Magnetic Iron Oxide Nanoparticle (IONP) Synthesis to Applications: Present and Future
Abstract
1. Introduction
2. Synthesis of IONPs
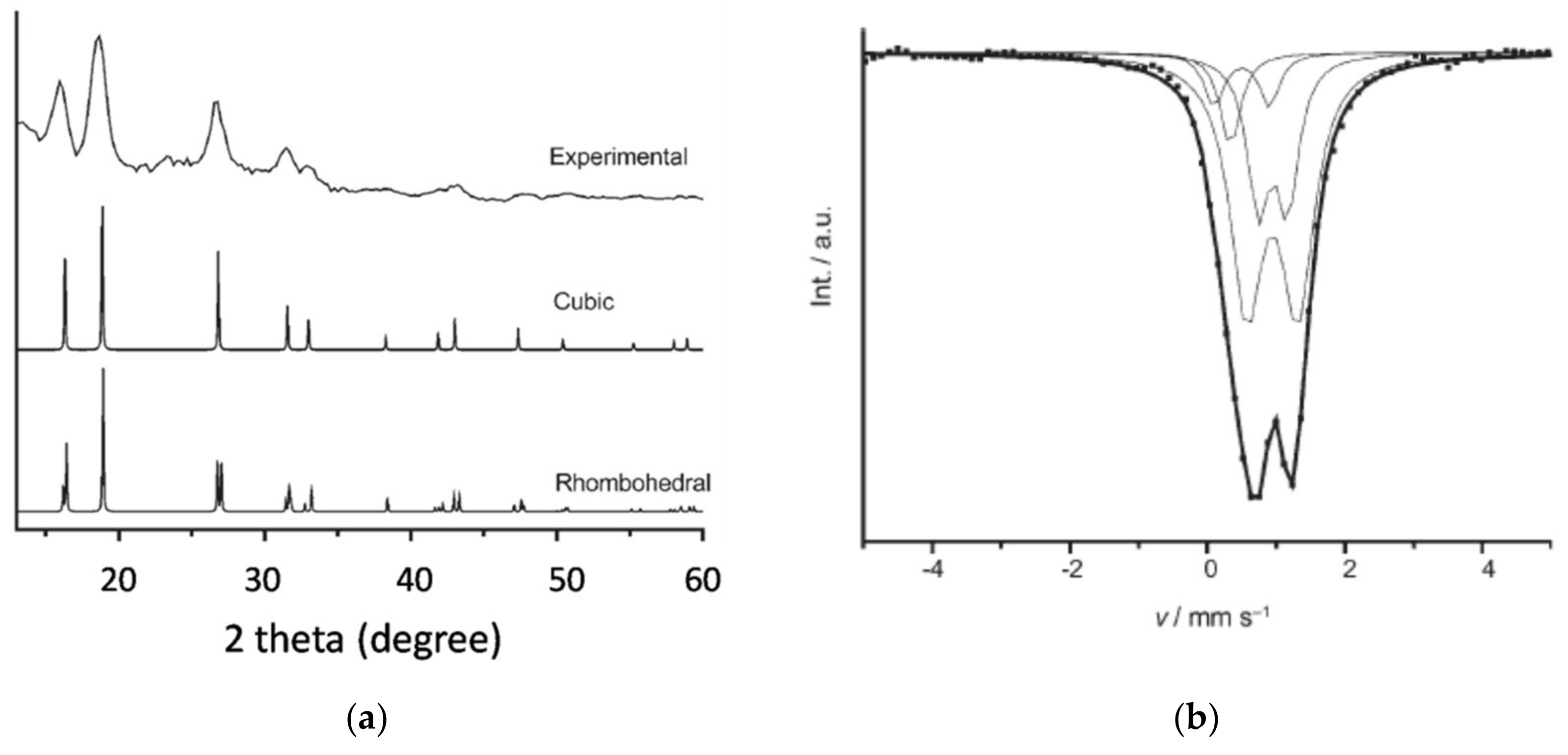
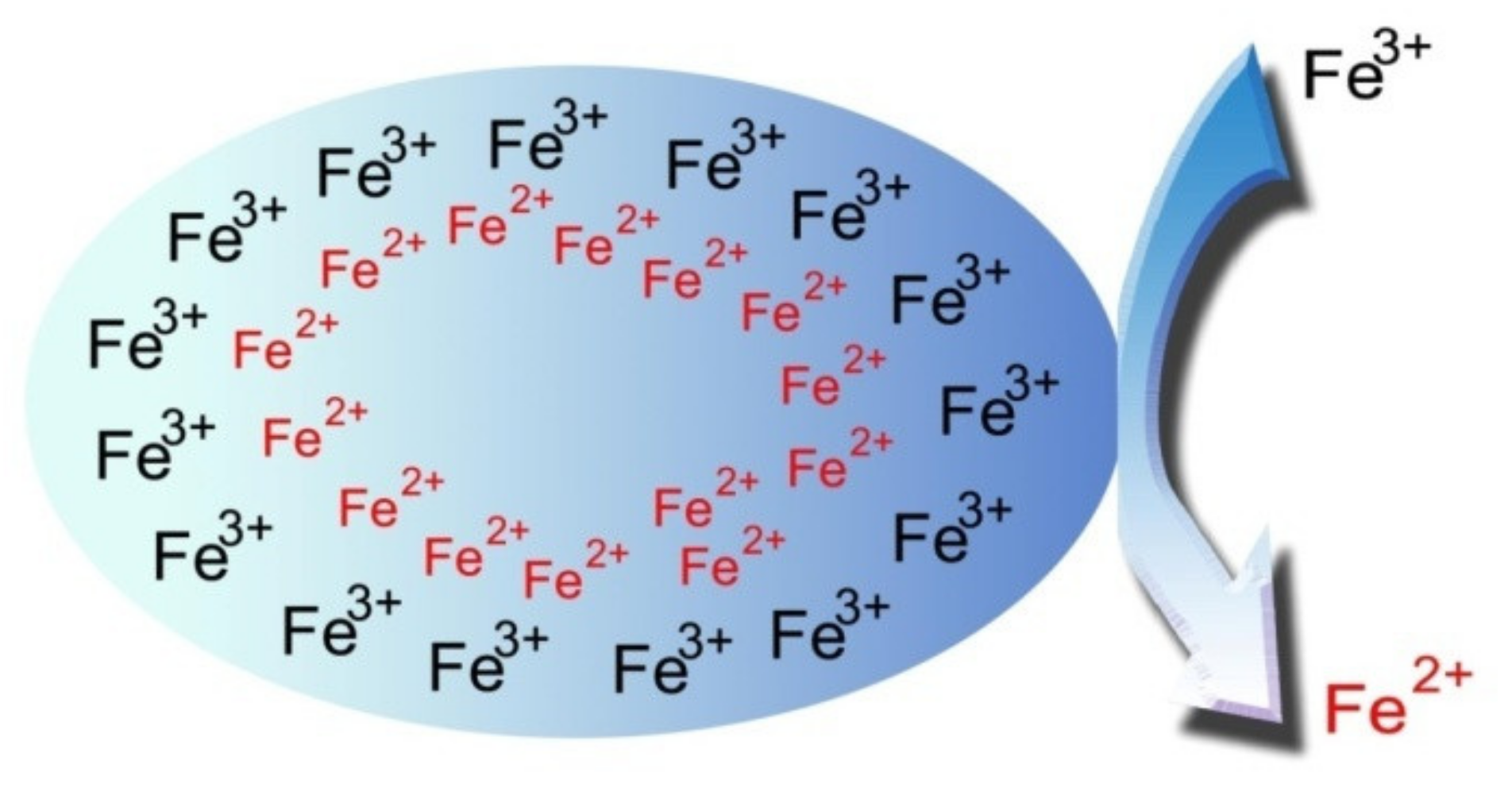
2.1. FeO Nanoparticle Synthesis
2.2. α-Fe2O3 Nanoparticle Synthesis
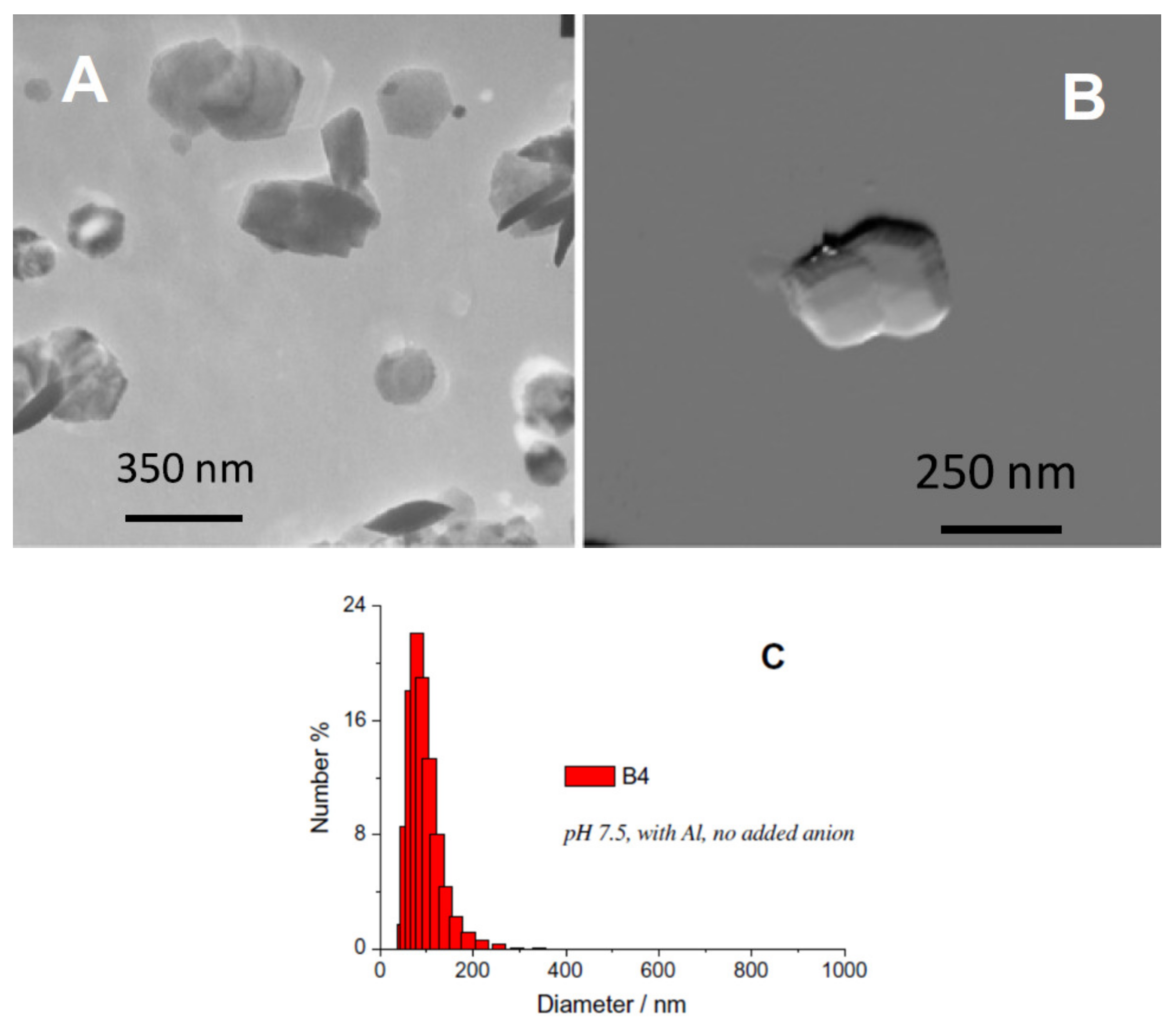
2.3. γ-Fe2O3 Nanoparticle Synthesis
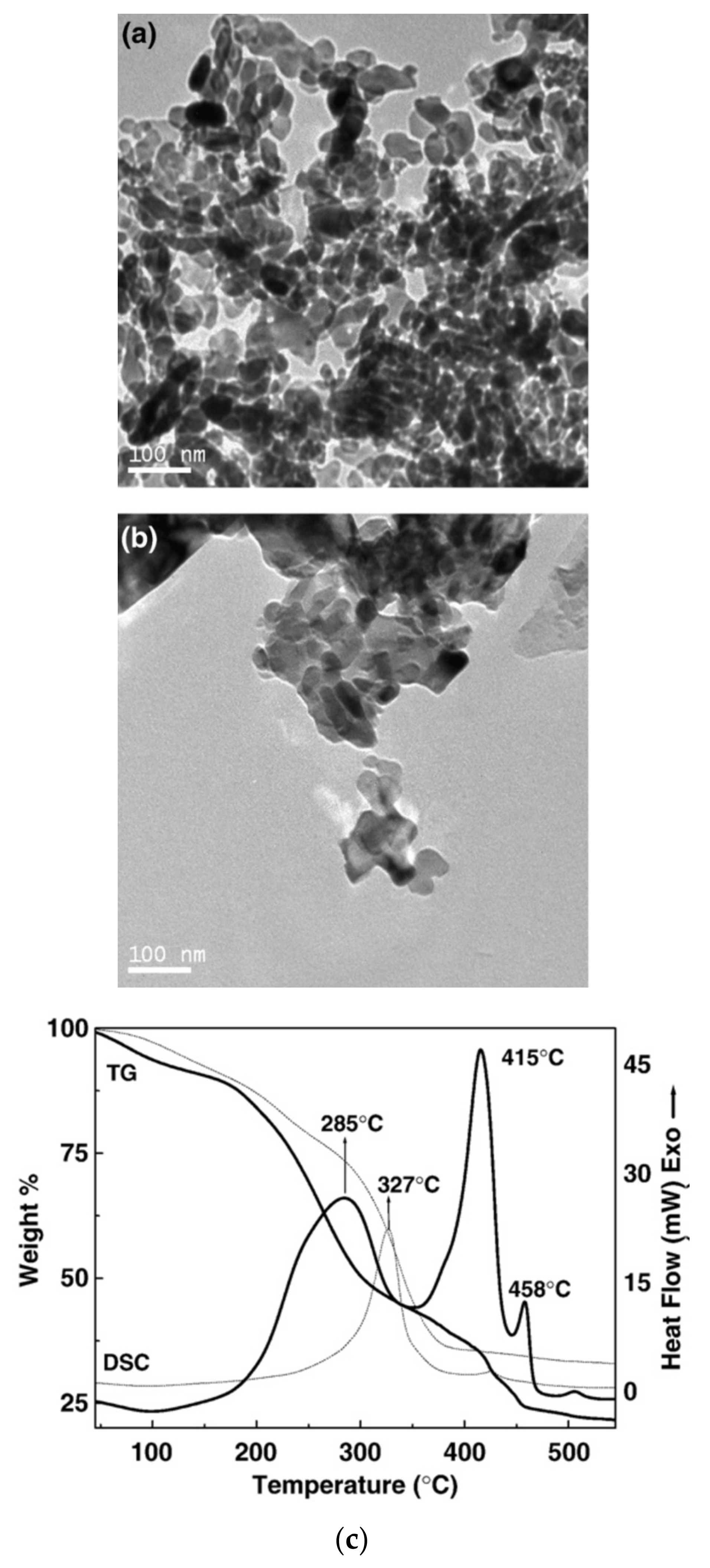
2.4. Fe3O4 Nanoparticle Synthesis
3. Different Mechanisms of Iron Oxide Nanoparticle Synthesis
| HOOC-PEG-COOH (Polyethylene glycol) | + | 2R-NH2 (Oleamine) | → | RNH3+−OOC-PEG-COO−+H3NR (Amine salt) |
| + | ||||
| Fe(acac)3 | ||||
| ↓ | ||||
| Fe3O4 NPs | ||||
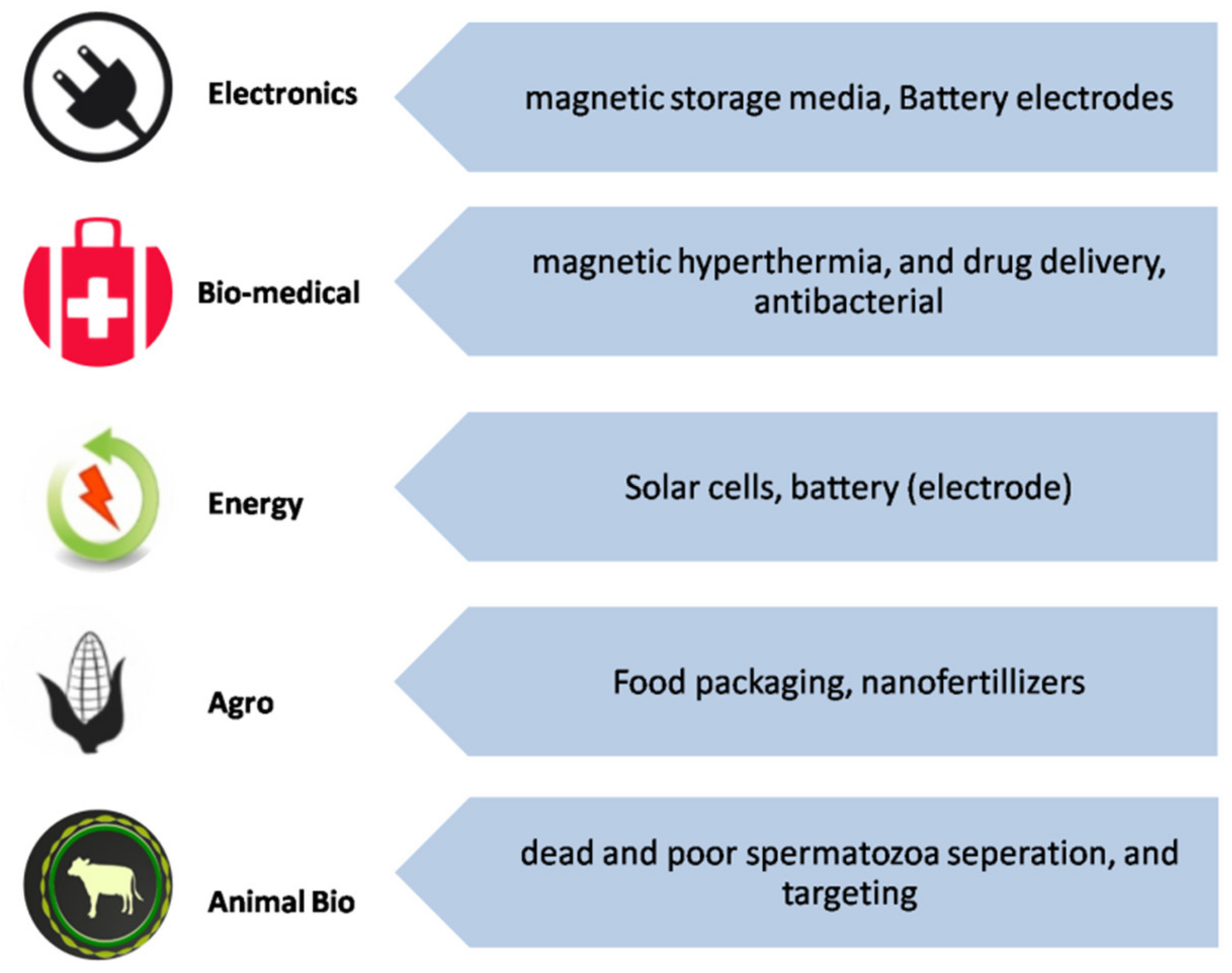
4. Application of IONPs
4.1. Biomedical Application
4.1.1. Biosensing Application of IONPs as Nanozymes
4.1.2. Hyperthermia to Cure Cancer Using IONPs
4.1.3. Drug Delivery
4.1.4. Alternative Immunosuppressive Activity of IONPs
4.1.5. Anticonvulsant Activity of IONPs
4.1.6. Antifungal Activity of IONPs
4.1.7. Antibiotic Activity of IONPs
4.1.8. IONPs for Imaging
4.1.9. Cellular Labeling/Cell Separation Using IONPs
4.1.10. IONP Tissue Engineering/Tissue Repair
4.1.11. Stem Cell Tracking by IONPs
4.1.12. Transplant Monitoring by IONPs
4.1.13. Fluorescence Techniquesand Encapsulated IONPs
5. Future Directions
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Roca, A.G.; Gutiérrez, L.; Gavilán, H.; Fortes Brollo, M.E.; Veintemillas-Verdaguer, S.; del Puerto Morales, M. Design strategies for shape-controlled magnetic iron oxide nanoparticles. Adv. Drug Deliv. Rev. 2019, 138, 68–104. [Google Scholar] [CrossRef]
- Noqta, O.A.; Aziz, A.A.; Usman, I.A.; Bououdina, M. Recent Advances in Iron Oxide Nanoparticles (IONPs): Synthesis and Surface Modification for Biomedical Applications. J. Supercond. Nov. Magn. 2019, 32, 779–795. [Google Scholar] [CrossRef]
- Iriarte-Mesa, C.; López, Y.C.; Matos-Peralta, Y.; de la Vega-Hernández, K.; Antuch, M. Gold, Silver and Iron Oxide Nanoparticles: Synthesis and Bionanoconjugation Strategies Aimed at Electrochemical Applications; Springer International Publishing: Berlin/Heidelberg, Germany, 2020; Volume 378, ISBN 0123456789. [Google Scholar]
- Trindade, T.; Thomas, P.J. Defining and Using Very Small Crystals; Elsevier Ltd.: Amsterdam, The Netherlands, 2013; Volume 4, ISBN 9780080965291. [Google Scholar] [CrossRef]
- Tringides, M.C.; Jałochowski, M.; Bauer, E. Quantum size effects in metallic nanostructures. Phys. Today 2007, 60, 50–54. [Google Scholar] [CrossRef]
- Li, Q.; Kartikowati, C.W.; Horie, S.; Ogi, T.; Iwaki, T.; Okuyama, K. Correlation between particle size/domain structure and magnetic properties of highly crystalline Fe3O4 nanoparticles. Sci. Rep. 2017, 7, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Gray, S.K.; Peng, S. Surface chemistry: A non-negligible parameter in determining optical properties of small colloidal metal nanoparticles. Phys. Chem. Chem. Phys. 2011, 13, 11814–11826. [Google Scholar] [CrossRef] [PubMed]
- Sudhakara, K.; Kumar, A.P.; Kumara, B.P.; Raghavendera, A.; Ravia, S.; Kenie, D.N.; Lee, Y.-I. Synthesis of γ-Fe2O3 Nanoparticles and Catalytic activity of Azide-Alkyne Cycloaddition Reactions. Asian J. Nanosci. Mater. 2018, 1, 172–182. [Google Scholar] [CrossRef]
- Cao, D.; Li, H.; Pan, L.; Li, J.; Wang, X.; Jing, P.; Cheng, X.; Wang, W.; Wang, J.; Liu, Q. High saturation magnetization of γ 3-Fe2 O3 nano-particles by a facile one-step synthesis approach. Sci. Rep. 2016, 6, 1–9. [Google Scholar] [CrossRef]
- Mohapatra, M.; Anand, S. Synthesis and applications of nano-structured iron oxides/hydroxides—A review. Int. J. Eng. Sci. Technol. 2011, 2, 127–146. [Google Scholar] [CrossRef]
- Fang, S.; Bresser, D.; Passerini, S. Transition Metal Oxide Anodes for Electrochemical Energy Storage in Lithium- and Sodium-Ion Batteries. Adv. Energy Mater. 2020, 10, 10. [Google Scholar] [CrossRef]
- Magro, M.; Baratella, D.; de Bonaiuto, E.A.; Roger, J.; Vianello, F. New Perspectives on Biomedical Applications of Iron Oxide Nanoparticles. Curr. Med. Chem. 2018, 25, 540–555. [Google Scholar] [CrossRef]
- Andrade, R.G.D.; Veloso, S.R.S.; Castanheira, E.M.S. Shape anisotropic iron oxide-based magnetic nanoparticles: Synthesis and biomedical applications. Int. J. Mol. Sci. 2020, 21, 2455. [Google Scholar] [CrossRef]
- Arakha, M.; Pal, S.; Samantarrai, D.; Panigrahi, T.K.; Mallick, B.C.; Pramanik, K.; Mallick, B.; Jha, S. Antimicrobial activity of iron oxide nanoparticle upon modulation of nanoparticle-bacteria interface. Sci. Rep. 2015, 5, 14813. [Google Scholar] [CrossRef] [PubMed]
- Kostyantyn, T.; Turek, Z.; Zanáška, M.; Kudrna, P.; Tichý, M. Iron Oxide and Iron Sulfide Films Prepared for Dye-Sensitized Solar Cells. Materials 2020, 13, 1797. [Google Scholar] [CrossRef]
- Wang, Q.; Ma, Y.; Liu, L.; Yao, S.; Wu, W.; Wang, Z.; Lv, P.; Zheng, J.; Yu, K.; Wei, W. Plasma enabled Fe2O3/Fe3O4 nano-aggregates anchored on nitrogen-doped graphene as anode for sodium-ion batteries. Nanomaterials 2020, 10, 782. [Google Scholar] [CrossRef]
- Cichello, S.A. Oxygen absorbers in food preservation: A review. J. Food Sci. Technol. 2015, 52, 1889–1895. [Google Scholar] [CrossRef]
- Rui, M.; Ma, C.; Hao, Y.; Guo, J.; Rui, Y.; Tang, X.; Zhao, Q.; Fan, X.; Zhang, Z.; Hou, T. Iron oxide nanoparticles as a potential iron fertilizer for peanut (Arachis hypogaea). Front. Plant Sci. 2016, 7, 815. [Google Scholar] [CrossRef]
- Durfey, C.L.; Swistek, S.E.; Liao, S.F.; Crenshaw, M.A.; Clemente, H.J.; Thirumalai, R.V.K.G.; Steadman, C.S.; Ryan, P.L.; Willard, S.T.; Feugang, J.M. Nanotechnology-based approach for safer enrichment of semen with best spermatozoa. J. Anim. Sci. Biotechnol. 2019, 10, 1–12. [Google Scholar] [CrossRef]
- Magdanz, V.; Gebauer, J.; Sharan, P.; Eltoukhy, S.; Voigt, D.; Simmchen, J. Sperm–Particle Interactions and Their Prospects for Charge Mapping. Adv. Biosyst. 2019, 3, 1–23. [Google Scholar] [CrossRef]
- Arias, L.S.; Pessan, J.P.; Vieira, A.P.M.; De Lima, T.M.T.; Delbem, A.C.B.; Monteiro, D.R. Iron oxide nanoparticles for biomedical applications: A perspective on synthesis, drugs, antimicrobial activity, and toxicity. Antibiotics 2018, 7, 46. [Google Scholar] [CrossRef]
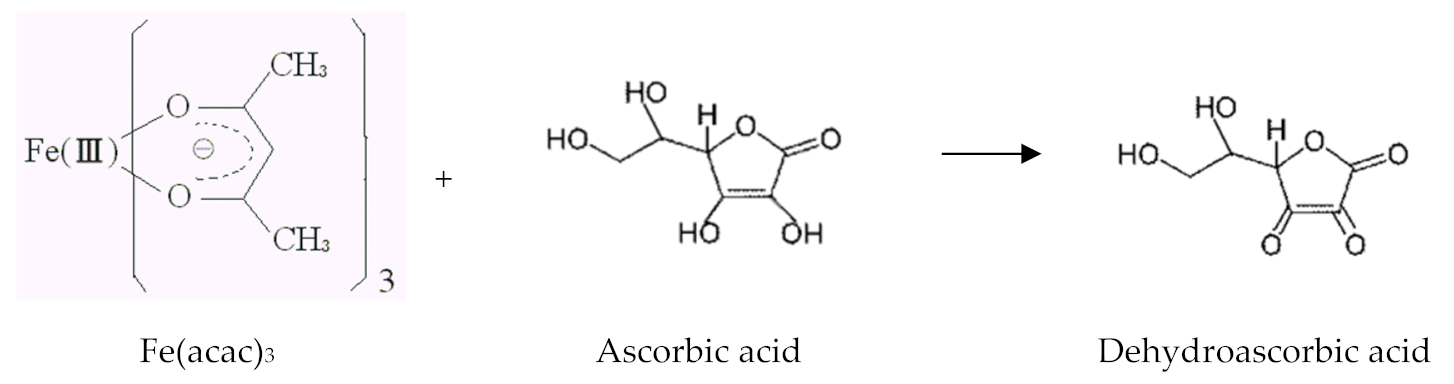
- Nene, A.G.; Takahashi, M.; Wakita, K.; Umeno, M. Size controlled synthesis of Fe3O4 nanoparticles by ascorbic acid mediated reduction of Fe(acac)3 without using capping agent. J. Nano Res. 2016, 40, 8–19. [Google Scholar] [CrossRef]
- Huang, P.H.; Zhao, S.; Bachman, H.; Nama, N.; Li, Z.; Chen, C.; Yang, S.; Wu, M.; Zhang, S.P.; Huang, T.J. Acoustofluidic Synthesis of Particulate Nanomaterials. Adv. Sci. 2019, 6, 6. [Google Scholar] [CrossRef]
- Jeevanandam, J.; Barhoum, A.; Chan, Y.S.; Dufresne, A.; Danquah, M.K. Review on nanoparticles and nanostructured materials: History, sources, toxicity and regulations. Beilstein J. Nanotechnol. 2018, 9, 1050–1074. [Google Scholar] [CrossRef] [PubMed]
- Nene, A.G.; Takahashi, M.; Somani, P.R.; Aryal, H.R.; Wakita, K.; Umeno, M. Synthesis and characterization of graphene-Fe3O4 nanocomposite. Carbon Sci. Technol. 2016, 8, 13–24. [Google Scholar]
- Nene, A.G.; Somani, P.R.; Takahashi, M.; Umeno, M.; Technologies, G. Effect of experimental parameters on the synthesis of Fe3O4 nanoparticles by ascorbic acid mediated reduction of Fe(acac)3. Carbon Sci. Technol. 2019, 3, 6–26. [Google Scholar]
- Nene, A.G.; Takahashi, M.; Somani, P.R. Fe3O4 and Fe Nanoparticles by Chemical Reduction of Fe(acac) 3 by Ascorbic Acid: Role of Water Keywords Fe 3 O 4 Nanoparticles, Fe-Nanoparticles, Iron Oxide, Chemical Reduction Method. World J. Nano Sci. Eng. 2016, 6, 20–28. [Google Scholar] [CrossRef]
- Ambro, Ž.Č.G.; Orel, Z.C.; Žigon, M. Microwave-assisted non-aqueous synthesis of ZnO nanoparticles. Mater. Tehnol. 2011, 45, 173–177. [Google Scholar]
- Morán-Lázaro, J.P.; Guillen-López, E.S.; López-Urias, F.; Muñoz-Sandoval, E.; Blanco-Alonso, O.; Guillén-Bonilla, H.; Guillén-Bonilla, A.; Rodríguez-Betancourtt, V.M.; Sanchez-Tizapa, M.; Olvera-Amador, M.D.L.L. Synthesis of znmn2 o4 nanoparticles by a microwave-assisted colloidal method and their evaluation as a gas sensor of propane and carbon monoxide. Sensors 2018, 18, 701. [Google Scholar] [CrossRef]
- Jung, D.S.; Ko, Y.N.; Kang, Y.C.; Park, S. Bin. Recent progress in electrode materials produced by spray pyrolysis for next-generation lithium ion batteries. Adv. Powder Technol. 2014, 25, 18–31. [Google Scholar] [CrossRef]
- Hasany, S.F.; Abdurahman, N.H.; Sunarti, A.R.; Jose, R. Magnetic iron oxide nanoparticles: Synthesis and applications. Curr. Nanosci. 2013, 9, 1–15. [Google Scholar]
- Eslamian, M.; Ahmed, M.; Ashgriz, N. Modelling of nanoparticle formation during spray pyrolysis. Nanotechnology 2006, 17, 1674–1685. [Google Scholar] [CrossRef]
- Umer, A.; Naveed, S.; Ramzan, N.; Rafique, M.S. Selection of a suitable method for the synthesis of copper nanoparticles. Nano 2012, 7, 1230005. [Google Scholar] [CrossRef]
- Gao, X.; Yokota, N.; Oda, H.; Tanaka, S.; Hokamoto, K.; Chen, P. One step preparation of Fe–FeO–graphene nanocomposite through pulsed wire discharge. Crystals 2018, 8, 104. [Google Scholar] [CrossRef]
- Dong, X.L.; Choi, C.J.; Kim, B.K. Structural and magnetic characterization of Fe nanoparticles synthesized by chemical vapor condensation process. J. Appl. Phys. 2002, 92, 5380–5385. [Google Scholar] [CrossRef]
- Kumar, H.; Sangwan, P. Synthesis and Characterization of MnO 2 Nanoparticles using Co-precipitation Technique. Int. J. Chem. Chem. Eng. 2013, 3, 155–160. [Google Scholar]
- Wang, B.; Wei, Q.; Qu, S. Synthesis and characterization of uniform and crystalline magnetite nanoparticles via oxidation-precipitation and modified co-precipitation methods. Int. J. Electrochem. Sci. 2013, 8, 3786–3793. [Google Scholar]
- Hariani, P.L.; Faizal, M.; Ridwan, R.; Marsi, M.; Setiabudidaya, D. Synthesis and Properties of Fe3O4 Nanoparticles by Co-precipitation Method to Removal Procion Dye. Int. J. Environ. Sci. Dev. 2013, 4, 336–340. [Google Scholar] [CrossRef]
- Wu, W.; He, Q.; Jiang, C. Magnetic iron oxide nanoparticles: Synthesis and surface functionalization strategies. Nanoscale Res. Lett. 2008, 3, 397–415. [Google Scholar] [CrossRef]
- Singamaneni, S.; Bliznyuk, V.N.; Binek, C.; Tsymbal, E.Y. Magnetic nanoparticles: Recent advances in synthesis, self-assembly and applications. J. Mater. Chem. 2011, 21, 16819–16845. [Google Scholar] [CrossRef]
- Campanini, M.; Ciprian, R.; Bedogni, E.; Mega, A.; Chiesi, V.; Casoli, F.; De Julián Fernández, C.; Rotunno, E.; Rossi, F.; Secchi, A.; et al. Lorentz microscopy sheds light on the role of dipolar interactions in magnetic hyperthermia. Nanoscale 2015, 7, 7717–7725. [Google Scholar] [CrossRef]
- Ansari, S.A.M.K.; Ficiarà, E.; Ruffinatti, F.A.; Stura, I.; Argenziano, M.; Abollino, O.; Cavalli, R.; Guiot, C.; D’Agata, F. Magnetic iron oxide nanoparticles: Synthesis, characterization and functionalization for biomedical applications in the Central Nervous System. Materials 2019, 12, 465. [Google Scholar] [CrossRef]
- Takai, Z.I.; Mustafa, M.K.; Asman, S.; Sekak, K.A. Preparation and characterization of magnetite (Fe3O4) nanoparticles by sol-gel method. Int. J. Nanoelectron. Mater. 2019, 12, 37–46. [Google Scholar]
- Rasheed, R.T.; Al-Algawi, S.D.; Kareem, H.H.; Mansoor, H.S. Preparation and Characterization of Hematite Iron Oxide (?-Fe2O3) by Sol-Gel Method. Chem. Sci. J. 2018, 9, 1000197. [Google Scholar] [CrossRef]
- Thiagarajan, S.; Sanmugam, A.; Vikraman, D. Facile Methodology of Sol-Gel Synthesis for Metal Oxide Nanostructures. Recent Appl. Sol-Gel Synth. 2017, 1–17. [Google Scholar] [CrossRef]
- Hassanjani-Roshan, A.; Vaezi, M.R.; Shokuhfar, A.; Rajabali, Z. Synthesis of iron oxide nanoparticles via sonochemical method and their characterization. Particuology 2011, 9, 95–99. [Google Scholar] [CrossRef]
- Singh, P.; Kim, Y.J.; Zhang, D.; Yang, D.C. Biological Synthesis of Nanoparticles from Plants and Microorganisms. Trends Biotechnol. 2016, 34, 588–599. [Google Scholar] [CrossRef]
- Varshney, R.; Bhadauria, S.; Gaur, M.S. A review: Biological synthesis of silver and copper nanoparticles. Nano Biomed. Eng. 2012, 4, 99–106. [Google Scholar] [CrossRef]
- Yin, M.; Chen, Z.; Deegan, B.; O’Brien, S. Wüstite nanocrystals: Synthesis, structure and superlattice formation. J. Mater. Res. 2007, 22, 1987–1995. [Google Scholar] [CrossRef]
- Ullrich, A.; Rölle, N.; Horn, S. From wustite to hematite: Thermal transformation of differently sized iron oxide nanoparticles in air. J. Nanoparticle Res. 2019, 21, 1–8. [Google Scholar] [CrossRef]
- Khurshid, H.; Li, W.; Chandra, S.; Phan, M.H.; Hadjipanayis, G.C.; Mukherjee, P.; Srikanth, H. Mechanism and controlled growth of shape and size variant core/shell FeO/Fe3O4 nanoparticles. Nanoscale 2013, 5, 7942–7952. [Google Scholar] [CrossRef]
- Strobel, R.; Pratsinis, S.E. Direct synthesis of maghemite, magnetite and wustite nanoparticles by flame spray pyrolysis. Adv. Powder Technol. 2009, 20, 190–194. [Google Scholar] [CrossRef]
- Chen, C.J.; Chiang, R.K.; Lai, H.Y.; Lin, C.R. Characterization of monodisperse wüstite nanoparticles following partial oxidation. J. Phys. Chem. C 2010, 114, 4258–4263. [Google Scholar] [CrossRef]
- Glaria, A.; Kahn, M.L.; Lecante, P.; Barbara, B.; Chaudret, B. Fe1-yO nanoparticles: Organometallic synthesis and magnetic properties. ChemPhysChem 2008, 9, 776–780. [Google Scholar] [CrossRef]
- Pichon, B.P.; Gerber, O.; Lefevre, C.; Florea, I.; Fleutot, S.; Baaziz, W.; Pauly, M.; Ohlmann, M.; Ulhaq, C.; Ersen, O.; et al. Microstructural and magnetic investigations of Wüstite-spinel core-shell cubic-shaped nanoparticles. Chem. Mater. 2011, 23, 2886–2900. [Google Scholar] [CrossRef]
- Guntlin, C.P.; Ochsenbein, S.T.; Wörle, M.; Erni, R.; Kravchyk, K.V.; Kovalenko, M.V. Popcorn-Shaped FexO (Wüstite) Nanoparticles from a Single-Source Precursor: Colloidal Synthesis and Magnetic Properties. Chem. Mater. 2018, 30, 1249–1256. [Google Scholar] [CrossRef] [PubMed]
- Akbar, S.; Hasanain, S.K.; Azmat, N.; Nadeem, M. Synthesis of Fe2O3 nanoparticles by new Sol-Gel method and their structural and magnetic characterizations. arXiv, 2004; arXiv:cond-mat/0408480. [Google Scholar]
- Morales Morales, J.A. Synthesis of hematite α-Fe2O3 nano powders by the controlled precipitation method. Cienc. Desarro. 2017, 8, 99–107. [Google Scholar] [CrossRef]
- Sarangi, P.P.; Naik, B.; Ghosh, N.N. Low temperature synthesis of single-phase α-Fe2O3 nano-powders by using simple but novel chemical methods. Powder Technol. 2009, 192, 245–249. [Google Scholar] [CrossRef]
- Colombo, C.; Palumbo, G.; Ceglie, A.; Angelico, R. Characterization of synthetic hematite (α-Fe2O3) nanoparticles using a multi-technique approach. J. Colloid Interface Sci. 2012, 374, 118–126. [Google Scholar] [CrossRef]
- Štarha, P.; Smola, D.; Tuček, J.; Trávníček, Z. Efficient synthesis of a maghemite/gold hybrid nanoparticle system as a magnetic carrier for the transport of platinum-based metallotherapeutics. Int. J. Mol. Sci. 2015, 16, 2034–2051. [Google Scholar] [CrossRef]
- Girod, M.; Vogel, S.; Szczerba, W.; Thünemann, A.F. How temperature determines formation of maghemite nanoparticles. J. Magn. Magn. Mater. 2015, 380, 163–167. [Google Scholar] [CrossRef]
- Schwaminger, S.P.; Syhr, C.; Berensmeier, S. Controlled synthesis of magnetic iron oxide nanoparticles: Magnetite or maghemite? Crystals 2020, 10, 214. [Google Scholar] [CrossRef]
- Karami, H. Synthesis and characterization of iron oxide nanoparticles by solid state chemical reaction method. J. Clust. Sci. 2010, 21, 11–20. [Google Scholar] [CrossRef]
- Zulfikar, M.A.; Suri, F.I.; Rusnadi; Setiyanto, H.; Mufti, N.; Ledyastuti, M.; Wahyuningrum, D. Fe3O4 nano-particles prepared by co-precipitation method using local sands as a raw material and their application for humic acid removal. Int. J. Environ. Stud. 2016, 73, 79–94. [Google Scholar] [CrossRef]
- Predoi, D.; Andronescub, E.; Raduc, M.; Munteanuc, M.C.; Dinischiotu, A. Synthesis and Characterization of. Dig. J. Nanomater. Biostruct. 2010, 5, 779–786. [Google Scholar]
- Nurdin, I.; Johan, M.R.; Yaacob, I.I.; Ang, B.C. Effect of nitric acid concentrations on synthesis and stability of maghemite nanoparticles suspension. Sci. World J. 2014, 2014, 589479. [Google Scholar] [CrossRef]
- Múzquiz-Ramos, E.M.; Guerrero-Chávez, V.; Macías-Martínez, B.I.; López-Badillo, C.M.; García-Cerda, L.A. Synthesis and characterization of maghemite nanoparticles for hyperthermia applications. Ceram. Int. 2015, 41, 397–402. [Google Scholar] [CrossRef]
- Tural, B.; Özenbaş, M.; Atalay, S.; Volkan, M. Rapid synthesis and characterization of maghemite nanoparticles. J. Nanosci. Nanotechnol. 2008, 8, 861–866. [Google Scholar] [CrossRef]
- Roca, A.G.; Morales, M.P.; O’Grady, K.; Serna, C.J. Structural and magnetic properties of uniform magnetite nanoparticles prepared by high temperature decomposition of organic precursors. Nanotechnology 2006, 17, 2783–2788. [Google Scholar] [CrossRef]
- Koo, K.N.; Ismail, A.F.; Othman, M.H.D.; Rahman, M.A.; Sheng, T.Z. Preparation and characterization of superparamagnetic magnetite (Fe3O4) nanoparticles: A short review. Malaysian J. Fundam. Appl. Sci. 2019, 15, 23–31. [Google Scholar] [CrossRef]
- Toyos-Rodríguez, C.; Calleja-García, J.; Torres-Sánchez, L.; López, A.; Abu-Dief, A.M.; Costa, A.; Elbaile, L.; Crespo, R.D.; Garitaonandia, J.S.; Lastra, E. A Simple and Reliable Synthesis of Superparamagnetic Magnetite Nanoparticles by Thermal Decomposition of Fe(acac)3. J. Nanomater. 2019, 2019, 2464010. [Google Scholar] [CrossRef]
- Kumar, B.; Smita, K.; Cumbal, L.; Debut, A. Biogenic synthesis of iron oxide nanoparticles for 2-arylbenzimidazole fabrication. J. Saudi Chem. Soc. 2014, 18, 364–369. [Google Scholar] [CrossRef]
- Motoyama, J.; Hakata, T.; Kato, R.; Yamashita, N.; Morino, T.; Kobayashi, T.; Honda, H. Size dependent heat generation of magnetite nanoparticles under AC magnetic field for cancer therapy. Biomagn. Res. Technol. 2008, 6, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.C.; Lee, C.Y. Adsorption of ciprofloxacin in water using Fe3O4 nanoparticles formed at low temperature and high reactant concentrations in a rotating packed bed with co-precipitation. Mater. Chem. Phys. 2020, 240, 122049. [Google Scholar] [CrossRef]
- Salviano, L.B.; da Silva Cardoso, T.M.; Silva, G.C.; Silva Dantas, M.S.; de Mello Ferreira, A. Microstructural assessment of magnetite nanoparticles (Fe3O4) obtained by chemical precipitation under different synthesis conditions. Mater. Res. 2018, 21, 2–8. [Google Scholar] [CrossRef]
- Besenhard, M.O.; LaGrow, A.P.; Hodzic, A.; Kriechbaum, M.; Panariello, L.; Bais, G.; Loizou, K.; Damilos, S.; Margarida Cruz, M.; Thanh, N.T.K.; et al. Co-precipitation synthesis of stable iron oxide nanoparticles with NaOH: New insights and continuous production via flow chemistry. Chem. Eng. J. 2020, 399, 125740. [Google Scholar] [CrossRef]
- Daou, T.J.; Pourroy, G.; Bégin-Colin, S.; Grenèche, J.M.; Ulhaq-Bouillet, C.; Legaré, P.; Bernhardt, P.; Leuvrey, C.; Rogez, G. Hydrothermal synthesis of monodisperse magnetite nanoparticles. Chem. Mater. 2006, 18, 4399–4404. [Google Scholar] [CrossRef]
- Mascolo, M.C.; Pei, Y.; Ring, T.A. Room Temperature Co-Precipitation Synthesis of Magnetite Nanoparticles in a Large ph Window with Different Bases. Materials 2013, 6, 5549–5567. [Google Scholar] [CrossRef]
- Panta, P.C.; Bergmann, C.P. Obtention by Coprecipitation and Magnetic Characterization of Fe3O4 Nanoparticles Coated with Surfactants. Nano Res. Appl. 2015, 1, 1–4. [Google Scholar]
- Bhuiyan, M.S.H.; Miah, M.Y.; Paul, S.C.; Das Aka, T.; Saha, O.; Rahaman, M.M.; Sharif, M.J.I.; Habiba, O.; Ashaduzzaman, M. Green synthesis of iron oxide nanoparticle using Carica papaya leaf extract: Application for photocatalytic degradation of remazol yellow RR dye and antibacterial activity. Heliyon 2020, 6, e04603. [Google Scholar] [CrossRef]
- Yew, Y.P.; Shameli, K.; Miyake, M.; Ahmad Khairudin, N.B.B.; Mohamad, S.E.B.; Naiki, T.; Lee, K.X. Green biosynthesis of superparamagnetic magnetite Fe3O4 nanoparticles and biomedical applications in targeted anticancer drug delivery system: A review. Arab. J. Chem. 2020, 13, 2287–2308. [Google Scholar] [CrossRef]
- Grasso, G.; Zane, D.; Dragone, R. Microbial nanotechnology: Challenges and prospects for green biocatalytic synthesis of nanoscale materials for sensoristic and biomedical applications. Nanomaterials 2020, 10, 11. [Google Scholar] [CrossRef]
- Elblbesy, M.A.-A.; Madbouly, A.K.; Hamdan, T.A.-A. Bio-Synthesis of Magnetite Nanoparticles by Bacteria. Sci. Publ. Gr. 2014, 2, 98–103. [Google Scholar] [CrossRef]
- Lassenberger, A.; Grünewald, T.A.; Van Oostrum, P.D.J.; Rennhofer, H.; Amenitsch, H.; Zirbs, R.; Lichtenegger, H.C.; Reimhult, E. Monodisperse Iron Oxide Nanoparticles by Thermal Decomposition: Elucidating Particle Formation by Second-Resolved in Situ Small-Angle X-ray Scattering. Chem. Mater. 2017, 29, 4511–4522. [Google Scholar] [CrossRef] [PubMed]
- Varanda, L.C.; Souza, C.G.S.; Moraes, D.A.; Neves, H.R.; Souza Junior, J.B.; Silva, M.F.; Bini, R.A.; Albers, R.F.; Silva, T.L.; Beck, W. Size and shape-controlled nanomaterials based on modified polyol and thermal decomposition approaches. Brief Rev. 2019, 91. [Google Scholar] [CrossRef]
- Qiu, G.; Wang, Q.; Nie, M. Polypyrrole-Fe3O4 magnetic nanocomposite prepared by ultrasonic irradiation. Macromol. Mater. Eng. 2006, 291, 68–74. [Google Scholar] [CrossRef]
- Abedini, A.; Daud, A.R.; Hamid, M.A.A.; Othman, N.K. Radiolytic formation of Fe3O4 nanoparticles: Influence of radiation dose on structure and magnetic properties. PLoS ONE 2014, 9, e90055. [Google Scholar] [CrossRef]
- Sundar, S.; Piraman, S. Nanospheres of Fe3O4 Synthesis through Sol-gel Technique and Their Structural & Magnetic Characterization. Indian J. Appl. Res. 2011, 3, 123–126. [Google Scholar] [CrossRef]
- Kurchania, R.; Sawant, S.S.; Ball, R.J. Synthesis and characterization of magnetite/polyvinyl alcohol core-shell composite nanoparticles. J. Am. Ceram. Soc. 2014, 97, 3208–3215. [Google Scholar] [CrossRef]
- Bagherzadeh, M.; Mousavi, O.; Shams Ghahfarokhi, Z. Fabrication and characterization of Fe3O4/polyvinylpyrrolidone (Fe3O4/PVP) nanocomposite as a coating for Carbon steel in saline media. New J. Chem. 2020. [Google Scholar] [CrossRef]
- Antarnusa, G.; Suharyadi, E. A synthesis of polyethylene glycol (PEG)-coated magnetite Fe3O4 nanoparticles and their characteristics for enhancement of biosensor. Mater. Res. Express 2020, 7, 7. [Google Scholar] [CrossRef]
- Zhang, L.; He, R.; Gu, H.C. Oleic acid coating on the monodisperse magnetite nanoparticles. Appl. Surf. Sci. 2006, 253, 2611–2617. [Google Scholar] [CrossRef]
- Feijoo, S.; González-Rodríguez, J.; Fernández, L.; Vázquez-Vázquez, C.; Feijoo, G.; Moreira, M.T. Fenton and photo-fenton nanocatalysts revisited from the perspective of life cycle assessment. Catalysts 2020, 10, 23. [Google Scholar] [CrossRef]
- Soenen, S.J.H.; Brisson, A.R.; De Cuyper, M. Addressing the Problem of Cationic Lipid-Mediated Toxicity: The Magnetoliposome Model. Biomaterials 2009, 30, 3691–3701. [Google Scholar] [CrossRef] [PubMed]
- Roonasi, P.; Holmgren, A. A study on the mechanism of magnetite formation based on iron isotope fractionation. In Proceedings of the TMS 2009 Annual Meeting and Exhibition, San Francisco, CA, USA, 15–19 February 2009; pp. 829–836. [Google Scholar]
- Jia, Q.; Zeng, J.; Qiao, R.; Jing, L.; Peng, L.; Gu, F.; Gao, M. Gelification: An effective measure for achieving differently sized biocompatible Fe3O4 nanocrystals through a single preparation recipe. J. Am. Chem. Soc. 2011, 133, 19512–19523. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Shi, Y.F.; Chen, Z.W. Facile synthesis and growth mechanism of uniform Fe3O4 nanorod with strong magnetic response. Adv. Mater. Res. 2013, 699, 616–619. [Google Scholar] [CrossRef]
- Gao, L.; Fan, K.; Yan, X. Iron Oxide Nanozyme: A Multifunctional Enzyme Mimetic for Biomedical Applications. Theranostics 2017, 7, 3207–3227. [Google Scholar] [CrossRef] [PubMed]
- Xu, P.; Zeng, G.M.; Huang, D.L.; Feng, C.L.; Hu, S.; Zhao, M.H.; Lai, C.; Wei, Z.; Huang, C.; Xie, G.X. Use of Iron Oxide Nanomaterials in Wastewater Treatment: A Review. Sci. Total Environ. 2012, 424, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, M.; Sant, S.; Wang, B.; Laurent, S.; Sen, T. Superparamagnetic Iron Oxide Nanoparticles (SPIONs): Development, Surface Modification and Applications in Chemotherapy. Adv. Drug Deliv. Rev. 2011, 63, 24–46. [Google Scholar] [CrossRef]
- Zhang, W. Nanoscale Iron Particles for Environments Remediation: An Overview. J. Nanoparticle Res. 2003, 5, 323–332. [Google Scholar] [CrossRef]
- Bystrzejewska-Piotrowska, G.; Golimowski, J.; Urban, P.L. Nanoparticles: Their Potential Toxicity, Waste and Environmental Management. Waste Manag. 2009, 29, 2587–2595. [Google Scholar] [CrossRef]
- Gupta, A.K.; Gupta, M. Synthesis and Surface Engineering of Iron Oxide Nanoparticles for Biomedical Applications. Biomaterials 2005, 26, 3995–4021. [Google Scholar] [CrossRef] [PubMed]
- Edwards, A.L. APTBLIBE; Lawrence Livermore National Lab.: Livermore, CA, USA, 1990. [Google Scholar] [CrossRef][Green Version]
- Soenen, S.J.H.; Himmelreich, U.; Nuytten, N.; De Cuyper, M. Cytotoxic Effects of Iron Oxide Nanoparticles and Implications for Safety in Cell Labelling. Biomaterials 2011, 32, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Busquets, M.A.; Estelrich, J.; Sánchez-Martín, M.J. Nanoparticles in Magnetic Resonance Imaging: From Simple to Dual Contrast Agents. IJN 2015, 10, 1727. [Google Scholar] [CrossRef]
- Matsui, I. Nanoparticles for Electronic Device Applications: A Brief Review. J. Chem. Eng. Jpn./JCEJ 2005, 38, 535–546. [Google Scholar] [CrossRef]
- Pisanic, T.R.; Blackwell, J.D.; Shubayev, V.I.; Fiñones, R.R.; Jin, S. Nanotoxicity of Iron Oxide Nanoparticle Internalization in Growing Neurons. Biomaterials 2007, 28, 2572–2581. [Google Scholar] [CrossRef]
- Zhu, X.; Zhu, Y.; Murali, S.; Stoller, M.D.; Ruoff, R.S. Nanostructured Reduced Graphene Oxide/Fe2O3 Composite As a High-Performance Anode Material for Lithium Ion Batteries. ACS Nano 2011, 5, 3333–3338. [Google Scholar] [CrossRef]
- Foley, R.W.; Wiek, A. Scenarios of Nanotechnology Innovation Vis-à-Vis Sustainability Challenges. Futures 2014, 64, 1–14. [Google Scholar] [CrossRef]
- Choi, H.C.; Kundaria, S.; Wang, D.; Javey, A.; Wang, Q.; Rolandi, M.; Dai, H. Efficient Formation of Iron Nanoparticle Catalystson Silicon Oxide by Hydroxylamine for Carbon Nanotube Synthesis and Electronics. Nano Lett. 2003, 3, 157–161. [Google Scholar] [CrossRef]
- Park, S.-J.; Kim, S.; Lee, S.; Khim, Z.G.; Char, K.; Hyeon, T. Synthesis and Magnetic Studies of Uniform Iron Nanorods and Nanospheres. J. Am. Chem. Soc. 2000, 122, 8581–8582. [Google Scholar] [CrossRef]
- Rodner, M.; Puglisi, D.; Ekeroth, S.; Helmersson, U.; Shtepliuk, I.; Yakimova, R.; Skallberg, A.; Uvdal, K.; Schütze, A.; Eriksson, J. Graphene Decorated with Iron Oxide Nanoparticles for Highly Sensitive Interaction with Volatile Organic Compounds. Sensors 2019, 19, 918. [Google Scholar] [CrossRef]
- Ali, A.; Zafar, H.; Zia, M.; ul Haq, I.; Phull, A.R.; Ali, J.S.; Hussain, A. Synthesis, Characterization, Applications, and Challenges of Iron Oxide Nanoparticles. NSA 2016, 9, 49–67. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.K.A.; Espinosa, A.; Kolosnjaj-Tabi, J.; Wilhelm, C.; Gazeau, F. Medical Applications of Iron Oxide Nanoparticles. Iron Oxides Nat. Appl. 2016, 425–472. [Google Scholar] [CrossRef]
- Servin, A.; Elmer, W.; Mukherjee, A.; De la Torre-Roche, R.; Hamdi, H.; White, J.C.; Bindraban, P.; Dimkpa, C. A Review of the Use of Engineered Nanomaterials to Suppress Plant Disease and Enhance Crop Yield. J. Nanopart Res. 2015, 17, 92. [Google Scholar] [CrossRef]
- Kumar, S.; Layek, S.; Pandey, B.; Verma, H.C. Magnetic structure of Fe-Fe oxide nanoparticles made by electrodeposition. Int. J. Eng. Sci. Technol. 2010, 2, 66–72. [Google Scholar]
- Kamzin, A.S.; Valiullin, A.A.; Khurshid, H.; Nemati, Z.; Srikanth, H.; Phan, M.H. Mössbauer Studies of Core–Shell FeO/Fe3O4 Nanoparticles. Phys. Solid State 2018, 60, 382–389. [Google Scholar] [CrossRef]
- Cursaru, L.M.; Piticescu, R.M.; Dragut, D.V.; Tudor, I.A.; Kuncser, V.; Iacob, N.; Stoiciu, F. The Influence of Synthesis Parameters on Structural and Magnetic Properties of Iron Oxide Nanomaterials. Nanomaterials 2020, 10, 85. [Google Scholar] [CrossRef]
- Gawande, M.B.; Goswami, A.; Felpin, F.-X.; Asefa, T.; Huang, X.; Silva, R.; Zou, X.; Zboril, R.; Varma, R.S. Cu and Cu-Based Nanoparticles: Synthesis and Applications in Catalysis. Chem. Rev. 2016, 116, 3722–3811. [Google Scholar] [CrossRef]
- Lin, T.; Zhong, L.; Guo, L.; Fu, F.; Chen, G. Seeing Diabetes: Visual Detection of Glucose Based on the Intrinsic Peroxidase-like Activity of MoS2 Nanosheets. Nanoscale 2014, 6, 11856–11862. [Google Scholar] [CrossRef]
- Mahato, K.; Kumar, A.; Maurya, P.K.; Chandra, P. Shifting Paradigm of Cancer Diagnoses in Clinically Relevant Samples Based on Miniaturized Electrochemical Nanobiosensors and Microfluidic Devices. Biosens. Bioelectron. 2018, 100, 411–428. [Google Scholar] [CrossRef]
- Wang, S.; Cazelles, R.; Liao, W.-C.; Vázquez-González, M.; Zoabi, A.; Abu-Reziq, R.; Willner, I. Mimicking Horseradish Peroxidase and NADH Peroxidase by Heterogeneous Cu2+-Modified Graphene Oxide Nanoparticles. Nano Lett. 2017, 17, 2043–2048. [Google Scholar] [CrossRef]
- Yu, F.; Huang, Y.; Cole, A.J.; Yang, V.C. The Artificial Peroxidase Activity of Magnetic Iron Oxide Nanoparticles and Its Application to Glucose Detection. Biomaterials 2009, 30, 4716–4722. [Google Scholar] [CrossRef] [PubMed]
- Giustini, A.J.; Petryk, A.A.; Cassim, S.M.; Tate, J.A.; Baker, I.; Hoopes, P.J. Magnetic Nanoparticle Hyperthermia in Cancer Treatment. Nano Life 2010, 1, 17–32. [Google Scholar] [CrossRef] [PubMed]
- Roussakow, S. The History of Hyperthermia Rise and Decline. Conf. Pap. Med. 2013, 2013, 1–40. [Google Scholar] [CrossRef]
- Chertok, B.; Moffat, B.A.; David, A.E.; Yu, F.; Bergemann, C.; Ross, B.D.; Yang, V.C. Iron Oxide Nanoparticles 9as a Drug Delivery Vehicle for MRI Monitored Magnetic Targeting of Brain Tumors. Biomaterials 2008, 29, 487–496. [Google Scholar] [CrossRef]
- Hu, J.; Qian, Y.; Wang, X.; Liu, T.; Liu, S. Drug-Loaded and Superparamagnetic Iron Oxide Nanoparticle Surface-Embedded Amphiphilic Block Copolymer Micelles for Integrated Chemotherapeutic Drug Delivery and MR Imaging. Langmuir 2012, 28, 2073–2082. [Google Scholar] [CrossRef]
- Sun, C.; Fang, C.; Stephen, Z.; Veiseh, O.; Hansen, S.; Lee, D.; Ellenbogen, R.G.; Olson, J.; Zhang, M. Tumor-Targeted Drug Delivery and MRI Contrast Enhancement by Chlorotoxin-Conjugated Iron Oxide Nanoparticles. Nanomedicine 2008, 3, 495–505. [Google Scholar] [CrossRef]
- Nigam, S.; Bahadur, D. Doxorubicin-Loaded Dendritic-Fe3O4 Supramolecular Nanoparticles for Magnetic Drug Targeting and Tumor Regression in Spheroid Murine Melanoma Model. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 759–768. [Google Scholar] [CrossRef]
- Pillai, G. Nanomedicines for Cancer Therapy: An Update of FDA Approved and Those under Various Stages of Development. SOJPPS 2014. [Google Scholar] [CrossRef]
- Abbasi Aval, N.; Pirayesh Islamian, J.; Hatamian, M.; Arabfirouzjaei, M.; Javadpour, J.; Rashidi, M.-R. Doxorubicin Loaded Large-Pore Mesoporous Hydroxyapatite Coated Superparamagnetic Fe 3 O 4 Nanoparticles for Cancer Treatment. Int. J. Pharm. 2016, 509, 159–167. [Google Scholar] [CrossRef]
- Dutta, B.; Shetake, N.G.; Barick, B.K.; Barick, K.C.; Pandey, B.N.; Priyadarsini, K.I.; Hassan, P.A. PH Sensitive Surfactant-Stabilized Fe3O4 Magnetic Nanocarriers for Dual Drug Delivery. Colloids Surf. B Biointerfaces 2018, 162, 163–171. [Google Scholar] [CrossRef]
- Mu, Q.; Kievit, F.M.; Kant, R.J.; Lin, G.; Jeon, M.; Zhang, M. Anti-HER2/Neu Peptide-Conjugated Iron Oxide Nanoparticles for Targeted Delivery of Paclitaxel to Breast Cancer Cells. Nanoscale 2015, 7, 18010–18014. [Google Scholar] [CrossRef] [PubMed]
- Hsiao, M.-H.; Mu, Q.; Stephen, Z.R.; Fang, C.; Zhang, M. Hexanoyl-Chitosan-PEG Copolymer Coated Iron Oxide Nanoparticles for Hydrophobic Drug Delivery. ACS Macro Lett. 2015, 4, 403–407. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Li, Y.; Li, Y.; Wu, H.; Yu, F.; Zhou, S.; Xie, L.; Luo, F.; Lin, C.; Hou, Z. Drug/Dye-Loaded, Multifunctional PEG–Chitosan–Iron Oxide Nanocomposites for Methotraxate Synergistically Self-Targeted Cancer Therapy and Dual Model Imaging. ACS Appl. Mater. Interfaces 2015, 7, 11908–11920. [Google Scholar] [CrossRef] [PubMed]
- Mu, Q.; Lin, G.; Patton, V.K.; Wang, K.; Press, O.W.; Zhang, M. Gemcitabine and Chlorotoxin Conjugated Iron Oxide Nanoparticles for Glioblastoma Therapy. J. Mater. Chem. B 2016, 4, 32–36. [Google Scholar] [CrossRef]
- Barar, J.; Kafil, V.; Majd, M.H.; Barzegari, A.; Khani, S.; Johari-Ahar, M.; Asgari, D.; Cokous, G.; Omidi, Y. Multifunctional mitoxantrone-conjugated magnetic nanosystem for targeted therapy of folate receptor-overexpressing malignant cells. J. Nanobiotechnol. 2015, 13, 26. [Google Scholar]
- Rao, Y.; Chen, W.; Liang, X.; Huang, Y.; Miao, J.; Liu, L.; Lou, Y.; Zhang, X.; Wang, B.; Tang, R. Epirubicin-Loaded Superparamagnetic Iron-Oxide Nanoparticles for Transdermal Delivery: Cancer Therapy by Circumventing the Skin Barrier. Small 2015, 11, 239–247. [Google Scholar] [CrossRef]
- Akilo, O.D.; Choonara, Y.E.; Strydom, A.M.; du Toit, L.C.; Kumar, P.; Modi, G.; Pillay, V. AN in Vitro Evaluation of a Carmustine-Loaded Nano-Co-Plex for Potential Magnetic-Targeted Intranasal Delivery to the Brain. Int. J. Pharm. 2016, 500, 196–209. [Google Scholar] [CrossRef]
- Shahabadi, N.; Falsafi, M.; Mansouri, K. Improving Antiproliferative Effect of the Anticancer Drug Cytarabine on Human Promyelocytic Leukemia Cells by Coating on Fe3O4@SiO2 Nanoparticles. Colloids Surf. B Biointerfaces 2016, 141, 213–222. [Google Scholar] [CrossRef]
- Alupei, L.; Peptu, C.A.; Lungan, A.-M.; Desbrieres, J.; Chiscan, O.; Radji, S.; Popa, M. New Hybrid Magnetic Nanoparticles Based on Chitosan-Maltose Derivative for Antitumor Drug Delivery. Int. J. Biol. Macromol. 2016, 92, 561–572. [Google Scholar] [CrossRef]
- Al-Jamal, K.T.; Bai, J.; Wang, J.T.-W.; Protti, A.; Southern, P.; Bogart, L.; Heidari, H.; Li, X.; Cakebread, A.; Asker, D. Magnetic Drug Targeting: Preclinical in Vivo Studies, Mathematical Modeling, and Extrapolation to Humans. Nano Lett. 2016, 16, 5652–5660. [Google Scholar] [CrossRef]
- Shelat, R.; Chandra, S.; Khanna, A. Detailed Toxicity Evaluation of β-Cyclodextrin Coated Iron Oxide Nanoparticles for Biomedical Applications. Int. J. Biol. Macromol. 2018, 110, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Sollinger, H.W. Mycophenolate Mofetil for the Prevention of Acute Rejection in Primary Cadaveric Renal Allograft Recipients. Transplantation 1995, 60, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.; Lee, E.; Kim, J.; Seo, Y.; Lee, K.H.; Hong, J.W.; Gilad, A.A.; Park, H.; Choi, J. Effective Delivery of Immunosuppressive Drug Molecules by Silica Coated Iron Oxide Nanoparticles. Colloids Surf. B Biointerfaces 2016, 142, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Fu, T.; Kong, Q.; Sheng, H.; Gao, L. Value of Functionalized Superparamagnetic Iron Oxide Nanoparticles in the Diagnosis and Treatment of Acute Temporal Lobe Epilepsy on MRI. Neural Plast. 2016, 2016, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Rosillo-de la Torre, A.; Zurita-Olvera, L.; Orozco-Suárez, S.; Garcia Casillas, P.E.; Salgado-Ceballos, H.; Luna-Bárcenas, G.; Rocha, L. Phenytoin Carried by Silica Core Iron Oxide Nanoparticles Reduces the Expression of Pharmacoresistant Seizures in Rats. Nanomedicine 2015, 10, 3563–3577. [Google Scholar] [CrossRef]
- Hussein-Al-Ali, S.H.; El Zowalaty, M.E.; Kura, A.U.; Geilich, B.; Fakurazi, S.; Webster, T.J.; Hussein, M.Z. Antimicrobial and Controlled Release Studies of a Novel Nystatin Conjugated Iron Oxide Nanocomposite. Biomed Res. Int. 2014, 2014, 651831. [Google Scholar] [CrossRef]
- Maltas, E.; Ozmen, M.; Yildirimer, B.; Kucukkolbasi, S.; Yildiz, S. Interaction Between Ketoconazole and Human Serum Albumin on Epoxy Modified Magnetic Nanoparticles for Drug Delivery. J. Nanosci. Nanotech. 2013, 13, 6522–6528. [Google Scholar] [CrossRef]
- Niemirowicz, K.; Durnaś, B.; Tokajuk, G.; Głuszek, K.; Wilczewska, A.Z.; Misztalewska, I.; Mystkowska, J.; Michalak, G.; Sodo, A.; Wątek, M. Magnetic Nanoparticles as a Drug Delivery System That Enhance Fungicidal Activity of Polyene Antibiotics. Nanomed. Nanotechnol. Biol. Med. 2016, 12, 2395–2404. [Google Scholar] [CrossRef]
- El Zowalaty, M.; Hussein-Al-Ali, S.; Husseiny, M.I.; Geilich, B.; Webster, T.; Hussein, M.Z. The Ability of Streptomycin-Loaded Chitosan-Coated Magnetic Nanocomposites to Possess Antimicrobial and Antituberculosis Activities. IJN 2015, 10, 3269. [Google Scholar] [CrossRef]
- Ivashchenko, O.; Lewandowski, M.; Peplińska, B.; Jarek, M.; Nowaczyk, G.; Wiesner, M.; Załęski, K.; Babutina, T.; Warowicka, A.; Jurga, S. Synthesis and Characterization of Magnetite/Silver/Antibiotic Nanocomposites for Targeted Antimicrobial Therapy. Mater. Sci. Eng. C 2015, 55, 343–359. [Google Scholar] [CrossRef]
- Ivashchenko, O.; Jurga-Stopa, J.; Coy, E.; Peplinska, B.; Pietralik, Z.; Jurga, S. Fourier Transform Infrared and Raman Spectroscopy Studies on Magnetite/Ag/Antibiotic Nanocomposites. Appl. Surf. Sci. 2016, 364, 400–409. [Google Scholar] [CrossRef]
- Istrate, C.M.; Holban, A.M.; Grumezescu, A.M.; Mogoanta, L.; Mogosanu, G.D.; Savopol, T.; Moisescu, M.; Iordache, M.; Vasile, B.S.; Kovacs, E. Iron oxide nanoparticles modulate the interaction of different antibiotics with cellular membranes. Rom. J. Morphol. Embryol. 2014, 55, 849–856. [Google Scholar] [PubMed]
- Allafchian, A.; Hosseini, S.S. Antibacterial Magnetic Nanoparticles for Therapeutics: A Review. IET Nanobiotechnol. 2019, 13, 786–799. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Yang, J.; Yan, Y.; Li, J.; Shen, M.; Zhang, G.; Mignani, S.; Shi, X. RGD-Functionalized Ultrasmall Iron Oxide Nanoparticles for Targeted T1-Weighted MR Imaging of Gliomas. Nanoscale 2015, 7, 14538–14546. [Google Scholar] [CrossRef] [PubMed]
- Nunn, A.V.W.; Barnard, M.L.; Bhakoo, K.; Murray, J.; Chilvers, E.J.; Bell, J.D. Characterisation of Secondary Metabolites Associated with Neutrophil Apoptosis. FEBS Lett. 1996, 392, 295–298. [Google Scholar] [CrossRef]
- Johnson, G.A.; Benveniste, H.; Black, R.D.; Hedlund, L.W.; Maronpot, R.R.; Smith, B.R. Histology by magnetic resonance microscopy. Magn Reson Q. 1993, 9, 1–30. [Google Scholar]
- Olsvik, O.; Popovic, T.; Skjerve, E.; Cudjoe, K.S.; Hornes, E.; Ugelstad, J.; Uhlén, M. Magnetic Separation Techniques in Diagnostic Microbiology. Clin. Microbiol. Rev. 1994, 7, 43–54. [Google Scholar] [CrossRef]
- Yeh, T.-C.; Zhang, W.; Ildstad, S.T.; Ho, C. Intracellular Labeling of T-Cells with Superparamagnetic Contrast Agents. Magn. Reson. Med. 1993, 30, 617–625. [Google Scholar] [CrossRef]
- Schoepf, U.; Marecos, E.M.; Melder, R.J.; Jain, R.K.; Weissleder, R. Intracellular Magnetic Labeling of Lymphocytes for In Vivo Trafficking Studies. BioTechniques 1998, 24, 642–651. [Google Scholar] [CrossRef]
- King, A.C.; Cuatrecasas, P. Adsorptive Pinocytosis of Epidermal Growth Factor: Studies of Its Relevance to Mitogenesis. In Receptor-Mediated Endocytosis; Springer: Dordrecht, The Netherlands, 1983; pp. 45–81. [Google Scholar] [CrossRef]
- Weissleder, R.; Cheng, H.-C.; Bogdanova, A.; Bogdanov, A. Magnetically Labeled Cells Can Be Detected by MR Imaging. J. Magn. Reson. Imaging 1997, 7, 258–263. [Google Scholar] [CrossRef]
- Qian, Z.M. Targeted Drug Delivery via the Transferrin Receptor-Mediated Endocytosis Pathway. Pharmacol. Rev. 2002, 54, 561–587. [Google Scholar] [CrossRef] [PubMed]
- Cima, L.G. Receptors: Models for Binding, Trafficking and Signaling. By Douglas A. Lauffenburger and Jennifer J. Linderman, Oxford University Press, 1993, $70.00. AICHE J. 1994, 40, 1089. [Google Scholar] [CrossRef]
- Shengfa, F.; Liao, J.M.F. Lectin-Functionalized Magnetic Iron Oxide Nanoparticles for Reproductive Improvement. JFIV Reprod. Med. Genet. 2014, 3, 17–19. [Google Scholar] [CrossRef]
- Sutovsky, P.; Kennedy, C.E. Biomarker-Based Nanotechnology for the Improvement of Reproductive Performance in Beef and Dairy Cattle. Ind. Biotechnol. 2013, 9, 24–30. [Google Scholar] [CrossRef]
- Odhiambo, J.F.; DeJarnette, J.M.; Geary, T.W.; Kennedy, C.E.; Suarez, S.S.; Sutovsky, M.; Sutovsky, P. Increased Conception Rates in Beef Cattle Inseminated with Nanopurified Bull Semen1. Biol. Reprod. 2014, 91. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Akakuru, O.U.; Zheng, J.; Wu, A. Applications of Iron Oxide-Based Magnetic Nanoparticles in the Diagnosis and Treatment of Bacterial Infections. Front. Bioeng. Biotechnol. 2019, 7. [Google Scholar] [CrossRef]
- Hjiri, M.; Aida, M.; Neri, G. NO2 Selective Sensor Based on α-Fe2O3 Nanoparticles Synthesized via Hydrothermal Technique. Sensors 2019, 19, 167. [Google Scholar] [CrossRef]
- He, L.; Mao, Y.; Zhang, L.; Wang, H.; Gao, B.; Wei, D. Functional expression of a novel α-amylase from Antarctic psychrotolerant fungus for baking industry and its magnetic immobilization. BMC Biotechnol. 2017, 17, 22. [Google Scholar] [CrossRef]
- Lobel, B.; Eyal, O.; Kariv, N.; Katzir, A. Temperature Controlled CO2 Laser Welding of Soft Tissues: Urinary Bladder Welding in Different Animal Models (Rats, Rabbits, and Cats). Lasers Surg. Med. 2000, 26, 4–12. [Google Scholar] [CrossRef]
- Anderson, S.A.; Glod, J.; Arbab, A.S.; Noel, M.; Ashari, P.; Fine, H.A.; Frank, J.A. Noninvasive MR Imaging of Magnetically Labeled Stem Cells to Directly Identify Neovasculature in a Glioma Model. Blood 2005, 105, 420–425. [Google Scholar] [CrossRef]
- Bull, E.; Madani, S.Y.; Sheth, R.; Seifalian, A.; Green, M.; Seifalian, A. Stem cell tracking using iron oxide nanoparticles. Int. J. Nanomed. 2014, 9, 1641–1653. [Google Scholar] [CrossRef]
- Pinaud, F.; King, D.; Moore, H.-P.; Weiss, S. Bioactivation and Cell Targeting of Semiconductor CdSe/ZnS Nanocrystals with Phytochelatin-Related Peptides. J. Am. Chem. Soc. 2004, 126, 6115–6123. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Liu, H.; Liu, J.; Haley, K.N.; Treadway, J.A.; Larson, J.P.; Ge, N.; Peale, F.; Bruchez, M.P. Immunofluorescent Labeling of Cancer Marker Her2 and Other Cellular Targets with Semiconductor Quantum Dots. Nat. Biotechnol. 2003, 21, 41–46. [Google Scholar] [CrossRef]
- Dumestre, F.; Chaudret, B.; Amiens, C.; Fromen, M.-C.; Casanove, M.-J.; Renaud, P.; Zurcher, P. Shape Control of Thermodynamically Stable Cobalt Nanorods through Organometallic Chemistry. Angew. Chem. Int. Ed. 2002, 41, 4286–4289. [Google Scholar] [CrossRef]
- Savva, M.; Duda, E.; Huang, L. A Genetically Modified Recombinant Tumor Necrosis Factor-α Conjugated to the Distal Terminals of Liposomal Surface Grafted Polyethyleneglycol Chains. Int. J. Pharm. 1999, 184, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Yuyama, Y.; Tsujimoto, M.; Fujimoto, Y.; Oku, N. Potential Usage of Thermosensitive Liposomes for Site-Specific Delivery of Cytokines. Cancer Lett. 2000, 155, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Bazile, D.; Prud’homme, C.; Bassoullet, M.; Marlard, M.; Spenlehauer, G.; Veillard, M. Stealth Me. PEG-PLA Nanoparticles Avoid Uptake by the Mononuclear Phagocytes System. J. Pharm. Sci. 1995, 84, 493–498. [Google Scholar] [CrossRef]
- Arias, J.L.; López-Viota, M.; Sáez-Fernández, E.; Ruiz, M.A.; Delgado, Á.V. Engineering of an Antitumor (Core/Shell) Magnetic Nanoformulation Based on the Chemotherapy Agent Ftorafur. Colloids Surf. A Physicochem. Eng. Asp. 2011, 384, 157–163. [Google Scholar] [CrossRef]
- Bronstein, L.M.; Shtykova, E.V.; Malyutin, A.; Dyke, J.C.; Gunn, E.; Gao, X.; Stein, B.; Konarev, P.V.; Dragnea, B.; Svergun, D.I. Hydrophilization of Magnetic Nanoparticles with Modified Alternating Copolymers. Part 1: The Influence of the Grafting. J. Phys. Chem. C 2010, 114, 21900–21907. [Google Scholar] [CrossRef]
- Kheiri Manjili, H.; Ma’mani, L.; Tavaddod, S.; Mashhadikhan, M.; Shafiee, A.; Naderi-Manesh, H. D, L-Sulforaphane Loaded Fe3O4@Gold Core Shell Nanoparticles: A Potential Sulforaphane Delivery System. PLoS ONE 2016, 11, e0151344. [Google Scholar] [CrossRef]
- Zhao, Z.; Cui, H.; Song, W.; Ru, X.; Zhou, W.; Yu, X. A Simple Magnetic Nanoparticles-Based Viral RNA Extraction Method for Efficient Detection of SARS-CoV-2. bioRxiv 2020. [Google Scholar] [CrossRef]
- Sur, U.K. Surface-Enhanced Raman Spectroscopy. Reson 2010, 15, 154–164. [Google Scholar] [CrossRef]
- Liang, Y.-Y.; Zhang, L.-M.; Jiang, W.; Li, W. Embedding Magnetic Nanoparticles into Polysaccharide-Based Hydrogels for Magnetically Assisted Bioseparation. ChemPhysChem 2007, 8, 2367–2372. [Google Scholar] [CrossRef] [PubMed]
- Nosrati, H.; Salehiabar, M.; Davaran, S.; Ramazani, A.; Manjili, H.K.; Danafar, H. New advances strategies for surface functionalization of iron oxide magnetic nanoparticles (IONPs). Res. Chem. Intermed 2017, 43, 7423–7442. [Google Scholar] [CrossRef]
- Dave, P.N.; Chopda L., V. Application of Iron Oxide Nanomaterials for the Removal of Heavy Metals. J. Nanotechnol. 2014. [Google Scholar] [CrossRef]
- Buonassisi, T.; Istratov, A.A.; Marcus, M.A.; Lai, B.; Cai, Z.; Heald, S.M.; Weber, E.R. Engineering Metal-Impurity Nanodefects for Low-Cost Solar Cells. Nat. Mater. 2005, 4, 676–679. [Google Scholar] [CrossRef]
- Seki, M. Bandgap-Engineered Iron Oxides for Solar Energy Harvesting. Iron Ores Iron Oxide Mater. 2018. [Google Scholar] [CrossRef]
- Rabiee, N.; Bagherzadeh, M.; Ghasemi, A.; Zare, H.; Ahmadi, S.; Fatahi, Y.; Dinarvand, R.; Rabiee, M.; Ramakrishna, S.; Shokouhimehr, M. Point-of-Use Rapid Detection of SARS-CoV-2: Nanotechnology-Enabled Solutions for the COVID-19 Pandemic. IJMS 2020, 21, 5126. [Google Scholar] [CrossRef]
- Rajasundari, K.; Ilamurugu, K. Nanotechnology and its applications in medical diagnosis. J. Basic. Appl. Chem. 2011, 1, 26–32. [Google Scholar]
- Sun, Y.; Xu, L.; Zhang, F.; Song, Z.; Hu, Y.; Ji, Y.; Shen, J.; Li, B.; Lu, H.; Yang, H. A Promising Magnetic SERS Immunosensor for Sensitive Detection of Avian Influenza Virus. Biosens. Bioelectron. 2017, 89, 906–912. [Google Scholar] [CrossRef]
- Deng, Z.-Y.; Chen, K.-L.; Wu, C.-H. Improving the SERS Signals of Biomolecules Using a Stacked Biochip Containing Fe2O3/Au Nanoparticles and a DC Magnetic Field. Sci. Rep. 2019, 9. [Google Scholar] [CrossRef] [PubMed]
- Mayer, A.C.; Ulrich, A.; Czerwinski, J.; Mooney, J.J. Metal-Oxide Particles in Combustion Engine Exhaust. SAE Tech. Paper 2010. [Google Scholar] [CrossRef]
- Siddiqui, M.H.; Al-Whaibi, M.H.; Sakran, A.M.; Ali, H.M.; Basalah, M.O.; Faisal, M.; Alatar, A.; Al-Amri, A.A. Calcium-Induced Amelioration of Boron Toxicity in Radish. J. Plant Growth Regul. 2013, 32, 61–71. [Google Scholar] [CrossRef]
- Aurich, K.; Schwalbe, M.; Clement, J.H.; Weitschies, W.; Buske, N. Polyaspartate Coated Magnetite Nanoparticles for Biomedical Applications. J. Magn. Magn. Mater. 2007, 311, 1–5. [Google Scholar] [CrossRef]
- Aslani, F.; Bagheri, S.; Muhd Julkapli, N.; Juraimi, A.S.; Hashemi, F.S.G.; Baghdadi, A. Effects of Engineered Nanomaterials on Plants Growth: An Overview. Sci. World J. 2014, 2014, 641759. [Google Scholar] [CrossRef]
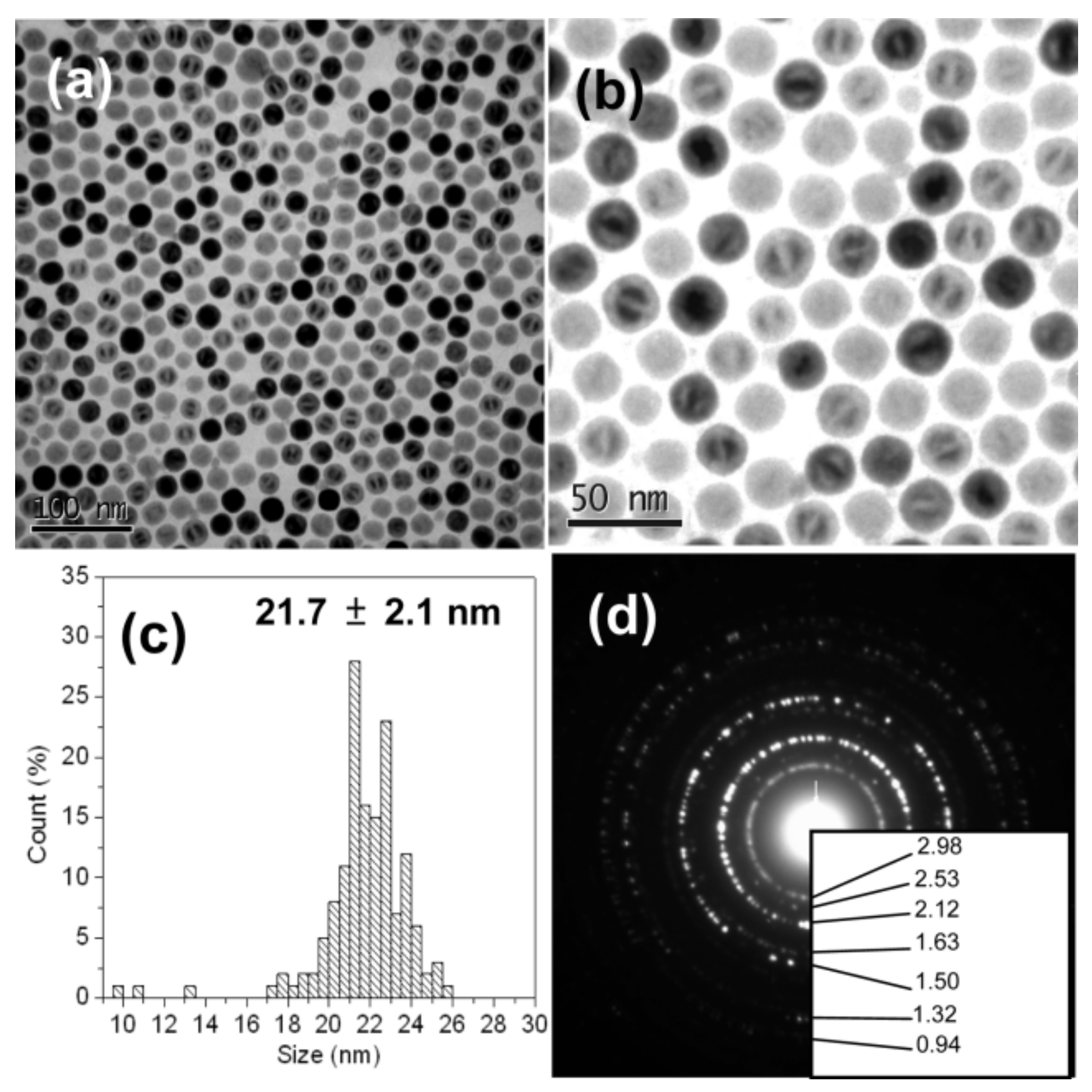





| Type of Synthesis | Pros | Cons | Reference |
|---|---|---|---|
| Microwave | Short reaction time, higher yields, excellent reproducibility, easy handling | Expensive, unsuitable for scale-up and reaction monitoring | [28,29] |
| Spray pyrolysis | Finely dispersed particles of predictable size, shape and variable composition | Aggregated particles, expensive | [30,31,32] |
| Laser pyrolysis | Small particle size, narrow particle size distribution, near absence of aggregation | Complicated, very expensive | [21,31] |
| Pulsed wire discharge method | Fast process, higher purity of NPs | Batch process, limited production, high vacuum systems, costly process, contaminations in product | [33,34] |
| Chemical vapor condensation | Suitable for preparing small quantities to demonstrate desired properties in the laboratory | Low production, difficult to control size and particle size distribution | [35] |
| Co-precipitation | Convenient method, simple and rapid preparative method, easy control of particle size and composition | Extensive agglomeration, poor morphology and particle size distribution | [36,37,38] |
| Thermal decomposition | Producing highly monodispersed particles with a narrow size distribution | High cost, long-time synthesis reaction, high temperature | [39,40,41] |
| Microemulsion | Monodispersed nanoparticles with various morphology can be produced | Not very efficient and difficult to scale up | [39,42] |
| Polyol | Uniform size particles can be prepared, easy to scaleup | Needs high temperature, long time | [22,31] |
| Sol–Gel | Low processing cost, energy efficiency, high production rate, and rapid productivity | Limited efficiency, high cost | [43,44,45] |
| Sonochemical | Simple, low cost, safe, environment friendly, absence of many reactants | Very small concentration of prepared NPs, particle agglomeration is very narrow | [33,46] |
| Biological synthesis of nanoparticles using plants and bacteria | Selectivity and precision for nanoparticle formation, cost effective, eco friendly | Limited knowledge, difficulty in controlling size and properties | [47,48] |
| Co-Precipitation Method | Polyol Method |
|---|---|
|
|
| Reagents/Precursors | Used Concentration | Synthesized Nanoparticles | Reference |
|---|---|---|---|
| Precursor A: Fe(III)(NO3)3·9H2O Precursor B: Fe(II)–naphthenate | 0.65 M 0.9 M | FeO | [52] |
| Goethite FeO(OH) Oleic acid | 3 mM 13.5 mM | FeO | [53] |
| Fe(stearate)2 i.e., ferric stearate oleic acid | 2.22 mM 4.44 mmol | FeO | [55] |
| Fe(III) acetylacetonate Oleic acid Oleylamine | 0.75 mM 5 mM 5 mM | core/shell FeO/Fe3O4 | [51] |
| Fe(NO3)3· 9H2O C6H8O7·H2O | 0.1M 0.05 to 0.2M | Fe2O3 (admixture of α and ɤ) | [57] |
| Precursor solution of Fe (III) NaOH | 1 M 6 M | α-Fe2O3 | [58] |
| Poly vinyl alcohol Fe(NO3)3 EDTA | 0.25 M 0.1 M 0.1 M | α-Fe2O3 | [59] |
| Fe2(SO4)3 and FeSO4 | 2:1 Fe(III) to Fe(II) | ||
| Total Fe of 8.6 × 10−3M | ɤ-Fe2O3 | [62] | |
| NH4OH | 25% aqueous solution | ||
| FeCl3·6H2O FeCl2·4H2O KCl KOH | 1.35 g 0.50 g 3.9 g 1.22 g | IONPs | [64] |
| FeCl2·4H2O and FeCl3·6H2O | Fe2+/Fe3+=1/2 | ||
| dissolved in 2 M HCl | ɤ-Fe2O3 | [66] | |
| NaOH | 2 M | ||
| FeCl2·4H2O and FeCl3·6H2O NH4OH HNO3 HCl | Molar ratio 2:1 28% 65% 37% | ɤ-Fe2O3 | [67] |
| FeCl3·6H2O FeCl2·4H2O NH4OH | 0.1 M 0.1 M 10% solution | ɤ-Fe2O3 | [68] |
| FeCl3∙6H2O FeCl2∙4H2O HCl N(CH3)4OH. | 1 M 2 M 2 M 25 % aqueous | Fe3O4 | [78] |
| FeCl2∙4H2O FeCl3∙6H2O NaOH | 0.01 M 0.02 M 0.08 M | Fe3O4 | [79] |
| Fe(acac)3 C6H8O6 Ultrapure water | 30 mM/15 mM/50 mM 0.025 M/0.005 M/0.05 M 12 M/1.2 M | Fe3O4 | [22,25,26,27] |
| Discipline | Application | Reference |
|---|---|---|
| Environmental Remediation |
| [95,100,101,102,103] |
| Biomedical | Magnetic hyperthermia, controlled drug release, magnetic separation, MRI contrast agent, controlled drug release, cell separation and handling of cells, cell labeling, tissue repair, antiviral. | [104,105,106,107] |
| Defense and Aerospace | Fuel additives, energy devices, nanocomposites, sensors, nanocoatings and electronics. | [108,109,110] |
| Construction |
| [105,111] |
| Electronics | Nanoscale memory, spintronics, printed electronics, nanowires and NEMS (nano electro mechanical systems) | [112,113] |
| Healthcare | Nanoscale biosensors and imaging, antimicrobial activities; nanophotothermolysis with pulsed lasers, antiviral, preventing skin aging; implants; nanocarrier for vaccination | [114,115] |
| Automotive Textiles | Additive in catalysts and lubricants, sensors, nanofibers, coatings, composite fillers, fuel cells and smart materials | [116] |
| Agriculture and Food | Nanopesticides, nanofungicides, nanofertilizers, nanosensors, nanofood, encapsulation, gene transfer (crop improvements), food packing | [117,118,119,120] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ajinkya, N.; Yu, X.; Kaithal, P.; Luo, H.; Somani, P.; Ramakrishna, S. Magnetic Iron Oxide Nanoparticle (IONP) Synthesis to Applications: Present and Future. Materials 2020, 13, 4644. https://doi.org/10.3390/ma13204644
Ajinkya N, Yu X, Kaithal P, Luo H, Somani P, Ramakrishna S. Magnetic Iron Oxide Nanoparticle (IONP) Synthesis to Applications: Present and Future. Materials. 2020; 13(20):4644. https://doi.org/10.3390/ma13204644
Chicago/Turabian StyleAjinkya, Nene, Xuefeng Yu, Poonam Kaithal, Hongrong Luo, Prakash Somani, and Seeram Ramakrishna. 2020. "Magnetic Iron Oxide Nanoparticle (IONP) Synthesis to Applications: Present and Future" Materials 13, no. 20: 4644. https://doi.org/10.3390/ma13204644
APA StyleAjinkya, N., Yu, X., Kaithal, P., Luo, H., Somani, P., & Ramakrishna, S. (2020). Magnetic Iron Oxide Nanoparticle (IONP) Synthesis to Applications: Present and Future. Materials, 13(20), 4644. https://doi.org/10.3390/ma13204644








