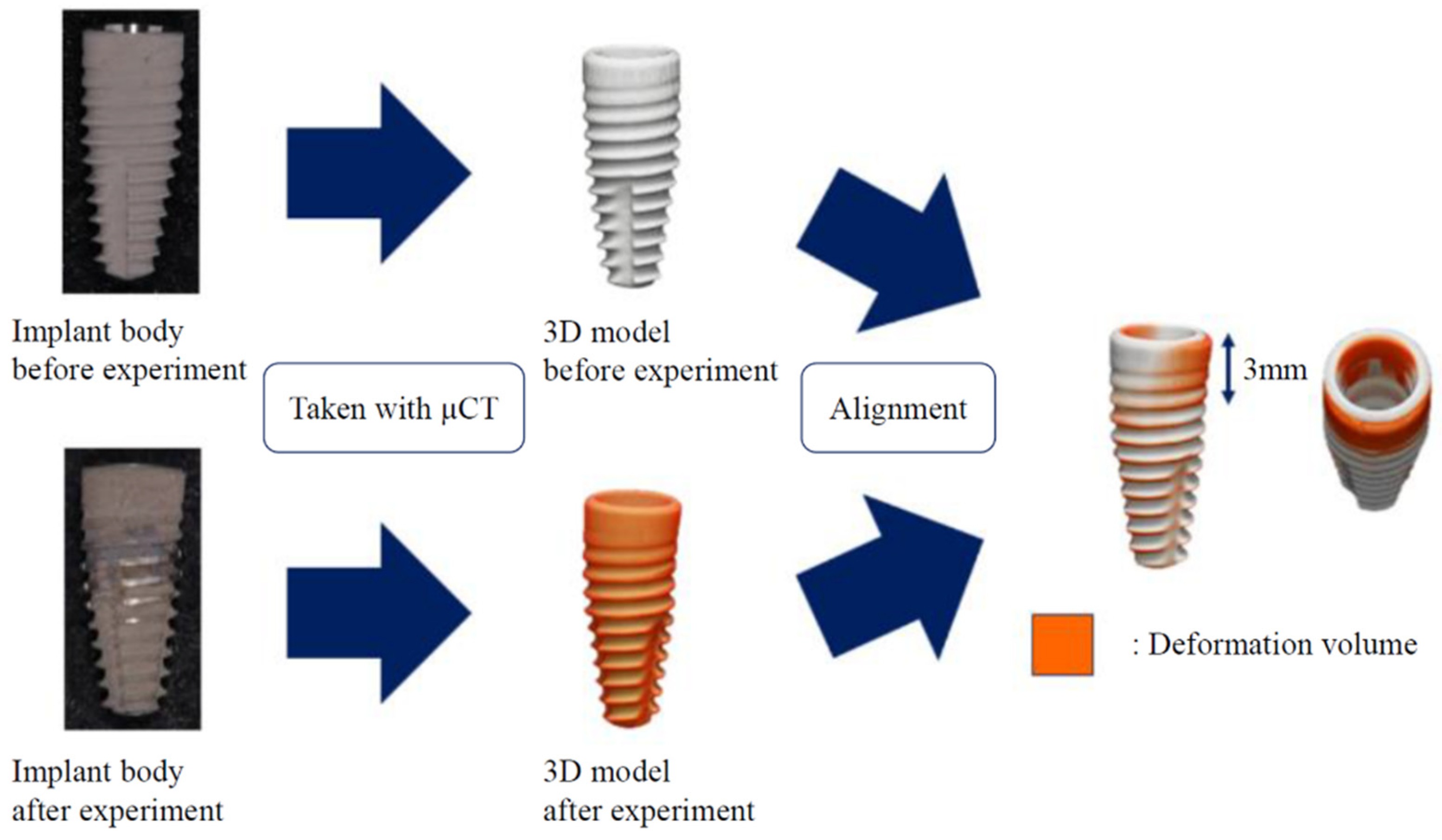
2. Materials and Methods
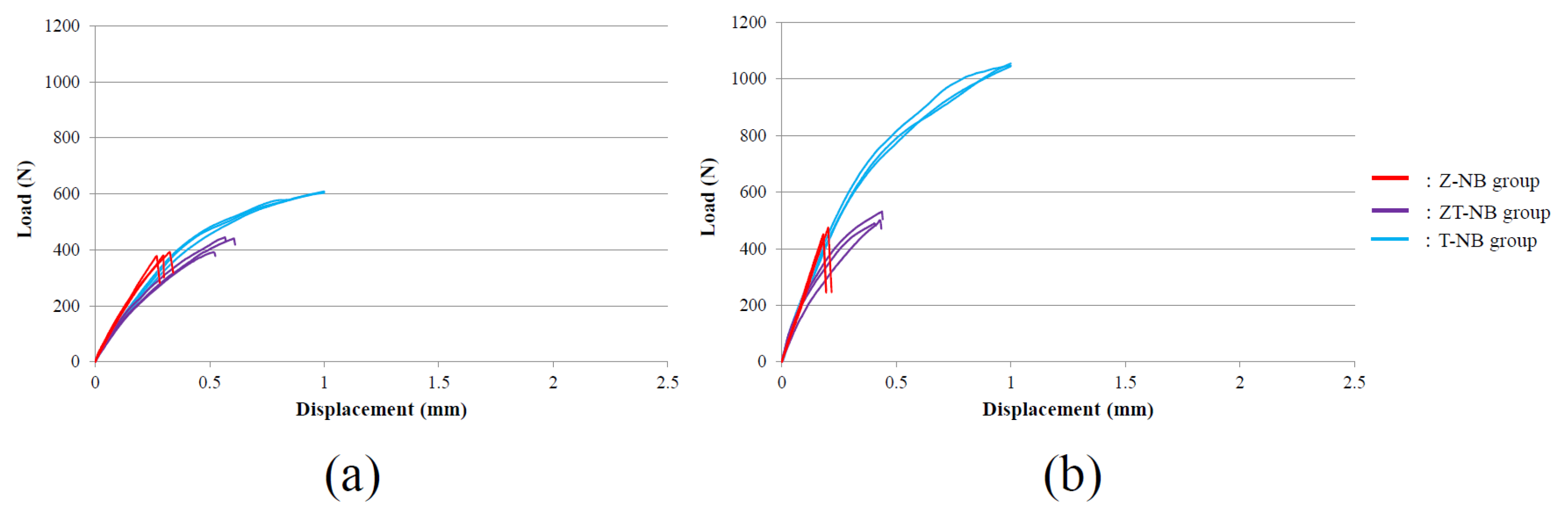
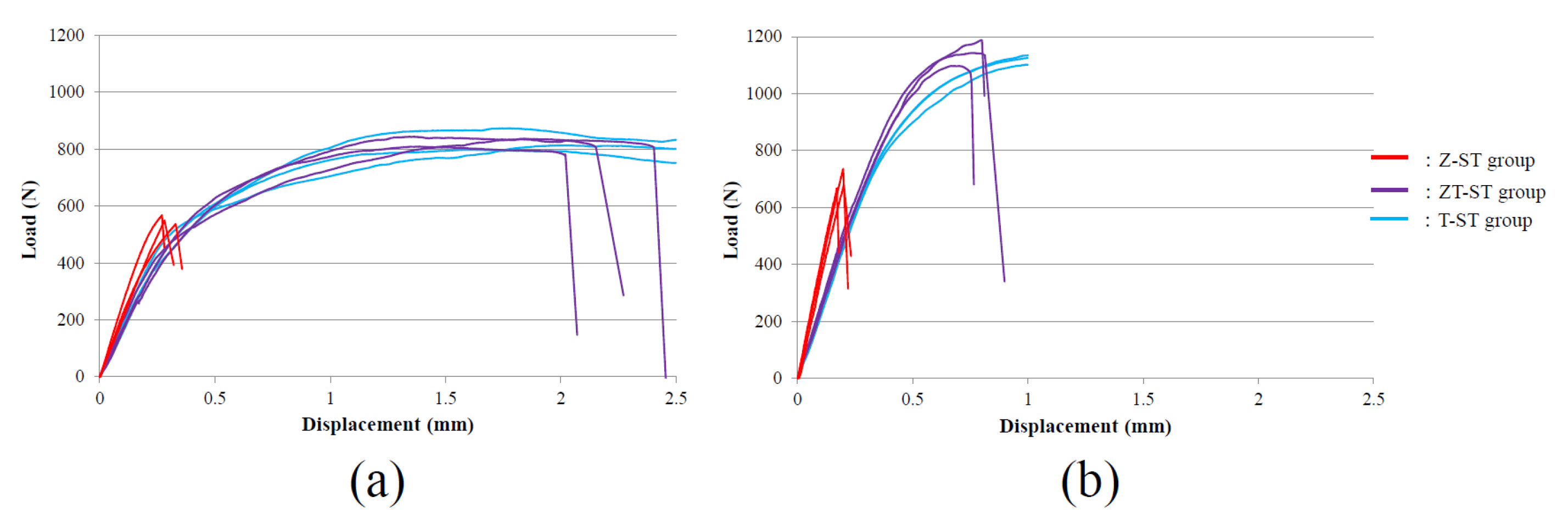
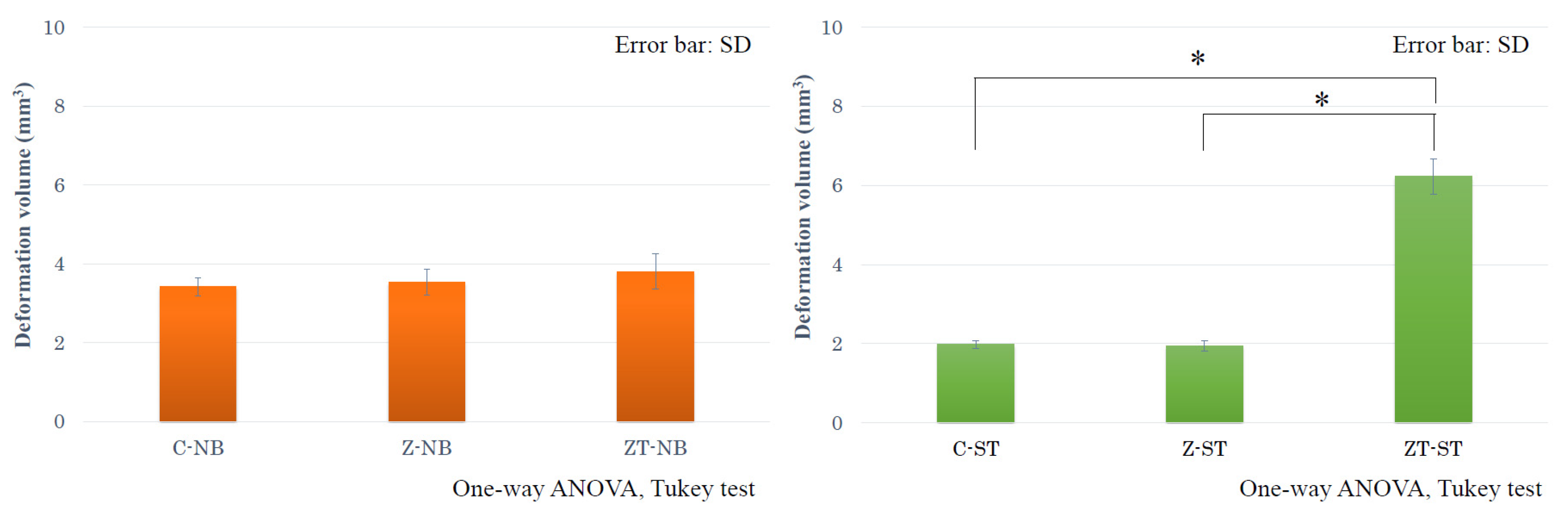
3. Results
4. Discussion
5. Conclusions
- The presence of a titanium base could increase the fracture resistance of zirconia abutments.
- The titanium base abutment showed higher fracture resistance than the full zirconia abutment but might damage the implant body.
- The full zirconia abutment showed lower fracture resistance than the titanium base abutment but is less likely to damage the implant body.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Nakamura, K.; Kanno, T.; Milleding, P.; Ortengren, U. Zirconia as a dental implant abutment material. Int. J. Prosthodont. 2010, 23, 299–309. [Google Scholar]
- Kim, J.S.; Raigrodski, A.J.; Flinn, B.D.; Rubenstein, J.E.; Chung, K.H.; Mancl, L.A. In vitro assessment of three types of zirconia implant abutments under static load. J. Prosthet. Dent. 2013, 109, 255–263. [Google Scholar] [CrossRef]
- Gracis, S.; Michalakis, K.; Vigolo, P.; Vult von Steyern, P.; Zwahlen, M.; Sailer, I. Internal vs. external connections for abutments/reconstructions: A systematic review. Clin. Oral Implants Res. 2012, 23, 202–216. [Google Scholar] [CrossRef]
- Jarman, J.; Hamalian, T.; Randi, A. Comparing the fracture resistance of alternatively engineered zirconia abutments with original equipment manufactured abutments with different implant connection designs. Int. J. Oral Maxillofac. Implants 2017, 32, 992–1000. [Google Scholar] [CrossRef][Green Version]
- Chun, H.J.; Yeo, I.S.; Lee, J.H.; Kim, S.K.; Heo, S.J.; Koak, J.Y.; Han, J.S.; Lee, S.J. Fracture strength study of internally connected zirconia abutments reinforced with titanium inserts. Int. J. Oral Maxillofac. Implants 2015, 30, 346–350. [Google Scholar] [CrossRef]
- Sailer, I.; Sailer, T.; Stawarczyk, B.; Jung, R.E.; Hämmerle, C.H.F. In vitro study of the influence of the type of connection on the fracture load of zirconia abutments with internal and external implant-abutment connections. Int. J. Oral Maxillofac. Implants 2009, 24, 850–858. [Google Scholar]
- Mühlemann, S.; Truninger, T.C.; Stawarczyk, B.; Hämmerle, C.H.F.; Sailer, I. Bending moments of zirconia and titanium implant abutments supporting all-ceramic crowns after aging. Clin. Oral Implants Res. 2014, 25, 74–81. [Google Scholar] [CrossRef]
- Yilmaz, B.; Salaita, L.G.; Seidt, J.D.; Clelland, N.L.; McGlumphy, E.A. Load to failure of different zirconia abutments for an internal hexagon implant. J. Prosthet. Dent. 2015, 114, 373–377. [Google Scholar] [CrossRef]
- Kelly, J.R.; Rungruanganunt, P. Fatigue behavior of computer-aided design/ computer-assisted manufacture ceramic abutments as a function of design and ceramics processing. Int. J. Oral Maxillofac. Implants 2016, 31, 601–609. [Google Scholar] [CrossRef]
- Sailer, I.; Asgeirsson, A.G.; Thoma, D.S.; Fehmer, V.; Aspelund, T.; Özcan, M.; Pjetursson, B.E. Fracture strength of zirconia implant abutments on narrow diameter implants with internal and external implant abutment connections: A study on the titanium resin base concept. Clin. Oral Implants Res. 2018, 29, 411–423. [Google Scholar] [CrossRef]
- Stimmelmayr, M.; Edelhoff, D.; Güth, J.F.; Erdelt, K.; Happe, A.; Beuer, F. Wear at the titanium-titanium and the titanium-zirconia implant-abutment interface: A comparative in vitro study. Dent. Mater. 2012, 28, 1215–1220. [Google Scholar] [CrossRef]
- Nam, R.K.; Lee, S.J.; Park, E.J.; Kwon, H.B.; Yoon, H.I. Three-dimensional deformation and wear of internal implant-abutment connection: A comparative biomechanical study using titanium and zirconia. Int. J. Oral Maxillofac. Implants 2018, 33, 1279–1286. [Google Scholar] [CrossRef]
- Zeynep, Ö.; Ender, K. Zirconia dental implants: A literature review. J. Oral Implantol. 2011, 37, 367–376. [Google Scholar]
- Apratim, A.; Eachempati, P.; Salian, K.; Singh, V.; Chhabra, S.; Shah, S. Zirconia in dental implantology: A review. J. Int. Soc. Prev. Community Dent. 2015, 5, 147–156. [Google Scholar] [CrossRef]
- Fabbri, G.; Fradeani, M.; Dellificorelli, G.; De Lorenzi, M.; Zarone, F.; Sorrentino, R. Clinical evaluation of the influence of connection type and restoration height on the reliability of zirconia abutments: A retrospective study on 965 abutments with a mean 6-year follow-up. Int. J. Periodontics Restorative Dent. 2017, 37, 19–31. [Google Scholar] [CrossRef]
- Annibali, S.; Bignozzi, I.; Cristalli, M.P.; Graziani, F.; La Monaca, G.; Polimeni, A. Peri-implant marginal bone level: A systematic review and meta-analysis of studies comparing platform switching versus conventionally restored implants. J. Clin. Periodontol. 2012, 39, 1097–1113. [Google Scholar] [CrossRef]
- Monje, A.; Pommer, B. The concept of platform switching to preserve peri-implant bone level: Assessment of methodologic quality of systematic reviews. Int. J. Oral Maxillofac. Implants 2015, 30, 1084–1092. [Google Scholar] [CrossRef][Green Version]
- Kaminaka, A.; Nakano, T.; Ono, S.; Kato, T.; Yatani, H. Cone-beam computed tomograph evaluation of horizontal and vertical dimensional changes in buccal peri-implant alveolar bone and soft tissue: A 1-year prospective clinical study. Clin. Implant Dent. Relat. Res. 2015, 17, 576–585. [Google Scholar] [CrossRef]
- International Organization for Standardization. Dentistry-implants-dynamic loading test for endosseous dental implants. In ISO 14801:2016; iTeh, Inc.: Newark, NJ, USA, 2016. [Google Scholar]
- Nguyen, H.Q.; Tan, K.B.; Nicholls, J.I. Load fatigue performance of implant-ceramic abutment combinations. Int. J. Oral Maxillofac. Implants 2009, 24, 636–646. [Google Scholar]
- Zembic, A.; Sailer, I.; Jung, R.E.; Hämmerle, C.H. Randomized-controlled clinical trial of customized zirconia and titanium implant abutments for single-tooth implants in canine and posterior regions: 3-year results. Clin. Oral Implants Res. 2009, 20, 802–808. [Google Scholar] [CrossRef]
- Jung, R.E.; Zembic, A.; Pjetursson, B.E.; Zwahlen, M.; Thoma, D.S. Systematic review of the survival rate and the incidence of biological, technical, and aesthetic complications of single crowns on implants reported in longitudinal studies with a mean follow-up of 5 years. Clin. Oral Implants Res. 2012, 23, 2–21. [Google Scholar] [CrossRef]
- Rosentritt, M.; Hagemann, A.; Hahnel, S.; Behr, M.; Preis, V. In vitro performance of zirconia and titanium implant/abutment systems for anterior application. J. Dent. 2014, 42, 1019–1026. [Google Scholar] [CrossRef]
- Sailer, I.; Philipp, A.; Zembic, A.; Pjetursson, B.E.; Hämmerle, C.H.F.; Zwahlen, M. A systematic review of the performance of ceramic and metal implant abutments supporting fixed implant reconstructions. Clin. Oral Implants Res. 2009, 20, 4–31. [Google Scholar] [CrossRef]
- Nilsson, A.; Johansson, L.Å.; Lindh, C.; Ekfeldt, A. One-piece internal zirconia abutments for single-tooth restorations on narrow and regular diameter implants: A 5-year prospective follow-up study. Clin. Implant Dent Relat. Res. 2017, 19, 916–925. [Google Scholar] [CrossRef]
- Apicella, D.; Veltri, M.; Balleri, P.; Apicella, A.; Ferrari, M. Influence of abutment material on the fracture strength and failure modes of abutment-fixture assemblies when loaded in a bio-faithful simulation. Clin. Oral Implants Res. 2011, 22, 182–188. [Google Scholar] [CrossRef]
- Mascarenhas, F.; Yilmaz, B.; McGlumphy, E.; Clelland, N.; Seidt, J. Load to failure of different zirconia implant abutments with titanium components. J. Prosthet. Dent. 2017, 117, 749–754. [Google Scholar] [CrossRef]
- Blanco, J.; Pico, A.; Caneiro, L.; Nóvoa, L.; Batalla, P.; Martín-Lancharro, P. Effect of abutment height on interproximal implant bone level in the early healing: A randomized clinical trial. Clin. Oral Implants Res. 2018, 29, 108–117. [Google Scholar] [CrossRef]
- Spinato, S.; Galindo-Moreno, P.; Bernardello, F.; Zaffe, D. Minimum abutment height to eliminate bone loss: Influence of implant neck design and platform switching. Int. J. Oral Maxillofac. Implants 2018, 33, 405–411. [Google Scholar] [CrossRef]
- Spinato, S.; Stacchi, C.; Lombardi, T.; Bernardello, F.; Messina, M.; Zaffe, D. Biological width establishment around dental implants is influenced by abutment height irrespective of vertical mucosal thickness: A cluster randomized controlled trial. Clin. Oral Implants Res. 2019, 30, 649–659. [Google Scholar] [CrossRef]
- Ferrario, V.F.; Sforza, C.; Serrao, G.; Dellavia, C.; Tartaglia, G.M. Single tooth bite forces in healthy young adults. J. Oral Rehabil. 2004, 31, 18–22. [Google Scholar] [CrossRef]







Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Watanabe, S.; Nakano, T.; Ono, S.; Yamanishi, Y.; Matsuoka, T.; Ishigaki, S. Fracture Resistance of Zirconia Abutments with or without a Titanium Base: An In Vitro Study for Tapered Conical Connection Implants. Materials 2022, 15, 364. https://doi.org/10.3390/ma15010364
Watanabe S, Nakano T, Ono S, Yamanishi Y, Matsuoka T, Ishigaki S. Fracture Resistance of Zirconia Abutments with or without a Titanium Base: An In Vitro Study for Tapered Conical Connection Implants. Materials. 2022; 15(1):364. https://doi.org/10.3390/ma15010364
Chicago/Turabian StyleWatanabe, Shota, Tamaki Nakano, Shinji Ono, Yasufumi Yamanishi, Takashi Matsuoka, and Shoichi Ishigaki. 2022. "Fracture Resistance of Zirconia Abutments with or without a Titanium Base: An In Vitro Study for Tapered Conical Connection Implants" Materials 15, no. 1: 364. https://doi.org/10.3390/ma15010364
APA StyleWatanabe, S., Nakano, T., Ono, S., Yamanishi, Y., Matsuoka, T., & Ishigaki, S. (2022). Fracture Resistance of Zirconia Abutments with or without a Titanium Base: An In Vitro Study for Tapered Conical Connection Implants. Materials, 15(1), 364. https://doi.org/10.3390/ma15010364





