Azaacenes Based Electroactive Materials: Preparation, Structure, Electrochemistry, Spectroscopy and Applications—A Critical Review †
Abstract
:1. Acenes vs. Azaacenes—Generalities
2. Synthesis of Azaacenes
3. Spectroscopic and Electrochemical Properties of Linear Azaacenes
4. Supramolecular Organization of Linear Azaacenes
5. Degradation of Linear Azaacenes
6. Azaacenes Containing Pyrene-Type Units
7. Spatially Extended Azaacenes
8. Nonlinear Azaacenes
9. Applications of Azaacenes
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bunz, U.H.F. The larger linear N-heteroacenes. Acc. Chem. Res. 2015, 48, 1676–1686. [Google Scholar] [CrossRef] [PubMed]
- Pron, A.; Gawrys, P.; Zagorska, M.; Djurado, D.; Demadrille, R. Electroactive materials for organic electronics: Preparation strategies, structural aspects and characterization techniques. Chem. Soc. Rev. 2010, 39, 2577–2632. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Li, B.; Cai, X.; Li, M.; Qiao, Z.; Tang, X.; Liu, K.; Gu, C.; Ma, Y.; Su, S.-J. Thiophene disubstituted benzothiadiazole derivatives: An effective planarization strategy toward deep-red to near-infrared (NIR) organic light-emitting diodes. Front. Chem. 2019, 7, 276. [Google Scholar] [CrossRef] [PubMed]
- Ahrens, L.; Hahn, S.; Rominger, F.; Freudenberg, J.; Bunz, U.H.F. N-Acenoacenes. Chem. Eur. J. 2019, 25, 14522–14526. [Google Scholar] [CrossRef]
- Takeya, J.; Yamagishi, M.; Tominari, Y.; Hirahara, R.; Nakazawa, Y.; Nishikawa, T.; Kawase, T.; Shimoda, T.; Ogawa, S. Very high-mobility organic single-crystal transistors with in-crystal conduction channels. Appl. Phys. Lett. 2007, 90, 102120. [Google Scholar] [CrossRef]
- Ono, K.; Totani, H.; Hiei, T.; Yoshino, A.; Saito, K.; Eguchi, K.; Tomura, M.; Nishida, J.; Yamashita, Y. Photooxidation and reproduction of pentacene derivatives substituted by aromatic groups. Tetrahedron 2007, 63, 9699–9704. [Google Scholar] [CrossRef]
- Malloci, G.; Mulas, G.; Cappellini, G.; Joblin, C. Time-dependent density functional study of the electronic spectra of oligoacenes in the charge states −1, 0, +1, and +2. Chem. Phys. 2007, 340, 43–58. [Google Scholar] [CrossRef] [Green Version]
- Hajgató, B.; Deleuze, M.S.; Tozer, D.J.; De Proft, F. A Benchmark theoretical study of the electron affinities of benzene and linear acenes. J. Chem. Phys. 2008, 129, 084308. [Google Scholar] [CrossRef]
- Winkler, M.; Houk, K.N. Nitrogen-rich oligoacenes: Candidates for n-channel organic semiconductors. J. Am. Chem. Soc. 2007, 129, 1805–1815. [Google Scholar] [CrossRef]
- Bunz, U.H.F.; Engelhart, J.U.; Lindner, B.D.; Schaffroth, M. Large N-heteroacenes: New tricks for very old dogs? Angew. Chem. Int. Ed. 2013, 52, 3810–3821. [Google Scholar] [CrossRef]
- Tang, X.-D. Theoretical study on electron structure and charge transport properties of tetraazapentacene derivatives. J. Mol. Graph. Model. 2017, 76, 535–542. [Google Scholar] [CrossRef]
- Gao, J.; Zhang, Q. Understanding the electronic structure of larger azaacenes through DFT calculations. Isr. J. Chem. 2014, 54, 699–702. [Google Scholar] [CrossRef]
- Parker, V.D. Energetics of electrode reactions. II. The relationship between redox potentials, ionization potentials, electron affinities, and solvation energies of aromatic hydrocarbons. J. Am. Chem. Soc. 1976, 98, 98–103. [Google Scholar] [CrossRef]
- Costa, J.C.S.; Taveira, R.J.S.; Lima, C.F.R.A.C.; Mendes, A.; Santos, L.M.N.B.F. Optical band gaps of organic semiconductor materials. Opt. Mater. 2016, 58, 51–60. [Google Scholar] [CrossRef]
- Bard, A.J. Encyclopedia of Electrochemistry of the Elements; Lund, H., Ed.; Taylor & Francis: New York, NY, USA, 1984; Volume 15, pp. 168–280. ISBN1 0824725158. ISBN2 9780824725150. [Google Scholar]
- Snyder, R.; Testa, A.C. Influence of electron-donor-acceptor complexation on electronic relaxation of quinoline. J. Phys. Chem. 1984, 88, 5948–5950. [Google Scholar] [CrossRef]
- Yang, S.; Liu, D.; Xu, X.; Miao, Q. Molecular packing and N-channel thin film transistors of chlorinated cyclobuta[1,2-b:3,4-B′] diquinoxalines. Chem. Commun. 2015, 51, 4275–4278. [Google Scholar] [CrossRef]
- Zhou, C.; Zhang, T.; Zhang, S.; Liu, H.; Gao, Y.; Su, Q.; Wu, Q.; Li, W.; Chen, J.; Yang, B. Isomerization effect of triphenylamine-acridine derivatives on excited-state modification, photophysical property and electroluminescence performance. Dyes Pigm. 2017, 146, 558–566. [Google Scholar] [CrossRef]
- Plasseraud, L.; Cattey, H.; Richard, P.; Ballivet-Tkatchenko, D. A novel two-dimensional organostannoxane coordination network promoted by phenazine: Synthesis, characterization and X-ray structure of 2∞{[n-Bu2(μ-OH)SnOSn(μ-H2-O3SCF3)n-Bu2]2[n-Bu2(H1-O3SCF3)SnOSn(μ-OH)n-Bu2]2}. J. Organomet. Chem. 2009, 694, 2386–2394. [Google Scholar] [CrossRef]
- Kobayashi, T.; Kobayashi, S. Cyclovoltammetric investigation into the homoconjugation of plural pyrazine rings linked by bicyclo[2.2.2]octadiene spacers. Eur. J. Org. Chem. 2002, 2002, 2066–2073. [Google Scholar] [CrossRef]
- Herrera, H.; de Echegaray, P.; Urdanpilleta, M.; Mancheño, M.J.; Mena-Osteritz, E.; Bäuerle, P.; Segura, J.L. Linear and star-shaped naphthalimide-fused pyrazinacenes. Chem. Commun. 2012, 49, 713–715. [Google Scholar] [CrossRef] [PubMed]
- Sworakowski, J.; Lipiński, J.; Janus, K. On the reliability of determination of energies of HOMO and LUMO levels in organic semiconductors from electrochemical measurements. A simple picture based on the electrostatic model. Org. Electron. 2016, 33, 300–310. [Google Scholar] [CrossRef]
- Krygowski, T.M.; Szatylowicz, H.; Stasyuk, O.A.; Dominikowska, J.; Palusiak, M. Aromaticity from the viewpoint of molecular geometry: Application to planar systems. Chem. Rev. 2014, 114, 6383–6422. [Google Scholar] [CrossRef]
- Chen, Z.; Wannere, C.S.; Corminboeuf, C.; Puchta, R.; von Schleyer, P.R. Nucleus-independent chemical shifts (NICS) as an aromaticity criterion. Chem. Rev. 2005, 105, 3842–3888. [Google Scholar] [CrossRef] [PubMed]
- Cardona, C.M.; Li, W.; Kaifer, A.E.; Stockdale, D.; Bazan, G.C. Electrochemical considerations for determining absolute frontier orbital energy levels of conjugated polymers for solar cell applications. Adv. Mater. 2011, 23, 2367–2371. [Google Scholar] [CrossRef] [PubMed]
- Sworakowski, J.; Janus, K. On the reliability of determination of energies of HOMO levels in organic semiconducting polymers from electrochemical measurements. Org. Electron. 2017, 48, 46–52. [Google Scholar] [CrossRef]
- Sworakowski, J. How accurate are energies of HOMO and LUMO levels in small-molecule organic semiconductors determined from cyclic voltammetry or optical spectroscopy? Synth. Met. 2018, 235, 125–130. [Google Scholar] [CrossRef]
- Miao, Q. Ten years of N-heteropentacenes as semiconductors for organic thin-film transistors. Adv. Mater. 2014, 26, 5541–5549. [Google Scholar] [CrossRef]
- Li, J.; Zhang, Q. Linearly fused azaacenes: Novel approaches and new applications beyond field-effect transistors (FETs). ACS Appl. Mater. Interfaces 2015, 7, 28049–28062. [Google Scholar] [CrossRef]
- Anthony, J.E. The larger acenes: Versatile organic semiconductors. Angew. Chem. Int. Ed. 2008, 47, 452–483. [Google Scholar] [CrossRef]
- Hayashi, H.; Kato, Y.; Matsumoto, A.; Shikita, S.; Aizawa, N.; Suzuki, M.; Aratani, N.; Yasuda, T.; Yamada, H. Synthesis of anthracene derivatives with azaacene-containing iptycene wings and the utilization as a dopant for solution-processed organic light-emitting diodes. Chem. Eur. J. 2019, 25, 15565–15571. [Google Scholar] [CrossRef]
- Sych, G.; Volyniuk, D.; Bezvikonnyi, O.; Lytvyn, R.; Grazulevicius, J.V. Dual interface exciplex emission of quinoline and carbazole derivatives for simplified nondoped white OLEDs. J. Phys. Chem. C 2019, 123, 2386–2397. [Google Scholar] [CrossRef]
- Gu, P.-Y.; Zhao, Y.; He, J.-H.; Zhang, J.; Wang, C.; Xu, Q.-F.; Lu, J.-M.; Sun, X.W.; Zhang, Q. Synthesis, physical properties, and light-emitting diode performance of phenazine-based derivatives with three, five, and nine fused six-membered rings. J. Org. Chem. 2015, 80, 3030–3035. [Google Scholar] [CrossRef] [PubMed]
- Tonzola, C.J.; Kulkarni, A.P.; Gifford, A.P.; Kaminsky, W.; Jenekhe, S.A. Blue-light-emitting oligoquinolines: Synthesis, properties, and high-efficiency blue-light-emitting diodes. Adv. Funct. Mater. 2007, 17, 863–874. [Google Scholar] [CrossRef]
- Ahrens, L.; Butscher, J.; Brosius, V.; Rominger, F.; Freudenberg, J.; Vaynzof, Y.; Bunz, U.H.F. Azaacene dimers: Acceptor materials with a twist. Chem. Eur. J. 2020, 26, 412–418. [Google Scholar] [CrossRef] [PubMed]
- Lami, V.; Leibold, D.; Fassl, P.; Hofstetter, Y.J.; Becker-Koch, D.; Biegger, P.; Paulus, F.; Hopkinson, P.E.; Adams, M.; Bunz, U.H.F.; et al. N-Heteroacenes as a new class of non-fullerene electron acceptors for organic bulk-heterojunction photovoltaic devices. Sol. RRL 2017, 1, 1700053. [Google Scholar] [CrossRef]
- Leibold, D.; Lami, V.; Hofstetter, Y.J.; Becker-Koch, D.; Weu, A.; Biegger, P.; Paulus, F.; Bunz, U.H.F.; Hopkinson, P.E.; Bakulin, A.A.; et al. Triptycenyl-phenazino-thiadiazole as acceptor in organic bulk-heterojunction solar cells. Org. Electron. 2018, 57, 285–291. [Google Scholar] [CrossRef]
- Fischer, O.; Hepp, E. Oxydation des orthophenylendiamins. Ber. Dtsch. Chem. Ges. 1889, 22, 355–359. [Google Scholar] [CrossRef] [Green Version]
- Fischer, O.; Hepp, E. Ueber die fluorindine II. Ber. Dtsch. Chem. Ges. 1895, 28, 293–301. [Google Scholar] [CrossRef]
- Ahrens, L.; Tverskoy, O.; Weigold, S.; Ganschow, M.; Rominger, F.; Freudenberg, J.; Bunz, U.H.F. (Aza)pentacenes clipped into a ring: Stabilization of large (aza)acenes. Angew. Chem. Int. Ed. 2021, 60, 9270–9273. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, Q. Recent progress in well-defined higher azaacenes (n ≥ 6): Synthesis, molecular packing, and applications. Mater. Chem. Front. 2020, 4, 3419–3432. [Google Scholar] [CrossRef]
- Weng, S.-Z.; Shukla, P.; Kuo, M.-Y.; Chang, Y.-C.; Sheu, H.-S.; Chao, I.; Tao, Y.-T. Diazapentacene derivatives as thin-film transistor materials: Morphology control in realizing high-field-effect mobility. ACS Appl. Mater. Interfaces 2009, 1, 2071–2079. [Google Scholar] [CrossRef]
- Tverskoy, O.; Rominger, F.; Peters, A.; Himmel, H.-J.; Bunz, U.H.F. An efficient synthesis of tetraazapentacenes. Angew. Chem. Int. Ed. 2011, 50, 3557–3560. [Google Scholar] [CrossRef]
- Tong, C.; Zhao, W.; Luo, J.; Mao, H.; Chen, W.; Chan, H.S.O.; Chi, C. Large-Size linear and star-shaped dihydropyrazine fused pyrazinacenes. Org. Lett. 2012, 14, 494–497. [Google Scholar] [CrossRef]
- Seillan, C.; Brisset, H.; Siri, O. Efficient synthesis of substituted dihydrotetraazapentacenes. Org. Lett. 2008, 10, 4013–4016. [Google Scholar] [CrossRef] [PubMed]
- Chou, T.-C.; Lin, K.-C.; Kon-no, M.; Lee, C.-C.; Shinmyozu, T. Quinoxaline-embedded polyacenoquinone esters: Synthesis, electronic properties, and crystal structure. Org. Lett. 2011, 13, 4588–4591. [Google Scholar] [CrossRef] [PubMed]
- Brosius, V.; Müller, M.; Borstelmann, J.; Rominger, F.; Freudenberg, J.; Bunz, U.H.F. Azaacenodibenzosuberones. J. Org. Chem. 2020, 85, 296–300. [Google Scholar] [CrossRef]
- Zhu, G.; Zhang, G. Access to a phthalazine derivative through an angular cis-quinacridone. J. Org. Chem. 2021, 86, 1198–1203. [Google Scholar] [CrossRef] [PubMed]
- Gawrys, P.; Marszalek, T.; Bartnik, E.; Kucinska, M.; Ulanski, J.; Zagorska, M. Novel, low-cost, highly soluble n-type semiconductors: Tetraazaanthracene tetraesters. Org. Lett. 2011, 13, 6090–6093. [Google Scholar] [CrossRef] [PubMed]
- Lindner, B.D.; Engelhart, J.U.; Märken, M.; Tverskoy, O.; Appleton, A.L.; Rominger, F.; Hardcastle, K.I.; Enders, M.; Bunz, U.H.F. Synthesis and optical properties of diaza-and tetraazatetracenes. Chem. Eur. J. 2012, 18, 4627–4633. [Google Scholar] [CrossRef]
- Engelhart, J.U.; Lindner, B.D.; Schaffroth, M.; Schrempp, D.; Tverskoy, O.; Bunz, U.H.F. Substituted tetraaza-and hexaazahexacenes and their N,N′-dihydro derivatives: Syntheses, properties, and structures. Chem. Eur. J. 2015, 21, 8121–8129. [Google Scholar] [CrossRef]
- Bunz, U.H.F.; Engelhart, J.U. The palladium way to N-heteroacenes. Chem. Eur. J. 2016, 22, 4680–4689. [Google Scholar] [CrossRef]
- Schwaben, J.; Münster, N.; Klues, M.; Breuer, T.; Hofmann, P.; Harms, K.; Witte, G.; Koert, U. Efficient syntheses of novel fluoro-substituted pentacenes and azapentacenes: Molecular and solid-state properties. Chem. Eur. J. 2015, 21, 13758–13771. [Google Scholar] [CrossRef]
- Zhang, X.-X.; Buchwald, S.L. Efficient synthesis of N-aryl-aza-crown ethers via palladium-catalyzed amination. J. Org. Chem. 2000, 65, 8027–8031. [Google Scholar] [CrossRef]
- Lindner, B.D.; Engelhart, J.U.; Tverskoy, O.; Appleton, A.L.; Rominger, F.; Peters, A.; Himmel, H.-J.; Bunz, U.H.F. Stable hexacenes through nitrogen substitution. Angew. Chem. Int. Ed. 2011, 50, 8588–8591. [Google Scholar] [CrossRef]
- He, Z.; Mao, R.; Liu, D.; Miao, Q. Highly electron-deficient hexaazapentacenes and their dihydro precursors. Org. Lett. 2012, 14, 4190–4193. [Google Scholar] [CrossRef] [PubMed]
- China Raju, B.; Veera Prasad, K.; Saidachary, G.; Sridhar, B. A novel approach for C–C, C–N, and C–O bond formation reactions: A facile synthesis of benzophenazine, quinoxaline, and phenoxazine derivatives via ring opening of benzoxepines. Org. Lett. 2014, 16, 420–423. [Google Scholar] [CrossRef]
- Sikari, R.; Chakraborty, G.; Guin, A.K.; Paul, N.D. Nickel-catalyzed [4 + 2] annulation of nitriles and benzylamines by C–H/N–H activation. J. Org. Chem. 2021, 86, 279–290. [Google Scholar] [CrossRef] [PubMed]
- Kotwica, K.; Bujak, P.; Wamil, D.; Materna, M.; Skorka, L.; Gunka, P.A.; Nowakowski, R.; Golec, B.; Luszczynska, B.; Zagorska, M.; et al. Indanthrone dye revisited after sixty years. Chem. Commun. 2014, 50, 11543–11546. [Google Scholar] [CrossRef] [Green Version]
- Kotwica, K.; Bujak, P.; Wamil, D.; Pieczonka, A.; Wiosna-Salyga, G.; Gunka, P.A.; Jaroch, T.; Nowakowski, R.; Luszczynska, B.; Witkowska, E.; et al. Structural, Spectroscopic, electrochemical, and electroluminescent properties of tetraalkoxydinaphthophenazines: New solution-processable nonlinear azaacenes. J. Phys. Chem. C 2015, 119, 10700–10708. [Google Scholar] [CrossRef]
- Kotwica, K.; Bujak, P.; Data, P.; Krzywiec, W.; Wamil, D.; Gunka, P.A.; Skorka, L.; Jaroch, T.; Nowakowski, R.; Pron, A.; et al. Soluble flavanthrone derivatives: Synthesis, characterization, and application to organic light-emitting diodes. Chem. Eur. J. 2016, 22, 7978–7986. [Google Scholar] [CrossRef] [PubMed]
- Kotwica, K.; Bujak, P.; Skorka, L.; Jaroch, T.; Nowakowski, R. Luminophore from forgotten dye: Di(alkylthiophene) derivative of benzo[h]benz[5,6]acridino[2,1,9,8-klmna]acridine. Synth. Met. 2017, 232, 117–122. [Google Scholar] [CrossRef]
- Goto, K.; Yamaguchi, R.; Hiroto, S.; Ueno, H.; Kawai, T.; Shinokubo, H. Intermolecular oxidative annulation of 2-aminoanthracenes to diazaacenes and aza[7]helicenes. Angew. Chem. Int. Ed. 2012, 51, 10333–10336. [Google Scholar] [CrossRef] [PubMed]
- Takeda, Y.; Okazaki, M.; Minakata, S. Oxidative skeletal rearrangement of 1,1′-binaphthalene-2,2′-diamines (BINAMs) via C–C bond cleavage and nitrogen migration: A versatile synthesis of U-shaped azaacenes. Chem. Commun. 2014, 50, 10291–10294. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wohl, A.; Aue, W. Ueber die einwirkung von nitrobenzol auf anilin bei gegenwart von alkali. Ber. Dtsch. Chem. Ges. 1901, 34, 2442–2450. [Google Scholar] [CrossRef]
- Haddadin, M.J.; Issidorides, C.H. Enamines with isobenzofuroxan: A novel synthesis of quinoxaline-di-n-oxides. Tetrahedron Lett. 1965, 6, 3253–3256. [Google Scholar] [CrossRef]
- Kotwica, K. Nowe Półprzewodnikowe Pochodne Indantronu i Flawantronu: Synteza, Właściwości Strukturalne, Spektroskopowe i Elektrochemiczne oraz Zastosowanie w Elektronice Organicznej. Ph.D. Thesis, Warsaw University of Technology, Warsaw, Poland, 2018. [Google Scholar]
- Ji, L.; Haehnel, M.; Krummenacher, I.; Biegger, P.; Geyer, F.L.; Tverskoy, O.; Schaffroth, M.; Han, J.; Dreuw, A.; Marder, T.B.; et al. The radical anion and dianion of tetraazapentacene. Angew. Chem. Int. Ed. 2016, 55, 10498–10501. [Google Scholar] [CrossRef]
- Müller, M.; Beglaryan, S.S.; Koser, S.; Hahn, S.; Tverskoy, O.; Rominger, F.; Bunz, U.H.F. Dicyano-substituted diazaacenes. Chem. Eur. J. 2017, 23, 7066–7073. [Google Scholar] [CrossRef]
- Biegger, P.; Stolz, S.; Intorp, S.N.; Zhang, Y.; Engelhart, J.U.; Rominger, F.; Hardcastle, K.I.; Lemmer, U.; Qian, X.; Hamburger, M.; et al. Soluble diazaiptycenes: Materials for solution-processed organic electronics. J. Org. Chem. 2015, 80, 582–589. [Google Scholar] [CrossRef]
- Porz, M.; Rominger, F.; Bunz, U.H.F. Halogen bonding in diaza-triisopropylsilyl-tetracene crystals? Cryst. Growth Des. 2014, 14, 5962–5965. [Google Scholar] [CrossRef]
- Engelhart, J.U.; Paulus, F.; Schaffroth, M.; Vasilenko, V.; Tverskoy, O.; Rominger, F.; Bunz, U.H.F. Halogenated symmetrical tetraazapentacenes: Synthesis, structures, and properties. J. Org. Chem. 2016, 81, 1198–1205. [Google Scholar] [CrossRef]
- Lu, X.; Fan, S.; Wu, J.; Jia, X.; Wang, Z.-S.; Zhou, G. Controlling the charge transfer in D–A–D chromophores based on pyrazine derivatives. J. Org. Chem. 2014, 79, 6480–6489. [Google Scholar] [CrossRef]
- Isoda, K.; Abe, T.; Kawamoto, I.; Tadokoro, M. Self-organized superstructure and electronic properties of a liquid-crystalline tetraazapentacene derivative. Chem. Lett. 2015, 44, 126–128. [Google Scholar] [CrossRef]
- Kolmer-Anderl, N.; Kolmer, A.; Thiele, C.M.; Rehahn, M. Exploration of the photodegradation of naphtho[2,3-g] quinoxalines and pyrazino[2,3-b]phenazines. Chem. Eur. J. 2016, 22, 5277–5287. [Google Scholar] [CrossRef] [PubMed]
- More, S.; Bhosale, R.; Mateo-Alonso, A. Low-LUMO pyrene-fused azaacenes. Chem. Eur. J. 2014, 20, 10626–10631. [Google Scholar] [CrossRef]
- García, R.; Melle-Franco, M.; Mateo-Alonso, A. A short pyrene-fused pyrazaacene with red to near-infrared photoluminescence. Chem. Commun. 2015, 51, 8037–8040. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- More, S.; Bhosale, R.; Choudhary, S.; Mateo-Alonso, A. Versatile 2,7-substituted pyrene synthons for the synthesis of pyrene-fused azaacenes. Org. Lett. 2012, 14, 4170–4173. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Li, P.; Wu, J.; Gao, J.; Xiong, W.-W.; Zhang, G.; Zhao, Y.; Zhang, Q. [4 + 2] cycloaddition reaction to approach diazatwistpentacenes: Synthesis, structures, physical properties, and self-assembly. J. Org. Chem. 2014, 79, 4438–4445. [Google Scholar] [CrossRef]
- Gao, B.; Wang, M.; Cheng, Y.; Wang, L.; Jing, X.; Wang, F. Pyrazine-containing acene-type molecular ribbons with up to 16 rectilinearly arranged fused aromatic rings. J. Am. Chem. Soc. 2008, 130, 8297–8306. [Google Scholar] [CrossRef]
- Wang, M.; Li, Y.; Tong, H.; Cheng, Y.; Wang, L.; Jing, X.; Wang, F. Hexaazatriphenylene derivatives with tunable lowest unoccupied molecular orbital levels. Org. Lett. 2011, 13, 4378–4381. [Google Scholar] [CrossRef]
- Segura, J.L.; Juárez, R.; Ramos, M.; Seoane, C. Hexaazatriphenylene (HAT) derivatives: From synthesis to molecular design, self-organization and device applications. Chem. Soc. Rev. 2015, 44, 6850–6885. [Google Scholar] [CrossRef]
- Shao, J.; Chang, J.; Chi, C. Linear and star-shaped pyrazine-containing acene dicarboximides with high electron-affinity. Org. Biomol. Chem. 2012, 10, 7045–7052. [Google Scholar] [CrossRef]
- Hahn, S.; Alrayyani, M.; Sontheim, A.; Wang, X.; Rominger, F.; Miljanić, O.Š.; Bunz, U.H.F. Synthesis and characterization of heterobenzenacyclo-octaphanes derived from cyclotetrabenzoin. Chem. Eur. J. 2017, 23, 10543–10550. [Google Scholar] [CrossRef] [PubMed]
- Hahn, S.; Koser, S.; Hodecker, M.; Seete, P.; Rominger, F.; Miljanić, O.Š.; Dreuw, A.; Bunz, U.H.F. Phenylene bridged cyclic azaacenes: Dimers and trimers. Chem. Eur. J. 2018, 24, 6968–6974. [Google Scholar] [CrossRef]
- Ding, F.; Xia, D.; Ge, C.; Kang, Z.; Yang, Y.; Fan, R.; Lin, K.; Gao, X. Indenone-fused N-heteroacenes. J. Mater. Chem. C 2019, 7, 14314–14319. [Google Scholar] [CrossRef]
- Alagna, N.; Lustres, J.L.P.; Roozbeh, A.; Han, J.; Hahn, S.; Berger, F.J.; Zaumseil, J.; Dreuw, A.; Bunz, U.H.F.; Buckup, T. Ultrafast singlet fission in rigid azaarene dimers with negligible orbital overlap. J. Phys. Chem. B 2020, 124, 9163–9174. [Google Scholar] [CrossRef] [PubMed]
- Hahn, S.; Biegger, P.; Bender, M.; Rominger, F.; Bunz, U.H.F. Synthesis of alkynylated benzo[a]naphtho[2,3-i]phenazine derivatives. Chem. Eur. J. 2016, 22, 869–873. [Google Scholar] [CrossRef] [PubMed]
- Hahn, S.; Koser, S.; Hodecker, M.; Tverskoy, O.; Rominger, F.; Dreuw, A.; Bunz, U.H.F. Alkyne-substituted N-heterophenes. Chem. Eur. J. 2017, 23, 8148–8151. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Fonari, A.; Zhang, Y.; Zhao, G.; Coropceanu, V.; Hu, W.; Parkin, S.; Brédas, J.-L.; Briseno, A.L. Triisopropylsilylethynyl-functionalized graphene-like fragment semiconductors: Synthesis, crystal packing, and density functional theory calculations. Chem. Eur. J. 2013, 19, 17907–17916. [Google Scholar] [CrossRef] [PubMed]
- Schneider, J.A.; Perepichka, D.F. A new approach to polycyclic azaarenes: Visible-light photolysis of vinyl azides in the synthesis of diazabenzopyrene and diazaperylene. J. Mater. Chem. C 2016, 4, 7269–7276. [Google Scholar] [CrossRef]
- Liang, Z.; Tang, Q.; Mao, R.; Liu, D.; Xu, J.; Miao, Q. The position of nitrogen in N-heteropentacenes matters. Adv. Mater. 2011, 23, 5514–5518. [Google Scholar] [CrossRef]
- Lindner, B.D.; Zhang, Y.; Höfle, S.; Berger, N.; Teusch, C.; Jesper, M.; Hardcastle, K.I.; Qian, X.; Lemmer, U.; Colsmann, A.; et al. N-fused quinoxalines and benzoquinoxalines as attractive emitters for organic light emitting diodes. J. Mater. Chem. C 2013, 1, 5718–5724. [Google Scholar] [CrossRef]
- Li, J.; Yan, F.; Gao, J.; Li, P.; Xiong, W.-W.; Zhao, Y.; Sun, X.W.; Zhang, Q. Synthesis, physical properties and OLED performance of azatetracenes. Dyes Pigm. 2015, 112, 93–98. [Google Scholar] [CrossRef]
- Chou, H.-H.; Shih, H.-H.; Cheng, C.-H. Triptycene derivatives as high-Tg host materials for various electrophosphorescent devices. J. Mater. Chem. 2010, 20, 798–805. [Google Scholar] [CrossRef]
- Sasabe, H.; Kido, J. Multifunctional materials in high-performance OLEDs: Challenges for solid-state lighting. Chem. Mater. 2011, 23, 621–630. [Google Scholar] [CrossRef]
- Andruleviciene, V.; Leitonas, K.; Volyniuk, D.; Sini, G.; Grazulevicius, J.V.; Getautis, V. TADF versus TTA emission mechanisms in acridan and carbazole-substituted dibenzo[a,c]phenazines: Towards triplet harvesting emitters and hosts. Chem. Eng. J. 2021, 417, 127902. [Google Scholar] [CrossRef]
- Yang, W.; Ning, W.; Gong, S.; Yang, C. Deep-red thermally activated delayed fluorescence emitters based on a phenanthroline-containing planar acceptor. Dyes Pigm. 2021, 192, 109474. [Google Scholar] [CrossRef]
- Yan, C.; Barlow, S.; Wang, Z.; Yan, H.; Jen, A.K.-Y.; Marder, S.R.; Zhan, X. Non-fullerene acceptors for organic solar cells. Nat. Rev. Mater. 2018, 3, 1–19. [Google Scholar] [CrossRef]
- Wadsworth, A.; Moser, M.; Marks, A.; Little, M.S.; Gasparini, N.; Brabec, C.J.; Baran, D.; McCulloch, I. Critical review of the molecular design progress in non-fullerene electron acceptors towards commercially viable organic solar cells. Chem. Soc. Rev. 2019, 48, 1596–1625. [Google Scholar] [CrossRef]
- Hahn, S.; Butscher, J.; An, Q.; Jocic, A.; Tverskoy, O.; Richter, M.; Feng, X.; Rominger, F.; Vaynzof, Y.; Bunz, U.H.F. Azaarene dimers. Chem. Eur. J. 2019, 25, 7285–7291. [Google Scholar] [CrossRef]
- Saliba, M.; Correa-Baena, J.-P.; Grätzel, M.; Hagfeldt, A.; Abate, A. Perovskite solar cells: From the atomic level to film quality and device performance. Angew. Chem. Int. Ed. 2018, 57, 2554–2569. [Google Scholar] [CrossRef]
- Boyd, C.C.; Cheacharoen, R.; Leijtens, T.; McGehee, M.D. Understanding degradation mechanisms and improving stability of perovskite photovoltaics. Chem. Rev. 2019, 119, 3418–3451. [Google Scholar] [CrossRef]
- Gu, P.-Y.; Wang, N.; Wang, C.; Zhou, Y.; Long, G.; Tian, M.; Chen, W.; Sun, X.W.; Kanatzidis, M.G.; Zhang, Q. Pushing up the efficiency of planar perovskite solar cells to 18.2% with organic small molecules as the electron transport layer. J. Mater. Chem. A 2017, 5, 7339–7344. [Google Scholar] [CrossRef]
- Gu, P.-Y.; Wang, N.; Wu, A.; Wang, Z.; Tian, M.; Fu, Z.; Sun, X.W.; Zhang, Q. An azaacene derivative as promising electron-transport layer for inverted perovskite solar cells. Chem. Asian J. 2016, 11, 2135–2138. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Zhao, K.; Ding, T.; Liu, W.; Ahmed, A.S.; Wang, Z.; Tian, M.; Sun, X.W.; Zhang, Q. Improving interfacial charge recombination in planar heterojunction perovskite photovoltaics with small molecule as electron transport layer. Adv. Energy Mater. 2017, 7, 1700522. [Google Scholar] [CrossRef]
- Liang, Z.; Tang, Q.; Xu, J.; Miao, Q. Soluble and stable N-heteropentacenes with high field-effect mobility. Adv. Mater. 2011, 23, 1535–1539. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-Y.; Song, C.-L.; Zeng, W.-J.; Zhou, K.-G.; Shi, Z.-F.; Ma, C.-B.; Yang, F.; Zhang, H.-L.; Gong, X. High and balanced hole and electron mobilities from ambipolar thin-film transistors based on nitrogen-containing oligoacences. J. Am. Chem. Soc. 2010, 132, 16349–16351. [Google Scholar] [CrossRef] [PubMed]
- Song, C.-L.; Ma, C.-B.; Yang, F.; Zeng, W.-J.; Zhang, H.-L.; Gong, X. Synthesis of Tetrachloro-azapentacene as an ambipolar organic semiconductor with high and balanced carrier mobilities. Org. Lett. 2011, 13, 2880–2883. [Google Scholar] [CrossRef]
- Liao, H.; Xiao, C.; Ravva, M.K.; Yao, L.; Yu, Y.; Yang, Y.; Zhang, W.; Zhang, L.; Li, Z.; McCulloch, I.; et al. Fused pyrazine-and carbazole-containing azaacenes: Synthesis and properties. ChemPlusChem 2019, 84, 1257–1262. [Google Scholar] [CrossRef]
- Chu, M.; Fan, J.-X.; Yang, S.; Liu, D.; Ng, C.F.; Dong, H.; Ren, A.-M.; Miao, Q. Halogenated tetraazapentacenes with electron mobility as high as 27.8 cm2 V−1 s−1 in solution-processed n-channel organic thin-film transistors. Adv. Mater. 2018, 30, 1803467. [Google Scholar] [CrossRef]
- Wu, Y.; Yin, Z.; Xiao, J.; Liu, Y.; Wei, F.; Tan, K.J.; Kloc, C.; Huang, L.; Yan, Q.; Hu, F.; et al. Crystal structure and phototransistor behavior of N-substituted heptacence. ACS Appl. Mater. Interfaces 2012, 8517, 1–4. [Google Scholar] [CrossRef]
- Ganschow, M.; Koser, S.; Hodecker, M.; Rominger, F.; Freudenberg, J.; Dreuw, A.; Bunz, U.H.F. Azaacenes bearing five-membered rings. Chem. Eur. J. 2018, 24, 13667–13675. [Google Scholar] [CrossRef] [PubMed]
- He, J.-L.; Kong, F.-C.; Sun, B.; Wang, X.-J.; Tian, Q.-S.; Fan, J.; Liao, L.-S. Highly efficient deep-red TADF organic light-emitting diodes via increasing the acceptor strength of fused polycyclic aromatics. Chem. Eng. J. 2021, 424, 130470. [Google Scholar] [CrossRef]
- Hu, Y.; Zhang, Y.; Han, W.; Li, J.; Pu, X.; Wu, D.; Bin, Z.; You, J. Orange–red organic light emitting diodes with high efficiency and low efficiency roll-off: Boosted by a fused acceptor composed of pyrazine and maleimide. Chem. Eng. J. 2022, 428, 131186. [Google Scholar] [CrossRef]
- Maeng, J.H.; Braveenth, R.; Jung, Y.H.; Hwang, S.J.; Lee, H.; Min, H.L.; Kim, J.Y.; Han, C.W.; Kwon, J.H. Efficiency enhancement in orange red thermally activated delayed fluorescence OLEDs by using a rigid di-indolocarbazole donor moiety. Dyes Pigm. 2021, 194, 109580. [Google Scholar] [CrossRef]
- Ha, J.M.; Shin, H.B.; Joung, J.F.; Chung, W.J.; Jeong, J.-E.; Kim, S.; Hur, S.H.; Bae, S.-Y.; Kim, J.-Y.; Lee, J.Y.; et al. Rational molecular design of azaacene-based narrowband green-emitting fluorophores: Modulation of spectral bandwidth and vibronic transitions. ACS Appl. Mater. Interfaces 2021, 13, 26227–26236. [Google Scholar] [CrossRef] [PubMed]
- Ganschow, M.; Koser, S.; Hahn, S.; Rominger, F.; Freudenberg, J.; Bunz, U.H.F. Dibenzobarrelene-based azaacenes: Emitters in organic light-emitting diodes. Chem. Eur. J. 2017, 23, 4415–4421. [Google Scholar] [CrossRef]
- Li, W.; Pan, Y.; Yao, L.; Liu, H.; Zhang, S.; Wang, C.; Shen, F.; Lu, P.; Yang, B.; Ma, Y. A Hybridized local and charge-transfer excited state for highly efficient fluorescent OLEDs: Molecular design, spectral character, and full exciton utilization. Adv. Opt. Mater. 2014, 2, 892–901. [Google Scholar] [CrossRef]
- Huang, W.; Einzinger, M.; Zhu, T.; Chae, H.S.; Jeon, S.; Ihn, S.-G.; Sim, M.; Kim, S.; Su, M.; Teverovskiy, G.; et al. Molecular design of deep blue thermally activated delayed fluorescence materials employing a homoconjugative triptycene scaffold and dihedral angle tuning. Chem. Mater. 2018, 30, 1462–1466. [Google Scholar] [CrossRef]
- Park, Y.; Kim, S.; Lee, J.-H.; Jung, D.H.; Wu, C.-C.; Park, J. New blue-violet emitters based on an indenopyrazine core for OLEDs: Effects of the position of m-terphenyl side group substitution on optical and electroluminescence properties. Org. Electron. 2010, 11, 864–871. [Google Scholar] [CrossRef]
- Chen, M.; Perzon, E.; Andersson, M.R.; Marcinkevicius, S.; Jönsson, S.K.M.; Fahlman, M.; Berggren, M. 1 Micron wavelength photo-and electroluminescence from a conjugated polymer. Appl. Phys. Lett. 2004, 84, 3570–3572. [Google Scholar] [CrossRef]
- Selzer, F.; Falkenberg, C.; Hamburger, M.; Baumgarten, M.; Müllen, K.; Leo, K.; Riede, M. Improved organic p-i-n type solar cells with n-doped fluorinated hexaazatrinaphthylene derivatives HATNA-F6 and HATNA-F12 as transparent electron transport material. J. Appl. Phys. 2014, 115, 054515. [Google Scholar] [CrossRef]
- Zhao, D.; Zhu, Z.; Kuo, M.-Y.; Chueh, C.-C.; Jen, A.K.-Y. Hexaazatrinaphthylene derivatives: Efficient Electron-transporting materials with tunable energy levels for inverted perovskite solar cells. Angew. Chem. Int. Ed. 2016, 55, 8999–9003. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Zhao, D.; Chueh, C.-C.; Shi, X.; Li, Z.; Jen, A.K.-Y. Highly efficient and stable perovskite solar cells enabled by all-crosslinked charge-transporting layers. Joule 2018, 2, 168–183. [Google Scholar] [CrossRef] [Green Version]
- Lindner, B.D.; Paulus, F.; Appleton, A.L.; Schaffroth, M.; Engelhart, J.U.; Schelkle, K.M.; Tverskoy, O.; Rominger, F.; Hamburger, M.; Bunz, U.H.F. Electron-transporting phenazinothiadiazoles with engineered microstructure. J. Mater. Chem. C 2014, 2, 9609–9612. [Google Scholar] [CrossRef]
- Thiadiazolo-Azaacenes-Müller-2019-Chemistry–A European Journal-Wiley Online Library. Available online: https://chemistry-europe.onlinelibrary.wiley.com/doi/full/10.1002/chem.201900462 (accessed on 20 August 2021).
- Xu, X.; Yao, Y.; Shan, B.; Gu, X.; Liu, D.; Liu, J.; Xu, J.; Zhao, N.; Hu, W.; Miao, Q. Electron mobility exceeding 10 cm2 V−1 s−1 and band-like charge transport in solution-processed n-channel organic thin-film transistors. Adv. Mater. 2016, 28, 5276–5283. [Google Scholar] [CrossRef] [PubMed]
- Paulus, F.; Porz, M.; Schaffroth, M.; Rominger, F.; Leineweber, A.; Vaynzof, Y.; Bunz, U.H.F. Side-group engineering: The influence of norbornadienyl substituents on the properties of ethynylated pentacene and tetraazapentacene. Org. Electron. 2016, 33, 102–109. [Google Scholar] [CrossRef]
- Reiss, H.; Ji, L.; Han, J.; Koser, S.; Tverskoy, O.; Freudenberg, J.; Hinkel, F.; Moos, M.; Friedrich, A.; Krummenacher, I.; et al. Bromination improves the electron mobility of tetraazapentacene. Angew. Chem. Int. Ed. 2018, 57, 9543–9547. [Google Scholar] [CrossRef] [PubMed]
- Brosius, V.; Weigold, S.; Hippchen, N.; Rominger, F.; Freudenberg, J.; Bunz, U.H.F. Diindenopyrazines: Electron-deficient arenes. Chem. Eur. J. 2021, 27, 10001–10005. [Google Scholar] [CrossRef]
- Gu, P.-Y.; Wang, Z.; Liu, G.; Yao, H.; Wang, Z.; Li, Y.; Zhu, J.; Li, S.; Zhang, Q. Synthesis, full characterization, and field effect transistor behavior of a stable pyrene-fused N-heteroacene with twelve linearly annulated six-membered rings. Chem. Mater. 2017, 29, 4172–4175. [Google Scholar] [CrossRef]
- Endres, A.H.; Schaffroth, M.; Paulus, F.; Reiss, H.; Wadepohl, H.; Rominger, F.; Krämer, R.; Bunz, U.H.F. Coronene-containing N-heteroarenes: 13 rings in a row. J. Am. Chem. Soc. 2016, 138, 1792–1795. [Google Scholar] [CrossRef]
- Rohnacher, V.; Benneckendorf, F.S.; Münch, M.; Sauter, E.; Asyuda, A.; Barf, M.-M.; Tisserant, J.-N.; Hillebrandt, S.; Rominger, F.; Jänsch, D.; et al. Functionalized tetrapodal diazatriptycenes for electrostatic dipole engineering in N-type organic thin film transistors. Adv. Mater. Technol. 2021, 6, 2000300. [Google Scholar] [CrossRef]
- Gu, P.-Y.; Wang, Z.; Xiao, F.-X.; Lin, Z.; Song, R.; Xu, Q.-F.; Lu, J.-M.; Liu, B.; Zhang, Q. An ambipolar azaacene as a stable photocathode for metal-free light-driven water reduction. Mater. Chem. Front. 2017, 1, 495–498. [Google Scholar] [CrossRef]
- Wang, C.; Wang, J.; Li, P.; Gao, J.; Tan, Y.; Xiong, W.; Hu, B.; Lee, S.; Zhao, Y.; Zhang, Q. Synthesis, characterization, and non-volatile memory device application of an N-substituted heteroacene. Chem. Asian J. 2014, 9, 779–783. [Google Scholar] [CrossRef] [PubMed]
- Zhao, K.; Yu, F.; Liu, W.; Huang, Y.; Said, A.A.; Li, Y.; Zhang, Q. Unexpected synthesis, properties, and nonvolatile memory device application of imidazole-fused azaacenes. J. Org. Chem. 2020, 85, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Zheng, K.; Wang, C.; Leck, K.S.; Hu, F.; Sun, X.W.; Zhang, Q. Synthesis and nonvolatile memory behaviors of dioxatetraazapentacene derivatives. ACS Appl. Mater. Interfaces 2013, 5, 6458–6462. [Google Scholar] [CrossRef] [PubMed]
- De Girolamo, J.; Reiss, P.; Pron, A. Hybrid materials from diaminopyriminide-functionalized poly(hexylthiophene) and thymine-capped CdSe nanocrystals: Part II—Hydrogen bond assisted layer-by-layer molecular level processing. J. Phys. Chem. C 2008, 112, 8797–8801. [Google Scholar] [CrossRef]
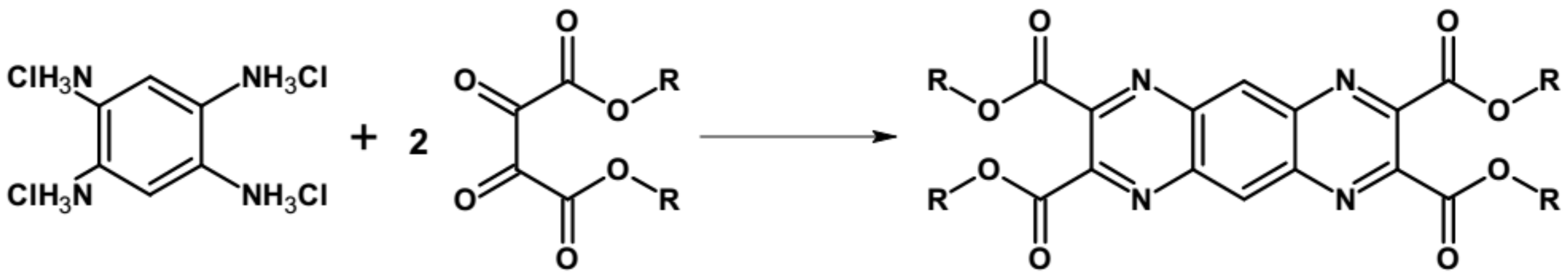
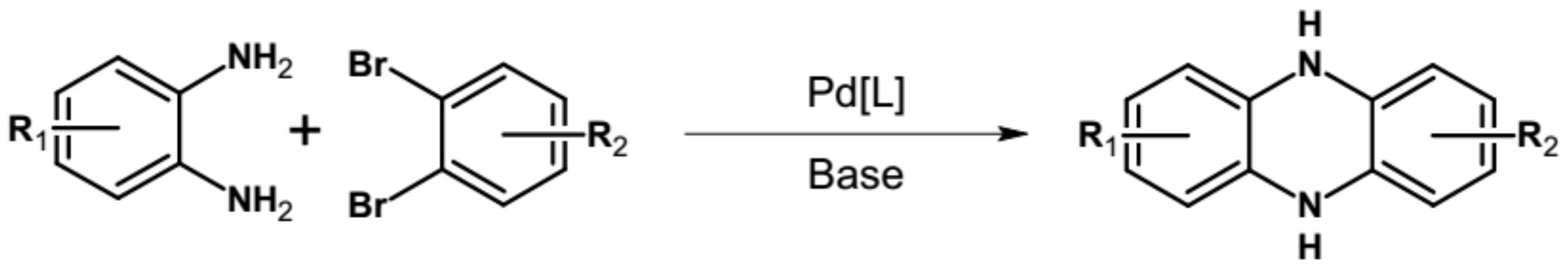
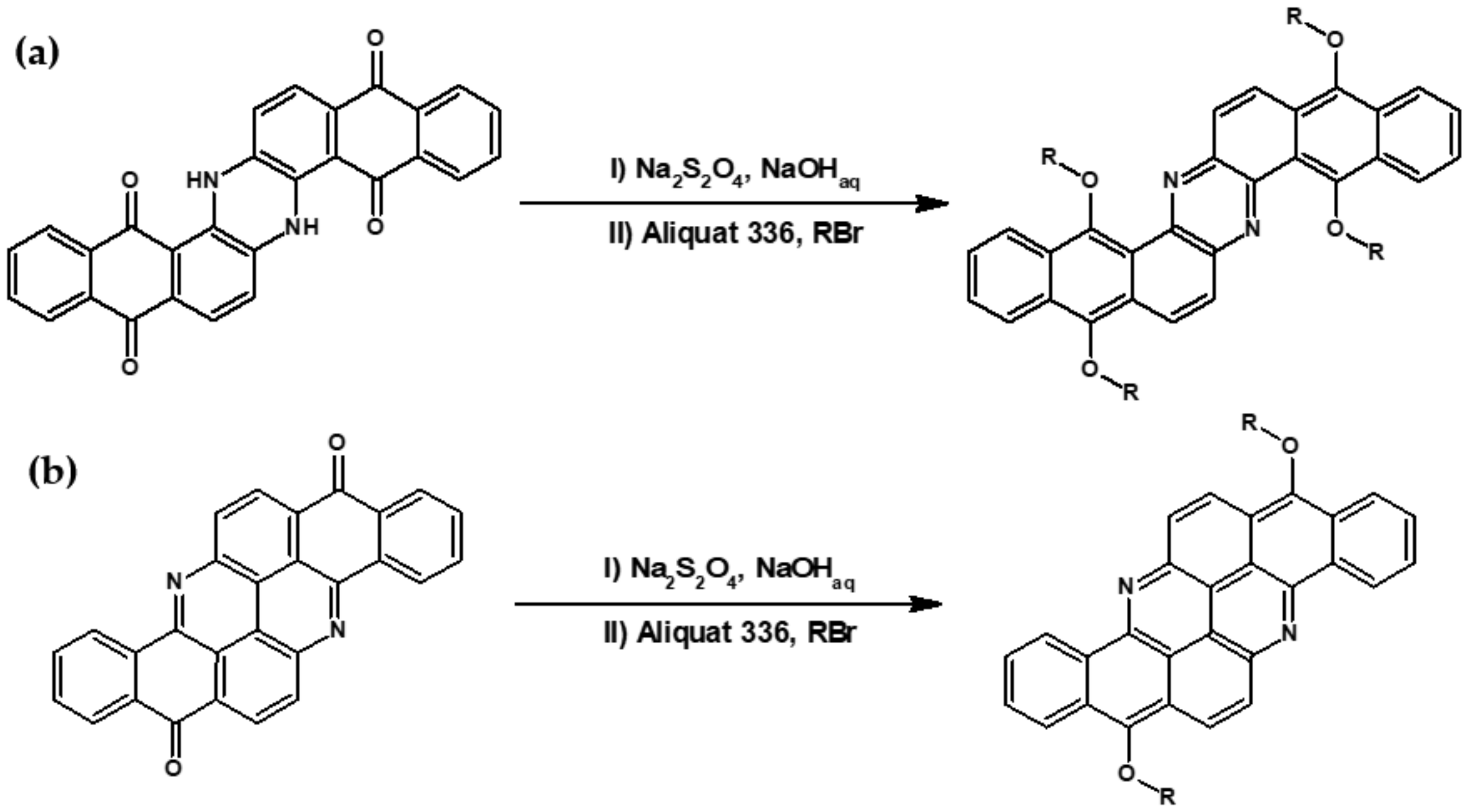
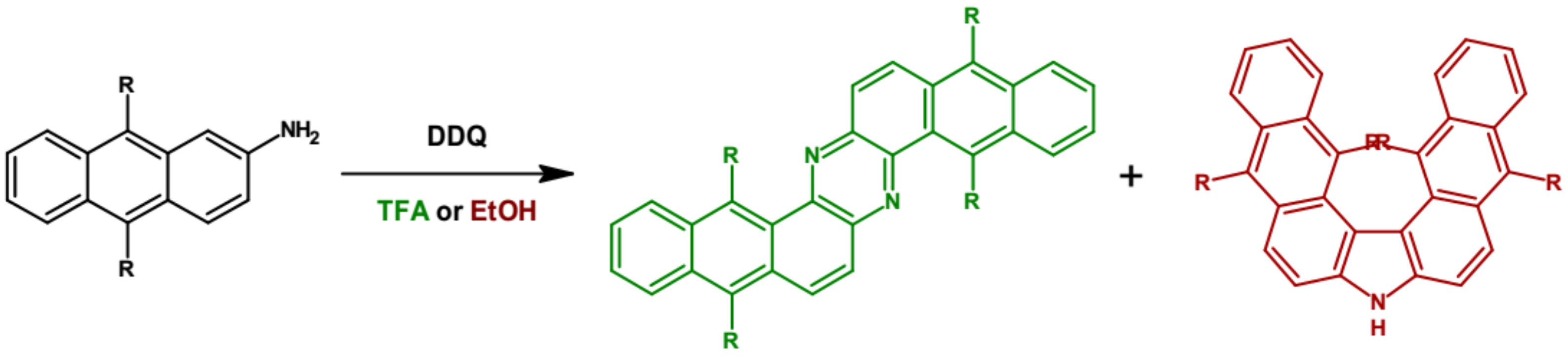
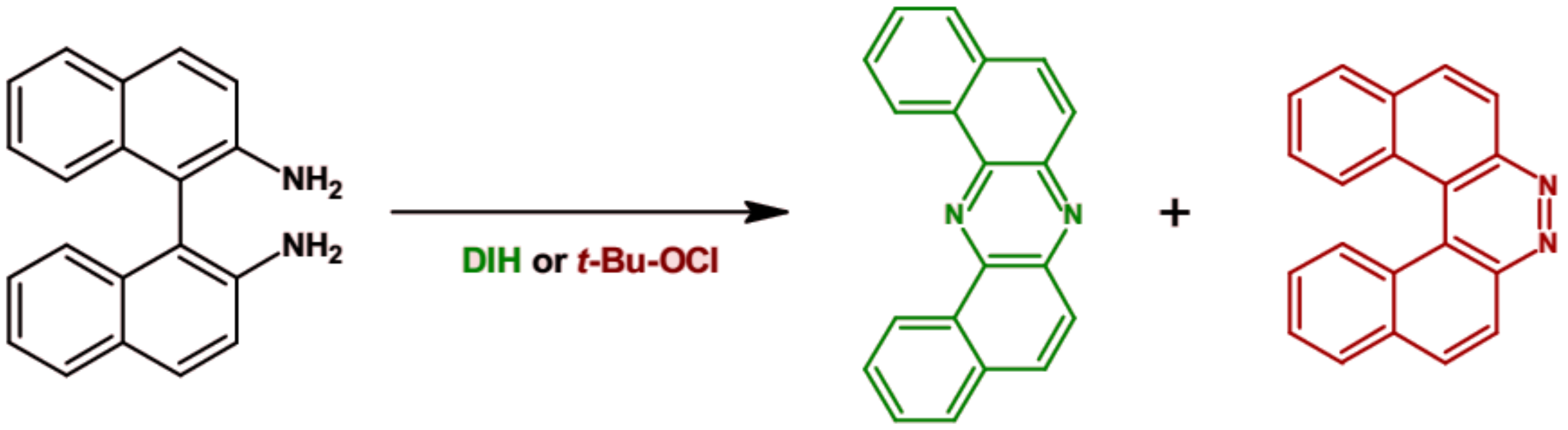


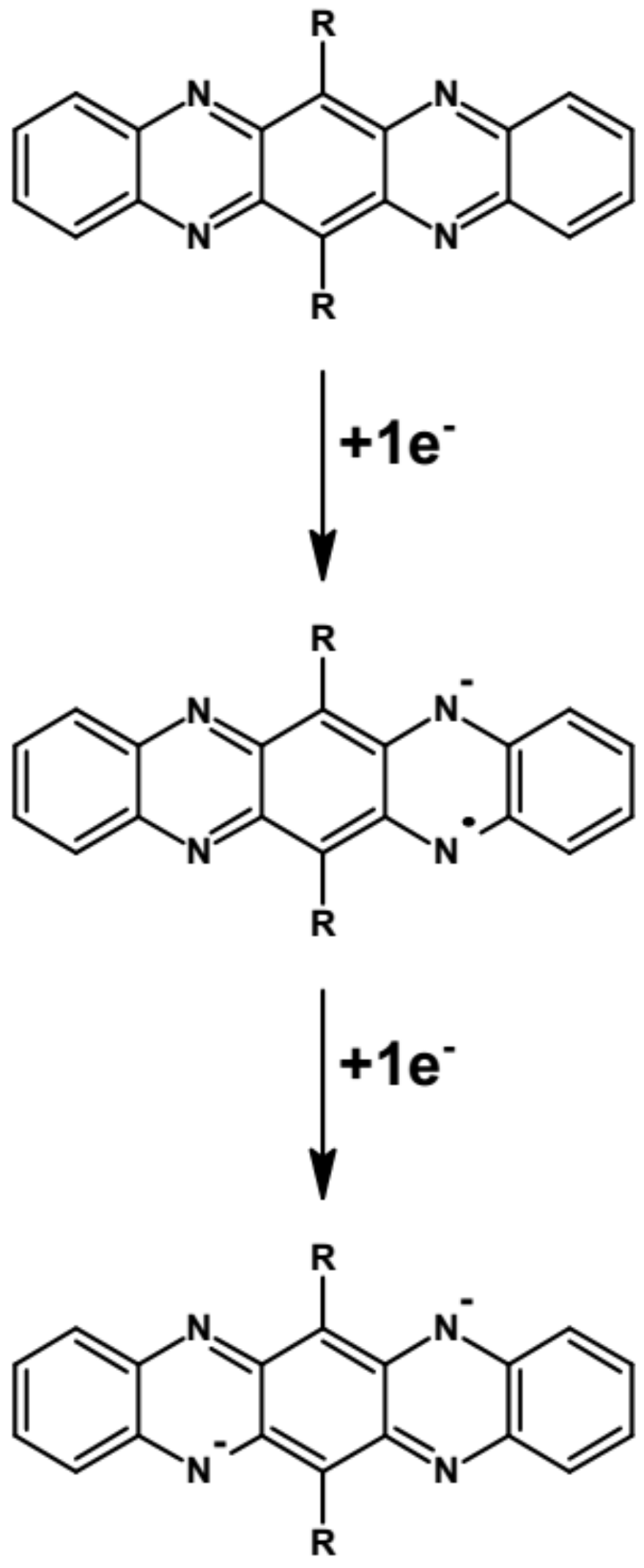
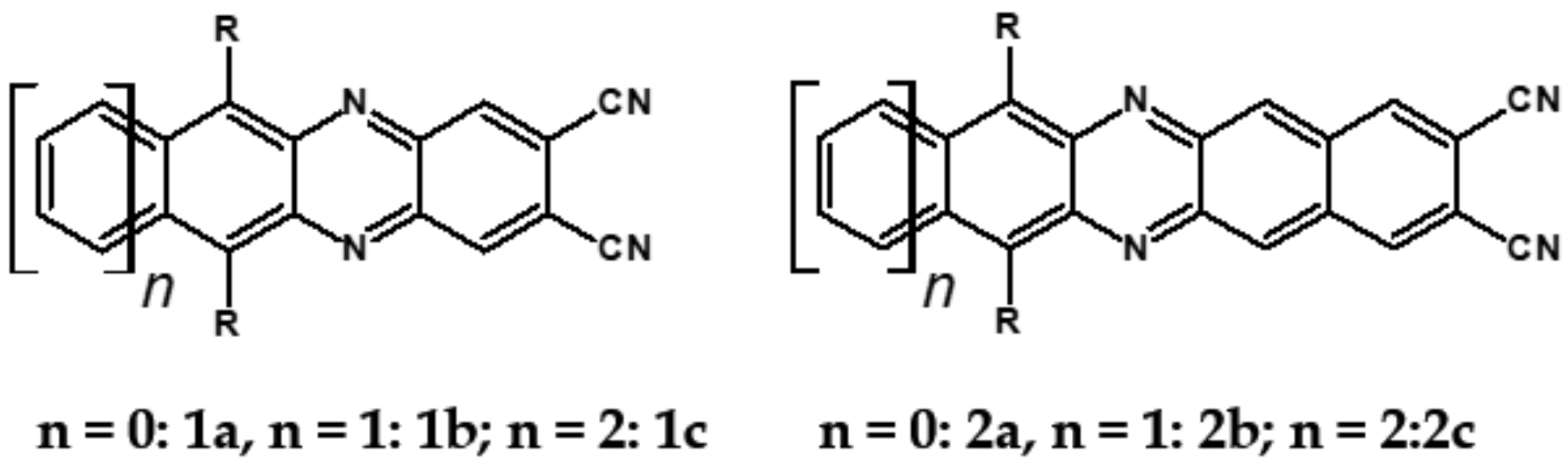
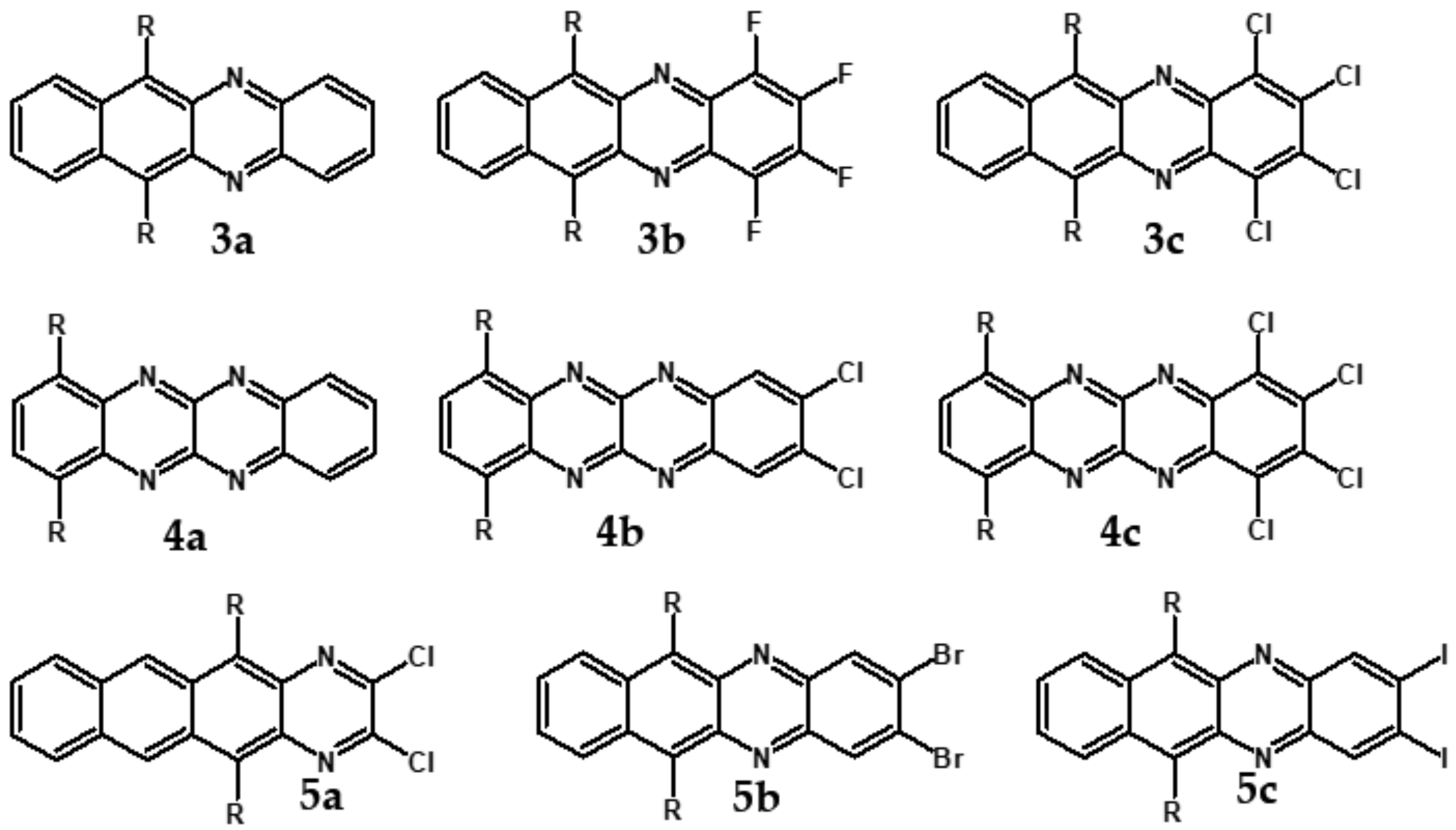
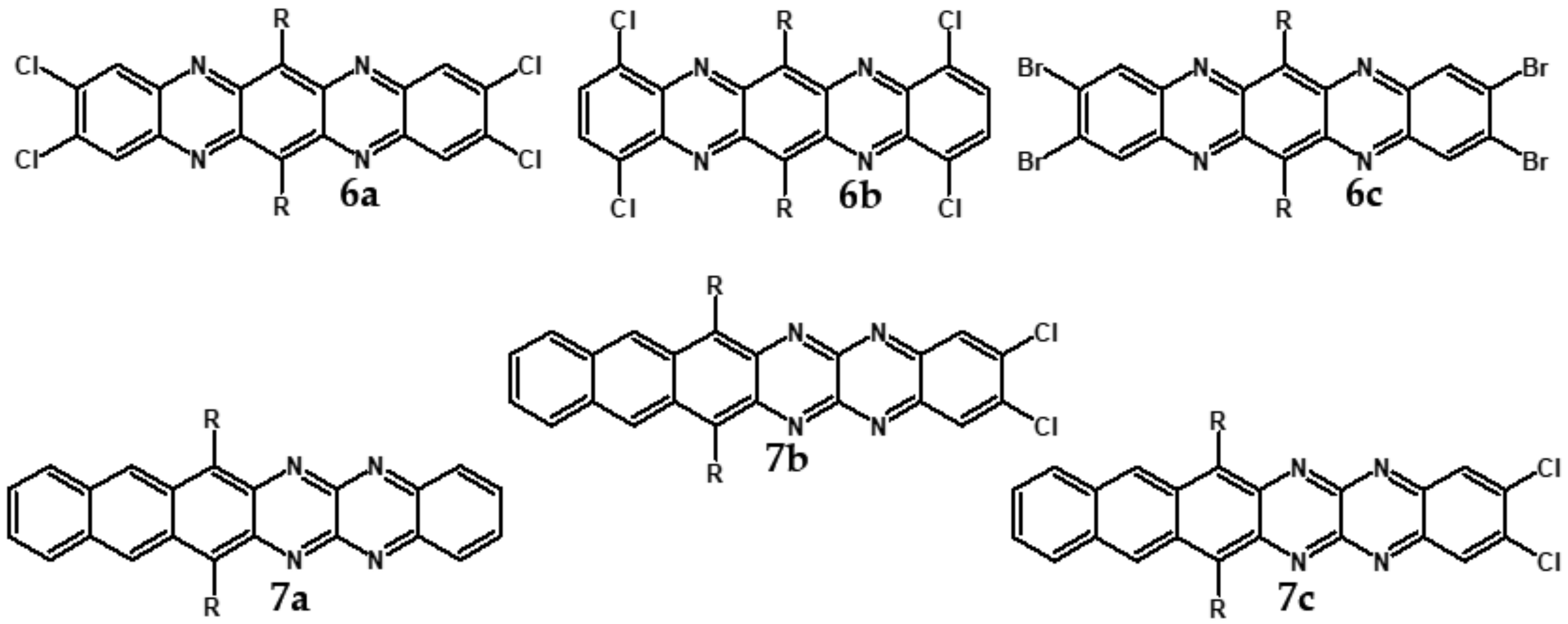

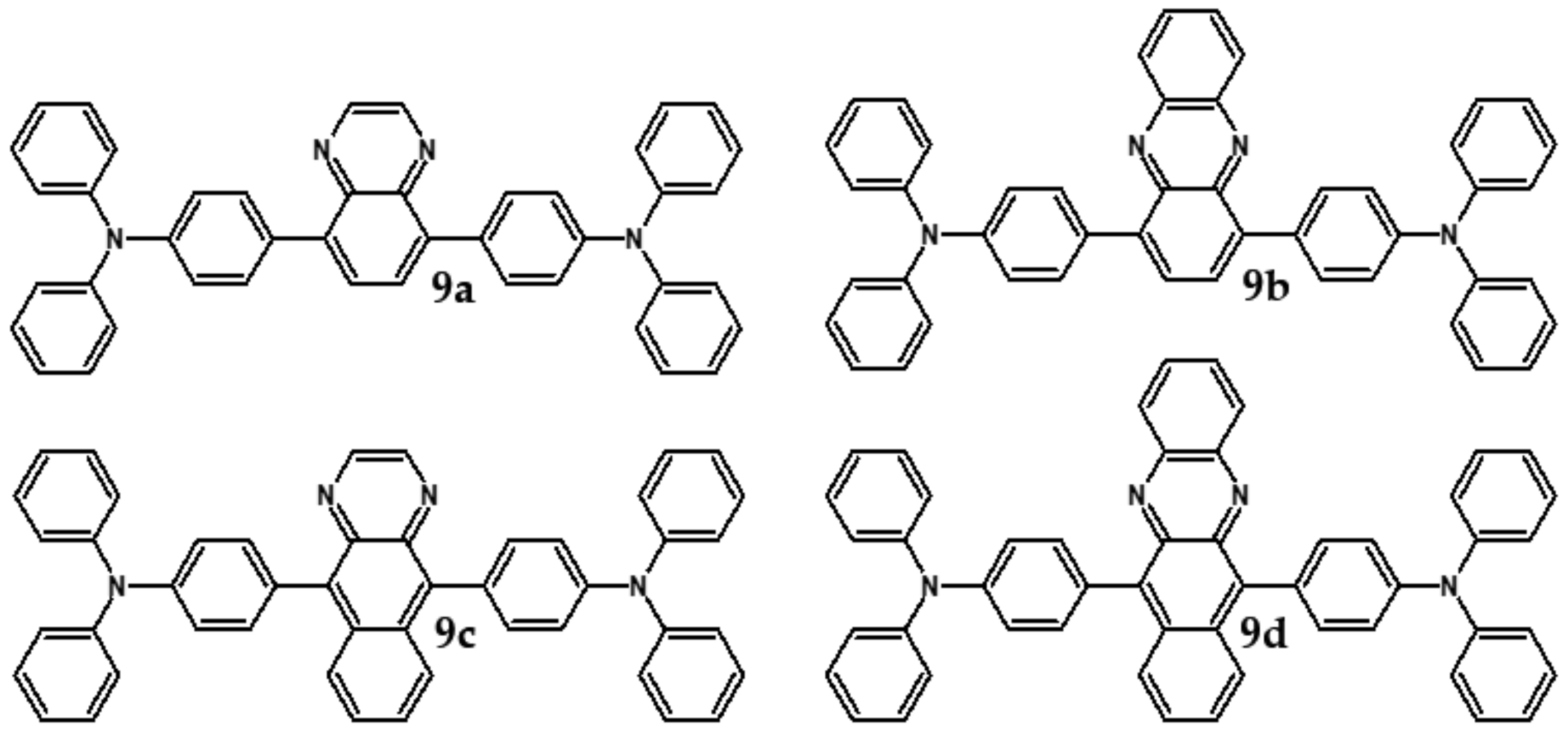

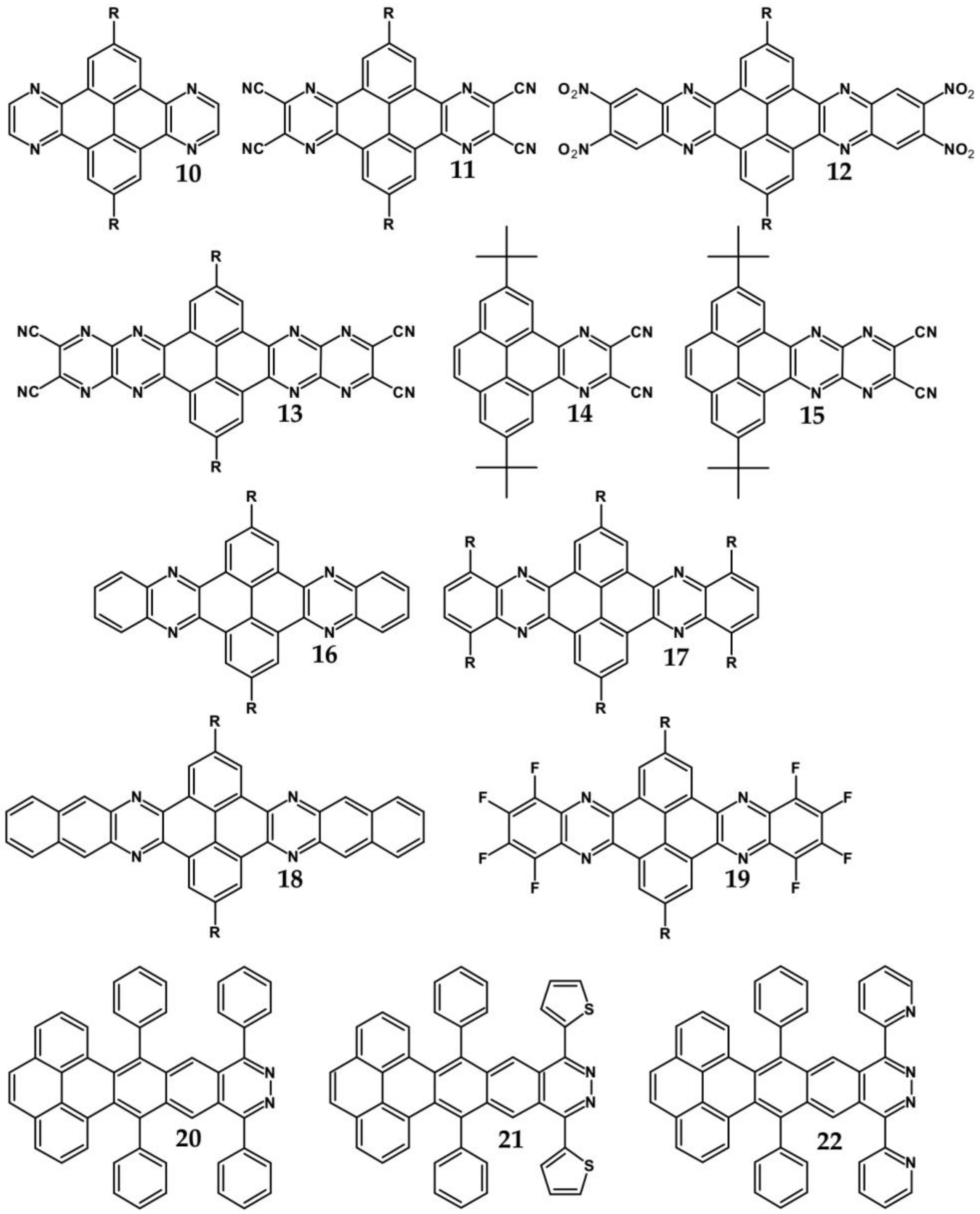
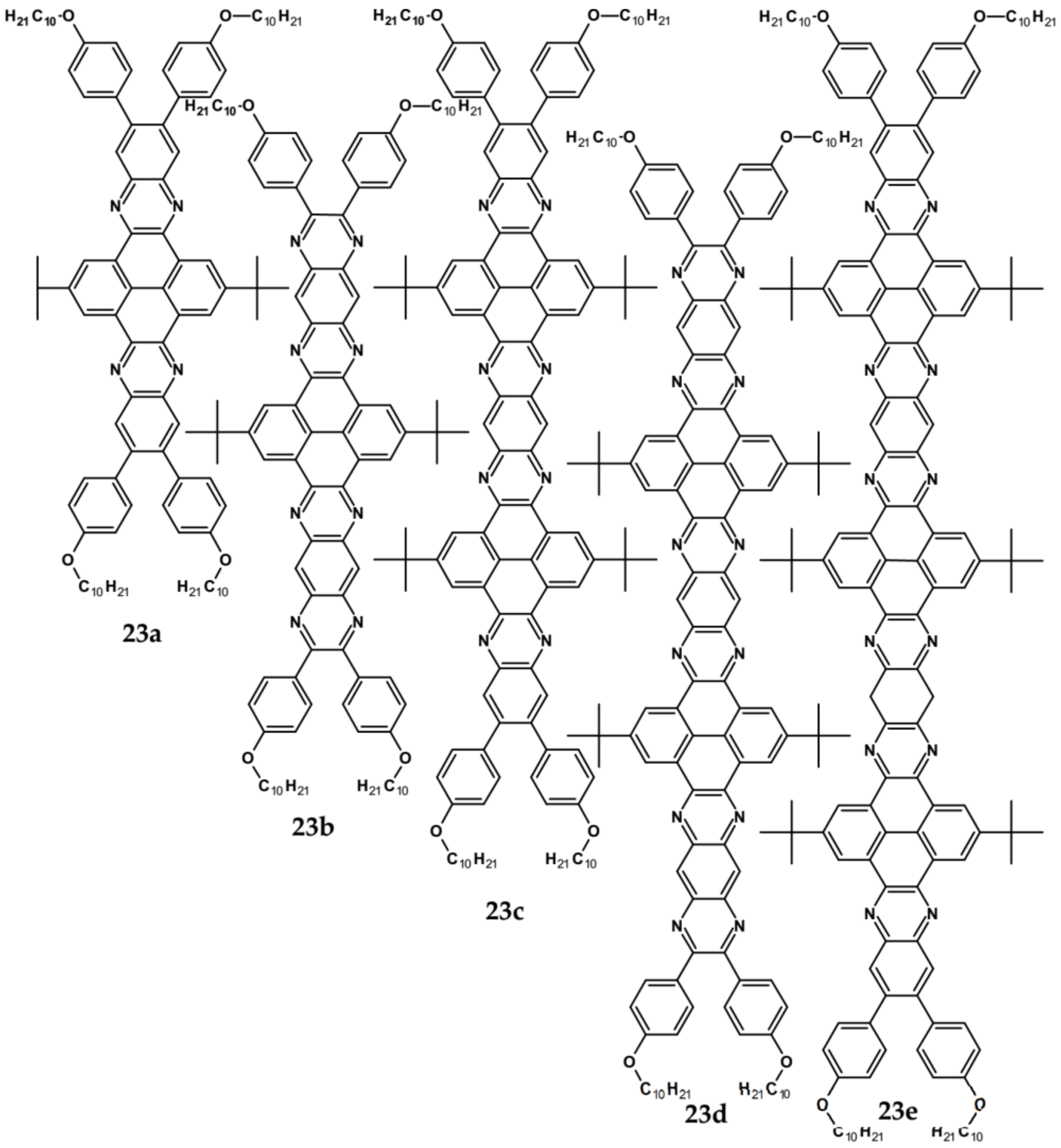
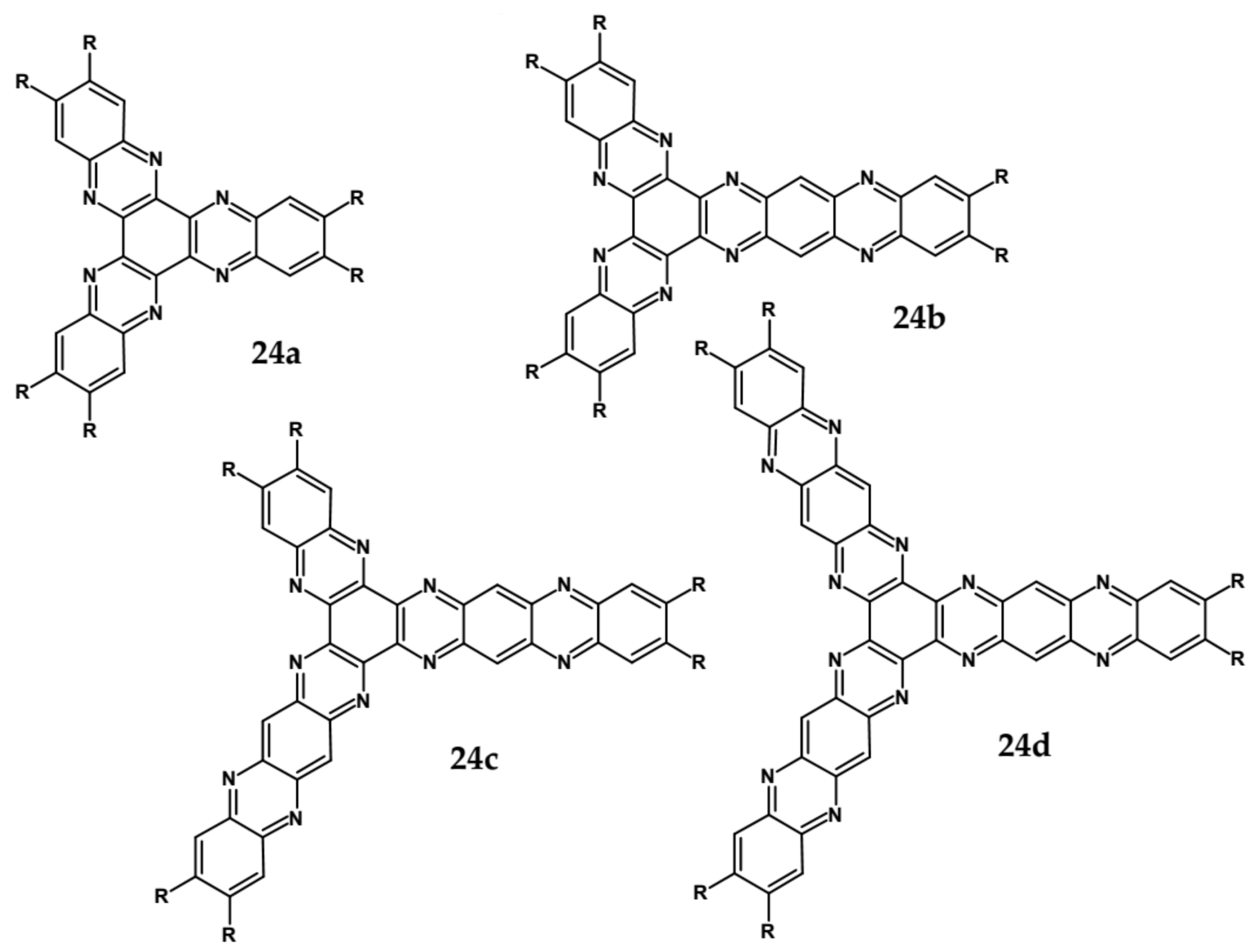


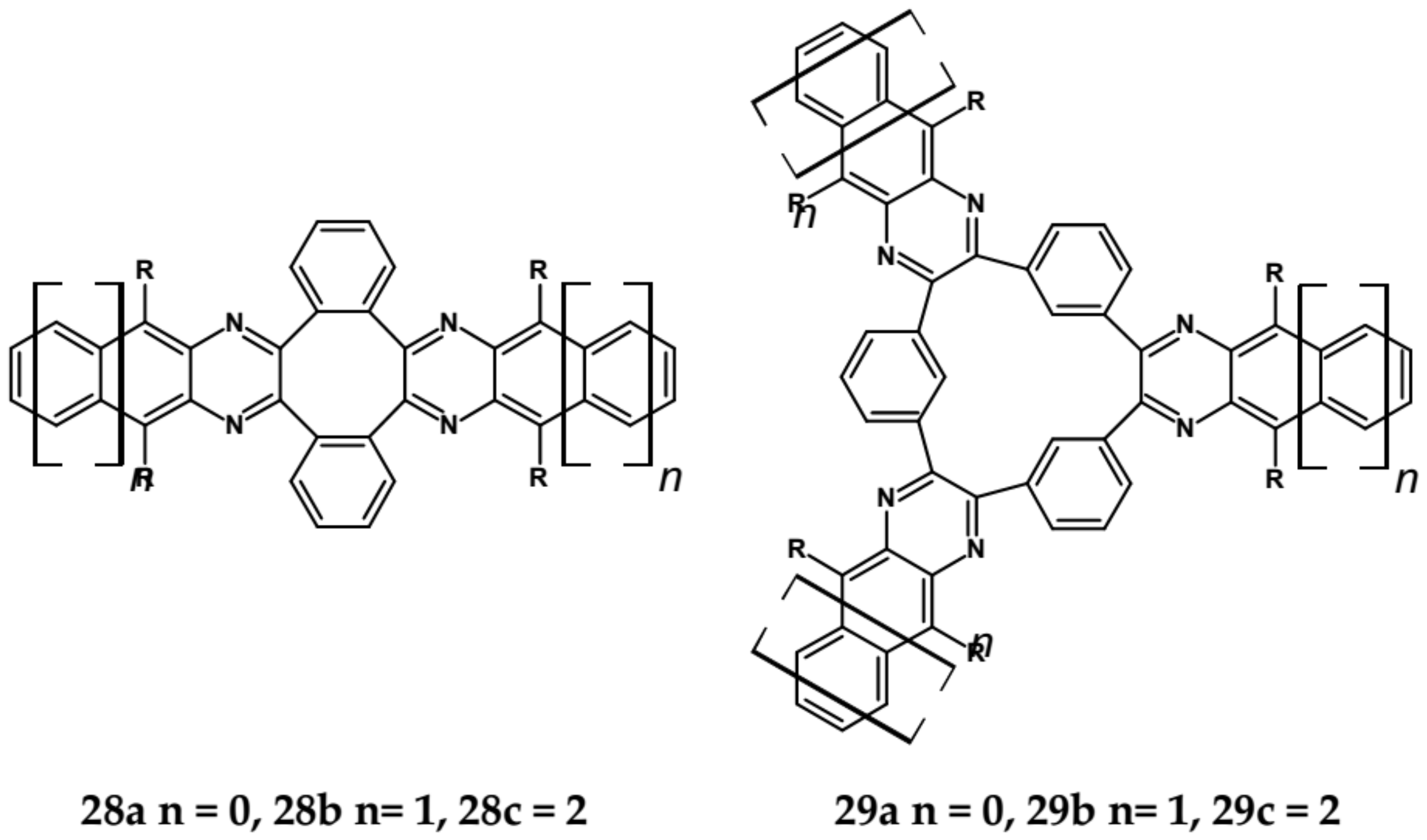
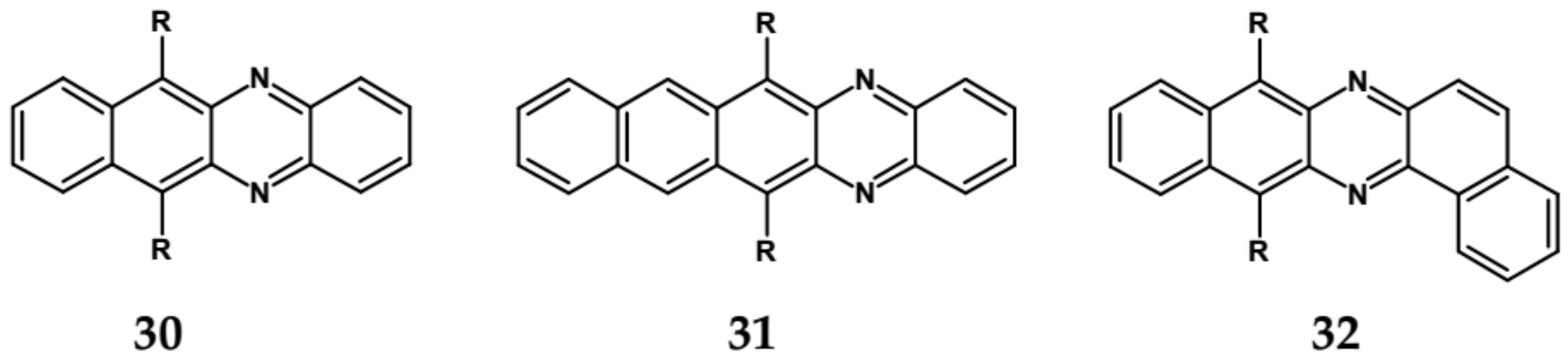
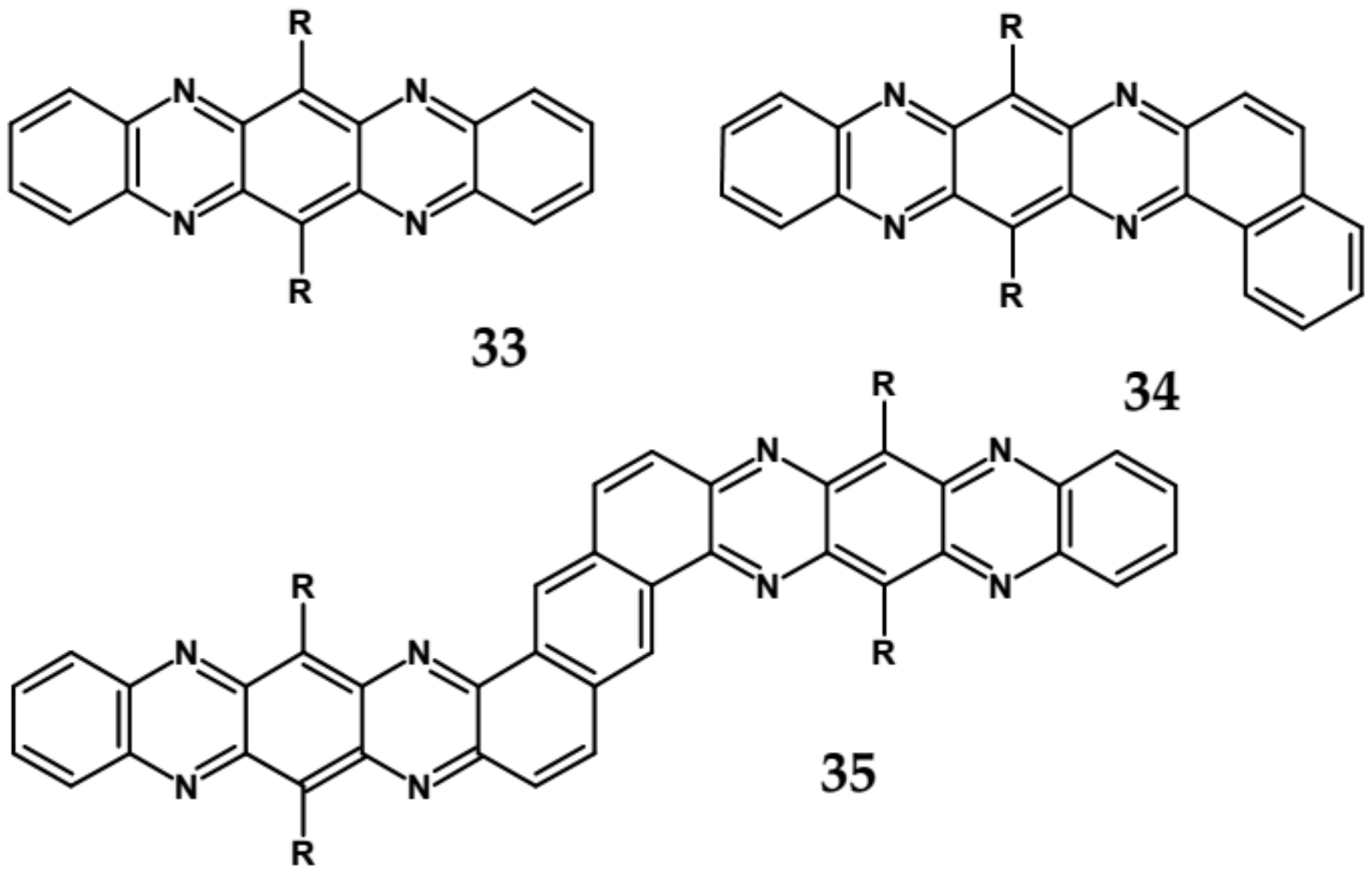
| Compound | E1/2red # vs. Fc/Fc+ [V] | EA * [eV] | Eg opt [eV] |
|---|---|---|---|
| Acenes | |||
| Naphthalene | −3.44 [13] | 0.77 | 4.14 [14] |
| Anthracene | −2.91 [13] | 1.40 | 3.20 [14] |
| Azaacenes | |||
| Quinoline (1N) | −2.77 [15] | 1.56 | 3.94 [16] ** |
| Quinoxaline (2N) | −2.26 [15] | 2.16 | 3.85 [17] ** |
| Acridine (1N) | −2.26 [15] | 2.16 | 3.22 [18] ** |
| Phenazine (2N) | −1.83 [15] | 2.67 | 3.07 [19] ** |
| Pyrazinoquinoxaline (4N) | −1.60 [20] | 2.94 | 2.95 [21] ** |
| Compounds | E1/2red vs. Fc/Fc+ [V] | EA * [eV] | Eg opt [eV] |
|---|---|---|---|
| 1a | −1.1 | 3.53 | 2.38 |
| 1b | −0.8 | 3.89 | 1.84 |
| 1c | −0,7 | 4.00 | 1.49 |
| 2a | −1.1 | 3.53 | 2.20 |
| 2b | −0.8 | 3.89 | 1.69 |
| 2c | −0.48 | 4.26 | 1.35 |
| Compound | λmax abs [nm] | λmax emi [nm] | Stokes Shift [nm] |
|---|---|---|---|
| 3a | 572 | 577 | 5 |
| 3b | 602 | 616 | 14 |
| 3c | 620 | 627 | 7 |
| 4a | 548 | ||
| 4b | 574 | ||
| 4c | 598 | ||
| 5a | 586 | 590 | 4 |
| 5b | 602 | 643 | 44 |
| 5c | 611 | 657 | 42 |
| Compound | Eg opt [nm]/[eV] | Ered vs. Fc/Fc+ [V] | EA * [eV] |
|---|---|---|---|
| 6a | 737/1.68 | −0.60 | 4.12 |
| 6b | 752/1.65 | −0.60 | 4.12 |
| 6c | 744/1.67 | −0.70 | 4.00 |
| 7a | 862/1.44 | −0.55 | 4.18 |
| 7b | 1039/1.19 | −0.48 | 4.26 |
| 7c | 1000/1.24 | −0.53 | 4.20 |
| 8a | 780/1.59 ** | −0.52 | 4.22 |
| 8b | 867/1.43 ** | −0.27 | 4.51 |
| Compounds | λmax abs [nm] | Eg opt [eV] | Eox1 [V] | Eox2 [V] |
|---|---|---|---|---|
| 9a | 413 | 2.60 | 0.26 | 0.48 |
| 9b | 493 | 2.17 | 0.24 | 0.42 |
| 9c | 455 | 2.36 | 0.25 | 0.44 |
| 9d | 560 | 1.88 | 0.22 | 0.37 |
| Compounds | λmax abs [nm] | Eg opt [eV] | Ered vs. Fc/Fc+ [V] | EA *** [eV] | Compounds | λmax abs [nm] | Eg opt [eV] | Ered vs. Fc/Fc+ [V] | EA *** [eV] |
|---|---|---|---|---|---|---|---|---|---|
| 10 | 417 | 2.81 | −0.79 | 3.90 | 19 | 485 | 2.42 | −1.51 * | 3.05 |
| 11 | 435 | 2.76 | −0.76 | 3.93 | 20 | 441 | 2.51 | −1.69 ** | 2.84 |
| 12 | 440 | 2.70 | −0.73 | 3.97 | 21 | 471 | 2.37 | −1.54 ** | 3.01 |
| 13 | 482 | 2.37 | −0.21 | 4.58 | 22 | 464 | 2.41 | −1.62 ** | 2.92 |
| 14 | 455 | 2.09 | −1.42 * | 3.15 | 23a | 443 | 2.65 | −1.68 ** | 2.85 |
| 15 | 571 | 1.85 | −0.71 * | 3.99 | 23b | 500 | 2.34 | −1.22 ** | 3.39 |
| 16 | 421 | 2.88 | −1.73 * | 2.79 | 23c | 525 | 2.25 | −1.19 ** | 3.43 |
| 17 | 436 | 2.69 | −1.57 * | 2.98 | 23d | 529 | 2.22 | −1.16 ** | 3.46 |
| 18 | 425 | 2.78 | −1.44 * | 3.13 | 23e | 538 | 2.18 | −1.14 ** | 3.48 |
| Compounds | λmax abs [nm] | λmax emi [nm] | Eg opt [eV] | Ered vs. Fc/Fc+ [V] | EA * [eV] |
|---|---|---|---|---|---|
| 24a | 471 | 552 | 2.43 | −1.01 | 3.64 |
| 24b | 508 | 593 | 2.23 | −0.71 | 3.99 |
| 24c | 530 | 605 | 2.13 | −0.60 | 4.12 |
| 24d | 543 | 615 | 2.08 | −0.53 | 4.20 |
| 25 | 399 | 504 | 2.95 | −0.91 | 3.90 |
| Compounds | λmax abs [nm] | λmax emi [nm] | Eg opt [eV] | Ered vs. Fc/Fc+ [V] | EA * [eV] |
|---|---|---|---|---|---|
| 26a | 378 | 422 | 2.99 | −1.50 | 3.90 |
| 26b | 497 | 508 | 2.42 | −1.63 | 3.06 |
| 26c | 597 | 604 | 1.98 | −1.50 | 2.91 |
| 26d | 595 | 607 | 2.01 | −1.09 | 3.06 |
| 27a | 369 | 427 | 3.06 | −1.85 | 3.54 |
| 27b | 485 | 497 | 2.47 | −1.56 | 2.65 |
| 27c | 582 | 590 | 2.07 | −1.40 | 2.99 |
| 27d | 586 | 599 | 2.05 | −1.03 | 3.18 |
| Compounds | λmax abs [nm] | λmax emi [nm] | Eg opt [nm]/[eV] | Ered vs. Fc/Fc+ [V] | EA * [eV] |
|---|---|---|---|---|---|
| 28a | 391 | 413 | 409/3.03 | −1.58 | 2.97 |
| 28b | 490 | 500 | 504/2.46 | −1.55 | 3.00 |
| 28c | 589 | 594 | 603/2.06 | −1.33 | 3.26 |
| 29a | 388 | 433 | 411/3.02 | −1.82 | 2.68 |
| 29b | 493 | 506 | 510/2.43 | −1.57 | 2.98 |
| 29c | 596 | 601 | 610/2.03 | −1.34 | 3.25 |
| Compound | λmax abs [nm] | λmax emi [nm] | Eg opt [nm]/[eV] | Ered vs. Fc/Fc+ [V] | EA * [eV] |
|---|---|---|---|---|---|
| 30 | 570 | 577 | 585/2.12 | −1.23 ** | 3.38 |
| 31 | 693 | 699 | 709/1.75 | −1.05 ** | 3.59 |
| 32 | 544 | 553 | 558/2.22 | −1.79 ** | 2.72 |
| 33 | 681 | 694 | −/1.94 | −0.68 | 4.03 |
| 34 | 653 | 699 | −/1.75 | −0.76 | 3.93 |
| 35 | 712 | 688 | −/1.75 | −0.51 | 4.23 |
| Compounds | λmax abs. [nm] | λmax emi [nm] | Eg opt [eV] | Ered vs. Fc/Fc+ [V] | EA [eV] |
|---|---|---|---|---|---|
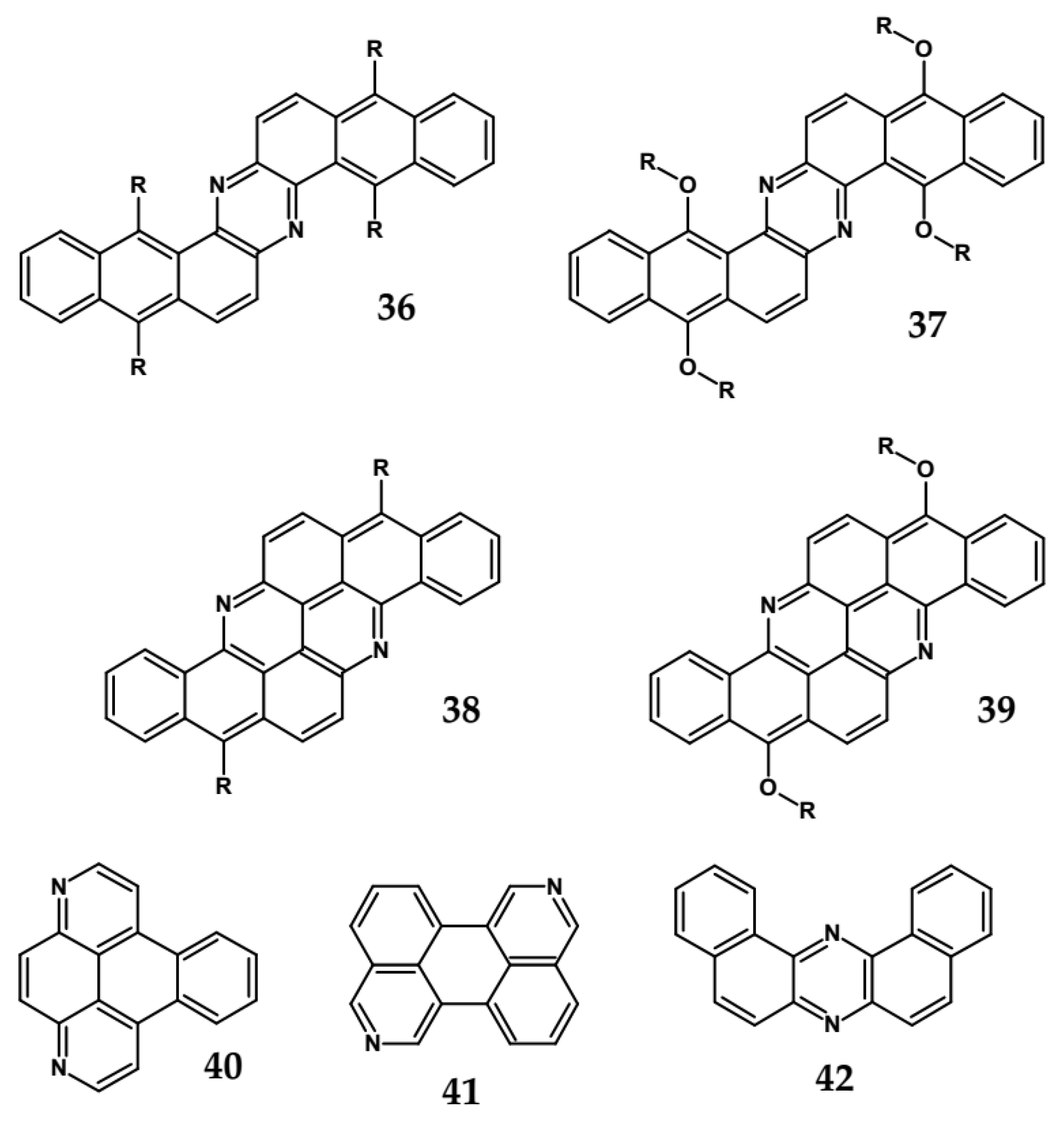
| 36 | 499 | 501 | - | −1.20 | 3.41 |
| 37 | 493 | 515 | 2.40 | −1.63 | 2.91 |
| 38 | 554 | - | 2.23 | - | - |
| 39 | 515 | 524 | 2.33 | −1.62 | 2.92 |
| 40 | 369 | 370 | 4.11 | −2.02 | 2.45 |
| 41 | 423 | 431 | 3.18 | −1.77 | 2.74 |
| 42 | 416 | 425 | - | −1.88 | 2.61 |
| Compound | Device’s Properties |
|---|---|
| Emitters in organic light emitting diode | |
43  | maximum luminance 1000 cd m−2 * efficiency 5 cd A−1 EQE = 0.53%, λmax emi = 540 nm * [93] |
44  | maximum luminance 2321 cd m−2 efficiency 0.79 cd A−1, λmax emi = 547 nm [85] |
45 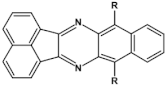 | maximum luminance 5792 cd m−2 * efficiency 2.88 cd A−1 λmax emi = 502 nm [113] |
46  | maximum luminance > 600 cd m−2 * efficiency 5.4 cd A−1 EQE = 7.2%, λmax emi = 640 nm [98] |
47  | maximum luminance > 300 cd m−2 * efficiency 0.8 cd A−1 EQE = 2.0%, λmax emi = 665 nm [98] |
48 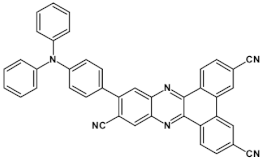 | maximum luminance > 300 cd m−2 * EQE = 22.80%, λmax emi = 698 nm [114] |
49 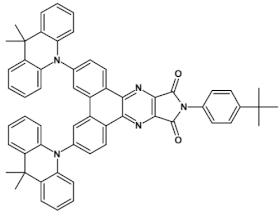 | maximum luminance > 10,000 cd m−2 EQE = 26.00%, [115] |
50  | efficiency 51.8 cd A−1 EQE = 21.8%, λmax emi = 577 nm [116] |
51 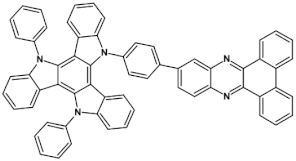 | efficiency 47.1 cd A−1 EQE = 23.8%, λmax emi = 587 nm [116] |
52  | maximum luminance > 6000 cd m−2 * EQE = 19.4%, λmax emi = 580nm [97] |
53  | maximum luminance > 10,000 cd m−2 * EQE = 22.1%, λmax emi = 547 nm [97] |
54 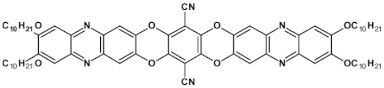 | maximum luminance > 1000 cd m−2 * EQE = 3.3%, λmax emi = 536 nm phosphorescence [33] |
55  | maximum luminance 8600 cd m−2, EQE = 0.33%, λmax emi = 511 nm [117] |
56 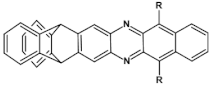 | maximum luminance > 200 cd m−2, efficiency 1 cd A−1, λmax emi = 590 nm [70] |
57  | maximum luminance 593 cd m−2 *, efficiency 0.85 cd A−1, λmax emi = 502 nm [118] |
58  | maximum luminance 20780 cd m−2, EQE = 3.1%, λmax emi = 500 nm [119] |
59 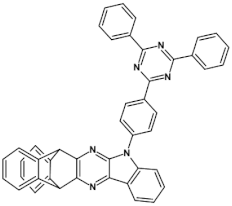 | maximum luminance > 500 cd m−2 *, efficiency 11.6 cd A−1, EQE = 10.4%, λmax emi = 456 nm [120] |
60 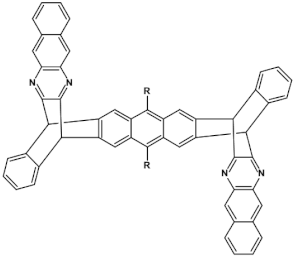 | maximum luminance 920 cd m−2 *, efficiency 80 cd A−1, EQR = 0.53%, λmax emi = 495 nm [31] |
61 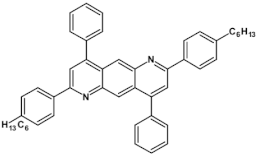 | maximum luminance 595 cd m−2 *, efficiency 7.0 cd A−1, EQE = 2.0%, λmax emi = 520 nm* [15] |
62 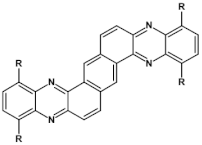 | maximum luminance 80 cd m−2, λmax emi = 508 nm [89] |
63 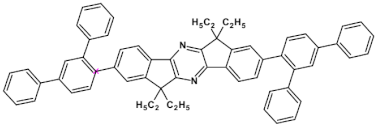 | efficiency 2.13 cd A−1, EQE = 4.61%, λmax emi = 440 nm [121] |
64 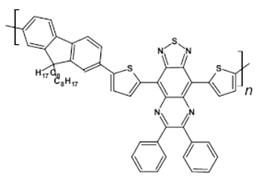 | EQE = 0.05%, λmax emi = 970 nm [122] |
| Acceptors for solar cells | |
65  | VOC = 0.81 V, FF = 37.3%, JSC = −6,77 mA cm2 PCE = 2.03% [101] |
66 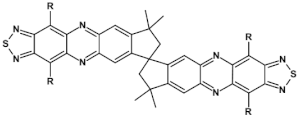 | VOC = 0.75 V, FF = 38.3%, JSC = −5.66 mA cm2, PCE = 1.62% [35] |
67 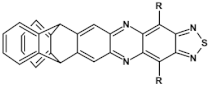 | VOC = 0.80 V, FF = 41%, JSC = −6,6 mA cm2, PCE = 2.19% [37] |
68 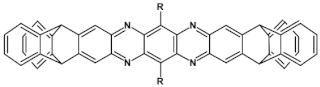 | VOC = 0.85 V, FF = 36%, JSC = −7.51 mA cm2, PCE = 2.28% [36] |
| Electron Transporting Layers for Perovskite Solar Cells | |
69 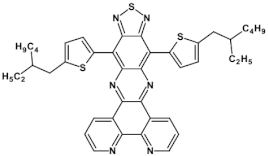 | μe = 2.8 × 10−4 cm2 V−1 s−1, μe = 2.7 × 10−3 cm2 V−1 s−1 (100 °C) [104] |
70 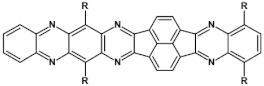 | μe = 4.7 × 10−4 cm2 V−1 s−1 [105] |
71 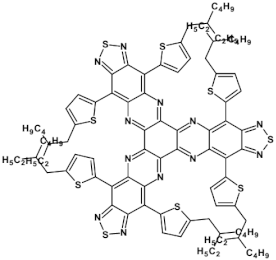 | μe = 1.73 × 10−2 cm2 V−1 s−1 [106] |
72 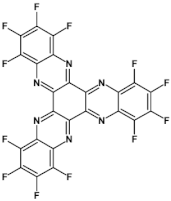 | μe = 1.9 × 10−4 cm2 V−1 s−1 [123] |
73 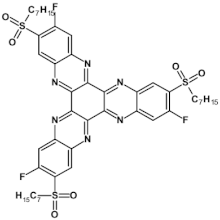 | μe = 5.13 × 10−3 cm2 V−1 s−1 [124] |
74 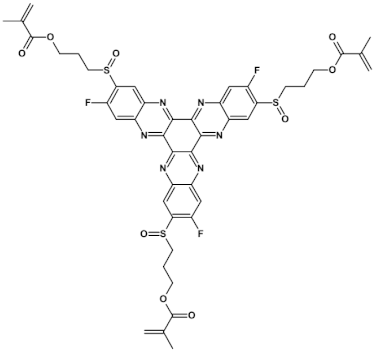 | μe = 1.68 × 10−3 cm2 V−1 s−1 (doped by Et3N) [125] |
| Active layers in organic field effect transistors | |
75 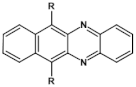 | μe = 7 × 10−3 cm2 V−1 s−1 [69] |
76 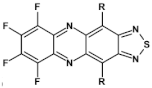 | μe = 7 × 10−2 cm2 V−1 s−1 [126] |
77  | μe = 9 × 10−4 cm2 V−1 s−1 [127] |
78  | μe = 0.14 cm2 V−1 s−1, μh = 0.12 cm2 V−1 s−1 [109] |
79 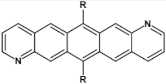 | μe = 0.15 cm2 V−1 s−1, μh = 0.11 cm2 V−1 s−1 [108] |
80 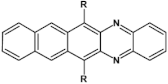 | μe = 0.05 cm2 V−1 s−1, μh = 4 × 10−4 cm2 V−1 s−1 [107] |
81 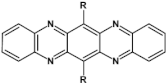 | μe = from 1 do 3.3 cm2 V−1 s−1 (depend of temperatures of substrate—from 25 °C to 100 °C) [107]μe = 11 cm2 V−1 s−1 (measured in vacuum) [128] |
82 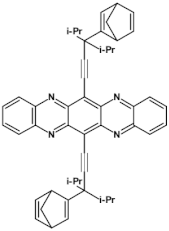 | μe = 3.5 × 10−4 cm2 V−1 s−1 [129] |
83 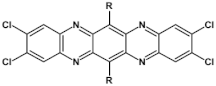 | μe = 27.8 cm2 V−1 s−1 (measured in vacuum) [111] |
84 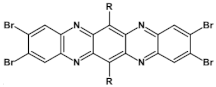 | μe = 0.56 cm2 V−1 s−1 [130] |
85 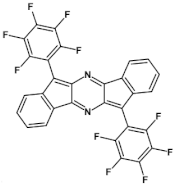 | μe = 3.7 × 10−2 cm2 V−1 s−1 [131] |
86  | μh = 0.06 cm2 V−1 s−1 [110] |
87  | μh = 8.1 × 10−3 cm2 V−1 s−1 [132] |
88 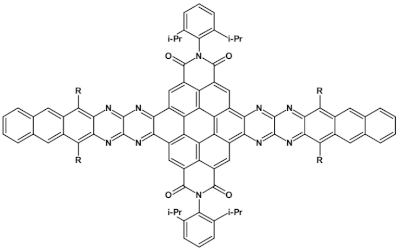 | μe = 8.1 × 10−4 cm2 V−1 s−1, μh = 2 × 10−4 cm2 V−1 s−1 [133] |
| Self-assembled monolayers (SAM) for functionalization of gold electrodes for organic field effect transistors | |
89 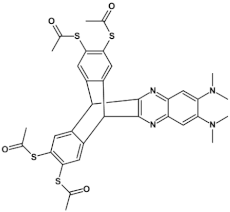 | on/off ratio increased from 102 to 104, μe = from 10−3 cm2 V−1 s−1 to 10−2 cm2 V−1 s−1 [134] |
| Photocatode | |
90  | current density 0.13 mA cm−2 at −0.13 V (Evs RHE) [135] |
91  | Detected [112] |
| Memory device | |
92 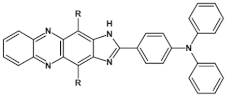 | “ON” state a 2.25 V, “OFF” state −0.95 V, 14 cycles of working [136] |
93 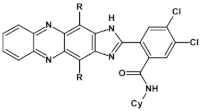 | “ON” state a ~3.00 V, “OFF” state −1.65 V *, [137] |
94 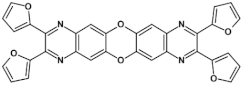 | “ON” state a ~2.2 V, “OFF” state −1.2 V * [138] |
95  | “ON” state a ~1.8 V, “OFF” state −2.0 V * [139] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kotwica, K.; Wielgus, I.; Proń, A. Azaacenes Based Electroactive Materials: Preparation, Structure, Electrochemistry, Spectroscopy and Applications—A Critical Review. Materials 2021, 14, 5155. https://doi.org/10.3390/ma14185155
Kotwica K, Wielgus I, Proń A. Azaacenes Based Electroactive Materials: Preparation, Structure, Electrochemistry, Spectroscopy and Applications—A Critical Review. Materials. 2021; 14(18):5155. https://doi.org/10.3390/ma14185155
Chicago/Turabian StyleKotwica, Kamil, Ireneusz Wielgus, and Adam Proń. 2021. "Azaacenes Based Electroactive Materials: Preparation, Structure, Electrochemistry, Spectroscopy and Applications—A Critical Review" Materials 14, no. 18: 5155. https://doi.org/10.3390/ma14185155
APA StyleKotwica, K., Wielgus, I., & Proń, A. (2021). Azaacenes Based Electroactive Materials: Preparation, Structure, Electrochemistry, Spectroscopy and Applications—A Critical Review. Materials, 14(18), 5155. https://doi.org/10.3390/ma14185155






