Silane-Coating Strategy for Titanium Functionalization Does Not Impair Osteogenesis In Vivo
Abstract
1. Introduction
2. Materials and Methods
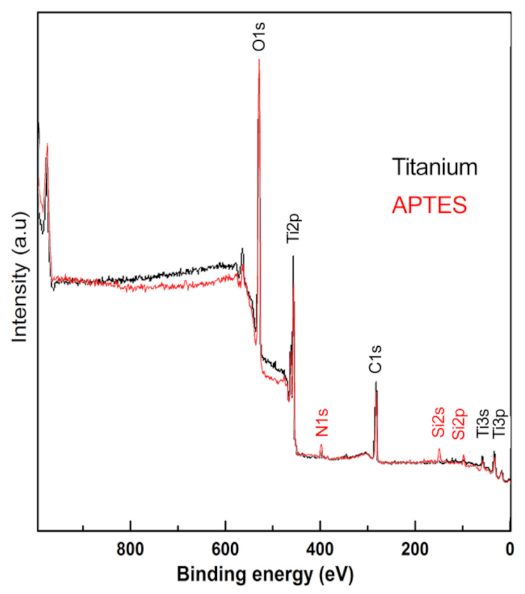
2.1. Silanization
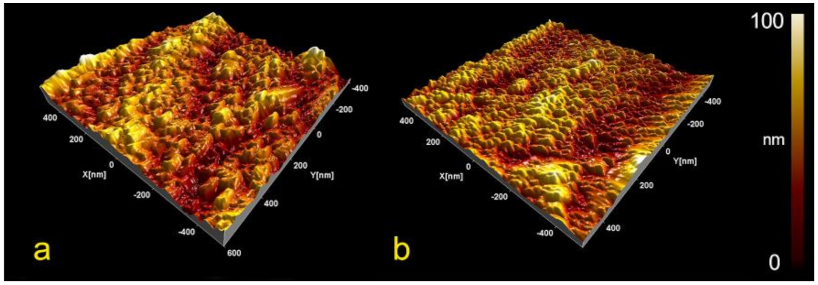
2.2. Surface Morphology
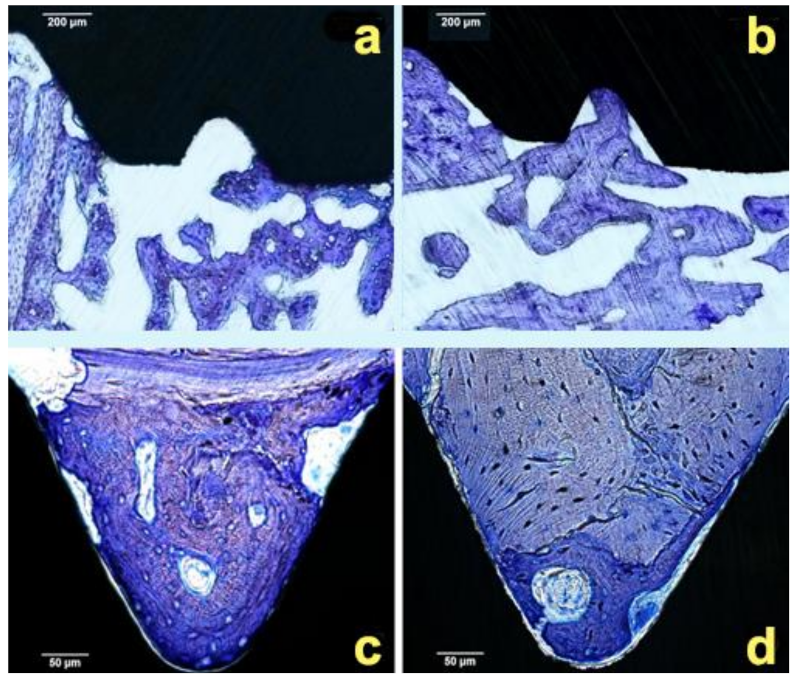
2.3. Bone Formation Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cai, K.; Frant, M.; Bossert, J.; Hildebrand, G.; Liefeith, K.; Jandt, K.D. Surface functionalized titanium thin films: Zeta-potential, protein adsorption and cell proliferation. Colloids Surf. B Biointerfaces 2006, 50, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.-C.; Ko, C.-L. Roughened titanium surfaces with silane and further RGD peptide modification in vitro. Mater. Sci. Eng. C 2013, 33, 2713–2722. [Google Scholar] [CrossRef]
- van Steenberghe, D.; Jacobs, R.; Desnyder, M.; Maffei, G.; Quirynen, M. The relative impact of local and endogenous pa-tient-related factors on implant failure up to the abutment stage. Clin. Oral. Implants Res. 2002, 13, 617–622. [Google Scholar] [CrossRef]
- Johnsen, S.P.; Sørensen, H.T.; Lucht, U.; Søballe, K.; Overgaard, S.; Pedersen, A.B. Patient-related predictors of implant failure after primary total hip replacement in the initial, short- and long-terms. J. Bone Jt. Surgery. Br. Vol. 2006, 88, 1303–1308. [Google Scholar] [CrossRef]
- Capparé, P.; Teté, G.; Romanos, G.E.; Nagni, M.; Sannino, G.; Gherlone, E.F. The ’All-on-four’ protocol in HIV-positive patients: A prospective, longitudinal 7-year clinical study. Int. J. Oral. Implantol. 2019, 12, 501–510. [Google Scholar]
- Gherlone, E.F.; Capparé, P.; Tecco, S.; Polizzi, E.; Pantaleo, G.; Gastaldi, G.; Grusovin, M.G. Implant Prosthetic Rehabilitation in Controlled HIV-Positive Patients: A Prospective Longitudinal Study with 1-Year Follow-Up. Clin. Implant. Dent. Relat. Res. 2016, 18, 725–734. [Google Scholar] [CrossRef] [PubMed]
- Sannino, G.; Montemezzi, P.; Pantaleo, G.; Agliardi, E. Dental implants survival rate in controlled type I diabetic patients: A prospective longitudinal study with a 2-year follow-up. J. Biol. Regul. Homeost. Agents 2021, 34, 35–43. [Google Scholar]
- Bagno, A.; Piovan, A.; Dettin, M.; Brun, P.; Gambaretto, R.; Palu, G.; Di Bello, C.; Castagliuolo, I. Improvement of Anselme’s adhesion model for evaluating human osteoblast response to peptide-grafted titanium surfaces. Bone 2007, 41, 704–712. [Google Scholar] [CrossRef] [PubMed]
- Shah, F.A.; Stenlund, P.; Martinelli, A.; Thomsen, P.; Palmquist, A. Direct communication between osteocytes and acid-etched titanium implants with a sub-micron topography. J. Mater. Sci. Mater. Electron. 2016, 27, 167. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.; Park, K.-H.; Jung, N.; Shim, J.-S.; Moon, H.-S.; Kim, H.-J.; Oh, S.-H.; Kim, Y.; Ku, S.-Y.; Park, Y.-B. In Vivo Study for Clinical Application of Dental Stem Cell Therapy Incorporated with Dental Titanium Implants. Materials 2021, 14, 381. [Google Scholar] [CrossRef] [PubMed]
- Capparè, P.; Tetè, G.; Sberna, M.T.; Panina-Bordignon, P. The Emerging Role of Stem Cells in Regenerative Dentistry. Curr. Gene Ther. 2020, 20, 259–268. [Google Scholar] [CrossRef]
- Park, J.-W.; Kurashima, K.; Tustusmi, Y.; An, C.-H.; Suh, J.-Y.; Doi, H.; Nomura, N.; Noda, K.; Hanawa, T. Bone healing of commercial oral implants with RGD immobilization through electrodeposited poly(ethylene glycol) in rabbit cancellous bone. Acta Biomater. 2011, 7, 3222–3229. [Google Scholar] [CrossRef] [PubMed]
- Felgueiras, H.P.; Evans, M.D.M.; Migonney, V. Contribution of fibronectin and vitronectin to the adhesion and morphology of MC3T3-E1 osteoblastic cells to poly(NaSS) grafted Ti6Al4V. Acta Biomater. 2015, 28, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Rezania, A.; Johnson, R.; Lefkow, A.R.; Healy, K.E. Bioactivation of Metal Oxide Surfaces. 1. Surface Characterization and Cell Response. Langmuir 1999, 15, 6931–6939. [Google Scholar] [CrossRef]
- Forget, G.; Latxague, L.; Heroguez, V.; Labrugere, C.; Durrieu, M.C. RGD nanodomains grafting onto titanium surface. In Proceedings of the 2007 29th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Lyon, France, 22–26 August 2007; Institute of Electrical and Electronics Engineers (IEEE): New York, NY, USA, 2007; Volume 2007, pp. 5107–5110. [Google Scholar]
- Ryu, J.J.; Park, K.; Kim, H.S.; Jeong, C.M.; Huh, J.B. Effects of anodized titanium with Arg-Gly-Asp (RGD) peptide immobi-lized via chemical grafting or physical adsorption on bone cell adhesion and differentiation. Int. J. Oral Maxillofac. Implant. 2013, 28, 963–972. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Huh, J.-B.; Lee, J.-Y.; Jeon, Y.-C.; Shin, S.-W.; Ahn, J.-S.; Ryu, J.-J. Physical stability of arginine-glycine-aspartic acid peptide coated on anodized implants after installation. J. Adv. Prosthodont. 2013, 5, 84–91. [Google Scholar] [CrossRef]
- Senna, P.; Dds, A.A.D.B.C.; Kates, S.; Meirelles, L. Surface Damage on Dental Implants with Release of Loose Particles after Insertion into Bone. Clin. Implant. Dent. Relat. Res. 2015, 17, 681–692. [Google Scholar] [CrossRef] [PubMed]
- Nanci, A.; Wuest, J.D.; Peru, L.; Brunet, P.; Sharma, V.; Zalzal, S.; McKee, M.D. Chemical modification of titanium surfaces for covalent attachment of biological molecules. J Biomed Mater Res 1998, 40, 324–335. [Google Scholar] [CrossRef]
- Marín-Pareja, N.; Cantini, M.; González-García, C.; Salvagni, E.; Salmerón-Sánchez, M.; Ginebra, M.-P. Different Organization of Type I Collagen Immobilized on Silanized and Nonsilanized Titanium Surfaces Affects Fibroblast Adhesion and Fibronectin Secretion. ACS Appl. Mater. Interfaces 2015, 7, 20667–20677. [Google Scholar] [CrossRef] [PubMed]
- Godoy-Gallardo, M.; Guillem-Marti, J.; Sevilla, P.; Manero, J.M.; Gil, F.J.; Rodriguez, D. Anhydride-functional silane immobi-lized onto titanium surfaces induces osteoblast cell differentiation and reduces bacterial adhesion and biofilm formation. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 59, 524–532. [Google Scholar] [CrossRef]
- Zheng, D.; Neoh, K.G.; Shi, Z.; Kang, E.-T. Assessment of stability of surface anchors for antibacterial coatings and immobilized growth factors on titanium. J. Colloid Interface Sci. 2013, 406, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Pfeufer, N.Y.; Hofmann-Peiker, K.; Muhle, M.; Warnke, P.H.; Weigel, M.C.; Kleine, M. Bioactive coating of titanium surfaces with recombinant human beta-defensin-2 (rHubetaD2) may prevent bacterial colonization in orthopaedic surgery. J. Bone Joint Surg. Am. 2011, 93, 840–846. [Google Scholar]
- Dettin, M.; Bagno, A.; Gambaretto, R.; Iucci, G.; Conconi, M.T.; Tuccitto, N.; Menti, A.M.; Grandi, C.; Di Bello, C.; Licciardello, A.; et al. Covalent surface modification of titanium oxide with different adhesive peptides: Surface characterization and osteoblast-like cell adhesion. J. Biomed. Mater. Res. Part A 2009, 90, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Adden, N.; Gamble, L.J.; Castner, D.G.; Hoffmann, A.; Gross, G.; Menzel, H. Phosphonic Acid Monolayers for Binding of Bioactive Molecules to Titanium Surfaces. Langmuir 2006, 22, 8197–8204. [Google Scholar] [CrossRef] [PubMed]
- Porte-Durrieu, M.C.; Guillemot, F.; Pallu, S.; Labrugere, C.; Brouillaud, B.; Bareille, R.; Amedee, J.; Barthe, N.; Dard, M.; Baquey, C. Cyclo-(DfKRG) peptide grafting onto Ti-6Al-4V: Physical characterization and interest towards human osteoprogenitor cells adhesion. Biomaterials 2004, 25, 4837–4846. [Google Scholar] [CrossRef] [PubMed]
- Marín-Pareja, N.; Salvagni, E.; Guillem-Marti, J.; Aparicio, C.; Ginebra, M.-P. Collagen-functionalised titanium surfaces for biological sealing of dental implants: Effect of immobilisation process on fibroblasts response. Colloids Surf. B:Biointerfaces 2014, 122, 601–610. [Google Scholar] [CrossRef] [PubMed]
- Pegg, E.C.; Walker, G.S.; Scotchford, C.A.; Farrar, D.; Grant, D. Mono-functional aminosilanes as primers for peptide func-tionalization. J. Biomed. Mater. Res. Part A 2009, 90, 947–958. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Lerum, M.Z.; Chen, W. How To Prepare Reproducible, Homogeneous, and Hydrolytically Stable Aminosilane-Derived Layers on Silica. Langmuir 2011, 28, 416–423. [Google Scholar] [CrossRef]
- Pasternack, R.M.; Amy, S.R.; Chabal, Y.J. Attachment of 3-(Aminopropyl)triethoxysilane on Silicon Oxide Surfaces: Dependence on Solution Temperature. Langmuir 2008, 24, 12963–12971. [Google Scholar] [CrossRef]
- Meirelles, L.; Brånemark, P.-I.; Albrektsson, T.; Feng, C.; Johansson, C. Histological Evaluation of Bone Formation Adjacent to Dental Implants with a Novel Apical Chamber Design: Preliminary Data in the Rabbit Model. Clin. Implant. Dent. Relat. Res. 2013, 17, 453–460. [Google Scholar] [CrossRef]
- Xia, Z.; Nanjo, H.; Aizawa, T.; Kanakubo, M.; Fujimura, M.; Onagawa, J. Growth process of atomically flat anodic films on titanium under potentiostatical electrochemical treatment in H2SO4 solution. Surf. Sci. 2007, 601, 5133–5141. [Google Scholar] [CrossRef]
- Xing, J.-H.; Xia, Z.-B.; Hu, J.-F.; Zhang, Y.-H.; Zhong, L. Growth and Crystallization of Titanium Oxide Films at Different Anodization Modes. J. Electrochem. Soc. 2013, 160, C239–C246. [Google Scholar] [CrossRef]
- Elias, C.N.; Fernandes, D.J.; De Souza, F.M.; Monteiro, E.D.S.; De Biasi, R.S. Mechanical and clinical properties of titanium and titanium-based alloys (Ti G2, Ti G4 cold worked nanostructured and Ti G5) for biomedical applications. J. Mater. Res. Technol. 2019, 8, 1060–1069. [Google Scholar] [CrossRef]
- Cordeiro, J.M.; Barão, V.A. Is there scientific evidence favoring the substitution of commercially pure titanium with titanium alloys for the manufacture of dental implants? Mater. Sci. Eng. C 2017, 71, 1201–1215. [Google Scholar] [CrossRef] [PubMed]
- Nicholson. Titanium Alloys for Dental Implants: A Review. Prosthesis 2020, 2, 100–116. [Google Scholar] [CrossRef]
- Wennerberg, A.; Albrektsson, T. On implant surfaces: A review of current knowledge and opinions. Int. J. Oral Maxillofac. Implant. 2010, 25, 63–74. [Google Scholar]
- Sakeye, M.; Smått, J.-H. Comparison of Different Amino-Functionalization Procedures on a Selection of Metal Oxide Microparticles: Degree of Modification and Hydrolytic Stability. Langmuir 2012, 28, 16941–16950. [Google Scholar] [CrossRef]
- Rosenholm, J.M.; Lindén, M. Wet-Chemical Analysis of Surface Concentration of Accessible Groups on Different Ami-no-Functionalized Mesoporous SBA-15 Silicas. Chem. Mater. 2007, 19, 5023–5034. [Google Scholar] [CrossRef]
- Zhang, F.; Srinivasan, M.P. Self-Assembled Molecular Films of Aminosilanes and Their Immobilization Capacities. Langmuir 2004, 20, 2309–2314. [Google Scholar] [CrossRef]
- Imamura, K.; Shimomura, M.; Nagai, S.; Akamatsu, M.; Nakanishi, K. Adsorption characteristics of various proteins to a titanium surface. J. Biosci. Bioeng. 2008, 106, 273–278. [Google Scholar] [CrossRef]
- Dodo, C.G.; Senna, P.M.; Custodio, W.; Leme, A.F.P.; Cury, A.A.D.B. Proteome analysis of the plasma protein layer adsorbed to a rough titanium surface. Biofouling 2013, 29, 549–557. [Google Scholar] [CrossRef] [PubMed]
- Smith, E.A.; Chen, W. How To Prevent the Loss of Surface Functionality Derived from Aminosilanes. Langmuir 2008, 24, 12405–12409. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Senna, P.M.; de Almeida Barros Mourão, C.F.; Mello-Machado, R.C.; Javid, K.; Montemezzi, P.; Del Bel Cury, A.A.; Meirelles, L. Silane-Coating Strategy for Titanium Functionalization Does Not Impair Osteogenesis In Vivo. Materials 2021, 14, 1814. https://doi.org/10.3390/ma14071814
Senna PM, de Almeida Barros Mourão CF, Mello-Machado RC, Javid K, Montemezzi P, Del Bel Cury AA, Meirelles L. Silane-Coating Strategy for Titanium Functionalization Does Not Impair Osteogenesis In Vivo. Materials. 2021; 14(7):1814. https://doi.org/10.3390/ma14071814
Chicago/Turabian StyleSenna, Plinio Mendes, Carlos Fernando de Almeida Barros Mourão, Rafael Coutinho Mello-Machado, Kayvon Javid, Pietro Montemezzi, Altair Antoninha Del Bel Cury, and Luiz Meirelles. 2021. "Silane-Coating Strategy for Titanium Functionalization Does Not Impair Osteogenesis In Vivo" Materials 14, no. 7: 1814. https://doi.org/10.3390/ma14071814
APA StyleSenna, P. M., de Almeida Barros Mourão, C. F., Mello-Machado, R. C., Javid, K., Montemezzi, P., Del Bel Cury, A. A., & Meirelles, L. (2021). Silane-Coating Strategy for Titanium Functionalization Does Not Impair Osteogenesis In Vivo. Materials, 14(7), 1814. https://doi.org/10.3390/ma14071814






