Microstructural Analysis of Fractured Orthopedic Implants
Abstract
1. Introduction
2. Materials and Methods
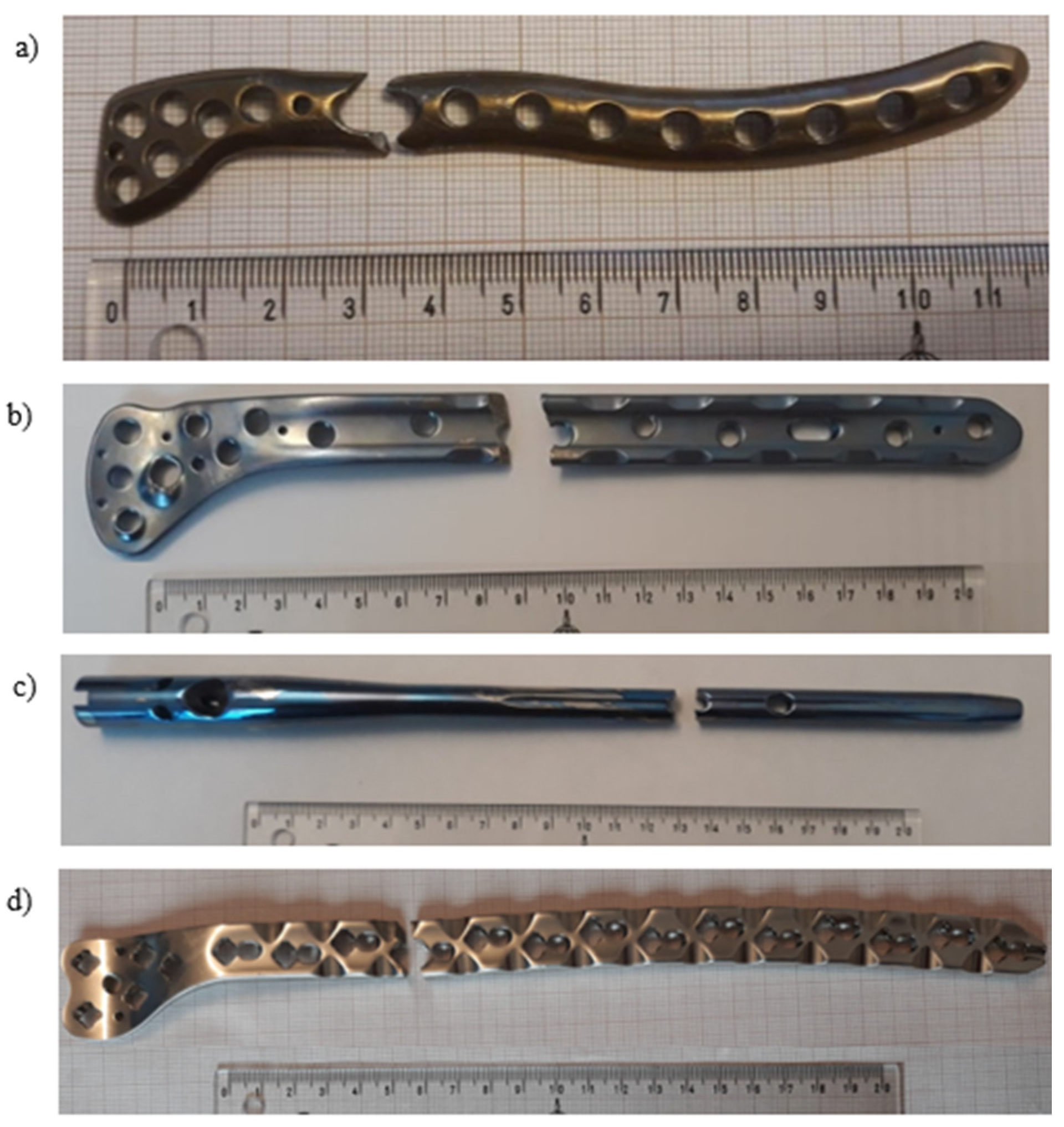
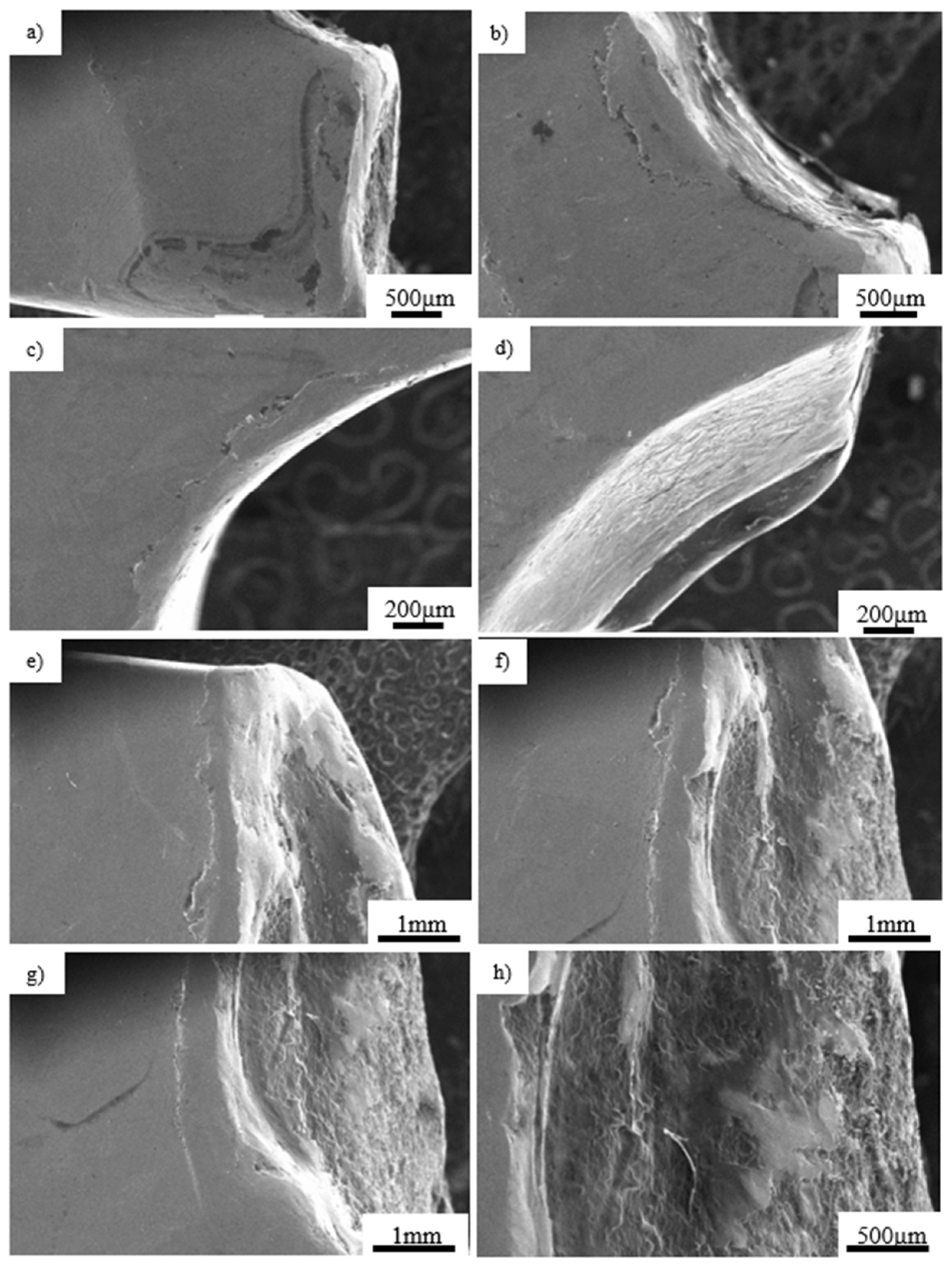
- Pure titanium, angularly stabile, clavicular, locking plate (5.0 ChLP; ChM, Juchnowiec Kościelny, Poland; Figure 1a);
- Pure titanium, angularly stabile, femoral, condylar plate (7.0 ChLP, ChM, Juchnowiec Kościelny, Poland; Figure 1b);
- Ti-6Al-4V intramedullary nail (Triple Proximal Femoral Nail, Medgal, Poland; Księżyno, Figure 1c);
- X2CrNiMo18-14-3 steel femoral plate (4.5 mm VA LCP®Condylar Plate, DePuy Synthes, Warsaw, IN, USA; Figure 1d).
3. Results and Discussion
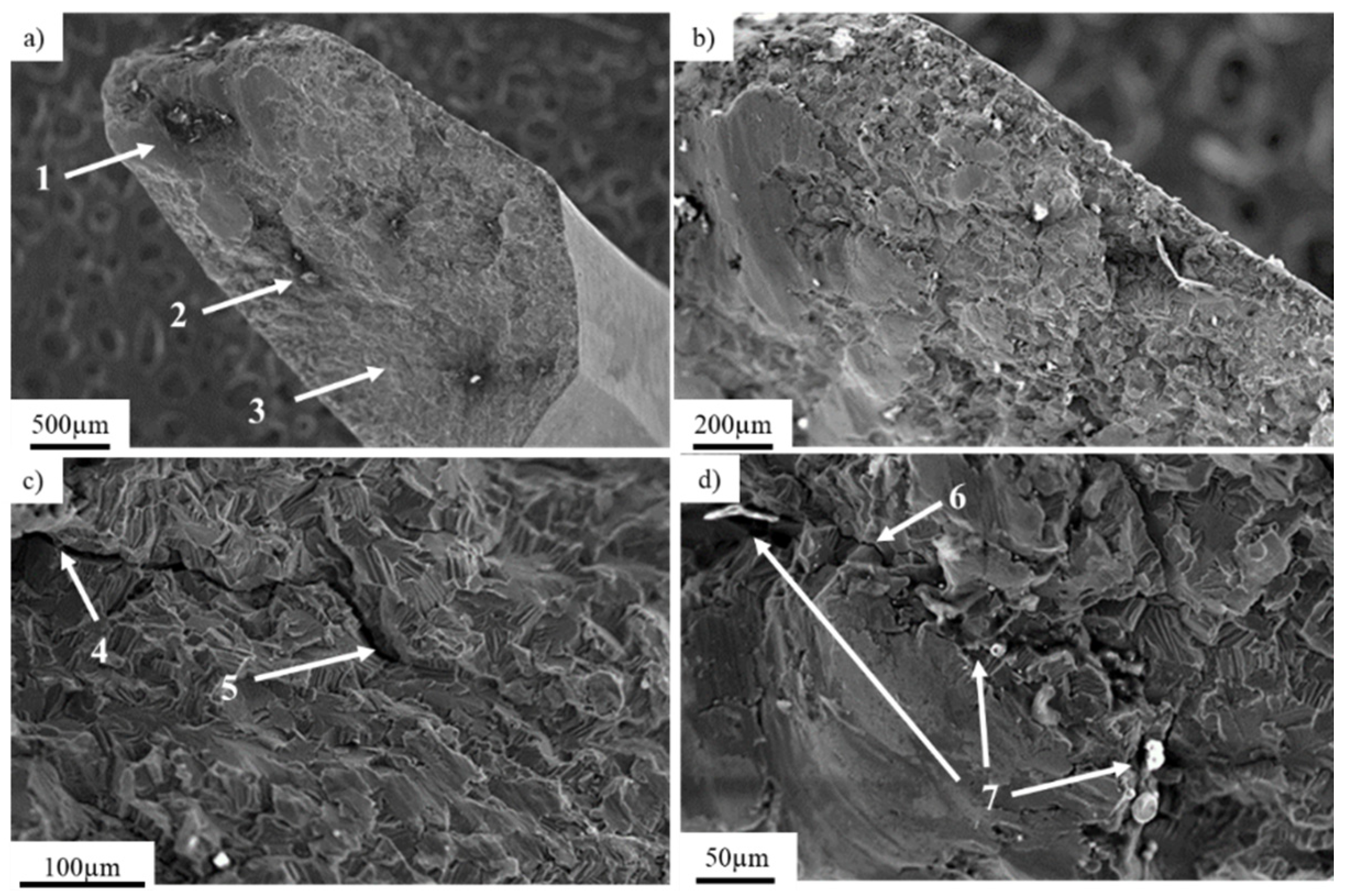
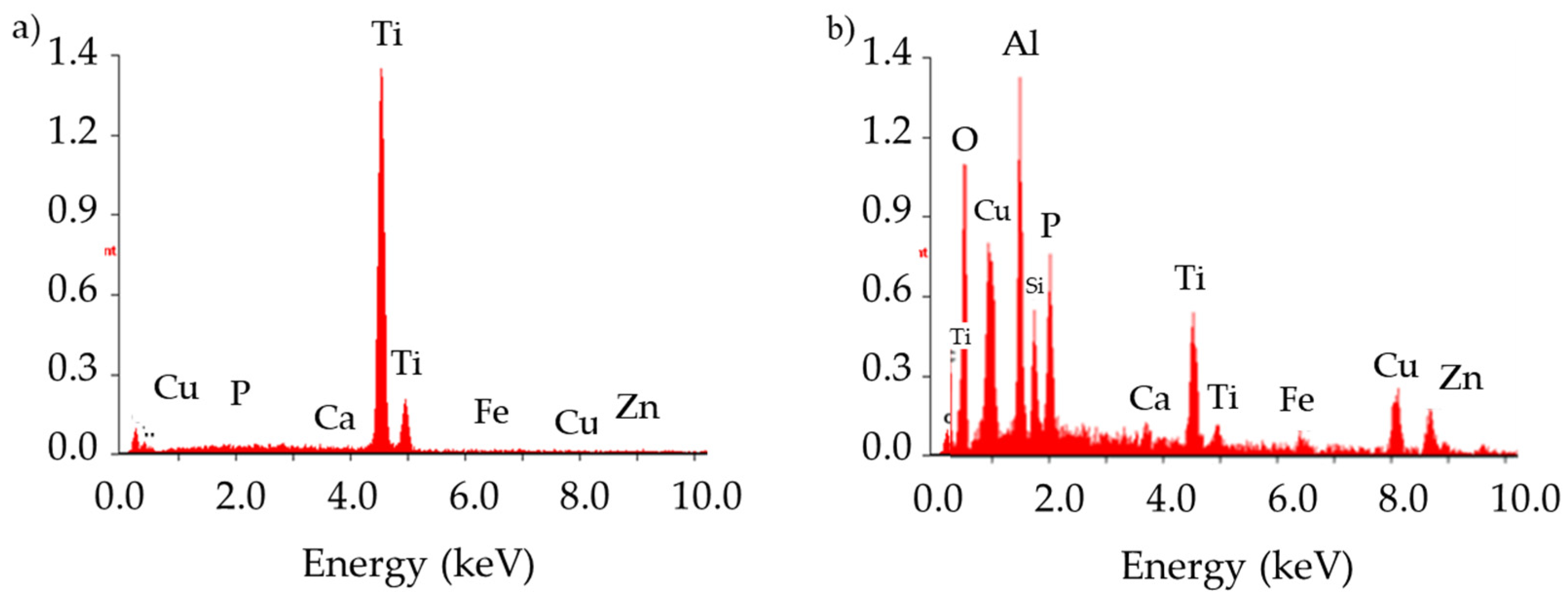
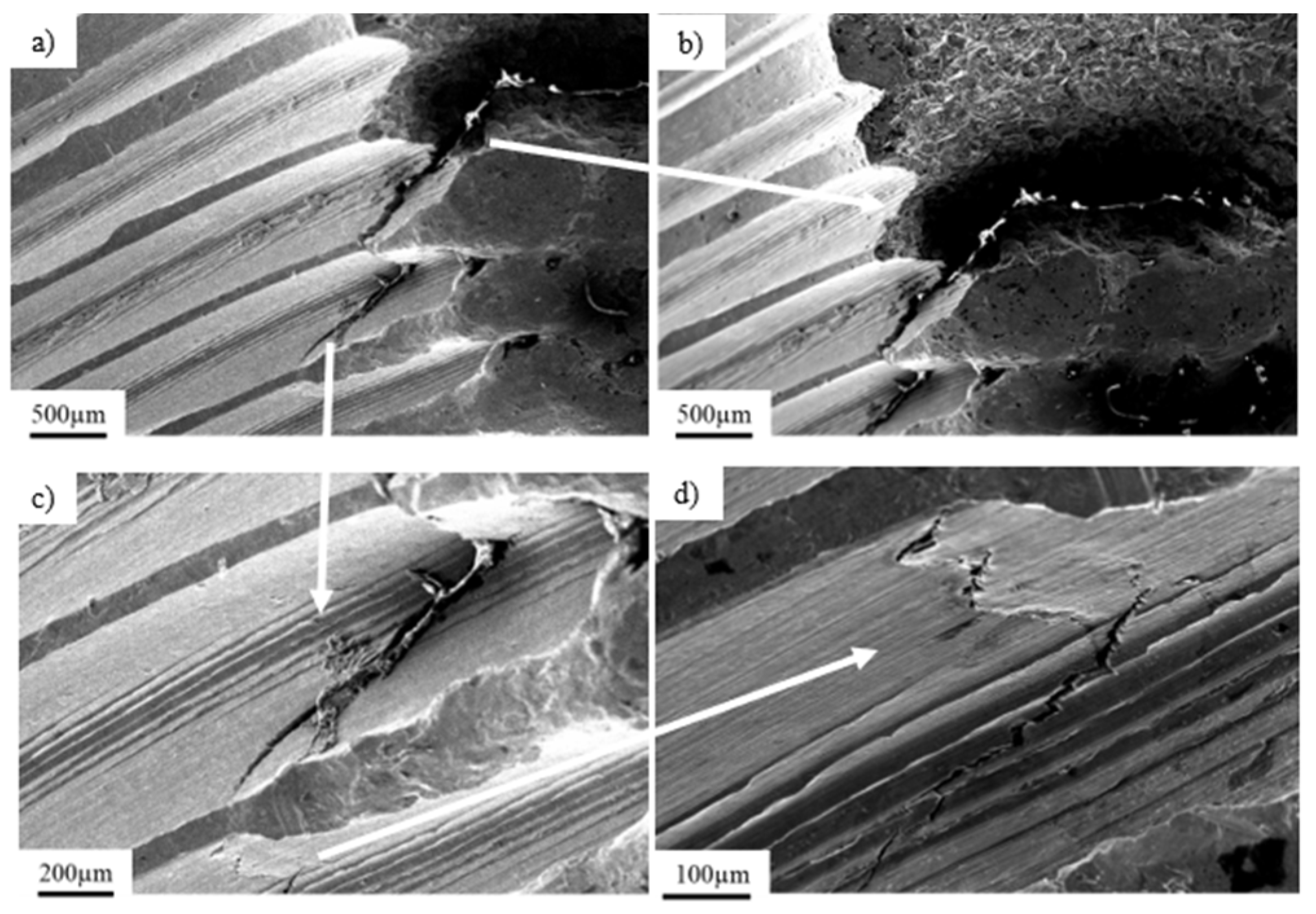
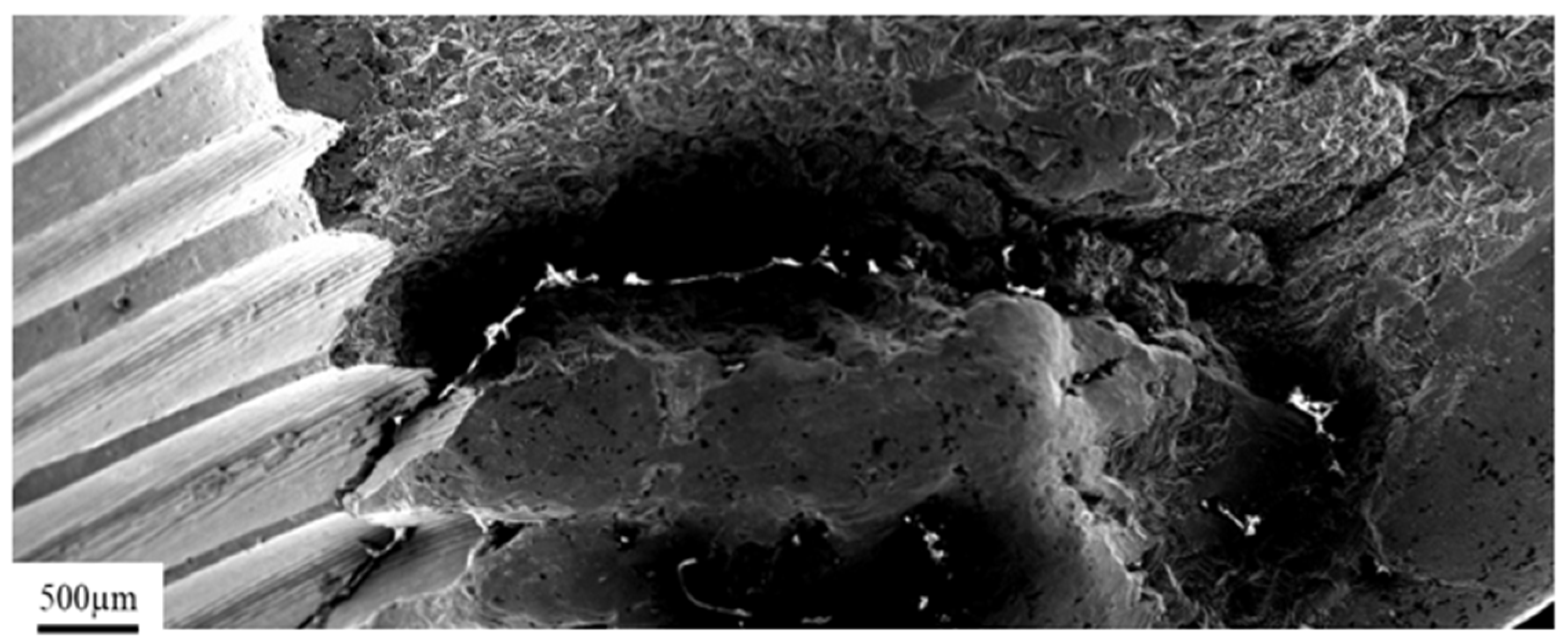
3.1. Pure Titanium Plates
3.2. Pure Titanium Femoral Implant
3.3. Ti-6Al-4V Titanium Alloy
3.4. X2CrNiMo18-14-3 Steel Femoral Implant
4. Summary and Concluding Remarks
- material impurities, where super-hard aluminum oxide acts as a mechanical strain concentrator, serving as the center of cracks propagating between the structure of an implant,
- the deficient adjustment of implant designs to the stresses acting on it,
- improper technology of implant production, especially in the case of drilling and threading of the holes serving as mounting areas of screws attaching it to the bone,
- inappropriate surgical technique during implantation,
- its mechanical overloads caused by excessive forces acting on limbs treated with this particular implant.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Özcan, M.; Hämmerle, C. Titanium as a Reconstruction and Implant Material in Dentistry: Advantages and Pitfalls. Materials 2012, 5, 1528–1545. [Google Scholar] [CrossRef]
- Hayes, J.S.; Richards, R.G. The use of titanium and stainless steel in fracture fixation. Expert Rev. Med. Devices 2010, 7, 843–853. [Google Scholar] [CrossRef] [PubMed]
- Becerikli, M.; Jaurich, H.; Wallner, C.; Wagner, J.M.; Dadras, M.; Jettkant, B. P2000—A high-nitrogen austenitic steel for application in bone surgery. PLoS ONE 2019, 14. [Google Scholar] [CrossRef]
- Nowacki, J.; Dobrzański, L.A.; Gustavo, L. Implanty śródszpikowe w osteosyntezie kości długich. Open Access Libr. 2012, 72, 1–150. [Google Scholar]
- Wiliams, D.F. Definitions in Biomaterials; Elsevier: Amsterdam, The Netherlands, 1987; Volume 26, p. 414. [Google Scholar]
- Hollander, D.A.; Von Walter, M.; Wirtz, T.; Sellei, R.; Schmidt-Rohlfing, B.; Paar, O.; Erli, H.-J. Structural, mechanical and in vitro characterization of individually structured Ti–6Al–4V produced by direct laser forming. Biomaterials 2006, 27, 955–963. [Google Scholar] [CrossRef]
- Zhou, Y.L.; Niinomi, M.; Akahori, T.; Fukui, H.; Toda, H. Corrosion resistance and biocompatibility of Ti–Ta alloys for biomedical applications. Mater. Sci. Eng. A 2005, 398, 28–36. [Google Scholar] [CrossRef]
- Gepreel, M.A.-H.; Niinomi, M. Biocompatibility of Ti-alloys for long-term implantation. J. Mech. Behav. Biomed. Mater. 2013, 20, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Bruni, S.; Martinesi, M.; Stio, M.; Treves, C.; Bacci, T.; Borgioli, F. Effects of surface treatment of Ti–6Al–4V titanium alloy on biocompatibility in cultured human umbilical vein endothelial cells. Acta Biomater. 2005, 1, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Kumari, R.; Scharnweber, T.; Pfleging, W.; Besser, H.; Majumdar, J.D. Laser surface textured titanium alloy (Ti–6Al–4V)—Part II—Studies on bio-compatibility. Appl. Surf. Sci. 2015, 357, 750–758. [Google Scholar] [CrossRef]
- Dobrzański, L.A. Materiały Inżynierskie i Projektowanie Materiałowe. Podstawy Nauki o Materiałach i Metaloznawstwo; WNT: Warsaw, Poland, 2006. [Google Scholar]
- Dobrzański, L.A. Metaloznawstwo Opisowe Stopów Metali Nieżelaznych; Wydawnictwo Politechniki Śląskiej: Gliwice, Poland, 2008. [Google Scholar]
- Dobrzański, L.A. Metaloznawstwo Opisowe Stopów Żelaza; Wydawnictwo Politechniki Śląskiej: Gliwice, Poland, 2007. [Google Scholar]
- Gaadzik, T.S. Gwoździowanie Śródszpikowe; Wydawnictwo Medyczne G-Medica press: Bielsko-Biała, Poland, 2000. [Google Scholar]
- Károly, D.; Asztalos, L.; Micsík, T.; Szabó, P.J. Non-Destructive Analysis of Explanted Coronary Artery Stents. Acta Polytech. Hung. 2017, 14. [Google Scholar] [CrossRef]
- Disegi, J.A.; Eschbach, L. Stainless steel in bone surgery. Injury 2000, 31, 2–6. [Google Scholar] [CrossRef]
- Banovetz, J.M.; Sharp, R.; Probe, R.A.; Anglen, J.O. Titanium plate fixation: A review of implant failures. J. Orthop. Trauma 1996, 10, 389–394. [Google Scholar] [CrossRef]
- Gosain, A.K.; Song, L.; Corrao, M.A.; Pintar, F.A. Biomechanical evaluation of titanium, biodegradable plate and screw, and cyanoacrylate glue fixation systems in craniofacial surgery. Plast. Reconstr. Surg. 1998, 101, 582–591. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Smith, W.R.; Stahel, P.F.; Morgan, S.J.; Baron, A.J.; Hak, D.J. Technical problems and complications in the removal of the less invasive stabilization system. J. Orthop. Trauma 2010, 24, 369–373. [Google Scholar] [CrossRef] [PubMed]
- Elias, C.N.; Lima, J.H.C.; Valiev, R.; Meyers, M.A. Biomedical Applications of Titanium and its Alloys. JOM 2008. [Google Scholar] [CrossRef]
- Zizzari, V.L.; Zara, S.; Tetè, G.; Vinci, R.; Gherlone, E.; Cataldi, A. Biologic and clinical aspects of integration of different bone substitutes in oral surgery: A literature review. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2016, 122, 392–402. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.H.; Lee, J.H. Absorbable Plate-Related Infection after Facial Bone Fracture Reduction. Arch. Craniofac. Surg. 2016, 17, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Liang, H.; Yang, Y.; Xie, D.; Li, L.; Mao, N.; Wang, C.; Tian, Z.; Zhang, P.; Shen, L. Trabecular-like Ti-6Al-4V scaffolds for orthopedic: Fabrication by selective laser melting and in vitro biocompatibility. J. Mater. Sci. Technol. 2019, 35, 1284–1297. [Google Scholar] [CrossRef]
- Kruth, J.-P.; Levy, G.; Klocke, F.; Childs, T. Consolidation phenomena in laser and powder-bed based layered manufacturing. CIRP Ann. 2007, 56, 730–759. [Google Scholar] [CrossRef]
- Yasa, E.; Deckers, J.; Kruth, J.-P.; Rombouts, M.; Luyten, J. Charpy impact testing of metallic selective laser melting parts. Virtual Phys. Prototyp. 2010, 5, 89–98. [Google Scholar] [CrossRef]
- Azarniya, A.; Colera, X.G.; Mirzaali, M.J.; Sovizi, S.; Bartolomeu, F.; Weglowski, M.S.; Wits, W.W.; Yap, C.Y.; Ahn, J.; Miranda, G.; et al. Additive manufacturing of Ti–6Al–4V parts through laser metal deposition (LMD): Process, microstructure, and mechanical properties. J. Alloys Compd. 2019, 804, 163–191. [Google Scholar] [CrossRef]
- Everton, S.K.; Hirsch, M.; Stravroulakis, P.; Leach, R.K.; Clare, A.T. Review of in-situ process monitoring and in-situ metrology for metal additive manufacturing. Mater. Des. 2016, 431–445. [Google Scholar] [CrossRef]
- De Formanoir, C.; Michotte, S.; Rigo, O.; Germain, L.; Godet, S. Electron beam melted Ti–6Al–4V: Microstructure, texture and mechanical behavior of the as-built and heat-treated material. Mater. Sci. Eng. A 2016, 652, 105–119. [Google Scholar] [CrossRef]
- Yu, H.-C.; Kim, Y.-K. Fractures of implant fixtures: A retrospective clinical study. Maxillofac. Plast. Reconstr. Surg. 2020, 42, 13–16. [Google Scholar] [CrossRef] [PubMed]
- Yi, Y.; Koak, J.-Y.; Kim, S.-K.; Lee, S.-J.; Heo, S.-J. Comparison of implant component fractures in external and internal type: A 12-year retrospective study. J. Adv. Prosthodont. 2018, 10, 155–162. [Google Scholar] [CrossRef]
- Lee, D.-W.; Kim, N.-H.; Lee, Y.; Oh, Y.-A.; Lee, J.-H.; You, H.-K. Implant fracture failure rate and potential associated risk indicators: An up to 12-year retrospective study of implants in 5124 patients. Clin. Oral Implant. Res. 2019, 30, 206–217. [Google Scholar] [CrossRef]
- Stoichkov, B.; Kirov, D. Analysis of the causes of dental implant fracture: A retrospective clinical study. Quintessence Int. 2018, 49, 279–286. [Google Scholar] [PubMed]
- Oh, S.-L.; Barnes, D. Managing a fractured implant. J. Prosthet. Dent. 2016, 115, 397–401. [Google Scholar] [CrossRef]
- Gealh, W.C.; Mazzo, V.; Barbi, F.; Camarini, E.T. Osseointegrated Implant Fracture: Causes and Treatment. J. Oral Implant. 2011, 37, 499–503. [Google Scholar] [CrossRef]
- Sanivarapu, S.; Moogla, S.; Kuntcham, R.S.; Kolaparthy, L.K. Implant fractures: Rare but not exceptional. J. Indian Soc. Periodontol. 2016, 20, 6–11. [Google Scholar] [CrossRef]
- Gibney, K. Fracture of the body of an implant and its management—A case history. Br. Dent. J. 2004, 197, 615–617. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.S.; Choi, B.R.; Na, Y.C. Fracture of dental implants: Case report. J. Dent. Implant. Res. 2012, 31, 84–87. [Google Scholar]
- Gargallo Albiol, J.; Satorres-Nieto, M.; Puyuelo Capablo, J.L.; Sanchez Garces, M.A.; Pi Urgell, J.; Gay Escoda, C. Endosseous dental implant fractures: An analysis of 21 cases. Med. Oral Patol. Oral Cir. Bucal 2008, 13, 124–128. [Google Scholar]
- Tabrizi, R.; Behnia, H.; Taherian, S.; Hesami, N. What are the incidence and factors associated with implant fracture? J. Oral Maxillofac. Surg. 2017, 75, 1866–1872. [Google Scholar] [CrossRef] [PubMed]
- Chrcanovic, B.R.; Kisch, J.; Albrektsson, T.; Wennerberg, A. Factors influencing the fractures of dental implants. Clin. Implant. Dent. Relat. Res. 2018, 20, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Kyehee Wee, M.S.; Bo-Ah, L. Implant Fracture After Screw Fracture in a TissueLevel Implant: A Case Report. Implantology 2019, 23, 229–234. [Google Scholar] [CrossRef]
- ASM International. ASM Handbook. Fatigue and Fracture; ASM International: Novelty, OH, USA, 1996; Volume 19. [Google Scholar] [CrossRef]
- Hearn, E.J. Chapter 15—Theories of elastic failure. In Mechanics of Materials 1, 3rd ed.; Butterworth-Heinemann: Oxford, UK, 1997; pp. 401–429. [Google Scholar]
- ISO. Implants for Surgery—Metallic Materials—Part 1: Wrought Stainless Steel PN-ISO 5832-1 Standard; ISO: Geneva, Switzerland, 2016. [Google Scholar]
- Nica, M.; Cretu, B.; Ene, D.; Antoniac, I.; Gheorghita, D.; Ene, R. Failure Analysis of Retrieved Osteosynthesis Implants. Materials 2020, 13, 1201. [Google Scholar] [CrossRef] [PubMed]
- Gervais, B.; Vadean, A.; Raison, M.; Brochu, M. Failure analysis of a 316L stainless steel femoral orthopedic implant. Case Stud. Eng. Fail. Anal. 2016, 5, 30–38. [Google Scholar] [CrossRef]
- Hou, S.M.; Wang, J.L.; Lin, J. Mechanical strength, fatigue life, and failure analysis of two prototypes and five conventional tibial locking screws. J. Orthop. Trauma 2002, 16, 701–708. [Google Scholar] [CrossRef]











| Element | Ti | Impurities |
|---|---|---|
| Wt % | 99.20 | 00.80 |
| Element | Ti | Impurities |
|---|---|---|
| Wt % | 99.12 | 00.88 |
| Element | C | O | P | Mo | Cl | K | Ca | Ti |
|---|---|---|---|---|---|---|---|---|
| Wt % | 17.34 | 04.77 | 12.65 | 02.27 | 01.40 | 01.56 | 51.95 | 08.04 |
| Element | Ti | Al | V |
|---|---|---|---|
| Wt % | 90.12 | 06.72 | 03.16 |
| Element | C | Si | Mo | Cr | Fe | Ni |
|---|---|---|---|---|---|---|
| Wt % | 2.11 | 0.64 | 3.18 | 17.45 | 61.73 | 14.89 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kopec, M.; Brodecki, A.; Szczęsny, G.; Kowalewski, Z.L. Microstructural Analysis of Fractured Orthopedic Implants. Materials 2021, 14, 2209. https://doi.org/10.3390/ma14092209
Kopec M, Brodecki A, Szczęsny G, Kowalewski ZL. Microstructural Analysis of Fractured Orthopedic Implants. Materials. 2021; 14(9):2209. https://doi.org/10.3390/ma14092209
Chicago/Turabian StyleKopec, Mateusz, Adam Brodecki, Grzegorz Szczęsny, and Zbigniew L. Kowalewski. 2021. "Microstructural Analysis of Fractured Orthopedic Implants" Materials 14, no. 9: 2209. https://doi.org/10.3390/ma14092209
APA StyleKopec, M., Brodecki, A., Szczęsny, G., & Kowalewski, Z. L. (2021). Microstructural Analysis of Fractured Orthopedic Implants. Materials, 14(9), 2209. https://doi.org/10.3390/ma14092209







