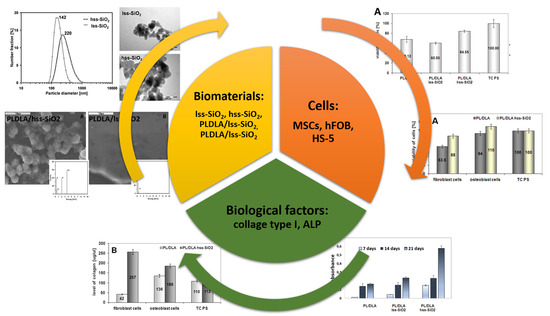
How Surface Properties of Silica Nanoparticles Influence Structural, Microstructural and Biological Properties of Polymer Nanocomposites
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
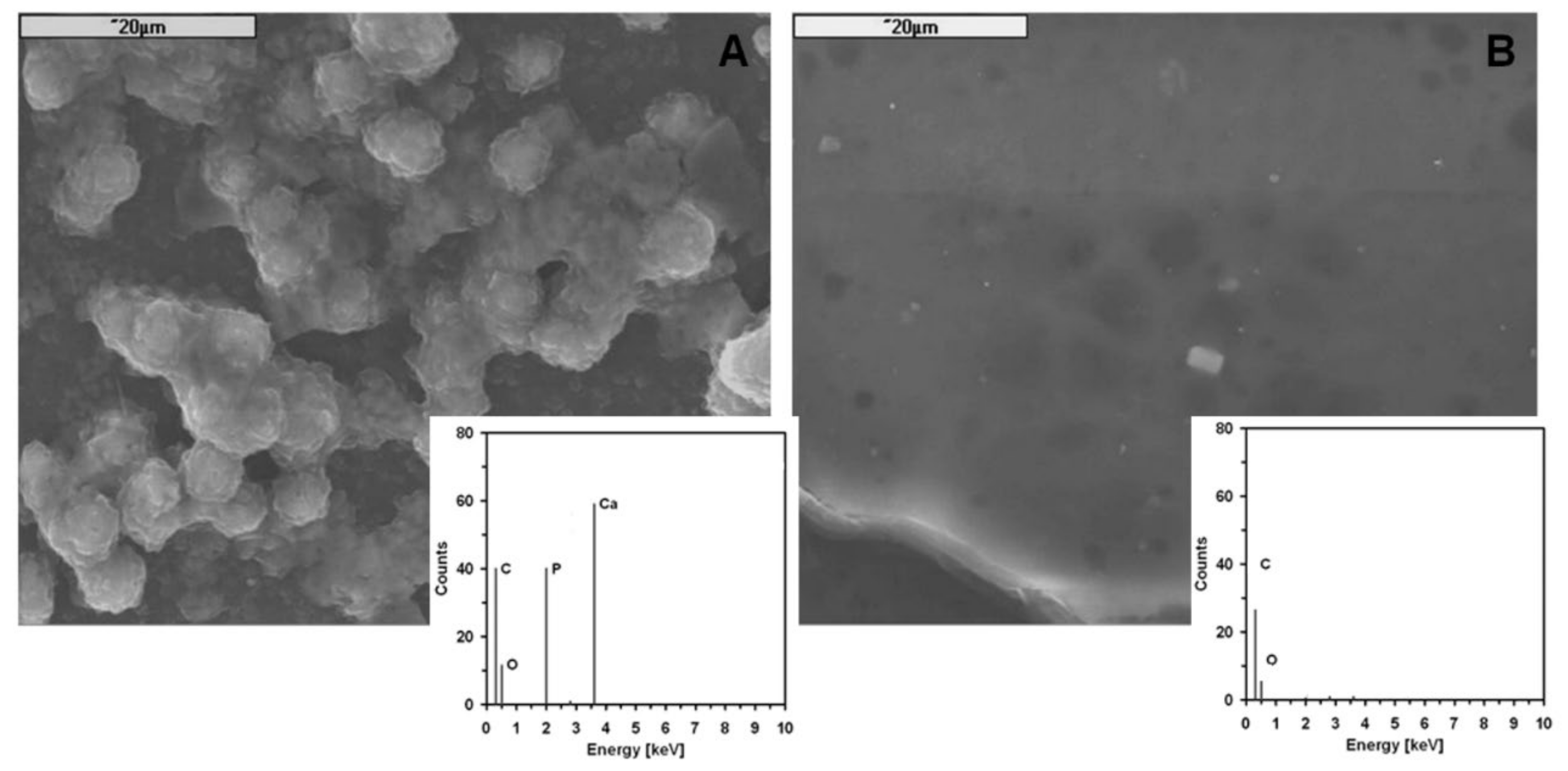
- the particles of the high specific surface silica (hss-SiO2) in polymer matrix (PLDLA) significantly enhanced the formation of apatite-like structures at the surface during a bioactivity test (in vitro conditions); this feature can be monitored by zeta potential in SBF;
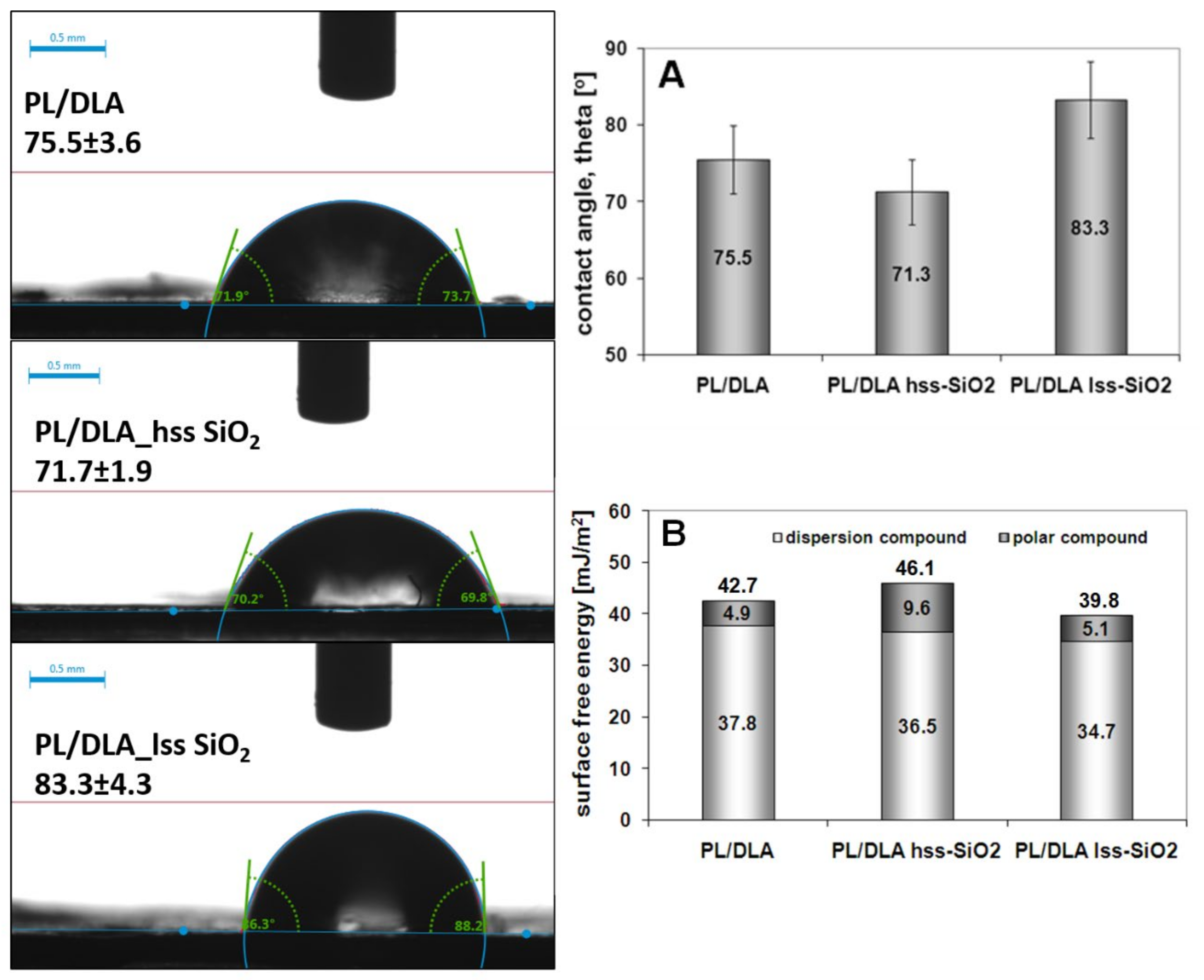
- the nanofillers such as hss-SiO2 significantly influence physicochemical properties such as wetting contact angle and surface free energy of polymer matrix, and slightly influence the crystallinity of nanocomposite materials (PLDLA/hss-SiO2). These parameters may be relevant to the biological response from both the mesenchymal and somatic cells;
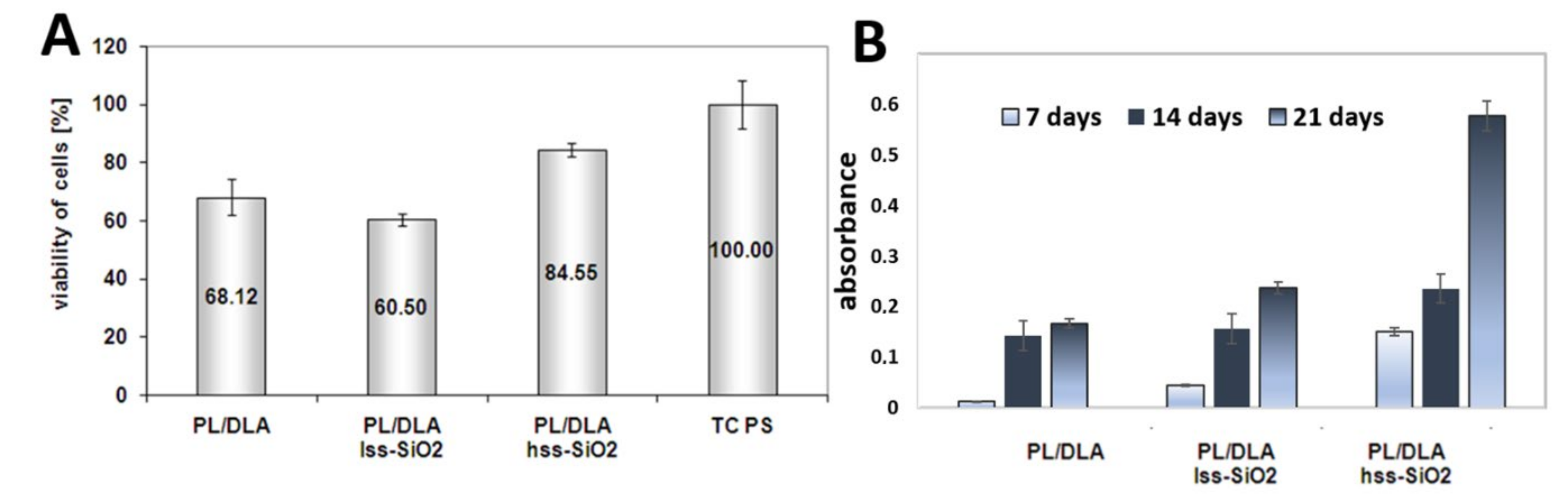
- the results of biological investigations, realised with the use of mesenchymal stem cells, show that the bioactive nanocomposite where hss-SiO2 was used as a filler may stimulate differentiation of mesenchymal cells into osteoblasts—resulting in higher proliferation stage of cells in in vitro conditions and higher alkaline phosphatase activity. Somatic cells: fibroblast and osteoblast contacted with nanocomposite with hss-SiO2 confirm that this material is much more suitable for promotion of cells’ proliferation than PLDLA/lss-SiO2.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fernandez de Grado, G.; Keller, L.; Idoux-Gillet, Y.; Wagner, Q.; Musset, A.-M.; Benkirane-Jessel, N.; Bornert, F.; Offner, D. Bone substitutes: A review of their characteristics, clinical use, and perspectives for large bone defects management. J. Tissue. Eng. 2018, 9, 2041731418776819. [Google Scholar] [CrossRef]
- Faour, O.; Dimitriou, R.; Cousins, C.A.; Giannoudis, P.V. The use of bone graft substitutes in large cancellous voids: Any specific needs? Injury 2011, 42, 87–90. [Google Scholar] [CrossRef] [PubMed]
- Campana, V.; Milano, G.; Pagano, E.; Barba, M.; Cicione, C.; Salonna, G.; Lattanzi, W.; Logroscino, G. Bone substitutes in orthopaedic surgery: From basic science to clinical practice. J. Mater. Sci. Mater. Med. 2014, 25, 2445–2461. [Google Scholar] [CrossRef]
- Giannoudis, P.V.; Dinopoulos, H.; Tsiridis, E. Bone substitutes: An update. Injury 2005, 36, 20–27. [Google Scholar] [CrossRef]
- Wagner, Q.; Offner, D.; Idoux-Gillet, Y.; Saleem, I.; Somavarapu, S.; Schwinté, P.; Benkirane-Jessel, N.; Keller, L. Advanced nanostructured medical device combining mesenchymal cells and VEGF nanoparticles for enhanced engineered tissue vascularization. Nanomed. (Lond.) 2016, 11, 2419–2430. [Google Scholar] [CrossRef]
- Schmidt-Bleek, K.; Schell, H.; Schulz, N.; Hoff, P.; Perka, C.; Buttgereit, F.; Volk, H.-D.; Lienau, J.; Duda, G.N. Inflammatory phase of bone healing initiates the regenerative healing cascade. Cell Tissue Res. 2012, 347, 567–573. [Google Scholar] [CrossRef] [PubMed]
- Offner, D.; Wagner, Q.; Idoux-Gillet, Y.; Gegout, H.; Ferrandon, A.; Schwinté, P.; Musset, A.-M.; Benkirane-Jessel, N.; Keller, L. Hybrid collagen sponge and stem cells as a new combined scaffold able to induce the re-organization of endothelial cells into clustered networks. Biomed. Mater. Eng. 2017, 28, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Götz, W.; Reichert, C.; Canullo, L.; Jäger, A.; Heinemann, F. Coupling of osteogenesis and angiogenesis in bone substitute healing—A brief overview. Ann. Anat. 2012, 194, 171–173. [Google Scholar] [CrossRef]
- Lu, J.X.; Flautre, B.; Anselme, K.; Hardouin, P.; Gallur, A.; Descamps, M.; Thierry, B. Role of interconnections in porous bioceramics on bone recolonization in vitro and in vivo. J. Mater. Sci. Mater. Med. 1999, 10, 111–120. [Google Scholar] [CrossRef]
- Glowacki, J. Angiogenesis in fracture repair. Clin. Orthop Relat. Res. 1998, 82–89. [Google Scholar] [CrossRef]
- Geiger, F.; Beverungen, M.; Lorenz, H.; Wieland, J.; Fehr, M.; Kasten, P. Bone Substitute Effect on Vascularization and Bone Remodeling after Application of phVEGF165 Transfected BMSC. J. Funct. Biomater. 2012, 3, 313–326. [Google Scholar] [CrossRef] [PubMed]
- Le Huec, J.C.; Schaeverbeke, T.; Clement, D.; Faber, J.; Le Rebeller, A. Influence of porosity on the mechanical resistance of hydroxyapatite ceramics under compressive stress. Biomaterials 1995, 16, 113–118. [Google Scholar] [CrossRef]
- Yuan, H.; Kurashina, K.; de Bruijn, J.D.; Li, Y.; de Groot, K.; Zhang, X. A preliminary study on osteoinduction of two kinds of calcium phosphate ceramics. Biomaterials 1999, 20, 1799–1806. [Google Scholar] [CrossRef]
- Galois, L.; Mainard, D. Bone ingrowth into two porous ceramics with different pore sizes: An experimental study. Acta Orthop. Belg. 2004, 70, 598–603. [Google Scholar]
- Roberts, T.T.; Rosenbaum, A.J. Bone grafts, bone substitutes and orthobiologics: The bridge between basic science and clinical advancements in fracture healing. Organogenesis 2012, 8, 114–124. [Google Scholar] [CrossRef]
- Kargozar, S.; Montazerian, M.; Fiume, E.; Baino, F. Multiple and Promising Applications of Strontium (Sr)-Containing Bioactive Glasses in Bone Tissue Engineering. Front. Bioeng. Biotechnol. 2019, 7, 161. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, M.D.; Hill, R.G. Influence of strontium and the importance of glass chemistry and structure when designing bioactive glasses for bone regeneration. Acta Biomater. 2010, 6, 2382–2385. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Zhou, Y.; Lin, C.; Chang, J.; Xiao, Y. Strontium-containing mesoporous bioactive glass scaffolds with improved osteogenic/cementogenic differentiation of periodontal ligament cells for periodontal tissue engineering. Acta Biomater. 2012, 8, 3805–3815. [Google Scholar] [CrossRef] [PubMed]
- Li, H.C.; Wang, D.G.; Chen, C.Z. Effect of zinc oxide and zirconia on structure, degradability and in vitro bioactivity of wollastonite. Ceram. Int. 2015, 41, 10160–10169. [Google Scholar] [CrossRef]
- Webster, T.J.; Ergun, C.; Doremus, R.H.; Siegel, R.W.; Bizios, R. Enhanced functions of osteoblasts on nanophase ceramics. Biomaterials 2000, 21, 1803–1810. [Google Scholar] [CrossRef]
- Hong, Z.; Zhang, P.; He, C.; Qiu, X.; Liu, A.; Chen, L.; Chen, X.; Jing, X. Nano-composite of poly(L-lactide) and surface grafted hydroxyapatite: Mechanical properties and biocompatibility. Biomaterials 2005, 26, 6296–6304. [Google Scholar] [CrossRef]
- Carson, J.S.; Bostrom, M.P.G. Synthetic bone scaffolds and fracture repair. Injury 2007, 38, 33–37. [Google Scholar] [CrossRef]
- Zhang, P.; Hong, Z.; Yu, T.; Chen, X.; Jing, X. In Vivo mineralization and osteogenesis of nanocomposite scaffold of poly(lactide-co-glycolide) and hydroxyapatite surface-grafted with poly(L-lactide). Biomaterials 2009, 30, 58–70. [Google Scholar] [CrossRef] [PubMed]
- Kotela, I.; Podporska, J.; Soltysiak, E.; Konsztowicz, K.J.; Blazewicz, M. Polymer nanocomposites for bone tissue substitutes. Ceram. Int. 2009, 35, 2475–2480. [Google Scholar] [CrossRef]
- Stodolak, E.; Szumera, M.; Blazewicz, M. Osteoconductive Nanocomposite Materials for Bone Regeneration. Mater. Sci. Forum. 2012, 730–732, 38–43. [Google Scholar] [CrossRef]
- Stodolak-Zych, E.; Łuszcz, A.; Menaszek, E.; Ścisłowska-Czarencka, A. Resorbable Polymer Membranes for Medical Applications. J. Biomim. Biomater. Biomed. Eng. 2014, 19, 99–108. [Google Scholar] [CrossRef]
- Yan, S.; Yin, J.; Yang, Y.; Dai, Z.; Ma, J.; Chen, X. Surface-grafted silica linked with l-lactic acid oligomer: A novel nanofiller to improve the performance of biodegradable poly(l-lactide). Polymer 2007, 48, 1688–1694. [Google Scholar] [CrossRef]
- Lord, M.S.; Cousins, B.G.; Doherty, P.J.; Whitelock, J.M.; Simmons, A.; Williams, R.L.; Milthorpe, B.K. The effect of silica nanoparticulate coatings on serum protein adsorption and cellular response. Biomaterials 2006, 27, 4856–4862. [Google Scholar] [CrossRef]
- Vijayalakshmi, D.; Balamurugan, A.; Rajeswari, S. Synthesis and Characterization of Porous Silica Gels for Biomedical Applications. Trends Biomater. Artif. Organs 2005, 18, 101–106. [Google Scholar]
- Ballarre, J.; López, A.J.; Cavalieri, A. Nano-indentation of hybrid silica coatings on surgical grade stainless steel. Thin Solid Films 2008, 516, 1082–1087. [Google Scholar] [CrossRef]
- Perry, C.C. An overview of silica in biology: Its chemistry and recent technological advances. Prog. Mol. Subcell. Biol. 2009, 47, 295–313. [Google Scholar] [CrossRef] [PubMed]
- Niu, L.-N.; Jiao, K.; Ryou, H.; Diogenes, A.; Yiu, C.K.Y.; Mazzoni, A.; Chen, J.-H.; Arola, D.D.; Hargreaves, K.M.; Pashley, D.H.; et al. Biomimetic silicification of demineralized hierarchical collagenous tissues. Biomacromolecules 2013, 14, 1661–1668. [Google Scholar] [CrossRef] [PubMed]
- Zhu, A.; Diao, H.; Rong, Q.; Cai, A. Preparation and properties of polylactide–silica nanocomposites. J. Appl. Polym. Sci. 2010, 116, 2866–2873. [Google Scholar] [CrossRef]
- Li, P.; Ohtsuki, C.; Kokubo, T.; Nakanishi, K.; Soga, N.; Nakamura, T.; Yamamuro, T. Apatite Formation Induced by Silica Gel in a Simulated Body Fluid. J. Am. Ceram. Soc. 1992, 75, 2094–2097. [Google Scholar] [CrossRef]
- Coreño, J.; Rivera, E.; Castaño, V.; Rodríguez, R. Apatite growth on calcium adsorbed surface of wet flocculated silica particles immersed in a modified simulated body fluid. J. Biomed. Mater. Res. 2000, 53, 44–50. [Google Scholar] [CrossRef]
- Galli, C.; Collaud Coen, M.; Hauert, R.; Katanaev, V.L.; Gröning, P.; Schlapbach, L. Creation of nanostructures to study the topographical dependency of protein adsorption. Colloids Surf. B 2002, 26, 255–267. [Google Scholar] [CrossRef]
- Lu, H.H.; Pollack, S.R.; Ducheyne, P. Temporal zeta potential variations of 45S5 bioactive glass immersed in an electrolyte solution. J. Biomed. Mater. Res. 2000, 51, 80–87. [Google Scholar] [CrossRef]
- Kokubo, T.; Kim, H.-M.; Kawashita, M. Novel bioactive materials with different mechanical properties. Biomaterials 2003, 24, 2161–2175. [Google Scholar] [CrossRef]
- Rainer, A.; Giannitelli, S.M.; Abbruzzese, F.; Traversa, E.; Licoccia, S.; Trombetta, M. Fabrication of bioactive glass–ceramic foams mimicking human bone portions for regenerative medicine. Acta Biomat. 2008, 4, 362–369. [Google Scholar] [CrossRef]
- Kokubo, T.; Takadama, H. How useful is SBF in predicting in vivo bone bioactivity? Biomaterials 2006, 27, 2907–2915. [Google Scholar] [CrossRef]
- Robey, P.G.; Termine, J.D. Human bone cellsin vitro. Calcif. Tissue Int. 1985, 37, 453–460. [Google Scholar] [CrossRef]
- Sinha, R.K.; Morris, F.; Shah, S.A.; Tuan, R.S. Surface Composition of Orthopaedic Implant Metals Regulates Cell Attachment, Spreading, and Cytoskeletal Organization of Primary Human Osteoblasts In Vitro. Clin. Orthop. Related Res. 1994, 305, 258–272. [Google Scholar] [CrossRef]
- Stodolak, E.; Paluszkiewicz, C.; Bogun, M.; Blazewicz, M. Nanocomposite fibres for medical applications. J. Mol. Struc. 2009, 924–926, 208–213. [Google Scholar] [CrossRef]
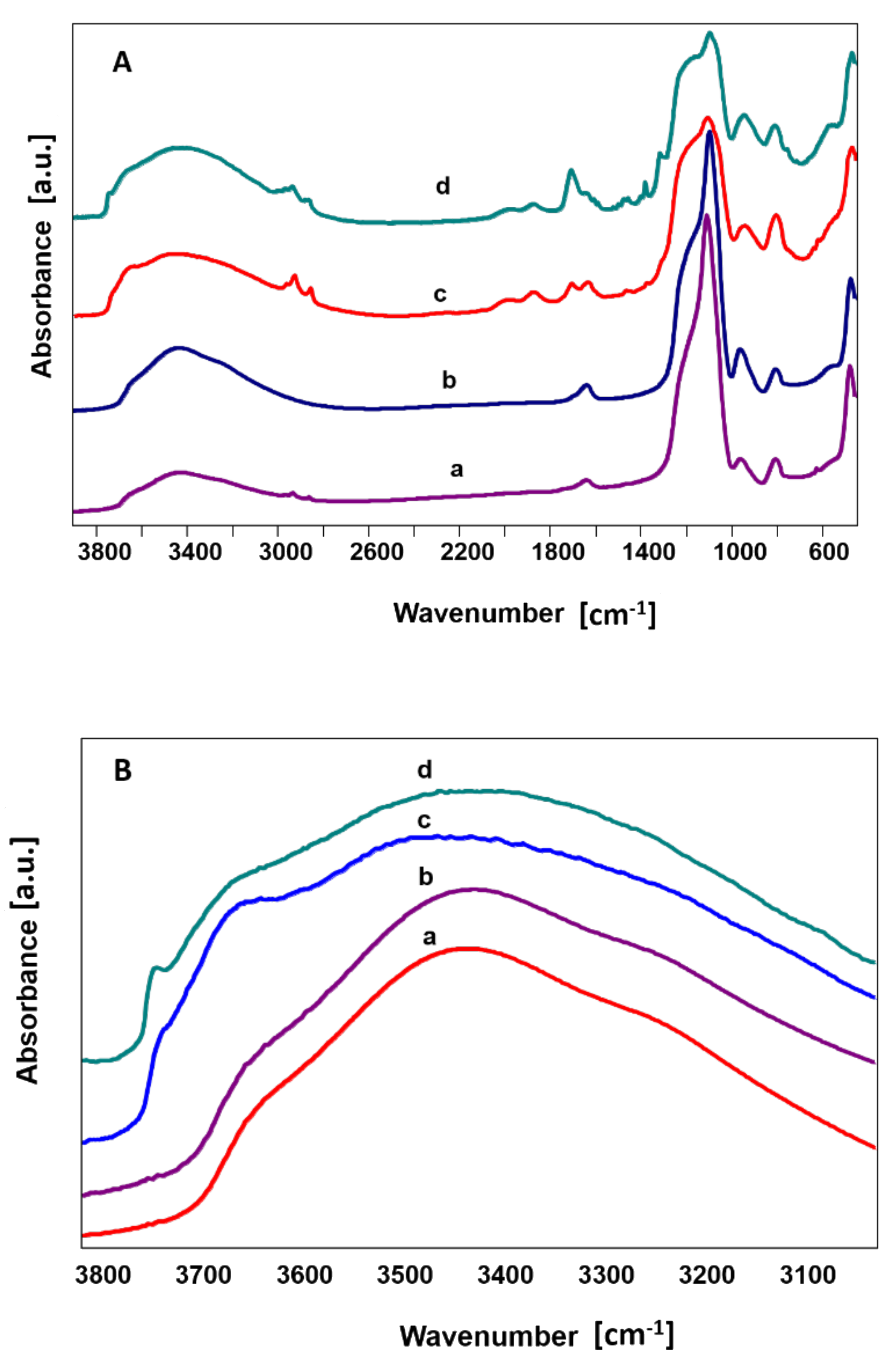
- Sitarz, M.; Handke, M.; Mozgawa, W. Identification of silicooxygen rings in SiO2 based on IR spectra. Spectrochim. Acta Part. A 2000, 56, 1819–1823. [Google Scholar] [CrossRef]
- Xu, T.; Zhang, N.; Nichols, H.; Shi, D.; Wen, X. Modification of nanostructured materials for biomedical applications. J. Mater. Sci. Eng. C. 2007, 27, 579–594. [Google Scholar] [CrossRef]
- Beck, G.R.; Ha, S.-W.; Camalier, C.E.; Yamaguchi, M.; Li, Y.; Lee, J.-K.; Weitzmann, M.N. Bioactive silica-based nanoparticles stimulate bone-forming osteoblasts, suppress bone-resorbing osteoclasts, and enhance bone mineral density in vivo. Nanomed. Nanotechnol. Biol. Med. 2012, 8, 793–803. [Google Scholar] [CrossRef]
- Cho, S.-B.; Nakanishi, K.; Kokubo, T.; Soga, N.; Ohtsuki, C.; Nakamura, T. Apatite formation on silica gel in simulated body fluid: Its dependence on structures of silica gels prepared in different media. J. Biomed. Mater. Res. 1996, 33, 145–151. [Google Scholar] [CrossRef]
- Yamamuro, T.; Hench, L.L.; Wilson, J. Handbook of Bioactive Ceramics; CRC-Press: Oxfordshire, UK, 1990; ISBN 978-0-8493-3241-8. [Google Scholar]
- Ohtsuki, C.; Kokubo, T.; Yamamuro, T. Mechanism of apatite formation on CaOSiO2P2O5 glasses in a simulated body fluid. J. Non-Cryst. Solids 1992, 143, 84–92. [Google Scholar] [CrossRef]
- Andersson, Ö.H.; Karlsson, K.H. On the bioactivity of silicate glass. J. Non-Cryst. Solids 1991, 129, 145–151. [Google Scholar] [CrossRef]
- Iler, R.K. Coagulation of colloidal silica by calcium ions, mechanism, and effect of particle size. J. Colloid. Interface Sci. 1975, 53, 476–488. [Google Scholar] [CrossRef]
- Kokubo, T. Bioactive glass ceramics: Properties and applications. Biomaterials 1991, 12, 155–163. [Google Scholar] [CrossRef]
- Wu, T.-M.; Wu, C.-Y. Biodegradable poly (lactic acid)/chitosan-modified montmorillonite nanocomposites: Preparation and characterization. Polym. Degrad. Stab. 2006, 91, 2198–2204. [Google Scholar] [CrossRef]
- Maiti, P.; Yamada, K.; Okamoto, M.; Ueda, K.; Okamoto, K. New Polylactide/Layered Silicate Nanocomposites: Role of Organoclays. Chem. Mater. 2002, 14, 4654–4661. [Google Scholar] [CrossRef]
- Moreno, A.J.; Bacova, P.; Verso, F.L.; Arbe, A.; Colmenero, J.; Pomposo, J.A. Effect of chain stiffness on the structure of single-chain polymer nanoparticles. J. Phys. Condens. Matter. 2017, 30, 034001. [Google Scholar] [CrossRef] [PubMed]
- Khan, J.; Harton, S.E.; Akcora, P.; Benicewicz, B.C.; Kumar, S.K. Polymer Crystallization in Nanocomposites: Spatial Reorganization of Nanoparticles. Macromolecules 2009, 42, 5741–5744. [Google Scholar] [CrossRef]
- Zare, Y. Study of nanoparticles aggregation/agglomeration in polymer particulate nanocomposites by mechanical properties. Compos. Part. A 2016, 84, 158–164. [Google Scholar] [CrossRef]
- Hench, L.L. Bioceramics: From Concept to Clinic. J. Am. Ceram. Soc. 1991, 74, 1487–1510. [Google Scholar] [CrossRef]
- Ducheyne, P. Bioceramics: Material characteristics versus in vivo behavior. J. Biomed. Mater. Res. 1987, 21, 219–236. [Google Scholar] [PubMed]
- Cho, S.B.; Miyaji, F.; Kokubo, T.; Nakanishi, K.; Soga, N.; Nakamura, T. Apatite formation on silica gel in simulated body fluid: Effects of structural modification with solvent-exchange. J. Mater. Sci. Mater. Med. 1998, 9, 279–284. [Google Scholar] [CrossRef]
- Coreño, J.; Martínez, A.; Coreño, O.; Bolarín, A.; Sánchez, F. Calcium and phosphate adsorption as initial steps of apatite nucleation on sol-gel-prepared titania surface. J. Biomed. Mat. Res. Part. A 2003, 64, 131–137. [Google Scholar] [CrossRef]
- Goddard, J.M.; Hotchkiss, J.H. Polymer surface modification for the attachment of bioactive compounds. Prog. Polym. Sci. 2007, 32, 698–725. [Google Scholar] [CrossRef]
- Webster, T.J.; Siegel, R.W.; Bizios, R. Nanoceramic surface roughness enhances osteoblast and osteoclast functions for improved orthopaedic/dental implant efficacy. Scrip. Mater. 2001, 44, 1639–1642. [Google Scholar] [CrossRef]
- Hong, Z.; Qiu, X.; Sun, J.; Deng, M.; Chen, X.; Jing, X. Grafting polymerization of l-lactide on the surface of hydroxyapatite nano-crystals. Polymer 2004, 45, 6699–6706. [Google Scholar] [CrossRef]
- Gomillion, C.T.; Burg, K.J.L. Stem cells and adipose tissue engineering. Biomaterials 2006, 27, 6052–6063. [Google Scholar] [CrossRef] [PubMed]
- Cenni, E.; Ciapetti, G.; Granchi, D.; Arciola, C.R.; Savarino, L.; Stea, S.; Montanaro, L.; Pizzoferrato, A. Established Cell Lines and Primary Cultures in Testing Medical Devices In Vitro. Toxicol. In Vitro 1999, 13, 801–810. [Google Scholar] [CrossRef]
- De Boer, J.; Siddappa, R.; Gaspar, C.; van Apeldoorn, A.; Fodde, R.; van Blitterswijk, C. Wnt signaling inhibits osteogenic differentiation of human mesenchymal stem cells. Bone 2004, 34, 818–826. [Google Scholar] [CrossRef]
- Zhou, X.; Feng, W.; Qiu, K.; Chen, L.; Wang, W.; Nie, W.; Mo, X.; He, C. BMP-2 Derived Peptide and Dexamethasone Incorporated Mesoporous Silica Nanoparticles for Enhanced Osteogenic Differentiation of Bone Mesenchymal Stem Cells. ACS Appl. Mater. Interfaces 2015, 7, 15777–15789. [Google Scholar] [CrossRef] [PubMed]
- Gandhimathi, C.; Quek, Y.J.; Ezhilarasu, H.; Ramakrishna, S.; Bay, B.-H.; Srinivasan, D.K. Osteogenic Differentiation of Mesenchymal Stem Cells with Silica-Coated Gold Nanoparticles for Bone Tissue Engineering. Int. J. Mol. Sci. 2019, 20, 5135. [Google Scholar] [CrossRef]





| Fluid | Concentration of Ion (mM) | |||||||
|---|---|---|---|---|---|---|---|---|
| Cl− | HCO3− | HPO42− | SO42− | Na+ | K+ | Mg2+ | Ca2+ | |
| Body Plasma | 103.0 | 27.0 | 1.0 | 0.5 | 142.0 | 5.0 | 1.5 | 2.5 |
| Simulated Body Fluid, SBF | 148.8 | 4.2 | 1.0 | 0.5 | 142.0 | 5.0 | 1.5 | 2.5 |
| Nanoparticles Characteristics | hss-SiO2 | lss-SiO2 |
|---|---|---|
| Modal particle diameter, DLS (nm) | 220 | 142 |
| Mean particle size, TEM (nm) | 60 | 50 |
| Specific surface area, BET (m2/g) | 582.8 | 65.8 |
| Material | Tg, °C | Tm, °C | Tdeg, °C | λ, % |
|---|---|---|---|---|
| PL/DLA | 57.3 | 162.5 | 356.7 | 36.5 |
| PL/DLA hss-SiO2 | 59.8 | 168.2 | 350.3 | 40.2 |
| PL/DLA lss-SiO2 | 57.2 | 161.8 | 348.2 | 37.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zych, Ł.; Osyczka, A.M.; Łacz, A.; Różycka, A.; Niemiec, W.; Rapacz-Kmita, A.; Dzierzkowska, E.; Stodolak-Zych, E. How Surface Properties of Silica Nanoparticles Influence Structural, Microstructural and Biological Properties of Polymer Nanocomposites. Materials 2021, 14, 843. https://doi.org/10.3390/ma14040843
Zych Ł, Osyczka AM, Łacz A, Różycka A, Niemiec W, Rapacz-Kmita A, Dzierzkowska E, Stodolak-Zych E. How Surface Properties of Silica Nanoparticles Influence Structural, Microstructural and Biological Properties of Polymer Nanocomposites. Materials. 2021; 14(4):843. https://doi.org/10.3390/ma14040843
Chicago/Turabian StyleZych, Łukasz, Anna Maria Osyczka, Agnieszka Łacz, Agnieszka Różycka, Wiktor Niemiec, Alicja Rapacz-Kmita, Ewa Dzierzkowska, and Ewa Stodolak-Zych. 2021. "How Surface Properties of Silica Nanoparticles Influence Structural, Microstructural and Biological Properties of Polymer Nanocomposites" Materials 14, no. 4: 843. https://doi.org/10.3390/ma14040843
APA StyleZych, Ł., Osyczka, A. M., Łacz, A., Różycka, A., Niemiec, W., Rapacz-Kmita, A., Dzierzkowska, E., & Stodolak-Zych, E. (2021). How Surface Properties of Silica Nanoparticles Influence Structural, Microstructural and Biological Properties of Polymer Nanocomposites. Materials, 14(4), 843. https://doi.org/10.3390/ma14040843