Green Nanocoatings Based on the Deposition of Zirconium Oxide: The Role of the Substrate
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation and Cleaning of Aluminium Substrates
2.3. Zirconium Oxide Nanocoating Depositions (DC and Zr-EAD)
2.4. Physical and Chemical Characterization
2.5. Electrochemical Characterization of Zirconium Oxide Nanocoating
3. Results and Discussion
3.1. Influence of the Substrate in the Deposition of Zr-EAD Films
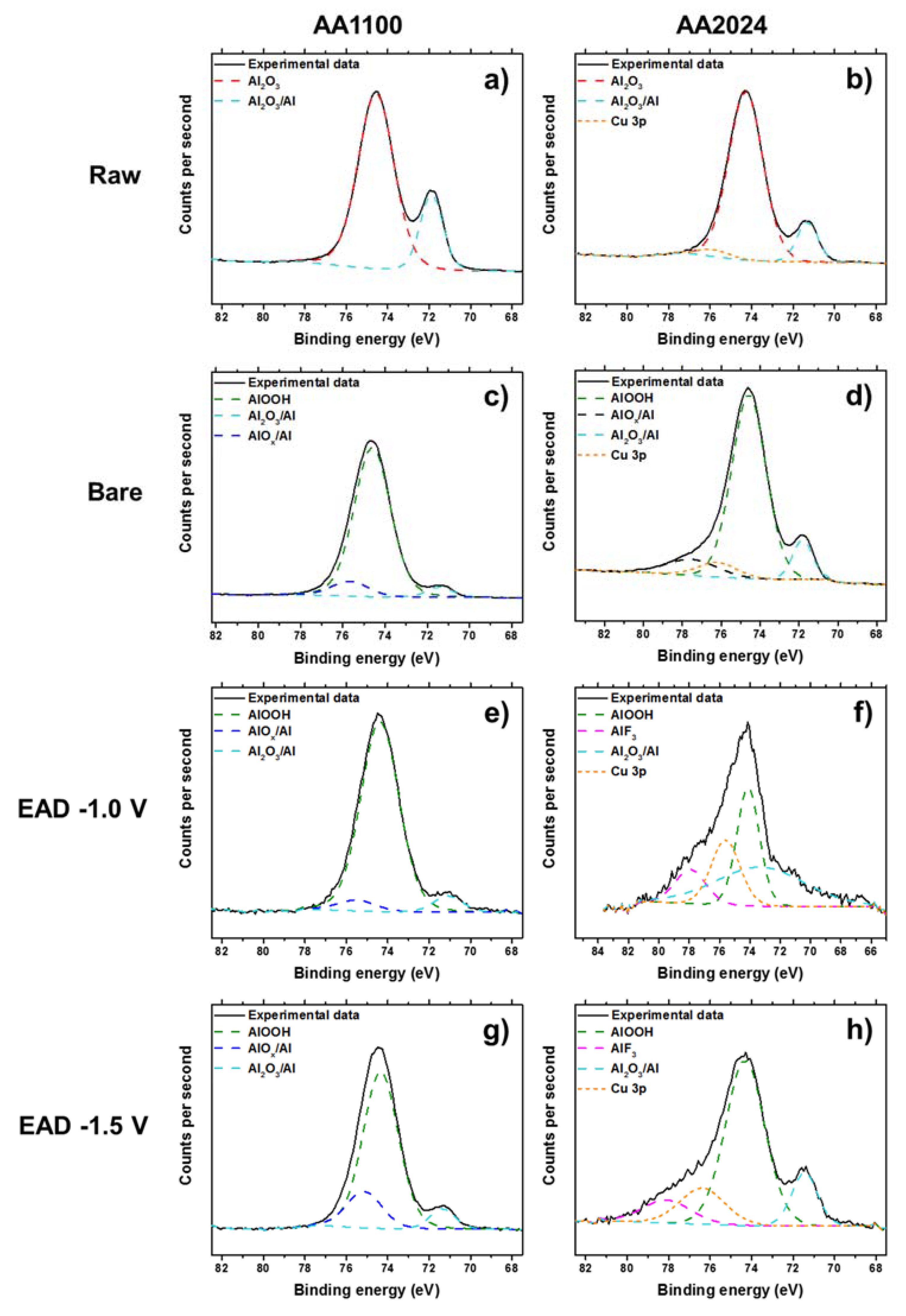
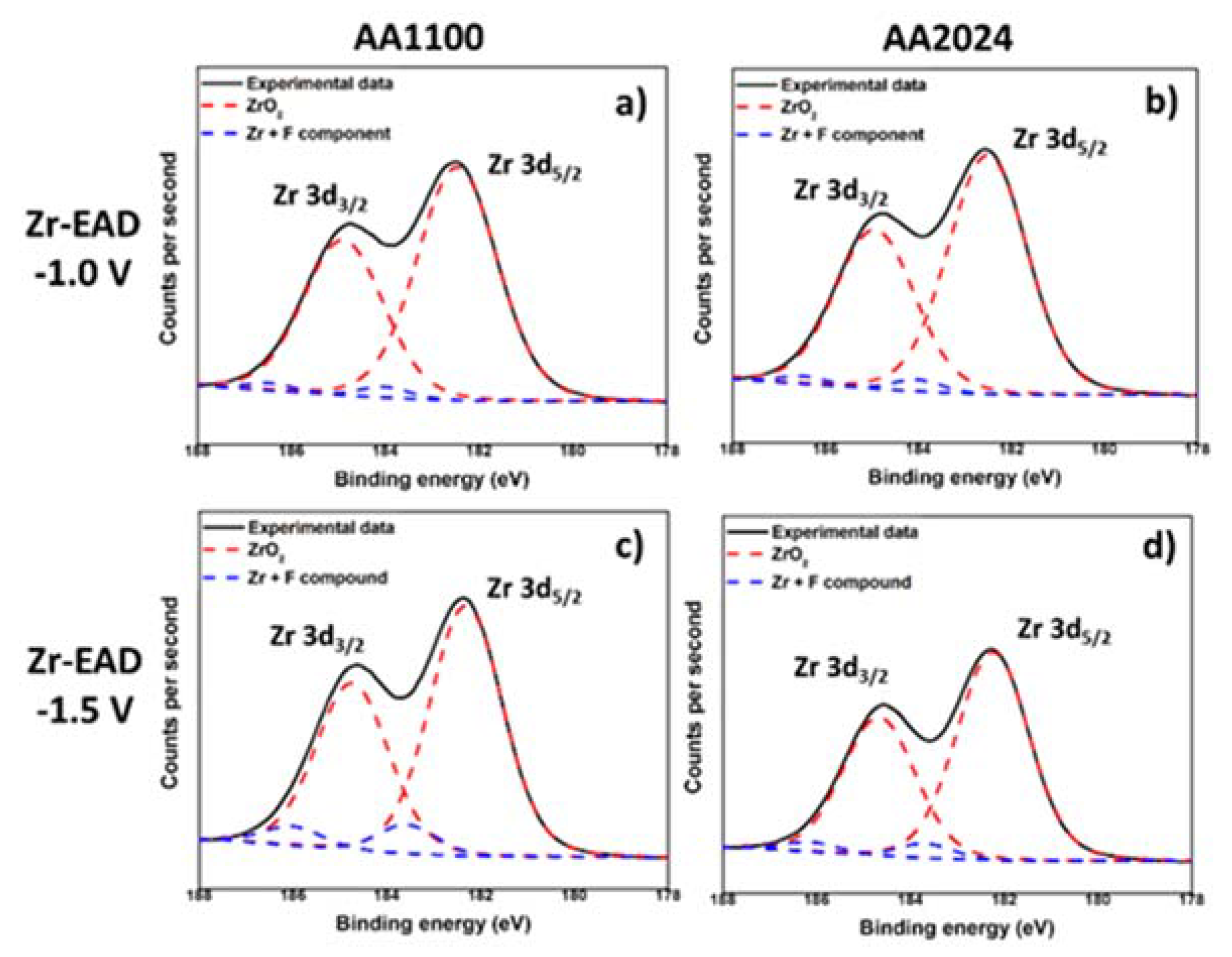
3.2. Zr-EAD Nanocoating Composition
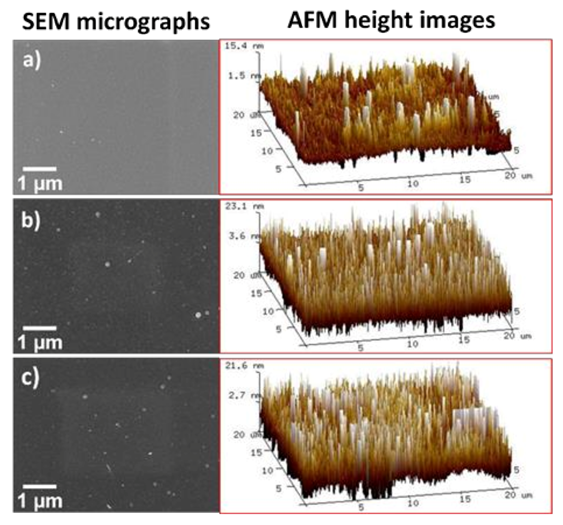
3.3. Zr-EAD Nanocoating Surface Topography
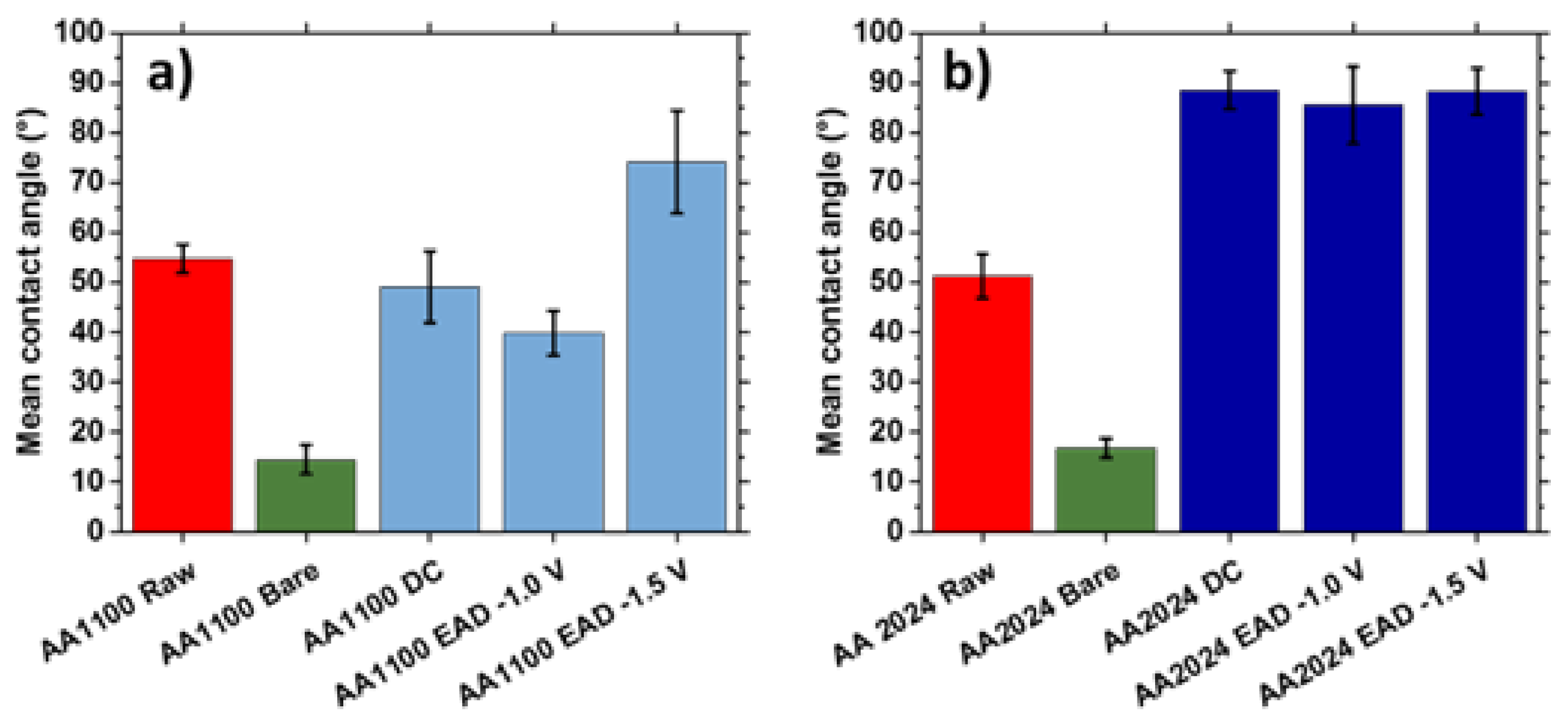
3.4. Wettability
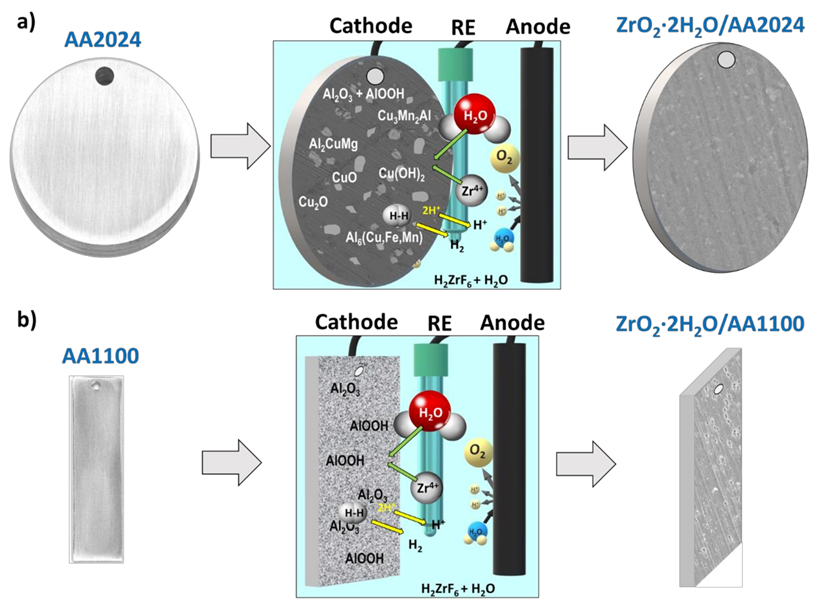
3.5. Mechanism of ZrO2 Deposition in AA1100 and AA2024 Surfaces
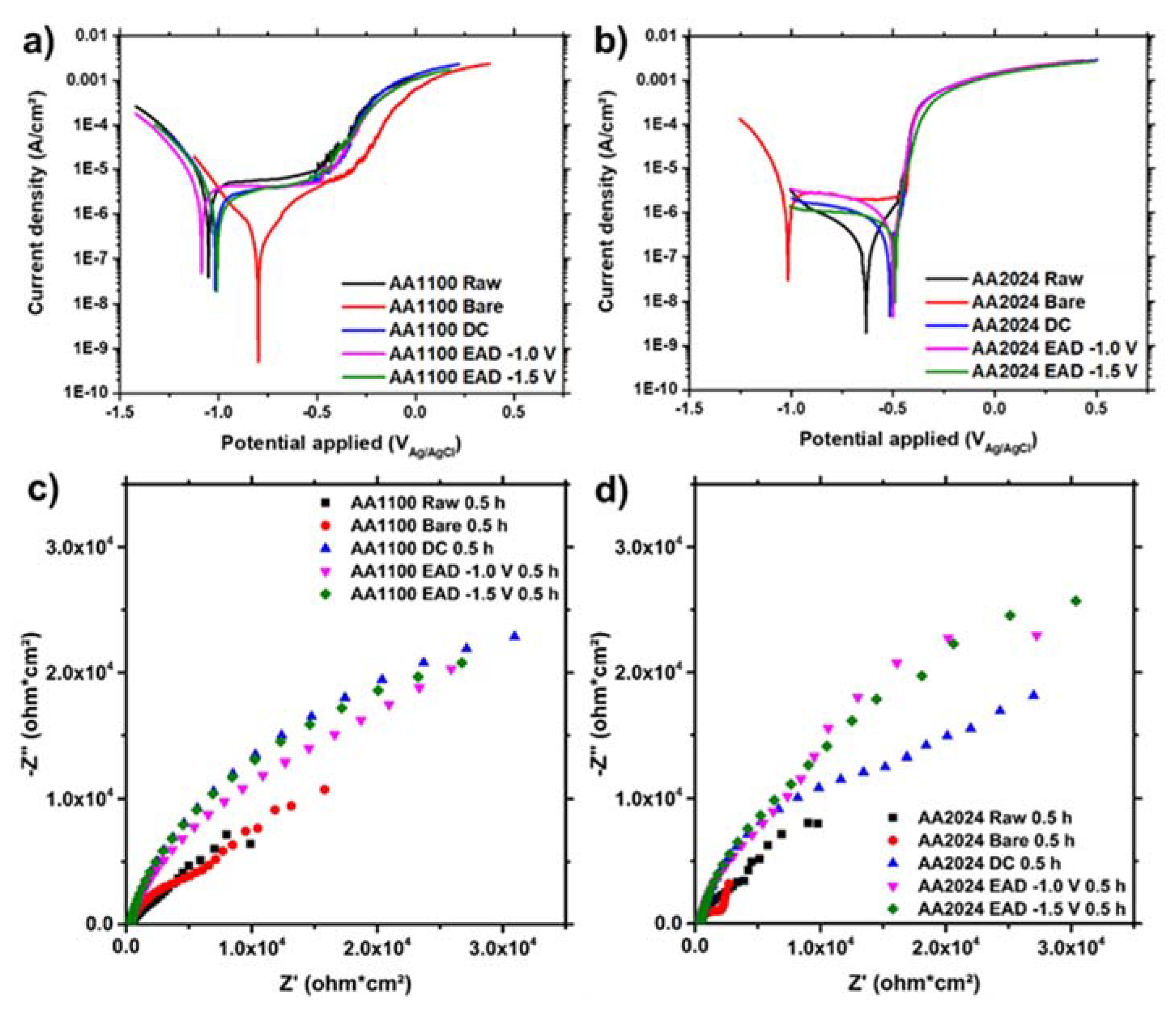
3.6. ZrO2 Nanocoating Stability in Sodium Chloride Solution
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Streitberger, H.-J.; Goldschmidt, A. BASF Handbook Basics of Coating Technology, 3rd ed.; Streitberger, H.-J., Goldschmidt, A., Eds.; European Coatings: Munster, Germany, 2018. [Google Scholar]
- Schütze, M.; Wieser, D.; Bender, B. Corrosion Resistance of Aluminium and Aluminium Alloys; Schütze, M., Wieser, D., Bender, B., Eds.; John Wiley & Sons, Ltd.: Frankfurt, Germany, 2010. [Google Scholar]
- Narayanan, T.S.N.S. Surface pretreatment by phosphate conversion coatings—A review. Rev. Adv. Mater. Sci. 2005, 9, 130–177. [Google Scholar]
- Freeman, D.B. Phosphating and Metal Pre-Treatment: A Guide to Modern Processes and Practice; Freeman, D.B., Ed.; Industrial Press: Michigan, USA, 1986; ISBN 0831111682. [Google Scholar]
- Martínez-Viademonte, M.P.; Abrahami, S.T.; Hack, T.; Burchardt, M.; Terryn, H. A review on anodizing of aerospace aluminum alloys for corrosion protection. Coatings 2020, 10, 1106. [Google Scholar] [CrossRef]
- Sander, J.; Kirmaier, L.; Manea, M.; Shchukin, D.; Skorb, E. Anticorrosive Coatings: Fundamentals and New Concepts; Sander, J., Kirmaier, L., Manea, M., Shchukin, D., Skorb, E., Eds.; Vincentz Network: Hanover, Germany, 2010; ISBN 9783866309111. [Google Scholar]
- Nicole, L.; Laberty-Robert, C.; Rozes, L.; Sanchez, C. Hybrid materials science: A promised land for the integrative design of multifunctional materials. Nanoscale 2014, 6, 6267–6292. [Google Scholar] [CrossRef]
- Kim, M.M.; Kapun, B.; Tiringer, U.; Šekularac, G.; Milošev, I. Protection of aluminum alloy 3003 in sodium chloride and simulated acid rain solutions by commercial conversion coatings containing Zr and Cr. Coatings 2019, 9, 563. [Google Scholar] [CrossRef]
- Milošev, I.; Frankel, G.S. Conversion Coatings Based on Zirconium and/or Titanium. J. Electrochem. Soc. 2018, 165, C127–C144. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, X.; Zhao, X.; Tang, Y.; Zuo, Y. Preparation of Ti-Zr-based conversion coating on 5052 aluminum alloy, and its corrosion resistance and antifouling performance. Coatings 2018, 8, 397. [Google Scholar] [CrossRef]
- Eivaz Mohammadloo, H.; Sarabi, A.A.; Asemani, H.R.; Ahmadi, P. A comparative study of eco-friendly hybrid thin films: With and without organic coating application. Prog. Org. Coat. 2018, 125, 432–442. [Google Scholar] [CrossRef]
- Cardoso, H.R.P.; Rapacki, C.; Ferreira, J.Z. Monitoring of a Zr-based conversion coating on galvanised steel and its performance against corrosion. Corros. Eng. Sci. Technol. 2019, 54, 726–730. [Google Scholar] [CrossRef]
- Liu, X.; Vonk, D.; Jiang, H.; Kisslinger, K.; Tong, X.; Ge, M.; Nazaretski, E.; Ravel, B.; Foster, K.; Petrash, S.; et al. Environmentally Friendly Zr-Based Conversion Nanocoatings for Corrosion Inhibition of Metal Surfaces Evaluated by Multimodal X-ray Analysis. ACS Appl. Nano Mater. 2019, 2, 1920–1929. [Google Scholar] [CrossRef]
- Han, J.; Thierry, D.; Ogle, K. Zr-based conversion coating on Zn and Zn-Al-Mg alloy coating: Understanding the accelerating effect of Cu (II) and NO3−. Surf. Coat. Technol. 2020, 402, 126236. [Google Scholar] [CrossRef]
- Ogle, K.; Buchheit, R.G. Conversion Coatings. Encyclopedia of Electrochemistry; Wiley-VCH GmbH & Co. KGaA: Weinheim, Germany, 2007; pp. 460–499. [Google Scholar]
- Ramanathan, E.; Balasubramanian, S. Comparative study on polyester epoxy powder coat and amide cured epoxy liquid paint over nano-zirconia treated mild steel. Prog. Org. Coat. 2016, 93, 68–76. [Google Scholar] [CrossRef]
- Cerezo, J.; Vandendael, I.; Posner, R.; Lill, K.; de Wit, J.H.W.; Mol, J.M.C.; Terryn, H. Initiation and growth of modified Zr-based conversion coatings on multi-metal surfaces. Surf. Coat. Technol. 2013, 236, 284–289. [Google Scholar] [CrossRef]
- Giacomelli, F.C.; Giacomelli, C.; De Oliveira, A.G.; Spinelli, A. Effect of electrolytic ZrO2 coatings on the breakdown potential of NiTi wires used as endovascular implants. Mater. Lett. 2005, 59, 754–758. [Google Scholar] [CrossRef]
- Dong, X.; Argekar, S.; Wang, P.; Schaefer, D.W. In situ Evolution of Trivalent Chromium Process Passive Film on Al in a Corrosive Aqueous Environment. ACS Appl. Mater. Interfaces 2011, 3, 4206–4214. [Google Scholar] [CrossRef]
- Moreira, V.B.; Ferreira, J.Z.; Meneguzzi, A. Pré-tratamento por deposição eletroassistida de viniltrietoxissilano (VTES) para a pintura de alumínio. Tecnol. Metal. Mater. Mineração 2018, 15, 68–74. [Google Scholar] [CrossRef][Green Version]
- Moreira, V.B.; Cardoso, H.R.P.; Ferreira, J.Z.; Meneguzzi, Á. DEPOSIÇÃO ELETROASSISTIDA DE ORTOSSILICATO DE TETRAETILA (TEOS) COMO PRÉ-TRATAMENTO PARA A PINTURA DE ALUMÍNIO AA1100. Tecnol. Metal. Mater. Mineração 2018, 15, 56–62. [Google Scholar] [CrossRef][Green Version]
- Li, L.; Swain, G.M. Effects of Aging Temperature and Time on the Corrosion Protection Provided by Trivalent Chromium Process Coatings on AA2024-T3. ACS Appl. Mater. Interfaces 2013, 5, 7923–7930. [Google Scholar] [CrossRef] [PubMed]
- Oliver, W.C.; Pharr, G.M. Measurement of hardness and elastic modulus by instrumented indentation: Advances in understanding and refinements to methodology. J. Mater. Res. 2004, 19, 3–20. [Google Scholar] [CrossRef]
- Roa, J.J.; Jiménez-Piqué, E.; Anglada, M.J. Nanoindentation of Advanced Ceramics: Applications to ZrO2 Materials. In Applied Nanoindentation in Advanced Materials; Tiwari, A., Natarajan, S., Eds.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2017; pp. 459–480. ISBN 1119084490. [Google Scholar]
- Sarfraz, A.; Posner, R.; Lange, M.M.; Lill, K.; Erbe, A. Role of Intermetallics and Copper in the Deposition of ZrO 2 Conversion Coatings on AA6014. J. Electrochem. Soc. 2014, 161, C509–C516. [Google Scholar] [CrossRef]
- Gusmano, G.; Montesperelli, G.; Rapone, M.; Padeletti, G.; Cusmà, A.; Kaciulis, S.; Mezzi, A.; Maggio, R. Di Zirconia primers for corrosion resistant coatings. Surf. Coat. Technol. 2007, 201, 5822–5828. [Google Scholar] [CrossRef]
- Andreatta, F.; Paussa, L.; Lanzutti, A.; Rosero Navarro, N.C.; Aparicio, M.; Castro, Y.; Duran, A.; Ondratschek, D.; Fedrizzi, L. Development and industrial scale-up of ZrO2 coatings and hybrid organic–inorganic coatings used as pre-treatments before painting aluminium alloys. Prog. Org. Coat. 2011, 72, 3–14. [Google Scholar] [CrossRef]
- Gaillard, Y.; Anglada, M.; Jiménez-Piqué, E. Nanoindentation of yttria-doped zirconia: Effect of crystallographic structure on deformation mechanisms. J. Mater. Res. 2009, 24, 719–727. [Google Scholar] [CrossRef]
- Jiménez-Piqué, E.; González-García, L.; Rico, V.J.; González-Elipe, A.R. Nanoindentation of nanocolumnar TiO2 thin films with single and stacked zig-zag layers. Thin Solid Film. 2014, 550, 444–449. [Google Scholar] [CrossRef]
- Gaillard, Y.; Rico, V.J.; Jiménez-Piqué, E.; González-Elipe, A.R. Nanoindentation of TiO2 thin films with different microstructures. J. Phys. D Appl. Phys. 2009, 42, 145305. [Google Scholar] [CrossRef]
- Kassavetis, S.; Logothetidis, S.; Zyganitidis, I. Nanomechanical testing of the barrier thin film adhesion to a flexible polymer substrate. J. Adhes. Sci. Technol. 2012, 26, 2393–2404. [Google Scholar] [CrossRef]
- Fockaert, L.I.; Taheri, P.; Abrahami, S.T.; Boelen, B.; Terryn, H.; Mol, J.M.C. Zirconium-based conversion film formation on zinc, aluminium and magnesium oxides and their interactions with functionalized molecules. Appl. Surf. Sci. 2017, 423, 817–828. [Google Scholar] [CrossRef]
- Šekularac, G.; Kovač, J.; Milošev, I. Comparison of the Electrochemical Behaviour and Self-sealing of Zirconium Conversion Coatings Applied on Aluminium Alloys of series 1xxx to 7xxx. J. Electrochem. Soc. 2020, 167, 111506. [Google Scholar] [CrossRef]
- George, F.O.; Skeldon, P.; Thompson, G.E. Formation of zirconium-based conversion coatings on aluminium and Al–Cu alloys. Corros. Sci. 2012, 65, 231–237. [Google Scholar] [CrossRef]
- Cerezo, J.; Vandendael, I.; Posner, R.; de Wit, J.H.W.; Mol, J.M.C.; Terryn, H. The effect of surface pre-conditioning treatments on the local composition of Zr-based conversion coatings formed on aluminium alloys. Appl. Surf. Sci. 2016, 366, 339–347. [Google Scholar] [CrossRef]
- Torras, J.; Azambuja, D.S.; Wolf, J.M.; Alemán, C.; Armelin, E. How organophosphonic acid promotes silane deposition onto aluminum surface: A detailed investigation on adsorption mechanism. J. Phys. Chem. C 2014, 118, 17724–17736. [Google Scholar] [CrossRef]
- Cerezo, J.; Taheri, P.; Vandendael, I.; Posner, R.; Lill, K.; de Wit, J.H.W.; Mol, J.M.C.; Terryn, H. Influence of surface hydroxyls on the formation of Zr-based conversion coatings on AA6014 aluminum alloy. Surf. Coat. Technol. 2014, 254, 277–283. [Google Scholar] [CrossRef]
- Buchheit, R.G.; Martinez, M.A.; Montes, L.P. Evidence for Cu Ion Formation by Dissolution and Dealloying the Al2CuMg Intermetallic Compound in Rotating Ring-Disk Collection Experiments. J. Electrochem. Soc. 2000, 147, 119. [Google Scholar] [CrossRef]
- Vukmirovic, M.B.; Dimitrov, N.; Sieradzki, K. Dealloying and Corrosion of Al Alloy 2024-T3. J. Electrochem. Soc. 2002, 149, B428. [Google Scholar] [CrossRef]
- Hashimoto, T.; Zhang, X.; Zhou, X.; Skeldon, P.; Haigh, S.J.; Thompson, G.E. Investigation of dealloying of S phase (Al2CuMg) in AA 2024-T3 aluminium alloy using high resolution 2D and 3D electron imaging. Corros. Sci. 2016, 103, 157–164. [Google Scholar] [CrossRef]
- Zhang, X.; Hashimoto, T.; Lindsay, J.; Zhou, X. Investigation of the de-alloying behaviour of θ-phase (Al2Cu) in AA2024-T351 aluminium alloy. Corros. Sci. 2016, 108, 85–93. [Google Scholar] [CrossRef]
- Šekularac, G.; Kovač, J.; Milošev, I. Prolonged protection, by zirconium conversion coatings, of AlSi7Mg0.3 aluminium alloy in chloride solution. Corros. Sci. 2020, 169, 108615. [Google Scholar] [CrossRef]
- Lostak, T.; Krebs, S.; Maljusch, A.; Gothe, T.; Giza, M.; Kimpel, M.; Flock, J.; Schulz, S. Formation and characterization of Fe3+-/Cu2+-modified zirconium oxide conversion layers on zinc alloy coated steel sheets. Electrochim. Acta 2013, 112, 14–23. [Google Scholar] [CrossRef]
- Yen, S.; Guo, M.; Zan, H. Characterization of electrolytic ZrO2 coating on Co–Cr–Mo implant alloys of hip prosthesis. Biomaterials 2001, 22, 125–133. [Google Scholar] [CrossRef]
- Eivaz Mohammadloo, H.; Sarabi, A.A.; Sabbagh Alvani, A.A.; Sameie, H.; Salimi, R. Nano-ceramic hexafluorozirconic acid based conversion thin film: Surface characterization and electrochemical study. Surf. Coat. Technol. 2012, 206, 4132–4139. [Google Scholar] [CrossRef]
- Zhang, F.; Jacobi, A.M. Aluminum surface wettability changes by pool boiling of nanofluids. Colloids Surf. Physicochem. Eng. Asp. 2016, 506, 438–444. [Google Scholar] [CrossRef]
- Gonzalez-Canche, N.G.; Flores-Johnson, E.A.; Cortes, P.; Carrillo, J.G. Evaluation of surface treatments on 5052-H32 aluminum alloy for enhancing the interfacial adhesion of thermoplastic-based fiber metal laminates. Int. J. Adhes. Adhes. 2018, 82, 90–99. [Google Scholar] [CrossRef]
- Mrad, M.; Ben Amor, Y.; Dhouibi, L.; Montemor, M.F. Effect of AA2024-T3 surface pretreatment on the physicochemical properties and the anticorrosion performance of poly(γ-glycidoxypropyltrimethoxysilane) sol-gel coating. Surf. Interface Anal. 2018, 50, 335–345. [Google Scholar] [CrossRef]
- Doerre, M.; Hibbitts, L.; Patrick, G.; Akafuah, N. Advances in Automotive Conversion Coatings during Pretreatment of the Body Structure: A Review. Coatings 2018, 8, 405. [Google Scholar] [CrossRef]
- Li, L.; Desouza, A.L.; Swain, G.M. In situ pH measurement during the formation of conversion coatings on an aluminum alloy (AA2024). Analyst 2013, 138, 4398. [Google Scholar] [CrossRef] [PubMed]
- Parvizi, R.; Marceau, R.K.W.; Hughes, A.E.; Tan, M.Y.; Forsyth, M. Atom probe tomography study of the nanoscale heterostructure around an Al20Mn3Cu2 dispersoid in aluminum alloy 2024. Langmuir 2014, 30, 14817–14823. [Google Scholar] [CrossRef]
- Li, L.; Whitman, B.W.; Swain, G.M. Characterization and Performance of a Zr/Ti Pretreatment Conversion Coating on AA2024-T3. J. Electrochem. Soc. 2015, 162, C279–C284. [Google Scholar] [CrossRef]
- Shruthi, T.K.; Swain, G.M. Communication—Role of Trivalent Chromium on the Anti-Corrosion Properties of a TCP Conversion Coating on Aluminum Alloy 2024-T3. J. Electrochem. Soc. 2018, 165, C103–C105. [Google Scholar] [CrossRef]
- Dong, X.; Wang, P.; Argekar, S.; Schaefer, D.W. Structure and Composition of Trivalent Chromium Process (TCP) Films on Al Alloy. Langmuir 2010, 26, 10833–10841. [Google Scholar] [CrossRef] [PubMed]
- Adhikari, S.; Unocic, K.A.; Zhai, Y.; Frankel, G.S.; Zimmerman, J.; Fristad, W. Hexafluorozirconic acid based surface pretreatments: Characterization and performance assessment. Electrochim. Acta 2011, 56, 1912–1924. [Google Scholar] [CrossRef]
- Van Ingelgem, Y.; Hubin, A.; Vereecken, J. Investigation of the first stages of the localized corrosion of pure copper combining EIS, FE-SEM and FE-AES. Electrochim. Acta 2007, 52, 7642–7650. [Google Scholar] [CrossRef]
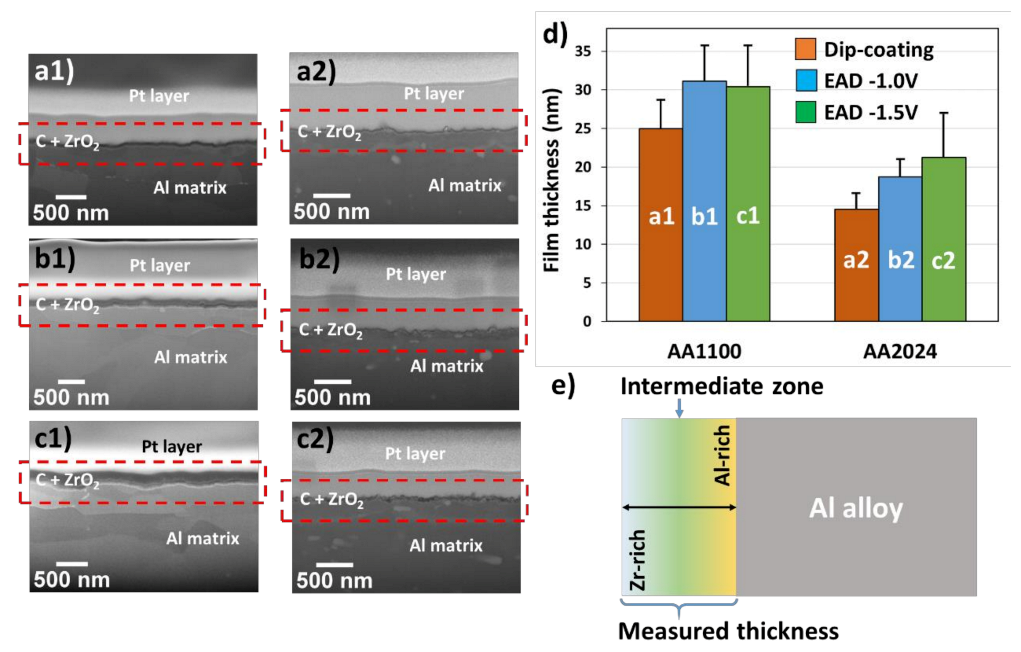


| Substrate | ZrO2 Deposition Method | Thickness and SD (nm) |
|---|---|---|
| AA1100 | DC | 24.9 ± 6.7 |
| EAD at −1.0 V | 31.1 ± 8.2 | |
| EAD at −1.5 V | 30.4 ± 9.8 | |
| AA2024 | DC | 14.5 ± 4.1 |
| EAD at −1.0 V | 18.7 ± 4.0 | |
| EAD at −1.5 V | 21.2 ± 10.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bonamigo Moreira, V.; Puiggalí-Jou, A.; Jiménez-Piqué, E.; Alemán, C.; Meneguzzi, A.; Armelin, E. Green Nanocoatings Based on the Deposition of Zirconium Oxide: The Role of the Substrate. Materials 2021, 14, 1043. https://doi.org/10.3390/ma14041043
Bonamigo Moreira V, Puiggalí-Jou A, Jiménez-Piqué E, Alemán C, Meneguzzi A, Armelin E. Green Nanocoatings Based on the Deposition of Zirconium Oxide: The Role of the Substrate. Materials. 2021; 14(4):1043. https://doi.org/10.3390/ma14041043
Chicago/Turabian StyleBonamigo Moreira, Vitor, Anna Puiggalí-Jou, Emilio Jiménez-Piqué, Carlos Alemán, Alvaro Meneguzzi, and Elaine Armelin. 2021. "Green Nanocoatings Based on the Deposition of Zirconium Oxide: The Role of the Substrate" Materials 14, no. 4: 1043. https://doi.org/10.3390/ma14041043
APA StyleBonamigo Moreira, V., Puiggalí-Jou, A., Jiménez-Piqué, E., Alemán, C., Meneguzzi, A., & Armelin, E. (2021). Green Nanocoatings Based on the Deposition of Zirconium Oxide: The Role of the Substrate. Materials, 14(4), 1043. https://doi.org/10.3390/ma14041043