Finite Element Analysis of Customized Acetabular Implant and Bone after Pelvic Tumour Resection throughout the Gait Cycle
Abstract
:1. Introduction
2. Materials and Methods
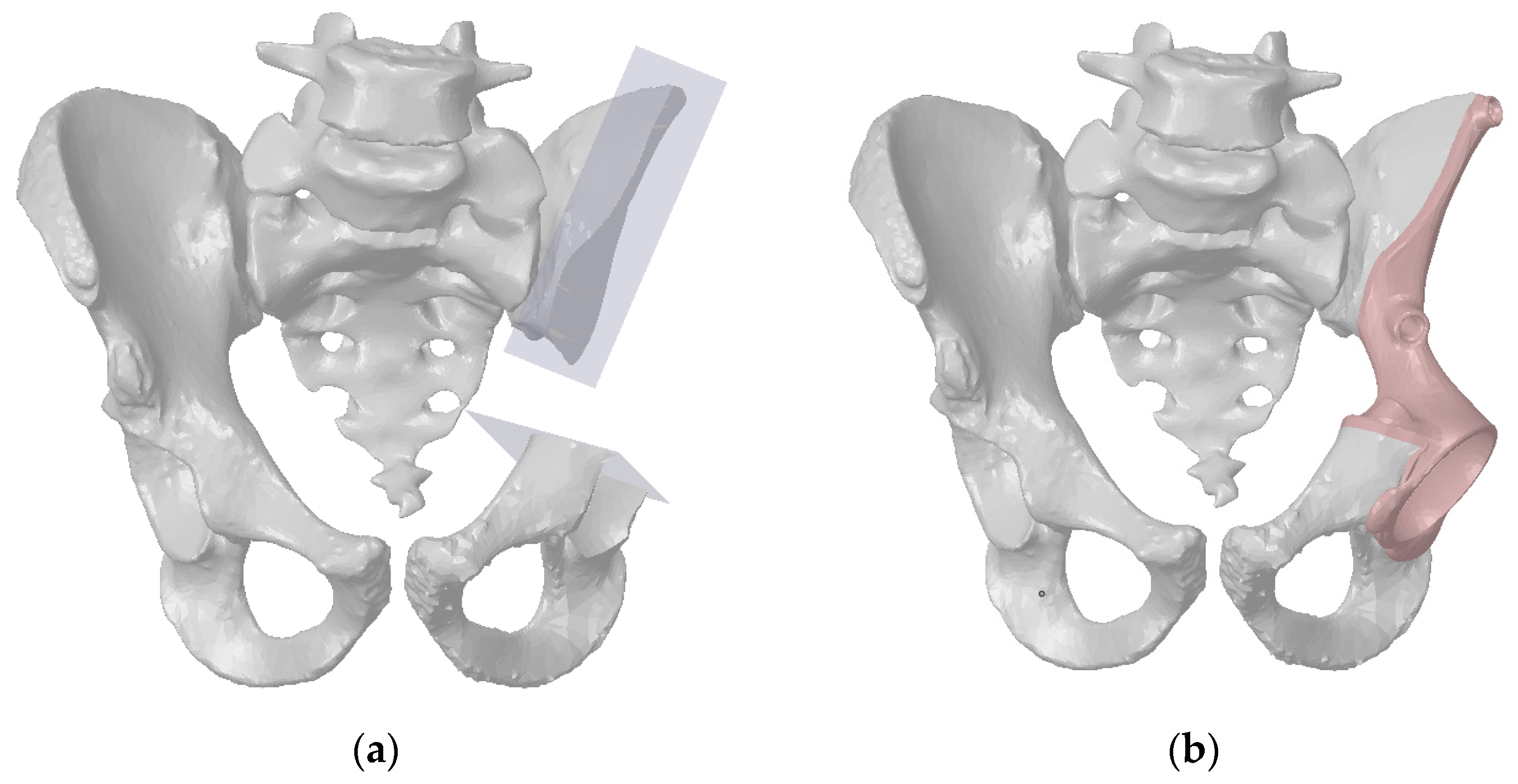
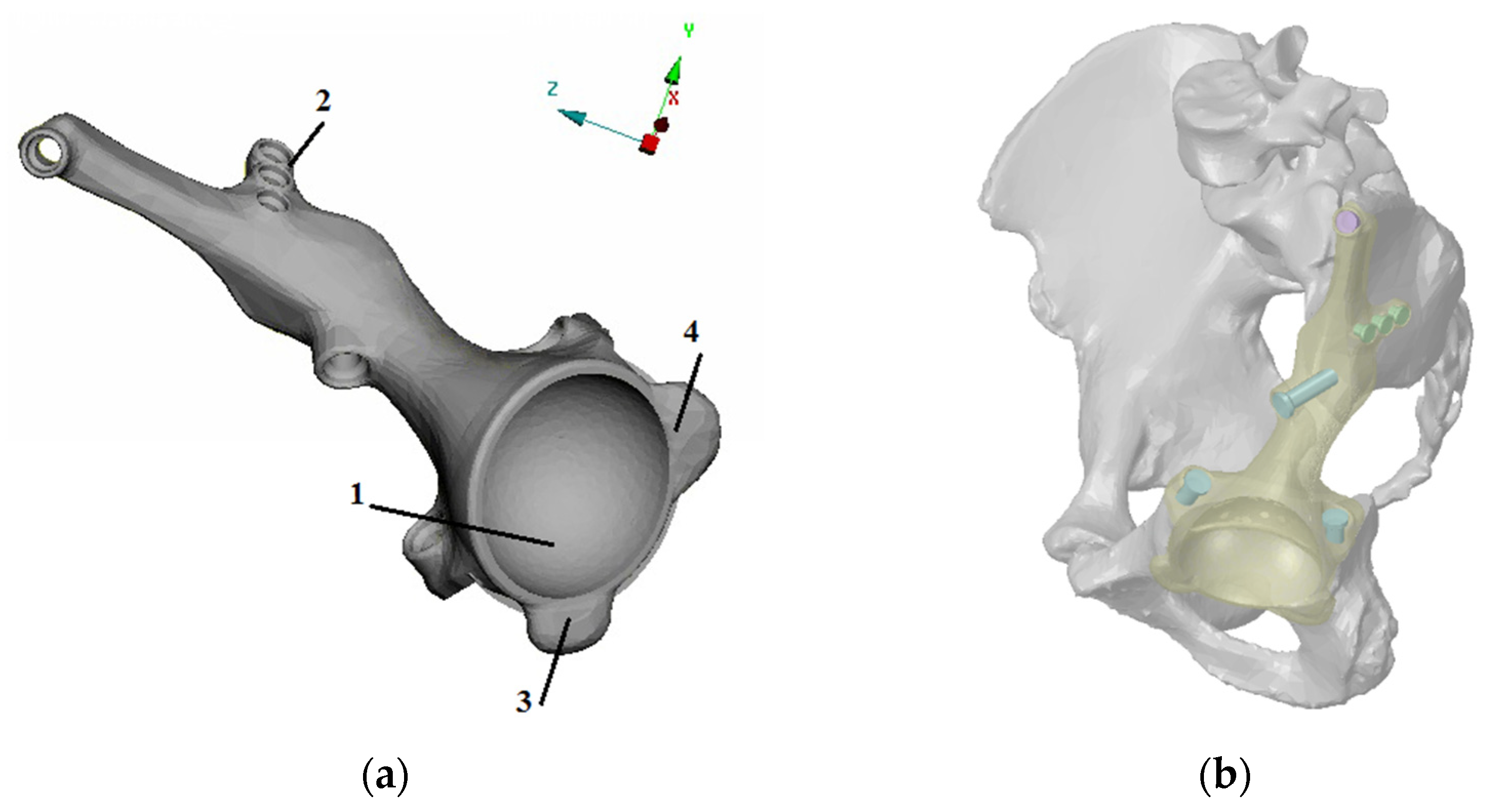
2.1. Finite Element Models
2.2. Material Properties
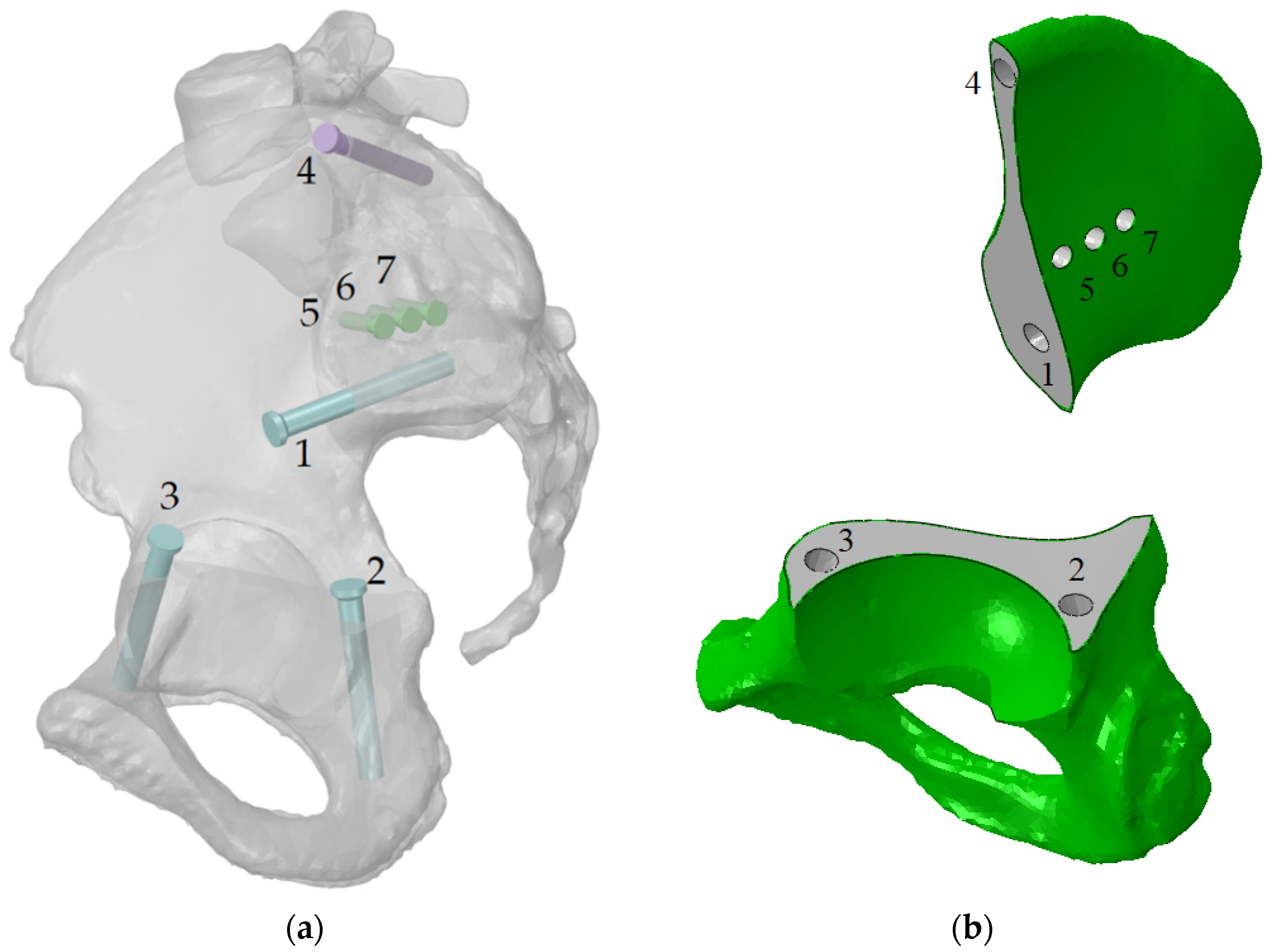
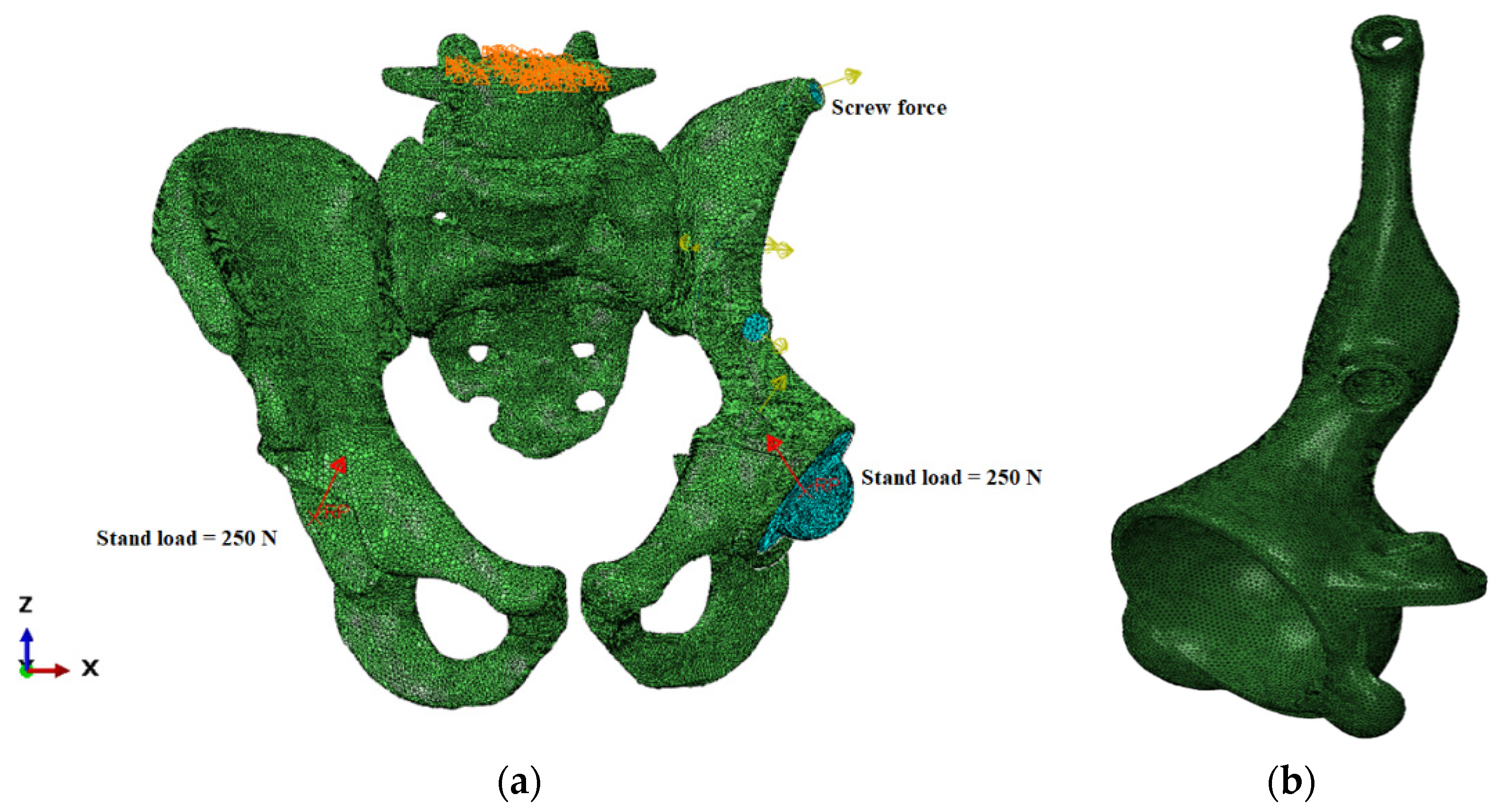
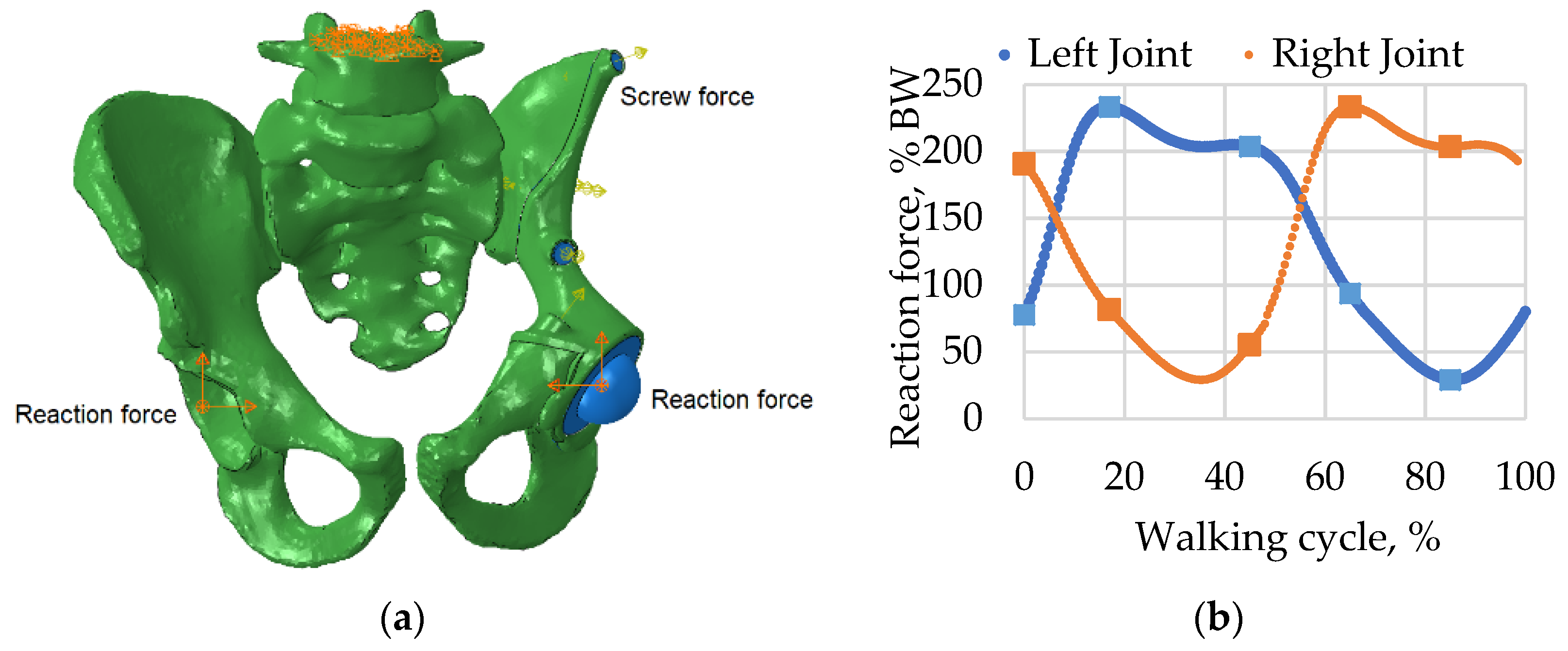
2.3. Loads and Boundary Conditions
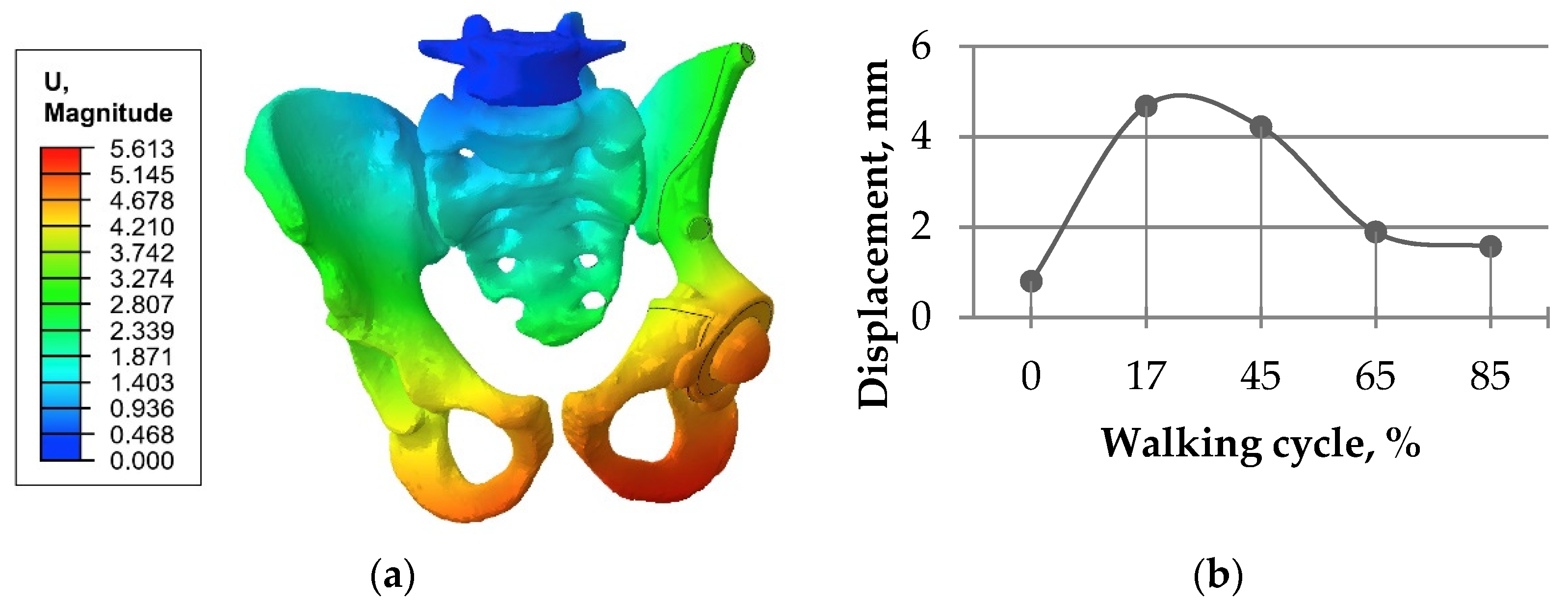
2.4. Model Validation
3. Results
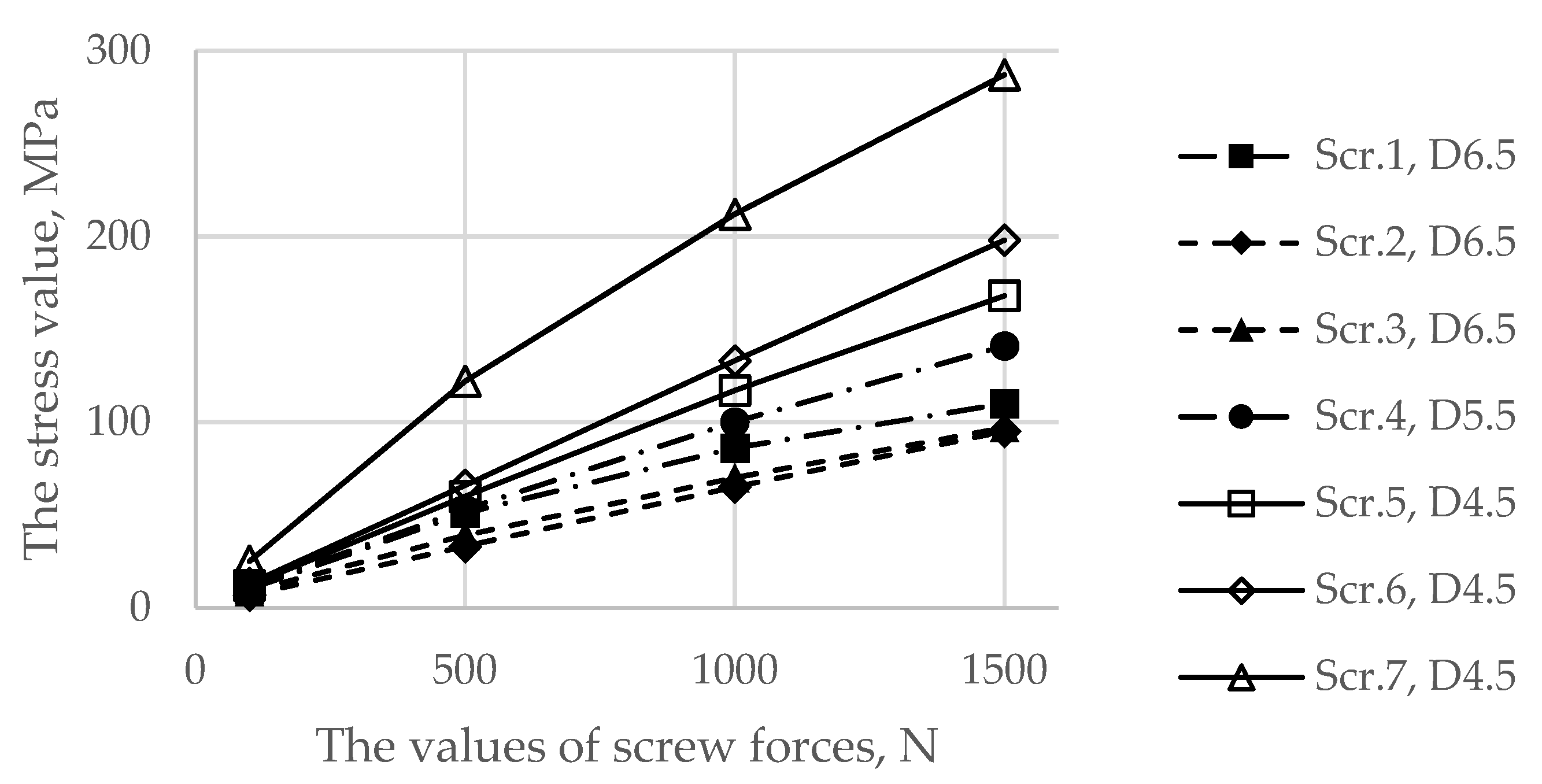
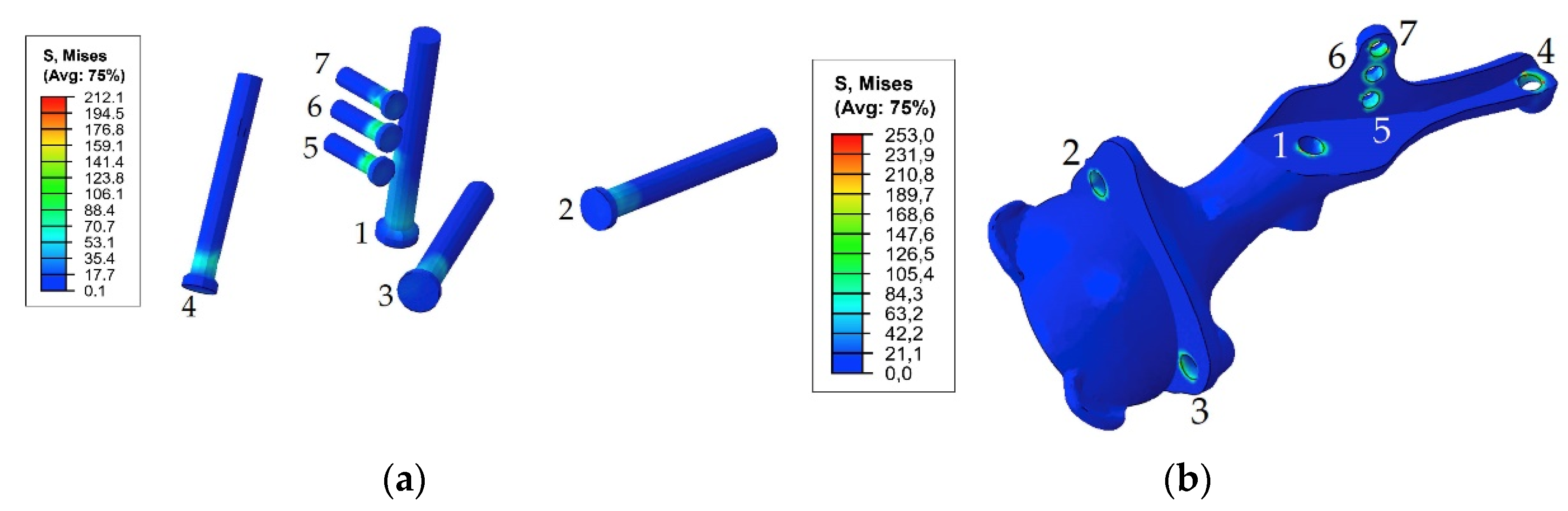
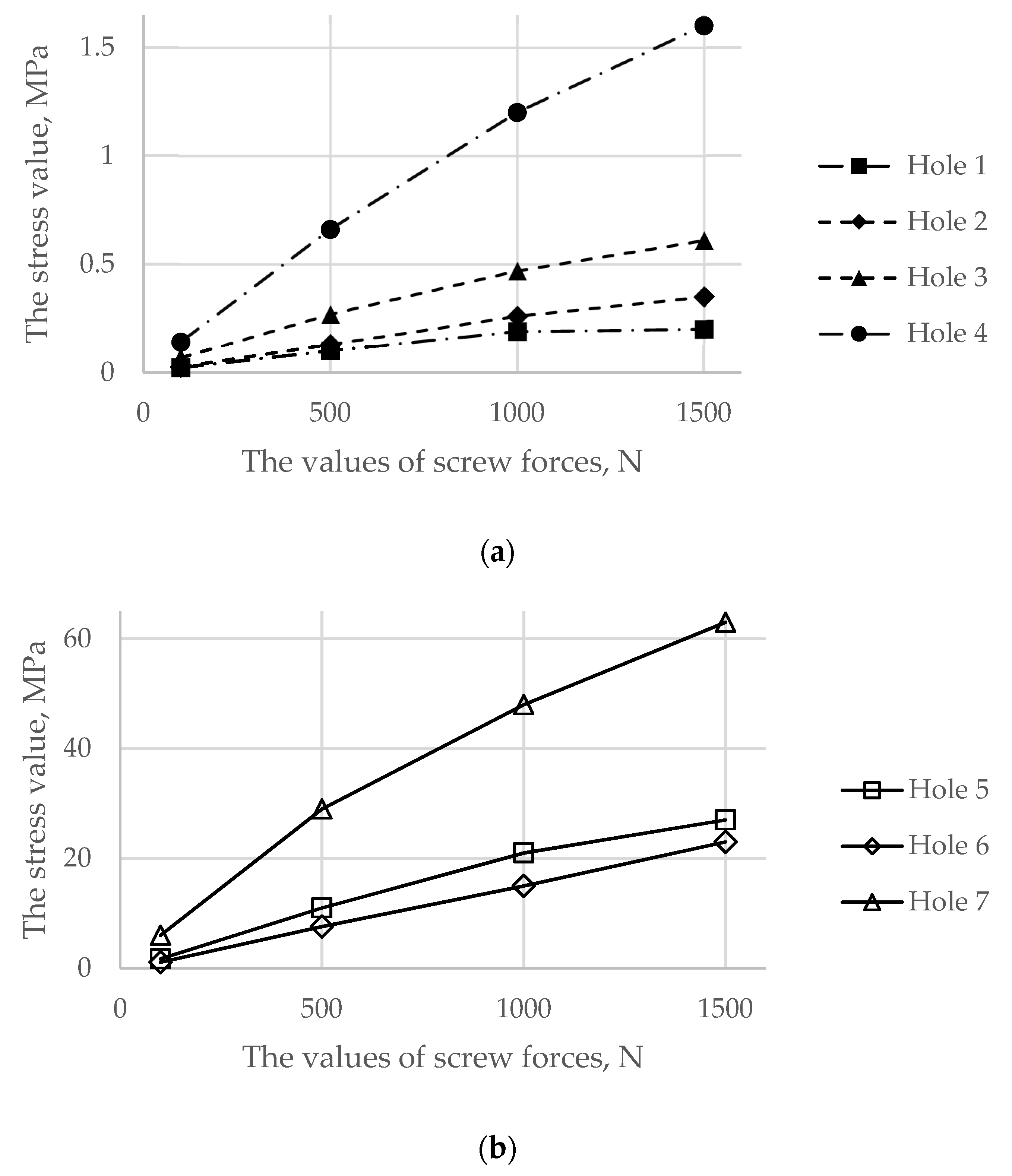
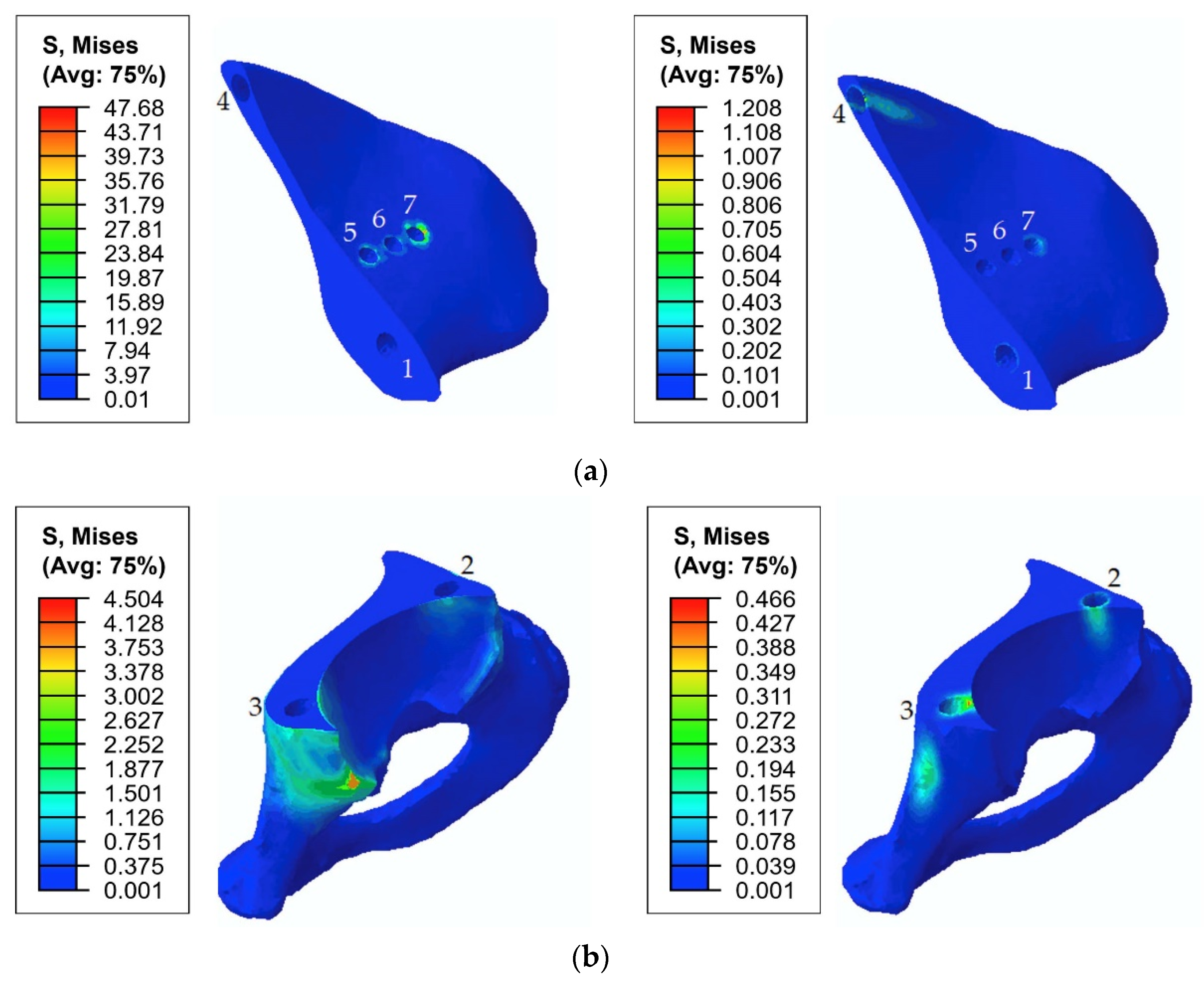
3.1. Results for the Stage of Tightening the Screw Simulation
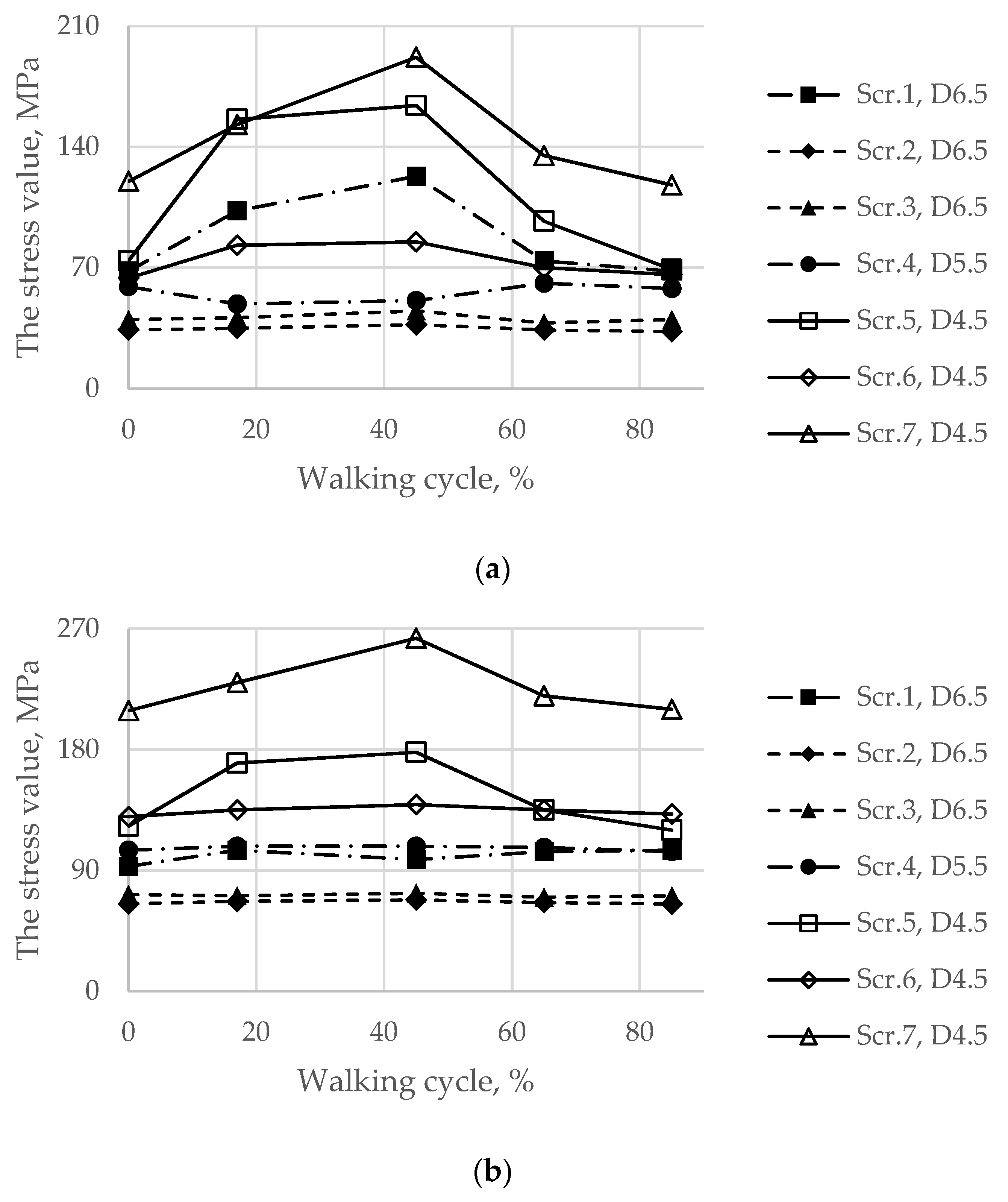
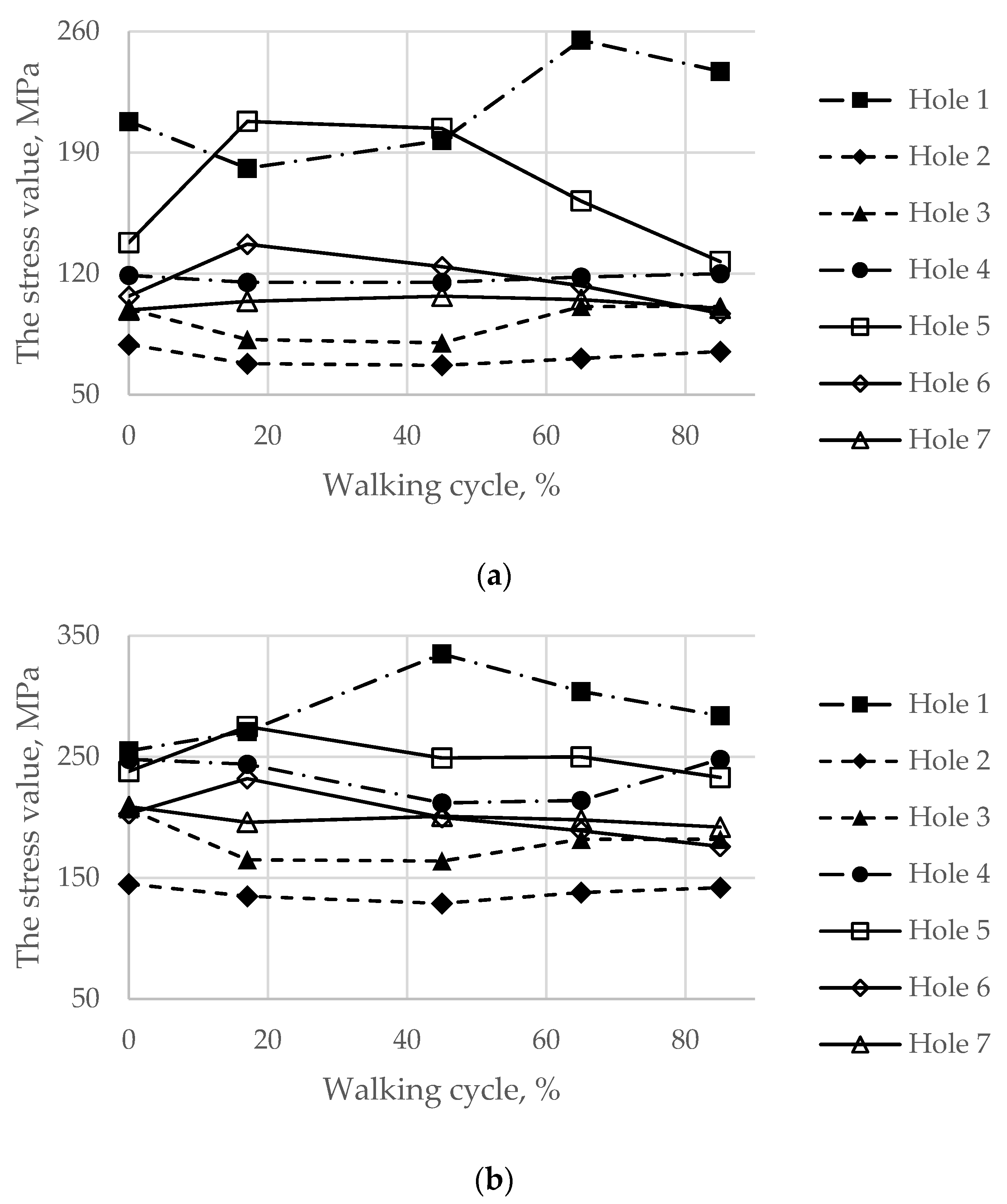
3.2. Results of the Walking Cycle Simulation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Iqbal, T.; Shi, L.; Wang, L.; Liu, Y.; Li, D.; Qin, M.; Jin, Z. Development of Finite Element Model for Customized Prostheses Design for Patient with Pelvic Bone Tumor. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 2017, 231, 525–533. [Google Scholar] [CrossRef] [PubMed]
- Dong, E.; Wang, L.; Iqbal, T.; Li, D.; Liu, Y.; He, J.; Zhao, B.; Li, Y. Finite Element Analysis of the Pelvis after Customized Prosthesis Reconstruction. J. Bionic Eng. 2018, 15, 443–451. [Google Scholar] [CrossRef]
- Jia, Y.; Cheng, L.; Yu, G.; Ding, Z. Finite Element Analysis of Pelvic Reconstruction Using Fibular Transplantation Fixed with Rod-Screw System after Type I Resection. In Proceedings of the 2007 1st International Conference on Bioinformatics and Biomedical Engineering, Wuhan, China, 6–8 July 2007; pp. 430–433. [Google Scholar] [CrossRef]
- Shim, V.; Höch, A.; Grunert, R.; Peldschus, S.; Böhme, J. Development of a Patient-Specific Finite Element Model for Predicting Implant Failure in Pelvic Ring Fracture Fixation. Comput. Math. Methods Med. 2017, 2017, 9403821. [Google Scholar] [CrossRef] [Green Version]
- Hu, P.; Wu, T.; Wang, H.Z.; Qi, X.Z.; Yao, J.; Cheng, X.D.; Chen, W.; Zhang, Y.Z. Influence of Different Boundary Conditions in Finite Element Analysis on Pelvic Biomechanical Load Transmission. Orthop. Surg. 2017, 9, 115–122. [Google Scholar] [CrossRef] [Green Version]
- Kluess, D.; Wieding, J.; Souffrant, R.; Mittelmeier, W.; Bader, R. Finite Element Analysis in Orthopedic Biomechanics. In Finite Element Analysis; Moratal, D., Ed.; Sciyo: Rijeka, Croatia, 2010; pp. 151–170. [Google Scholar]
- Hao, Z.; Wan, C.; Gao, X.; Ji, T. The Effect of Boundary Condition on the Biomechanics of a Human Pelvic Joint under an Axial Compressive Load: A Three-Dimensional Finite Element Model. J. Biomech. Eng. 2011, 133, 101006. [Google Scholar] [CrossRef]
- Sushentsov, E.A.; Musaev, E.R.; Maslov, L.B.; Zhmaylo, M.A.; Sofronov, D.I.; Agaev, D.K.; Dzampaev, A.Z.; Romantsova, O.M.; Fedorova, A.V.; Aliev, M.D. Computer simulation, 3d-printing and custom-made prosthetics in treatment of a patient with osteosarcoma of the pelvis. Bone Soft Tissue Sarcomas Tumors Skin 2019, 11, 53–61. [Google Scholar]
- Dalstra, M.; Huiskes, R.; van Erning, L. Development and Validation of a Three-dimensional Finite Element Model of the Pelvic Bone. J. Biomech. Eng. 1995, 117, 272–278. [Google Scholar] [CrossRef] [Green Version]
- Liu, D.; Hua, Z.; Yan, X.; Jin, Z. Design and Biomechanical Study of a Novel Adjustable Hemipelvic Prosthesis. Med. Eng. Phys. 2016, 38, 1416–1425. [Google Scholar] [CrossRef] [PubMed]
- Phillips, A.T.M.; Pankaj, P.; Howie, C.R.; Usmani, A.S.; Simpson, A.H.R.W. Finite Element Modelling of the Pelvis: Inclusion of Muscular and Ligamentous Boundary Conditions. Med. Eng. Phys. 2007, 29, 739–748. [Google Scholar] [CrossRef] [PubMed]
- Viceconti, M.; Olsen, S.; Nolte, L.P.; Burton, K. Extracting Clinically Relevant Data from Finite Element Simulations. Clin. Biomech. 2005, 20, 451–454. [Google Scholar] [CrossRef]
- Zhou, Y.; Min, L.; Liu, Y.; Shi, R.; Zhang, W.; Zhang, H.; Duan, H.; Tu, C. Finite Element Analysis of the Pelvis after Modular Hemipelvic Endoprosthesis Reconstruction. Int. Orthop. 2013, 37, 653–658. [Google Scholar] [CrossRef] [Green Version]
- Dalstra, M.; Huiskes, R.; Odgaard, A.; van Erning, L. Mechanical and Textural Properties of Pelvic Trabecular Bone. J. Biomech. 1993, 26, 523–535. [Google Scholar] [CrossRef] [Green Version]
- Kukin, I.A.; Kirpichev, I.V.; Maslov, L.B.; Vikhrev, S.V. Characteristics of the trabecular bone strength properties of people with hip diseases. Fundam. Res. 2013, 7, 328–333. [Google Scholar]
- Morgan, E.F.; Unnikrisnan, G.U.; Hussein, A.I. Bone Mechanical Properties in Healthy and Diseased States. Annu. Rev. Biomed. Eng. 2018, 20, 119–143. [Google Scholar] [CrossRef]
- Tikhilov, R.M.; Shubnyakov, I.I.; Mazurenko, A.V.; Mitryaykin, V.I.; Sachenkov, O.A.; Kuzin, A.K.; Denisov, A.O.; Pliev, D.G.; Boyarov, A.A.; Kovalenko, A.N. Experimental substantiation of acetabular component impaction with undercoverage in arthoplasty of patients with severe hip dysplasia. Travmatol. Ortop. Ross. 2013, 4, 42–51. [Google Scholar] [CrossRef] [Green Version]
- Reilly, D.T.; Burstein, A.H. The elastic and ultimate properties of compact bone tissue. J. Biomech. 1975, 8, 393–405. [Google Scholar] [CrossRef]
- Wirtz, D.C.; Schiffers, N.; Forst, R.; Pandorf, T.; Weichert, D.; Radermacher, K. Critical evaluation of known bone material properties to realize anisotropic FE-simulation of the proximal femur. J. Biomech. 2000, 33, 1325–1330. [Google Scholar] [CrossRef]
- Keaveny, T.M.; Wachtel, E.F.; Kopperdahl, D.L. Mechanical behavior of human trabecular bone after overloading. J. Orthop. Res. 1999, 17, 346–353. [Google Scholar] [CrossRef]
- Long, M.; Rack, H.J. Titanium alloys in total joint replacement—A materials science perspective. Biomaterials 1998, 19, 1621–1639. [Google Scholar] [CrossRef]
- Borovkov, A.; Maslov, L.; Tarasenko, F.; Zhmaylo, M.; Maslova, I.; Solovev, D. Development of elastic–plastic model of additively produced titanium for personalised endoprosthetics. Int. J. Adv. Manuf. Technol. 2021, 117, 2117–2132. [Google Scholar] [CrossRef]
- Greitemeier, D.; Palm, F.; Syassen, F.; Melz, T. Fatigue performance of additive manufactured TiAl6V4 using electron and laser beam melting. Int. J. Fatigue 2017, 94, 211–217. [Google Scholar] [CrossRef]
- MatWeb Material Property Data. Overview of Materials for High Density Polyethylene (HDPE), Injection Molded. Available online: http://www.matweb.com/search/datasheet.aspx?MatGUID=fce23f90005d4fbe8e12a1bce53ebdc8 (accessed on 12 September 2021).
- Bergmann, G.; Deuretzbacher, G. Hip contact forces and gait patterns from routine activities. J. Biomech. 2001, 34, 859–871. [Google Scholar] [CrossRef]
- Borovkov, A.; Maslov, L.; Zhmaylo, M.; Zelinskiy, I.; Voinov, I.; Keresten, I.; Mamchits, D.; Tikhilov, R.; Kovalenko, A.; Bilyk, S.; et al. Finite element stress analysis of a total hip replacement in a two-legged standing. Russ. J. Biomech. 2018, 4, 382–400. [Google Scholar] [CrossRef]
- Maslov, L.; Surkova, P.; Maslova, I.; Solovev, D.; Zhmaylo, M.; Kovalenko, A.; Bilyk, S. Finite-Element Study of the Customized Implant for Revision Hip Replacement. Vibroengineering Procedia 2019, 26, 40–45. [Google Scholar] [CrossRef]
- Dong, E.; Iqbal, T.; Fu, J.; Li, D.; Liu, B.; Guo, Z.; Cuadrado, A. Preclinical Strength Checking for Artificial Pelvic Prosthesis under Multi-activities—A Case Study. J. Bionic Eng. 2019, 16, 1092–1102. [Google Scholar] [CrossRef]
- Wong, K.C.; Kumta, S.M.; Gee, N.V.L.; Demol, J. One-Step Reconstruction with a 3D-Printed, Biomechanically Evaluated Custom Implant after Complex Pelvic Tumor Resection. Comput. Aided Surg. 2015, 20, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Akulich, Y.V.; Podgayets, R.M.; Scryabin, V.L.; Sotin, A.V. The Investigation of Stresses and Strains in the Hip Joint after Operation of Endoprosthetics. Russ. J. Biomech. 2007, 11, 9–35. [Google Scholar]
- Maslov, L.B. Biomechanical model and numerical analysis of tissue regeneration within a porous scaffold. Mech. Solids 2020, 55, 1115–1134. [Google Scholar] [CrossRef]
- Hao, Z.; Wan, C.; Gao, X.; Ji, T.; Wang, H. The Effect of Screw Fixation Type on a Modular Hemi-Pelvic Prosthesis: A 3-D Finite Element Model. Disabil. Rehabil. Assist. Technol. 2013, 8, 125–128. [Google Scholar] [CrossRef]
| Part | Number of Finite Elements | Finite Element Type | FEM Verification | ||
|---|---|---|---|---|---|
| Aspect | Skewness | Warping | |||
| Top of the damaged half of the pelvic bone | 77,392 | Four-node linear solid tetrahedral C3D4 type | No violations | 2892 elements off | No violations |
| Bottom of the damaged half of the pelvic bone | 122,131 | 3031 elements off | |||
| Healthy pelvic bone | 265,775 | 8807 elements off | |||
| Sacrum | 44,746 | 2009 elements off | |||
| Implant | 329,828 | 12,615 elements off | |||
| Material | Young’s Modulus, GPa | Poisson’s Ratio | Ultimate Stress, MPa | |
|---|---|---|---|---|
| Yield | Fatigue | |||
| Cortical tissue [9,18] | 17 | 0.3 | 80–150 [18] | the same as the yield stress |
| Spongy tissue [14,20] | 0.07 | 0.2 | 1.4–2.1 [20] | |
| Normal Ti-6Al-4V [21] | 113.8 | 0.34 | 950 | 310–610 [21] |
| 3D printed Ti-6Al-4V [22,23] | 123.4 | 0.26 | 910 | 200–500 [23] |
| Polyethylene [24] | 1 | 0.35 | 26 | - |
| Assembly Components | Maximum Von Mises Stresses (MPa) and Their Location | |||
|---|---|---|---|---|
| Pretension Force of 500 N | Pretension Force of 1000 N | |||
| Pretension Stage | Walking Cycle (45% Phase) | Pretension Stage | Walking Cycle (45% Phase) | |
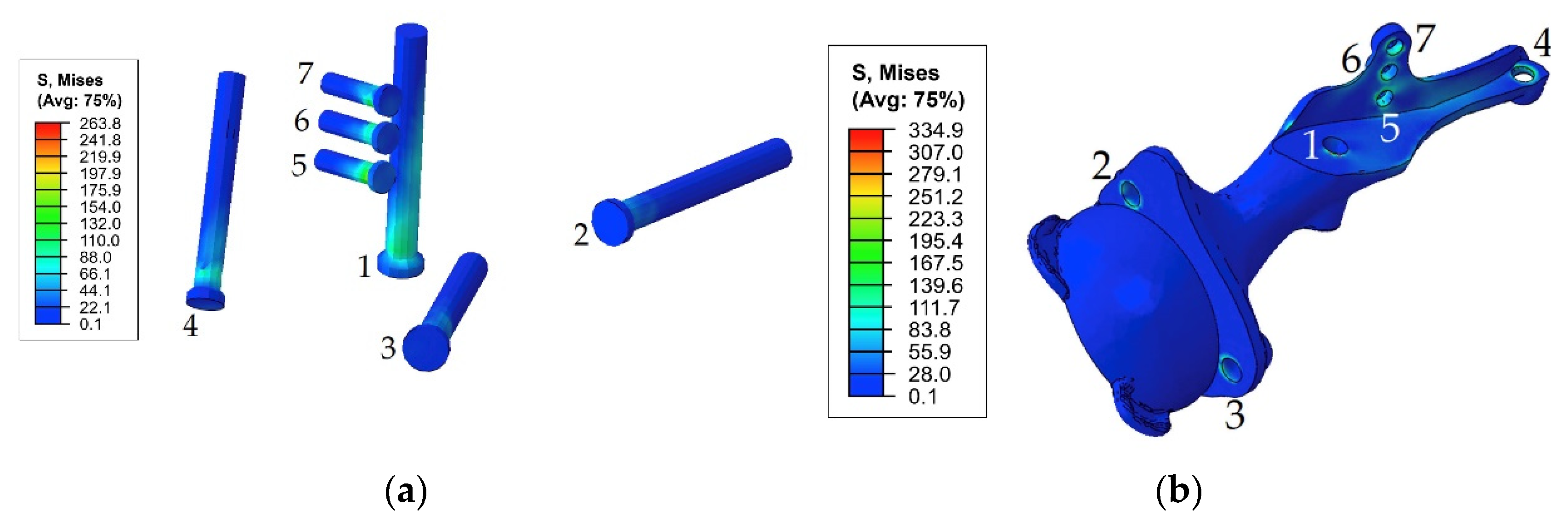
| Screw system | 122 MPa, screw 7 | 192 MPa, screw 7 | 212 MPa, screw 7 | 263 MPa, screw 7 |
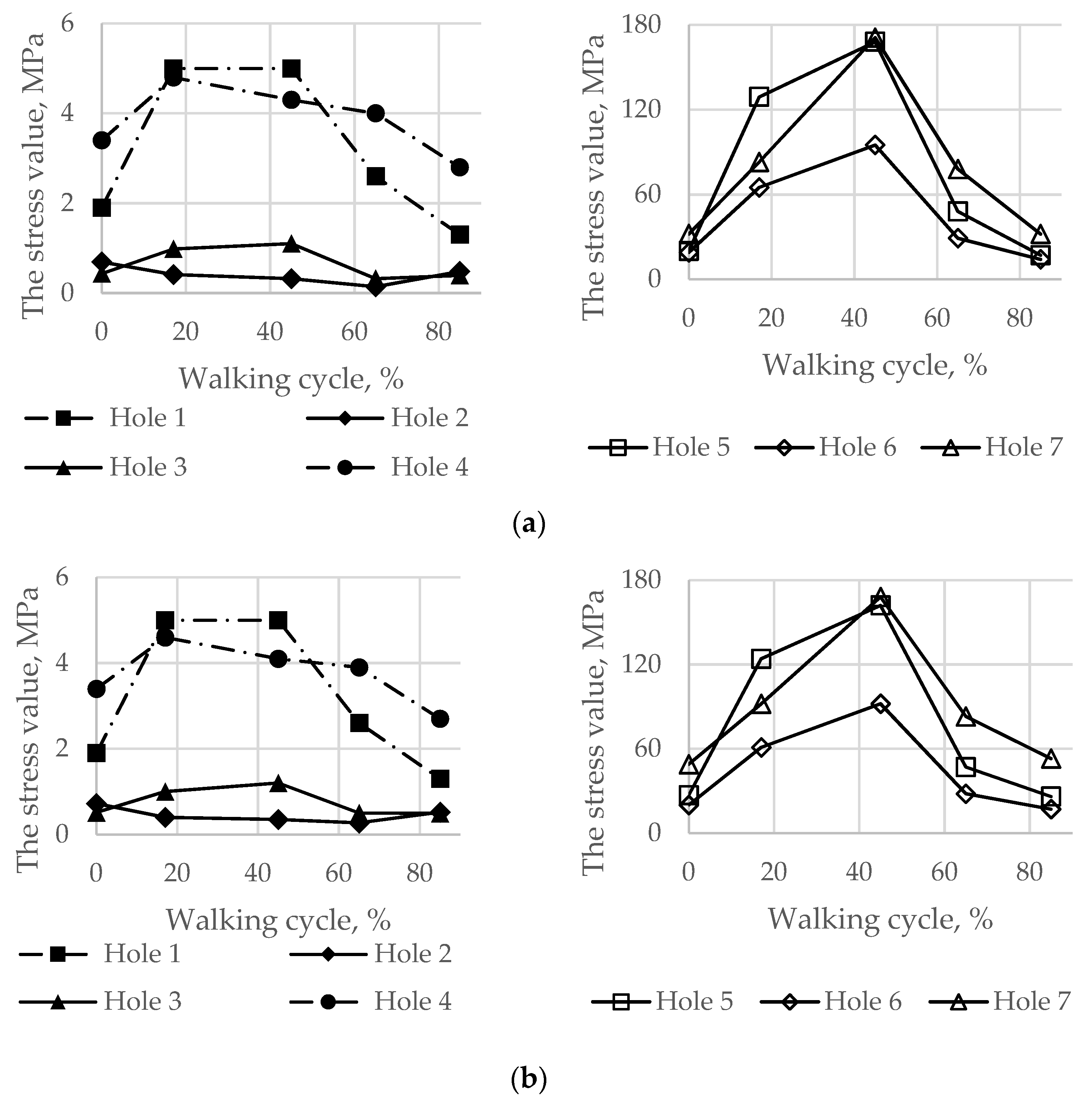
| Implant | 151 MPa, hole 1 | 197 MPa, hole 1 255 MPa, hole 1 (65% phase) | 253 MPa, hole 4 | 335 MPa, hole 1 |
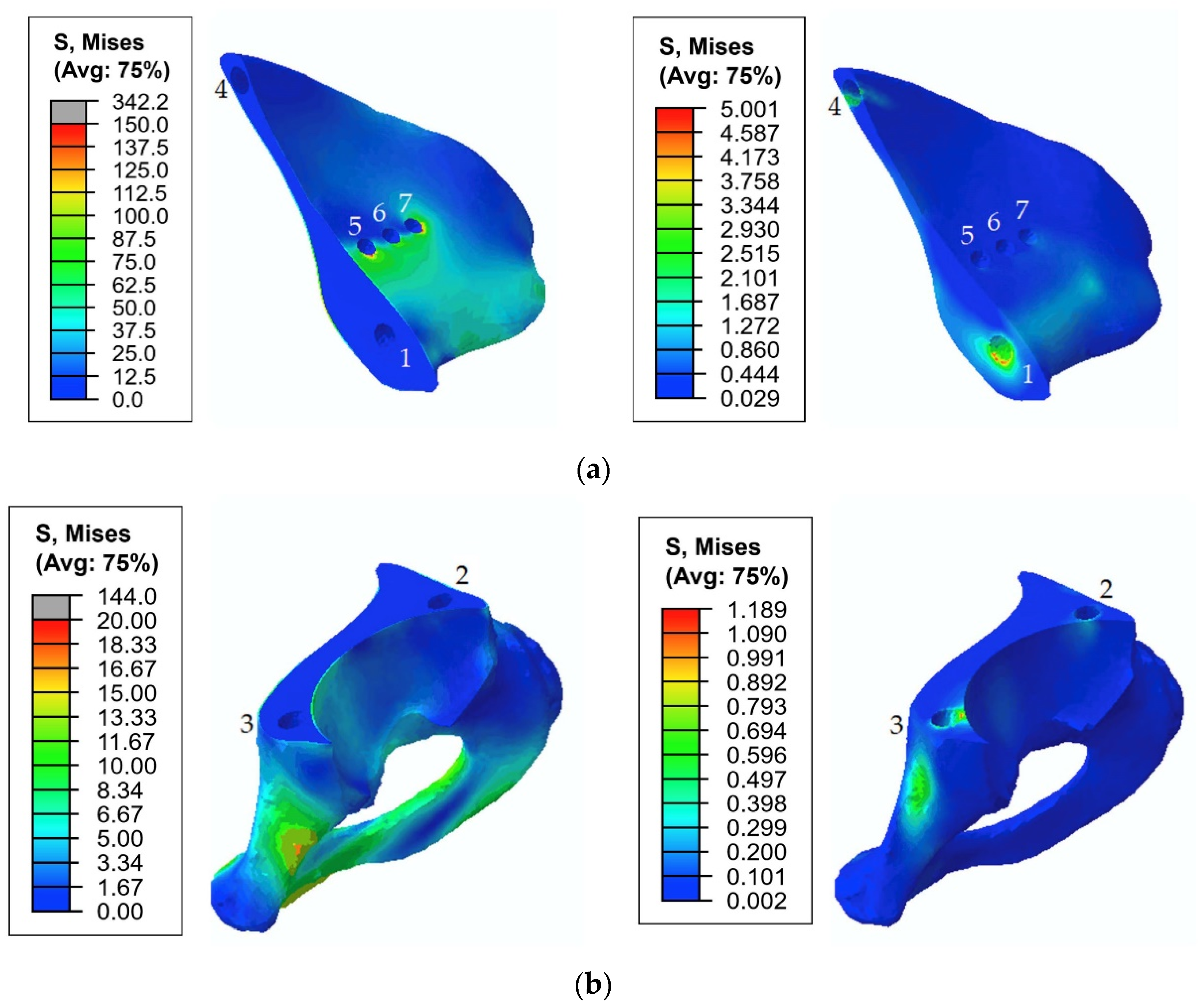
| Pelvic cortical tissue, top part of resected bone | 29 MPa, hole 7 | 171 MPa, hole 7 168 MPa, hole 5 | 48 MPa, hole 7 | 168 MPa, hole 7 162 MPa, hole 5 |
| Pelvic spongy tissue, top part of resected bone | 0.68 MPa, hole 4 | 4.98 MPa, hole 1 4.3 MPa, hole 4 | 1.25 MPa, hole 4 | 5.0 MPa, hole 1 4.1 MPa, hole 4 |
| Pelvic cortical tissue, bottom part of resected bone | 2.7 MPa | 19 MPa | 4.5 MPa | 19 MPa |
| Pelvic spongy tissue, bottom part of resected bone | 0.28 MPa, hole 3 | 1.1 MPa, hole 3 | 0.47 MPa, hole 3 | 1.2 MPa, hole 3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maslov, L.; Borovkov, A.; Maslova, I.; Soloviev, D.; Zhmaylo, M.; Tarasenko, F. Finite Element Analysis of Customized Acetabular Implant and Bone after Pelvic Tumour Resection throughout the Gait Cycle. Materials 2021, 14, 7066. https://doi.org/10.3390/ma14227066
Maslov L, Borovkov A, Maslova I, Soloviev D, Zhmaylo M, Tarasenko F. Finite Element Analysis of Customized Acetabular Implant and Bone after Pelvic Tumour Resection throughout the Gait Cycle. Materials. 2021; 14(22):7066. https://doi.org/10.3390/ma14227066
Chicago/Turabian StyleMaslov, Leonid, Alexey Borovkov, Irina Maslova, Dmitriy Soloviev, Mikhail Zhmaylo, and Fedor Tarasenko. 2021. "Finite Element Analysis of Customized Acetabular Implant and Bone after Pelvic Tumour Resection throughout the Gait Cycle" Materials 14, no. 22: 7066. https://doi.org/10.3390/ma14227066
APA StyleMaslov, L., Borovkov, A., Maslova, I., Soloviev, D., Zhmaylo, M., & Tarasenko, F. (2021). Finite Element Analysis of Customized Acetabular Implant and Bone after Pelvic Tumour Resection throughout the Gait Cycle. Materials, 14(22), 7066. https://doi.org/10.3390/ma14227066







