Characterization of Newly Developed Zinc Composite with the Content of 8 wt.% of Hydroxyapatite Particles Processed by Extrusion
Abstract
1. Introduction
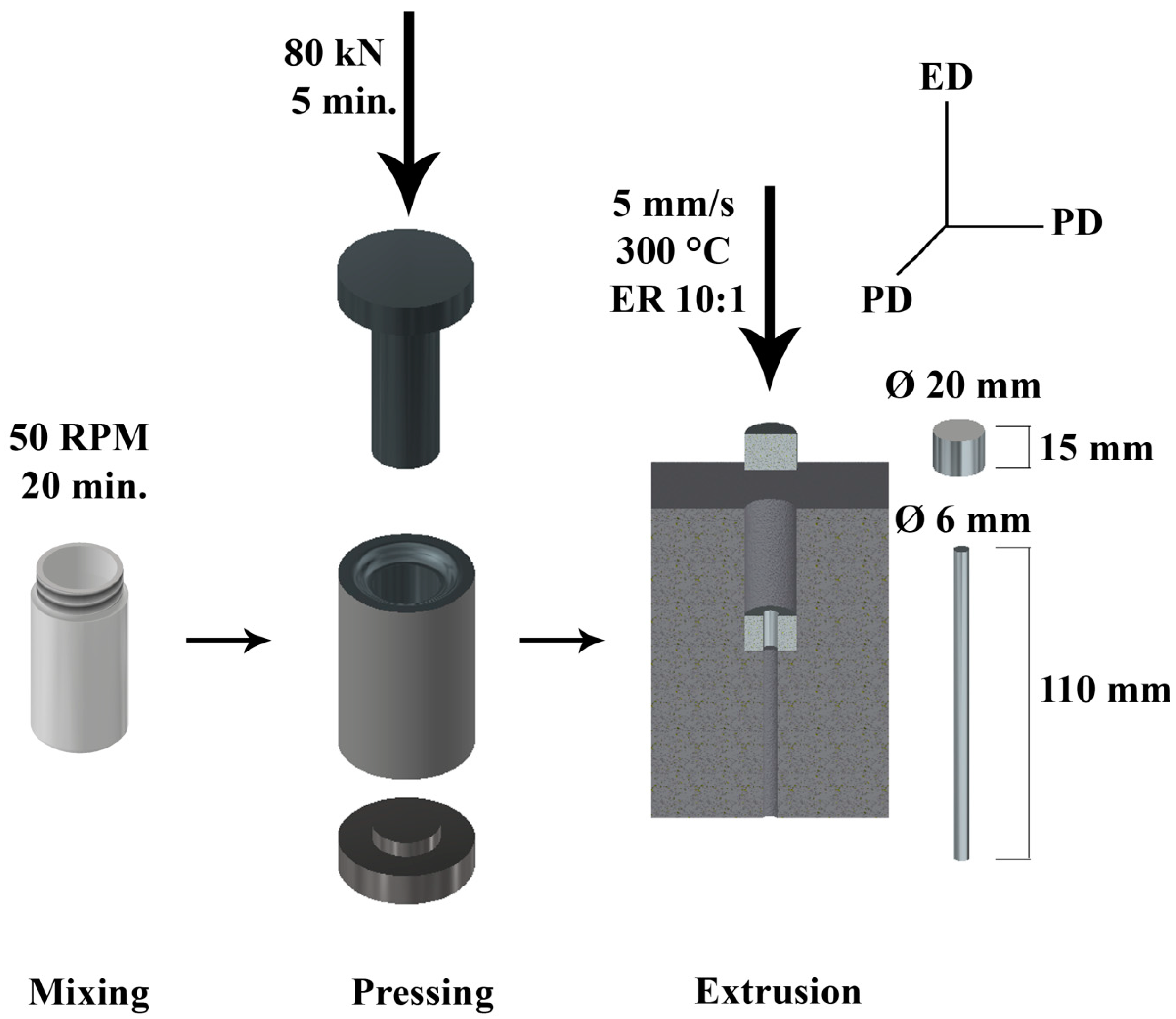
2. Materials and Methods
3. Results and Discussion
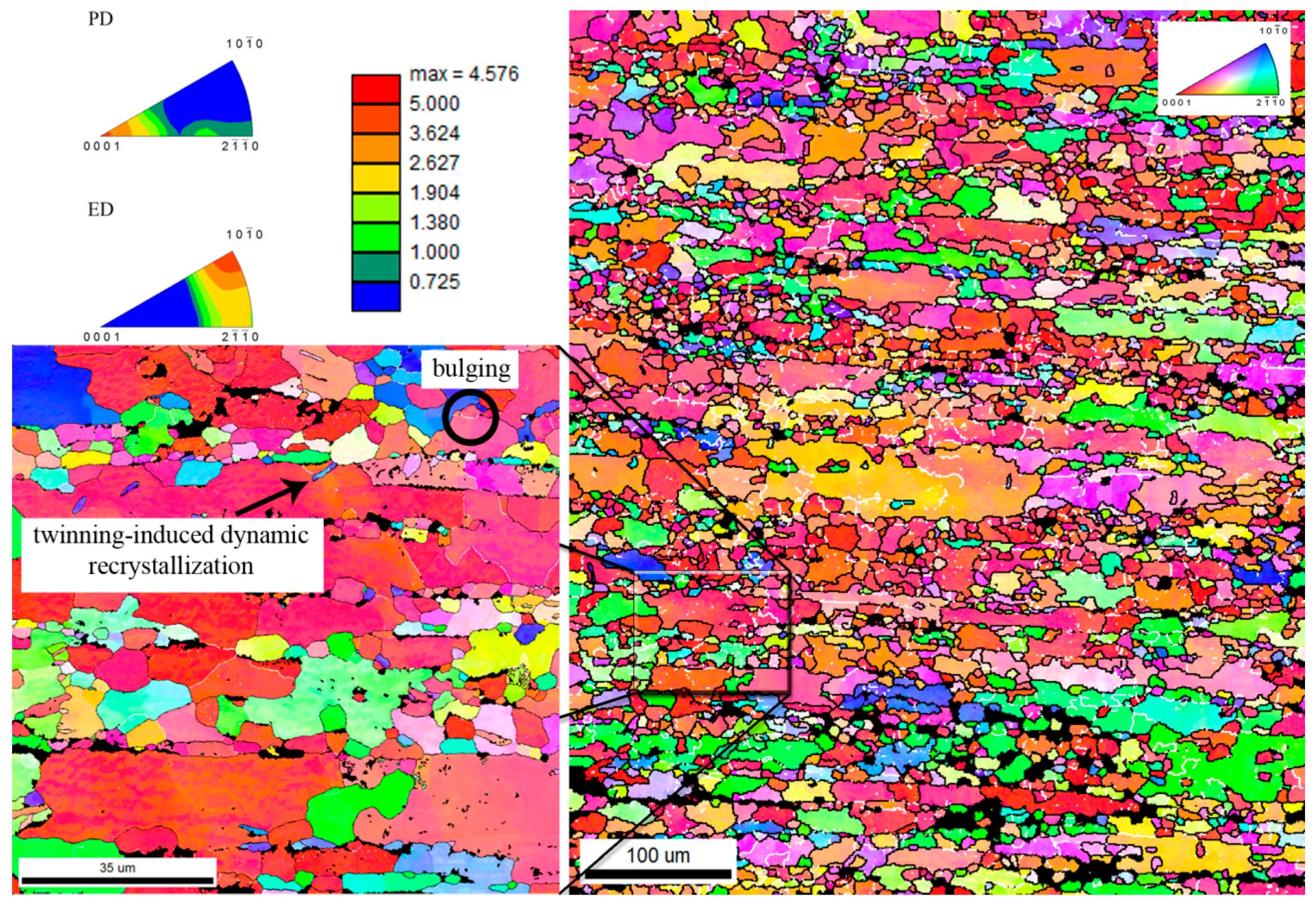
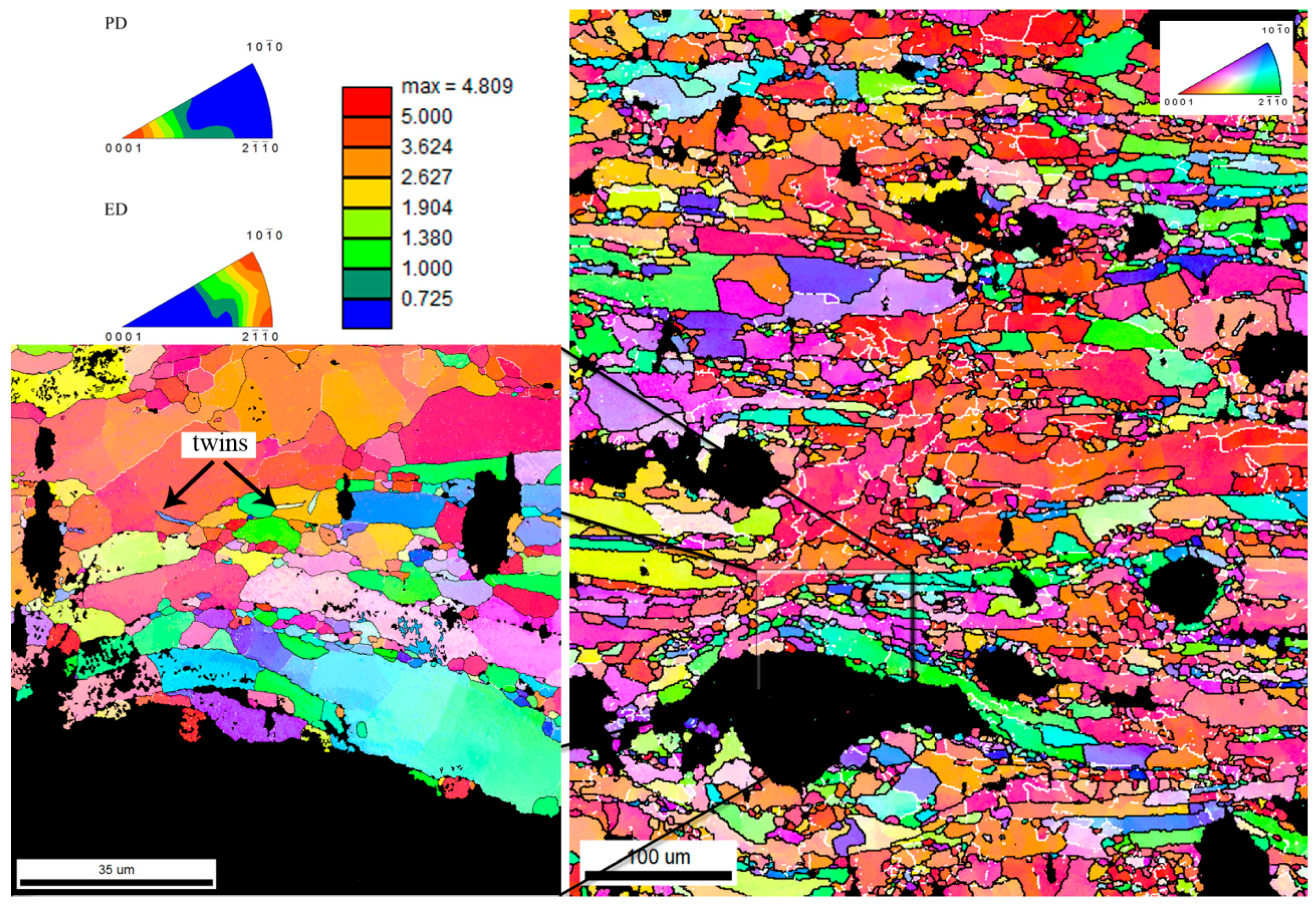
3.1. Microstructure
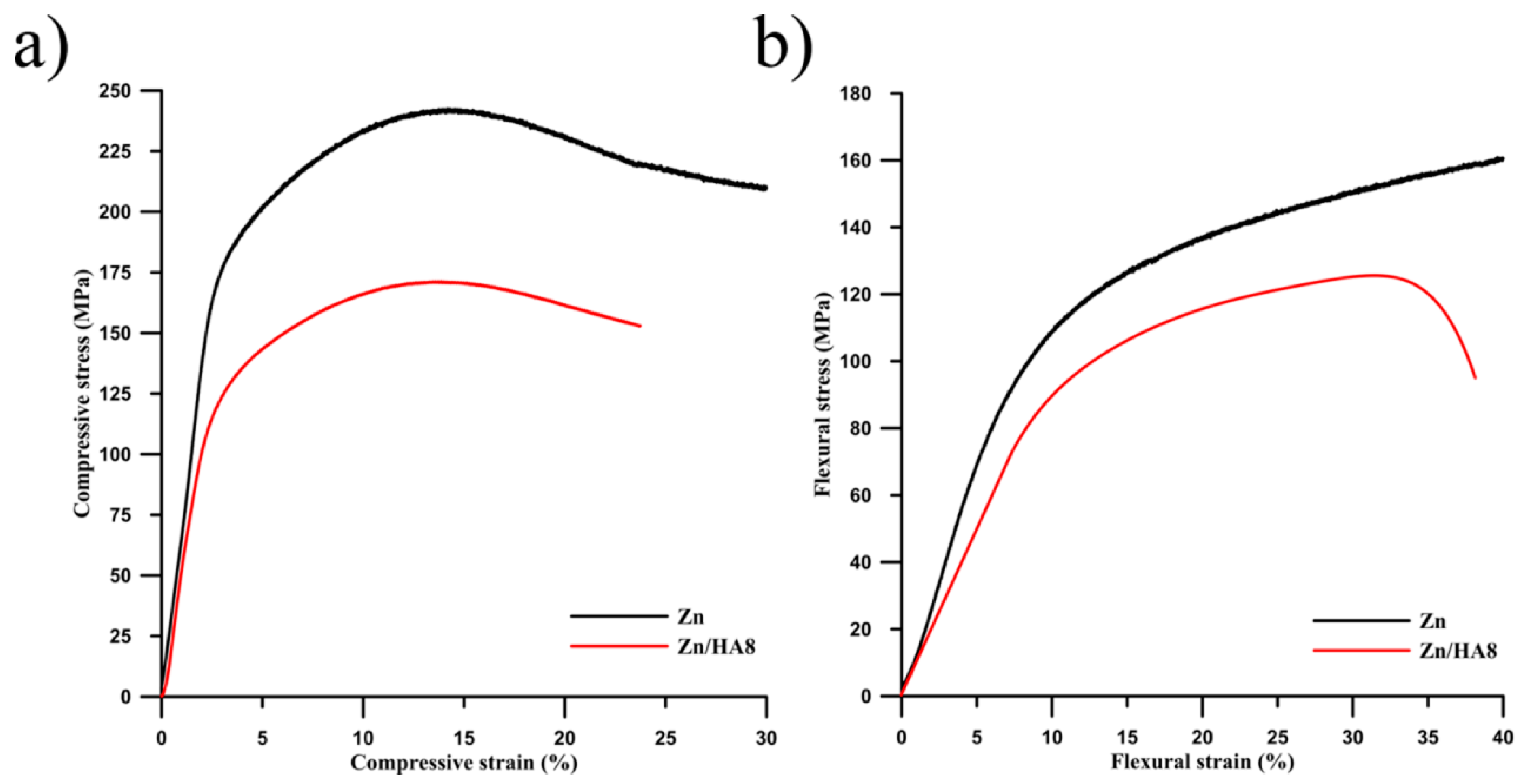
3.2. Mechanical Properties
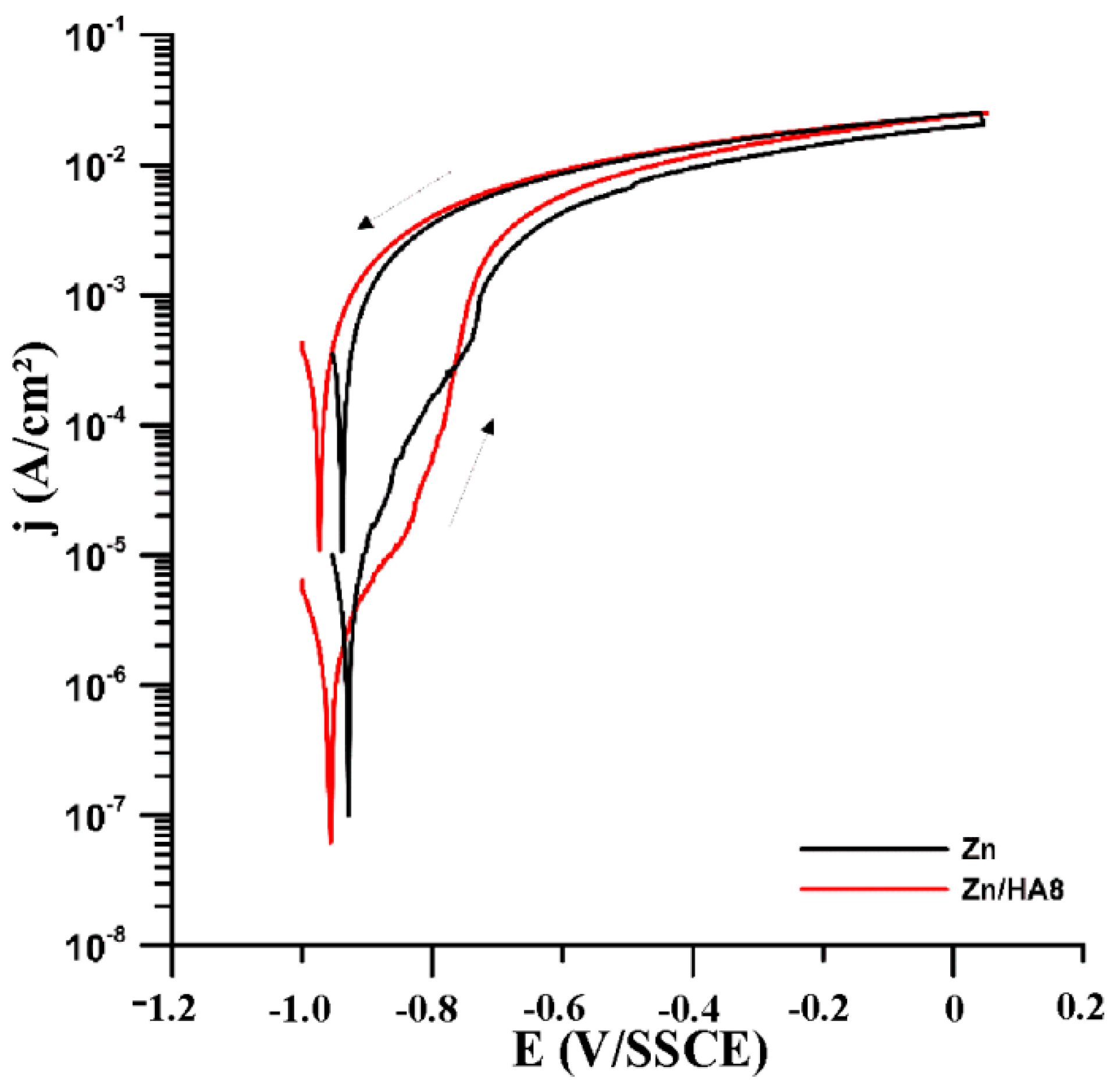
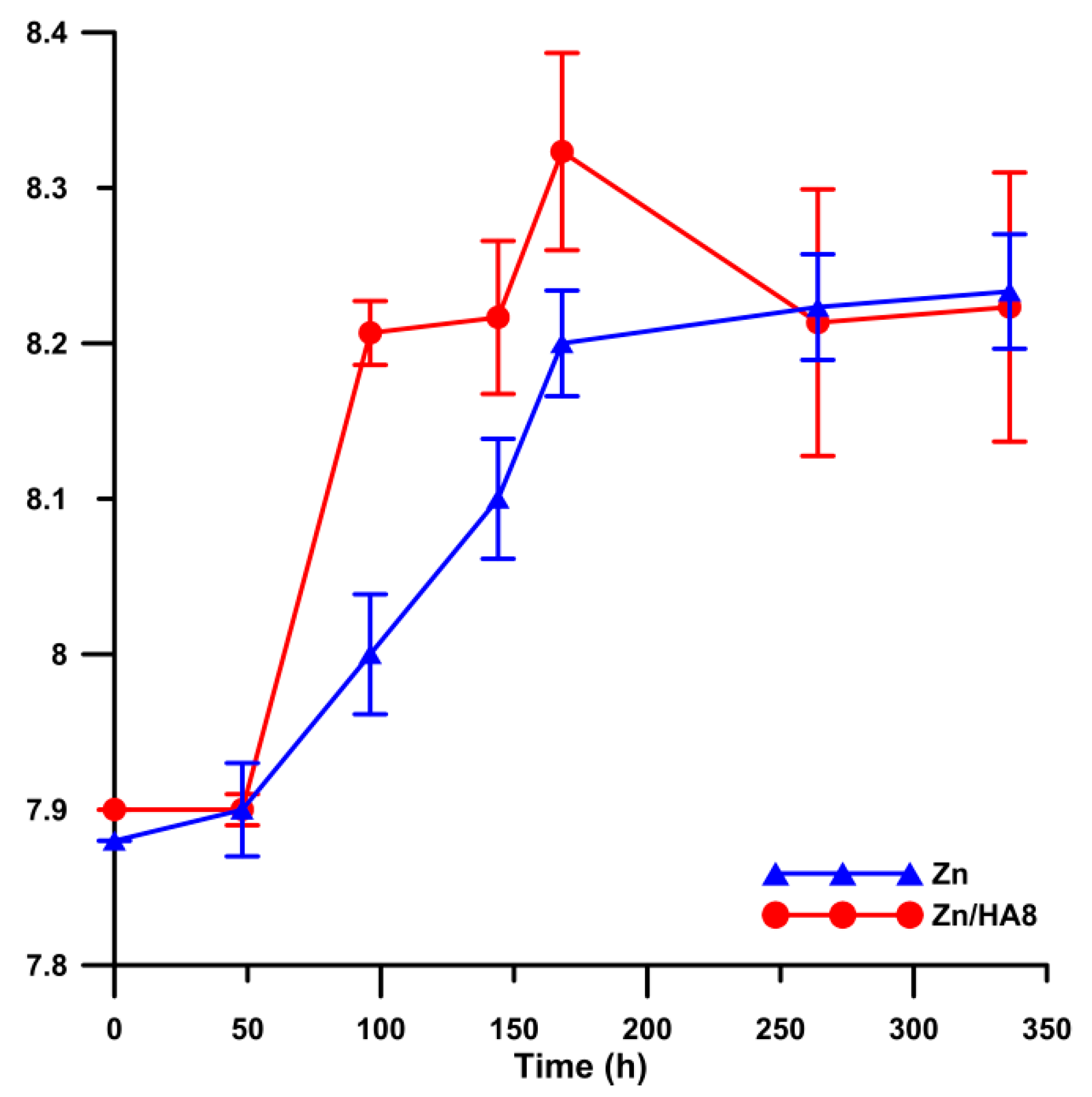
3.3. Corrosion Properties
4. Conclusions
- The extrusion process significantly enhances the plastic behavior of the material in comparison to other methods, which were studied previously.
- Recrystallization of the Zn matrix during the extrusion was enhanced in the surrounding of the HA particles.
- The HA particles act as defects in the structure from the point of view of the mechanical behavior.
- The presence of the HA particles decreases a mechanical performance (compressive and flexural) approximately by 30% in comparison to pure zinc prepared in the same way.
- The HA particles simplify the precipitation of HA from the solution.
- The corrosion rates of studied materials seem to be appropriate in relation to potential applications.
- All the mentioned results suggest the usability of those materials for specific biodegradable applications.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ma, J.; Zhao, N.; Zhu, D. Endothelial cellular responses to biodegradable metal zinc. ACS Biomater. Sci. Eng. 2015, 1, 1174–1182. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zheng, Y.; Qin, L. Progress of biodegradable metals. Prog. Nat. Sci. Mater. Int. 2014, 24, 414–422. [Google Scholar] [CrossRef]
- Bowen, P.K.; Shearier, E.R.; Zhao, S.; Guillory , R.J., II; Zhao, F.; Goldman, J.; Drelich, J.W. Biodegradable Metals for Cardiovascular Stents: From Clinical Concerns to Recent Zn-Alloys. Adv. Healthc. Mater. 2016, 5, 1121–1140. [Google Scholar] [CrossRef] [PubMed]
- Čapek, J.; Pinc, J.; Msallamová, Š.; Jablonská, E.; Veřtát, P.; Kubásek, J.; Vojtěch, D. Thermal Plasma Spraying as a New Approach for Preparation of Zinc Biodegradable Scaffolds: A Complex Material Characterization. J. Therm. Spray Technol. 2019, 28, 826–841. [Google Scholar] [CrossRef]
- Bowen, P.K.; Drelich, J.; Goldman, J. Zinc Exhibits Ideal Physiological Corrosion Behavior for Bioabsorbable Stents. Adv. Mater. 2013, 25, 2577–2582. [Google Scholar] [CrossRef]
- Vojtěch, D.; Kubásek, J.; Šerák, J.; Novák, P. Mechanical and corrosion properties of newly developed biodegradable Zn-based alloys for bone fixation. Acta Biomater. 2011, 7, 3515–3522. [Google Scholar] [CrossRef]
- Zheng, Y.F.; Gu, X.N.; Witte, F. Biodegradable metals. Mater. Sci. Eng. R Rep. 2014, 77, 1–34. [Google Scholar] [CrossRef]
- Cheng, J.; Liu, B.; Wu, Y.H.; Zheng, Y.F. Comparative in vitro Study on Pure Metals (Fe, Mn, Mg, Zn and W) as Biodegradable Metals. J. Mater. Sci. Technol. 2013, 29, 619–627. [Google Scholar] [CrossRef]
- Katarivas Levy, G.; Goldman, J.; Aghion, E. The Prospects of Zinc as a Structural Material for Biodegradable Implants—A Review Paper. Metals 2017, 7, 402. [Google Scholar] [CrossRef]
- Shearier, E.R.; Bowen, P.K.; He, W.; Drelich, A.; Drelich, J.; Goldman, J.; Zhao, F. In Vitro Cytotoxicity, Adhesion, and Proliferation of Human Vascular Cells Exposed to Zinc. ACS Biomater. Sci. Eng. 2016, 2, 634–642. [Google Scholar] [CrossRef]
- Roach, P.; Farrar, D.; Perry, C.C. Interpretation of Protein Adsorption: Surface-Induced Conformational Changes. J. Am. Chem. Soc. 2005, 127, 8168–8173. [Google Scholar] [CrossRef] [PubMed]
- Bidhendi, H.R.A.; Pouranvari, M. Corrosion study of metallic biomaterials in simulated body fluid. Metallur. Mater. Eng. 2012, 17, 13–22. [Google Scholar]
- Clark, G.C.F.; Williams, D.F. The effects of proteins on metallic corrosion. J. Biomed. Mater. Res. 1982, 16, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Sato, N. An overview on the passivity of metals. Corros. Sci. 1990, 31, 1–19. [Google Scholar] [CrossRef]
- Dunne, C.F.; Levy, G.K.; Hakimi, O.; Aghion, E.; Twomey, B.; Stanton, K.T. Corrosion behaviour of biodegradable magnesium alloys with hydroxyapatite coatings. Surf. Coat. Technol. 2016, 289, 37–44. [Google Scholar] [CrossRef]
- Tisdel, C.L.; Goldberg, V.M.; Parr, J.A.; Bensusan, J.S.; Staikoff, L.S.; Stevenson, S. The influence of a hydroxyapatite and tricalcium-phosphate coating on bone growth into titanium fiber-metal implants. J. Bone Jt. Surg. Am. Vol. 1994, 76, 159–171. [Google Scholar] [CrossRef]
- Deligianni, D.D.; Katsala, N.D.; Koutsoukos, P.G.; Missirlis, Y.F. Effect of surface roughness of hydroxyapatite on human bone marrow cell adhesion, proliferation, differentiation and detachment strength. Biomaterials 2000, 22, 87–96. [Google Scholar] [CrossRef]
- Ye, X.; Chen, M.; Yang, M.; Wei, J.; Liu, D. In vitro corrosion resistance and cytocompatibility of nano-hydroxyapatite reinforced Mg–Zn–Zr composites. J. Mater. Sci. Mater. Med. 2010, 21, 1321–1328. [Google Scholar] [CrossRef]
- Mensah-Darkwa, K.; Gupta, R.K.; Kumar, D. Mechanical and Corrosion Properties of Magnesium–Hydroxyapatite (Mg–HA) Composite Thin Films. J. Mater. Sci. Technol. 2013, 29, 788–794. [Google Scholar] [CrossRef]
- Gbureck, U.; Masten, A.; Probst, J.; Thull, R. Tribochemical structuring and coating of implant metal surfaces with titanium oxide and hydroxyapatite layers. Mater. Sci. Eng. C 2003, 23, 461–465. [Google Scholar] [CrossRef]
- Zheng, X.; Huang, M.; Ding, C. Bond strength of plasma-sprayed hydroxyapatite/Ti composite coatings. Biomaterials 2000, 21, 841–849. [Google Scholar] [CrossRef]
- Ishizawa, H.; Ogino, M. Thin hydroxyapatite layers formed on porous titanium using electrochemical and hydrothermal reaction. J. Mater. Sci. 1996, 31, 6279–6284. [Google Scholar] [CrossRef]
- Gu, X.; Zhou, W.; Zheng, Y.; Dong, L.; Xi, Y.; Chai, D. Microstructure, mechanical property, bio-corrosion and cytotoxicity evaluations of Mg/HA composites. Mater. Sci. Eng. C 2010, 30, 827–832. [Google Scholar] [CrossRef]
- Kim, J.Y.; Lee, J.W.; Lee, S.-J.; Park, E.K.; Kim, S.-Y.; Cho, D.-W. Development of a bone scaffold using HA nanopowder and micro-stereolithography technology. Microelectron. Eng. 2007, 84, 1762–1765. [Google Scholar] [CrossRef]
- Radin, S.R.; Ducheyne, P. Effect of bioactive ceramic composition and structure on in vitro behavior. III. Porous versus dense ceramics. J. Biomed. Mater. Res. 1994, 28, 1303–1309. [Google Scholar] [CrossRef]
- Ramesh, C.; Hirianiah, A.; Harishanad, K.; Noronha, N.P. A review on hot extrusion of Metal Matrix Composites (MMC’s). Int. J. Eng. Sci. 2012, 1, 30–35. [Google Scholar]
- Saravanan, R.A.; Surappa, M.K. Fabrication and characterisation of pure magnesium-30 vol% SiCP particle composite. Mater. Sci. Eng. A 2000, 276, 108–116. [Google Scholar] [CrossRef]
- Bauser, M.; Siegert, K. Extrusion, 2nd ed.; ASM International: Cleveland, OH, USA, 2006. [Google Scholar]
- Pinc, J.; Miklášová, E.; Průša, F.; Čapek, J.; Drahokoupil, J.; Vojtěch, D. Influence of Processing on the Microstructure and the Mechanical Properties of Zn/HA8 wt.% Biodegradable Composite. Manuf. Technol. 2019, 19, 836–841. [Google Scholar] [CrossRef]
- Müller, L.; Müller, F.A. Preparation of SBF with different HCO3- content and its influence on the composition of biomimetic apatites. Acta Biomater. 2006, 2, 181–189. [Google Scholar] [CrossRef]
- Thümmler, F.; Oberacker, R. Introduction to Powder Metallurgy; Maney Publishing for IOM3, the Institute of Materials, Minerals and Mining; Oxford Science Publications: Oxford, UK, 1993. [Google Scholar]
- Sakai, T.; Belyakov, A.; Kaibyshev, R.; Miura, H.; Jonas, J.J. Dynamic and post-dynamic recrystallization under hot, cold and severe plastic deformation conditions. Prog. Mater. Sci. 2014, 60, 130–207. [Google Scholar] [CrossRef]
- Al-Samman, T. Modification of texture and microstructure of magnesium alloy extrusions by particle-stimulated recrystallization. Mater. Sci. Eng. A 2013, 560, 561–566. [Google Scholar] [CrossRef]
- Barrett, C.D.; Imandoust, A.; Oppedal, A.L.; Inal, K.; Tschopp, M.A.; El Kadiri, H. Effect of grain boundaries on texture formation during dynamic recrystallization of magnesium alloys. Acta Mater. 2017, 128, 270–283. [Google Scholar] [CrossRef]
- Hing, K.A.; Best, S.M.; Bonfield, W. Characterization of porous hydroxyapatite. J. Mater. Sci. Mater. Med. 1999, 10, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Qu, X.; Lin, W.; Wang, C.; Zhu, D.; Dai, K.; Zheng, Y. In vitro and in vivo studies on zinc-hydroxyapatite composites as novel biodegradable metal matrix composite for orthopedic applications. Acta Biomater. 2018, 71, 200–214. [Google Scholar] [CrossRef] [PubMed]
- Helgason, B.; Perilli, E.; Schileo, E.; Taddei, F.; Brynjólfsson, S.; Viceconti, M. Mathematical relationships between bone density and mechanical properties: A literature review. Clin. Biomech. 2008, 23, 135–146. [Google Scholar] [CrossRef] [PubMed]
- Silva, V.V.; Domingues, R.Z.; Lameiras, F.S. Microstructural and mechanical study of zirconia-hydroxyapatite (ZH) composite ceramics for biomedical applications. Compos. Sci. Technol. 2001, 61, 301–310. [Google Scholar] [CrossRef]
- Wu, S.; Liu, X.; Yeung, K.W.K.; Liu, C.; Yang, X. Biomimetic porous scaffolds for bone tissue engineering. Mater. Sci. Eng. R. Rep. 2014, 80, 1–36. [Google Scholar] [CrossRef]
- Čapek, J.; Vojtěch, D. Effect of sintering conditions on the microstructural and mechanical characteristics of porous magnesium materials prepared by powder metallurgy. Mater. Sci. Eng. C 2014, 35, 21–28. [Google Scholar] [CrossRef]
- Zhang, X.; Li, X.W.; Li, J.G.; Sun, X.D. Preparation and mechanical property of a novel 3D porous magnesium scaffold for bone tissue engineering. Mater. Sci. Eng. C 2014, 42, 362–367. [Google Scholar] [CrossRef]
- Moser-Veillon, P.B. Zinc: Consumption patterns and dietary recommendations. J. Am. Diet. Assoc. 1990, 90, 1089–1093. [Google Scholar]
- Pinc, J.; Čapek, J.; Kubásek, J.; Průša, F.; Hybášek, V.; Veřtát, P.; Sedlářová, I.; Vojtěch, D. Characterization of a Zn-Ca5(PO4)3(OH) Composite with a High Content of the Hydroxyapatite Particles Prepared by the Spark Plasma Sintering Process. Metals 2020, 10, 372. [Google Scholar] [CrossRef]
- Bohner, M.; Lemaitre, J. Can bioactivity be tested in vitro with SBF solution? Biomaterials 2009, 30, 2175–2179. [Google Scholar] [CrossRef] [PubMed]
- Sopyan, I.; Mel, M.; Ramesh, S.; Khalid, K.A. Porous hydroxyapatite for artificial bone applications. Sci. Technol. Adv. Mater. 2007, 8, 116–123. [Google Scholar] [CrossRef]



| Measurement | Stabilization (s) | Initial Potential (V) | Apex Potential (V) | Finish Potential (V) | Scan Speed (mV/s) |
|---|---|---|---|---|---|
| Polarization resistance | 3600/39600 | −0.02 | − | 0.02 | 0.125 |
| Cyclic polarization | 3600 | −0.05 | 1 | −0.05 | 2 |
| Sample | Porosity (%) | Density (g/cm3) | ||
|---|---|---|---|---|
| Zn Matrix/Zn | HA | Whole Sample | ||
| Zn | 1.1 | − | 1.1 | 7.060 |
| Zn/HA8 | 8.5 | 1.5 | 10 | 6.094 |
| Samples | HV1 | FYS (MPa) | UFS (MPa) | CYS (MPa) | UCS (MPa) | Ref. |
|---|---|---|---|---|---|---|
| Zn/HA8 Ex | 44.7 ± 4.5 | 75.6 ± 8.2 | 127.6 ± 8.3 | 112.8 ± 5.1 | 168.9 ± 3.7 | This study |
| Zn/HA8 SPS | 34.3 ± 4.5 | 49.5 ± 4.4 | 63.6 ± 10.4 | 67.9 ± 7.4 | 88.9 ± 7.2 | [29] |
| Zn/HA10 SPS | 44.3 ± 2.6 | − | − | 45.2 ± 11.4 | 70.8 ± 6.3 | [36] |
| Zn Ex | 45.6 ± 2 | 106.4 ± 10.2 | − | 153.6 ± 11.1 | 243.8 ± 1.5 | This study |
| Zn SPS | 36.8 ± 1.4 | 78.8 ± 4.9 | 109.7 ± 9.88 | 92.1 ± 1.2 | 128.7 ± 1.7 | [29] |
| Cortical bone | − | − | 160–300 | 75–200 | 95–230 | [38,39] |
| Cancellous bone | − | − | − | 2–12 | 0.2–80 | [40,41] |
| Samples | Rp after 1 h (kΩ·cm2) | approx. corr. Rate (µm/a) | Rp after 12 h (kΩ·cm2) | approx. corr. Rate (µm/a) |
|---|---|---|---|---|
| Zn | 1.5 | 440.0 | 5.1 | 130.0 |
| Zn/HA8 | 1.7 | 380.0 | 9.1 | 70.0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pinc, J.; Čapek, J.; Hybášek, V.; Průša, F.; Hosová, K.; Maňák, J.; Vojtěch, D. Characterization of Newly Developed Zinc Composite with the Content of 8 wt.% of Hydroxyapatite Particles Processed by Extrusion. Materials 2020, 13, 1716. https://doi.org/10.3390/ma13071716
Pinc J, Čapek J, Hybášek V, Průša F, Hosová K, Maňák J, Vojtěch D. Characterization of Newly Developed Zinc Composite with the Content of 8 wt.% of Hydroxyapatite Particles Processed by Extrusion. Materials. 2020; 13(7):1716. https://doi.org/10.3390/ma13071716
Chicago/Turabian StylePinc, Jan, Jaroslav Čapek, Vojtěch Hybášek, Filip Průša, Klára Hosová, Jan Maňák, and Dalibor Vojtěch. 2020. "Characterization of Newly Developed Zinc Composite with the Content of 8 wt.% of Hydroxyapatite Particles Processed by Extrusion" Materials 13, no. 7: 1716. https://doi.org/10.3390/ma13071716
APA StylePinc, J., Čapek, J., Hybášek, V., Průša, F., Hosová, K., Maňák, J., & Vojtěch, D. (2020). Characterization of Newly Developed Zinc Composite with the Content of 8 wt.% of Hydroxyapatite Particles Processed by Extrusion. Materials, 13(7), 1716. https://doi.org/10.3390/ma13071716






