The Interaction of Biofoulants and Calcareous Deposits on Corrosion Performance of Q235 in Seawater
Abstract
1. Introduction
2. Materials and Methods
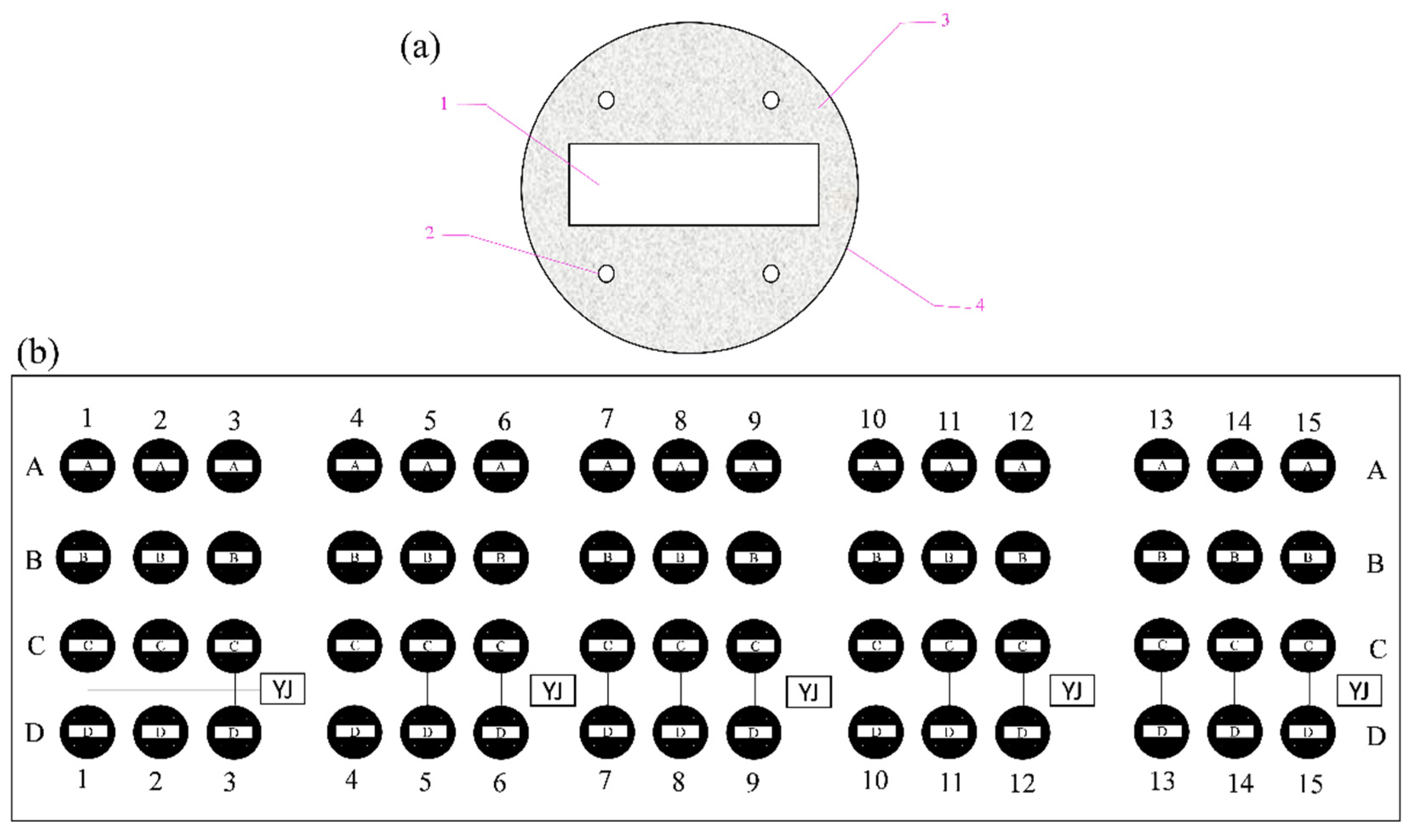
2.1. Experimental Conditions
2.2. The Chemical Composition of Q235
2.3. Electrochemical Procedure and Measurements
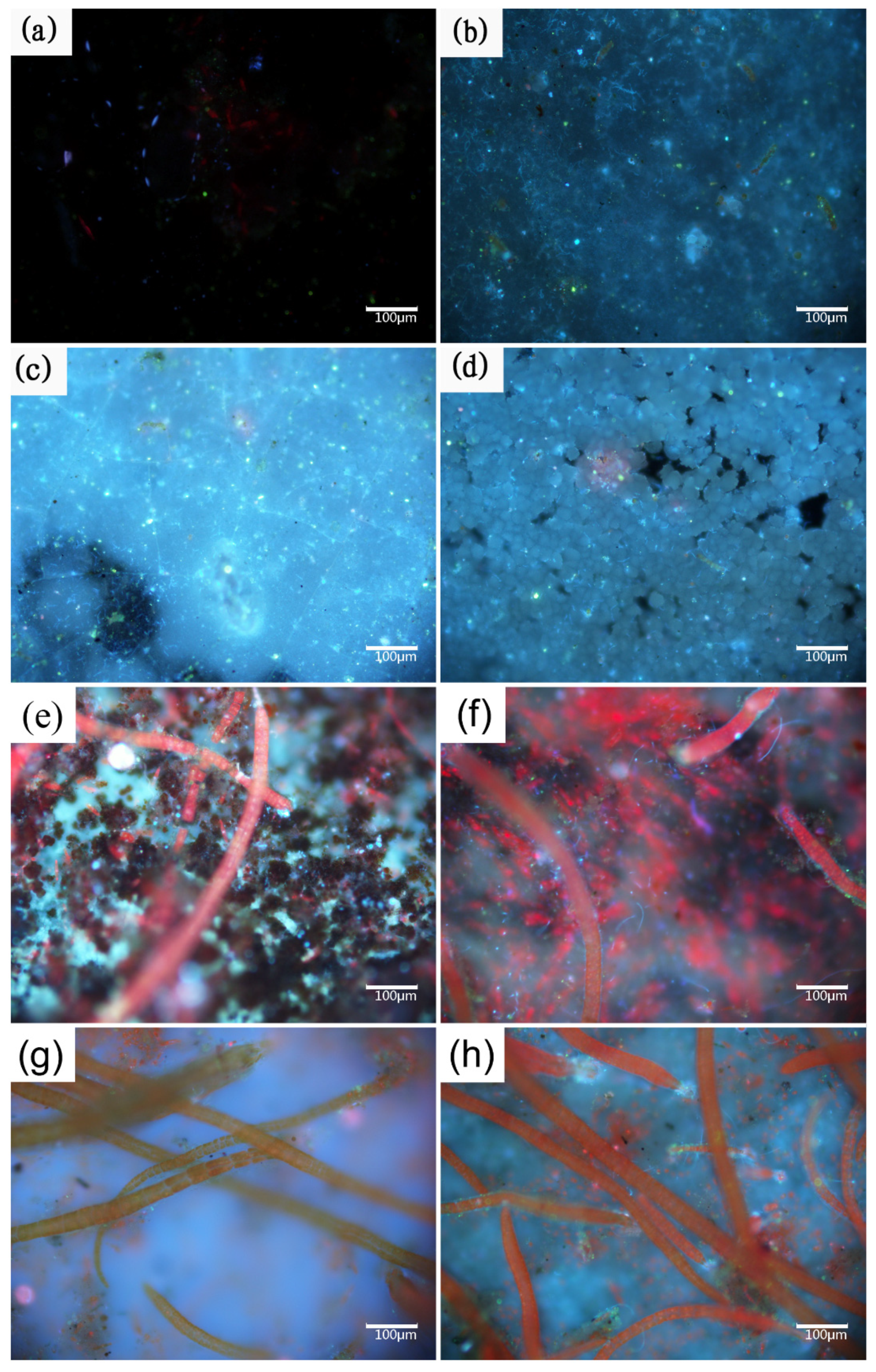
2.4. Observation of Fouling Organisms by Fluorescence Microscopy
2.5. SEM Observations of Microorganisms
2.6. SEM Analysis of the Corrosion Morphology
3. Results and Discussion
3.1. Biofouling Process
3.2. Calcareous Deposits Formation
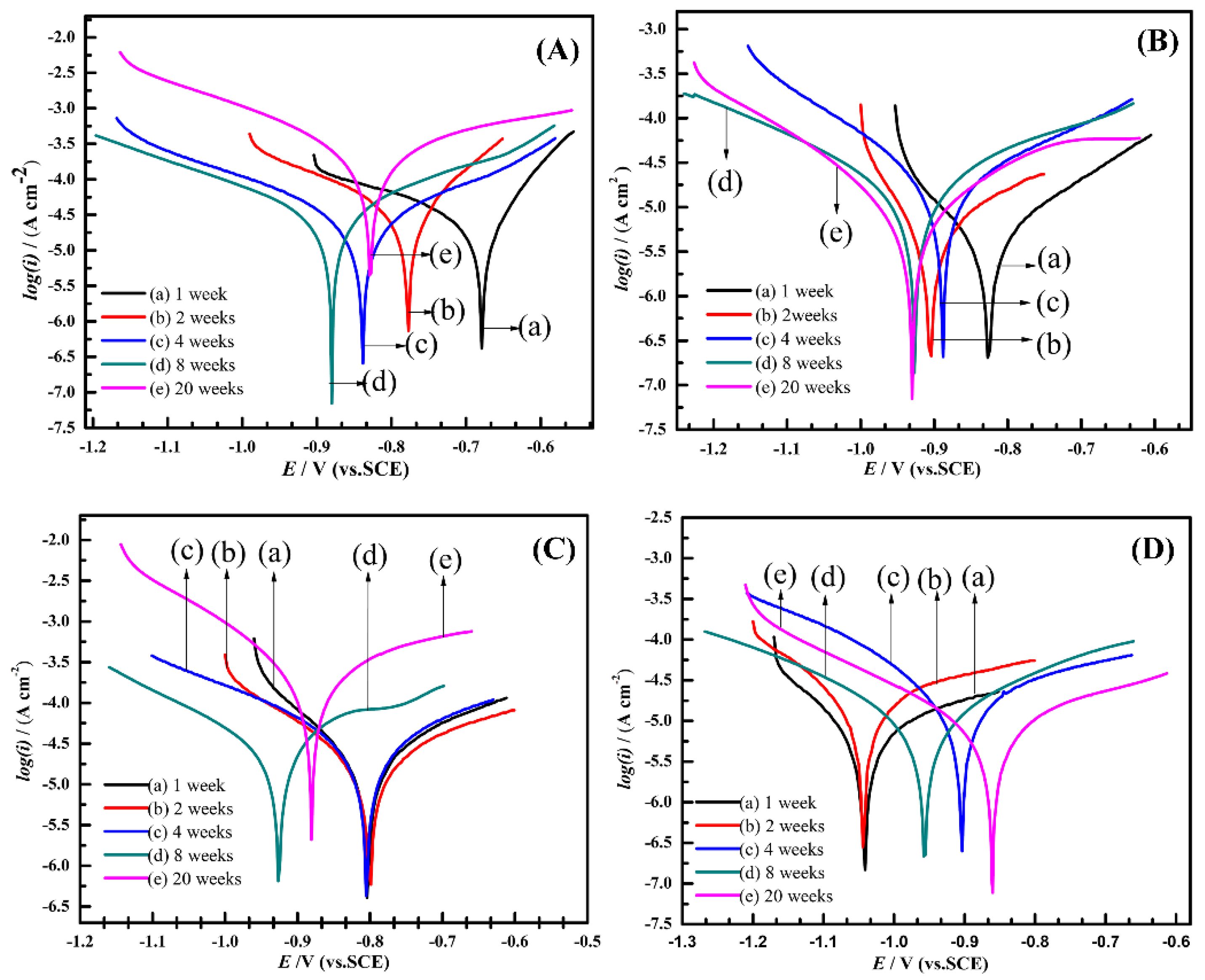
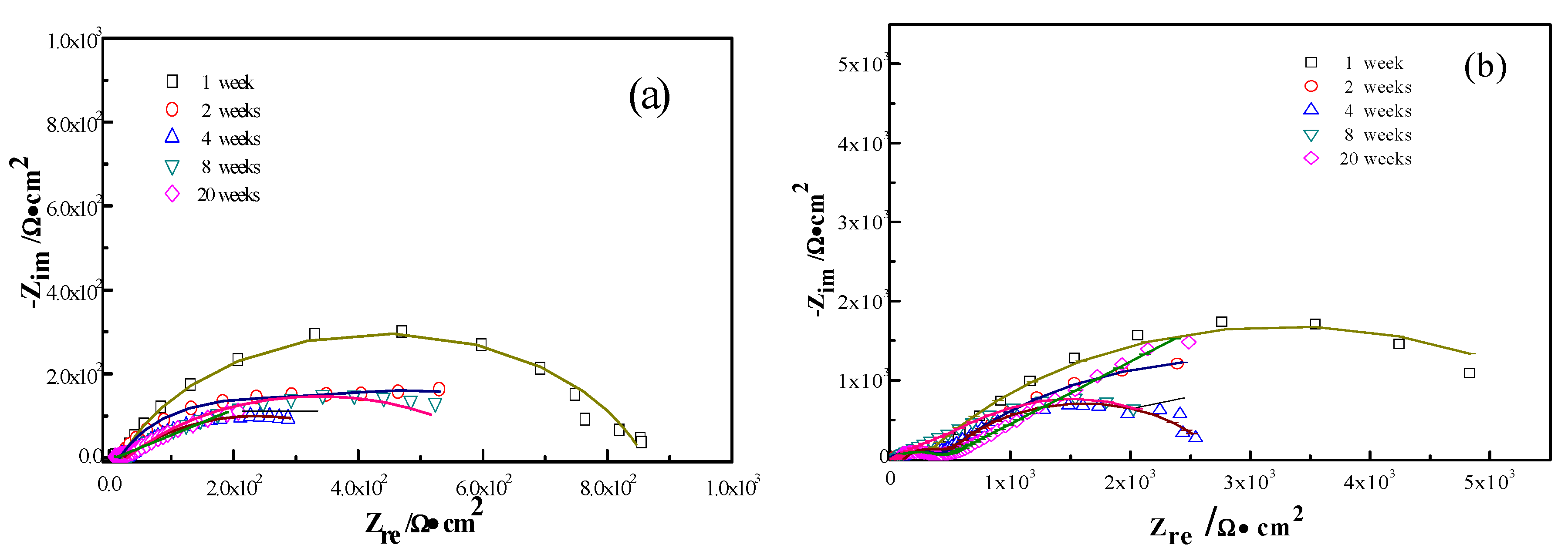
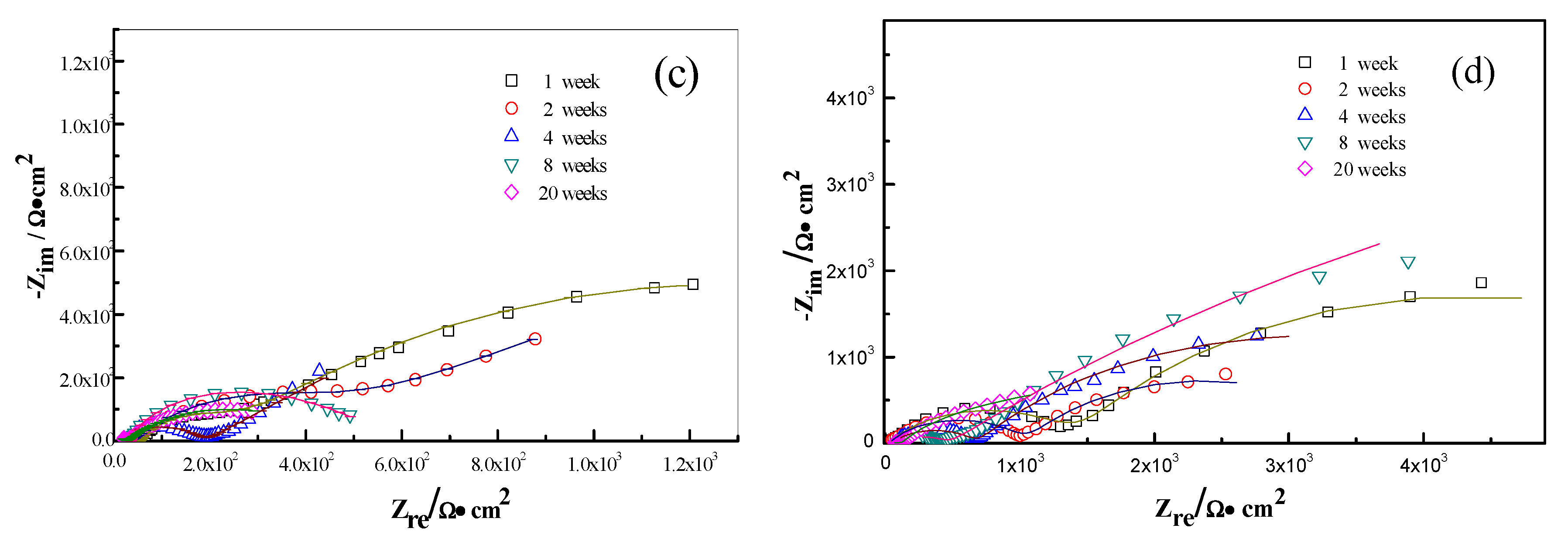
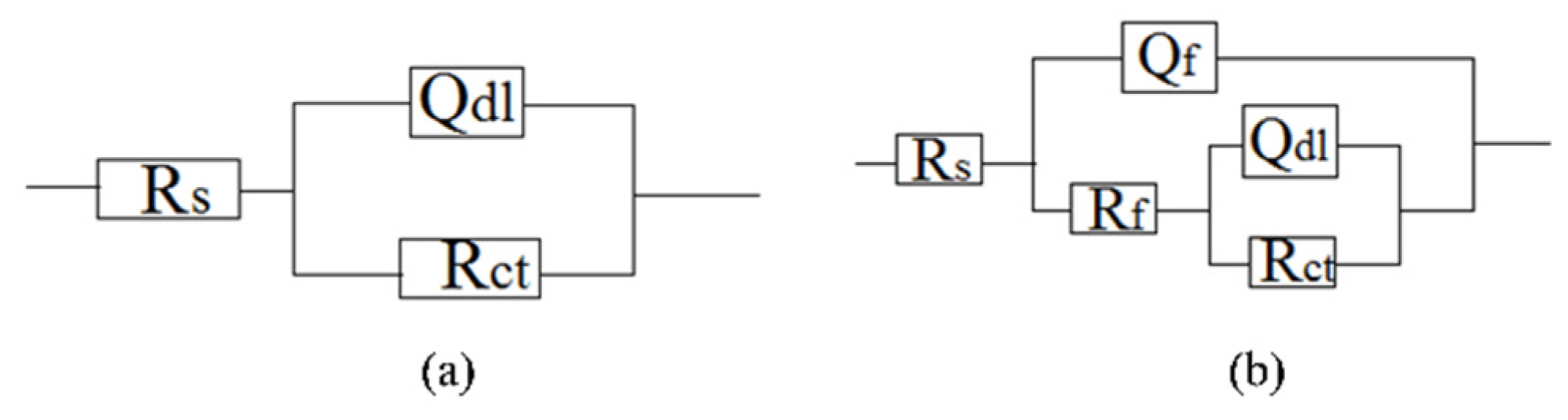
3.3. Electrochemical Analysis
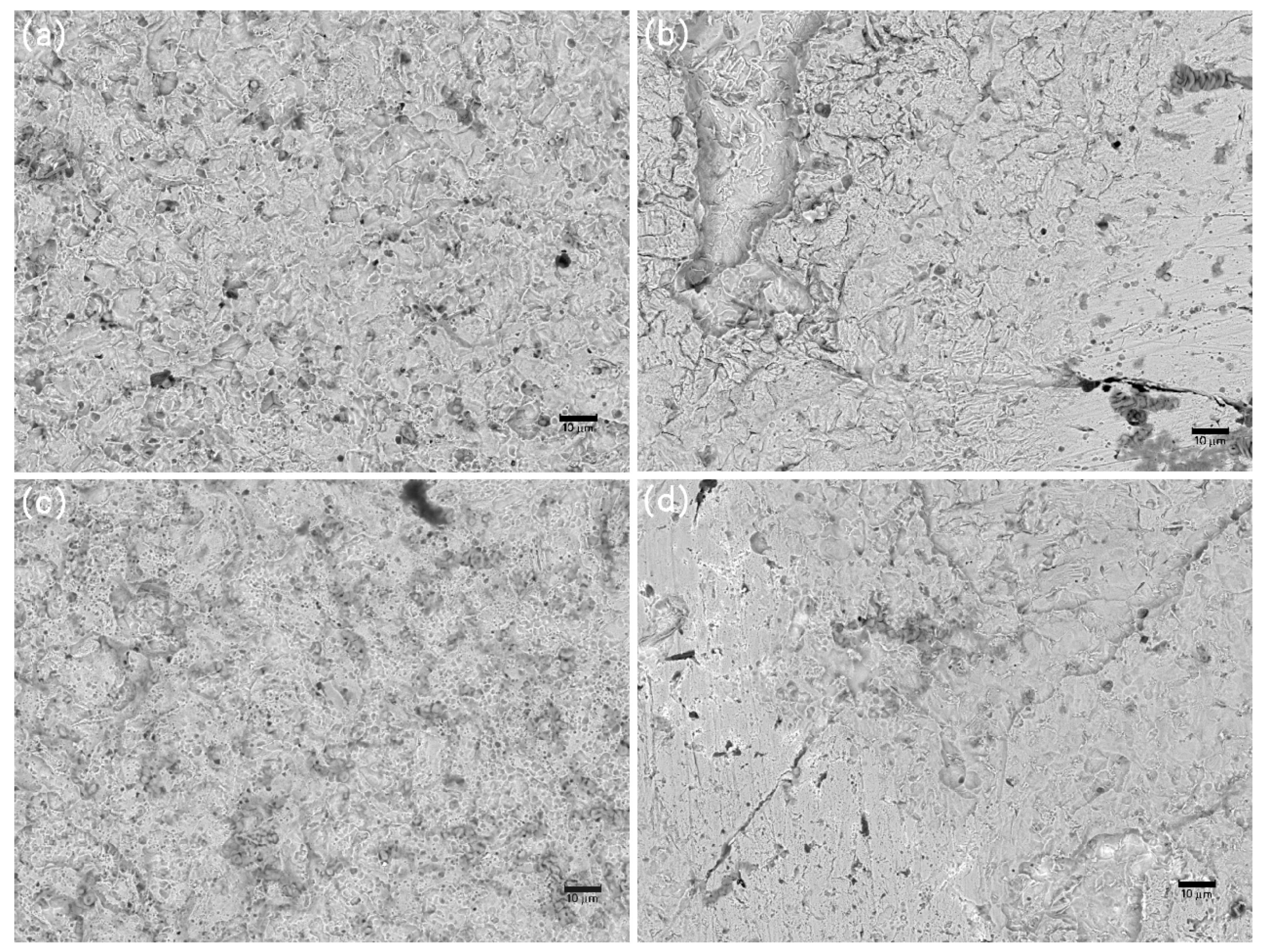
3.4. Corrosion Evolution
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Li, Y.C.; Xu, D.K.; Chen, C.F.; Li, X.G.; Jia, R.; Zhang, D.W.; Sand, W.; Wang, F.H.; Gu, T.Y. Anaerobic microbiologically influenced corrosion mechanisms interpreted using bioenergetics and bioelectrochemistry: A review. J. Mater. Sci. Technol. 2018, 34, 1713–1718. [Google Scholar] [CrossRef]
- Satheesh, S.; Ba-akdah, M.A.; Al-Sofyani, A.A. Natural antifouling compound production by microbes associated with marine macroorganisms—A review. Electron. J. Biotechnol. 2016, 21, 26–35. [Google Scholar] [CrossRef]
- Xu, D.K.; Zhou, E.Z.; Zhao, Y.; Li, H.B.; Liu, Z.Y.; Zhang, D.W.; Yang, C.G.; Lin, H.; Li, X.G.; Yang, K. Enhanced resistance of 2205 Cu-bearing duplex stainless steel towards microbiologically influenced corrosion by marine aerobic Pseudomonas aeruginosa biofilms. J. Mater. Sci. Technol. 2018, 34, 1325–1336. [Google Scholar] [CrossRef]
- Machuca, L.L.; Bailey, S.I.; Gubner, R.; Watkin, E.L.J.; Ginige, M.P.; Kaksonen, A.H.; Heidersbach, K. Effect of oxygen and biofilms on crevice corrosion of UNS S31803 and UNS N08825 in natural seawater. Corros. Sci. 2013, 67, 242–255. [Google Scholar] [CrossRef]
- Machuca, L.L.; Jeffrey, R.; Bailey, S.I.; Gubner, R.; Watkin, E.L.J.; Ginige, M.P.; Kaksonen, A.H.; Heidersbach, K. Filtration-UV irradiation as an option for mitigating the risk ofmicrobiologically influenced corrosion of subsea construction alloys in seawater. Corros. Sci. 2014, 79, 89–99. [Google Scholar] [CrossRef]
- Xu, D.K.; Li, Y.C.; Gu, T.Y. Mechanistic modeling of biocorrosion caused by biofilmsof sulfate reducing bacteria and acid producing bacteria. Bioelectrochemistry 2016, 110, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Li, H.B.; Zhou, E.Z.; Ren, Y.B.; Zhang, D.W.; Xu, D.K.; Yang, C.G.; Feng, H.; Jiang, Z.H.; Li, X.G.; Gu, T.Y.; et al. Investigation of microbiologically influenced corrosion of high nitrogen nickel-free stainless steel by Pseudomonas aeruginosa. Corros. Sci. 2016, 111, 811–821. [Google Scholar] [CrossRef]
- Callow, M.E.; Fletcher, R.L. The influence of low surface energy materials on bioadhesion—A review. Int. Biodeterior. Biodegrad. 1994, 34, 333–334. [Google Scholar] [CrossRef]
- Qian, P.Y.; Lau, S.C.K.; Dahms, H.-U.; Dobretsov, S.; Harder, T. Marine biofilms as mediators of colonization by marine macroorganisms: Implications for antifouling and aquaculture. Mar. Biol. 2007, 9, 399–410. [Google Scholar] [CrossRef]
- Relini, G.; Tixi, E.; Relini, M.; Torchia, G. The macrofouling on offshore platforms at Ravenna. Int. Biodeterior. Biodegrad. 1998, 41, 41–55. [Google Scholar] [CrossRef]
- Blackwood, D.J.; Lim, C.S.; Teo, S.L.M.; Hu, X.P.; Pang, J.J. Macrofouling induced localized corrosion of stainless steel in Singapore seawater. Corros. Sci. 2017, 129, 152–160. [Google Scholar] [CrossRef]
- De Brito, L.V.R.; Coutinho, R.; Cavalcanti, E.H.S.; Benchimol, M. The influence of macrofouling on the corrosion behaviour of API 5L X65 carbon steel. Biofouling 2007, 3, 193–201. [Google Scholar] [CrossRef]
- Stipanicev, M.; Turcu, F.; Esnault, L.; Rosas, O.; Basseguy, R.; Sztyler, M.; Beech, I.B. Corrosion of carbon steel by bacteria from North Sea offshore seawater injection systems: Laboratory investigation. Bioelectrochemistry 2014, 97, 76–88. [Google Scholar] [CrossRef] [PubMed]
- San, N.O.; Nazır, H.; Dönmez, G. Microbiologically influenced corrosion of NiZn alloy coatings by Delftia acidovorans bacterium. Corros. Sci. 2012, 64, 198–203. [Google Scholar] [CrossRef]
- Beale, D.J.; Dunn, M.S.; Marney, D. Application of GC–MS metabolic profiling to ‘blue-green water’ from microbial influenced corrosion in copper pipes. Corros. Sci. 2010, 52, 3140–3145. [Google Scholar] [CrossRef]
- Street, C.N.; Gibbs, A.J. Eradication of the corrosion-causing bacterial strains Desulfovibrio vulgaris and Desulfovibrio desulfuricans in planktonic and biofilm form using photodisinfection. Corros. Sci. 2010, 52, 1447–1452. [Google Scholar] [CrossRef]
- Wagner, P.; Little, B.; Hart, K.; Ray, R.; Thomas, D.; Trzaskoma-Paulette, P.; Lucas, K. Environmental fate of sacrificial zinc anodes and influence of a biofilm. Int. Biodeterior. Biodegrad. 1996, 37, 151–157. [Google Scholar] [CrossRef]
- Pierozynski, B.; Jankowski, J.; Sokolski, W. Application of nickel-coated carbon fibre material in cathodic protection of underground-buried steel structures. Corros. Sci. 2009, 51, 2605–2609. [Google Scholar] [CrossRef]
- Refait, P.; Jeannin, M.; Sabot, R.; Antony, H.; Pineau, S. Corrosion and cathodic protection of carbon steel in the tidal zone: Products, mechanisms and kinetics. Corros. Sci. 2015, 90, 375–382. [Google Scholar] [CrossRef]
- Zhang, L.; Shen, H.J.; Sun, J.Y.; Sun, Y.N.; Fang, Y.C.; Cao, W.H.; Xing, Y.Y.; Lu, M.X. Effect of calcareous deposits on hydrogen permeation in X80 steel under cathodic protection. Mater. Chem. Phys. 2018, 207, 123–129. [Google Scholar] [CrossRef]
- Simoni, L.; Caselani, J.Q.; Ramos, L.B.; Schroeder, R.M.; Malfatti, C.D.F. The influence of calcareous deposits on hydrogen uptake and embrittlement of API 5CT P110 steel. Corros. Sci. 2018, 118, 178–189. [Google Scholar] [CrossRef]
- Li, C.J.; Du, M. The growth mechanism of calcareous deposits under various hydrostatic pressures during the cathodic protection of carbon steel in seawater. RSC Adv. 2017, 7, 28819–28825. [Google Scholar] [CrossRef]
- Lee, R.U.; Ambrose, J.R. Influence of cathodic protection parameters on calcareous deposit formation. Corrosion 1988, 44, 887–891. [Google Scholar] [CrossRef]
- Rossi, S.; Bonora, P.L.; Pasinetti, R.; Benedetti, L.; Draghetti, M.; Sacco, E. Laboratory and field characterization of a new sacrificial anode for cathodic protection of offshore structures. Corrosion 1998, 54, 1018–1025. [Google Scholar] [CrossRef]
- Rousseau, C.; Baraud, F.; Leleyter, L.; Jeannin, M.; Gil, O. Calcareous deposit formed under cathodic protection in the presence of natural marine sediments: A 12 month experiment. Corros. Sci. 2010, 52, 2206–2218. [Google Scholar] [CrossRef]
- Dexter, S.C.; Lin, S.H. Effect of marine biofilms on cathodic protection. Int. Biodeterior. Biodegrad. 1992, 29, 231–249. [Google Scholar] [CrossRef]
- Möller, H. The influence of Mg2+ on the formation of calcareous deposits on a freely corroding low carbon steel in seawater. Corros. Sci. 2007, 49, 1992–2001. [Google Scholar] [CrossRef]
- Barchiche, C.; Deslouis, C.; Gil, O.; Refait, P.; Tribollet, B. Characterisation of calcareous deposits by electrochemical methods: Role of sulphates, calcium concentration and temperature. Electrochim. Acta 2004, 49, 2833–2839. [Google Scholar] [CrossRef]
- Blackwood, D.J.; Lim, C.S.; Teo, S.L.M. Influence of fouling on the efficiency of sacrificial anodes in providing cathodic protection in Southeast Asian tropical seawater. Biofouling 2010, 26, 779–785. [Google Scholar] [CrossRef]
- Eashwar, M.; Subramanian, G.; Palanichamy, S.; Rajagopal, G.; Madhu, S.; Kamaraj, P. Cathodic behaviour of stainless steel in coastal Indian seawater: Calcareous deposits overwhelm biofilms. Biofouling 2009, 25, 191–201. [Google Scholar] [CrossRef]
- Miyanaga, K.; Terashi, R.; Kawai, H.; Unno, H.; Tanji, Y. Biocidal effect of cathodic protection on bacterial viability in biofilm attached to carbon steel. Biotechnol. Bioeng. 2007, 97, 850–857. [Google Scholar] [CrossRef] [PubMed]
- Vedaprakash, L.; Dineshram, R.; Ratnam, K.; Lakshmi, K.; Jayaraj, K.; Babu, S.M.; Venkatesan, R.; Shanmugam, A. Experimental studies on the effect of different metallic substrates on marine biofouling. Colloids Surf. B Biointerfaces 2013, 106, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Compere, C.; Fontaine, M.N.B.; Bertrand, P.; Costa, D.; Marcus, P.; Poleunis, C.; Pradier, C.M.; Rondot, B.; Walls, M.G. Kinetics of conditioning layer formation on stainless steel immersed in seawater. Biofouling 2001, 17, 129–145. [Google Scholar] [CrossRef]
- Dexter, S.C. Biofouling and biocorrosion. Bull. Electrochem. 1996, 12, 1–7. [Google Scholar]
- Kim, S.J.; Jang, S.K. Corrosion characteristics of steel in seawater containing various chloride concentrations generated by electrochemical method. Trans. Nonferrous Met. Soc. China 2009, 19, 50–55. [Google Scholar] [CrossRef]
- Rosenhahn, A.; Sendra, G.H. Surface sensing and settlement strategies of marine biofouling organisms. Biointerphases 2012, 7, 1–13. [Google Scholar] [CrossRef]
- D’Onofrio, A.; Crawford, J.M.; Stewart, E.J.; Witt, K.; Gavrish, E.; Epstein, S.; Clardy, J.; Lewis, K. Siderophores from Neighboring Organisms Promote the Growth of Uncultured Bacteria. Chem. Biol. 2010, 17, 254–264. [Google Scholar] [CrossRef]
- López-Moreno, A.; Sepúlveda-Sánchez, J.D.; Mercedes Alonso Guzmán, E.M.; Le Borgne, S. Calcium carbonate precipitation by heterotrophic bacteria isolated from biofilms formed on deteriorated ignimbrite stones: Influence of calcium on EPS production and biofilm formation by these isolates. Biofouling 2014, 30, 547–560. [Google Scholar] [CrossRef]
- Cachet, H.; El Moustafid, T.; Herbert-Guillou, D.; Festy, D.; Touzain, S.; Tribollet, B. Characterization of deposits by direct observation and by electrochemical methods on a conductive transparent electrode. Application to biofilm and scale deposit under cathodic protection. Electrochim. Acta 2001, 46, 3851–3857. [Google Scholar] [CrossRef]
- Molino, P.J.; Wetherbee, R. The biology of biofouling diatoms and their role in the development of microbial slimes. Biofouling 2008, 24, 365–379. [Google Scholar] [CrossRef]
- Desai, D.V. Impact of Irgarol 1051 on the larval development and metamorphosis of Balanus Amphitrite Darwin, the diatom Amphora coffeaformis and natural biofilm. Biofouling 2008, 24, 393–403. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; Wang, J.D.; Chen, H.S.; Chen, D.R. Progress of marine biofouling and antifouling technologies. Mater. Sci. 2011, 56, 598–612. [Google Scholar] [CrossRef]
- Okhrimenko, D.V.; Nissenbaum, J.; Andersson, M.P.; Olsson, M.H.M.; Stipp, S.L.S. Energies of the adsorption of functional groups to calcium carbonate polymorphs: The importance of -OH and -COOH groups. Langmuir 2013, 29, 11062–11073. [Google Scholar] [CrossRef] [PubMed]
- Geesey, G.; Wigglesworth-Cooksey, B.; Cooksey, K.E. Influence of calcium and other cations on Surface adhesion of bacteria and diatoms: A review. Biofouling 2000, 15, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Little, B.J.; Wagner, P.A. The interrelationship between marine biofouling, cathodic protection and microbiologically influenced corrosion. Mater. Sci. Forum 1995, 192–194, 433–446. [Google Scholar] [CrossRef]
- Guezennec, J.; Dowling, N.J.E.; Bullen, J.; White, D.C. Relationship between bacterial colonization and cathodic current density associated with mild steel surfaces. Biofouling 1994, 8, 133–146. [Google Scholar] [CrossRef]
- Little, B.J.; Ray, R.I.; Wagner, P.A.; Jones-Meehan, J.; Lee, C.C.; Mansfeld, F. Spatial relationships between marine bacteria and localized corrosion on polymer coated steel. Biofouling 1999, 13, 301–321. [Google Scholar] [CrossRef]
- Wang, J.W.; Zhang, J.; Chen, S.G.; Duan, J.Z.; Hou, B.R. Influence of calcareous deposit on corrosion behavior of Q235 carbon steel in f/2 culture medium with amphora. J. Chin. Soc. Corros. Prot. 2015, 35, 535–542. [Google Scholar]
- Deslouis, C.; Falaras, P.; Gil, O.; Jeannin, M.; Maillot, V.; Tribollet, B. Influence of clay on calcareous deposit in natural and artificial sea water. Electrochim. Acta 2006, 51, 3173–3180. [Google Scholar] [CrossRef]
- Mansfeld, F. Tafel slopes and corrosion rates obtained in the pre-Tafel region of polarization curves. Corros. Sci. 2005, 47, 3178–3186. [Google Scholar] [CrossRef]
- Stern, M.; Geary, A.L. Electrochemical polarization: I. A theoretical analysis of the shape of polarization curves. J. Electrochem. Soc. 1957, 104, 56–63. [Google Scholar] [CrossRef]
- Poorqasemi, E.; Abootalebi, O.; Peikari, M.; Haqdar, F. Investigating accuracy of the Tafel extrapolation method in HCl solutions. Corros. Sci. 2009, 51, 1043–1054. [Google Scholar] [CrossRef]





| Element (wt %) Samples | C | O | Na | Mg | Si | P | Cl | Ca |
|---|---|---|---|---|---|---|---|---|
| anode-coupled bare samples (a) | 9.02 | 41.33 | 0.67 | 0.30 | 1.06 | 1.09 | - | 46.51 |
| anode-coupled samples with pre-existing calcareous deposits (b) | 10.70 | 54.39 | 1.04 | 0.48 | 0.67 | 1.08 | 0.12 | 31.50 |
| Samples | Exposure Time (Week) | -bc (mV/dec) | ba (mV/dec) | Rp (Ω·cm2) | Ecorr (mV/SCE) | icorr (mA/cm2) |
|---|---|---|---|---|---|---|
| A | 1 | 226 | 88 | 1193 | −679 | 0.0231 |
| 2 | 205 | 114 | 907 | −778 | 0.0351 | |
| 4 | 277 | 282 | 1671 | −839 | 0.0363 | |
| 8 | 288 | 232 | 1302 | −879 | 0.0429 | |
| 20 | 206 | 311 | 216 | −828 | 0.2491 | |
| B | 1 | 87 | 183 | 6243 | −826 | 0.0041 |
| 2 | 57 | 59 | 5700 | −905 | 0.0022 | |
| 4 | 157 | 269 | 2336 | −889 | 0.0184 | |
| 8 | 376 | 245 | 3063 | −926 | 0.0210 | |
| 20 | 177 | 228 | 4834 | −929 | 0.0090 | |
| C | 1 | 131 | 172 | 1612 | −803 | 0.0200 |
| 2 | 157 | 224 | 1988 | −800 | 0.0202 | |
| 4 | 314 | 181 | 1560 | −804 | 0.0320 | |
| 8 | 310 | 238 | 1268 | −861 | 0.0461 | |
| 20 | 139 | 224 | 199 | −880 | 0.1872 | |
| D | 1 | 132 | 235 | 5547 | −1042 | 0.0066 |
| 2 | 151 | 292 | 2529 | −1043 | 0.0171 | |
| 4 | 249 | 473 | 2628 | −903 | 0.0270 | |
| 8 | 153 | 183 | 4624 | −956 | 0.0078 | |
| 20 | 225 | 295 | 5477 | −925 | 0.0101 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; Yu, Z.; Zhao, X.; Lan, X.; Wang, J.; Lv, X.; Zhang, C.; Duan, J.; Hou, B. The Interaction of Biofoulants and Calcareous Deposits on Corrosion Performance of Q235 in Seawater. Materials 2020, 13, 850. https://doi.org/10.3390/ma13040850
Zhang J, Yu Z, Zhao X, Lan X, Wang J, Lv X, Zhang C, Duan J, Hou B. The Interaction of Biofoulants and Calcareous Deposits on Corrosion Performance of Q235 in Seawater. Materials. 2020; 13(4):850. https://doi.org/10.3390/ma13040850
Chicago/Turabian StyleZhang, Jie, Zhenhua Yu, Xia Zhao, Xiao Lan, Jiangwei Wang, Xianzi Lv, Chao Zhang, Jizhou Duan, and Baorong Hou. 2020. "The Interaction of Biofoulants and Calcareous Deposits on Corrosion Performance of Q235 in Seawater" Materials 13, no. 4: 850. https://doi.org/10.3390/ma13040850
APA StyleZhang, J., Yu, Z., Zhao, X., Lan, X., Wang, J., Lv, X., Zhang, C., Duan, J., & Hou, B. (2020). The Interaction of Biofoulants and Calcareous Deposits on Corrosion Performance of Q235 in Seawater. Materials, 13(4), 850. https://doi.org/10.3390/ma13040850





