Solubility of Carbon Dioxide in Deep Eutectic Solvents Based on 3-Amino-1-Propanol and Tetraalkylammonium Salts at Low Pressure
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of Deep Eutectic Solvents
2.2. Properties of Deep Eutectic Solvents
2.2.1. Viscosity Measurements
2.2.2. Polarity and Kamlet-Taft Parameters
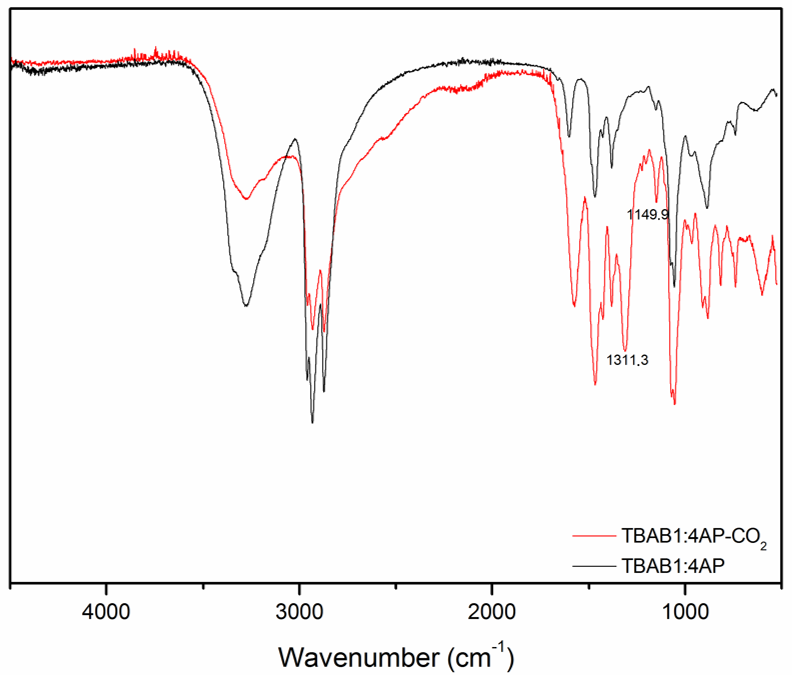
2.3. Infrared Spectroscopy Measurements
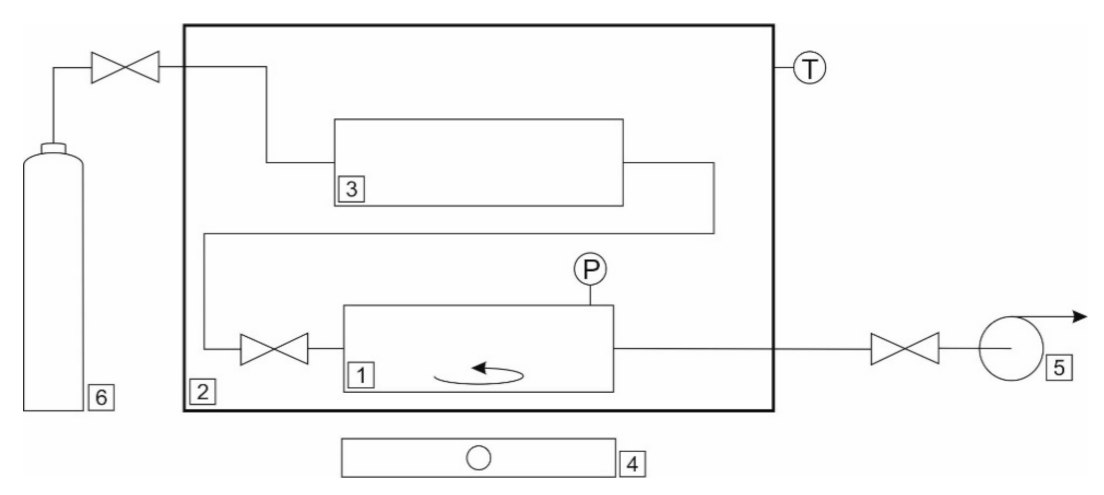
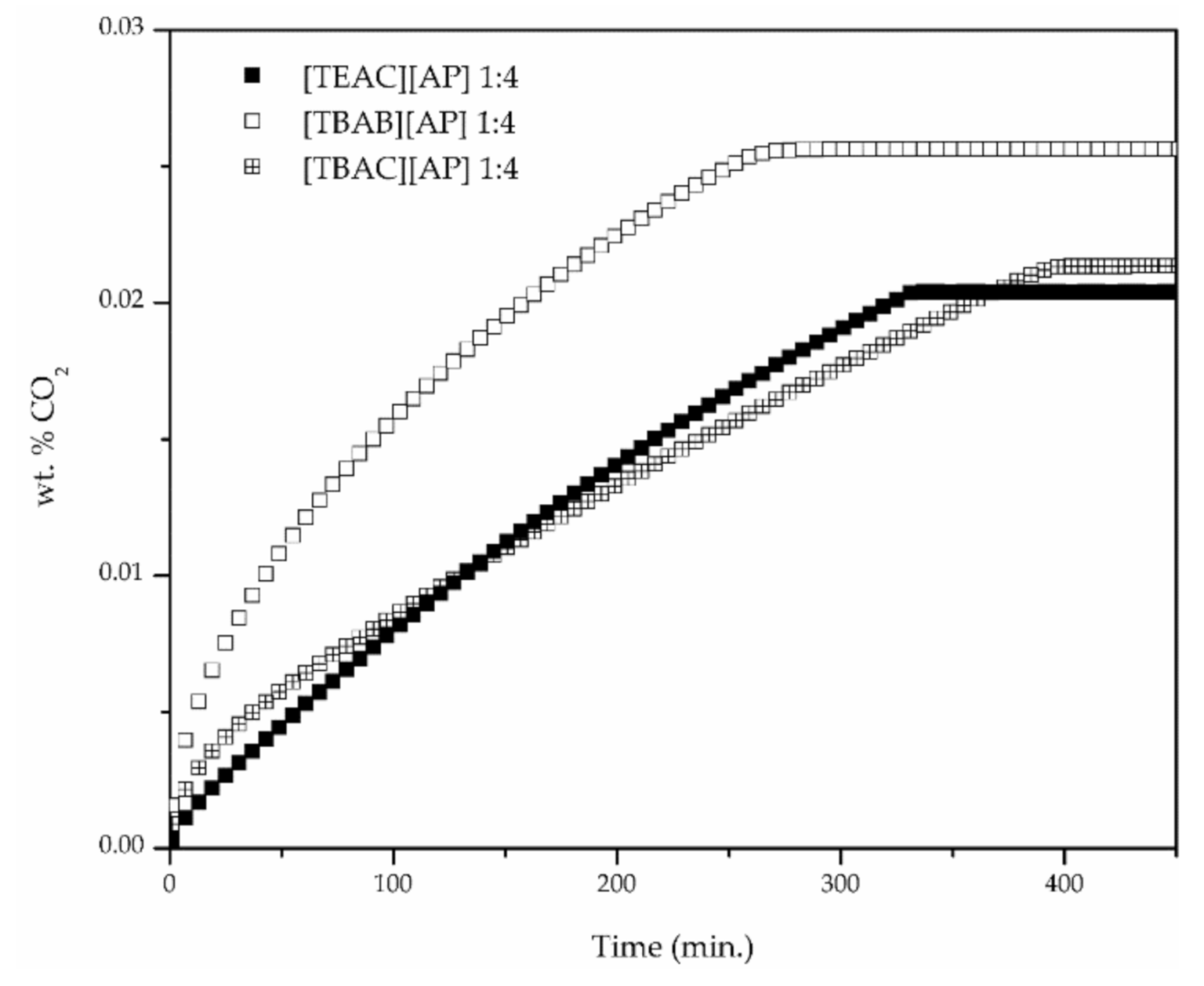
2.4. CO2 Solubility
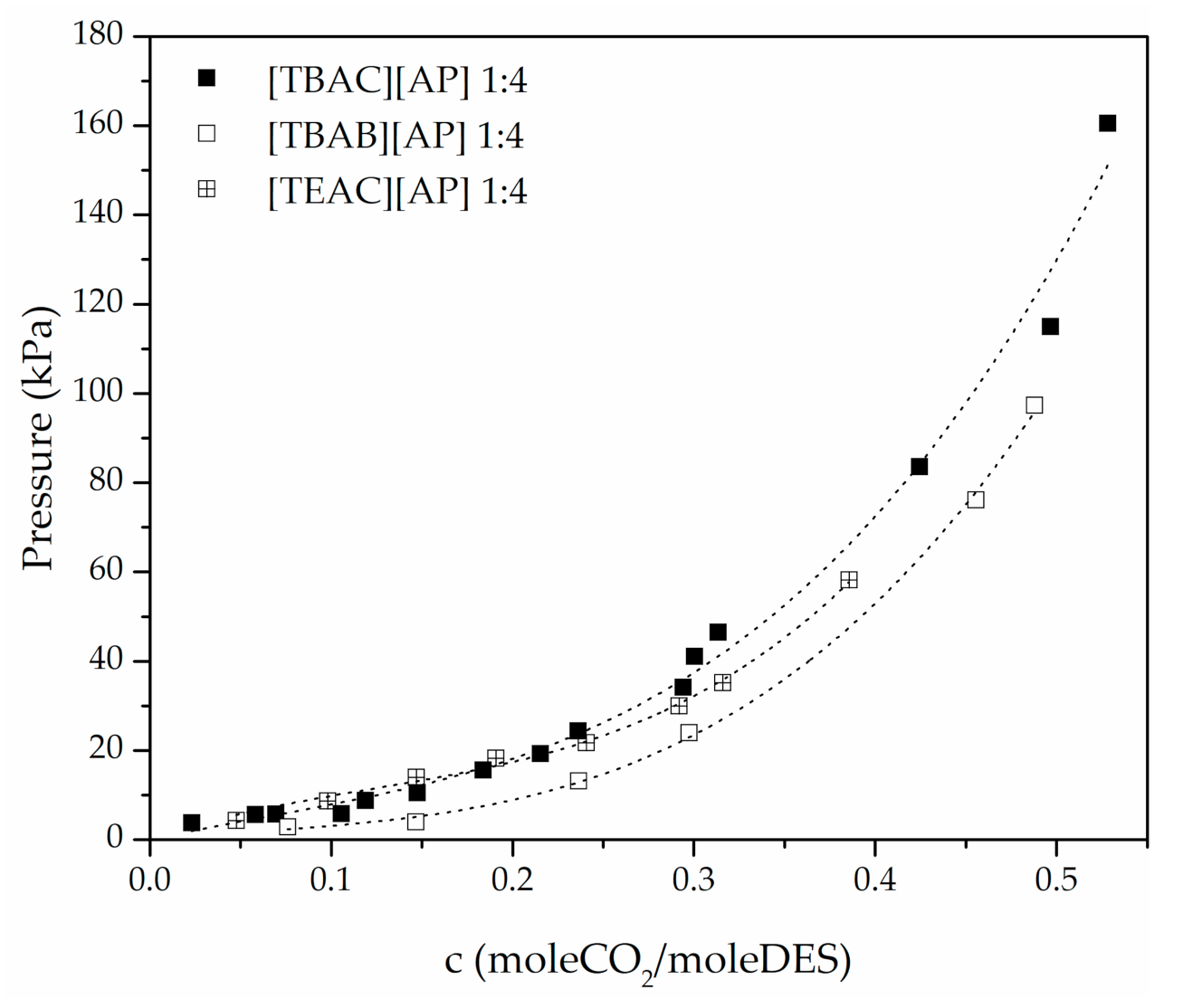
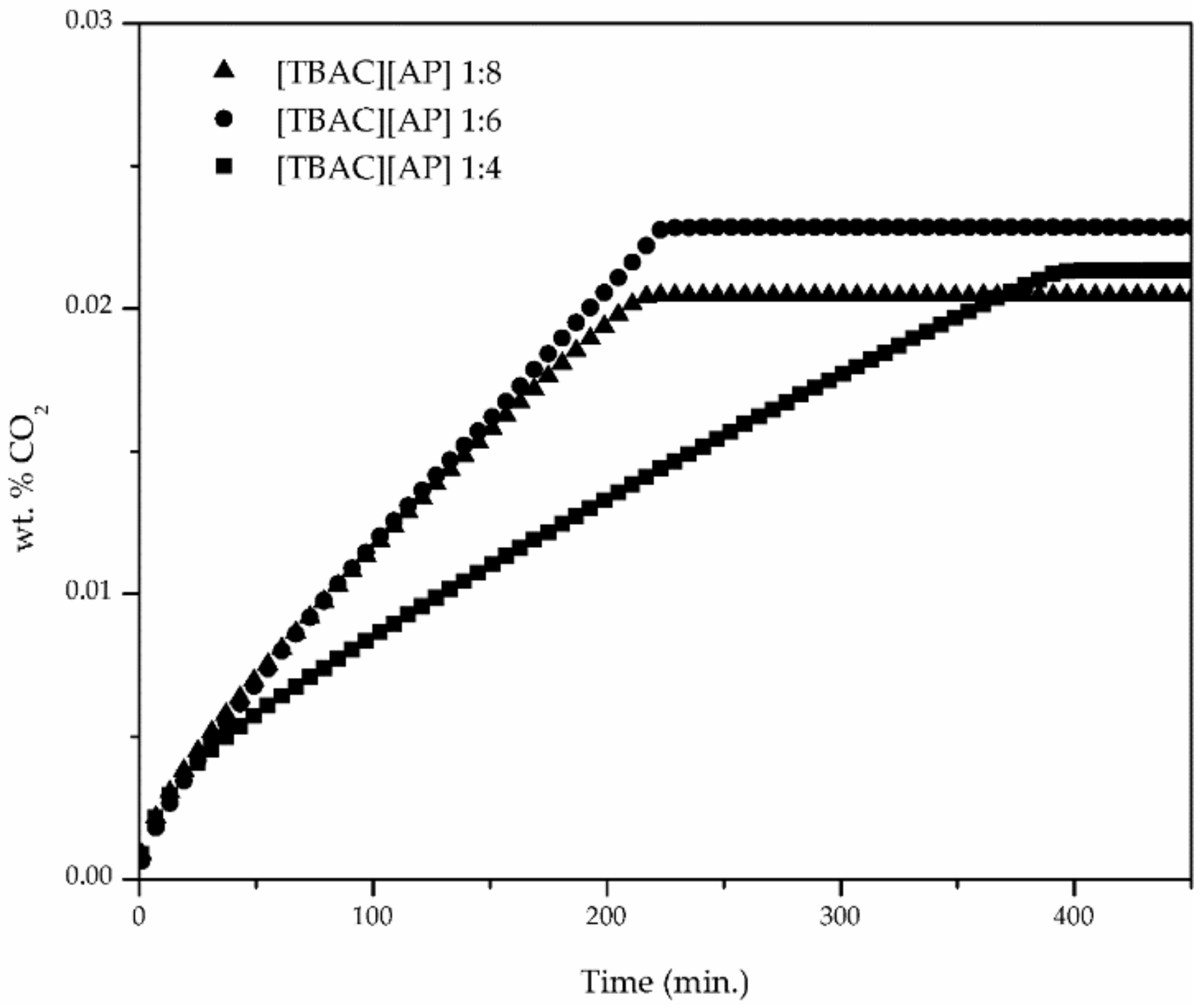
3. Results
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Spigarelli, B.P.; Kawatra, S.K. Opportunities and challenges in carbon dioxide capture. J. CO2 Util. 2013, 1, 69–87. [Google Scholar] [CrossRef]
- Kang, D.; Park, S.; Jo, H.; Min, J.; Park, J. Solubility of CO2 in Amino-acid-based solutions of (Potassium Sarcosinate), (Potassium Alaninate + Piperazine), and (Potassium Serinate + Piperazine). J. Chem. Eng. Data 2013, 58, 1787–1791. [Google Scholar] [CrossRef]
- Aronu, U.E.; Svendsen, H.; Hoff, K.A. Investigation of amine amino acid salts for carbon dioxide absorption. Int. J. Greenh. Gas Control 2010, 4, 771–775. [Google Scholar] [CrossRef]
- Bai, H.; Yeh, A.C. Removal of CO2 greenhouse gas by ammonia scrubbing. Ind. Eng. Chem. Res. 1997, 36, 2490–2493. [Google Scholar] [CrossRef]
- Wang, M.; Lawal, A.; Stephenson, P.; Sidders, J.A.; Ramshaw, C. Post-combustion CO2 capture with chemical absorption: A state-of-the-art review. Chem. Eng. Res. Des. 2011, 89, 1609–1624. [Google Scholar] [CrossRef]
- Olajire, A.A. CO2 capture and separation technologies for end-of-pipe applications—A review. Energy 2010, 35, 2610–2628. [Google Scholar] [CrossRef]
- Rochelle, G.T. Amine scrubbing for CO2 capture. Science 2009, 325, 1652–1654. [Google Scholar] [CrossRef]
- Alam Chowdhury, F.; Yamada, H.; Higashii, T.; Goto, K.; Onoda, M. CO2 capture by tertiary amine absorbents: A performance comparison study. Ind. Eng. Chem. Res. 2013, 52, 8323–8331. [Google Scholar] [CrossRef]
- Aghaie, M.; Rezaei, N.; Zendehboudi, S. A systematic review on CO2 capture with ionic liquids: Current status and future prospects. Renew. Sustain. Energy Rev. 2018, 96, 502–525. [Google Scholar] [CrossRef]
- Marsh, K.N.; Boxall, J.A.; Lichtenthaler, R. Room temperature ionic liquids and their mixtures—A review. Fluid Phase Equilibria 2004, 219, 93–98. [Google Scholar] [CrossRef]
- Raksajati, A.; Ho, M.; Wiley, D. Solvent development for post-combustion CO2 capture: Recent development and opportunities. MATEC Web Conf. 2018, 156, 03015. [Google Scholar] [CrossRef]
- Oliveira, M.V.; Vidal, B.T.; De Melo, C.M.; Miranda, R.D.C.D.; Soares, C.M.F.; Coutinho, J.A.; Ventura, S.P.M.; Mattedi, S.; Lima, Á.S. (Eco)toxicity and biodegradability of protic ionic liquids. Chemosphere 2016, 147, 460–466. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, A.; Gopinath, K.P.; Vo, D.-V.N.; Malolan, R.; Nagarajan, V.M.; Arun, J. Ionic liquids, deep eutectic solvents and liquid polymers as green solvents in carbon capture technologies: A review. Environ. Chem. Lett. 2020, 18, 1–24. [Google Scholar] [CrossRef]
- Sarmad, S.; Mikkola, J.-P.; Ji, X. Carbon dioxide capture with ionic liquids and deep eutectic solvents: A new generation of sorbents. ChemSusChem 2017, 10, 324–352. [Google Scholar] [CrossRef]
- Marcus, Y. Deep Eutectic Solvents; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2019. [Google Scholar]
- Zhang, Y.; Ji, X.; Lu, X. Choline-based deep eutectic solvents for CO2 separation: Review and thermodynamic analysis. Renew. Sustain. Energy Rev. 2018, 97, 436–455. [Google Scholar] [CrossRef]
- Leron, R.B.; Li, M.-H. Solubility of carbon dioxide in a eutectic mixture of choline chloride and glycerol at moderate pressures. J. Chem. Thermodyn. 2013, 57, 131–136. [Google Scholar] [CrossRef]
- Li, G.; Deng, D.; Chen, Y.; Shan, H.; Ai, N. Solubilities and thermodynamic properties of CO2 in choline-chloride based deep eutectic solvents. J. Chem. Thermodyn. 2014, 75, 58–62. [Google Scholar] [CrossRef]
- Ali, E.; Hadj-Kali, M.K.; Mulyono, S.; Alnashef, I.M.; Fakeeha, A.; Mjalli, F.S.; Hayyan, A. Solubility of CO2 in deep eutectic solvents: Experiments and modelling using the Peng–Robinson equation of state. Chem. Eng. Res. Des. 2014, 92, 1898–1906. [Google Scholar] [CrossRef]
- Francisco, M.; Bruinhorst, A.; Zubeir, L.F.; Peters, C.C.; Kroon, M.C. A new low transition temperature mixture (LTTM) formed by choline chloride + lactic acid: Characterization as solvent for CO2 capture. Fluid Phase Equilibria 2013, 340, 77–84. [Google Scholar] [CrossRef]
- Adeyemi, I.; Abu-Zahra, M.R.; Al-Nashef, I.M. Novel green solvents for CO2 capture. Energy Procedia 2017, 114, 2552–2560. [Google Scholar] [CrossRef]
- Haider, M.B.; Jha, D.; Sivagnanam, B.M.; Kumar, R. Thermodynamic and kinetic studies of CO2 capture by glycol and amine-based deep eutectic solvents. J. Chem. Eng. Data 2018, 63, 2671–2680. [Google Scholar] [CrossRef]
- Nowosielski, B.; Jamrógiewicz, M.; Łuczak, J.; Śmiechowski, M.; Warmińska, D. Experimental and predicted physicochemical properties of monopropanolamine-based deep eutectic solvents. J. Mol. Liq. 2020, 309, 113110–113122. [Google Scholar] [CrossRef]
- Dong, L.; Chen, J.; Gao, G. Solubility of carbon dioxide in aqueous solutions of 3-amino-1-propanol. J. Chem. Eng. Data 2010, 55, 1030–1034. [Google Scholar] [CrossRef]
- Shukla, S.K.; Mikkola, J.-P. Intermolecular interactions upon carbon dioxide capture in deep-eutectic solvents. Phys. Chem. Chem. Phys. 2018, 20, 24591–24601. [Google Scholar] [CrossRef]
- Zhang, K.; Hou, Y.; Wang, Y.; Wang, K.; Ren, S.; Wu, W. Efficient and reversible absorption of CO2 by functional deep eutectic solvents. Energy Fuels 2018, 32, 7727–7733. [Google Scholar] [CrossRef]
- Li, Z.; Wang, L.; Li, C.; Cui, Y.; Li, S.; Yang, G.; Shen, Y. Absorption of carbon dioxide using ethanolamine-based deep eutectic solvents. ACS Sustain. Chem. Eng. 2019, 7, 10403–10414. [Google Scholar] [CrossRef]
- Idris, Z.; Kummamuru, N.B.; Eimer, D.A. Viscosity measurement and correlation of unloaded and CO2-loaded 3-amino-1-propanol solution. J. Chem. Eng. Data 2018, 63, 1454–1459. [Google Scholar] [CrossRef]
- Saravanamurugan, S.; Kunov-Kruse, A.J.; Fehrmann, R.; Riisager, A. Amine-functionalized amino acid-based ionic liquids as efficient and high-capacity absorbents for CO2. ChemSusChem 2014, 7, 897–902. [Google Scholar] [CrossRef]
- Trivedi, T.J.; Lee, J.H.; Lee, H.J.; Jeong, Y.K.; Choi, J.W. Deep eutectic solvents as attractive media for CO2 capture. Green Chem. 2016, 18, 2834–2842. [Google Scholar] [CrossRef]
- Shukla, S.K.; Mikkola, J.-P. Unusual temperature-promoted carbon dioxide capture in deep-eutectic solvents: The synergistic interactions. Chem. Commun. 2019, 55, 3939–3942. [Google Scholar] [CrossRef]
- Sarmad, S.; Nikjoo, D.; Mikkola, J.-P. Amine functionalized deep eutectic solvent for CO2 capture: Measurements and modeling. J. Mol. Liq. 2020, 309, 113159. [Google Scholar] [CrossRef]
- Astarita, G.; Marrucci, G.; Gioia, F. The influence of carbonation ratio and total amine concentration on carbon dioxide absorption in aqueous monoethanolamine solutions. Chem. Eng. Sci. 1964, 19, 95–103. [Google Scholar] [CrossRef]
- Camacho, B.F.; Sunchez, S.; Pacheco, R. Thermal effects during the absorption of CO2 in aqueous solutions of 3-amino-1-propanol. Chem. Eng. Technol. 2000, 23, 1073–1080. [Google Scholar] [CrossRef]
- Benamor, A.; Al-Marri, M.J.; Hawari, A. Experimental determination of carbamate formation and amine protonation constants in 3-amino-1-propanol–CO2–H2O system and their temperature dependency. Int. J. Greenh. Gas Control 2015, 37, 237–242. [Google Scholar] [CrossRef]


| Chemical Name | Source | CAS Number | Purity/Mass Fraction a |
|---|---|---|---|
| 3-amino-1-propanol [AP] | Sigma Aldrich | 157-87-6 | 0.99 |
| Tetrabutylammonium bromide [TBAB] | Sigma Aldrich | 1643-19-2 | ≥0.99 |
| Tetrabutylammonium chloride [TBAC] | Sigma Aldrich | 1112-67-0 | ≥0.99 |
| Tetraethylammonium chloride [TEAC] | Sigma Aldrich | 56-34-8 | ≥0.99 |
| DES | Molar Ratio | ƞ/(mPa.s) | ET(30)/(kcal/mol) | π | α | β | |α−β| |
|---|---|---|---|---|---|---|---|
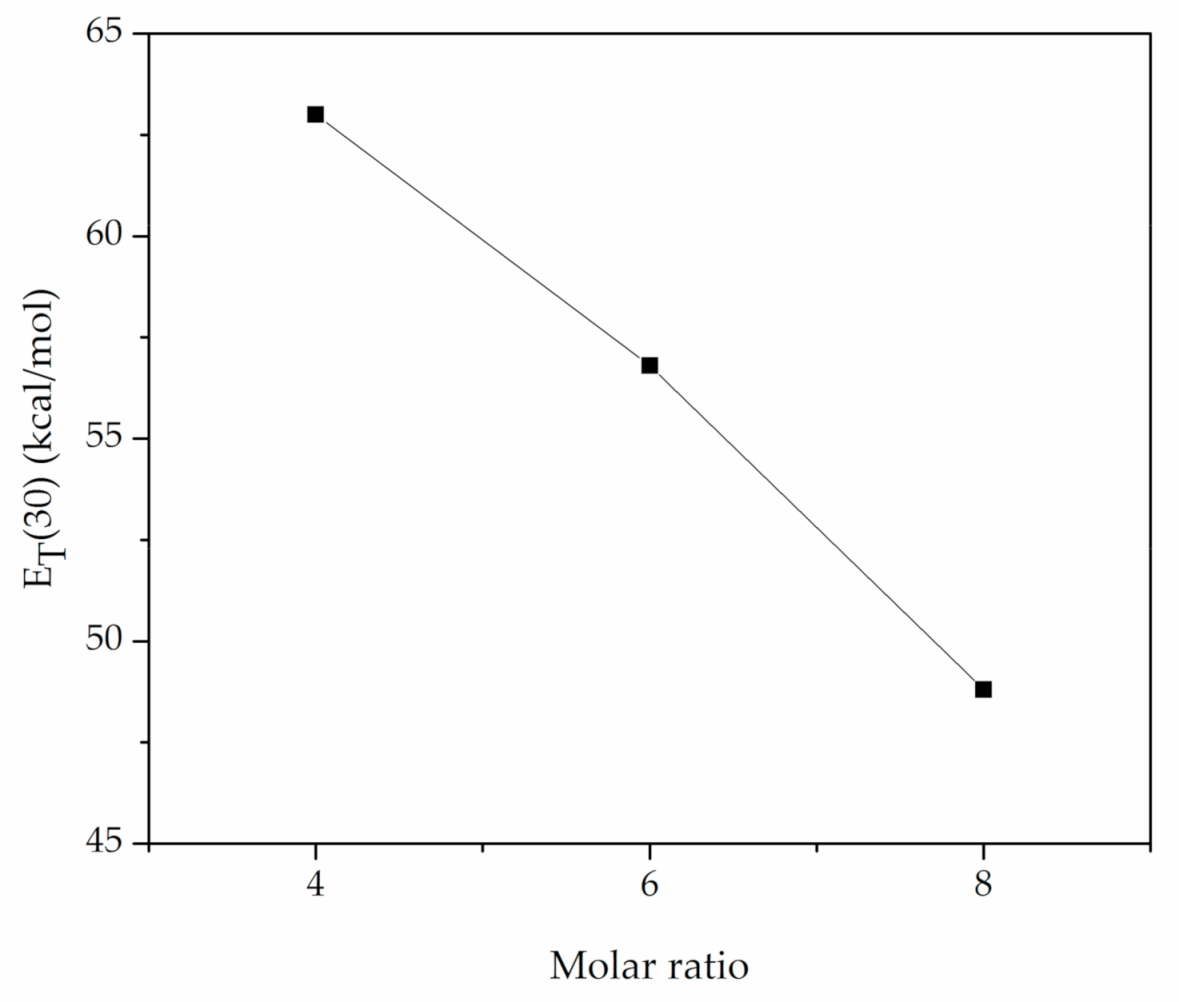
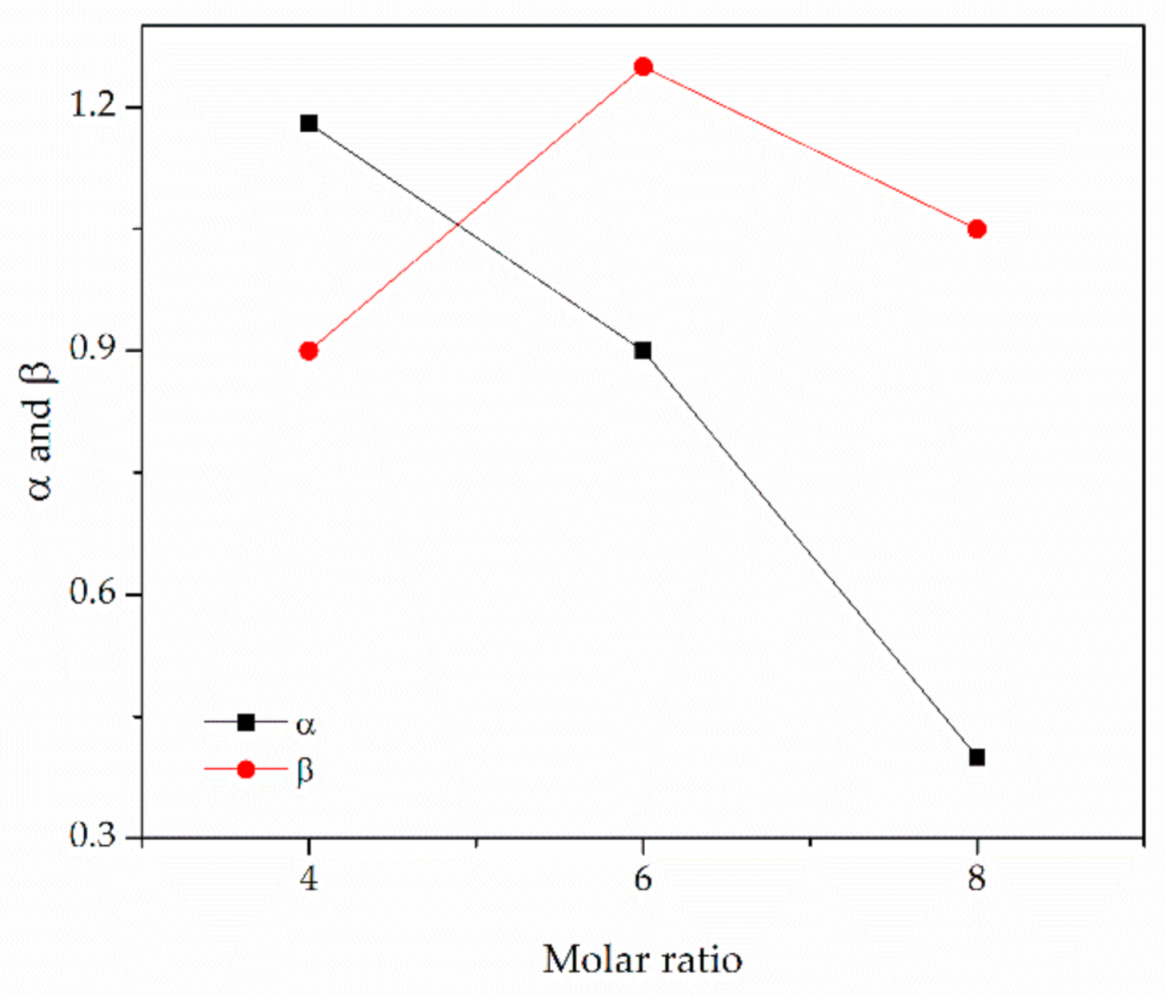
| TBAB-AP | 1:4 | 84.69 | 48.7 (48.5 a) | 1.07 (1.02 a) | 0.37 (0.39 a) | 1.00 (0.90 a) | 0.63 |
| TEAC-AP | 1:4 | 42.37 | 56.7 | 1.10 | 0.87 | 0.92 | 0.05 |
| TBAC-AP | 1:4 | 77.36 | 63.0 | 1.13 | 1.18 | 0.90 | 0.28 |
| TBAC-AP | 1:6 | 53.86 | 56.8 | 1.02 | 0.90 | 1.25 | 0.34 |
| TBAC-AP | 1:8 | 46.14 | 48.8 | 1.04 | 0.40 | 1.05 | 0.65 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cichowska-Kopczyńska, I.; Warmińska, D.; Nowosielski, B. Solubility of Carbon Dioxide in Deep Eutectic Solvents Based on 3-Amino-1-Propanol and Tetraalkylammonium Salts at Low Pressure. Materials 2021, 14, 594. https://doi.org/10.3390/ma14030594
Cichowska-Kopczyńska I, Warmińska D, Nowosielski B. Solubility of Carbon Dioxide in Deep Eutectic Solvents Based on 3-Amino-1-Propanol and Tetraalkylammonium Salts at Low Pressure. Materials. 2021; 14(3):594. https://doi.org/10.3390/ma14030594
Chicago/Turabian StyleCichowska-Kopczyńska, Iwona, Dorota Warmińska, and Bartosz Nowosielski. 2021. "Solubility of Carbon Dioxide in Deep Eutectic Solvents Based on 3-Amino-1-Propanol and Tetraalkylammonium Salts at Low Pressure" Materials 14, no. 3: 594. https://doi.org/10.3390/ma14030594
APA StyleCichowska-Kopczyńska, I., Warmińska, D., & Nowosielski, B. (2021). Solubility of Carbon Dioxide in Deep Eutectic Solvents Based on 3-Amino-1-Propanol and Tetraalkylammonium Salts at Low Pressure. Materials, 14(3), 594. https://doi.org/10.3390/ma14030594






