3. Results and Discussion
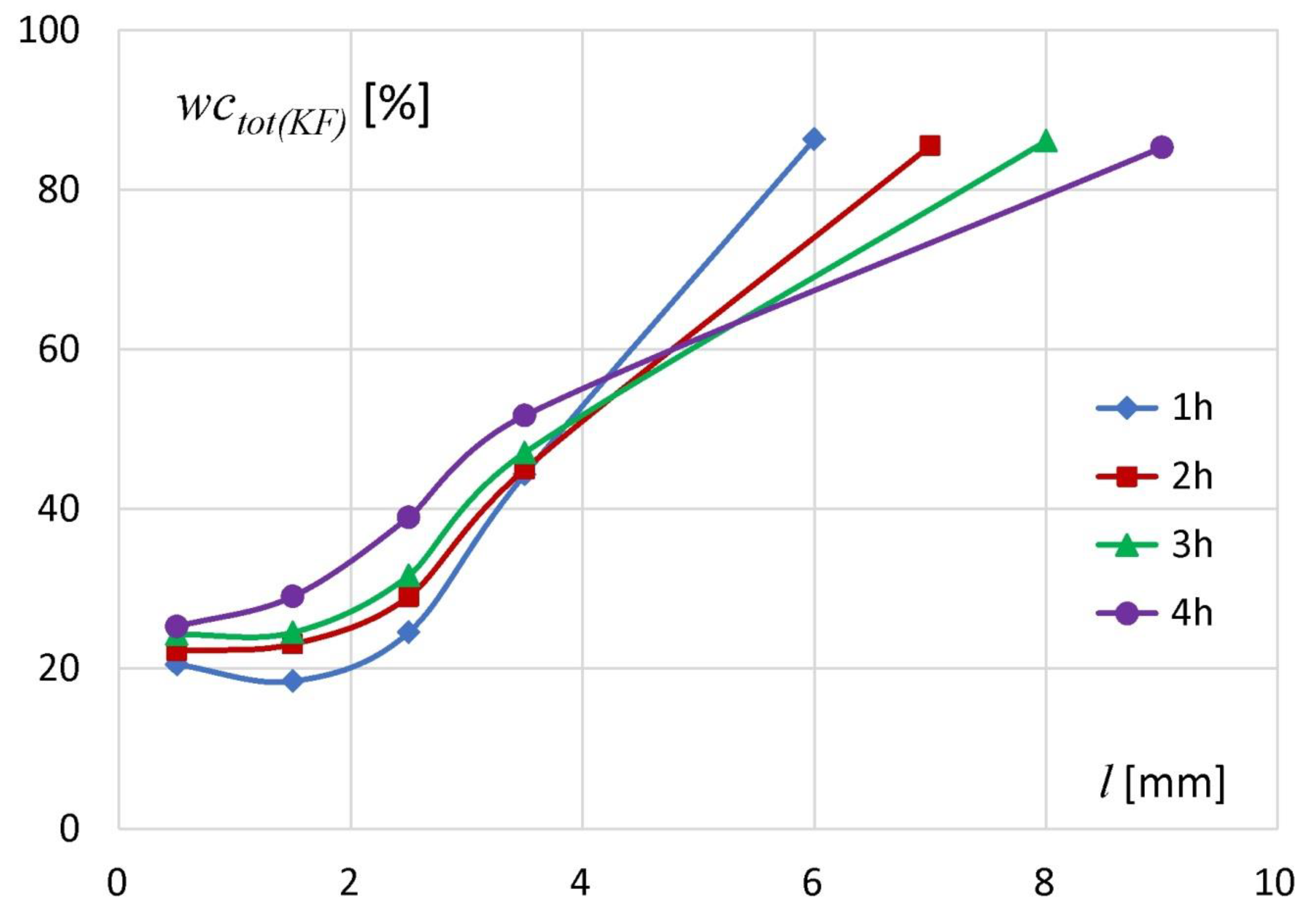
The results of the KF analysis are presented in
Figure 2. Position
l = 0 denotes the matrix bottom and spatial positions’ (
l) increase towards the hydration medium (water). The initial water content of the unhydrated tablets containing sodium alginate determined by the KF (
wc0(KF)) method was 15.2%. The values of total water content (
wctot(KF)) in all tablet slices were higher than the initial polymer water content, regardless of hydration time. It meant that the water penetrated into the deepest tablet regions even after the shortest measured hydration time (1 h). Along the axial section of the tablets, after the first hour of hydration the
wctot(KF) in particular slices was in the range of approximately 20–86%. After two hours of the experiment the increase of water content in particular slices was in the range 1–5%. The material from the slice located at
l = 0–1 mm (tablet bottom) contained 22% of water. In the next slices the water content was 22.9% for
l between 1 and 2 mm, 28.8% (for
l between 2 and 3 mm) and 44.8% (for
l between 3 and 4 mm). After three hours of the experiment, the changes in the amount of water in particular slices did not exceed 2.5% in comparison to the values obtained after 2 h of hydration. After four hours there was an increase in water content of approximately 1–7%. In subsequent slices starting from the tablet bottom (
l from 0 to 4 mm) following water concentrations were measured: 25.1% (
l between 0 and 1 mm), 28.9% (
l between 1 and 2 mm), 38.9% (
l between 2 and 3 mm) and 51.5% (
l between 3 and 4 mm). KF studies showed that the uptake of water by the matrix was the highest in its external part close to the hydration medium. Water content in that region (
l ~ 6–9 mm depending on incubation time) was approximately 85–86% and had similar values regardless of hydration time.
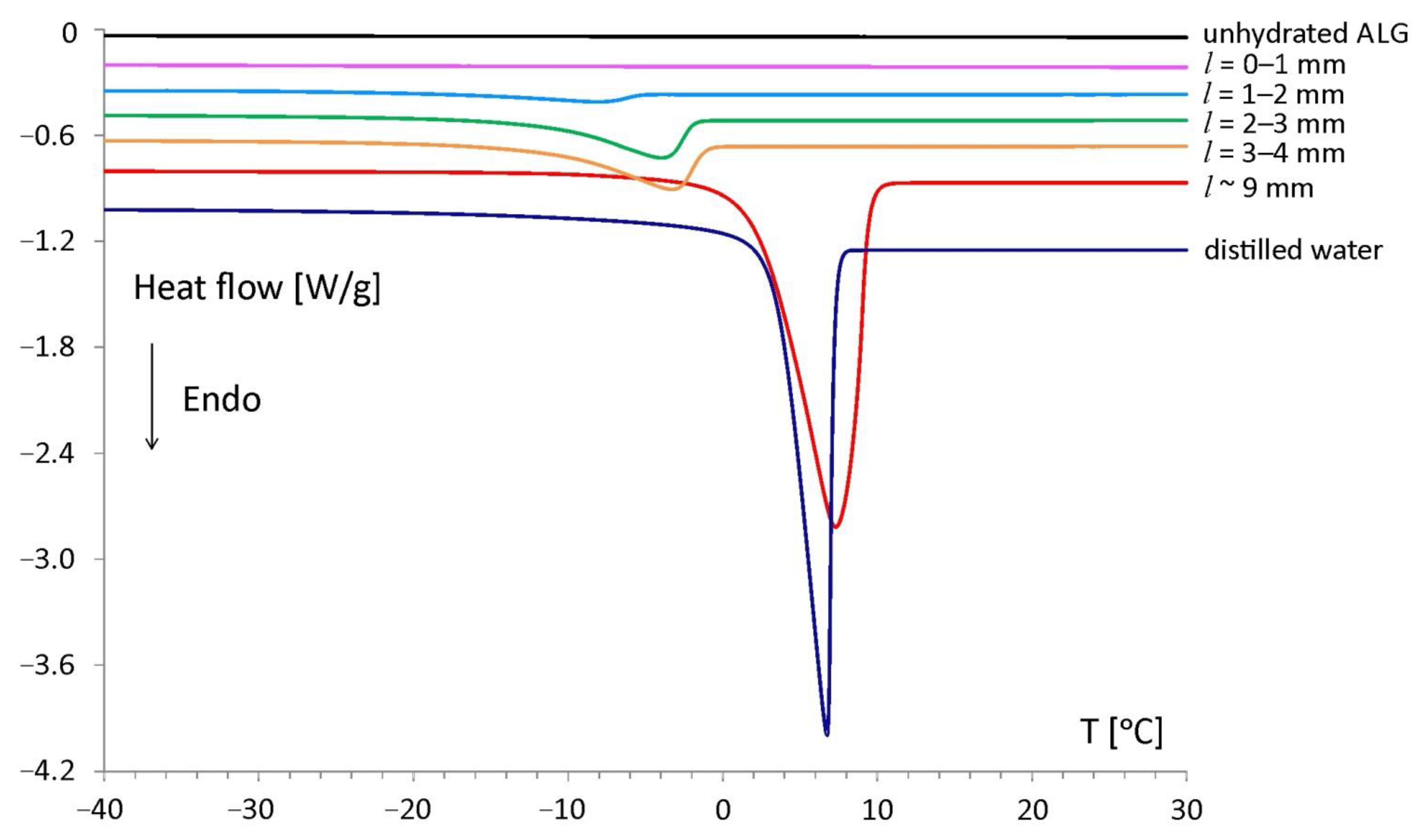
The DSC studies of the material taken from particular slices allowed for the characterization of the interaction between the water and the sodium alginate chains at 4 h of hydration. Endothermic peaks were observed during the heating run in the tablet for
l between 0 and 9 mm. The peaks were the result of first-order phase transition of water present in the sample and it were called “freezing water”. In the slice located between
l = 0 and 1 mm freezing water was not detected, as no peaks were registered according to experimental conditions used in the study [
10,
17].
The DSC curve of the unhydrated polymer (
Figure 3, black line) showed no phase transitions in the heating run. Therefore, the occurrence of DSC peaks in some slices of the hydrated sodium alginate tablet was associated with the phase transition of water penetrating the matrix during hydration. In spatial range (
l = 0–10 mm) only one endothermic peak was observed. The normalized peak areas were increasing upon the distance from the tablet bottom, and it was associated with increasing freezing water content in the sample. The highest freezing water content was determined in the external part of the tablet, which had direct contact with the surrounding hydration medium (matrix region centered at
l ~ 9 mm).
With decreasing water content, a gradual shift of onset temperatures towards lower temperatures was observed. The lowest temperature of the phase transition was observed at approximately −12 °C for the slice located between
l = 1 and 2 mm. The shift of melting onsets in hydrated sodium alginate samples is also reported in other works [
18,
19]. The DSC characteristics of the hydrated sodium alginate matrix could be affected by the presence of sodium ions, which stay in the vicinity of the polymer chains [
20,
21]. The highest absolute value of phase transition heat was recorded in the external part of the tablet (
l ~ 9 mm): −245 mJ/mg, and the lowest at a slice located between
l = 1 and 2 mm was −13 mJ/mg. The onset of the water melting peak for
l ~ 9 mm was recorded at +2 °C and was close to the melting temperature of pure water: +3 °C. This indicated the presence of free water in this matrix region.
Both the unhydrated polymer and the samples taken from the tablet bottom (
l between 0 and 1 mm) did not contain the freezing water fraction. However, the existence of water in these samples was confirmed by the difference in sample weight before and after its heating up to 180 °C according to the procedure described in the
Section 2.4. The water molecules present in those samples were strongly restricted by the alginate chains and did not form a solid phase. This type of water was designated as nonfreezing water.
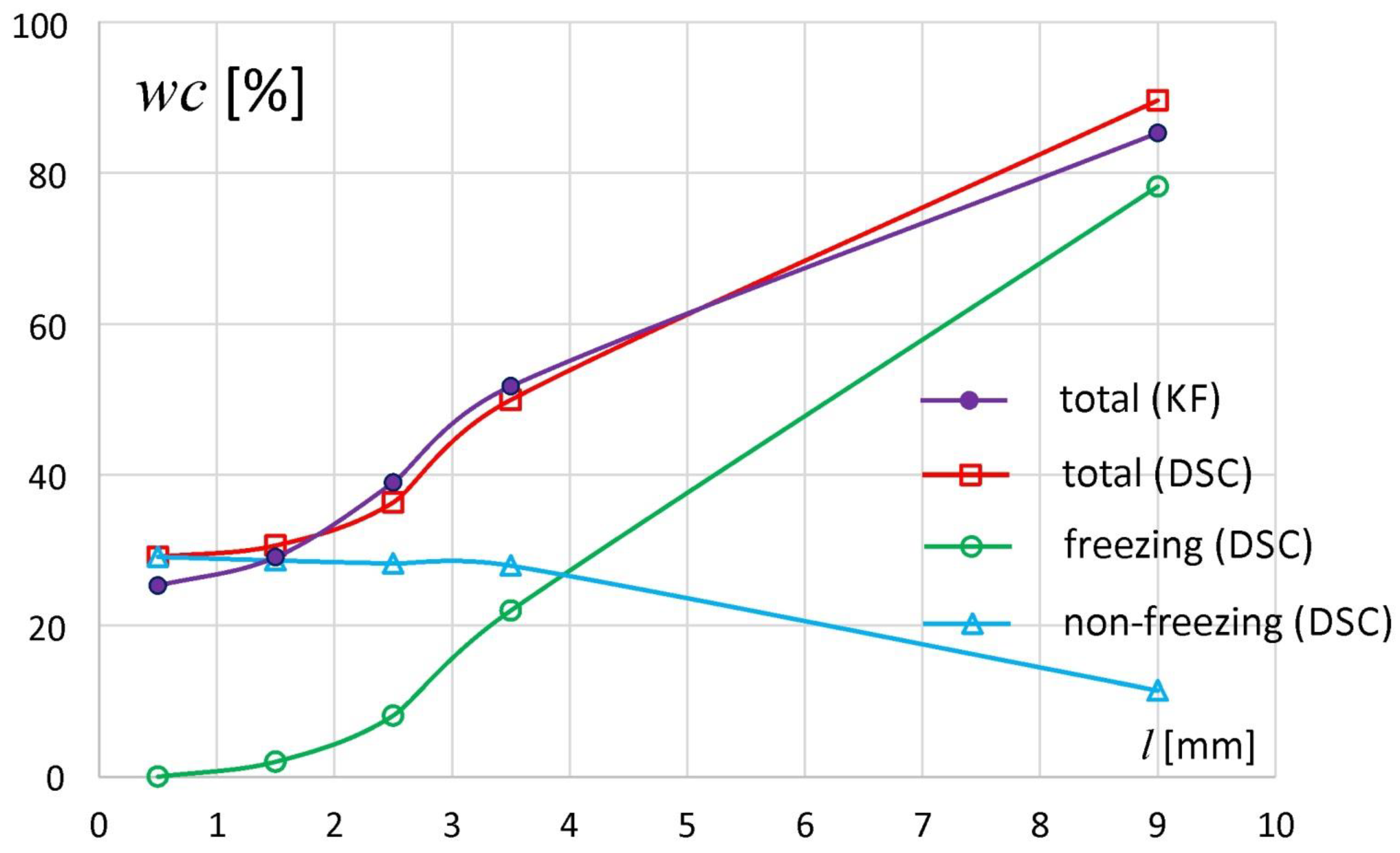
Figure 4 presents the individual water fractions in the samples of all tablet slices—total, freezing and nonfreezing water content. For comparison, the total water content determined by KF method is also presented. The results of the measurements of the total content of water by the KF and DSC methods were convergent.
The distribution of water in the sodium alginate tablets indicated that the samples taken from the matrix regions (slices) above
l = 1 mm contained both freezing and nonfreezing water in varying proportions. The highest content of freezing water was observed in the external part of the tablet and decreased gradually towards the tablet bottom. In the sample taken from the slice at
l ~ 9 mm,
wcf (DSC) = 79%, and the slice located between
l = 1 and 2 mm contained only ca. 1% of freezing water. In contrast, the nonfreezing water content in the external tablet region was about 11%, while in the rest of the tablet it remained at a constant level of 28–29%. A constant level of nonfreezing water with total water content below 50% (as happened here) is also reported by Ping et al. during the hydration of polyvinyl alcohol [
9].
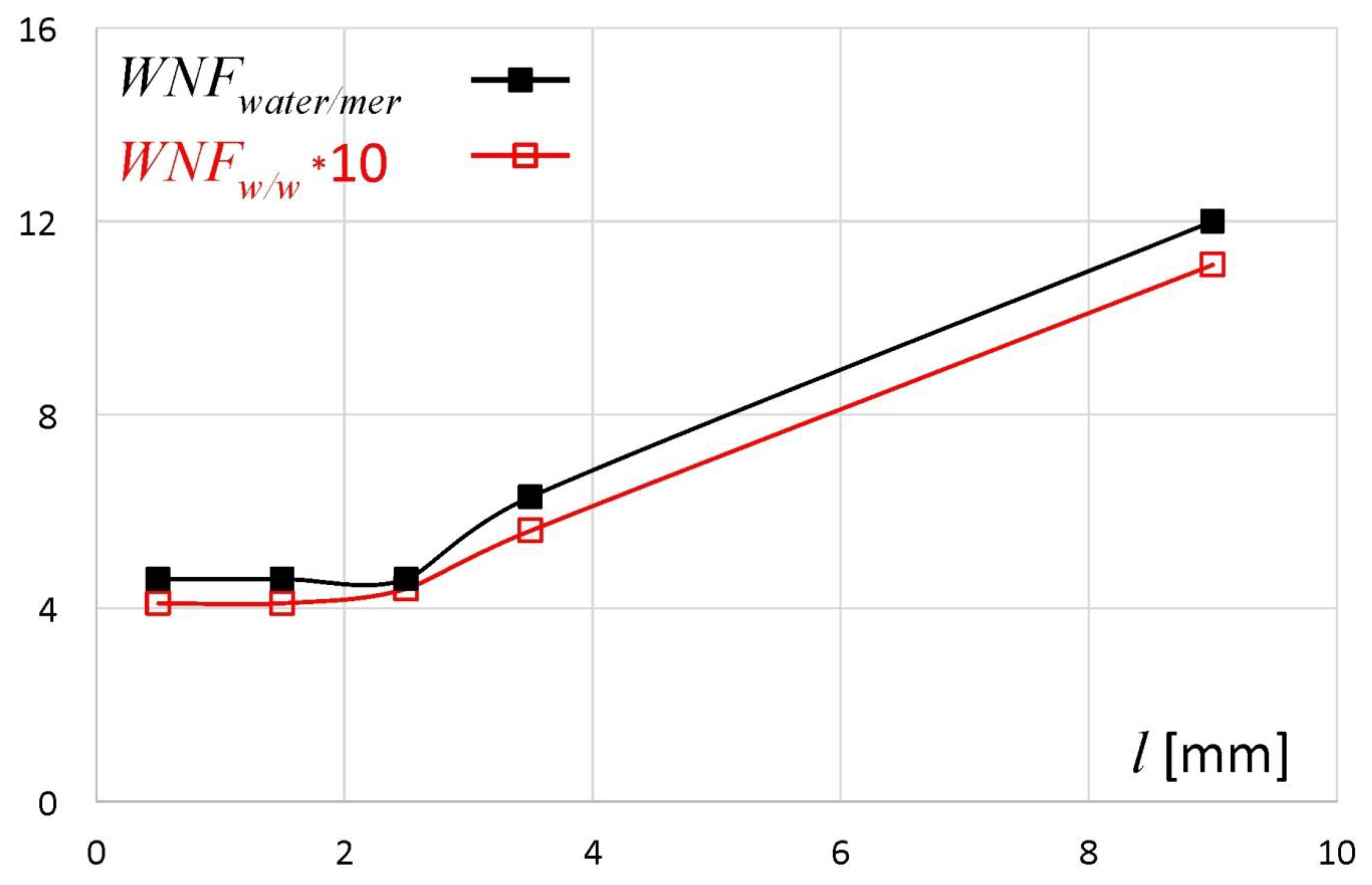
The nonfreezing water fraction described above was expressed as a percentage of total sample weight (%). However, in the literature the ratio of nonfreezing water weight and dry polymer weight (
WNFw/w) is commonly used. This is because the ratio
WNFw/w better shows the interaction between water and polymer binding sites and its value does not directly depend on the total water content in the sample. In
Figure 5 the fraction of nonfreezing water expressed as the ratio
WNFw/w is presented. The same ratio was also recalculated as the number of water molecules per one dry polymer unit (
WNFwater/mer), where the molar mass of water is 18 g/mol and the molar mass of one unit of sodium alginate is 198 g/mol, and presented in the same graph [
15,
22]. The ratio
WNFw/w remained at the level of ca. 0.4 in the slices/regions located between
l = 0 and 2 mm, and then it ranged from 0.44 for
l = 2–3 mm to 1.11 at
l ~ 9 mm. Similar
WNFw/w values were obtained for other water–sodium alginate systems [
8,
19,
22]. An increase of
WNFw/w above a certain total water content level was also observed for other polymer systems [
23]. The values of
WNFwater/mer indicated that for one unit of dry polymer there were between 5 (for
l = 0–1 mm) to approximately 12 (for
l ~ 9 mm) of nonfreezing water molecules. Similar results are reported by Nakamura et al. [
19]. Mazur et al. have estimated that in the first hydration shell of alginate anion in aqueous solutions there are six (± 2) water molecules with reduced rotational dynamics and five water molecules slowed down by the sodium ion (total: 11 ± 2 molecules per sodium alginate unit) [
21]. Based on their results it may be estimated that in the external part of the matrix (
l ~ 9 mm), the polymer and water formed a solution.
According to the theory of Peirce et al. and its extensions (Magne et al., Okubayashi et al.) concerning the mechanism of water sorption by polysaccharides, it could be assumed that at low total water content, it was adsorbed on polymer chains through hydrogen bonds with the sorption centers (hydrophilic groups, sodium ions, etc.) [
24,
25,
26]. When freezing water was observed (the matrix region for
l above 1 mm), the places capable of binding nonfreezing water in the matrix were already saturated. The uptake of additional water molecules spread apart the sodium alginate chains and caused the exposition of new polymer sorption sites in the regions situated closer to the matrix surface, resulting in higher values of
WNFw/w in those regions [
20,
27].
The KF and DSC data represented slices approximately one-millimeter thick. Higher resolution in the matrix volume region located at l = 0-4 mm was not feasible due to technical limitations of the device used for tablet hydration as well as destructive character of sample preparation. Moreover, the external part of the matrix protruding beyond the holder was treated as a source of one sample for KF and DSC analysis—it mainly contained a viscous “gel-like” substance unsuitable for slicing. However, it is worth noting that such a spatial characterization of a polymer matrix under hydration using KF and DSC was not presented previously. In MRI (MSME and UTE) analysis the whole matrix cross-section was easily visible. Nondestructive MRI methods allowed observing the changes of matrix properties through the tablet cross-section with an order of magnitude higher resolution (i.e., 0.1 mm).
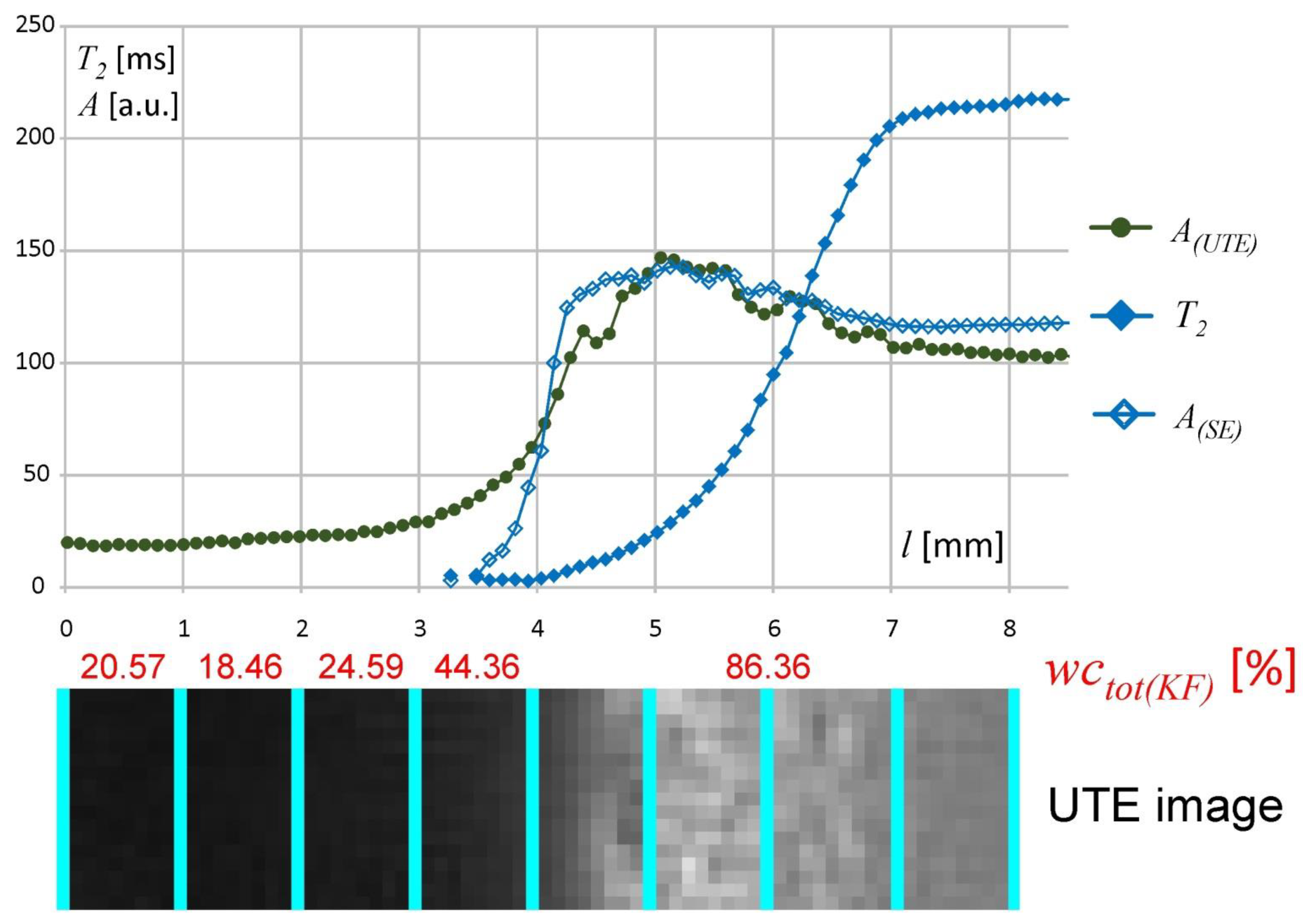
The UTE sequence application was proposed as a link between the KF, DSC and MSME results. Using UTE, the signal acquisition started a very short time after stimulation, i.e., 300 µs. This allowed protons to be imaged in regions with very short
T2 relaxation times in the least hydrated regions of the matrix (including mobilized polymer and bound water). Therefore, it was possible to acquire an MR signal from the inner part of the matrix starting from
l = 0 mm. UTE and multi-echo spin-echo (MSME) signal intensity profiles scaled in arbitrary units are juxtaposed in
Figure 6 together with the KF results and the corresponding UTE image (results obtained at 1 h of hydration). The UTE and MSME signal intensity profiles match each other in the
l = 4–7 mm spatial range. The MSME sequence allowed signal acquisition at 3.5 ms and only in freezing water (DSC based terminology), i.e., only higher mobility protons could be detected starting from
l ≈ 3 mm. Protons with less mobility were not detected in the MSME sequence due to detection limitations, but multi-echo spin-echo (MSME) data complemented KF and DSC data in the highly hydrated regions of the matrix. The signal intensity in the spatial range containing hydration medium (water) (
l > 7) differed between the UTE and multi-echo spin-echo (MSME) profiles due to different
T1 weighting. The apparently higher water content in the hydrated matrix than in the bulk solvent (water) is also a result of
T1 weighting (i.e., the bulk solvent was slightly attenuated). Despite detection limits (i.e., TE = 3.5 ms) the quantitative measurement of the effective
T2 relaxation and corresponding signal amplitude was performed and gave a link between water content and its molecular mobility. Using multi-echo spin-echo imaging one pool (nonfreezing) of water was found at single pixel level, characterized by signal amplitude (
A) and corresponding effective
T2 relaxation time. Both values decreased towards
l = 0 and
T2 had the highest values
T2 > 200 ms in the matrix regions adjacent to the bulk solvent (
T2 ≈ 250).
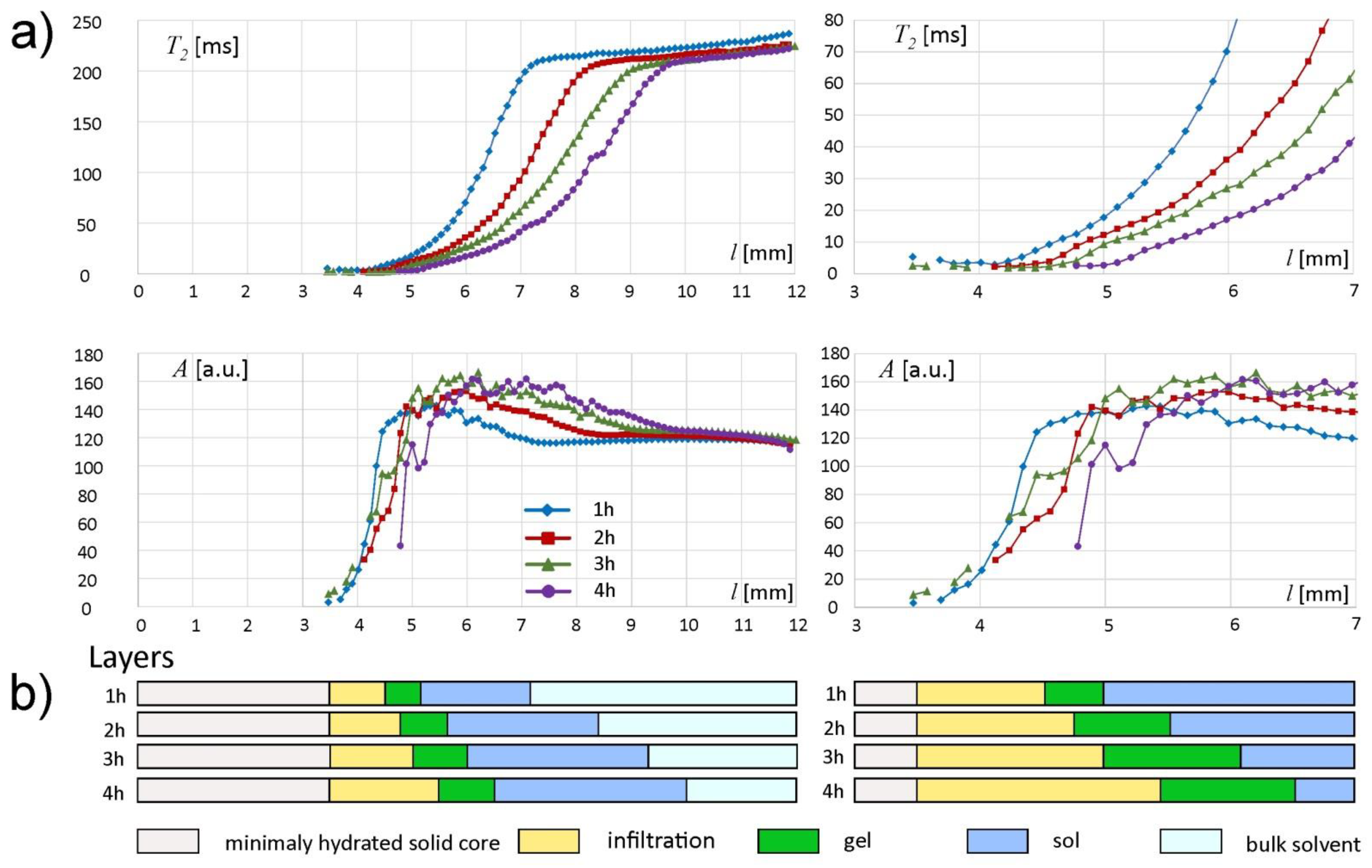
The complete results and interpretation of multi-echo spin-echo (MSME) imaging are presented in
Figure 7. The whole matrix spatial range was separated into layers (important note: “slices” refer to KF/DSC sampling, while “layers” denote interpretation results based on multi-echo spin-echo MRI). The deepest part of the matrix, i.e., for
l < 3 mm, gave no signal when using MSME sequence. This region was denoted as minimally hydrated solid core (
Figure 7b). According to KF results—starting from 1 h of incubation, the hydration level of the deepest regions of the matrix was higher than the initial one (i.e., as assessed for the unhydrated matrix). Two pools of water were present in this layer. The first, present in unhydrated matrix, was connected with polymer binding sites in its hydration shell and was assigned as nonfreezing according to the DSC results. The second pool was water absorbed by the matrix during hydration. According to the DSC results this second pool of water contributed both to nonfreezing and to freezing water and it preceded a steep hydration front. Starting from the spin-echo signal detection limit at
l ≈ 3 mm, the amplitude increased steeply towards the external border of the matrix—in this region
T2 < 10 ms (mostly set at the lower limit). This layer was denoted as an infiltration layer and was marked yellow in
Figure 7. The approximate width of the layer was 1, 1.25, 1.5 and 1.75 mm at 1, 2, 3 and 4 h of incubation, respectively. Despite high water content, its molecular mobility was still very low in terms of effective
T2 relaxation time. The water content reached almost maximum at corresponding
T2 ≈ 10 ms. These spatial locations can be assigned as a second (right) border of infiltration layer, very similar to those observed for HPMC-based matrices [
28,
29] and can be regarded as the beginning of the fully hydrated part of the matrix. The position of this front was 4.5 mm at 1 h, 4.75 mm at 2 h, 5.0 mm at 3 h and 5.25 mm at 4 h of hydration (
Figure 7a). The infiltration layer tended to be more gradual at consecutive hydration times. The fully hydrated part of the matrix could be further separated into two distinct layers. From this spatial position the
T2 of the detected water increased linearly. It should be noted that generally, increase in
T2 time means an increase in proton mobility. The layer characterized by linear spatial increase in
T2 was denoted as gel and marked green in
Figure 7. The approximate width of the layer was 0.5, 0.75, 1 and 1.25 mm at 1, 2, 3 and 4 h of incubation, respectively. The position of switch point in spatial
T2 increase (the change from linear to nonlinear) was approximately 5.0 mm at 1 h, 5.5 mm at 2 h, 6.0 mm at 3 h and 6.5 mm at 4 h of hydration. The layer characterized by nonlinear spatial
T2 increase was marked blue. It can be assumed that switch-point position marks the gel/sol transition. Therefore, the layer was denoted as sol. The approximate width of the layer was 2.25, 2.75, 3.25 and 3.25 mm at 1, 2, 3 and 4 h of incubation, respectively.
The beginning of the bulk solvent is marked by another switch-point, i.e., the blurry border between concentrated sol and bulk solvent— the beginning of plateaus both in amplitude and
T2 profiles could be identified (
Figure 7a). The
T2 plateau started at a value of about 210 ms for each hydration time. The plateau started at
l = 7.25 mm at 1 h of hydration,
l = 8.25 at 2 h, 9.25 at 3 h and
l = 9.75 at 4 h of hydration. The bulk solvent is marked light blue in
Figure 7b. The
T2 plateau rose slightly towards higher values of
l. It was related to a decrease in the content of dissolved polymer particles in bulk solvent (higher
T2 values means less polymer dissolved).
The maximum values of amplitude A of the spin echo envelope signals in the matrix region adjacent to the hydration medium increased from approximately 143 a.u. for l = 7.25 mm after the first hour to approximately 166 a.u. for l = 8.2 mm after the fourth hour of the experiment. This indicates that new water molecules penetrated the gel/sol regions of the matrix, increasing water content in these regions.
The extensive results presented above can be summarized to give a compact overview of the alginate matrix under unilateral hydration. The hydration in the minimally hydrated layer preceded the steep hydration front. The minimally hydrated layer gained water starting from the first hour of incubation (5–10% w/w) and at 4 h the total water content was 29–39% with a nonfreezing pool of 28–29% as assessed by KF and DSC. The infiltration layer with the steep hydration front was characterized by low water mobility in terms of effective T2 relaxation times (T2 < 10 ms). The water content at the beginning of the infiltration layer (l = 3–4 mm) was 45–47% (1–3 h of hydration) and at 4 h it reached about 50% with nonfreezing pool water molecules of 28%. The water content reached almost maximum at T2 circa 10 ms. Despite full hydration, the first part of the fully hydrated layer (gel) was characterized roughly by T2 < 20 ms (with linear spatial increase towards external part of the matrix), which reflected low water mobility. The second part of the fully hydrated layer with T2 between 20 and 200 ms (with nonlinear spatial increase) was denoted as sol. The water content at near the sol/bulk solvent interface was 85–86% from the first hour of hydration with a nonfreezing water pool of 11% at 4 h as assessed using KF and DSC.
The sodium alginate matrix tablet could not be regarded as a medicine itself because it did not contain any active substances. However, its analysis showed the complexity of the “picture” formed during hydration. It gives a good starting point for further studies of more complex systems (e.g., oral controlled release drug delivery systems), where the drugs and additional excipients could introduce additional factors modifying the water transport and interaction with the matrix.