Active Double-Layered Films Enriched with AgNPs in Great Water Dock Root and Pu-Erh Extracts
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Plant Extracts
2.3. Silver Nanoparticle Synthesis (AgNPs)
2.4. Double-Layered Film Preparation
2.5. UV-Vis Spectroscopy Analysis
2.6. Particle Size and Zeta Potential
2.7. X-ray Diffraction Patterns (XRD)
2.8. Scanning Electron Microscopy (SEM)
2.9. Colour Parameters
2.10. Water Vapour Transmission Rate (WVTR)
2.11. Determination of Contact Angle
2.12. Thermal Properties
2.13. Antioxidant Activity Measured via FRAP Method
2.14. Antimicrobial Measurements
2.15. Statistical Analysis
3. Results and Discussion
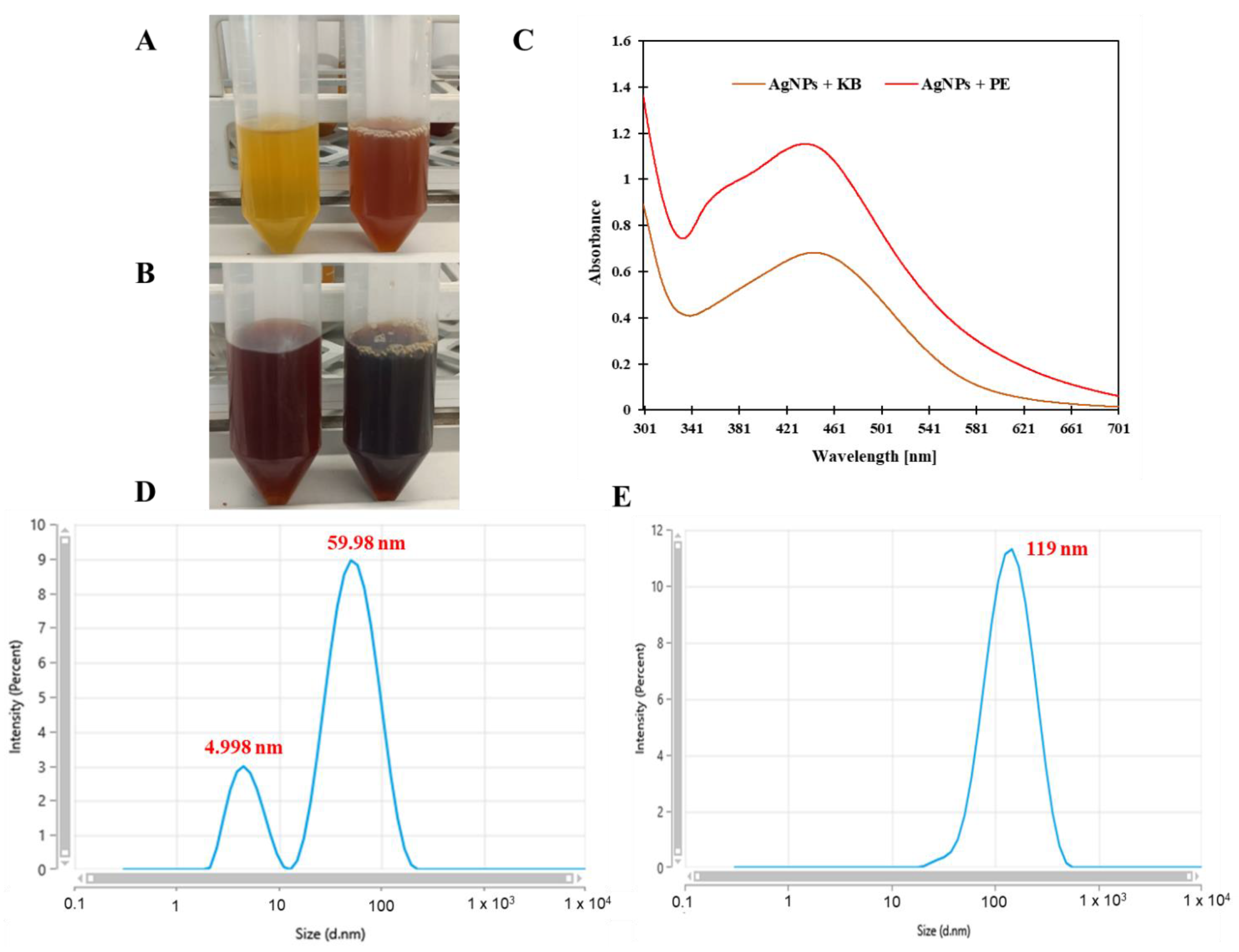
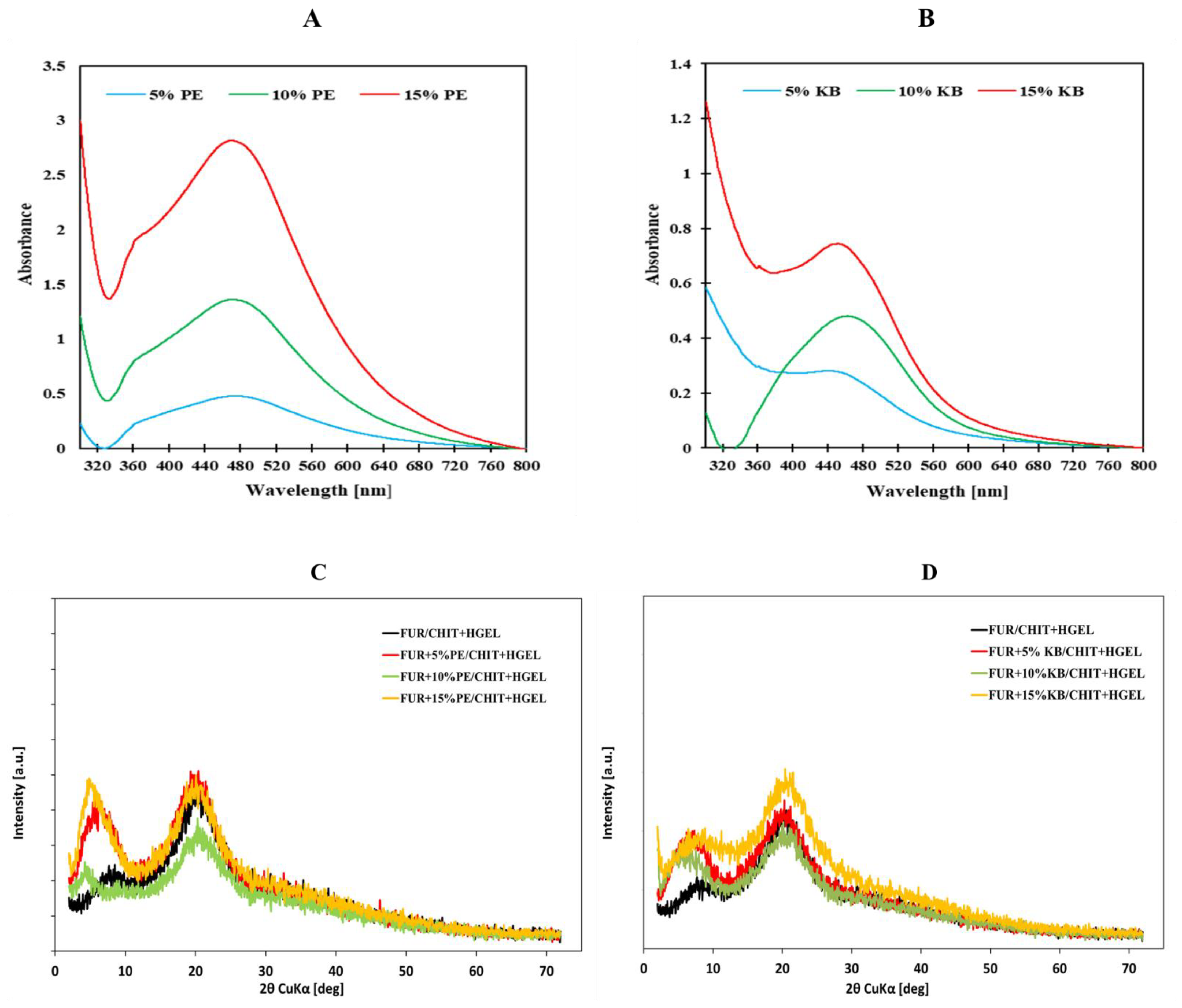
3.1. UV-Visible Spectral Study
3.2. Dynamic Light Scattering Analysis
3.3. Characterization of Double-Layered Films with AgNPs-Great Water Dock Root and AgNPs-Pu-Erh Extracts
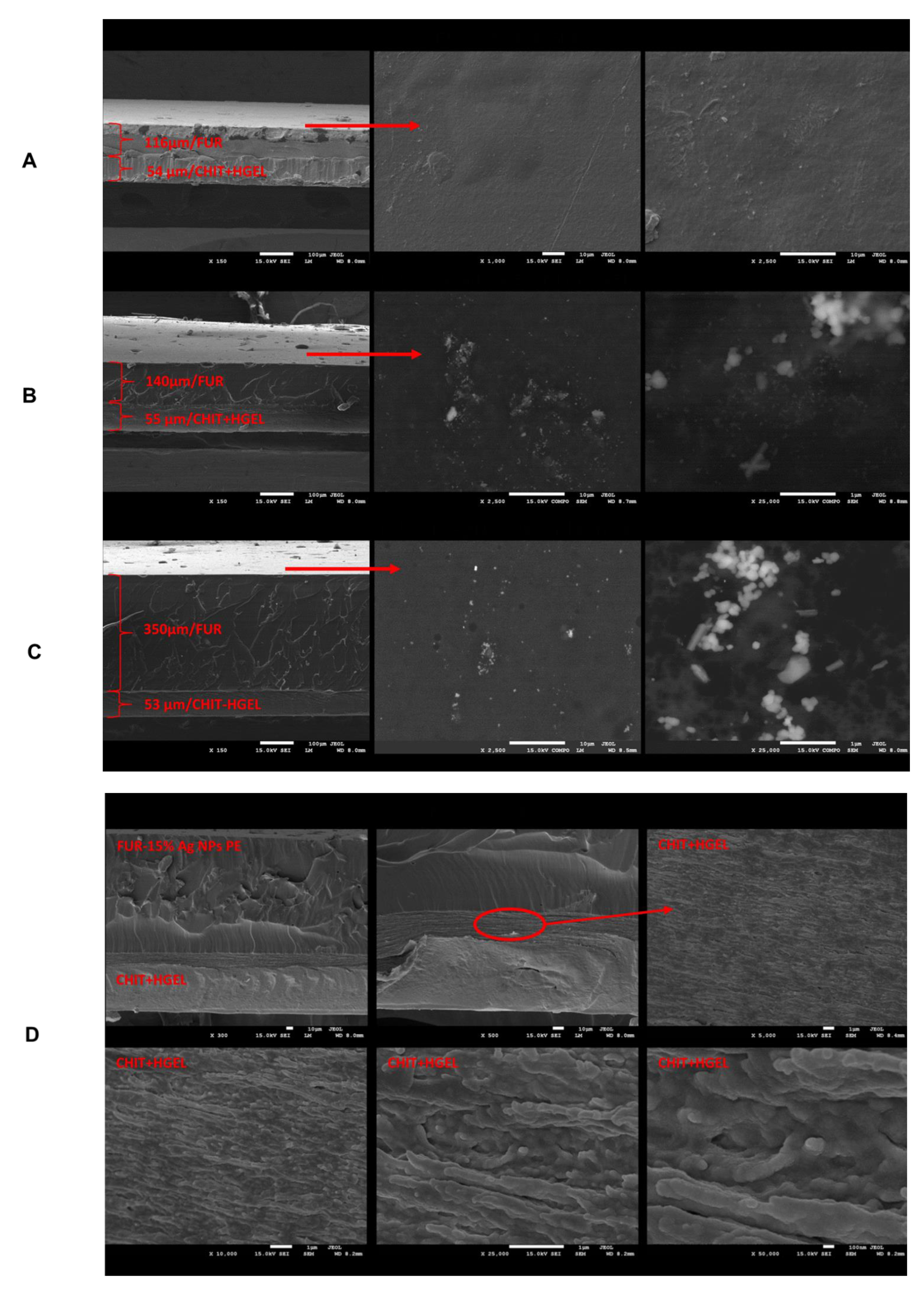
3.4. SEM Analysis
3.5. Physical Properties of Tested Films
3.6. Antioxidant Activity of the Tested Films
3.7. Antimicrobial Activity of the Tested Films
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- de Carvalho Bernardo, W.L.; Boriollo, M.F.G.; Tonon, C.C.; da Silva, J.J.; Cruz, F.M.; Martins, A.L.; Höfling, J.F.; Spolidorio, D.M.P. Antimicrobial effects of silver nanoparticles and extracts of Syzygium cumini flowers and seeds: Periodontal, cariogenic and opportunistic pathogens. Arch. Oral Biol. 2021, 125, 105101. [Google Scholar] [CrossRef]
- Wang, L.; Periyasami, G.; Aldalbahi, A.; Fogliano, V. The antimicrobial activity of silver nanoparticles biocomposite films depends on the silver ions release behaviour. Food Chem. 2021, 359, 129859. [Google Scholar] [CrossRef]
- Shah, Z.; Gul, T.; Ali Khan, S.; Shaheen, K.; Anwar, Y.; Suo, H.; Ismail, M.; Alghamdi, K.M.; Salman, S.M. Synthesis of high surface area AgNPs from Dodonaea viscosa plant for the removal of pathogenic microbes and persistent organic pollutants. Mater. Sci. Eng. B 2021, 263, 114770. [Google Scholar] [CrossRef]
- Rosman, N.S.R.; Harun, N.A.; Idris, I.; Ismail, W.I.W. Eco-friendly silver nanoparticles (AgNPs) fabricated by green synthesis using the crude extract of marine polychaete, Marphysa moribidii: Biosynthesis, characterisation, and antibacterial applications. Heliyon 2020, 6, e05462. [Google Scholar] [CrossRef]
- Mani, M.; Chang, J.H.; Dhanesh Gandhi, A.; Kayal Vizhi, D.; Pavithra, S.; Mohanraj, K.; Mohanbabu, B.; Babu, B.; Balachandran, S.; Kumaresan, S. Environmental and biomedical applications of AgNPs synthesized using the aqueous extract of Solanum surattense leaf. Inorg. Chem. Commun. 2020, 121, 108228. [Google Scholar] [CrossRef]
- Chandhirasekar, K.; Thendralmanikandan, A.; Thangavelu, P.; Nguyen, B.-S.; Nguyen, T.-A.; Sivashanmugan, K.; Nareshkumar, A.; Nguyen, V.-H. Plant-extract-assisted green synthesis and its larvicidal activities of silver nanoparticles using leaf extract of Citrus medica, Tagetes lemmonii, and Tarenna asiatica. Mater. Lett. 2021, 287, 129265. [Google Scholar] [CrossRef]
- Orbán-Gyapai, O.; Liktor-Busa, E.; Kúsz, N.; Stefkó, D.; Urbán, E.; Hohmann, J.; Vasas, A. Antibacterial screening of Rumex species native to the Carpathian Basin and bioactivity-guided isolation of compounds from Rumex aquaticus. Fitoterapia 2017, 118, 101–106. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, D.; Meng, J.; Xu, K.; Xiao, R.; Xu, M.; Liu, Y.; Zhao, Y.; Yao, P.; Yan, H.; Liu, L. Evaluation of oral subchronic toxicity of Pu-erh green tea (Camellia sinensis var. Assamica) extract in Sprague Dawley rats. J. Ethnopharmacol. 2012, 142, 836–844. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.-Y.; He, D.; Xing, Y.-F.; Zeng, W.; Ren, K.; Zhang, C.; Lu, Y.; Yang, S.; Ou, S.-J.; Wang, Y.; et al. Effects of bioactive components of Pu-erh tea on gut microbiomes and health: A review. Food Chem. 2021, 353, 129439. [Google Scholar] [CrossRef]
- Jamróz, E.; Kulawik, P.; Tkaczewska, J.; Guzik, P.; Zając, M.; Juszczak, L.; Krzyściak, P.; Turek, K. The effects of active double-layered furcellaran/gelatin hydrolysate film system with Ala-Tyr peptide on fresh Atlantic mackerel stored at −18 °C. Food Chem. 2021, 338, 127867. [Google Scholar] [CrossRef]
- Zhou, X.; Yu, X.; Xie, F.; Fan, Y.; Xu, X.; Qi, J.; Xiong, G.; Gao, X.; Zhang, F. pH-responsive double-layer indicator films based on konjac glucomannan/camellia oil and carrageenan/anthocyanin/curcumin for monitoring meat freshness. Food Hydrocoll. 2021, 106695. [Google Scholar] [CrossRef]
- Zhang, W.; Shu, C.; Chen, Q.; Cao, J.; Jiang, W. The multi-layer film system improved the release and retention properties of cinnamon essential oil and its application as coating in inhibition to penicillium expansion of apple fruit. Food Chem. 2019, 299, 125109. [Google Scholar] [CrossRef]
- Jamróz, E.; Tkaczewska, J.; Juszczak, L.; Zimowska, M.; Kawecka, A.; Krzyściak, P.; Skóra, M. The influence of lingonberry extract on the properties of novel, double-layered biopolymer films based on furcellaran, CMC and a gelatin hydrolysate. Food Hydrocoll. 2021, 107334. [Google Scholar] [CrossRef]
- Dai, L.; Long, Z.; Chen, J.; An, X.; Cheng, D.; Khan, A.; Ni, Y. Robust Guar Gum/Cellulose Nanofibrils Multilayer Films with Good Barrier Properties. ACS Appl. Mater. Interfaces 2017, 9, 5477–5485. [Google Scholar] [CrossRef] [PubMed]
- Marangoni, L.; Vieira, R.P.; Jamróz, E.; Anjos, C.A.R. Furcellaran: An innovative biopolymer in the production of films and coatings. Carbohydr. Polym. 2020, 117221. [Google Scholar] [CrossRef] [PubMed]
- Haghighi, H.; Licciardello, F.; Fava, P.; Siesler, H.W.; Pulvirenti, A. Recent advances on chitosan-based films for sustainable food packaging applications. Food Packag. Shelf Life 2020, 26, 100551. [Google Scholar] [CrossRef]
- Tkaczewska, J.; Kulawik, P.; Jamróz, E.; Guzik, P.; Zając, M.; Szymkowiak, A.; Turek, K. One- and double-layered furcellaran/carp skin gelatin hydrolysate film system with antioxidant peptide as an innovative packaging for perishable foods products. Food Chem. 2021, 351, 129347. [Google Scholar] [CrossRef]
- Tkaczewska, J.; Jamróz, E.; Kulawik, P.; Morawska, M.; Szczurowska, K. Evaluation of the potential use of a carp (Cyprinus carpio) skin gelatine hydrolysate as an antioxidant component. Food Funct. 2019, 10, 1038–1048. [Google Scholar] [CrossRef] [PubMed]
- Basumatary, K.; Daimary, P.; Das, S.K.; Thapa, M.; Singh, M.; Mukherjee, A.; Kumar, S. Lagerstroemia speciosa fruit-mediated synthesis of silver nanoparticles and its application as filler in agar based nanocomposite films for antimicrobial food packaging. Food Packag. Shelf Life 2018, 17, 99–106. [Google Scholar] [CrossRef]
- Pluta-Kubica, A.; Jamróz, E.; Kawecka, A.; Juszczak, L.; Krzyściak, P. Active edible furcellaran/whey protein films with yerba mate and white tea extracts: Preparation, characterization and its application to fresh soft rennet-curd cheese. Int. J. Biol. Macromol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Ghoshal, G.; Goyal, M. Development and characterization of corn starch based nanocomposite film with AgNPs and plant extract. Mater. Sci. Energy Technol. 2020, 3, 672–678. [Google Scholar] [CrossRef]
- Palem, R.R.; Ganesh, S.D.; Kronekova, Z.; Sláviková, M.; Saha, N.; Saha, P. Green synthesis of silver nanoparticles and biopolymer nanocomposites: A comparative study on physico-chemical, antimicrobial and anticancer activity. Bull. Mater. Sci. 2018, 41, 55. [Google Scholar] [CrossRef] [Green Version]
- Li, W.; Qu, F.; Chen, Y.; Sun, Y.; Zhang, J.; Xie, G.; You, Q.; Xu, H. Antimicrobial activity of sliver nanoparticles synthesized by the leaf extract of Cinnamomum camphora. Biochem. Eng. J. 2021, 172, 108050. [Google Scholar] [CrossRef]
- Jamróz, E.; Khachatryan, G.; Kopel, P.; Juszczak, L.; Kawecka, A.; Krzyściak, P.; Kucharek, M.; Bębenek, Z.; Zimowska, M. Furcellaran nanocomposite films: The effect of nanofillers on the structural, thermal, mechanical and antimicrobial properties of biopolymer films. Carbohydr. Polym. 2020, 116244. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Sanpui, P.; Chattopadhyay, A.; Ghosh, S.S. Fabrication of antibacterial silver nanoparticle—sodium alginate–chitosan composite films. Rsc Adv. 2012, 2, 5837–5843. [Google Scholar] [CrossRef]
- Kabekkodu, S.E. ICDD PDF-4+ 2015 00-067-1540; International Centre for Diffraction Data: Newtown Square, PA, USA, 2015. [Google Scholar]
- Li, X.; Xie, H.; Lin, J.; Xie, W.; Ma, X. Characterization and biodegradation of chitosan–alginate polyelectrolyte complexes. Polym. Degrad. Stab. 2009, 94, 1–6. [Google Scholar] [CrossRef]
- Yoksan, R.; Chirachanchai, S. Silver nanoparticle-loaded chitosan–starch based films: Fabrication and evaluation of tensile, barrier and antimicrobial properties. Mater. Sci. Eng. C 2010, 30, 891–897. [Google Scholar] [CrossRef]
- Das, B.; Dash, S.K.; Mandal, D.; Ghosh, T.; Chattopadhyay, S.; Tripathy, S.; Das, S.; Dey, S.K.; Das, D.; Roy, S. Green synthesized silver nanoparticles destroy multidrug resistant bacteria via reactive oxygen species mediated membrane damage. Arab. J. Chem. 2017, 10, 862–876. [Google Scholar] [CrossRef] [Green Version]
- Dara, P.K.; Mahadevan, R.; Digita, P.A.; Visnuvinayagam, S.; Kumar, L.R.G.; Mathew, S.; Ravishankar, C.N.; Anandan, R. Synthesis and biochemical characterization of silver nanoparticles grafted chitosan (Chi-Ag-NPs): In vitro studies on antioxidant and antibacterial applications. SN Appl. Sci. 2020, 2, 665. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.H.; Jeong, D.; Kanmani, P. Study on physical and mechanical properties of the biopolymer/silver based active nanocomposite films with antimicrobial activity. Carbohydr. Polym. 2019, 115159. [Google Scholar] [CrossRef]
- Kanmani, P.; Rhim, J.-W. Physical, mechanical and antimicrobial properties of gelatin based active nanocomposite films containing AgNPs and nanoclay. Food Hydrocoll. 2014, 35, 644–652. [Google Scholar] [CrossRef]
- Gudimalla, A.; Jose, J.; Varghese, R.J.; Thomas, S. Green Synthesis of Silver Nanoparticles Using Nymphae odorata Extract Incorporated Films and Antimicrobial Activity. J. Polym. Environ. 2021, 29, 1412–1423. [Google Scholar] [CrossRef]
- Shankar, S.; Tanomrod, N.; Rawdkuen, S.; Rhim, J.-W. Preparation of pectin/silver nanoparticles composite films with UV-light barrier and properties. Int. J. Biol. Macromol. 2016, 92, 842–849. [Google Scholar] [CrossRef]
- Tkaczewska, J.; Borawska-Dziadkiewicz, J.; Kulawik, P.; Duda, I.; Morawska, M.; Mickowska, B. The effects of hydrolysis condition on the antioxidant activity of protein hydrolysate from Cyprinus carpio skin gelatin. LWT 2020, 117, 108616. [Google Scholar] [CrossRef]
- Tkaczewska, J.; Bukowski, M.; Mak, P. Identification of antioxidant peptides in enzymatic hydrolysates of carp (Cyprinus carpio) skin gelatin. Molecules 2019, 24, 97. [Google Scholar] [CrossRef] [Green Version]
- Nunes, M.R.; de Souza Maguerroski Castilho, M.; de Lima Veeck, A.P.; da Rosa, C.G.; Noronha, C.M.; Maciel, M.V.O.B.; Barreto, P.M. Antioxidant and antimicrobial methylcellulose films containing Lippia alba extract and silver nanoparticles. Carbohydr. Polym. 2018, 192, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Bedlovičová, Z.; Strapáč, I.; Baláž, M.; Salayová, A. A brief overview on antioxidant activity determination of silver nanoparticles. Molecules 2020, 25, 3191. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, L.; do Carmo, M.A.V.; Wu, Y.; Esmerino, L.A.; Azevedo, L.; Zhang, L.; Granato, D. Optimizing the extraction of bioactive compounds from pu-erh tea (Camellia sinensis var. assamica) and evaluation of antioxidant, cytotoxic, antimicrobial, antihemolytic, and inhibition of α-amylase and α-glucosidase activities. Food Res. Int. 2020, 137, 109430. [Google Scholar] [CrossRef]
- Ropiak, H.M.; Ramsay, A.; Mueller-Harvey, I. Condensed tannins in extracts from European medicinal plants and herbal products. J. Pharm. Biomed. Anal. 2016, 121, 225–231. [Google Scholar] [CrossRef]
- Pereira de Abreu, D.A.; Paseiro Losada, P.; Maroto, J.; Cruz, J.M. Natural antioxidant active packaging film and its effect on lipid damage in frozen blue shark (Prionace glauca). Innov. Food Sci. Emerg. Technol. 2011, 12, 50–55. [Google Scholar] [CrossRef]
- Ortega, F.; Arce, V.B.; Garcia, M.A. Nanocomposite starch-based films containing silver nanoparticles synthesized with lemon juice as reducing and stabilizing agent. Carbohydr. Polym. 2021, 252, 117208. [Google Scholar] [CrossRef]
- Mathew, S.; Jayakumar, A.; Kumar, V.P.; Mathew, J.; Radhakrishnan, E.K. One-step synthesis of eco-friendly boiled rice starch blended polyvinyl alcohol bionanocomposite films decorated with in situ generated silver nanoparticles for food packaging purpose. Int. J. Biol. Macromol. 2019, 139, 475–485. [Google Scholar] [CrossRef]
- Feng, Q.L.; Wu, J.; Chen, G.Q.; Cui, F.; Kim, T.; Kim, J. A mechanistic study of the antibacterial effect of silver ions on Escherichia coli and Staphylococcus aureus. J. Biomed. Mater. Res. 2000, 52, 662–668. [Google Scholar] [CrossRef]
- Kim, J.S.; Kuk, E.; Yu, K.N.; Kim, J.-H.; Park, S.J.; Lee, H.J.; Kim, S.H.; Park, Y.K.; Park, Y.H.; Hwang, C.-Y. Antimicrobial effects of silver nanoparticles. Nanomed. Nanotechnol. Biol. Med. 2007, 3, 95–101. [Google Scholar] [CrossRef]
- Shankar, S.; Rhim, J.-W. Amino acid mediated synthesis of silver nanoparticles and preparation of antimicrobial agar/silver nanoparticles composite films. Carbohydr. Polym. 2015, 130, 353–363. [Google Scholar] [CrossRef] [PubMed]
- Priyadarshini, S.; Gopinath, V.; Priyadharsshini, N.M.; MubarakAli, D.; Velusamy, P. Synthesis of anisotropic silver nanoparticles using novel strain, Bacillus flexus and its biomedical application. Colloids Surf. B Biointerfaces 2013, 102, 232–237. [Google Scholar] [CrossRef] [PubMed]

| Control | 5% KB | 10% KB | 15% KB | 5% PE | 10% PE | 15% PE | |
|---|---|---|---|---|---|---|---|
| COLOUR PROPERTIES | |||||||
| L* | 89.78 g ± 0.23 | 77.76 f ± 0.52 | 66.13 e ± 0.34 | 61.89 d ± 0.86 | 55.06 c ± 0.42 | 41.00 b ± 0.76 | 35.52 a ± 0.24 |
| a* | −1.87 a ± 0.04 | 9.17 b ± 0.23 | 24.37 e ± 0.38 | 26.22 f ± 0.54 | 20.51 d ± 0.10 | 20.38 d ± 0.93 | 14.94 c ± 0.33 |
| b* | 23.86 c ± 0.94 | 45.86 e ± 1.51 | 55.07 f ± 0.52 | 55.12 f ± 0.66 | 33.84 d ± 0.53 | 21.22 b ± 1.22 | 12.93 a ± 0.34 |
| ΔE | - | 27.40 | 46.30 | 49.63 | 23.07 | 39.91 | 50.67 |
| Appearance |  |  |  |  |  |  |  |
| THERMAL PROPERTIES | |||||||
| Peak temperature (Tm) (°C) | 182.8 ab ± 2.3 | 178.2 a ± 4.8 | 189.3 bc ± 8.7 | 191.4 c ± 2.4 | 198.4 d ± 1.7 | 202.8 d ± 1.0 | 205.5 d ± 0.7 |
| Enthalpy (ΔHm) (J/g) | 235.3 ab ± 1.5 | 249.1 b ± 3.9 | 237.6 ab ± 6.4 | 249.5 b ± 15.1 | 227.3 a ± 8.4 | 237.0 ab ± 15.7 | 227.5 a ± 5.0 |
| WATER PROPERTIES | |||||||
| WVTR [g/m2× d] | 840.59 a ± 48.07 | 886.9 b ± 24.63 | 894.77 b ± 13.29 | 921.66 bc ± 14.94 | 941.41 c ± 14.04 | 967.87 c ± 18.53 | 944.42 c ± 46.29 |
| WCA [°] | CHIT+HGEL | CHIT+HGEL | CHIT+HGEL | CHIT+HGEL | CHIT+HGEL | CHIT+HGEL | CHIT+HGEL |
| 91.26 f ± 1.44 | 88.96 e ± 0.38 | 88.27 de ± 0.64 | 88.08 cd ± 0.43 | 87.46 c ± 0.77 | 85.70 b ± 1.09 | 81.08 a ± 1.29 | |
 |  |  |  |  |  |  | |
| FUR | FUR + 5% KB | FUR + 10% KB | FUR + 15% KB | FUR + 5% PE | FUR + 10% PE | FUR + 15% PE | |
| 86.34 a ± 0.23 | 86.32 a ± 1.11 | 87.57 b ± 1.16 | 88.98 b ± 0.39 | 91.86 c ± 0.59 | 94.11 d ± 0.60 | 94.68 d ± 0.70 | |
 |  |  |  |  |  |  | |
| ANTIOXIDANT ACTIVITY | |||||||
| FRAP iron ion reduction capacity (mMTrolox/mg) | 1.55 a ± 0.29 | 2.37 b ± 0.22 | 5.71 d ± 0.34 | 8.32 f ± 0.78 | 2.86 b ± 0.44 | 3.92 c ± 0.18 | 6.87 e ± 0.68 |
| 10% PE | 15% PE | 10% KB | 15% KB | |
|---|---|---|---|---|
| Staphylococcus aureus |  |  |  |  |
| very good effect with broad inhibition zone ZOI *-20 mm | very good effect with broad inhibition zone ZOI-16 mm | very good effect with broad inhibition zone ZOI-17 mm | very good effect with broad inhibition zone ZOI-17 mm | |
| Escherichia coli |  |  |  |  |
| very good effect with colony growth inside inhibition zone ZOI-17 mm (13 mm internal zone) | very good effect with colony growth inside inhibition zone ZOI-16 mm | very good effect with colony growth inside inhibition zone ZOI-16 mm | very good effect with colony growth inside inhibition zone ZOI-16 mm | |
| Salmonella enterica |  |  |  |  |
| very good effect with colony growth inside inhibition zone ZOI-17 mm (13 mm internal zone) | very good effect with colony growth inside inhibition zone ZOI-16 mm (13 mm internal zone) | very good effect with colony growth inside inhibition zone ZOI-17 mm | very good effect with colony growth inside inhibition zone ZOI-17 mm (14 mm internal) | |
| Enterococcus faecalis |  |  |  |  |
| very good effect with broad inhibition zone ZOI-16 mm | very good effect with narrow inhibition zone ZOI-14 mm | very good effect with narrow inhibition zone ZOI-13 mm | very good effect with narrow inhibition zone ZOI-13 mm | |
| Pseudomonas aeruginosa |  |  |  |  |
| very good effect with broad inhibition zone ZOI-21 mm | very good effect with broad inhibition zone ZOI-19 mm | very good effect with broad inhibition zone ZOI-20 mm | very good effect with broad inhibition zone ZOI-21 mm | |
| Candida krusei |  |  |  |  |
| very good effect with broad inhibition zone ZOI-17 mm | very good effect with narrow inhibition zone ZOI-11 mm | very good effect with narrow inhibition zone ZOI-12 mm | very good effect with broad inhibition zone ZOI-15 mm |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jamróz, E.; Cabaj, A.; Juszczak, L.; Tkaczewska, J.; Zimowska, M.; Cholewa-Wójcik, A.; Krzyściak, P.; Kopel, P. Active Double-Layered Films Enriched with AgNPs in Great Water Dock Root and Pu-Erh Extracts. Materials 2021, 14, 6925. https://doi.org/10.3390/ma14226925
Jamróz E, Cabaj A, Juszczak L, Tkaczewska J, Zimowska M, Cholewa-Wójcik A, Krzyściak P, Kopel P. Active Double-Layered Films Enriched with AgNPs in Great Water Dock Root and Pu-Erh Extracts. Materials. 2021; 14(22):6925. https://doi.org/10.3390/ma14226925
Chicago/Turabian StyleJamróz, Ewelina, Agnieszka Cabaj, Lesław Juszczak, Joanna Tkaczewska, Małgorzata Zimowska, Agnieszka Cholewa-Wójcik, Paweł Krzyściak, and Pavel Kopel. 2021. "Active Double-Layered Films Enriched with AgNPs in Great Water Dock Root and Pu-Erh Extracts" Materials 14, no. 22: 6925. https://doi.org/10.3390/ma14226925
APA StyleJamróz, E., Cabaj, A., Juszczak, L., Tkaczewska, J., Zimowska, M., Cholewa-Wójcik, A., Krzyściak, P., & Kopel, P. (2021). Active Double-Layered Films Enriched with AgNPs in Great Water Dock Root and Pu-Erh Extracts. Materials, 14(22), 6925. https://doi.org/10.3390/ma14226925








