Nanocarrier-Mediated Topical Insulin Delivery for Wound Healing
Abstract
1. Introduction
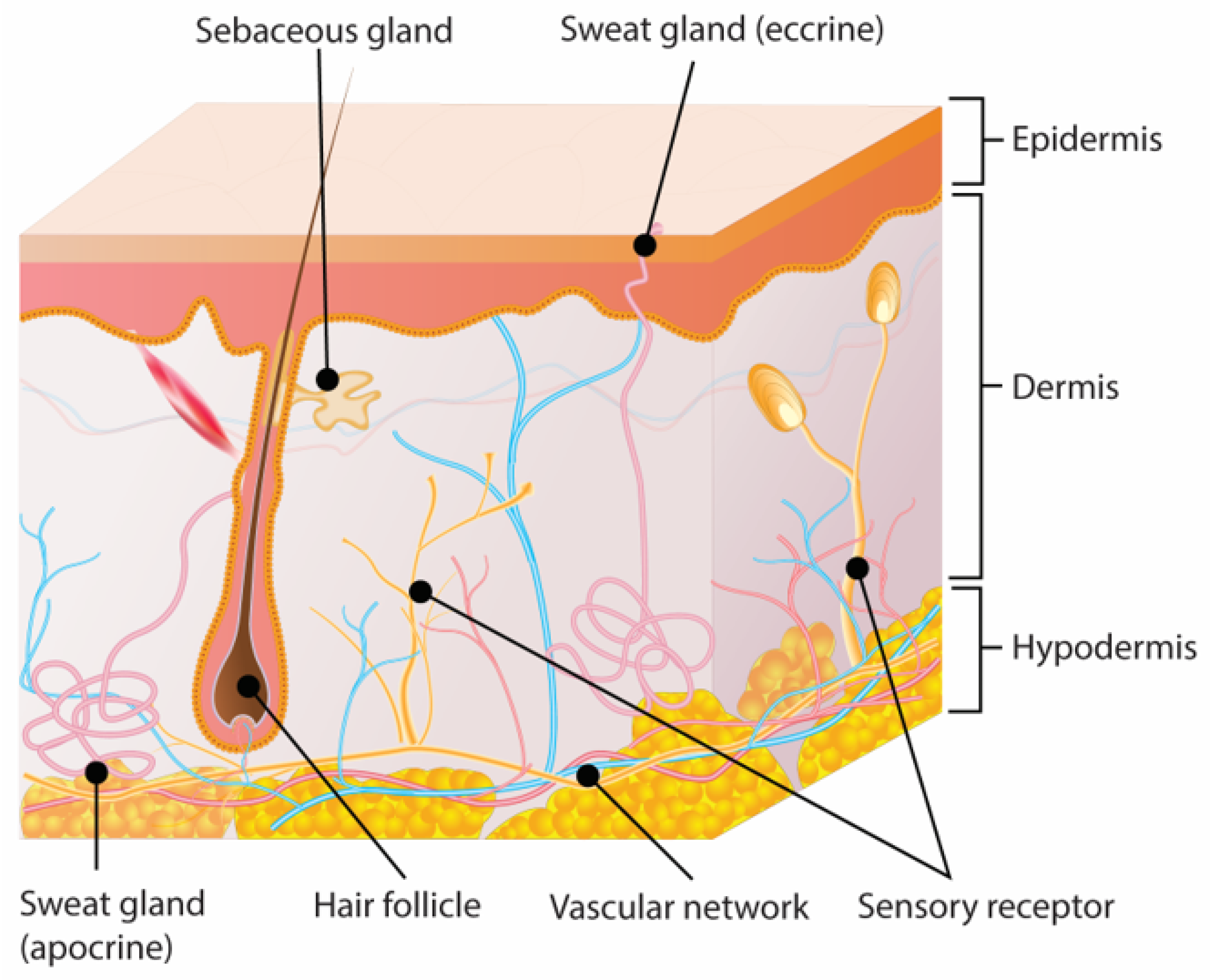
2. Anatomophysiology of Skin
2.1. Healthy Skin
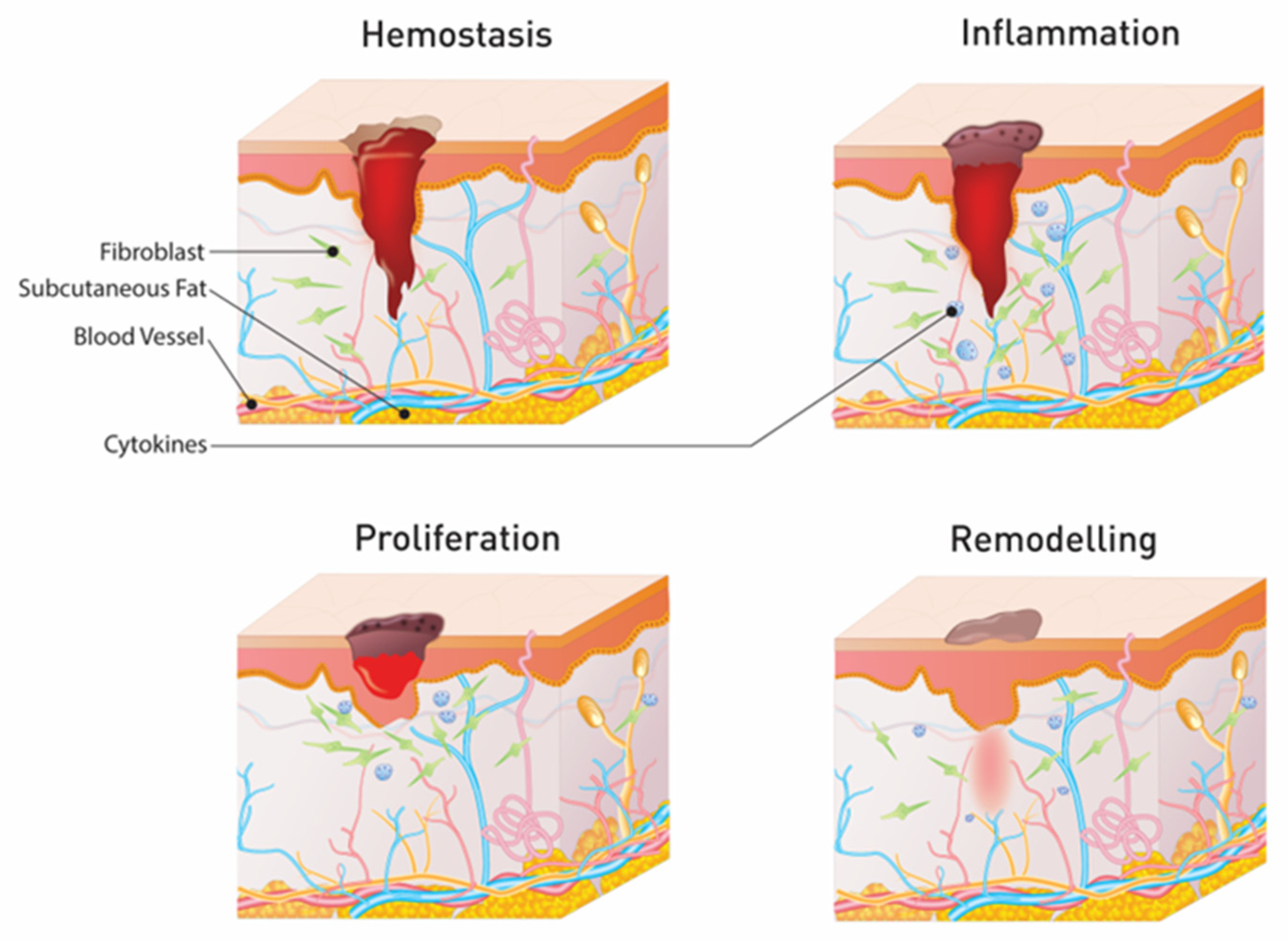
2.2. Skin Wounds
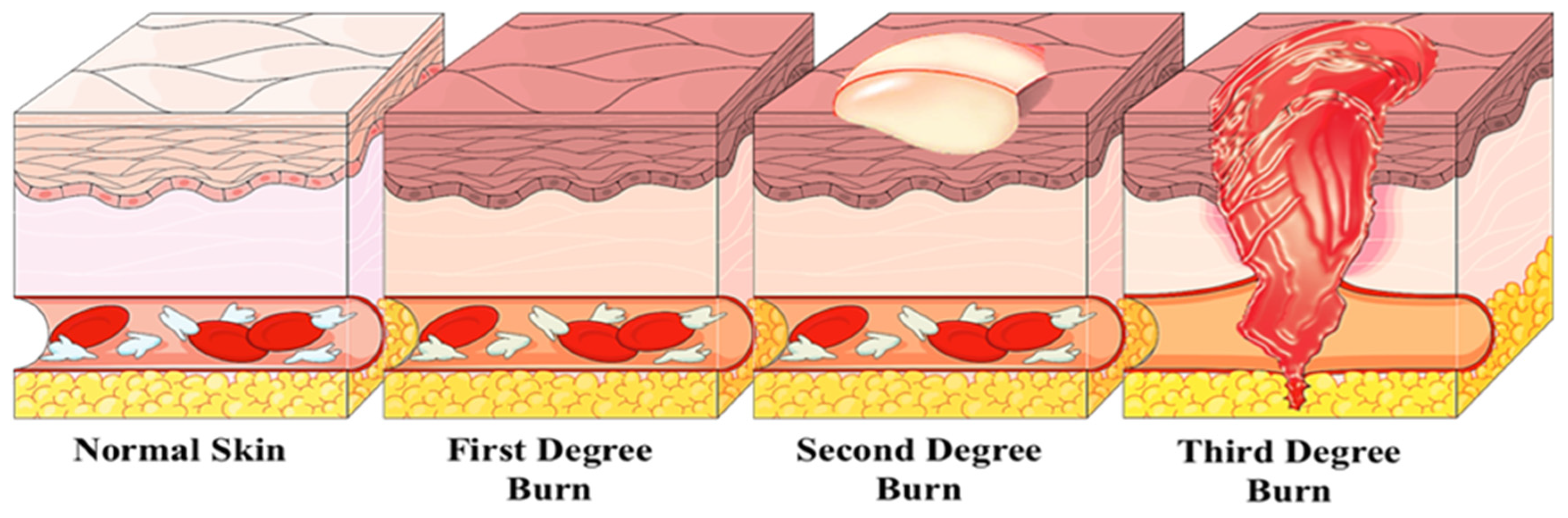
2.3. Burns
3. Effect of Insulin in Wound Healing
3.1. Diabetic Wounds
3.2. Non-Diabetic Wounds
3.3. Burns
4. Advantages of Nanocarriers for Topical Insulin Delivery
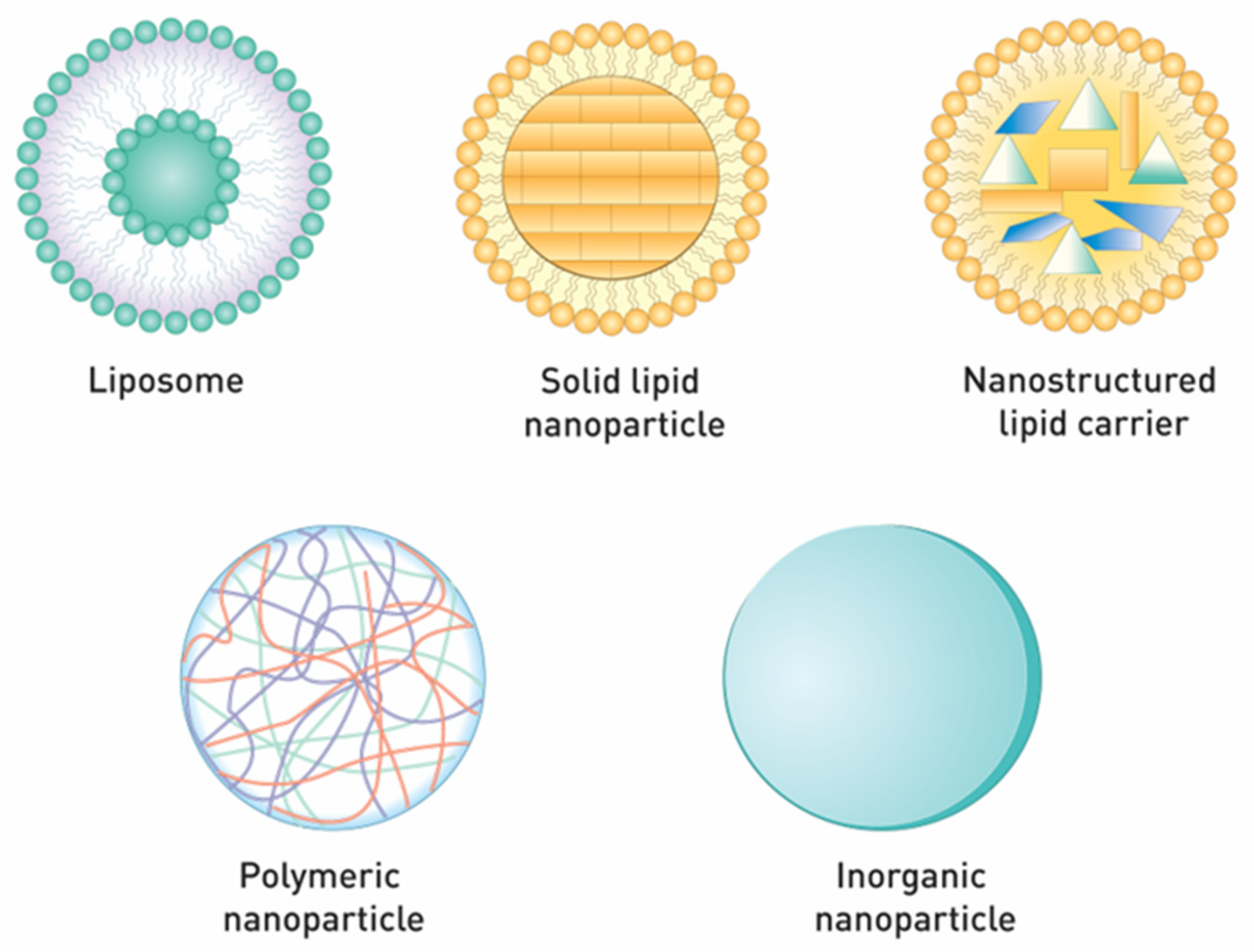
5. Nanocarrier Systems for Topical Insulin Delivery
5.1. Lipid-Based Nanoparticles
5.1.1. Liposomes
5.1.2. Solid Lipid Nanoparticles and Nanostructured Lipid Carriers
5.2. Polymer Nanoparticles
5.2.1. Natural Polymers
5.2.2. Synthetic Polymers
5.3. Inorganic Nanoparticles
6. Pipeline Products
7. Toxicity Concerns
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Frykberg, R.G.; Banks, J. Challenges in the treatment of chronic wounds. Adv. Wound Care 2015, 4, 560–582. [Google Scholar] [CrossRef]
- Menke, N.B.; Ward, K.R.; Witten, T.M.; Bonchev, D.G.; Diegelmann, R.F. Impaired wound healing. Clin. Dermatol. 2007, 25, 19–25. [Google Scholar] [CrossRef]
- Besson, J.C.F.; Hernandes, L.; Campos, J.M.; Morikawa, K.A.; Bersani-Amado, C.A.; Matioli, G. Insulin complexed with cyclodextrins stimulates epithelialization and neovascularization of skin wound healing in rats. Injury 2017, 48, 2417–2425. [Google Scholar] [CrossRef] [PubMed]
- Lai-Cheong, J.E.; McGrath, J.A. Structure and function of skin, hair and nails. Medicine 2013, 41, 317–320. [Google Scholar] [CrossRef]
- Gallo, R.L.; Nizet, V. Innate barriers against skin infection and associated disorders. Drug Discov. Today Dis. Mech. 2008, 5, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Sen, C.K.; Gordillo, G.M.; Roy, S.; Kirsner, R.; Lambert, L.; Hunt, T.K.; Gottrup, F.; Gurtner, G.C.; Longaker, M.T. Human skin wounds: A major and snowballing threat to public health and the economy. Wound Repair. Regen. 2009, 17, 763–771. [Google Scholar] [CrossRef]
- Menon, G.K.; Cleary, G.W.; Lane, M.E. The structure and function of the stratum corneum. Int. J. Pharm. 2012, 435, 3–9. [Google Scholar] [CrossRef]
- Salmon, J.K.; Armstrong, C.A.; Ansel, J.C. The skin as an immune organ. West. J. Med. 1994, 160, 146. [Google Scholar]
- Jarrett, A. The physiology of the skin. Practitioner 1969, 202, 12–22. [Google Scholar]
- Powell, J. Skin physiology. Found. Years 2007, 3, 193–196. [Google Scholar] [CrossRef]
- Gay, S.; Vijanto, J.; Raekallio, J.; Penttinen, R. Collagen types in early phases of wound healing in children. Acta Chir. Scand. 1978, 144, 205–211. [Google Scholar]
- Lee, D.Y.; Cho, K.H. The effects of epidermal keratinocytes and dermal fibroblasts on the formation of cutaneous basement membrane in three-dimensional culture systems. Arch. Dermatol. Res. 2005, 296, 296–302. [Google Scholar] [CrossRef]
- Gaboriau, H.P.; Murakami, C.S. Skin anatomy and flap physiology. Otolaryngol. Clin. N. Am. 2001, 34, 555–569. [Google Scholar] [CrossRef]
- Flanagan, M. Wound Healing and Skin Integrity: Principles and Practice; John Wiley and Sons Ltd.: Hoboken, NJ, USA, 2013. [Google Scholar]
- Broughton, G., 2nd; Janis, J.E.; Attinger, C.E. The basic science of wound healing. Plast. Reconstr. Surg. 2006, 117, 12–34. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Young, A.; McNaught, C.-E. The physiology of wound healing. Surgery (Oxford) 2017, 35, 473–477. [Google Scholar] [CrossRef]
- Bhushan, M.; Young, H.S.; Brenchley, P.E.; Griffiths, C.E. Recent advances in cutaneous angiogenesis. Br. J. Dermatol. 2002, 147, 418–425. [Google Scholar] [CrossRef] [PubMed]
- Baum, C.L.; Arpey, C.J. Normal cutaneous wound healing: Clinical correlation with cellular and molecular events. Dermatol. Surg. 2005, 31, 674–686. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, S.R.; Diegelmann, R.F. Wound healing primer. Surg. Clin. N. Am. 2010, 90, 1133–1146. [Google Scholar] [CrossRef]
- Velnar, T.; Bailey, T.; Smrkolj, V. The wound healing process: An overview of the cellular and molecular mechanisms. J. Int. Med. Res. 2009, 37, 1528–1542. [Google Scholar] [CrossRef]
- Eming, S.A.; Martin, P.; Tomic-Canic, M. Wound repair and regeneration: Mechanisms, signaling, and translation. Sci. Transl. Med. 2014, 6, 265sr6. [Google Scholar] [CrossRef]
- Xue, M.; Jackson, C.J. Extracellular matrix reorganization during wound healing and its impact on abnormal scarring. Adv. Wound Care 2015, 4, 119–136. [Google Scholar] [CrossRef]
- Das, S.; Baker, A.B. Biomaterials and nanotherapeutics for enhancing skin wound healing. Front. Bioeng. Biotechnol. 2016, 4, 82. [Google Scholar] [CrossRef] [PubMed]
- Reger, S.I.; Ranganathan, V.K.; Sahgal, V. Support surface interface pressure, microenvironment, and the prevalence of pressure ulcers: An analysis of the literature. Ostomy Wound Manag. 2007, 53, 50–58. [Google Scholar]
- Reger, S.I.; Ranganathan, V.K. The importance of the microenvironment of support surfaces in the prevalence of pressure ulcers. In Bioengineering Research of Chronic Wounds: A Multidisciplinary Study Approach; Gefen, A., Ed.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 85–100. [Google Scholar]
- Smith, D.M.; Snow, D.E.; Rees, E.; Zischkau, A.M.; Hanson, J.D.; Wolcott, R.D.; Sun, Y.; White, J.; Kumar, S.; Dowd, S.E. Evaluation of the bacterial diversity of pressure ulcers using bTEFAP pyrosequencing. BMC Med. Genom. 2010, 3, 41. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, M.L.B.; Jansen, R.B.; Wilbek Fabricius, T.; Jørgensen, B.; Svendsen, O.L. Healing of diabetic foot ulcers in patients treated at the Copenhagen Wound Healing Center in 1999/2000 and in 2011/2012. J. Diabetes Res. 2019, 2019. [Google Scholar] [CrossRef]
- Falanga, V. Wound healing and its impairment in the diabetic foot. Lancet 2005, 366, 1736–1743. [Google Scholar] [CrossRef]
- Banu, A.; Noorul Hassan, M.M.; Rajkumar, J.; Srinivasa, S. Spectrum of bacteria associated with diabetic foot ulcer and biofilm formation: A prospective study. Australas. Med. J. 2015, 8, 280–285. [Google Scholar] [CrossRef]
- Thomas, P.R.; Nash, G.B.; Dormandy, J.A. White cell accumulation in dependent legs of patients with venous hypertension: A possible mechanism for trophic changes in the skin. Br. Med. J. (Clin. Res. Ed.) 1988, 296, 1693–1695. [Google Scholar] [CrossRef]
- Pugliese, D.J. Infection in venous leg ulcers: Considerations for optimal management in the elderly. Drugs Aging 2016, 33, 87–96. [Google Scholar] [CrossRef]
- Johnson, C. Management of burns. Surgery (Oxford) 2018, 36, 435–440. [Google Scholar] [CrossRef]
- Church, D.; Elsayed, S.; Reid, O.; Winston, B.; Lindsay, R. Burn wound infections. Clin. Microbiol. Rev. 2006, 19, 403–434. [Google Scholar] [CrossRef] [PubMed]
- Dissanaike, S.; Ha, D.; Mitchell, D.; Larumbe, E. Socioeconomic status, gender, and burn injury: A retrospective review. Am. J. Surg. 2017, 214, 677–681. [Google Scholar] [CrossRef]
- Dhall, S.; Silva, J.P.; Liu, Y.; Hrynyk, M.; Garcia, M.; Chan, A.; Lyubovitsky, J.; Neufeld, R.J.; Martins-Green, M. Release of insulin from PLGA-alginate dressing stimulates regenerative healing of burn wounds in rats. Clin. Sci. 2015, 129, 1115–1129. [Google Scholar] [CrossRef] [PubMed]
- Hettiaratchy, S.; Dziewulski, P. ABC of burns. Introduction. BMJ 2004, 328, 1366–1368. [Google Scholar] [CrossRef]
- Garner, W.L.; Magee, W. Acute burn injury. Clin. Plast. Surg. 2005, 32, 187–193. [Google Scholar] [CrossRef]
- Nielson, C.B.; Duethman, N.C.; Howard, J.M.; Moncure, M.; Wood, J.G. Burns: Pathophysiology of systemic complications and current management. J. Burn. Care Res. Off. Publ. Am. Burn. Assoc. 2017, 38, 469–481. [Google Scholar] [CrossRef]
- Auger, C.; Samadi, O.; Jeschke, M.G. The biochemical alterations underlying post-burn hypermetabolism. Biochimica et biophysica acta. Mol. Basis Dis. 2017, 1863, 2633–2644. [Google Scholar] [CrossRef] [PubMed]
- Gary Sibbald, R.; Woo, K.Y. The biology of chronic foot ulcers in persons with diabetes. Diabetes Metab. Res. Rev. 2008, 24 (Suppl. 1), 25–30. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.; Srivastava, S.; Singh, M.R.; Singh, D. Mechanistic insight into diabetic wounds: Pathogenesis, molecular targets and treatment strategies to pace wound healing. Biomed. Pharmacother. Biomed. Pharmacother. 2019, 112, 108615. [Google Scholar] [CrossRef]
- May, K. Preventing foot ulcers. Aust. Prescr. 2008, 31, 94–96. [Google Scholar] [CrossRef]
- Rosenthal, S.P. Acceleration of primary wound healing by insulin. Arch. Surg. 1968, 96, 53–55. [Google Scholar] [CrossRef]
- Thalhimer, W. Insulin treatment of postoperative (nondiabetic) acidosis. J. Am. Med. Assoc. 1923, 81, 383–385. [Google Scholar] [CrossRef]
- Foster, N.B. Diabetic coma. J. Am. Med. Assoc. 1925, 84, 719–722. [Google Scholar] [CrossRef]
- Weringer, E.J.; Kelso, J.M.; Tamai, I.Y.; Arquilla, E.R. Effects of insulin on wound healing in diabetic mice. Acta Endocrinol. 1982, 99, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Lima, M.H.; Caricilli, A.M.; de Abreu, L.L.; Araújo, E.P.; Pelegrinelli, F.F.; Thirone, A.C.; Tsukumo, D.M.; Pessoa, A.F.; dos Santos, M.F.; de Moraes, M.A.; et al. Topical insulin accelerates wound healing in diabetes by enhancing the AKT and ERK pathways: A double-blind placebo-controlled clinical trial. PLoS ONE 2012, 7, e36974. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Liu, Y.; Zhang, J.; You, R.; Qu, J.; Li, M. Functionalized silk fibroin dressing with topical bioactive insulin release for accelerated chronic wound healing. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 72, 394–404. [Google Scholar] [CrossRef]
- Rohrmann, A.; Young, D.; Abbas, Z.; Kish, B.; Robinson, B. Profile of diabetic and non-diabetic patients treated for wounds or ulcers. Foot Care Can. J. Diabetes 2013, 1, 42–47. [Google Scholar]
- Joseph, B. Insulin in the treatment of non-diabetic bed sores. Ann. Surg. 1930, 92, 318. [Google Scholar] [CrossRef]
- Udupa, K.N.; Chansouria, J.P. The role of protamine zinc insulin in accelerating wound healing in the rat. Br. J. Surg. 1971, 58, 673–675. [Google Scholar] [CrossRef]
- Wilson, J.M.; Baines, R.; Babu, E.D.; Kelley, C.J. A role for topical insulin in the management problematic surgical wounds. Ann. R. Coll. Surg. Engl. 2008, 90, 160. [Google Scholar] [CrossRef][Green Version]
- Zhang, X.J.; Chinkes, D.L.; Sadagopa Ramanujam, V.M.; Wolfe, R.R. Local injection of insulin-zinc stimulates DNA synthesis in skin donor site wound. Wound Repair Regen. 2007, 15, 258–265. [Google Scholar] [CrossRef]
- Liu, Y.; Petreaca, M.; Yao, M.; Martins-Green, M. Cell and molecular mechanisms of keratinocyte function stimulated by insulin during wound healing. BMC Cell Biol. 2009, 10, 1–15. [Google Scholar] [CrossRef]
- Rowan, M.P.; Cancio, L.C.; Elster, E.A.; Burmeister, D.M.; Rose, L.F.; Natesan, S.; Chan, R.K.; Christy, R.J.; Chung, K.K. Burn wound healing and treatment: Review and advancements. Crit. Care 2015, 19, 243. [Google Scholar] [CrossRef] [PubMed]
- Sakurai, Y.; Aarsland, A.; Herndon, D.N.; Chinkes, D.L.; Pierre, E.; Nguyen, T.T.; Patterson, B.W.; Wolfe, R.R. Stimulation of muscle protein synthesis by long-term insulin infusion in severely burned patients. Ann. Surg. 1995, 222, 283. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.J.; Chinkes, D.L.; Wolf, S.E.; Wolfe, R.R. Insulin but not growth hormone stimulates protein anabolism in skin wound and muscle. Am. J. Physiol. 1999, 276, E712–E720. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.J.; Chinkes, D.L.; Irtun, O.; Wolfe, R.R. Anabolic action of insulin on skin wound protein is augmented by exogenous amino acids. Am. J. Physiol. 2002, 282, 1308–1315. [Google Scholar] [CrossRef] [PubMed]
- Van den Berghe, G.; Wouters, P.; Weekers, F.; Verwaest, C.; Bruyninckx, F.; Schetz, M.; Vlasselaers, D.; Ferdinande, P.; Lauwers, P.; Bouillon, R. Intensive insulin therapy in critically Ill patients. N. Engl. J. Med. 2001, 345, 1359–1367. [Google Scholar] [CrossRef] [PubMed]
- Jeschke, M.G.; Boehning, D.F.; Finnerty, C.C.; Herndon, D.N. Effect of insulin on the inflammatory and acute phase response after burn injury. Crit. Care Med. 2007, 35, 519–523. [Google Scholar] [CrossRef]
- Jeschke, M.G.; Kulp, G.A.; Kraft, R.; Finnerty, C.C.; Mlcak, R.; Lee, J.O.; Herndon, D.N. Intensive insulin therapy in severely burned pediatric patients: A prospective randomized trial. Am. J. Respir. Crit. Care Med. 2010, 182, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Jeschke, M.G.; Finnerty, C.C.; Herndon, D.N.; Song, J.; Boehning, D.; Tompkins, R.G.; Baker, H.V.; Gauglitz, G.G. Severe injury is associated with insulin resistance, endoplasmic reticulum stress response, and unfolded protein response. Ann. Surg. 2012, 255, 370–378. [Google Scholar] [CrossRef]
- Kabalak, A.A.; Ceylan, A.; Vural, Ç. Effect of insulin treatment to systemic inflammatory response in burn injury. N. Engl. J. Med. 2013, 30, 191–194. [Google Scholar]
- Fram, R.Y.; Cree, M.G.; Wolfe, R.R.; Mlcak, R.P.; Qian, T.; Chinkes, D.L.; Herndon, D.N. Intensive insulin therapy improves insulin sensitivity and mitochondrial function in severely burned children. Crit. Care Med. 2010, 38, 1475–1483. [Google Scholar] [CrossRef]
- Tuvdendorj, D.; Zhang, X.J.; Chinkes, D.L.; Aarsland, A.; Kulp, G.A.; Jeschke, M.G.; Herndon, D.N. Intensive insulin treatment increases donor site wound protein synthesis in burn patients. Surgery 2011, 149, 512–518. [Google Scholar] [CrossRef][Green Version]
- Azevedo, F.; Pessoa, A.; Moreira, G.; Dos Santos, M.; Liberti, E.; Araujo, E.; Carvalho, C.; Saad, M.; Lima, M.H. Effect of topical insulin on second-degree burns in diabetic rats. Biol. Res. Nurs. 2016, 18, 181–192. [Google Scholar] [CrossRef]
- Chen, R.R.; Mooney, D.J. Polymeric growth factor delivery strategies for tissue engineering. Pharm. Res. 2003, 20, 1103–1112. [Google Scholar] [CrossRef]
- Barrientos, S.; Brem, H.; Stojadinovic, O.; Tomic-Canic, M. Clinical application of growth factors and cytokines in wound healing. Wound Repair Regen. 2014, 22, 569–578. [Google Scholar] [CrossRef]
- Tan, M.L.; Choong, P.F.; Dass, C.R. Recent developments in liposomes, microparticles and nanoparticles for protein and peptide drug delivery. Peptides 2010, 31, 184–193. [Google Scholar] [CrossRef]
- Gainza, G.; Villullas, S.; Pedraz, J.L.; Hernandez, R.M.; Igartua, M. Advances in drug delivery systems (DDSs) to release growth factors for wound healing and skin regeneration. Nanomed. Nanotechnol. Biol. Med. 2015, 11, 1551–1573. [Google Scholar] [CrossRef] [PubMed]
- Kumar, C.S. Nanotechnology tools in pharmaceutical R&D. Mater. Today 2010, 12, 24–30. [Google Scholar] [CrossRef]
- Krausz, A.E.; Adler, B.L.; Cabral, V.; Navati, M.; Doerner, J.; Charafeddine, R.A.; Chandra, D.; Liang, H.; Gunther, L.; Clendaniel, A.; et al. Curcumin-encapsulated nanoparticles as innovative antimicrobial and wound healing agent. Nanomed. Nanotechnol. Biol. Med. 2015, 11, 195–206. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.; Chan, S.Y.; Lee, W.G.; Kang, L. Microfabricated particulate drug-delivery systems. Biotechnol. J. 2011, 6, 1477–1487. [Google Scholar] [CrossRef]
- Abdelkader, D.H.; Tambuwala, M.M.; Mitchell, C.A.; Osman, M.A.; El-Gizawy, S.A.; Faheem, A.M.; El-Tanani, M.; McCarron, P.A. Enhanced cutaneous wound healing in rats following topical delivery of insulin-loaded nanoparticles embedded in poly(vinyl alcohol)-borate hydrogels. Drug Deliv. Transl. Res. 2018, 8, 1053–1065. [Google Scholar] [CrossRef]
- Pardeike, J.; Hommoss, A.; Müller, R.H. Lipid nanoparticles (SLN, NLC) in cosmetic and pharmaceutical dermal products. Int. J. Pharm. 2009, 366, 170–184. [Google Scholar] [CrossRef] [PubMed]
- Prausnitz, M.R. A peptide chaperone for transdermal drug delivery. Nat. Biotechnol. 2006, 24, 416–417. [Google Scholar] [CrossRef] [PubMed]
- Stefánsson, E.; Loftsson, T. Microspheres and nanotechnology for drug delivery. In Retinal Pharmacotherapy; Nguyen, Q.D., Rodrigues, E.B., Farah, M.E., Mieler, W.F., Eds.; W.B. Saunders: Edinburgh, Scotland, 2010; pp. 86–90. [Google Scholar]
- Bozzuto, G.; Molinari, A. Liposomes as nanomedical devices. Int. J. Nanomed. 2015, 10, 975–999. [Google Scholar] [CrossRef]
- Raghavachari, N.; Fahl, W.E. Targeted gene delivery to skin cells In Vivo: A comparative study of liposomes and polymers as delivery vehicles. J. Pharm. Sci. 2002, 91, 615–622. [Google Scholar] [CrossRef]
- Kaplani, K.; Koutsi, S.; Armenis, V.; Skondra, F.G.; Karantzelis, N.; Champeris Tsaniras, S.; Taraviras, S. Wound healing related agents: Ongoing research and perspectives. Adv. Drug Deliv. Rev. 2018, 129, 242–253. [Google Scholar] [CrossRef] [PubMed]
- El Maghraby, G.M.; Barry, B.W.; Williams, A.C. Liposomes and skin: From drug delivery to model membranes. Eur. J. Pharm. Sci. Off. J. Eur. Fed. Pharm. Sci. 2008, 34, 203–222. [Google Scholar] [CrossRef]
- Ferreira, H.; Matamá, T.; Silva, R.; Silva, C.; Gomes, A.C.; Cavaco-Paulo, A. Functionalization of gauzes with liposomes entrapping an anti-inflammatory drug: A strategy to improve wound healing. React. Funct. Polym. 2013, 73, 1328–1334. [Google Scholar] [CrossRef]
- Toh, M.-R.; Chiu, G.N.C. Liposomes as sterile preparations and limitations of sterilisation techniques in liposomal manufacturing. Asian J. Pharm. Sci. 2013, 8, 88–95. [Google Scholar] [CrossRef]
- Dawoud, M.; Yassin, G.; Ghorab, D.M.; Morsi, N.M. Response Surface Optimization and In-vitro Evaluation of Sustained Release Topical Insulin Liposomal Spray for Wound Healing. J. Appl. Pharm. Sci. 2018, 8, 22–29. [Google Scholar] [CrossRef]
- Dawoud, M.H.S.; Yassin, G.E.; Ghorab, D.M.; Morsi, N.M. Insulin mucoadhesive liposomal gel for wound healing: A formulation with sustained release and extended stability using quality by design approach. AAPS Pharm. Sci. Tech. 2019, 20, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Puglia, C.; Bonina, F. Lipid nanoparticles as novel delivery systems for cosmetics and dermal pharmaceuticals. Expert Opin. Drug Deliv. 2012, 9, 429–441. [Google Scholar] [CrossRef] [PubMed]
- Fonte, P.; Araújo, F.; Reis, S.; Sarmento, B. Oral insulin delivery: How far are we? J. Diabetes Sci. Technol. 2013, 7, 520–531. [Google Scholar] [CrossRef]
- Müller, R.H.; Radtke, M.; Wissing, S.A. Solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) in cosmetic and dermatological preparations. Adv. Drug Deliv. Rev. 2002, 54 (Suppl. 1), S131–S155. [Google Scholar] [CrossRef]
- Gainza, G.; Pastor, M.; Aguirre, J.J.; Villullas, S.; Pedraz, J.L.; Hernandez, R.M.; Igartua, M. A novel strategy for the treatment of chronic wounds based on the topical administration of rhEGF-loaded lipid nanoparticles: In Vitro bioactivity and In Vivo effectiveness in healing-impaired db/db mice. J. Control. Release Off. J. Control. Release Soc. 2014, 185, 51–61. [Google Scholar] [CrossRef]
- Gainza, G.; Bonafonte, D.C.; Moreno, B.; Aguirre, J.J.; Gutierrez, F.B.; Villullas, S.; Pedraz, J.L.; Igartua, M.; Hernandez, R.M. The topical administration of rhEGF-loaded nanostructured lipid carriers (rhEGF-NLC) improves healing in a porcine full-thickness excisional wound model. J. Control. Release 2015, 197, 41–47. [Google Scholar] [CrossRef]
- Fonte, P.; Araújo, F.; Silva, C.; Pereira, C.; Reis, S.; Santos, H.A.; Sarmento, B. Polymer-based nanoparticles for oral insulin delivery: Revisited approaches. Biotechnol. Adv. 2015, 33, 1342–1354. [Google Scholar] [CrossRef]
- Patrulea, V.; Ostafe, V.; Borchard, G.; Jordan, O. Chitosan as a starting material for wound healing applications. Eur. J. Pharm. Biopharm. Off. J. Arb. Fur Pharm. Verfahr. 2015, 97, 417–426. [Google Scholar] [CrossRef]
- Elgadir, M.A.; Uddin, M.S.; Ferdosh, S.; Adam, A.; Chowdhury, A.J.; Sarker, M.Z. Impact of chitosan composites and chitosan nanoparticle composites on various drug delivery systems: A review. J. Food Drug Anal. 2015, 23, 619–629. [Google Scholar] [CrossRef]
- Ueno, H.; Mori, T.; Fujinaga, T. Topical formulations and wound healing applications of chitosan. Adv. Drug Deliv. Rev. 2001, 52, 105–115. [Google Scholar] [CrossRef]
- Ahmed, S.; Ikram, S. Chitosan based scaffolds and their applications in wound healing. Achiev. Life Sci. 2016, 10, 27–37. [Google Scholar] [CrossRef]
- Ishihara, M.; Nakanishi, K.; Ono, K.; Sato, M.; Kikuchi, M.; Saito, Y.; Yura, H.; Matsui, T.; Hattori, H.; Uenoyama, M.; et al. Photocrosslinkable chitosan as a dressing for wound occlusion and accelerator in healing process. Biomaterials 2002, 23, 833–840. [Google Scholar] [CrossRef]
- Ehterami, A.; Salehi, M.; Farzamfar, S.; Vaez, A.; Samadian, H.; Sahrapeyma, H.; Mirzaii, M.; Ghorbani, S.; Goodarzi, A. In Vitro and In Vivo study of PCL/COLL wound dressing loaded with insulin-chitosan nanoparticles on cutaneous wound healing in rats model. Int. J. Biol. Macromol. 2018, 117, 601–609. [Google Scholar] [CrossRef]
- Mohammed, M.A.; Syeda, J.T.M.; Wasan, K.M.; Wasan, E.K. An overview of chitosan nanoparticles and its application in non-parenteral drug delivery. Pharmaceutics 2017, 9, 53. [Google Scholar] [CrossRef]
- Bellich, B.; D’Agostino, I.; Semeraro, S.; Gamini, A.; Cesàro, A. The good, the bad and the ugly of chitosans. Mar. Drugs 2016, 14, 99. [Google Scholar] [CrossRef]
- Kavasi, R.M.; Berdiaki, A.; Spyridaki, I.; Corsini, E.; Tsatsakis, A.; Tzanakakis, G.; Nikitovic, D. HA metabolism in skin homeostasis and inflammatory disease. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2017, 101, 128–138. [Google Scholar] [CrossRef] [PubMed]
- Litwiniuk, M.; Krejner, A.; Speyrer, M.S.; Gauto, A.R.; Grzela, T. Hyaluronic acid in inflammation and tissue regeneration. Wounds A Compend. Clin. Res. Pract. 2016, 28, 78–88. [Google Scholar]
- Hirakura, T.; Yasugi, K.; Nemoto, T.; Sato, M.; Shimoboji, T.; Aso, Y.; Morimoto, N.; Akiyoshi, K. Hybrid hyaluronan hydrogel encapsulating nanogel as a protein nanocarrier: New system for sustained delivery of protein with a chaperone-like function. J. Control. Release 2010, 142, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Nyman, E.; Henricson, J.; Ghafouri, B.; Anderson, C.D.; Kratz, G. Hyaluronic acid accelerates re-epithelialization and alters protein expression in a human wound model. Plastic and reconstructive surgery. Glob. Open 2019, 7, e2221. [Google Scholar] [CrossRef]
- Aderibigbe, B.A.; Buyana, B. Alginate in wound dressings. Pharmaceutics 2018, 10, 42. [Google Scholar] [CrossRef]
- Borselli, C.; Storrie, H.; Benesch-Lee, F.; Shvartsman, D.; Cezar, C.; Lichtman, J.W.; Vandenburgh, H.H.; Mooney, D.J. Functional muscle regeneration with combined delivery of angiogenesis and myogenesis factors. Proc. Natl. Acad. Sci. USA 2010, 107, 3287–3292. [Google Scholar] [CrossRef]
- Fonte, P.; Lino, P.R.; Seabra, V.; Almeida, A.J.; Reis, S.; Sarmento, B. Annealing as a tool for the optimization of lyophilization and ensuring of the stability of protein-loaded PLGA nanoparticles. Int. J. Pharm. 2016, 503, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Fonte, P.; Andrade, F.; Azevedo, C.; Pinto, J.; Seabra, V.; van de Weert, M.; Reis, S.; Sarmento, B. Effect of the freezing step in the stability and bioactivity of protein-loaded PLGA nanoparticles upon lyophilization. Pharm. Res. 2016, 33, 2777–2793. [Google Scholar] [CrossRef] [PubMed]
- Ulery, B.D.; Nair, L.S.; Laurencin, C.T. Biomedical applications of biodegradable polymers. J. Polym. Sci. Part B Polym. Phys. 2011, 49, 832–864. [Google Scholar] [CrossRef] [PubMed]
- Hrynyk, M.; Martins-Green, M.; Barron, A.E.; Neufeld, R.J. Sustained prolonged topical delivery of bioactive human insulin for potential treatment of cutaneous wounds. Int. J. Pharm. 2010, 398, 146–154. [Google Scholar] [CrossRef] [PubMed]
- Hrynyk, M.; Martins-Green, M.; Barron, A.E.; Neufeld, R.J. Alginate-PEG sponge architecture and role in the design of insulin release dressings. Biomacromolecules 2012, 13, 1478–1485. [Google Scholar] [CrossRef] [PubMed]
- Abdelkader, D.H.; Osman, M.A.; El-Gizawy, S.A.; Hawthorne, S.J.; Faheem, A.M.; McCarron, P.A. Effect of poly(ethylene glycol) on insulin stability and cutaneous cell proliferation In Vitro following cytoplasmic delivery of insulin-loaded nanoparticulate carriers—A potential topical wound management approach. Eur. J. Pharm. Sci. Off. J. Eur. Fed. Pharm. Sci. 2018, 114, 372–384. [Google Scholar] [CrossRef]
- Mody, V.V.; Siwale, R.; Singh, A.; Mody, H.R. Introduction to metallic nanoparticles. J. Pharm. Bioallied Sci. 2010, 2, 282. [Google Scholar] [CrossRef]
- Rai, M.; Yadav, A.; Gade, A. Silver nanoparticles as a new generation of antimicrobials. Biotechnol. Adv. 2009, 27, 76–83. [Google Scholar] [CrossRef]
- Gunasekaran, T.; Nigusse, T.; Dhanaraju, M.D. Silver nanoparticles as real topical bullets for wound healing. J. Am. Coll. Clin. Wound Spec. 2012, 3, 82–96. [Google Scholar] [CrossRef]
- Tian, J.; Wong, K.K.; Ho, C.M.; Lok, C.N.; Yu, W.Y.; Che, C.M.; Chiu, J.F.; Tam, P.K. Topical delivery of silver nanoparticles promotes wound healing. Chem. Med. Chem. 2007, 2, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Lee, P.Y.; Ho, C.M.; Lui, V.C.; Chen, Y.; Che, C.M.; Tam, P.K.; Wong, K.K. Silver nanoparticles mediate differential responses in keratinocytes and fibroblasts during skin wound healing. Chem. Med. Chem. 2010, 5, 468–475. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Jun, B.H. Silver nanoparticles: Synthesis and application for nanomedicine. Int. J. Mol. Sci. 2019, 20, 865. [Google Scholar] [CrossRef] [PubMed]
- Kaur, P.; Sharma, A.K.; Nag, D.; Das, A.; Datta, S.; Ganguli, A.; Goel, V.; Rajput, S.; Chakrabarti, G.; Basu, B.; et al. Novel nano-insulin formulation modulates cytokine secretion and remodeling to accelerate diabetic wound healing. Nanomed. Nanotechnol. Biol. Med. 2019, 15, 47–57. [Google Scholar] [CrossRef]
- Rakhmetova, A.A.; Alekseeva, T.P.; Bogoslovskaya, O.A.; Leipunskii, I.O.; Ol’khovskaya, I.P.; Zhigach, A.N.; Glushchenko, N.N. Wound-healing properties of copper nanoparticles as a function of physicochemical parameters. Nanotechnol. Russ. 2010, 5, 271–276. [Google Scholar] [CrossRef]
- Chigurupati, S.; Mughal, M.R.; Okun, E.; Das, S.; Kumar, A.; McCaffery, M.; Seal, S.; Mattson, M.P. Effects of cerium oxide nanoparticles on the growth of keratinocytes, fibroblasts and vascular endothelial cells in cutaneous wound healing. Biomaterials 2013, 34, 2194–2201. [Google Scholar] [CrossRef]
- Sankar, R.; Dhivya, R.; Shivashangari, K.S.; Ravikumar, V. Wound healing activity of Origanum vulgare engineered titanium dioxide nanoparticles in Wistar Albino rats. J. Mater. Sci. Mater. Med. 2014, 25, 1701–1708. [Google Scholar] [CrossRef] [PubMed]
- Randeria, P.S.; Seeger, M.A.; Wang, X.-Q.; Wilson, H.; Shipp, D.; Mirkin, C.A.; Paller, A.S. siRNA-based spherical nucleic acids reverse impaired wound healing in diabetic mice by ganglioside GM3 synthase knockdown. Proc. Natl. Acad. Sci. USA 2015, 112, 5573–5578. [Google Scholar] [CrossRef]
- Ponnanikajamideen, M.; Shanmugam, R.; Vanaja, M.; Gurusamy, A. In Vivo anti-diabetic and wound healing effect of antioxidant gold nanoparticles synthesized using insulin plant (Chamaecostus Cuspidatus). Can. J. Diabetes 2018, 43, 1–8. [Google Scholar] [CrossRef]
- Mebert, A.M.; Baglole, C.J.; Desimone, M.F.; Maysinger, D. Nanoengineered silica: Properties, applications and toxicity. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2017, 109, 753–770. [Google Scholar] [CrossRef]
- Quignard, S.; Coradin, T.; Powell, J.J.; Jugdaohsingh, R. Silica nanoparticles as sources of silicic acid favoring wound healing In Vitro. Colloids Surf. B Biointerfaces 2017, 155, 530–537. [Google Scholar] [CrossRef]
- Ponnanikajamideen, M.; Rajeshkumar, S.; Vanaja, M.; Annadurai, G. In Vivo type 2 diabetes and wound-healing effects of antioxidant gold nanoparticles synthesized using the insulin plant Chamaecostus cuspidatus in Albino rats. Can. J. Diabetes 2019, 43, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Bobo, D.; Robinson, K.J.; Islam, J.; Thurecht, K.J.; Corrie, S.R. Nanoparticle-based medicines: A review of FDA-approved materials and clinical trials to date. Pharm. Res. 2016, 33, 2373–2387. [Google Scholar] [CrossRef] [PubMed]
- Fang, R.C.; Galiano, R.D. A review of becaplermin gel in the treatment of diabetic neuropathic foot ulcers. Biol. Targets Ther. 2008, 2, 1–12. [Google Scholar] [CrossRef][Green Version]
- Papanas, N.; Maltezos, E. Becaplermin gel in the treatment of diabetic neuropathic foot ulcers. Clin. Interv. Aging 2008, 3, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Okabe, K.; Hayashi, R.; Aramaki-Hattori, N.; Sakamoto, Y.; Kishi, K. Wound Treatment Using Growth Factors. Mod. Plastic Surg. 2013, 3, 108–112. [Google Scholar] [CrossRef]
- Hayashida, K.; Akita, S. Quality of pediatric second-degree burn wound scars following the application of basic fibroblast growth factor: Results of a randomized, controlled pilot study. Ostomy Wound Manag. 2012, 58, 32–36. [Google Scholar]
- Park, J.W.; Hwang, S.R.; Yoon, I.S. Advanced growth factor delivery systems in wound management and skin regeneration. Molecules 2017, 22, 1259. [Google Scholar] [CrossRef]
- Veves, A.; Sheehan, P.; Pham, H.T. A randomized, controlled trial of Promogran (a collagen/oxidized regenerated cellulose dressing) vs standard treatment in the management of diabetic foot ulcers. Arch. Surg. 2002, 137, 822–827. [Google Scholar] [CrossRef]
- Roukis, T.S.; Zgonis, T.; Tiernan, B. Autologous platelet-rich plasma for wound and osseous healing: A review of the literature and commercially available products. Adv. Ther. 2006, 23, 218–237. [Google Scholar] [CrossRef]
- Bowler, P.G.; Duerden, B.I.; Armstrong, D.G. Wound microbiology and associated approaches to wound management. Clin. Microbiol. Rev. 2001, 14, 244–269. [Google Scholar] [CrossRef]
- Gainza LaFuente, E.; Gainza Lucea, G.; Villullas Rincón, S.; Pastor Navarro, M.; Ibarrola Moreno, O.; Alonso Hornes, G.; Del Pozo Pérez, A.; Hernández Martín Rosa, M.; Igartua Olaechea, M.; Pedraz Muñoz, J.L. Lipid Nanoparticles for Wound Healing. U.S. Patent 20160199447A1, 14 July 2016. [Google Scholar]
- Paladini, F.; Pollini, M. Antimicrobial silver nanoparticles for wound healing application: Progress and future trends. Materials 2019, 12, 2540. [Google Scholar] [CrossRef] [PubMed]
- Kwan, K.H.; Liu, X.; To, M.K.; Yeung, K.W.; Ho, C.M.; Wong, K.K. Modulation of collagen alignment by silver nanoparticles results in better mechanical properties in wound healing. Nanomed. Nanotechnol. Biol. Med. 2011, 7, 497–504. [Google Scholar] [CrossRef]
- Rawat, M.; Singh, D.; Saraf, S.; Saraf, S. Nanocarriers: Promising vehicle for bioactive drugs. Biol. Pharm. Bull. 2006, 29, 1790–1798. [Google Scholar] [CrossRef]
- Riediker, M.; Zink, D.; Kreyling, W.; Oberdörster, G.; Elder, A.; Graham, U.; Lynch, I.; Duschl, A.; Ichihara, G.; Ichihara, S.; et al. Particle toxicology and health—Where are we? Part. Fibre Toxicol. 2019, 16, 1–33. [Google Scholar] [CrossRef] [PubMed]
- Schneider, M.; Stracke, F.; Hansen, S.; Schaefer, U.F. Nanoparticles and their interactions with the dermal barrier. Dermatoendocrinol 2009, 1, 197–206. [Google Scholar] [CrossRef]
- Nel, A.; Xia, T.; Mädler, L.; Li, N. Toxic potential of materials at the nanolevel. Science 2006, 311, 622–627. [Google Scholar] [CrossRef] [PubMed]
- Upton, Z.; Wallace, H.J.; Shooter, G.K.; van Lonkhuyzen, D.R.; Yeoh-Ellerton, S.; Rayment, E.A.; Fleming, J.M.; Broszczak, D.; Queen, D.; Sibbald, R.G.; et al. Human pilot studies reveal the potential of a vitronectin: Growth factor complex as a treatment for chronic wounds. Int. Wound J. 2011, 8, 522–532. [Google Scholar] [CrossRef]
- Manavitehrani, I.; Fathi, A.; Badr, H.; Daly, S.; Negahi Shirazi, A.; Dehghani, F. Biomedical applications of biodegradable polyesters. Polymers 2016, 8, 20. [Google Scholar] [CrossRef]
- Jia, Y.-P.; Ma, B.-Y.; Wei, X.-W.; Qian, Z.-Y. The In Vitro and In Vivo toxicity of gold nanoparticles. Chin. Chem. Lett. 2017, 28, 691–702. [Google Scholar] [CrossRef]
- Reidy, B.; Haase, A.; Luch, A.; Dawson, K.A.; Lynch, I. Mechanisms of silver nanoparticle release, transformation and toxicity: A critical review of current knowledge and recommendations for future studies and applications. Materials 2013, 6, 2295–2350. [Google Scholar] [CrossRef] [PubMed]
- Ryu, H.J.; Seong, N.W.; So, B.J.; Seo, H.S.; Kim, J.H.; Hong, J.S.; Park, M.K.; Kim, M.S.; Kim, Y.R.; Cho, K.B.; et al. Evaluation of silica nanoparticle toxicity after topical exposure for 90 days. Int. J. Nanomed. 2014, 9 (Suppl. 2), 127–136. [Google Scholar] [CrossRef]
- Pan, Z.; Lee, W.; Slutsky, L.; Clark, R.A.; Pernodet, N.; Rafailovich, M.H. Adverse effects of titanium dioxide nanoparticles on human dermal fibroblasts and how to protect cells. Small 2009, 5, 511–520. [Google Scholar] [CrossRef] [PubMed]
| Wound Type | Etiology | Microenvironment | Bacterial Environment | Average Healing Time | Ref. |
|---|---|---|---|---|---|
| Pressure ulcers | spinal cord injury paralysis senility substance abuse malnutrition stroke multiple sclerosis | shear stress friction high humidity temperature tissue atrophy | Enterococcus Staphylococcus Pseudomonas Serratia Proteus Corynebacterium Staphylococcus Anaerococcus Bacteroides | 2 weeks | [24,25,26] |
| Diabetic foot ulcers | hyperglycemia poor circulation neuropathy wounded feet | ischemia infection foot deformity callus trauma pressure | Enterococcus Pseudomonas Streptococcus Serratia Staphylococcus Anaerococcus Corynebacterium Pseudomonas | 6 months | [27,28,29] |
| Venous leg ulcers | age obesity physical inactivity trauma deep vein thrombosis phlebitis | chronic venous hypertension vein walls structural failure valve system failure | Staphylococcus Serratia Streptococcus Pseudomonas Corynebacterium Staphylococcus Bacteroides | 4 months | [30,31] |
| Type of Nanoparticles | Material | Size (nm) | PdI | Zeta Potential (mV) | In Vitro Model | In Vivo Model | Wound Decrease (%) | Ref. |
|---|---|---|---|---|---|---|---|---|
| Liposomes | Phosphatyl choline, cholesterol, chitosan | 184–701 | - | −22.9 to −18 | - | Human | 40 | [85] |
| Polymeric nanoparticles | Chitosan, poly (ε-caprolactone)/collagen | 256 | 0.23 | 17.89 | L929 cells | C57BL/6 mice | ~63 | [97] |
| PLGA, PEG | 297 | 0.15 | −3.94 | HaCat, human fibroblasts | - | - | [110] | |
| Inorganic nanoparticles | Silver | 10 | - | - | Mouse embryo fibroblast cell line (BALB/3T3; clone A31) | C57BL/6N mice | ~31 | [116] |
| Silver | 42 | - | −15.1 | Human epidermal keratinocyte cells (HEKa) | Wistar rats | ~73 | [118] | |
| Copper | 119 | - | - | - | Mice of the SHK line | ~52 | [119] | |
| Nanoceria | 5 | - | - | HMEC-1 cells | C57BL/6 mice | - | [120] | |
| Titanium dioxide | 341 | - | −27.3 | - | Wistar Albino rats | 94 | [121] | |
| Gold | 50 | - | - | - | Wistar rats | 97 | [126] | |
| Silica | 10 | - | −30 to +23 | CCD-25SK cells | - | - | [125] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Macedo, A.S.; Mendes, F.; Filipe, P.; Reis, S.; Fonte, P. Nanocarrier-Mediated Topical Insulin Delivery for Wound Healing. Materials 2021, 14, 4257. https://doi.org/10.3390/ma14154257
Macedo AS, Mendes F, Filipe P, Reis S, Fonte P. Nanocarrier-Mediated Topical Insulin Delivery for Wound Healing. Materials. 2021; 14(15):4257. https://doi.org/10.3390/ma14154257
Chicago/Turabian StyleMacedo, Ana S., Francisca Mendes, Patrícia Filipe, Salette Reis, and Pedro Fonte. 2021. "Nanocarrier-Mediated Topical Insulin Delivery for Wound Healing" Materials 14, no. 15: 4257. https://doi.org/10.3390/ma14154257
APA StyleMacedo, A. S., Mendes, F., Filipe, P., Reis, S., & Fonte, P. (2021). Nanocarrier-Mediated Topical Insulin Delivery for Wound Healing. Materials, 14(15), 4257. https://doi.org/10.3390/ma14154257









