Green Synthesis of Silver-Decorated Magnetic Particles for Efficient and Reusable Antimicrobial Activity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of the Ethanolic Extract of Green Tea Leaves
2.3. Synthesis of Fe2O3 NPs
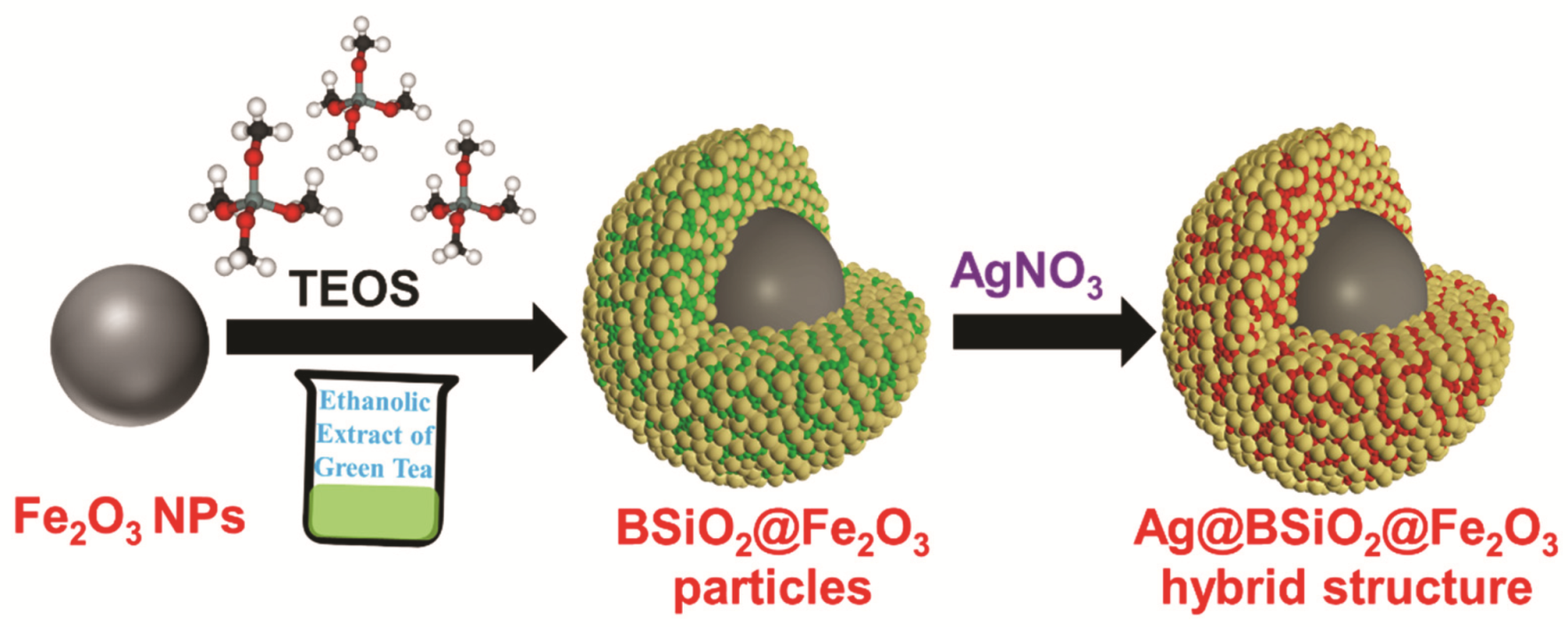
2.4. Synthesis of BSiO2-Fe2O3 Particles
2.5. Synthesis of the Ag-BSiO2-Fe2O3 Hybrid Microstructure
2.6. Antimicrobial Activity
2.7. Biochemical Analysis
2.7.1. Estimation of Total Phenolic Content
2.7.2. DPPH Radical Scavenging Activity
2.7.3. ABTS Radical Scavenging Assay
2.8. Analytical Instrumentation
3. Results and Discussion
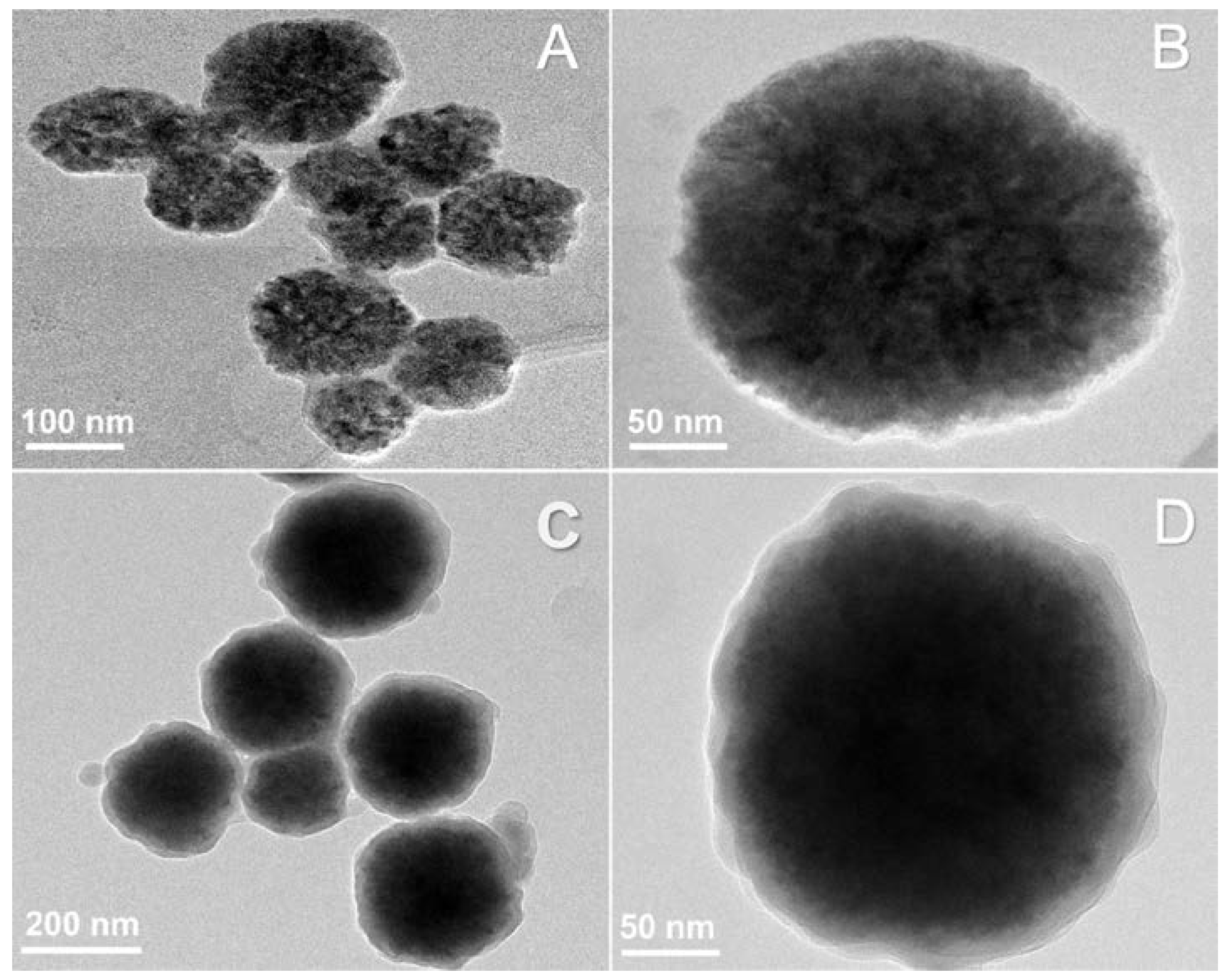
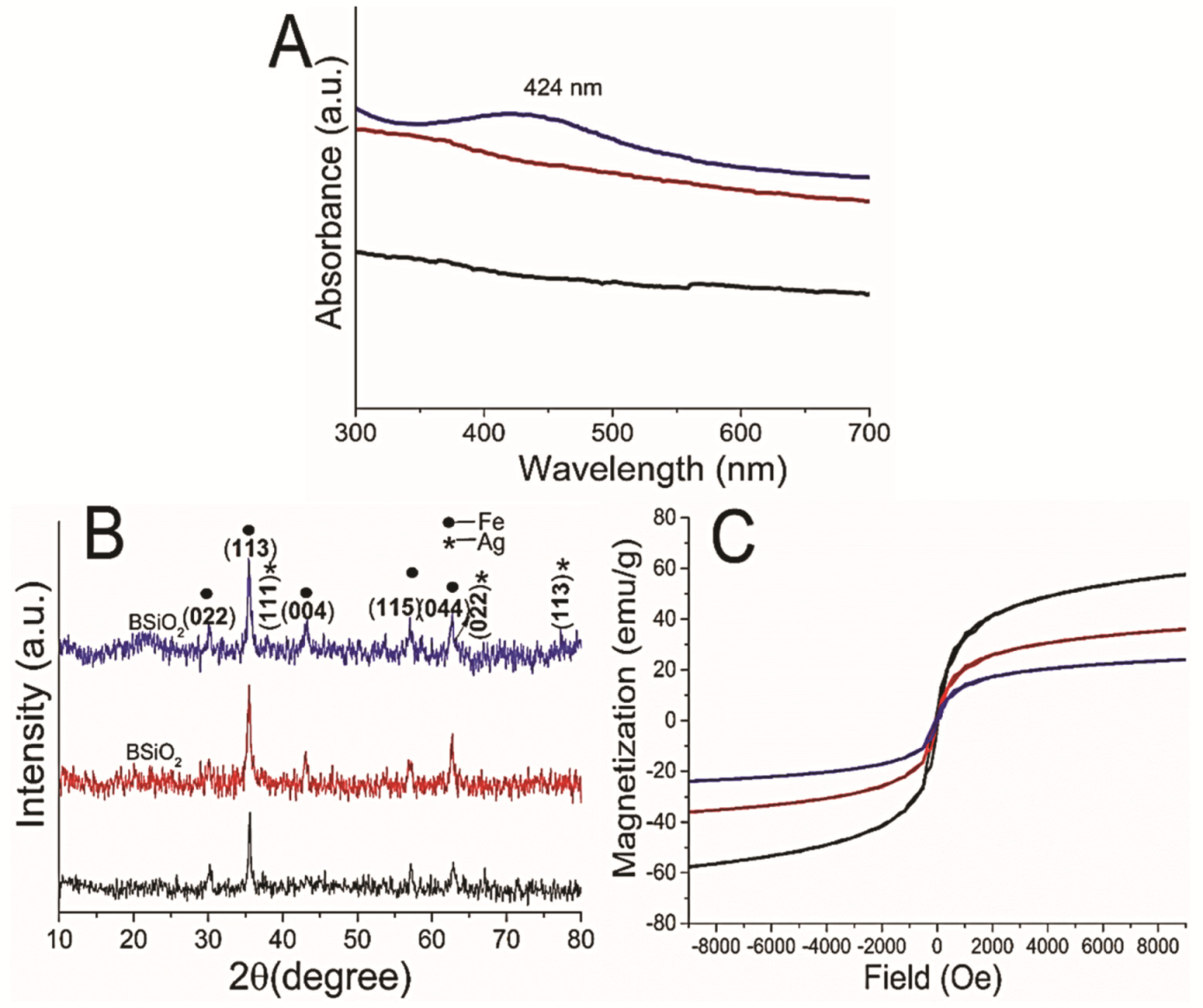
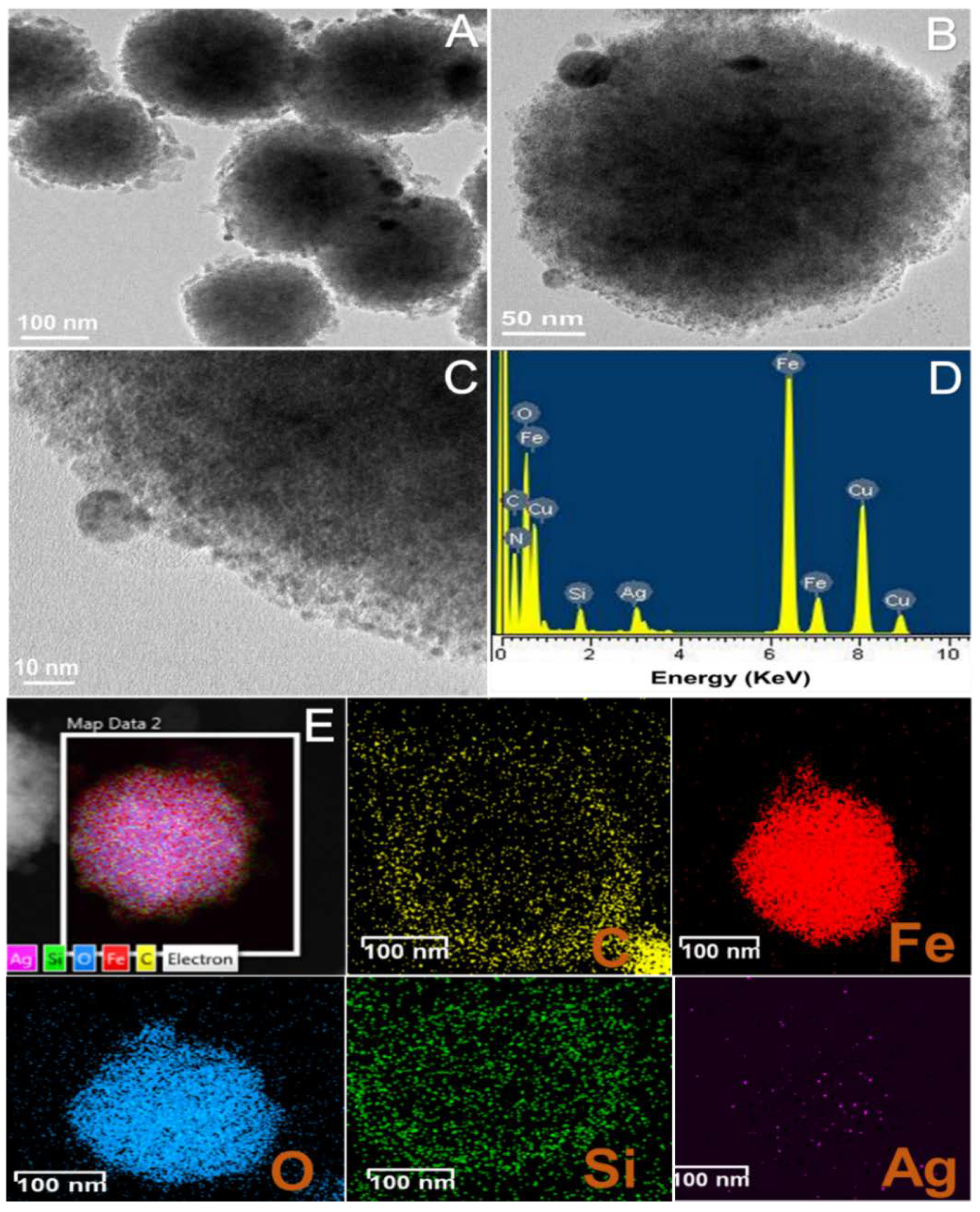
3.1. Synthesis of the Hybrid Microstructure
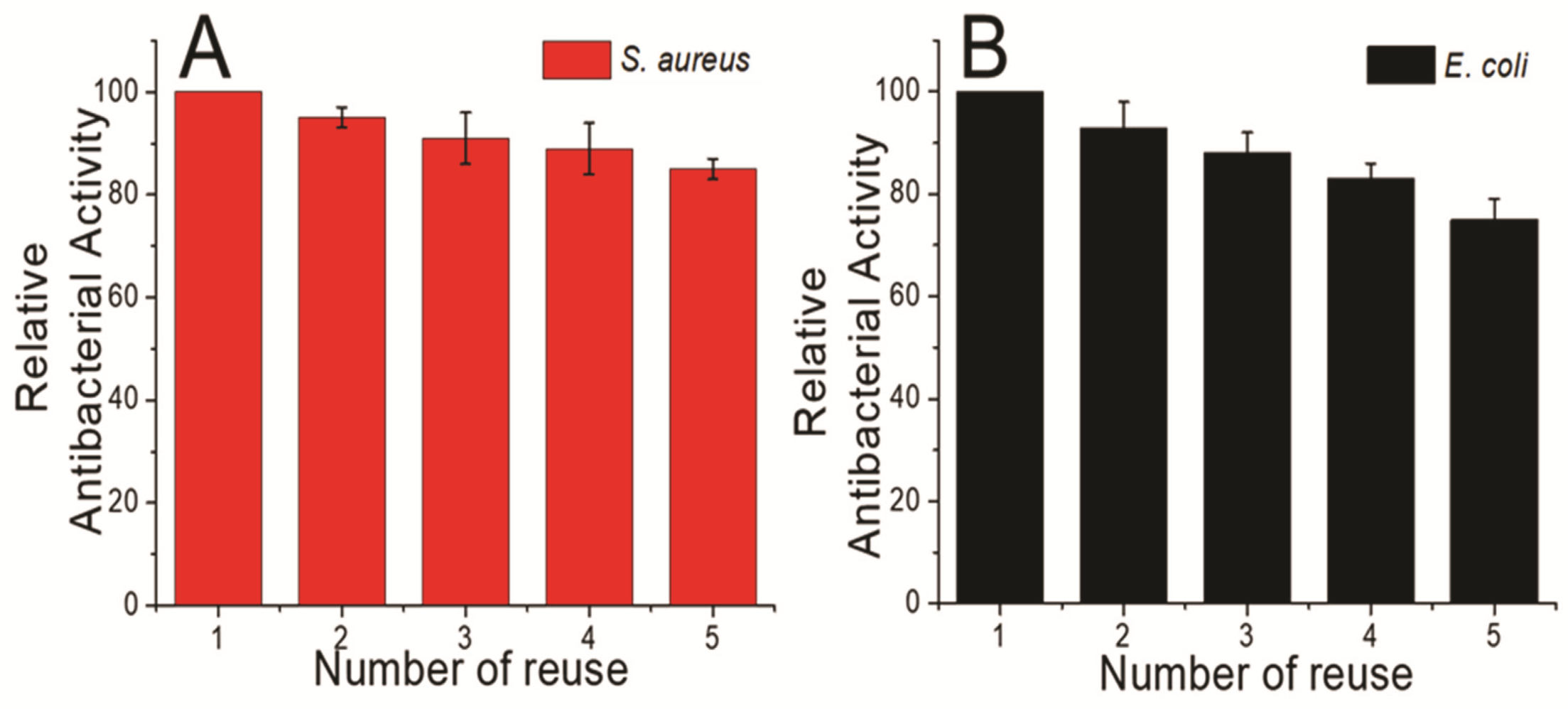
3.2. Antimicrobial Activity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tenover, F.C. Mechanisms of antimicrobial resistance in bacteria. Am. J. Infect. Med. 2006, 119, S3–S10. [Google Scholar] [CrossRef] [PubMed]
- Holmes, A.H.; Moore, L.S.P.; Sundsfjord, A.; Steinbakk, M.; Regmi, S.; Karkey, A.; Guerin, P.J.; Piddock, L.J.V. Understanding the mechanisms and drivers of antimicrobial resistance. Lancet 2016, 387, 176–187. [Google Scholar] [CrossRef]
- Kim, H.U.; Blin, K.; Lee, S.Y.; Weber, T. Recent development of computational resources for new antibiotics discovery. Curr. Opin. Microbiol. 2017, 39, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Mumtaz, S.; Li, C.H.; Hussain, I.; Rotello, V.M. Combatting antibiotic-resistant bacteria using nanomaterials. Chem. Soc. Rev. 2019, 48, 415–427. [Google Scholar] [CrossRef] [PubMed]
- Hemeg, H.A. Nanomaterials for alternative antibacterial therapy. Int. J. Nanomed. 2017, 12, 8211–8225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bandow, J.E.; Metzler-Nolte, N. New ways of killing the beast: Prospects for inorganic-organic hybrid nanomaterials as antibacterial agents. ChemBioChem 2009, 10, 2847–2850. [Google Scholar] [CrossRef] [PubMed]
- Yacoby, I.; Benhar, I. Antibacterial nanomedicine. Antibact. Nanomed. 2008, 3, 329–341. [Google Scholar] [CrossRef]
- Rai, M.; Yadav, A.; Gade, A. Silver nanoparticles as a new generation of antimicrobials. Biotechnol. Adv. 2009, 27, 76–83. [Google Scholar] [CrossRef]
- Otari, S.V.; Patil, R.M.; Nadaf, N.H.; Ghosh, S.J.; Pawar, S.H. Green biosynthesis of silver nanoparticles from an actinobacteria Rhodococcus sp. Mater. Lett. 2012, 72, 92–94. [Google Scholar] [CrossRef]
- Lok, C.N.; Ho, C.M.; Chen, R.; He, Q.Y.; Yu, W.Y.; Sun, H.; Tam, P.K.H.; Chiu, J.F.; Che, C.M. Silver nanoparticles: Partial oxidation and antibacterial activities. JBIC J. Biol. Inorg. Chem. 2007, 12, 527–534. [Google Scholar] [CrossRef] [PubMed]
- Choi, O.; Deng, K.K.; Kim, N.J.; Ross, L.; Surampalli, R.Y.; Hu, Z. The inhibitory effects of silver nanoparticles, silver ions, and silver chloride colloids on microbial growth. Water Res. 2008, 42, 3066–3074. [Google Scholar] [CrossRef] [PubMed]
- Ahamed, M.; AlSalhi, M.S.; Siddiqui, M.K.J. Silver nanoparticle applications and human health. Clin. Chim. Acta 2010, 411, 1841–1848. [Google Scholar] [CrossRef]
- Wu, F.; Harper, B.J.; Harper, S.L. Differential dissolution and toxicity of surface functionalized silver nanoparticles in small-scale microcosms: Impacts of community complexity. Environ. Sci. Nano 2017, 4, 359–372. [Google Scholar] [CrossRef]
- Zhao, X.S.; Bao, X.Y.; Guo, W.; Lee, F.Y. Immobilizing catalysts on porous materials. Mater. Today 2006, 9, 32–39. [Google Scholar] [CrossRef]
- Dalpozzo, R. Magnetic nanoparticle supports for asymmetric catalysts. Green Chem. 2015, 17, 3671–3686. [Google Scholar] [CrossRef]
- Tzounis, L.; Logothetidis, S. Fe3O4@SiO2 core shell particles as platforms for the decoration of Ag nanoparticles. Mater. Today Proc. 2017, 4, 7076–7082. [Google Scholar] [CrossRef]
- Sui, N.; Monnier, V.; Salvia, M.V.; Chevolot, Y.; Souteyrand, E. Magnetic and optical properties of Ag@SiO2-FITC-Fe3O4 hybrid nanoparticles. Mater. Sci. Eng. B 2014, 182, 92–95. [Google Scholar] [CrossRef]
- Das, S.K.; Khan, M.M.R.; Parandhaman, T.; Laffir, F.; Guha, A.K.; Sekaran, G.; Mandal, A.B. Nano-silica fabricated with silver nanoparticles: Antifouling adsorbent for efficient dye removal, effective water disinfection and biofouling control. Nanoscale 2013, 5, 5549–5560. [Google Scholar] [CrossRef]
- Kaloti, M.; Kumar, A.; Navani, N.K. Synthesis of glucose-mediated Ag-γ-Fe2O3 multifunctional nanocomposites in aqueous medium—A kinetic analysis of their catalytic activity for 4-nitrophenol reduction. Green Chem. 2015, 17, 4786–4799. [Google Scholar] [CrossRef]
- Tzounis, L.; Contreras-Caceres, R.; Schellkopf, L.; Jehnichen, D.; Fischer, D.; Cai, C.; Uhlmann, P.; Stamm, M. Controlled growth of Ag nanoparticles decorated onto the surface of SiO2 spheres: A nanohybrid system with combined SERS and catalytic properties. RSC Adv. 2014, 4, 17846–17855. [Google Scholar] [CrossRef]
- Muthukumar, H.; Palanirajan, S.K.; Shanmugam, M.K.; Gummadi, S.N. Plant extract mediated synthesis enhanced the functional properties of silver ferrite nanoparticles over chemical mediated synthesis. Biotechnol. Rep. 2020, 26, e00469. [Google Scholar] [CrossRef]
- Darezereshki, E. Synthesis of maghemite (γ-Fe2O3) nanoparticles by wet chemical method at room temperature. Mater. Lett. 2010, 64, 1471–1472. [Google Scholar] [CrossRef]
- Deng, Y.H.; Wang, C.C.; Hu, J.H.; Yang, W.L.; Fu, S.K. Investigation of formation of silica-coated magnetite nanoparticles via sol-gel approach. Colloids Surf. A Physicochem. Eng. Asp. 2005, 262, 87–93. [Google Scholar] [CrossRef]
- Chaurasia, A.K.; Thorat, N.D.; Tandon, A.; Kim, J.H.; Park, S.H.; Kim, K.K. Coupling of radiofrequency with magnetic nanoparticles treatment as an alternative physical antibacterial strategy against multiple drug resistant bacteria. Sci. Rep. 2016, 6, 33662. [Google Scholar] [CrossRef] [Green Version]
- de Oliveira, A.C.; Valentim, I.B.; Silva, C.A.; Bechara, E.J.H.; de Barros, M.P.; Mano, C.M.; Goulart, M.O.F. Total phenolic content and free radical scavenging activities of methanolic extract powders of tropical fruit residues. Food Chem. 2009, 115, 469–475. [Google Scholar] [CrossRef]
- Ahmed, S.; Ahmad, M.; Swami, B.L.; Ikram, S. A review on plants extract mediated synthesis of silver nanoparticles for antimicrobial applications: A green expertise. J. Adv. Res. 2016, 7, 17–28. [Google Scholar] [CrossRef] [Green Version]
- Rónavári, A.; Kovács, D.; Igaz, N.; Vágvölgyi, C.; Boros, I.M.; Kónya, Z.; Pfeiffer, I.; Kiricsi, M. Biological activity of green-synthesized silver nanoparticles depends on the applied natural extracts: A comprehensive study. Int. J. Nanomed. 2017, 12, 871–883. [Google Scholar] [CrossRef] [Green Version]
- Rónavári, A.; Bélteky, P.; Boka, E.; Zakupszky, D.; Igaz, N.; Szerencsés, B.; Pfeiffer, I.; Kónya, Z.; Kiricsi, M. Polyvinyl-pyrrolidone-coated silver nanoparticles—The colloidal, chemical and biological consequences of steric stabilization under biorelevant conditions. Int. J. Mol. Sci. 2021, 22, 8673. [Google Scholar] [CrossRef] [PubMed]
- Bélteky, P.; Rónavári, A.; Igaz, N.; Szerencsés, B.; Tóth, I.Y.; Pfeiffer, I.; Kiricsi, M.; Kónya, Z. Silver nanoparticles: Aggregation behavior in biorelevant conditions and its impact on biological activity. Int. J. Nanomed. 2019, 14, 667–687. [Google Scholar] [CrossRef] [Green Version]
- Hussain, S.; Khan, Z. Epigallocatechin-3-gallate-capped Ag nanoparticles: Preparation and characterization. Bioprocess Biosyst. Eng. 2014, 37, 1221–1231. [Google Scholar] [CrossRef] [PubMed]
- Moulton, M.C.; Braydich-Stolle, L.K.; Nadagouda, M.N.; Kunzelman, S.; Hussain, S.M.; Varma, R.S. Synthesis, characterization and biocompatibility of “green” synthesized silver nanoparticles using tea polyphenols. Nanoscale 2010, 2, 763–770. [Google Scholar] [CrossRef] [PubMed]
- Trouillas, P.; Marsal, P.; Siri, D.; Lazzaroni, R.; Duroux, J.L. A DFT study of the reactivity of OH groups in quercetin and taxifolin antioxidants: The specificity of the 3-OH site. Food Chem. 2006, 97, 679–688. [Google Scholar] [CrossRef]
- Navale, S.T.; Bandgar, D.K.; Nalage, S.R.; Khuspe, G.D.; Chougule, M.A.; Kolekar, Y.D.; Sen, S.; Patil, V.B. Synthesis of Fe2O3 nanoparticles for nitrogen dioxide gas sensing applications. Ceram. Int. 2013, 39, 6453–6460. [Google Scholar] [CrossRef]
- Otari, S.V.; Patil, R.M.; Waghmare, S.R.; Ghosh, S.J.; Pawar, S.H. A novel microbial synthesis of catalytically active Ag-alginate biohydrogel and its antimicrobial activity. Dalton Trans. 2013, 42, 9966–9975. [Google Scholar] [CrossRef] [PubMed]
- Shin, K.S.; Cho, Y.K.; Choi, J.-Y.; Kim, K. Facile synthesis of silver-deposited silanized magnetite nanoparticles and their application for catalytic reduction of nitrophenols. Appl. Catal. A Gen. 2012, 413–414, 170–175. [Google Scholar] [CrossRef]
- Yi, T.; Zhu, L.; Peng, W.L.; He, X.C.; Chen, H.L.; Li, J.; Yu, T.; Liang, Z.T.; Zhao, Z.Z.; Chen, H.B. Comparison of ten major constituents in seven types of processed tea using HPLC-DAD-MS followed by principal component and hierarchical cluster analysis. LWT—Food Sci. Technol. 2015, 62, 194–201. [Google Scholar] [CrossRef] [Green Version]
- Li, Z.; Zhu, Y. Surface-modification of SiO2 nanoparticles with oleic acid. Appl. Surf. Sci. 2003, 211, 315–320. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Otari, S.V.; Kalia, V.C.; Bisht, A.; Kim, I.-W.; Lee, J.-K. Green Synthesis of Silver-Decorated Magnetic Particles for Efficient and Reusable Antimicrobial Activity. Materials 2021, 14, 7893. https://doi.org/10.3390/ma14247893
Otari SV, Kalia VC, Bisht A, Kim I-W, Lee J-K. Green Synthesis of Silver-Decorated Magnetic Particles for Efficient and Reusable Antimicrobial Activity. Materials. 2021; 14(24):7893. https://doi.org/10.3390/ma14247893
Chicago/Turabian StyleOtari, Sachin V., Vipin Chandra Kalia, Aarti Bisht, In-Won Kim, and Jung-Kul Lee. 2021. "Green Synthesis of Silver-Decorated Magnetic Particles for Efficient and Reusable Antimicrobial Activity" Materials 14, no. 24: 7893. https://doi.org/10.3390/ma14247893
APA StyleOtari, S. V., Kalia, V. C., Bisht, A., Kim, I.-W., & Lee, J.-K. (2021). Green Synthesis of Silver-Decorated Magnetic Particles for Efficient and Reusable Antimicrobial Activity. Materials, 14(24), 7893. https://doi.org/10.3390/ma14247893





