The Structure, Vibrational Spectra, and Thermal Expansion Study of AVO4 (A=Bi, Fe, Cr) and Co2V2O7
Abstract
1. Introduction
2. Experimental and Methods
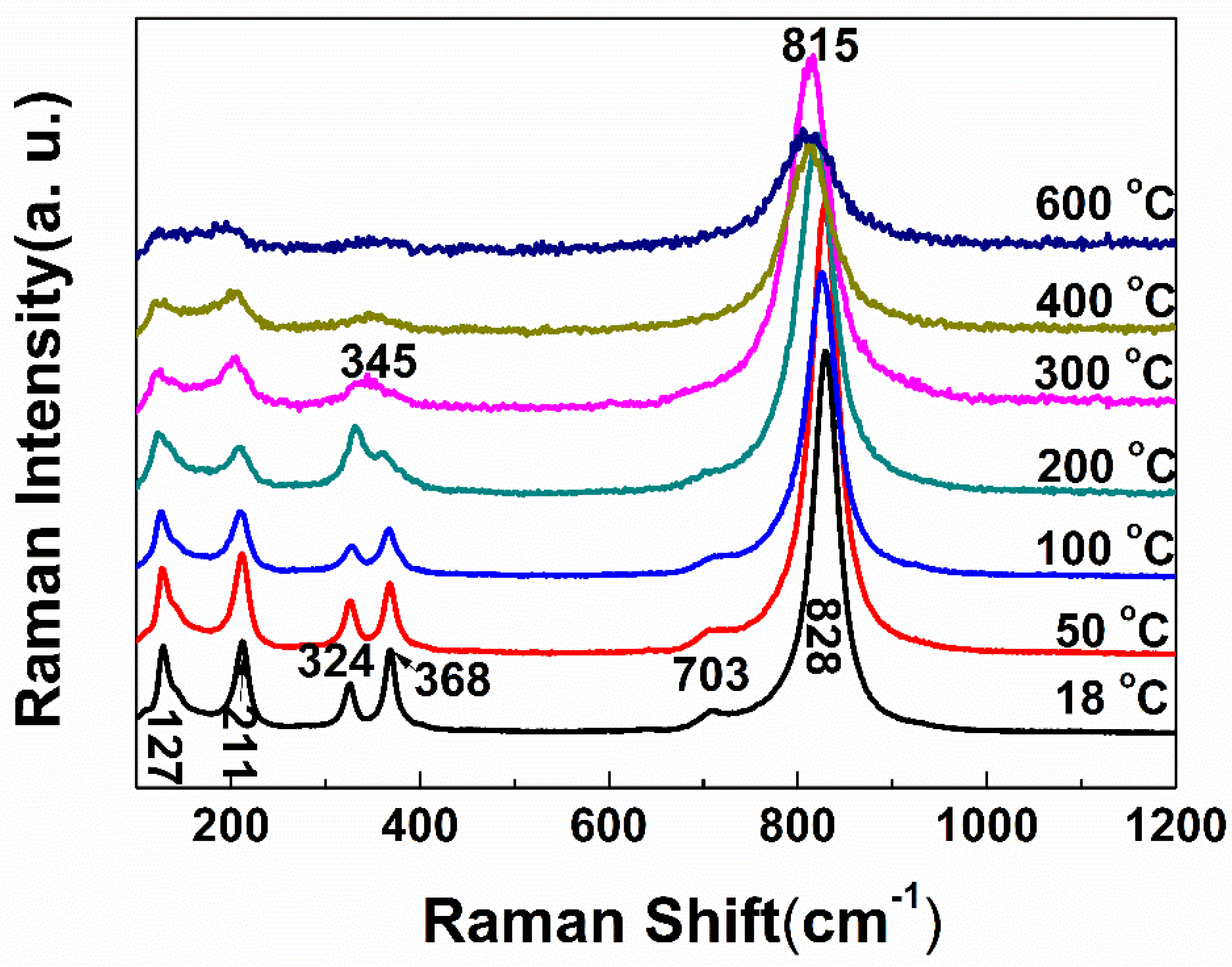
3. Results and Discussion
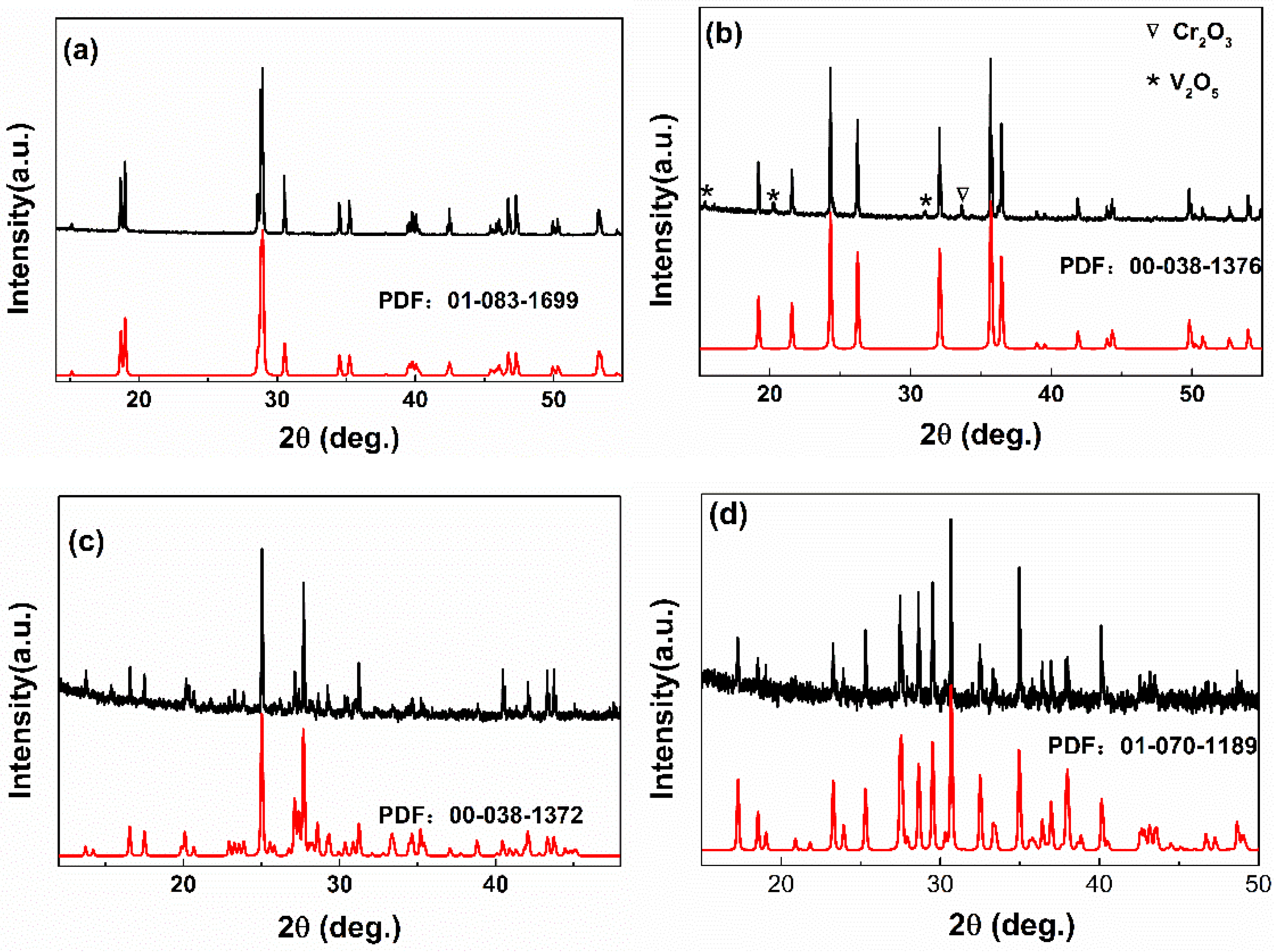
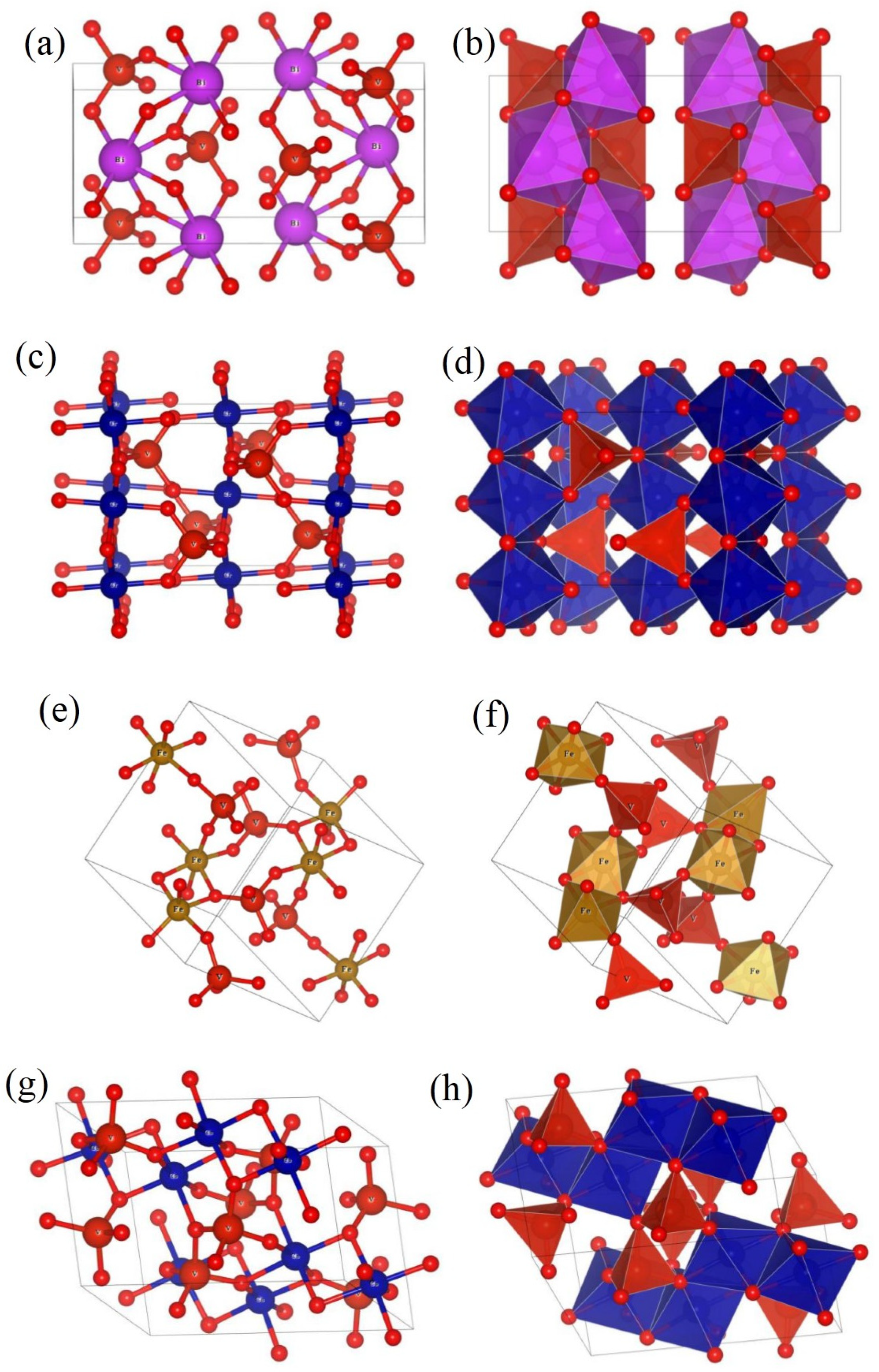
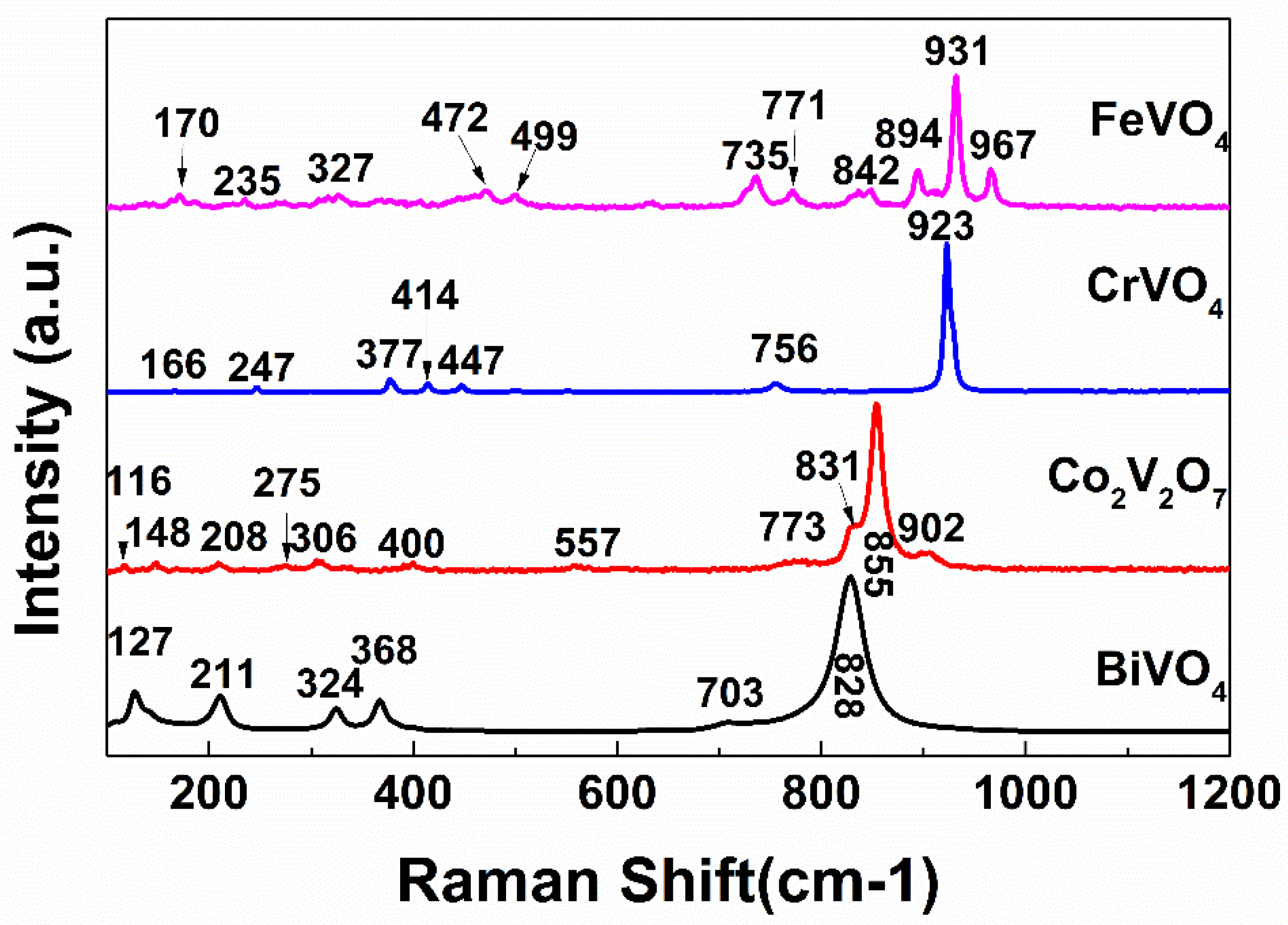
3.1. Crystal Structure Analysis
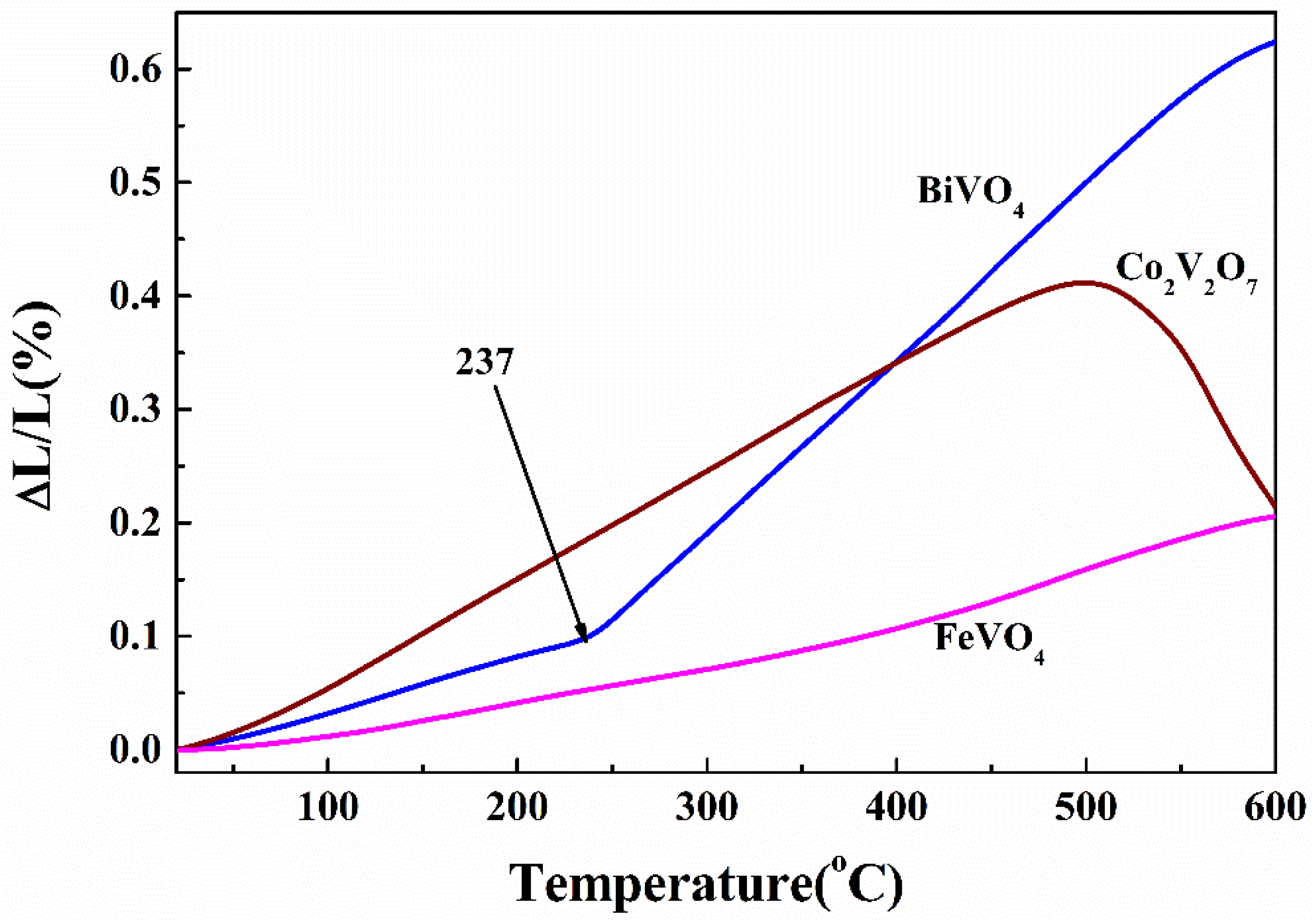
3.2. Thermal Expansion Property
4. Summary
Author Contributions
Funding
Conflicts of Interest
References
- Liu, C.; Su, J.; Zhou, J.; Guo, L. A Multi-step Ion Exchange Approach for Fabrication of Porous BiVO4 Nanorod Arrays on Transparent Conductive Substrate. ACS Sustain. Chem. Eng. 2016, 4, 4492–4497. [Google Scholar] [CrossRef]
- Huang, Z.F.; Pan, L.; Zou, J.J.; Zhang, X.; Wang, L. Nanostructured bismuth vanadate-based materials for solar-energy-driven water oxidation: A review on recent progress. Nanoscale 2014, 6, 14044–14063. [Google Scholar] [CrossRef]
- Xu, J.; Wang, W.Z.; Wang, J.; Liang, Y.J. Controlled fabrication and enhanced photocatalystic performance of BiVO4@CeO2 hollow microspheres for the visible-light-driven degradation of rhodamine B. Appl. Surf. Sci. 2015, 349, 529–537. [Google Scholar] [CrossRef]
- Wu, J.M.; Chen, Y.; Pan, L.; Wang, P.H.; Cui, Y.; Kong, D.C.; Wang, L.; Zhang, X.W.; Zou, J.J. Multilayer monoclinic BiVO4 with oxygen vacancies and V4+ species for highly efficient visible-light photoelectrochemical applications. Appl. Catal. B Environ. 2018, 221, 187–195. [Google Scholar] [CrossRef]
- Sleight, A.W.; Chen, H.-Y.; Ferretti, A.; Cox, D.E. Crystal growth and structure of BiVO4. Mater. Res. Bull. 1979, 14, 1571–1581. [Google Scholar] [CrossRef]
- Robertson, B.; Kostiner, E. Crystal structure and miissbauer effect investigation of FeVO4. J. Solid State Chem. 1972, 4, 29–37. [Google Scholar] [CrossRef]
- Lopez-Moreno, S.; Errandonea, D.; Pellicer-Porres, J.; Martinez-Garcia, D.; Patwe, S.J.; Achary, S.N.; Tyagi, A.K.; Rodriguez-Hernandez, P.; Munoz, A.; Popescu, C. Stability of FeVO4 under Pressure: An X-ray Diffraction and First-Principles Study. Inorg. Chem. 2018, 57, 7860–7876. [Google Scholar] [CrossRef]
- Sim, D.H.; Rui, X.H.; Chen, J. Direct growth of FeVO4 nanosheet arrays on stainless steel foil as high-performance binder-free Li ion battery anode. RSC Adv. 2012, 2, 3630–3633. [Google Scholar] [CrossRef]
- Liu, X.L.; Cao, Y.C.; Zheng, H.; Chen, X.; Feng, C.Q. Synthesis and modification of FeVO4 as novel anode for lithium-ion batteries. Appl. Surf. Sci. 2017, 394, 183–189. [Google Scholar] [CrossRef]
- Ghani, F.; Raza, A.; Kyung, D.G.; Kim, H.S.; Lim, C.J.; Nah, I.W. Optimization of synthesis conditions of high–tap density FeVO4 hollow microspheres via spray pyrolysis for lithium–ion batteries. Appl. Surf. Sci. 2019, 497, 143718. [Google Scholar] [CrossRef]
- Yan, N.; Xu, Y.; Li, H.J.; Chen, W. The preparation of FeVO4 as a new sort of anode material for lithium ion batteries. Mater. Lett. 2016, 165, 223–226. [Google Scholar] [CrossRef]
- Li, J.; Zhao, W.; Guo, Y.; Wei, Z.; Han, M.; He, H.; Yang, S.; Sun, C. Facile synthesis and high activity of novel BiVO4/FeVO4 hetero junction photocatalyst for degradation of metronidazole. Appl. Surf. Sci. 2015, 351, 270–279. [Google Scholar] [CrossRef]
- Wang, M.; Liu, Q.; Zhang, D. Synthesis and photocatalytic property of V2O5/FeVO4 composite novel photocatalyst. Adv. Mater. Res. 2011, 129–131, 784–788. [Google Scholar]
- Baran, E.J. Materials belonging to the CrVO4 structure type: Preparation, crystal chemistry and physicochemical properties. J. Mater. Sci. 1998, 33, 2479–2497. [Google Scholar] [CrossRef]
- Wu, F.F.; Yu, C.H.; Liu, W.X.; Wang, T.; Feng, J.K.; Xiong, S.L. Large-scale synthesis of Co2V2O7 hexagonal microplatelets under ambient conditions for highly reversible lithium storage. Mater. Chem. A 2015, 32, 16728–16736. [Google Scholar] [CrossRef]
- Sun, H.; Chen, X.; Chai, H.; Wang, Y.C.; Jia, D.Z.; Cao, Y.L.; Liu, A.J. 3D porous hydrated cobalt pyrovanadate microflowers with excellent cycling stability as cathode materials for asymmetric supercapacitor. Appl. Surf. Sci. 2019, 469, 118–124. [Google Scholar] [CrossRef]
- Ji, W.H.; Sun, Y.C.; Kumar, C.M.N.; Li, C.; Nandi, S.; Jin, W.T.; Su, Y.; Sun, X.; Lee, Y.; Harmon, B.; et al. Non-collinear magnetic structure and anisotropic magnetoelastic coupling in cobalt pyrovanadate, Co2V2O7. Phys. Rev. B 2019, 100, 134420. [Google Scholar] [CrossRef]
- Wachowski, S.; Kamecki, B.; Winiarz, P.; Dzierzgowski, K.; Mielewczyk-Gryń, A.; Gazda, M. Tailoring structural properties of lanthanum orthoniobates through an isovalent substitution on Nb-site. Inorg. Chem. Front. 2018, 5, 2157–2166. [Google Scholar] [CrossRef]
- Errandonea, D.; Manjón, F.J. Pressure effects on the structural and electronic properties of ABX4 scintillating crystals. Prog. Mater. Sci. 2008, 53, 711–773. [Google Scholar] [CrossRef]
- Bera, G.; Reddy, V.R.; Rambabu, P.; Mal, P.; Das, P.; Mohapatra, N.; Padmaja, G.; Turpu, G.R. Triclinic–monoclinic–orthorhombic (T–M–O) structural transitions in phase diagram of FeVO4-CrVO4 solid solutions. J. Appl. Phys. 2017, 122, 115101. [Google Scholar] [CrossRef]
- Løken, A.; Ricote, S.; Wachowski, S. Thermal and Chemical Expansion in Proton Ceramic Electrolytes and Compatible Electrodes. Crystals 2018, 8, 365. [Google Scholar] [CrossRef]
- Wang, N.L.; Qiu, J.; Wu, J.; You, K.Y.; Luo, H.A. A comparison of the redox properties of bulk vanadium mixed oxide catalysts. Catal. Lett. 2015, 145, 1792–1797. [Google Scholar] [CrossRef]
- Zhang, A.M.; Liu, K.; Ji, J.T.; He, C.Z.; Tian, Y.; Jineng, F.; Zhang, Q.M. Raman phonons in multiferroic FeVO4 crystals. Chin. Phys. B 2015, 24, 126301. [Google Scholar] [CrossRef]
- Yuan, H.L.; Yuan, B.H.; Li, F.; Liang, E.J. Phase transition and thermal expansion properties of ZrV2−xPxO7. Acta Phys. Sin. 2012, 22, 226502. [Google Scholar]
- Liang, E.J.; Liang, Y.; Zhao, Y.; Liu, J.; Jiang, Y.J. Low-frequency phonon modes and negative thermal expansion in A(MO4)2 (A = Zr, Hf and M = W, Mo) by Raman and Terahertz time-domain spectroscopy. J. Phys. Chem. A 2008, 112, 12582–12587. [Google Scholar] [CrossRef]
- Yu, J.; Kudo, A. Effects of Structural Variation on the Photocatalytic Performance of Hydrothermally Synthesized BiVO4. Adv. Funct. Mater. 2006, 16, 2163–2169. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, X.; Zhang, C.; Tian, D. The Structure, Vibrational Spectra, and Thermal Expansion Study of AVO4 (A=Bi, Fe, Cr) and Co2V2O7. Materials 2020, 13, 1628. https://doi.org/10.3390/ma13071628
He X, Zhang C, Tian D. The Structure, Vibrational Spectra, and Thermal Expansion Study of AVO4 (A=Bi, Fe, Cr) and Co2V2O7. Materials. 2020; 13(7):1628. https://doi.org/10.3390/ma13071628
Chicago/Turabian StyleHe, Xiaoke, Chenjun Zhang, and Ding Tian. 2020. "The Structure, Vibrational Spectra, and Thermal Expansion Study of AVO4 (A=Bi, Fe, Cr) and Co2V2O7" Materials 13, no. 7: 1628. https://doi.org/10.3390/ma13071628
APA StyleHe, X., Zhang, C., & Tian, D. (2020). The Structure, Vibrational Spectra, and Thermal Expansion Study of AVO4 (A=Bi, Fe, Cr) and Co2V2O7. Materials, 13(7), 1628. https://doi.org/10.3390/ma13071628



