Antibacterial Effect of Endodontic Disinfections on Enterococcus Faecalis in Dental Root Canals—An In-Vitro Model Study
Abstract
1. Introduction
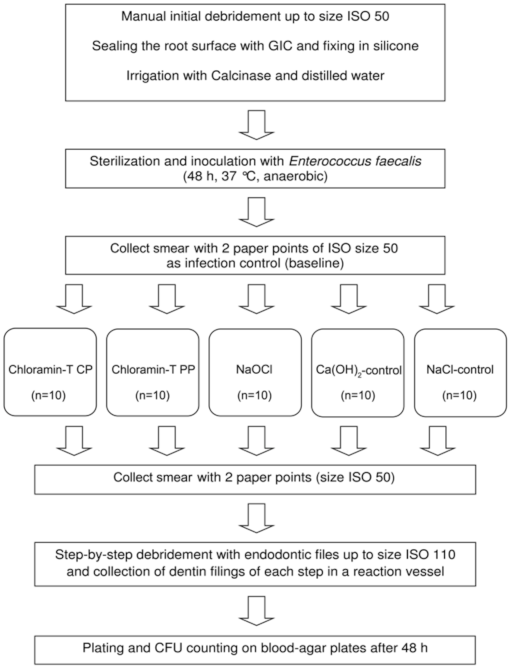
2. Materials and Methods
2.1. Cultivation of Enterococcus Faecalis
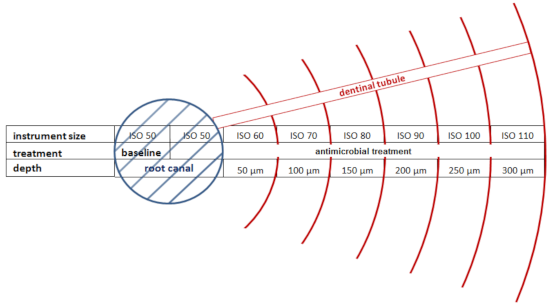
2.2. Preparation of the Dental Root Canals
2.3. Inoculation of the Root Canals with E. faecalis
2.4. Microscopic Evaluation of the Infected Root Specimens
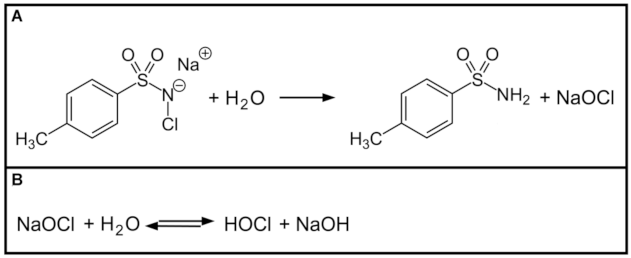
2.5. Root Canal Disinfection with Chloramine-T
2.6. Microbial Analysis
2.7. Data Analysis
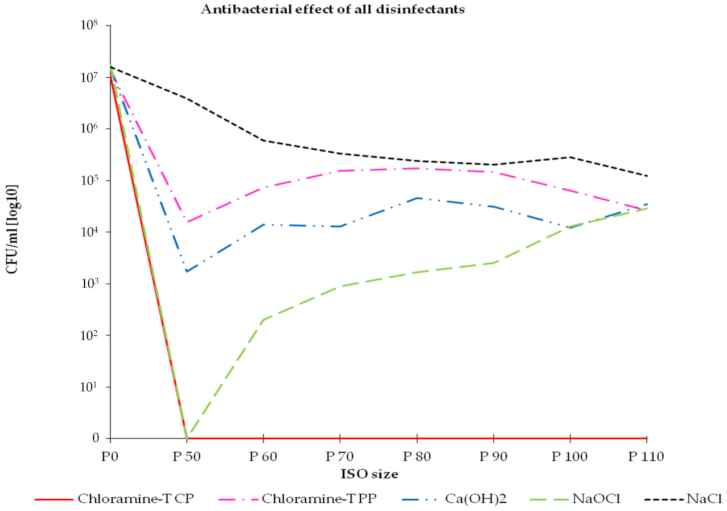
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nair, P.N.; Henry, S.; Cano, V.; Vera, J. Microbial status of apical root canal system of human mandibular first molars with primary apical periodontitis after “one-visit” endodontic treatment. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2005, 99, 231–252. [Google Scholar] [CrossRef] [PubMed]
- Prada, I.; Micó-Muñoz, P.; Giner-Lluesma, T.; Micó-Martínez, P.; Collado-Castellano, N.; Manzano-Saiz, A. Influence of microbiology on endodontic failure. Literature review. Med Oral Patol Oral Cir. Bucal 2019, 24, e364–e372. [Google Scholar] [CrossRef] [PubMed]
- Dioguardi, M.; Di Gioia, G.; Illuzzi, G.; Arena, C.; Caponio, V.C.A.; Caloro, G.A.; Zhurakivska, K.; Adipietro, I.; Troiano, G.; Lo Muzio, L. Inspection of the Microbiota in Endodontic Lesions. Dent. J. 2019, 7, 47. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Du, J.; Peng, Z. Correlation between Enterococcus faecalis and Persistent Intraradicular Infection Compared with Primary Intraradicular Infection: A Systematic Review. J. Endod. 2015, 41, 1207–1213. [Google Scholar] [CrossRef]
- Zargar, N.; Marashi, M.A.; Ashraf, H.; Hakopian, R.; Beigi, P. Identification of microorganisms in persistent/secondary endodontic infections with respect to clinical and radiographic findings: Bacterial culture and molecular detection. Iran J. Microbiol. 2019, 11, 120–128. [Google Scholar] [CrossRef]
- Komiyama, E.Y.; Lepesqueur, L.S.; Yassuda, C.G.; Samaranayake, L.P.; Parahitiyawa, N.B.; Balducci, I.; Koga-Ito, C.Y. Enterococcus Species in the Oral Cavity: Prevalence, Virulence Factors and Antimicrobial Susceptibility. PLoS ONE 2016, 11, e0163001. [Google Scholar] [CrossRef]
- Sedgley, C.M.; Nagel, A.C.; Shelburne, C.E.; Clewell, D.B.; Appelbe, O.; Molander, A. Quantitative real-time PCR detection of oral Enterococcus faecalis in humans. Arch. Oral Biol. 2005, 50, 575–583. [Google Scholar] [CrossRef]
- Siqueira, J.F.J.; Antunes, H.S.; Rocas, I.N.; Rachid, C.T.; Alves, F.R. Microbiome in the Apical Root Canal System of Teeth with Post-Treatment Apical Periodontitis. PLoS ONE 2016, 11, e0162887. [Google Scholar] [CrossRef]
- Du, T.; Wang, Z.; Shen, Y.; Ma, J.; Cao, Y.; Haapasalo, M. Effect of long-term exposure to endodontic disinfecting solutions on young and old Enterococcus faecalis biofilms in dentin canals. J. Endod. 2014, 40, 509–514. [Google Scholar] [CrossRef]
- Haseeb, R.; Lau, M.; Sheah, M.; Montagner, F.; Quiram, G.; Palmer, K.; Stefan, M.C.; Rodrigues, D.C. Synthesis and Characterization of New Chlorhexidine-Containing Nanoparticles for Root Canal Disinfection. Materials 2016, 9, 452. [Google Scholar] [CrossRef]
- Jose, J.; Krishnamma, S.; Peedikayil, F.; Aman, S.; Tomy, N.; Mariodan, J.P. Comparative Evaluation of Antimicrobial Activity of QMiX, 2.5% Sodium Hypochlorite, 2% Chlorhexidine, Guava Leaf Extract and Aloevera Extract Against Enterococcus faecalis and Candida albicans - An in-vitro Study. J. Clin. Diagn. Res. JCDR 2016, 10, Zc20. [Google Scholar] [CrossRef]
- Eneide, C.; Castagnola, R.; Martini, C.; Grande, N.M.; Bugli, F.; Patini, R.; Cordaro, M.; Sanguinetti, M.; Olivi, G.; Isola, G.; et al. Antibiofilm Activity of Three Different Irrigation Techniques: An in Vitro Study. Antibiotcs 2019, 8, 112. [Google Scholar] [CrossRef]
- Asnaashari, M.; Eghbal, M.J.; Sahba Yaghmayi, A.; Shokri, M.; Azari-Marhabi, S. Comparison of Antibacterial Effects of Photodynamic Therapy, Modified Triple Antibiotic Paste and Calcium Hydroxide on Root Canals Infected with Enterococcus faecalis: An In Vitro Study. J. Lasers Med. Sci. 2019, 10, S23–S29. [Google Scholar] [CrossRef]
- Ran, S.; He, Z.; Liang, J. Survival of Enterococcus faecalis during alkaline stress: Changes in morphology, ultrastructure, physiochemical properties of the cell wall and specific gene transcripts. Arch. Oral Biol. 2013, 58, 1667–1676. [Google Scholar] [CrossRef]
- George, M.; Ivancakova, R. Root canal microflora. Acta Med. 2007, 50, 7–15. [Google Scholar] [CrossRef][Green Version]
- Kim, R.J.; Kim, M.O.; Lee, K.S.; Lee, D.Y.; Shin, J.H. An in vitro evaluation of the antibacterial properties of three mineral trioxide aggregate (MTA) against five oral bacteria. Arch. Oral Biol. 2015, 60, 1497–1502. [Google Scholar] [CrossRef]
- Rocas, I.N.; Siqueira, J.F.J. Detection of antibiotic resistance genes in samples from acute and chronic endodontic infections and after treatment. Arch. Oral Biol. 2013, 58, 1123–1128. [Google Scholar] [CrossRef]
- Li, Y.; Wang, Y.; Chen, X.; Jiang, W.; Jiang, X.; Zeng, Y.; Li, X.; Feng, Z.; Luo, J.; Zhang, L. Antimicrobial peptide GH12 as root canal irrigant inhibits biofilm and virulence of Enterococcus faecalis. Int. Endod. J. 2020, 53, 948–961. [Google Scholar] [CrossRef]
- Ong, T.H.; Chitra, E.; Ramamurthy, S.; Siddalingam, R.P.; Yuen, K.H.; Ambu, S.P.; Davamani, F. Chitosan-propolis nanoparticle formulation demonstrates anti-bacterial activity against Enterococcus faecalis biofilms. PLoS ONE 2017, 12, e0174888. [Google Scholar]
- Walsh, L.J. Novel Approaches to Detect and Treat Biofilms within the Root Canals of Teeth: A Review. Antibiotcs 2020, 9, 129. [Google Scholar] [CrossRef]
- Ossmann, A.; Kranz, S.; Andre, G.; Volpel, A.; Albrecht, V.; Fahr, A.; Sigusch, B.W. Photodynamic killing of Enterococcus faecalis in dentinal tubules using mTHPC incorporated in liposomes and invasomes. Clin. Oral Investig. 2015, 19, 373–384. [Google Scholar] [CrossRef] [PubMed]
- Zilm, P.S.; Butnejski, V.; Rossi-Fedele, G.; Kidd, S.P.; Edwards, S.; Vasilev, K. D-amino acids reduce Enterococcus faecalis biofilms in vitro and in the presence of antimicrobials used for root canal treatment. PLoS ONE 2017, 12, e0170670. [Google Scholar] [CrossRef] [PubMed]
- Betancourt, P.; Sierra, J.M.; Camps-Font, O.; Arnabat-Domínguez, J.; Viñas, M. Er,Cr:YSGG Laser-Activation Enhances Antimicrobial and Antibiofilm Action of Low Concentrations of Sodium Hypochlorite in Root Canals. Antibiotcs 2019, 8, 232. [Google Scholar] [CrossRef] [PubMed]
- Kloth, L.C.; Berman, J.E.; Laatsch, L.J.; Kirchner, P.A. Bactericidal and cytotoxic effects of chloramine-T on wound pathogens and human fibroblasts in vitro. Adv. Ski. Wound Care 2007, 20, 331–345. [Google Scholar] [CrossRef]
- Gowda, N.M.; Trieff, N.M.; Stanton, G.J. Inactivation of poliovirus by chloramine-T. Appl. Environ. Microbiol. 1981, 42, 469–476. [Google Scholar] [CrossRef]
- Sattar, S.A.; Springthorpe, V.S.; Karim, Y.; Loro, P. Chemical disinfection of non-porous inanimate surfaces experimentally contaminated with four human pathogenic viruses. Epidemiol. Infect. 1989, 102, 493–505. [Google Scholar] [CrossRef]
- Tirapelli, C.; Landi, F.; Ribas, J.P.; Panzeri, H.; Lara, E.H. Evaluating an experimental dentifrice containing chloramine-T: A preliminary study. Oral Health Prev. Dent. 2010, 8, 375–381. [Google Scholar]
- Ferreira, G.L.S.; Rosalen, P.L.; Peixoto, L.R.; Perez, A.; Carlo, F.G.C.; Castellano, L.R.C.; Lima, J.M.; Freires, I.A.; Lima, E.O.; Castro, R.D. Antibiofilm Activity and Mechanism of Action of the Disinfectant Chloramine T on Candida spp., and Its Toxicity against Human Cells. Molecules 2017, 22, 1527. [Google Scholar] [CrossRef]
- Kneist, S.; Nietzsche, S.; Küpper, H.; Raser, G.; Willershausen, B.; Callaway, A. Penetration of Streptococcus sobrinus and Streptococcus sanguinis into dental enamel. Anaerobe 2015, 35, 54–59. [Google Scholar] [CrossRef]
- Sigusch, B.W.; Kranz, S.; Klein, S.; Volpel, A.; Harazim, S.; Sanchez, S.; Watts, D.C.; Jandt, K.D.; Schmidt, O.G.; Guellmar, A. Colonization of Enterococcus faecalis in a new SiO/SiO(2)-microtube in vitro model system as a function of tubule diameter. Dent. Mater. Off. Publ. Acad. Dent. Mater. 2014, 30, 661–668. [Google Scholar] [CrossRef]
- De Lucena, J.M.; Decker, E.M.; Walter, C.; Boeira, L.S.; Lost, C.; Weiger, R. Antimicrobial effectiveness of intracanal medicaments on Enterococcus faecalis: Chlorhexidine versus octenidine. Int. Endod. J. 2013, 46, 53–61. [Google Scholar] [CrossRef]
- Schafer, E.; Bossmann, K. Antimicrobial efficacy of chlorhexidine and two calcium hydroxide formulations against Enterococcus faecalis. J. Endod. 2005, 31, 53–56. [Google Scholar] [CrossRef]
- Vatkar, N.A.; Hegde, V.; Sathe, S. Vitality of Enterococcus faecalis inside dentinal tubules after five root canal disinfection methods. J. Conserv. Dent. 2016, 19, 445–449. [Google Scholar] [CrossRef]
- De Paula, V.A.; de Carvalho Ferreira, D.; Cavalcante, F.S.; do Carmo, F.L.; Rosado, A.S.; Primo, L.G.; dos Santos, K.R. Clinical signs and bacterial communities of deciduous necrotic root canals detected by PCR-DGGE analysis: Research association. Arch. Oral Biol. 2014, 59, 848–854. [Google Scholar] [CrossRef]
- Gomes, B.P.; Souza, S.F.; Ferraz, C.C.; Teixeira, F.B.; Zaia, A.A.; Valdrighi, L.; Souza-Filho, F.J. Effectiveness of 2% chlorhexidine gel and calcium hydroxide against Enterococcus faecalis in bovine root dentine in vitro. Int. Endod. J. 2003, 36, 267–275. [Google Scholar] [CrossRef]
- Love, R.M. Enterococcus faecalis--a mechanism for its role in endodontic failure. Int. Endod. J. 2001, 34, 399–405. [Google Scholar] [CrossRef]
- Love, R.M. Bacterial adhesins--their role in tubule invasion and endodontic disease. Aust. Endod. J. 2002, 28, 25–28. [Google Scholar] [CrossRef]
- Milroy, T.H. The Action of Hypochlorites and Allied Substances on Proteins, and their Behaviour on Injection. Biochem. J. 1916, 10, 453–465. [Google Scholar] [CrossRef]
- Stralfors, A.; Thilander, H.; Bergenholtz, A. Simultaneous inhibition of caries and periodontal disease in hamsterby disinfection, tooth-brushing or phosphate addition. Arch. Oral Biol. 1967, 12, 1367–1373. [Google Scholar] [CrossRef]
- Guiteras, A.F.; Schmelkes, F.C. The comparative action of sodium hypochlorite, chloramine-T, and azochloramid on organic substrates. J. Biol. Chem. 1934, 107, 235–239. [Google Scholar] [CrossRef]
- Mozayeni, M.A.; Haeri, A.; Dianat, O.; Jafari, A.R. Antimicrobial effects of four intracanal medicaments on enterococcus faecalis: An in vitro study. Iran. Endod. J. 2014, 9, 195–198. [Google Scholar]
- Kawashima, N.; Wadachi, R.; Suda, H.; Yeng, T.; Parashos, P. Root canal medicaments. Int. Dent. J. 2009, 59, 5–11. [Google Scholar]
- Siqueira, J.F.J.; Lopes, H.P. Mechanisms of antimicrobial activity of calcium hydroxide: A critical review. Int. Endod. J. 1999, 32, 361–366. [Google Scholar] [CrossRef]
- Athanassiadis, B.; Walsh, L.J. Aspects of Solvent Chemistry for Calcium Hydroxide Medicaments. Materials 2017, 10, 1219. [Google Scholar] [CrossRef]
- Teoh, Y.Y.; Athanassiadis, B.; Walsh, L.J. Comparison of Commercial Calcium Hydroxide Pastes for Prolonged Antibacterial Effect using a Colourimetric Assessment. Materials 2018, 11, 348. [Google Scholar] [CrossRef]
- Evans, M.; Davies, J.K.; Sundqvist, G.; Figdor, D. Mechanisms involved in the resistance of Enterococcus faecalis to calcium hydroxide. Int. Endod. J. 2002, 35, 221–228. [Google Scholar] [CrossRef]
- Borzini, L.; Condo, R.; De Dominicis, P.; Casaglia, A.; Cerroni, L. Root Canal Irrigation: Chemical Agents and Plant Extracts Against Enterococcus faecalis. Open Dent. J. 2016, 10, 692–703. [Google Scholar] [CrossRef]
- Sahebi, S.; Khosravifar, N.; Sedighshamsi, M.; Motamedifar, M. Comparison of the antibacterial effect of sodium hypochlorite and aloe vera solutions as root canal irrigants in human extracted teeth contaminated with enterococcus faecalis. J. Dent. 2014, 15, 39–43. [Google Scholar]
- Walsh, L.J.; George, R. Activation of Alkaline Irrigation Fluids in Endodontics. Materials 2017, 10, 1214. [Google Scholar] [CrossRef]
- Choi, H.W.; Park, S.Y.; Kang, M.K.; Shon, W.J. Comparative Analysis of Biofilm Removal Efficacy by Multisonic Ultracleaning System and Passive Ultrasonic Activation. Materials 2019, 12, 3492. [Google Scholar] [CrossRef]
- Estrela, C.; Estrela, C.R.; Barbin, E.L.; Spano, J.C.; Marchesan, M.A.; Pecora, J.D. Mechanism of action of sodium hypochlorite. Braz. Dent. J. 2002, 13, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Buttler, T.K.; Crawford, J.J. The detoxifying effect of varying concentrations of sodium hypochlorite on endotoxins. J. Endod. 1982, 8, 59–66. [Google Scholar] [CrossRef]
- Wang, D.M.; Gao, X.J.; Shen, S. Comparison of antimicrobial efficacy of four endodontic irrigants using an in vitro model infected by Enterococcus faecalis. Chin. J. Stomatol. 2007, 42, 223–224. [Google Scholar]
- Qi, P. An experimental study on cleaning efficacy of several root canal irrigants. Chin. J. Stomatol. 1991, 26, 205–207. [Google Scholar]
- Esposito, M.; Trullenque-Eriksson, A.; Tallarico, M. Endodontic retreatment versus dental implants of teeth with an uncertain endodontic prognosis: 3-year results from a randomised controlled trial. Eur. J. Oral Implantol. 2018, 11, 423–438. [Google Scholar]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kranz, S.; Guellmar, A.; Braeutigam, F.; Tonndorf-Martini, S.; Heyder, M.; Reise, M.; Sigusch, B. Antibacterial Effect of Endodontic Disinfections on Enterococcus Faecalis in Dental Root Canals—An In-Vitro Model Study. Materials 2021, 14, 2427. https://doi.org/10.3390/ma14092427
Kranz S, Guellmar A, Braeutigam F, Tonndorf-Martini S, Heyder M, Reise M, Sigusch B. Antibacterial Effect of Endodontic Disinfections on Enterococcus Faecalis in Dental Root Canals—An In-Vitro Model Study. Materials. 2021; 14(9):2427. https://doi.org/10.3390/ma14092427
Chicago/Turabian StyleKranz, Stefan, André Guellmar, Franziska Braeutigam, Silke Tonndorf-Martini, Markus Heyder, Markus Reise, and Bernd Sigusch. 2021. "Antibacterial Effect of Endodontic Disinfections on Enterococcus Faecalis in Dental Root Canals—An In-Vitro Model Study" Materials 14, no. 9: 2427. https://doi.org/10.3390/ma14092427
APA StyleKranz, S., Guellmar, A., Braeutigam, F., Tonndorf-Martini, S., Heyder, M., Reise, M., & Sigusch, B. (2021). Antibacterial Effect of Endodontic Disinfections on Enterococcus Faecalis in Dental Root Canals—An In-Vitro Model Study. Materials, 14(9), 2427. https://doi.org/10.3390/ma14092427







