Protection of LiFePO4 against Moisture
Abstract
1. Introduction
2. Experimental Section
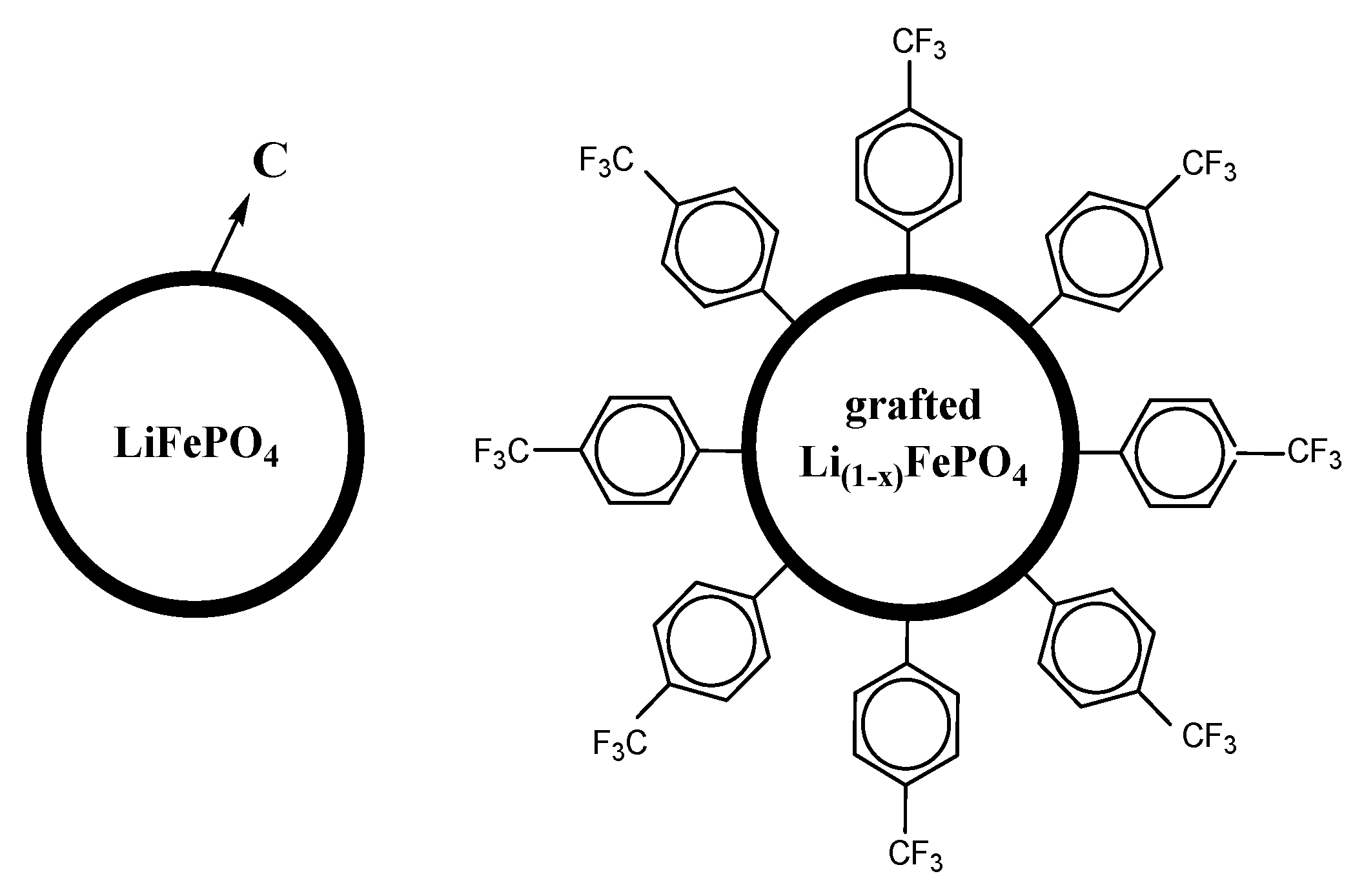
2.1. Surface Modification of LFP/C
2.2. Chemical Lithiation of Partially Oxidized LFP/C Powders
2.3. Preparation of the Samples for Iron Dissolution Experiments
2.4. Characterization
2.5. Battery Preparation and Electrochemical Testing
3. Results and Discussion
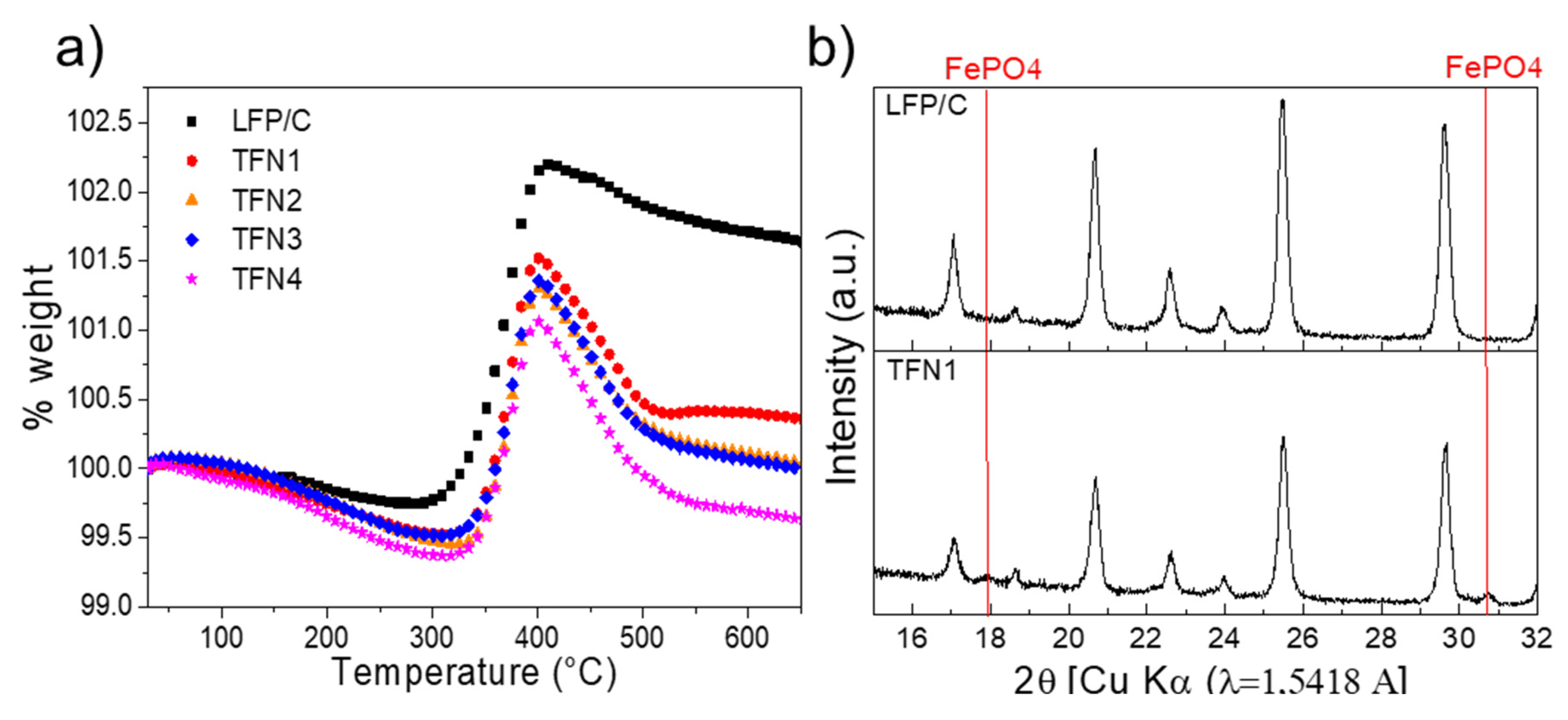
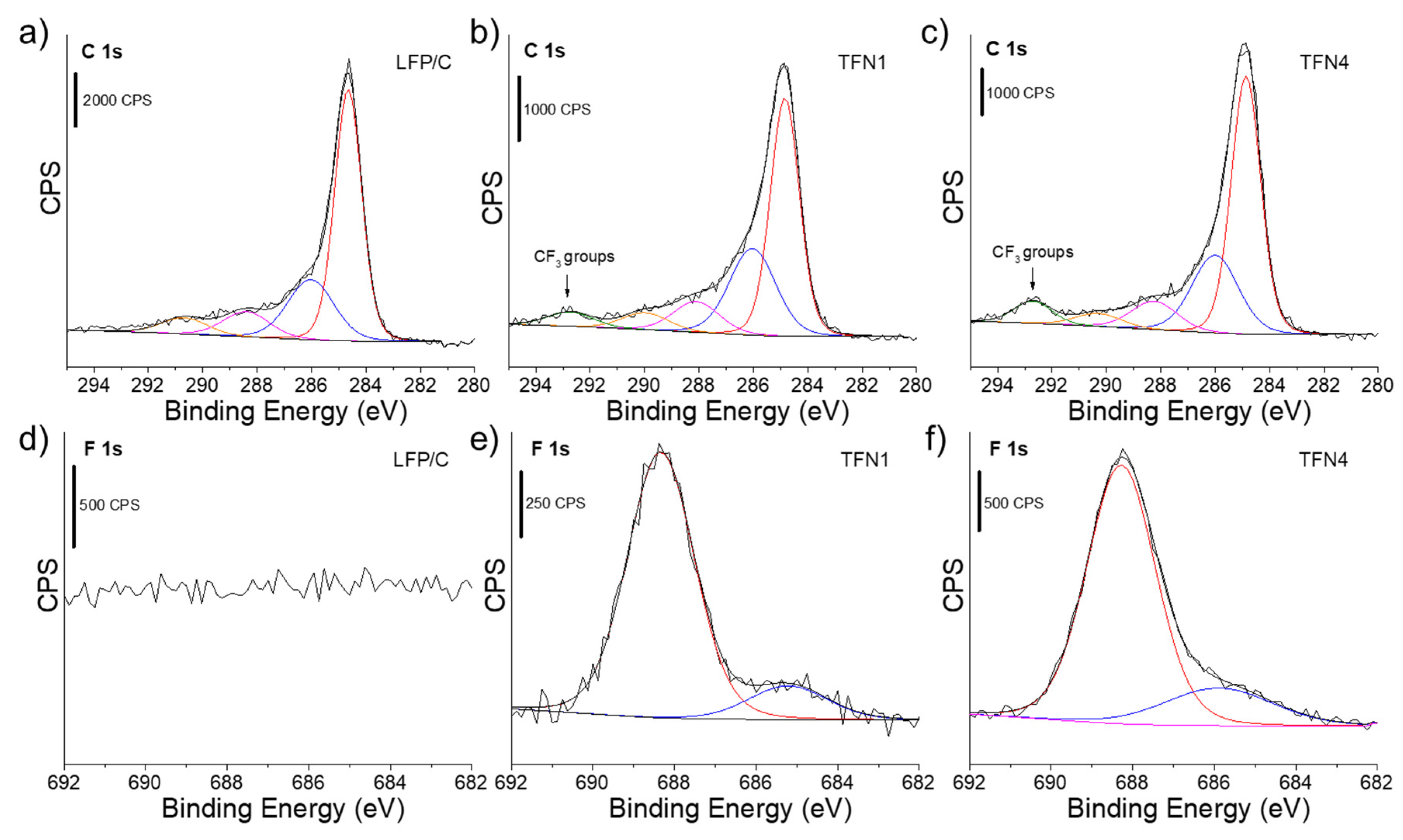
3.1. Characterization of the LFP/C Powders
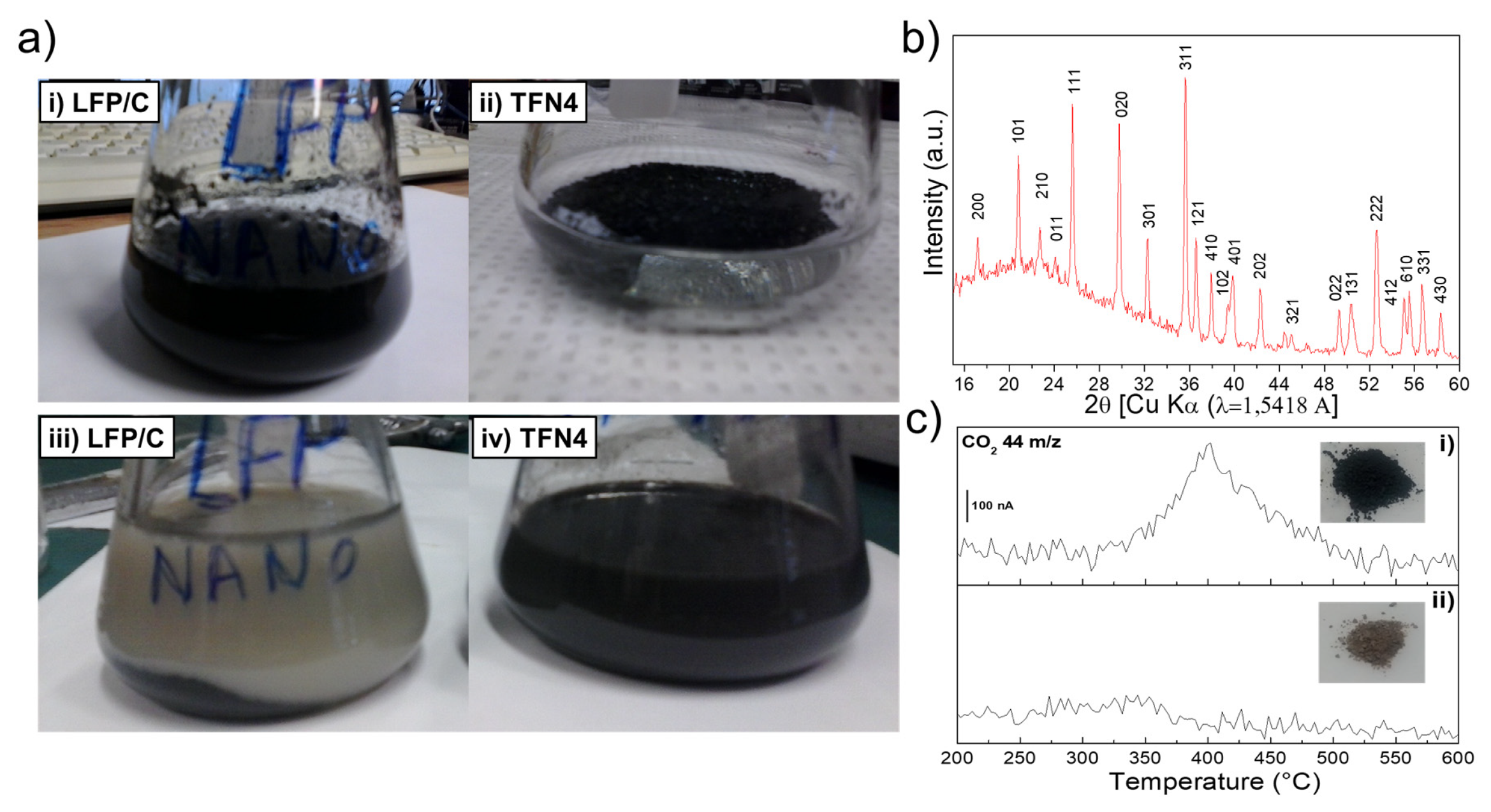
3.2. Stability of the LFP/C Powder in Water and Electrolyte
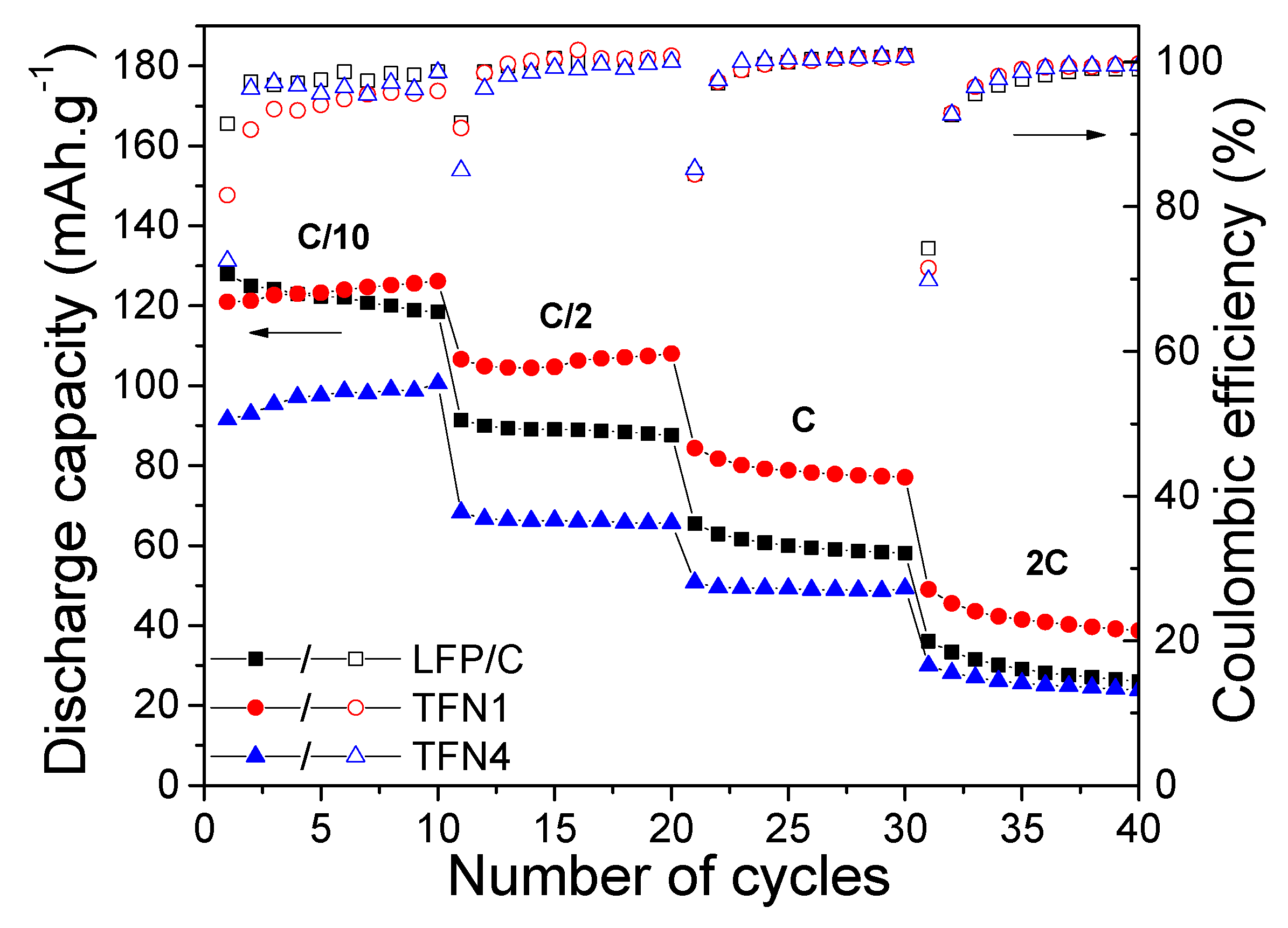
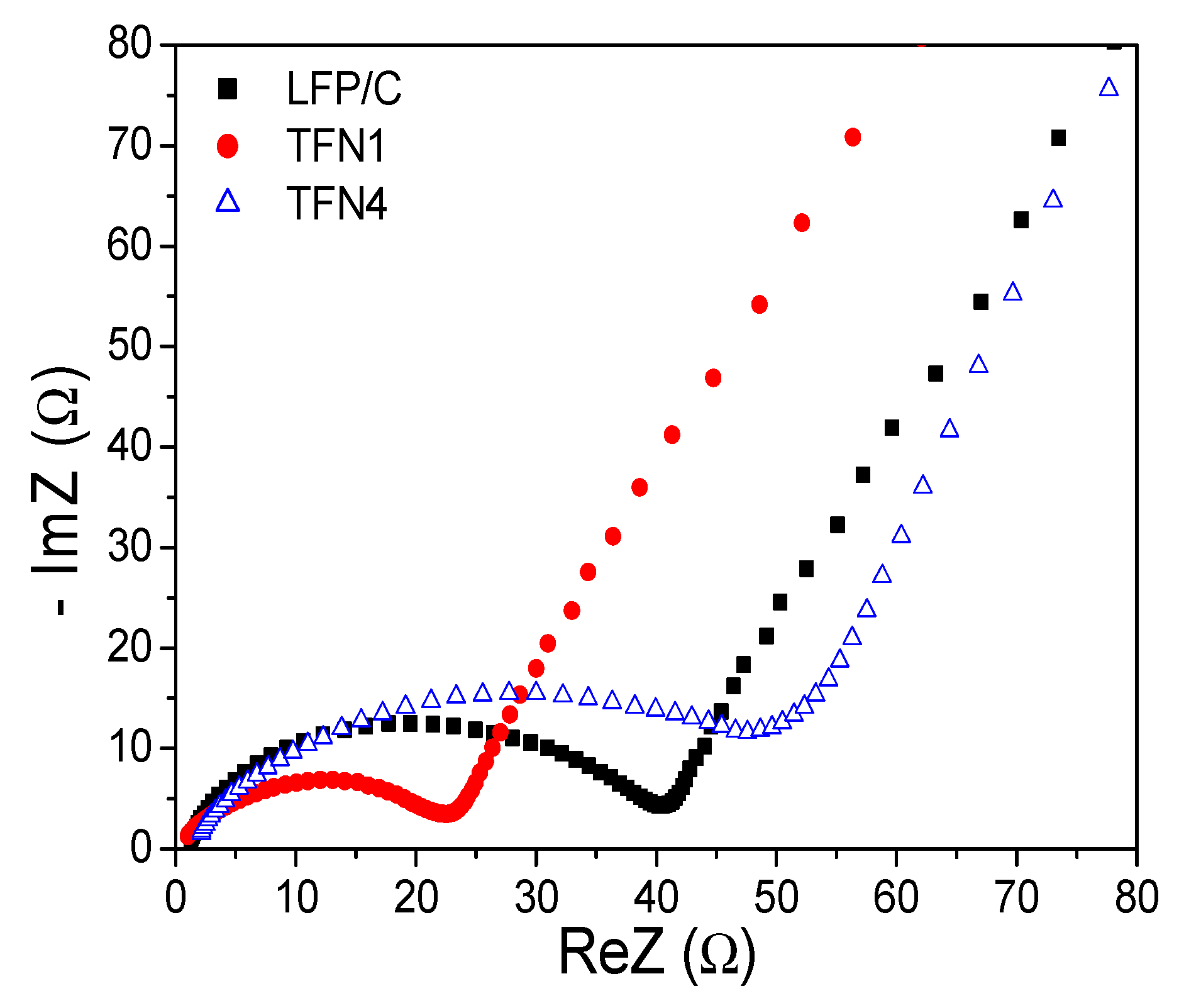
3.3. Electrochemical Performance of LFP/LTO Batteries
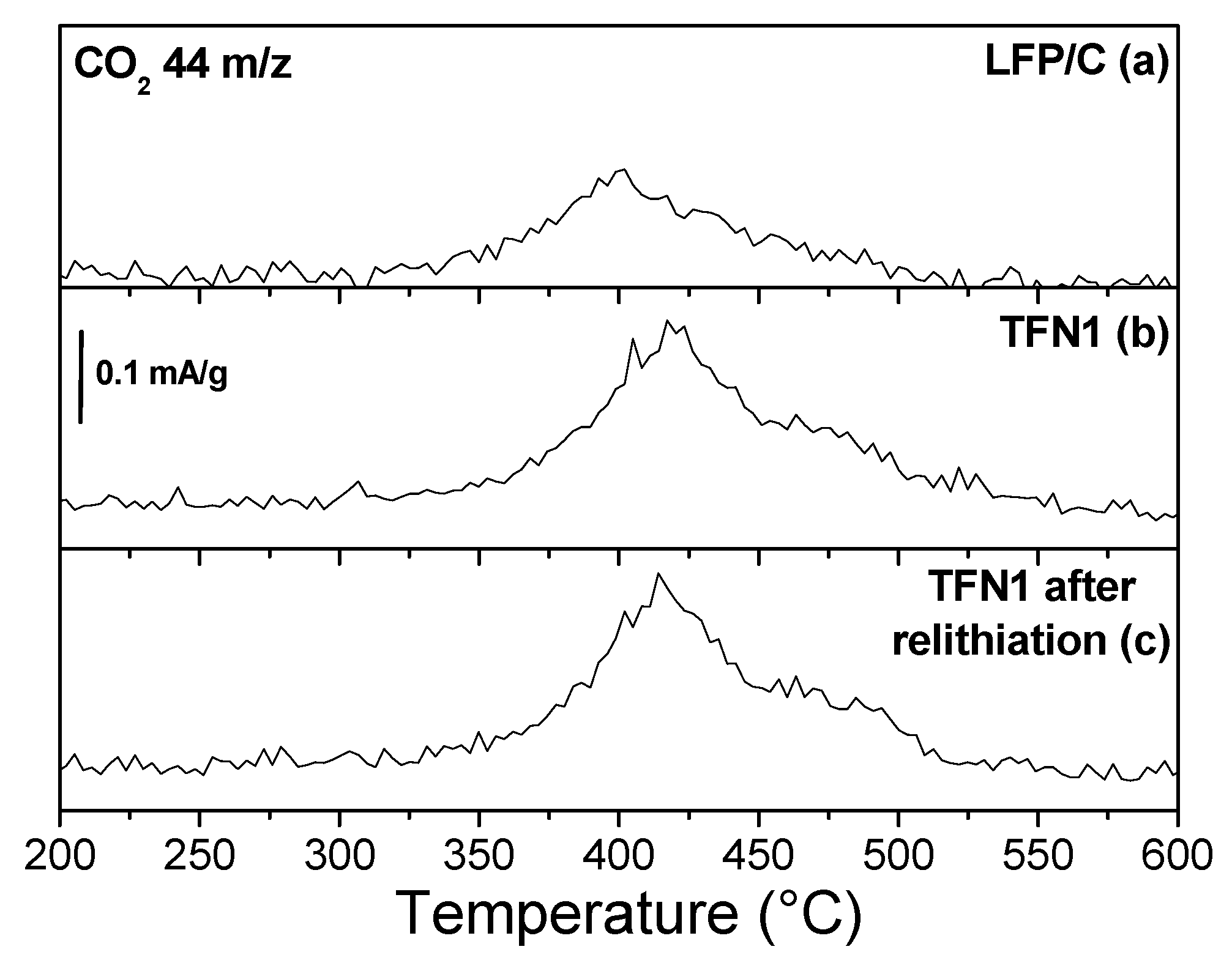
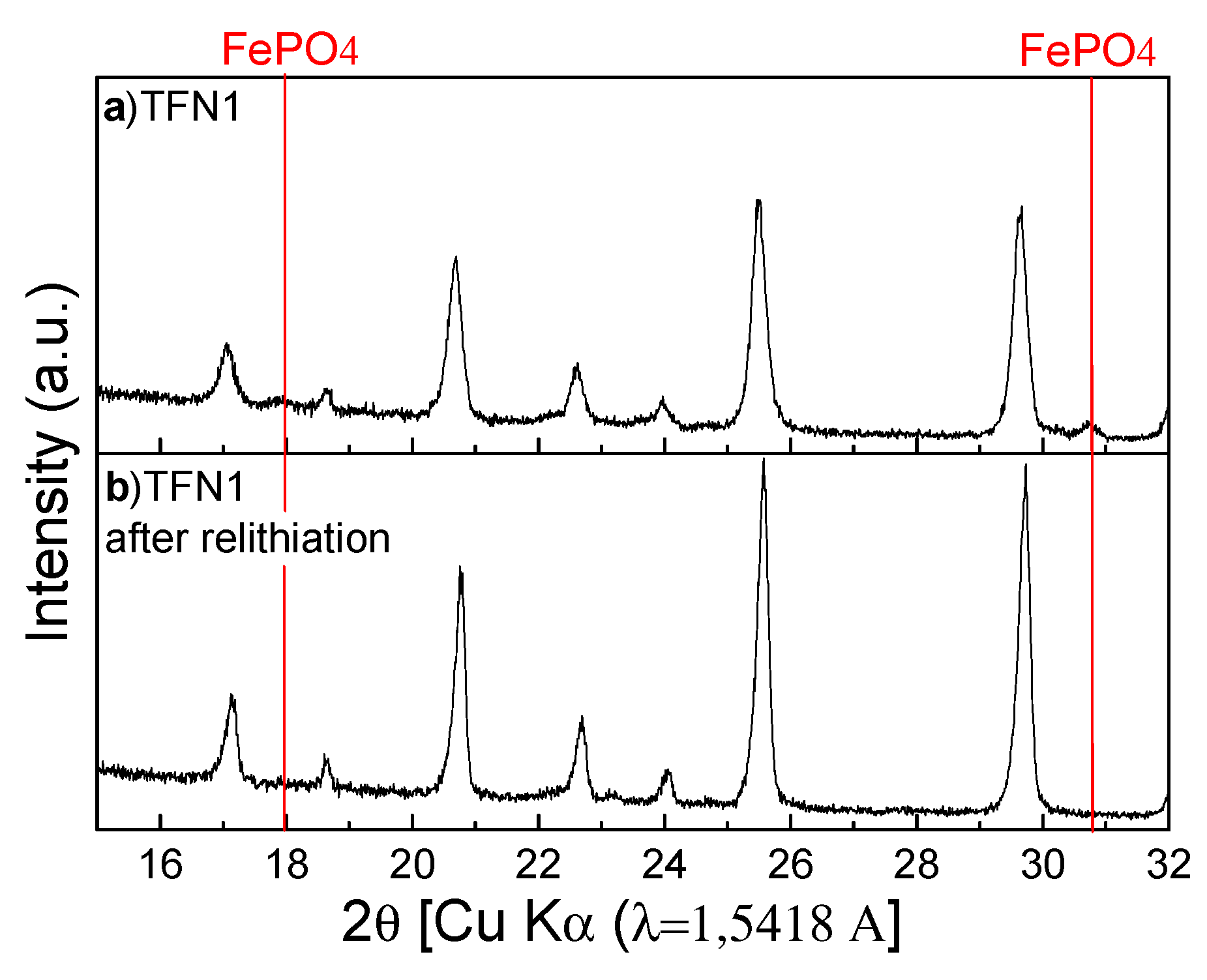
3.4. Chemical Lithiation of the Oxidized Samples
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Eftekhari, A. LiFePO4/C nanocomposites for lithium-ion batteries. J. Power Sources 2017, 343, 395–411. [Google Scholar] [CrossRef]
- Aurbach, D.; Markovsky, B.; Salitra, G.; Markevich, E.; Talyossef, Y.; Koltypin, M.; Nazar, L.F.; Ellis, B.; Kovacheva, D. Review on electrode–electrolyte solution interactions, related to cathode materials for Li-ion batteries. J. Power Sources 2007, 165, 491–499. [Google Scholar] [CrossRef]
- Aurbach, D.; Markovsky, B.; Rodkin, A.; Levi, E.; Cohen, Y.; Kim, H.-J.; Schmidt, M. On the capacity fading of LiCoO2 intercalation electrodes. Electrochim. Acta 2002, 47, 4291–4306. [Google Scholar] [CrossRef]
- Dupré, N.; Martin, J.-F.; Degryse, J.; Fernandez, V.; Soudan, P.; Guyomard, D. Aging of the LiFePO4 positive electrode interface in electrolyte. J. Power Sources 2010, 195, 7415–7425. [Google Scholar] [CrossRef]
- Zaghib, K.; Dontigny, M.; Charest, P.; Labrecque, J.; Guerfi, A.; Kopéc, M.; Mauger, A.; Gendron, F.; Julien, C.M. Aging of LiFePO4 upon exposure to H2O. J. Power Sources 2008, 185, 698–710. [Google Scholar] [CrossRef]
- Martin, J.-F.; Yamada, A.; Kobayashi, G.; Nishimura, S.; Kanno, R.; Guyomard, D.; Dupré, N. ir Exposure Effect on LiFePO4. Electrochem. Solid-State Lett. 2008, 11, A12–A16. [Google Scholar] [CrossRef]
- Martin, J.-F.; Cuisinier, M.; Dupré, N.; Yamada, A.; Kanno, R.; Guyomard, D. More on the reactivity of olivine LiFePO4 nano-particles with atmosphere at moderate temperature. J. Power Sources 2011, 196, 2155–2163. [Google Scholar] [CrossRef]
- Delaporte, N.; Zaghib, K.; Bélanger, D. Process for Protecting Electrode Materials against Moisture. U.S. Patent No. 20180138512, 27 May 2015. [Google Scholar]
- Weissmann, M.; Baranton, S.; Clacens, J.-M.; Coutanceau, C. Modification of hydrophobic/hydrophilic properties of Vulcan XC72 carbon powder by grafting of trifluoromethylphenyl and phenylsulfonic acid groups. Carbon 2010, 48, 2755–2764. [Google Scholar] [CrossRef]
- Shul, G.; Parent, R.; Mosqueda, A.H.; Bélanger, D. Localized In situ Generation of Diazonium Cations by Electrocatalytic Formation of a Diazotization Reagent. ACS Appl. Mater. Interfaces 2013, 5, 1468–1473. [Google Scholar] [CrossRef]
- Yu, Y.; Kosbach, L. Modified Carbon Products Useful in Gas Diffusion Electrodes. U.S. Patent No. 6,399,202, 12 October 1999. [Google Scholar]
- Bélanger, D.; Pinson, J. Electrografting: A powerful method for surface modification. Chem. Soc. Rev. 2011, 40, 3995. [Google Scholar] [CrossRef]
- Delamar, M.; Hitmi, R.; Pinson, J.; Saveant, J.M. Covalent modification of carbon surfaces by grafting of functionalized aryl radicals produced from electrochemical reduction of diazonium salts. J. Am. Chem. Soc. 1992, 114, 5883–5884. [Google Scholar] [CrossRef]
- Toupin, M.; Belanger, D. Thermal Stability Study of Aryl Modified Carbon Black by in Situ Generated Diazonium Salt. J. Phys. Chem. C 2007, 111, 5394–5401. [Google Scholar] [CrossRef]
- Adenier, A.; Barré, N.; Cabet-Deliry, E.; Chaussé, A.; Griveau, S.; Mercier, F.; Pinson, J.; Vautrin-Ul, C. Study of the spontaneous formation of organic layers on carbon and metal surfaces from diazonium salts. Surf. Sci. 2006, 600, 4801–4812. [Google Scholar] [CrossRef]
- Combellas, C.; Delamar, M.; Kanoufi, F.; Pinson, J.; Podvorica, F.I. Spontaneous Grafting of Iron Surfaces by Reduction of Aryldiazonium Salts in Acidic or Neutral Aqueous Solution. Application to the Protection of Iron against Corrosion. Chem. Mater. 2005, 17, 3968–3975. [Google Scholar] [CrossRef]
- Lehr, J.; Williamson, B.; Flavel, B.S.; Downard, A.J. Reaction of Gold Substrates with Diazonium Salts in Acidic Solution at Open-Circuit Potential. Langmuir 2009, 25, 13503–13509. [Google Scholar] [CrossRef]
- Belmont, J.A. Aqueous Inks and Coatings Containing Modified Carbon Products. U.S. Patent No. 5,672,198A, 15 December 1994. [Google Scholar]
- Adams, C.E.; Belmont, J.A.; Amici, R.M. Compositions of Modified Carbon Products and Amphiphilic Ions and Methods of Using the Same. U.S. Patent No. 5,698,016A, 14 June 1996. [Google Scholar]
- Adams, C.E.; Belmont, J.A. Modified Carbon Products and Inks and Coatings Containing Modified Carbon Products. U.S. Patent No. 5,707,432A, 14 June 1996. [Google Scholar]
- Barrière, F.; Downard, A.J. Covalent modification of graphitic carbon substrates by non-electrochemical methods. J. Solid State Electrochem. 2008, 12, 1231–1244. [Google Scholar] [CrossRef]
- Mahouche-Chergui, S.; Gam-Derouich, S.; Mangeney, C.; Chehimi, M.M. Aryl diazonium salts: A new class of coupling agents for bonding polymers, biomacromolecules and nanoparticles to surfaces. Chem. Soc. Rev. 2011, 40, 4143–4166. [Google Scholar] [CrossRef] [PubMed]
- Shaidarova, L.G.; Budnikov, G.K. Chemically modified electrodes based on noble metals, polymer films, or their composites in organic voltammetry. J. Anal. Chem. 2008, 63, 922–942. [Google Scholar] [CrossRef]
- Mandler, D.; Turyan, I. Applications of self-assembled monolayers in electroanalytical chemistry. Electroanalysis 1996, 8, 207–213. [Google Scholar] [CrossRef]
- Martin, C.; Alias, M.; Christien, F.; Crosnier, O.; Bélanger, D.; Brousse, T. Graphite-Grafted Silicon Nanocomposite as a Negative Electrode for Lithium-Ion Batteries. Adv. Mater. 2009, 21, 4735–4741. [Google Scholar] [CrossRef]
- Yang, S.; Li, G.; Zhu, Q.; Pan, Q. Covalent binding of Si nanoparticles to graphene sheets and its influence on lithium storage properties of Si negative electrode. J. Mater. Chem. 2012, 22, 3420–3425. [Google Scholar] [CrossRef]
- Yang, S.; Pan, Q.; Liu, J. Improving the cycleability of Si anodes by covalently grafting with 4-carboxyphenyl groups. Electrochem. Commun. 2010, 12, 479–482. [Google Scholar] [CrossRef]
- Martin, C.; Crosnier, O.; Retoux, R.; Bélanger, D.; Schleich, D.M.; Brousse, T. Chemical Coupling of Carbon Nanotubes and Silicon Nanoparticles for Improved Negative Electrode Performance in Lithium-Ion Batteries. Adv. Funct. Mater. 2011, 21, 3524–3530. [Google Scholar] [CrossRef]
- Assresahegn, B.D.; Bélanger, D. Synthesis of binder-like molecules covalently linked to silicon nanoparticles and application as anode material for lithium-ion batteries without the use of electrolyte additives. J. Power Sources 2017, 345, 190–201. [Google Scholar] [CrossRef]
- Tanguy, F.; Gaubicher, J.; Gaillot, A.-C.; Guyomard, D.; Pinson, J. Lowering interfacial chemical reactivity of oxide materials for lithium batteries. A molecular grafting approach. J. Mater. Chem. 2009, 19, 4771. [Google Scholar] [CrossRef]
- Madec, L.; Humbert, B.; Lestriez, B.; Brousse, T.; Cougnon, C.; Guyomard, D.; Gaubicher, J. Covalent vs. non-covalent redox functionalization of C–LiFePO4 based electrodes. J. Power Sources 2013, 232, 246–253. [Google Scholar] [CrossRef]
- Delaporte, N.; Perea, A.; Amin, R.; Zaghib, K.; Bélanger, D. Chemically grafted carbon-coated LiFePO4 using diazonium chemistry. J. Power Sources 2015, 280, 246–255. [Google Scholar] [CrossRef]
- Delaporte, N.; Perea, A.; Lebègue, E.; Ladouceur, S.; Zaghib, K.; Bélanger, D. Increasing the Affinity Between Carbon-Coated LiFePO4/C Electrodes and Conventional Organic Electrolyte by Spontaneous Grafting of a Benzene-Trifluoromethylsulfonimide Moiety. ACS Appl. Mater. Interfaces 2015, 7, 18519–18529. [Google Scholar] [CrossRef]
- Delaporte, N.; Zaghib, K.; Bélanger, D. In situ formation of bromobenzene diazonium ions and their spontaneous reaction with carbon-coated LiFePO4 in organic media. New J. Chem. 2016, 40, 6135–6140. [Google Scholar] [CrossRef]
- Aurbach, D.; Markovsky, B.; Rodkin, A.; Cojocaru, M.; Levi, E.; Kim, H.-J. An analysis of rechargeable lithium-ion batteries after prolonged cycling. Electrochim. Acta 2002, 47, 1899–1911. [Google Scholar] [CrossRef]
- Belharouak, I.; Johnson, C.; Amine, K. Synthesis and electrochemical analysis of vapor-deposited carbon-coated LiFePO4. Electrochem. Commun. 2005, 7, 983–988. [Google Scholar] [CrossRef]
- Hamelet, S.; Gibot, P.; Casas-Cabanas, M.; Bonnin, D.; Grey, C.P.; Cabana, J.; Leriche, J.-B.; Rodriguez-Carvajal, J.; Courty, M.; Levasseur, S.; et al. The effects of moderate thermal treatments under air on LiFePO4-based nano powders. J. Mater. Chem. 2009, 19, 3979–3991. [Google Scholar] [CrossRef]
- Ueda, M.; Ohe, M.; Kim, J.-H.; Yonezawa, S.; Takashima, M. Effects of surface fluorination on the electrochemical properties and thermal stability of LiFePO4 cathode for lithium-ion batteries. J. Fluor. Chem. 2013, 149, 88–94. [Google Scholar] [CrossRef]
- Delacourt, C.; Rodriguez-Carvajal, J.; Schmitt, B.; Tarascon, J.-M.; Masquelier, C. Crystal chemistry of the olivine-type LixFePO4 system (0 ⩽ x ⩽ 1) between 25 and 370 °C. Solid State Sci. 2005, 7, 1506–1516. [Google Scholar] [CrossRef]
- Zaghib, K.; Guerfi, A.; Hovington, P.; Vijh, A.; Trudeau, M.; Mauger, A.; Goodenough, J.B.; Julien, C. Review and analysis of nanostructured olivine-based lithium recheargeable batteries: Status and trends. J. Power Sources 2013, 232, 357–369. [Google Scholar] [CrossRef]
- Meethong, N.; Huang, H.-Y.S.; Carter, W.C.; Chiang, Y.-M. Size-Dependent Lithium Miscibility Gap in Nanoscale Li1-xFePO4. Electrochem. Solid-State Lett. 2007, 10, A134–A138. [Google Scholar] [CrossRef]
- Meethong, N.; Huang, H.-Y.S.; Speakman, S.A.; Carter, W.C.; Chiang, Y.-M. Strain Accommodation during Phase Transformations in Olivine-Based Cathodes as a Materials Selection Criterion for High-Power Rechargeable Batteries. Adv. Funct. Mater. 2007, 17, 1115–1123. [Google Scholar] [CrossRef]
- Kobayashi, G.; Nishimura, S.-I.; Park, M.-S.; Kanno, R.; Yashima, M.; Ida, T.; Yamada, A. Isolation of Solid Solution Phases in Size-Controlled LixFePO4 at Room Temperature. Adv. Funct. Mater. 2009, 19, 395–403. [Google Scholar] [CrossRef]
- Badi, S.-P.; Wagemaker, M.; Ellis, B.; Singh, D.P.; Borghols, W.J.H.; Kan, W.; Ryan, D.H.; Mulder, F.; Nazar, L.F. Direct synthesis of nanocrystalline Li0.90FePO4: Observation of phase segregation of anti-site defects on delithiation. J. Mater. Chem. 2011, 21, 10085–10093. [Google Scholar] [CrossRef]
- Lebègue, E.; Brousse, T.; Gaubicher, J.; Cougnon, C. Spontaneous arylation of activated carbon from aminobenzene organic acids as source of diazonium ions in mild conditions. Electrochim. Acta 2013, 88, 680–687. [Google Scholar] [CrossRef]
- Zhao, M.; Cao, Y.; Liu, X.; Deng, J.; Li, D.; Gu, H. Effect of nitrogen atomic percentage on N+-bombarded MWCNTs in cytocompatibility and hemocompatibility. Nanoscale Res. Lett. 2014, 9, 142. [Google Scholar] [CrossRef] [PubMed]
- Laforgue, A.; Addou, T.; Belanger, D. Characterization of the Deposition of Organic Molecules at the Surface of Gold by the Electrochemical Reduction of Aryldiazonium Cations. Langmuir 2005, 21, 6855–6865. [Google Scholar] [CrossRef] [PubMed]
- Yun, S.-M.; Kim, J.-W.; Jung, M.-J.; Nho, Y.-C.; Kang, P.H.; Lee, Y.S. An XPS Study of Oxyfluorinated Multiwalled Carbon Nano Tubes. Carbon Lett. 2007, 8, 292–298. [Google Scholar] [CrossRef]
- Veith, G.M.; Dudney, N.J. Current Collectors for Rechargeable Li-Air Batteries. J. Electrochem. Soc. 2011, 158, A658–A663. [Google Scholar] [CrossRef]
- He, P.; Liu, J.-L.; Cui, W.-J.; Luo, J.; Xia, Y.-Y. Investigation on capacity fading of LiFePO4 in aqueous electrolyte. Electrochim. Acta 2011, 56, 2351–2357. [Google Scholar] [CrossRef]
- Zaghib, K.; Simoneau, M.; Armand, M.; Gauthier, M. Electrochemical study of Li4Ti5O12 as negative electrode for Li-ion polymer rechargeable batteries. J. Power Sources 1999, 81, 300–305. [Google Scholar] [CrossRef]
- Lee, J.; Kumar, P.; Lee, G.; Moudgil, B.M.; Singh, R.K. Electrochemical performance of surfactant-processed LiFePO4 as a cathode material for lithium-ion rechargeable batteries. Ionics 2012, 19, 371–378. [Google Scholar] [CrossRef]
- Zhuang, Q.-C.; Qiu, X.-Y.; Xu, S.-D.; Qiang, Y.-H.; Su, S.-G. Diagnosis of Electrochemical Impedance Spectroscopy in Lithium-Ion Batteries. Lithium Ion Batter.-New Dev. 2012. [Google Scholar] [CrossRef]
- Galoustov, K.; Anthonisen, M.; Ryan, D.; MacNeil, D.D. Characterization of two lithiation reactions starting with an amorphous FePO4 precursor. J. Power Sources 2011, 196, 6893–6897. [Google Scholar] [CrossRef]
- Jeong, S.; Böckenfeld, N.; Balducci, A.; Winter, M.; Passerini, S. Natural cellulose as binder for lithium battery electrodes. J. Power Sources 2012, 199, 331–335. [Google Scholar] [CrossRef]
- Kvasha, A.; Urdampilleta, I.; De Meatza, I.; Bengoechea, M.; Blázquez, J.A.; Yate, L.; Miguel, O.; Grande, H.-J. Towards high durable lithium ion batteries with waterborne LiFePO4 electrodes. Electrochim. Acta 2016, 215, 238–246. [Google Scholar] [CrossRef]
- Porcher, W.; Lestriez, B.; Jouanneau, S.; Guyomard, D. Optimizing the surfactant for the aqueous processing of LiFePO4 composite electrodes. J. Power Sources 2010, 195, 2835–2843. [Google Scholar] [CrossRef]
- Kim, H.; Hong, J.; Park, K.-Y.; Kim, H.; Kim, S.-W.; Kang, K. Aqueous Rechargeable Li and Na Ion Batteries. Chem. Rev. 2014, 114, 11788–11827. [Google Scholar] [CrossRef] [PubMed]
- Wood, M.; Li, J.; Ruther, R.E.; Du, Z.; Self, E.C.; Meyer, H.; Daniel, C.; Belharouak, I.; Wood, D.L. Chemical stability and long-term cell performance of low-cobalt, Ni-Rich cathodes prepared by aqueous processing for high-energy Li-Ion batteries. Energy Storage Mater. 2020, 24, 188–197. [Google Scholar] [CrossRef]

| Sample | Amine (mmol) | Tert-butyl Nitrite (mmol) | wt.% of Grafted Groups (TGA) | x in Li(1-x)FePO4 by TGA | x in Li(1-x)FePO4 by ICP |
|---|---|---|---|---|---|
| LFP/C | - | - | - | 0 | ~ 0 |
| TFN1 | 2 | 6 | 0.55 | 0.20 | 0.20 |
| TFN2 | 4 | 12 | 0.60 | 0.27 | 0.25 |
| TFN3 | 6 | 18 | 0.71 | 0.26 | 0.27 |
| TFN4 | 8 | 24 | 0.78 | 0.32 | 0.28 |
| Sample | Atomic Concentration (%) | |||||||
|---|---|---|---|---|---|---|---|---|
| Amine (mmol) | Tert-butyl Nitrite (mmol) | C 1s | O 1s | N 1s | P 2p | Fe 2p | F 1s | |
| LFP/C | - | - | 42.5 | 41.6 | 0.2 | 10.4 | 5.3 | 0.0 |
| TFN1 | 2 | 6 | 38.0 | 43.3 | 0.5 | 9.5 | 5.7 | 3.0 |
| TFN4 | 8 | 24 | 43.9 | 37.5 | 0.6 | 8.2 | 4.3 | 5.5 |
| Sample-Conditions | x in Li(1-x)FePO4 by ICP * | Δx |
|---|---|---|
| LFP/C | ~ 0 | - |
| LFP/C–24 h of immersion, ambient temperature | 0.34 | 0.34 |
| LFP/C–18 h of immersion, 60 ˚C | 0.29 | 0.29 |
| TFN4 | 0.28 | - |
| TFN4–24 h of immersion, ambient temperature | 0.30 | 0.02 |
| TFN4–18 h of immersion, 60 ˚C | 0.32 | 0.04 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Delaporte, N.; Trudeau, M.L.; Bélanger, D.; Zaghib, K. Protection of LiFePO4 against Moisture. Materials 2020, 13, 942. https://doi.org/10.3390/ma13040942
Delaporte N, Trudeau ML, Bélanger D, Zaghib K. Protection of LiFePO4 against Moisture. Materials. 2020; 13(4):942. https://doi.org/10.3390/ma13040942
Chicago/Turabian StyleDelaporte, Nicolas, Michel L. Trudeau, Daniel Bélanger, and Karim Zaghib. 2020. "Protection of LiFePO4 against Moisture" Materials 13, no. 4: 942. https://doi.org/10.3390/ma13040942
APA StyleDelaporte, N., Trudeau, M. L., Bélanger, D., & Zaghib, K. (2020). Protection of LiFePO4 against Moisture. Materials, 13(4), 942. https://doi.org/10.3390/ma13040942








