Investigation of the Thermal Properties of Diesters from Methanol, 1-Pentanol, and 1-Decanol as Sustainable Phase Change Materials
Abstract
1. Introduction
2. Results and Discussion
2.1. Structural Characterization
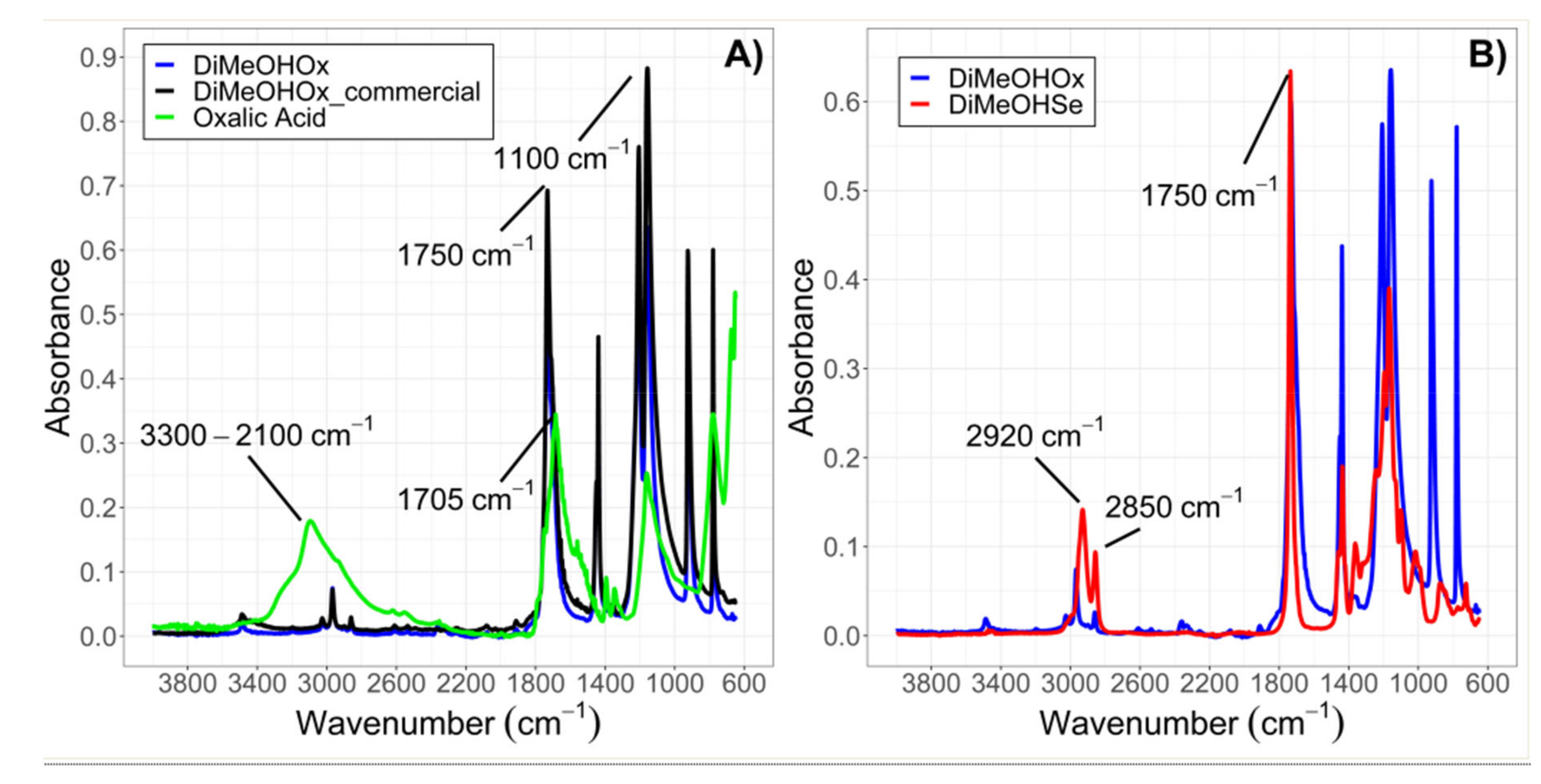
2.1.1. ATR-IR
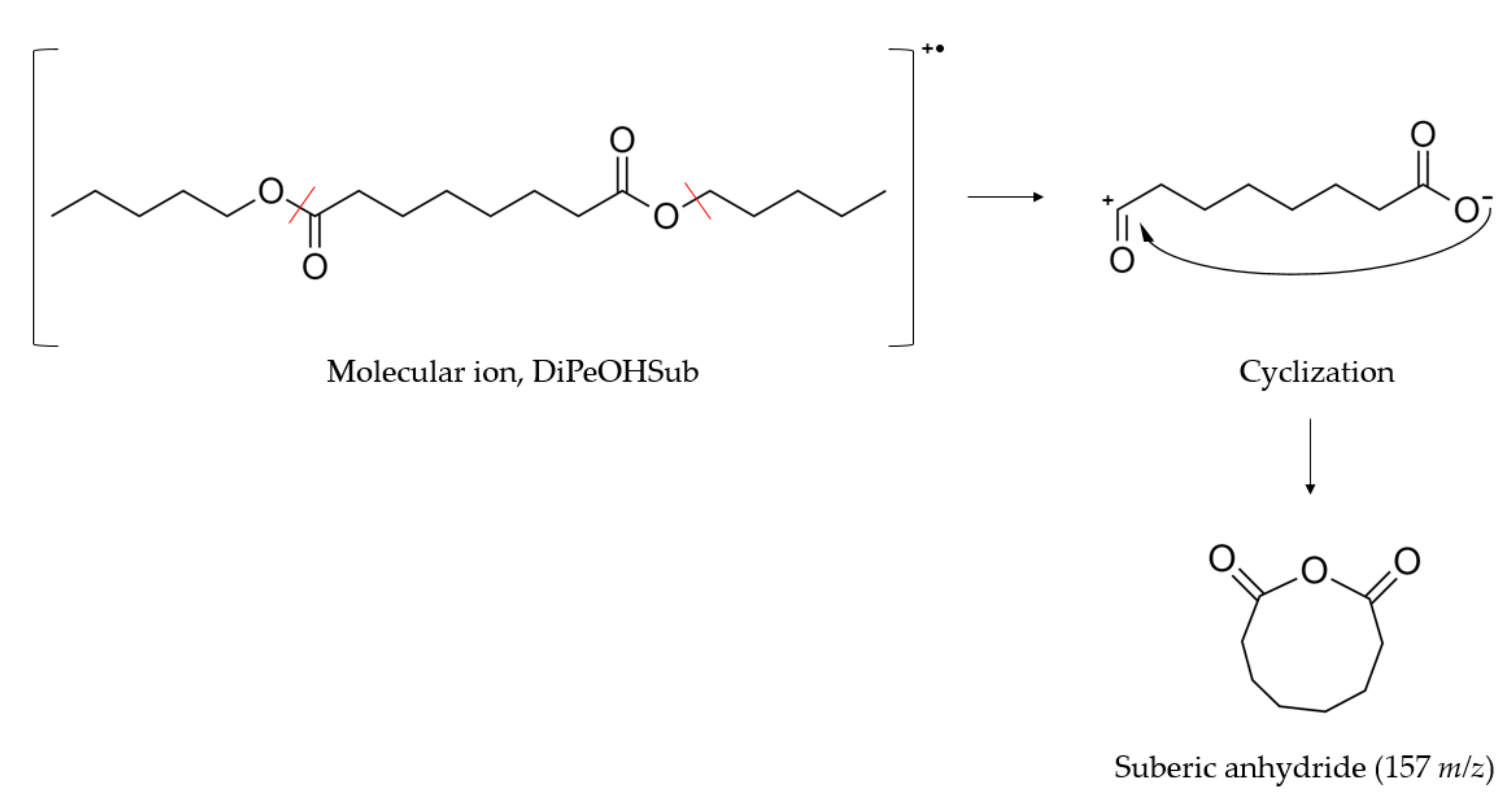
2.1.2. GC-MS
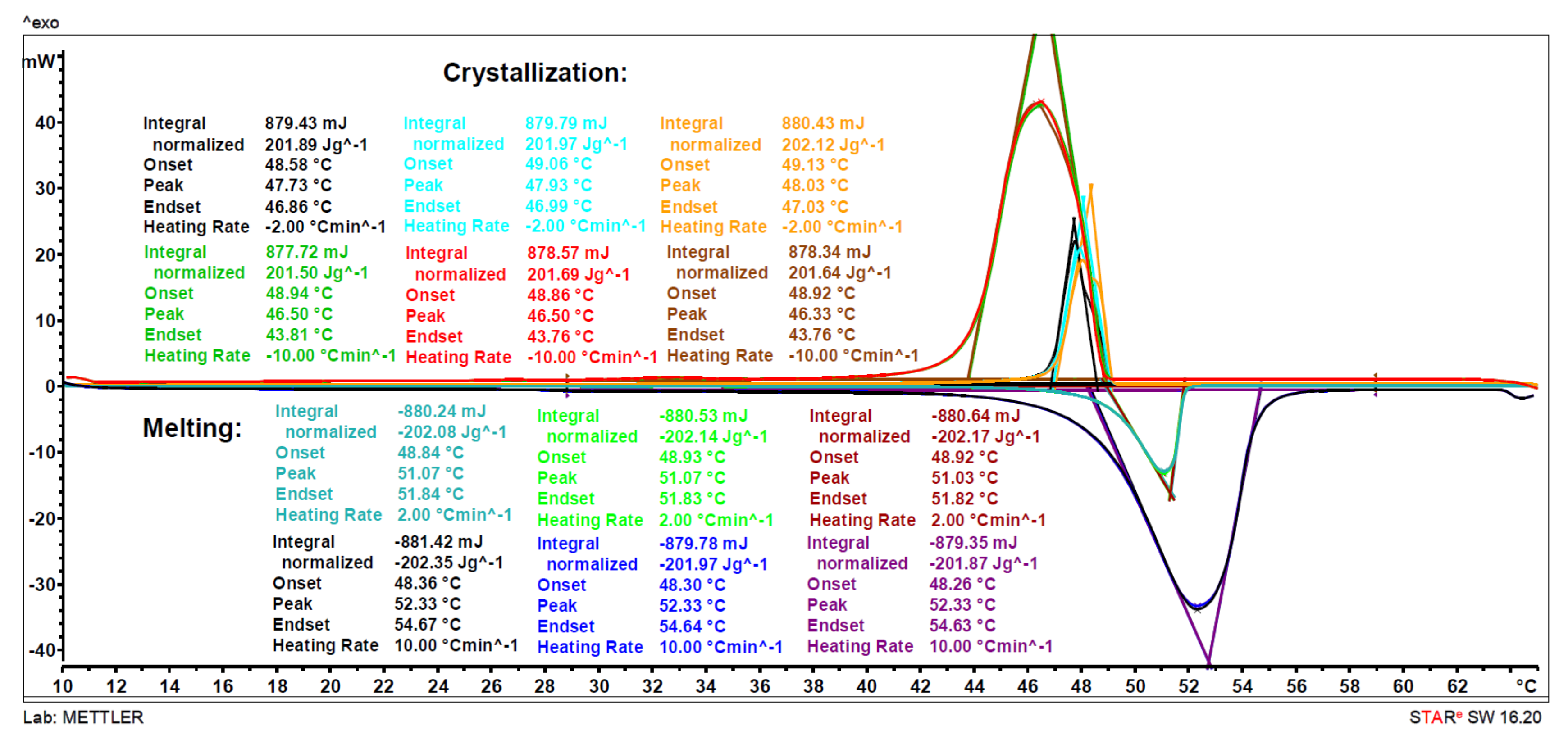
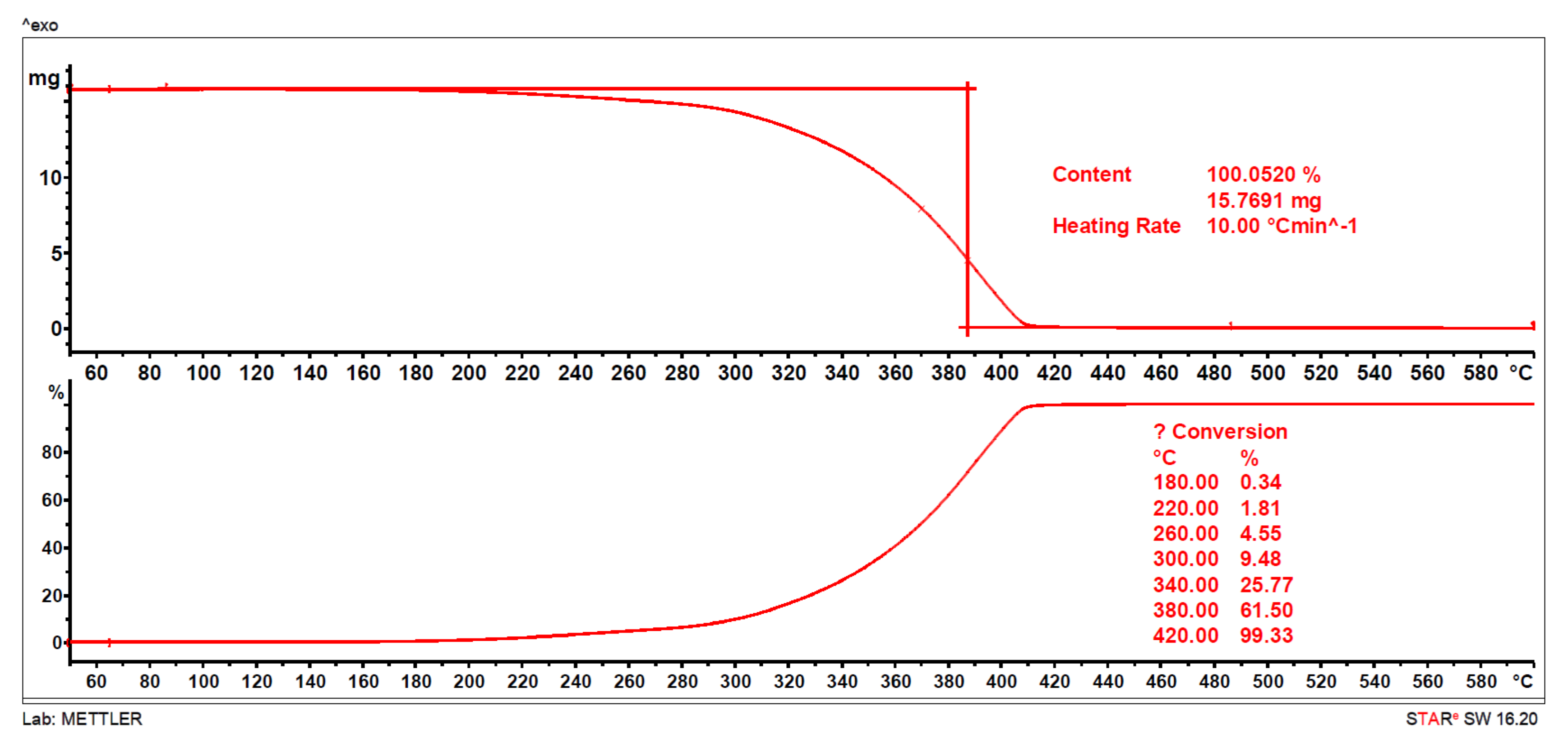
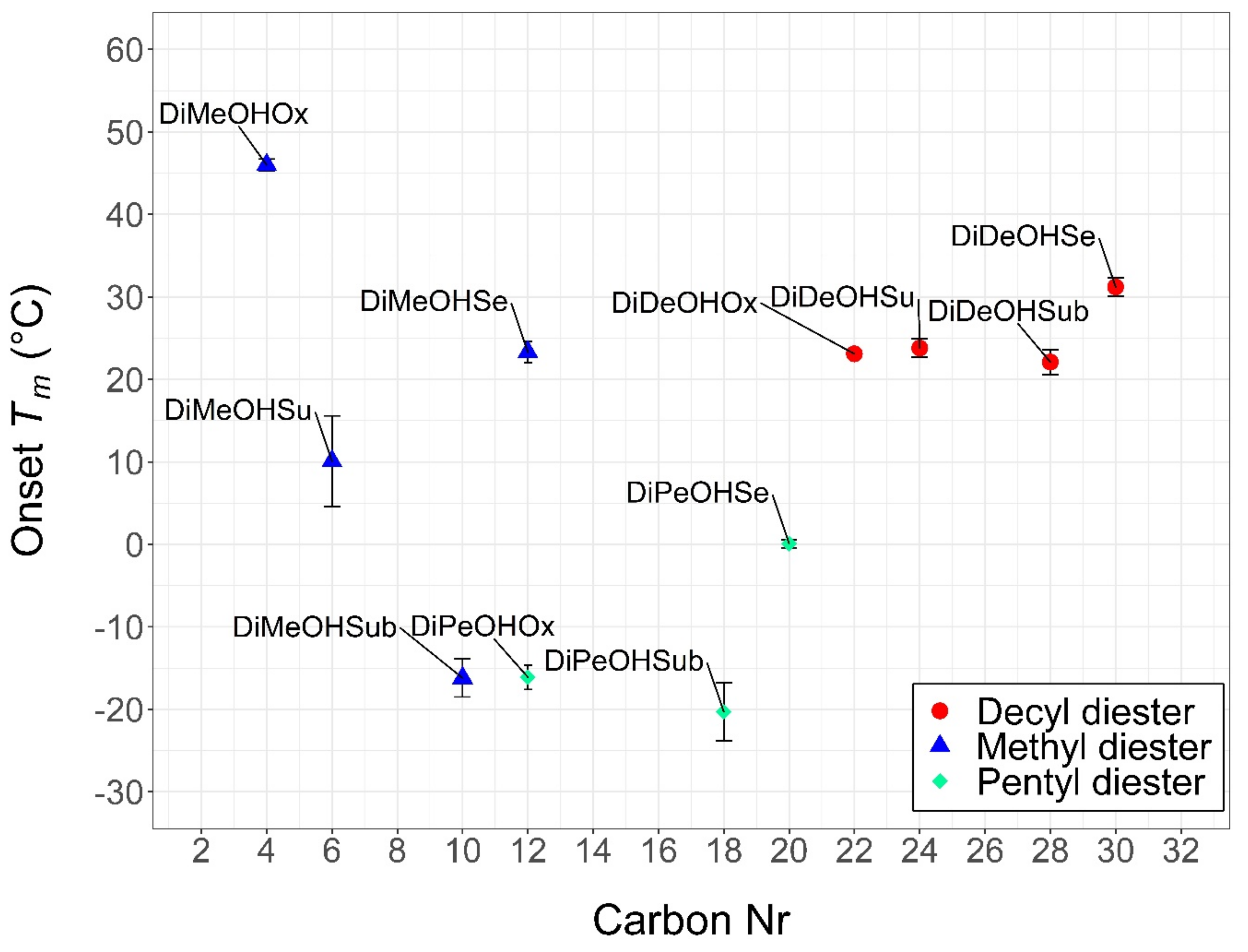
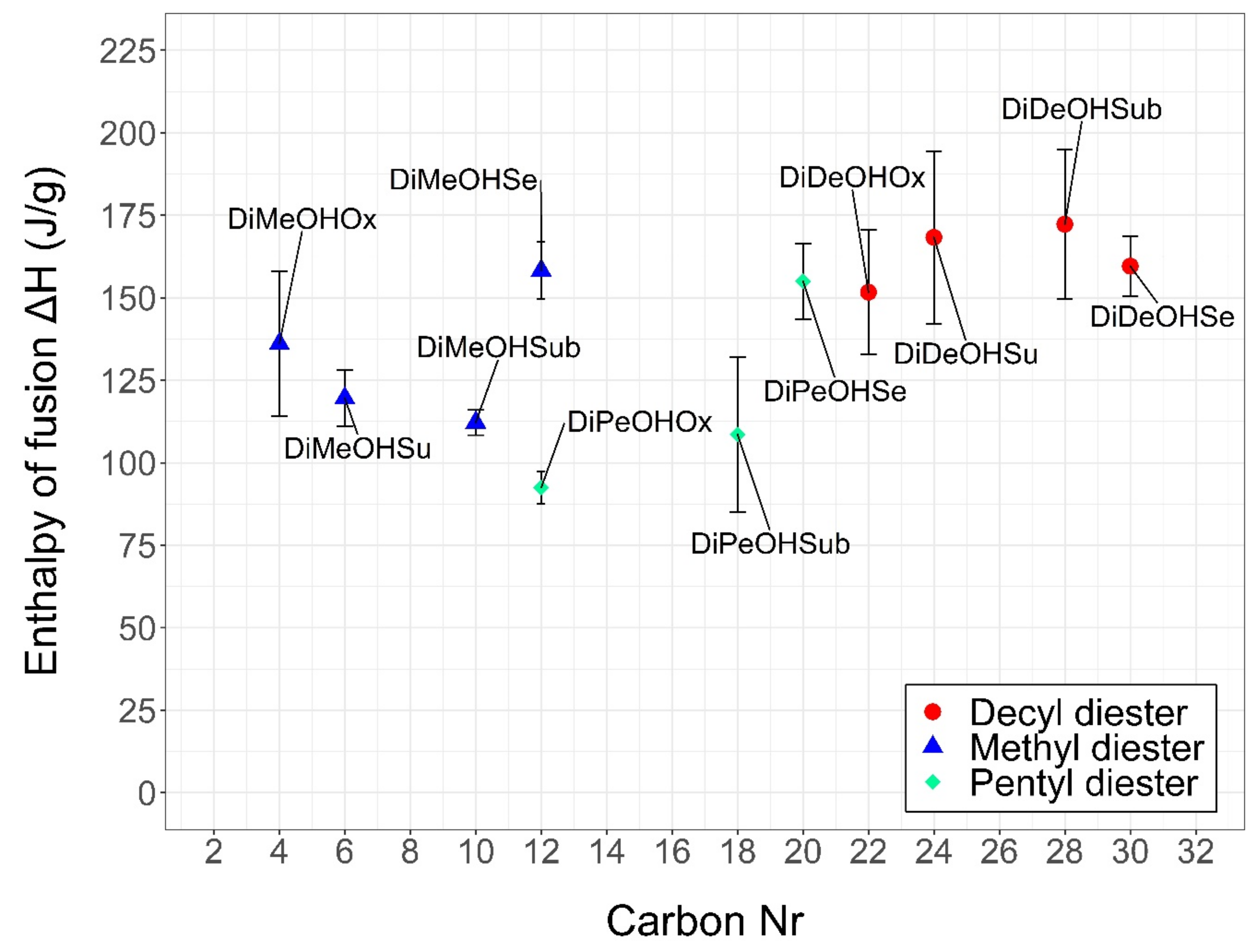
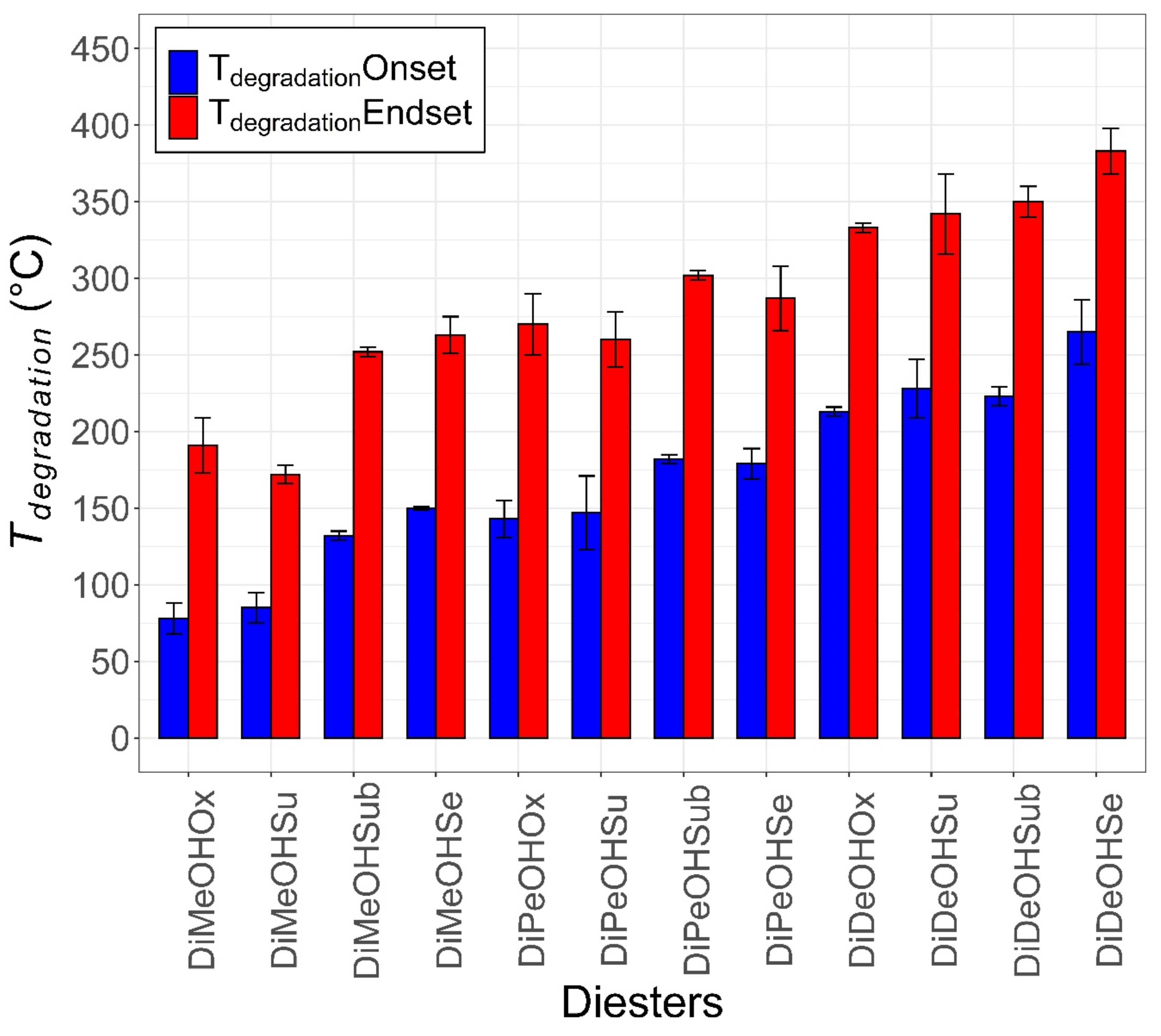
2.2. Thermal Characterization
3. Materials and Methods
3.1. Synthesis
3.2. Characterization
3.2.1. Differential Scanning Calorimetry (DSC)
3.2.2. Thermal Gravimetric Analysis (TGA)
3.2.3. Attenuated Total Reflectance Infrared Spectroscopy (ATR-IR)
3.2.4. Gas Chromatography Coupled with Mass Spectrometry (GC-MS)
4. Conclusions and Outlook
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ATR-IR | Attenuated Total Reflectance InfraRed Spectroscopy |
| DENSE | Direct contact ENergy StoragE |
| DeOH | 1-Decanol (C10) |
| DiDeOHOx | Didecyl Oxalate (10.2.10) |
| DiDeOHSe | Didecyl Sebacate (10.10.10) |
| DiDeOHSub | Didecyl Suberate (10.8.10) |
| DiDeOHSu | Didecyl Succinate (10.4.10) |
| DiMeOHOx | DiMethyl Oxalate (1.2.1) |
| DiMeOHSe | DiMethyl Sebacate (1.10.1) |
| DiMeOHSub | DiMethyl Suberate (1.8.1) |
| DiMeOHSu | DiMethyl Succinate (1.4.1) |
| DiPeOHOx | DiPentyl Oxalate (5.2.5) |
| DiPeOHSe | DiPentyl Sebacate (5.10.5) |
| DiPeOHSub | DiPentyl Suberate (5.8.5) |
| DiPeOHSu | DiPentyl Succinate (5.4.5) |
| DiTeOHSe | DiTetradecyl Sebacate (14.10.14) |
| DSC | Differential Scanning Calorimetry |
| EtOAc | Ethyl Acetate |
| GC | Gas Chromatography |
| GC-MS | Gas-Chromatography coupled with Mass Spectrometry |
| H2SO4 | Sulfuric Acid |
| IR | Infrared Spectroscopy |
| IUPAC | International Union of Pure and Applied Chemistry |
| LHS | Latent Heat Storage |
| MeOH | Methanol (C1) |
| MS | Mass spectrometry |
| MSD | Mass Selective Detector |
| Na2SO4 | Sodium Sulfate |
| NIST | National Institute of Standards and Technology |
| Ox | Oxalic acid (C2) |
| PCM | Phase Change Material |
| PeOH | 1-Pentanol (C5) |
| PFTBA | Perfluorotributylamine |
| Se | Sebacic acid (C10) |
| SNSF | Swiss National Science Foundation |
| Sub | Suberic acid (C8) |
| Su | Succinic acid (C4) |
| Tc | Crystallization Temperature |
| TeOH | 1-Tetradecanol (C14) |
| TES | Thermal Energy Storage |
| TGA | Thermogravimetric Analysis |
| Tm | Melting Temperature |
| ΔH | Enthalpy of fusion |
Appendix A
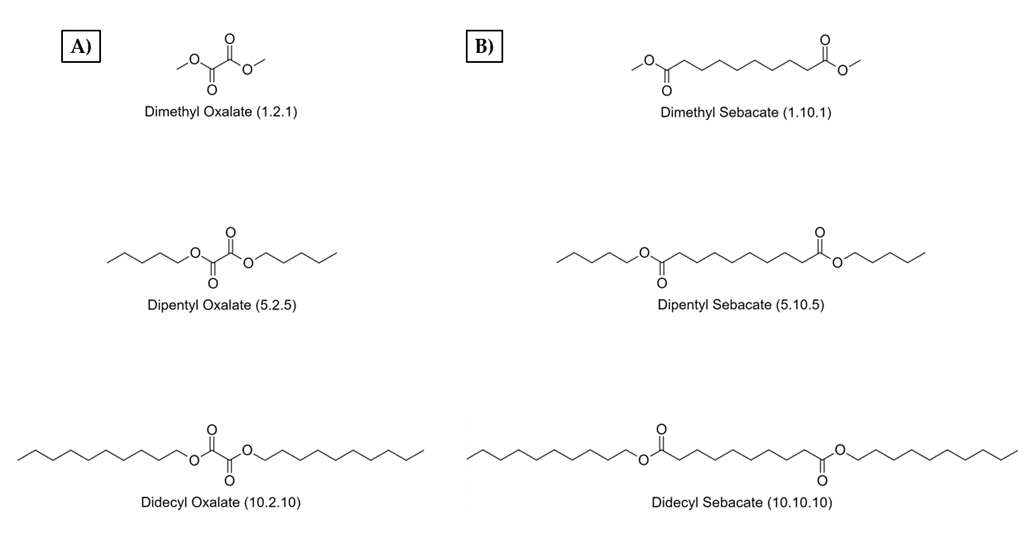
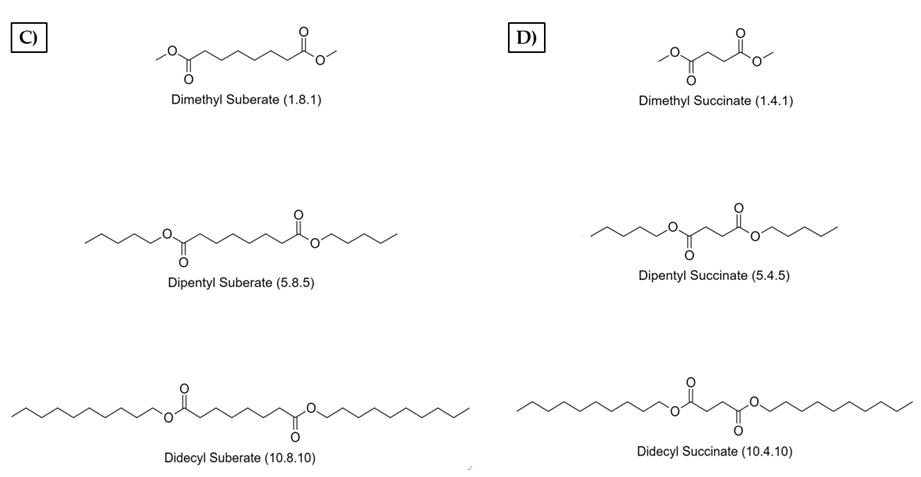
Appendix B
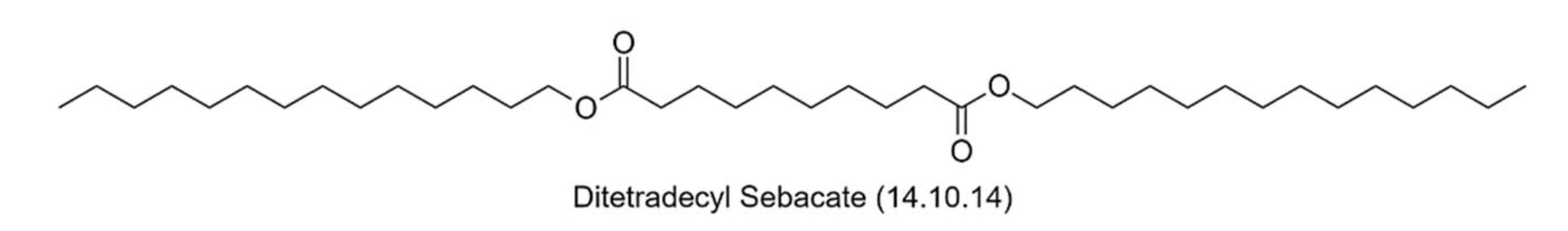
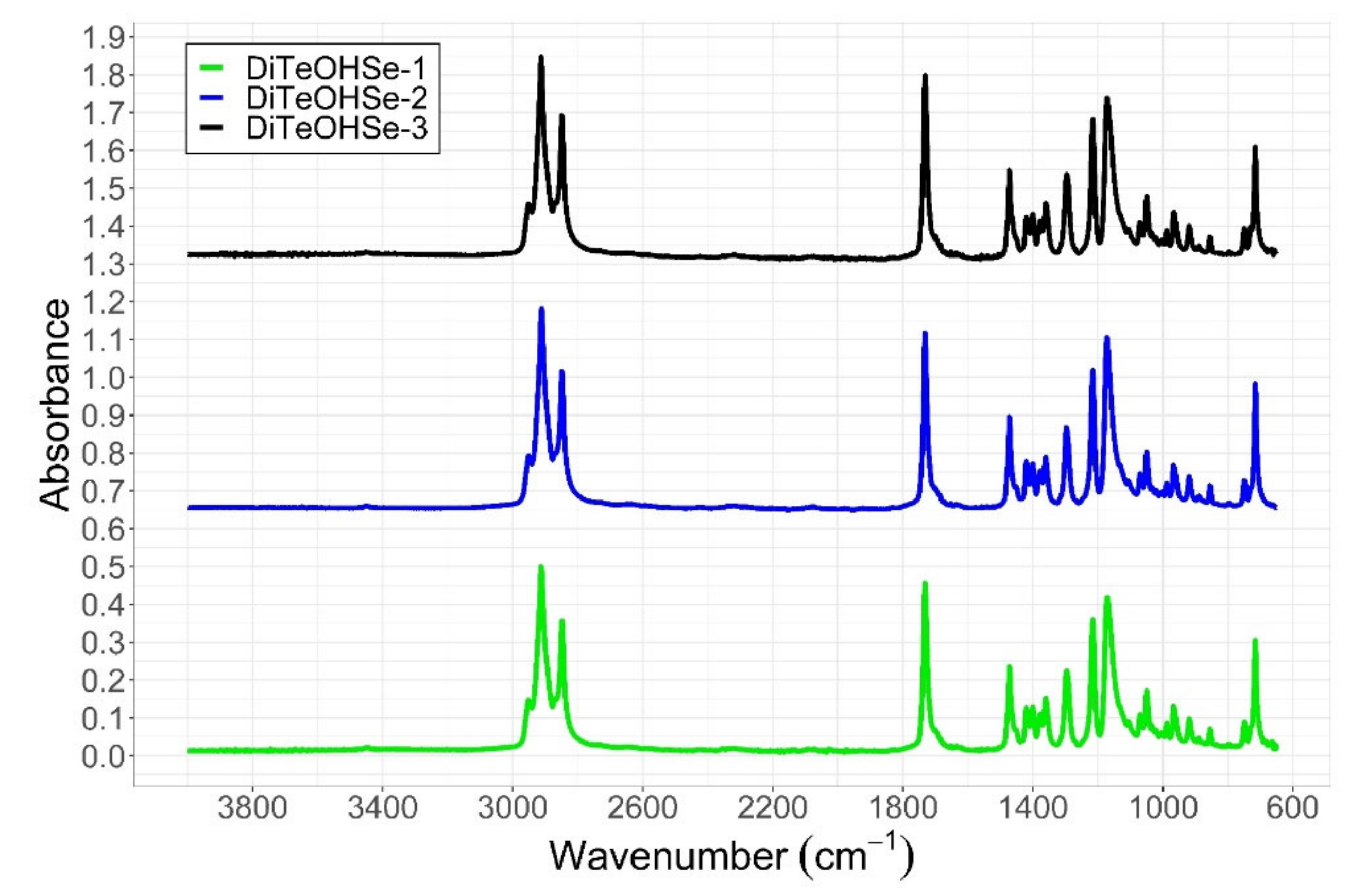
References
- European Commission, EU. Energy 2020—A Strategy for Competitive, Sustainable and Secure Energy; Publications office of the European Union: Luxembourg, 2010. [Google Scholar] [CrossRef]
- European Commission, EU. Energy Roadmap 2050; Publications office of the European Union: Luxembourg, 2012. [Google Scholar] [CrossRef]
- Connolly, D.; Lund, H.; Mathiesen, B.; Werner, S.; Möller, B.; Persson, U.; Boermans, T.; Trier, D.; Østergaard, P.A.; Nielsen, S. Heat roadmap Europe: Combining district heating with heat savings to decarbonise the EU energy system. Energy Policy 2014, 65, 475–489. [Google Scholar] [CrossRef]
- Yazdanie, M.; Densing, M.; Wokaun, A. The role of decentralized generation and storage technologies in future energy systems planning for a rural agglomeration in Switzerland. Energy Policy 2016, 96, 432–445. [Google Scholar] [CrossRef]
- Child, M.; Bogdanov, D.; Breyer, C. The role of storage technologies for the transition to a 100% renewable energy system in Europe. Energy Policy 2018, 155, 44–60. [Google Scholar] [CrossRef]
- Bakan, G.; Gerislioglu, B.; Dirisaglik, F.; Jurado, Z.; Sullivan, L.; Dana, A.; Lam, C.; Gokirkmak, A.; Silva, H. Extracting the temperature distribution on a phase-change memory cell during crystallization. J. Appl. Phys. 2016, 120, 164504. [Google Scholar] [CrossRef]
- Gerislioglu, B.; Ahmadivand, A. The Role of Electron Transfer in the Nonlinear Response of Ge2Sb2Te5—Mediated Plasmonic Dimers. Photonics 2019, 6, 52. [Google Scholar] [CrossRef]
- Choi, C.; Lee, S.; Mun, S.; Lee, G.; Sung, J.; Yun, H.; Yang, J.; Kim, H.; Hwang, C.; Lee, B. Metasurface with Nanostructured Ge2Sb2Te5 as a Platform for Broadband-Operating Wavefront Switch. Adv. Opt. Mater. 2019, 7, 1–8. [Google Scholar] [CrossRef]
- Avghad, S.N.; Keche, A.J.; Kousal, A. Thermal Energy Storage: A Review. J. Mech. Civ. Eng. 2016, 13, 72–77. [Google Scholar] [CrossRef]
- Konuklu, Y.; Şahan, N.; Paksoy, H. 2.14 Latent Heat Storage Systems. Compr. Energy Syst. 2018, 2, 396–434. [Google Scholar] [CrossRef]
- Mehling, H.; Cabeza, L.F. Heat and Cold Storage with PCM; Springer: Berlin, Germany, 2008; pp. 11–13. ISBN 354068557X. [Google Scholar]
- Nkwetta, D.N.; Haghighat, F. Thermal energy storage with phase change material—A state-of-the art review. Sustain. Cities Soc. 2014, 10, 87–100. [Google Scholar] [CrossRef]
- Tang, Y.R.; Gao, D.L.; Guo, Y.F.; Wang, S.Q.; Deng, T.L. Supercooling and Phase Separation of Inorganic Salt Hydrates as PCMs. Appl. Mech. Mater. 2011, 71–78, 2602–2605. [Google Scholar] [CrossRef]
- Cabeza, L.F.; Castell, A.; Barreneche, C.; de Gracia, A.; Fernández, A.I. Materials used as PCM in thermal energy storage in buildings: A review. Renew. Sustain. Energy Rev. 2011, 15, 1675–1695. [Google Scholar] [CrossRef]
- Feldman, D.; Banu, D.; Hawes, D. Low chain esters of stearic acid as phase change materials for thermal energy storage in buildings. Sol. Energy Mater. Sol. Cells 1995, 36, 311–322. [Google Scholar] [CrossRef]
- Sari, A.; Biçer, A. Karaipekli, Synthesis, characterization, thermal properties of a series of stearic acid esters as novel solid—Liquid phase change materials. Mat. Let. 2009, 13–14, 1213–1216. [Google Scholar] [CrossRef]
- Stamatiou, A.; Obermeyer, M.; Fischer, L.J.; Schuetz, P.; Worlitschek, J. Investigation of unbranched, saturated, carboxylic esters as phase change materials. Renew. Energy 2017, 108, 401–409. [Google Scholar] [CrossRef]
- Ravotti, R.; Fellmann, O.; Lardon, N.; Fischer, L.J.; Stamatiou, A.; Worlitschek, J. Synthesis and Investigation of Thermal Properties of Highly Pure Carboxylic Fatty Esters to Be Used as PCM. Appl. Sci. 2018, 8, 1069. [Google Scholar] [CrossRef]
- Ravotti, R.; Fellmann, O.; Lardon, N.; Fischer, L.J.; Stamatiou, A.; Worlitschek, J. Analysis of Bio-Based Fatty Esters PCM’s Thermal Properties and Investigation of Trends in Relation to Chemical Structures. Appl. Sci. 2019, 9, 225. [Google Scholar] [CrossRef]
- Ravotti, R.; Fellmann, O.; Lardon, N.; Fischer, L.J.; Stamatiou, A.; Worlitschek, J. Investigation of Lactones as Innovative Bio-sourced PCM for Latent Heat Storage. Molecules 2019, 24, 1300. [Google Scholar] [CrossRef]
- Suppes, G.J.; Goff, M.J.; Lopes, S. Latent heat characteristics of fatty acid derivatives pursuant phase change material applications. Chem. Eng. Sci. 2003, 58, 1751–1763. [Google Scholar] [CrossRef]
- Malkin, T. The Polymorphism of Glycerides. J. Chem. Soc. 1940, 103, 1–50. [Google Scholar] [CrossRef]
- Ferguson, R.H.; Lutton, E.S. The Polymorphic forms or phases of triglyceride fats. Symp. Mol. Struct. Fats Oils 1941, 29, 355–384. [Google Scholar] [CrossRef]
- Aydin, A. Diesters of high-chain dicarboxylic acids with 1-tetradecanol as novel organic phase change materials for thermal energy storage. Sol. Energy Mater. Sol. Cells 2012, 104, 102–108. [Google Scholar] [CrossRef]
- Feldman, D.; Shapiro, M.M.; Banu, D. Organic phase change materials for thermal energy storage. Sol. Energy Mater. 1986, 13, 1–10. [Google Scholar] [CrossRef]
- Tariq, M.; Ali, S.; Ahmad, F.; Ahmad, M.; Zafar, M.; Khalid, N.; Khan, M. Identification, FT-IR, NMR (1H and 13C) and GC/MS studies of fatty acid methyl esters in biodiesel from rocket seed oil. Fuel Process. Technol. 2011, 92, 336–341. [Google Scholar] [CrossRef]
- Liehr, J.G.; Larka, E.A.; Beynon, J.H. Cyclization of dimethyl oxalate upon electron impact. Org. Mass Spectrom. 1982, 16, 34–36. [Google Scholar] [CrossRef]
- Vajdi, M.; Nawar, W.W.; Merritt, C. GC/MS analysis of some long chain esters, ketones and propanediol diesters. J. Am. Oil Chem. Soc. 1981, 58, 106–110. [Google Scholar] [CrossRef]
- Bunn, C.W. The Melting Points of Chain Polymers. J. Polym. Sci. 1955, XVI, 323–343. [Google Scholar] [CrossRef]
- Crowe, R.W.; Smyth, C.P. Molecular Rotation in Some Long-chain Fatty Acid Esters in the Solid State. J. Am. Chem. Soc. 1951, 73, 5401–5406. [Google Scholar] [CrossRef]
- Malkin, T. Alternation in properties of long chain carbon compounds. Nature 1931, 127, 126–127. [Google Scholar] [CrossRef]
- Lutton, E.S.; Fehl, A.J. The Polymorphism of Odd and Even Saturated Single Acid Triglycerides C8-C22. Lipids 1969, 5, 90–99. [Google Scholar] [CrossRef]
- Zeberg-Mikkelsen, C.K.; Stenby, E.H. Predicting the melting points and the enthalpies of fusion of saturated triglycerides by a group contribution method. Fluid Phase Equilib. 1999, 162, 7–17. [Google Scholar] [CrossRef]
- Constantinou, L.; Gani, R. New group contribution method for estimating properties of pure compounds. AIChE J. 1994, 40, 1697–1710. [Google Scholar] [CrossRef]
- Oró, E.; de Gracia, A.; Castell, A.; Farid, M.M.; Cabeza, L.F. Review on phase change materials (PCMs) for cold thermal energy storage applications. Appl. Energy 2012, 99, 513–533. [Google Scholar] [CrossRef]
- Oró, E.; Barreneche, C.; Farid, M.M.; Cabeza, L.F. Experimental study on the selection of phase change materials for low temperature applications. Renew. Energy 2013, 57, 130–136. [Google Scholar] [CrossRef]
| Compound * | Retention Time GC, min | Molecular Weight (MW), g/mol | Fragmentation Peaks MS, m/z with Relative Intensities (%) |
|---|---|---|---|
| DiMeOHOx (1.2.1) | 1.84 | 118 | 118 (8), 59 (100), 45 (32) |
| DiPeOHOx (5.2.5) | 9.17 | 230 | 161 (1), 117 (5), 115 (8), 99 (1), 87 (3), 71 (100), 70 (55), 55(54) |
| DiDeOHOx (10.2.10) | 18.27 | 370 | 281 (1), 207 (4), 191 (3), 169 (1), 164 (3), 140 (2), 133 (3), 124 (2), 112 (1), 97 (4), 85 (8), 83 (9), 71 (14), 69 (17), 57 (85), 55 (100) |
| DiMeOHSu (1.4.1) | 2.73 | 146 | 116 (8), 115 (100), 114 (32), 101 (2), 87 (20), 86 (2), 60 (1), 59 (5), 57 (5), 56 (38), 55 (43) |
| DiPeOHSu (5.4.5) | 12.12 | 258 | 189 (7), 171 (14), 144 (1), 119 (10), 102 (5), 101 (100), 74 (4), 71 (13), 70 (10), 69 (3), 55 (10) |
| DiDeOHSu (10.4.10) | 19.71 | 398 | 259 (24), 241 (4), 207 (2), 141 (17), 119 (62), 111 (2), 101 (100), 97 (5), 85 (35), 83 (15), 71 (36), 69 (27), 57 (50), 55 (38) |
| DiMeOHSub (1.8.1) | 7.94 | 202 | 171 (54), 139 (19), 138 (88), 129 (100), 114 (10), 110 (22), 101 (6), 97 (78), 87 (43), 83 (43), 74 (96), 69 (95), 67 (15), 59 (64), 55 (98) |
| DiPeOHSub (5.8.5) | 15.30 | 326 | 227 (64), 200 (2), 185 (12), 157 (71), 139 (17), 138 (28), 129 (3), 115 (48), 111 (26), 97 (15), 83 (27), 70 (59), 55 (100) |
| DiDeOHSub (10.8.10) | 22.43 | 454 | 327 (1), 297 (27), 281 (1), 253 (2), 209 (1), 157 (49), 140 (10), 115 (16), 111 (15), 97 (20), 83 (32), 70 (43), 69 (65), 57 (58), 56 (67), 55 (100) |
| DiMeOHSe (1.10.1) | 10.30 | 230 | 200 (3), 199 (54), 170 (4), 166 (42), 157 (45), 148 (6), 139 (20), 138 (53), 125 (98), 121 (18), 111 (13), 107 (7), 101 (9), 98 (72), 97 (57), 87 (36), 84 (54), 83 (49), 79 (8), 74 (96), 73 (12), 69 (42), 67 (16), 59 (42), 55 (100) |
| DiPeOHSe (5.10.5) | 17.00 | 342 | 256 (7), 255 (72), 228 (2), 213 (15), 186 (10), 185 (100), 166 (12), 143 (32), 139 (18), 125 (26), 121 (10), 98 (25), 97 (21), 83 (12), 73 (7), 70 (38), 69 (20), 60 (4), 55 (39) |
| DiDeOHSe (10.10.10) | 23.88 | 483 | 325 (50), 283 (4), 253 (2), 207 (5), 186 (6), 185 (100), 166 (4), 143 (10), 139 (8), 125 (10), 111 (4), 97 (18), 83 (18), 70 (29), 69 (38), 57 (43), 55 (50) |
| Structure | Carbon Number | MW (g/mol) | Tc * (Onset, °C) | Tm * (Onset, °C) | Supercooling (°C) | ΔH * (J/g) | ΔH (KJ/mol) | Tdegradation * (Start, °C) | Tdegradation * (End, °C) | |
|---|---|---|---|---|---|---|---|---|---|---|
| DiMeOHOx | C4H6O4 | 4 | 118 | 30.2 ± 2.3 | 46.0 ± 0.7 | 15.7 | 136.1 ± 22.0 | 16.1 | 78 ± 10 | 191 ± 18 |
| DiDeOHSe | C30H58O4 | 30 | 483 | 30.1 ± 1.1 | 31.2 ± 1.1 | 1.2 | 159.6 ± 9.1 | 77.1 | 265 ± 21 | 383 ± 15 |
| DiDeOHSu | C24H46O4 | 24 | 398 | 17.5 ± 1.2 | 23.8 ± 1.1 | 6.3 | 168.3 ± 26.2 | 67.0 | 228 ± 19 | 342 ± 26 |
| DiMeOHSe | C12H22O4 | 12 | 230 | 5.3 ± 3.0 | 23.3 ± 1.3 | 18.0 | 158.3 ± 8.6 | 36.4 | 150 ± 1 | 263 ± 12 |
| DiDeOHOx | C22H42O4 | 22 | 370 | 20.5 ± 0.7 | 23.1 ± 0.3 | 2.6 | 151.7 ± 18.8 | 56.1 | 213 ± 3 | 333 ± 3 |
| DiDeOHSub | C28H54O4 | 28 | 454 | 18.2 ± 1.6 | 22.1 ± 1.5 | 3.9 | 172.3 ± 22.7 | 78.2 | 223 ± 6 | 350 ± 10 |
| DiMeOHSu | C6H10O4 | 6 | 146 | −6.8 ± 6.6 | 10.1 ± 5.5 | 16.9 | 119.6 ± 8.6 | 17.5 | 85 ± 10 | 172 ± 6 |
| DiPeOHSe | C20H38O4 | 20 | 342 | −3.3 ± 0.2 | 0.1 ± 0.5 | 3.4 | 155.0 ± 11.4 | 53.0 | 179 ± 10 | 287 ± 21 |
| DiPeOHOx | C12H22O4 | 12 | 230 | −36.3 ± 3.0 | −16.1 ± 1.5 | 20.2 | 92.5 ± 4.9 | 21.3 | 143 ± 12 | 270 ± 20 |
| DiMeOHSub | C10H18O4 | 10 | 202 | −26.0 ± 2.6 | −16.2 ± 2.3 | 9.8 | 112.2 ± 3.8 | 22.7 | 132 ± 3 | 252 ± 3 |
| DiPeOHSub | C18H34O4 | 18 | 326 | −28.4 ± 2.3 | −20.3 ± 3.5 | 8.1 | 108.6 ± 23.4 | 35.4 | 182 ± 3 | 302 ± 3 |
| DiPeOHSu | C14H26O4 | 14 | 258 | N.A. | N.A. | N.A. | N.A. | N.A. | 147 ± 24 | 260 ± 18 |
| Structure | Carbon Number | MW (g/mol) | Tc (Onset, °C) | Tm (Onset, °C) | Supercooling (°C) | ΔH (J/g) | ΔH (KJ/mol) | Tdegradation (Start, °C) | Tdegradation (End, °C) | |
|---|---|---|---|---|---|---|---|---|---|---|
| DiDeOHSe | C30H58O4 | 30 | 483 | 30.1 ± 1.1 | 31.2 ± 1.1 | 1.2 | 159.6 ± 9.1 | 77.1 | 265 ± 21 | 383 ± 15 |
| DiDeOHSu | C24H46O4 | 24 | 398 | 17.5 ± 1.2 | 23.8 ± 1.1 | 6.3 | 168.3 ± 26.2 | 67.0 | 228 ± 19 | 342 ± 26 |
| DiDeOHSub | C28H54O4 | 28 | 454 | 18.2 ± 1.6 | 22.1 ± 1.5 | 3.9 | 172.3 ± 22.7 | 78.2 | 223 ± 6 | 350 ± 10 |
| DiDeOHOx | C22H42O4 | 22 | 370 | 20.5 ± 0.7 | 23.1 ± 0.3 | 2.6 | 151.7 ± 18.8 | 56.1 | 213 ± 3 | 333 ± 3 |
| DiPeOHSub | C18H34O4 | 18 | 326 | −28.4 ± 2.3 | −20.3 ± 3.5 | 8.1 | 108.6 ± 23.4 | 35.4 | 182 ± 3 | 302 ± 3 |
| DiPeOHSe | C20H38O4 | 20 | 342 | −3.3 ± 0.2 | 0.1 ± 0.5 | 3.4 | 155.0 ± 11.4 | 53.0 | 179 ± 10 | 287 ± 21 |
| DiPeOHOx | C12H22O4 | 12 | 230 | −36.3 ± 3.0 | −16.1 ± 1.5 | 20.2 | 92.5 ± 4.9 | 21.3 | 143 ± 12 | 270 ± 20 |
| DiMeOHSe | C12H22O4 | 12 | 230 | 5.3 ± 3.0 | 23.3 ± 1.3 | 18.0 | 158.3 ± 8.6 | 36.4 | 150 ± 1 | 263 ± 12 |
| DiPeOHSu | C14H26O4 | 14 | 258 | N.A. | N.A. | N.A. | N.A. | N.A. | 147 ± 24 | 260 ± 18 |
| DiMeOHSub | C10H18O4 | 10 | 202 | −26.0 ± 2.6 | −16.2 ± 2.3 | 9.8 | 112.2 ± 3.8 | 22.7 | 132 ± 3 | 252 ± 3 |
| DiMeOHOx | C4H6O4 | 4 | 118 | 30.2 ± 2.3 | 46.0 ± 0.7 | 15.7 | 136.1 ± 22.0 | 16.1 | 78 ± 10 | 191 ± 18 |
| DiMeOHSu | C6H10O4 | 6 | 146 | −6.8 ± 6.6 | 10.1 ± 5.5 | 16.9 | 119.6 ± 8.6 | 17.5 | 85 ± 10 | 172 ± 6 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ravotti, R.; Fellmann, O.; Fischer, L.J.; Worlitschek, J.; Stamatiou, A. Investigation of the Thermal Properties of Diesters from Methanol, 1-Pentanol, and 1-Decanol as Sustainable Phase Change Materials. Materials 2020, 13, 810. https://doi.org/10.3390/ma13040810
Ravotti R, Fellmann O, Fischer LJ, Worlitschek J, Stamatiou A. Investigation of the Thermal Properties of Diesters from Methanol, 1-Pentanol, and 1-Decanol as Sustainable Phase Change Materials. Materials. 2020; 13(4):810. https://doi.org/10.3390/ma13040810
Chicago/Turabian StyleRavotti, Rebecca, Oliver Fellmann, Ludger J. Fischer, Jörg Worlitschek, and Anastasia Stamatiou. 2020. "Investigation of the Thermal Properties of Diesters from Methanol, 1-Pentanol, and 1-Decanol as Sustainable Phase Change Materials" Materials 13, no. 4: 810. https://doi.org/10.3390/ma13040810
APA StyleRavotti, R., Fellmann, O., Fischer, L. J., Worlitschek, J., & Stamatiou, A. (2020). Investigation of the Thermal Properties of Diesters from Methanol, 1-Pentanol, and 1-Decanol as Sustainable Phase Change Materials. Materials, 13(4), 810. https://doi.org/10.3390/ma13040810







