Secondary Crystalline Phases Influence on Optical Properties in Off-Stoichiometric Cu2S–ZnS–SnS2 Thin Films
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Chemical Composition
3.2. Structural Properties
3.2.1. GIXRD Characterization
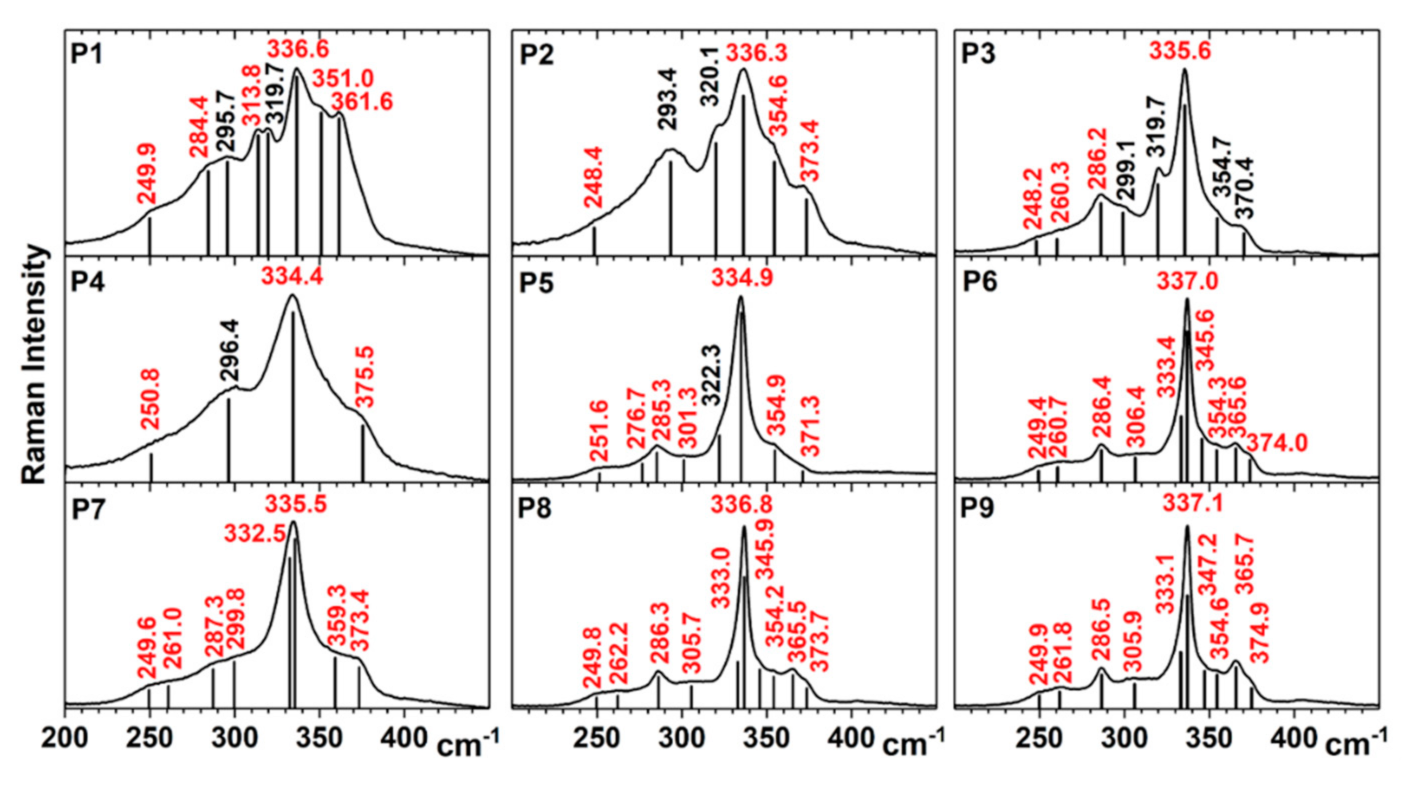
3.2.2. Raman Spectroscopy
- P1: CZTS (I-42m) (284.4 cm−1, 336.6 cm−1, 361.6 cm−1), CZTS (I-4) (249.9 cm−1, 284.4 cm−1, 313.8 cm−1, 336.6 cm−1, 351.0 cm−1), Cu2SnS3 (I-42m) (295.7 cm−1, 336.6 cm−1, 351.0 cm−1), orthorhombic Cu3SnS4 (319.7 cm−1) [20], hexagonal SnS2 (314 cm−1, determined by Raman scattering on our SnS2 target, data not shown);
- P2: CZTS (I-42m) (336.3 cm−1), CZTS (I-4) (248.4 cm−1, 336.3 cm−1, 354.6 cm−1, 373.4 cm−1), Cu2SnS3 (I-42m) (293.4 cm−1, 336.3 cm−1, 354.6 cm−1), orthorhombic Cu3SnS4 (320.1 cm−1);
- P3: CZTS (I-42m) (286.2, 336.3 cm−1), CZTS (I-4) (248.2, 260.3, 286.2, 335.6, 354.7, 370.4 cm−1), Cu2SnS3 (I-42m) (299.1, 335.6, 354.7 cm−1), orthorhombic Cu3SnS4: (319.7 cm−1) while for Cu–S the peaks are out of the measured range;
- P4: CZTS (I-4) (250.8, 334.4, 375.5 cm−1), Cu2SnS3 (I-42m) (296.4, 334.4 cm−1). The most prominent peak in the Raman spectra of P4 is red shifted to 334.4 cm−1, and a possible cause could be the smaller size of Cu–Zn–Sn–S crystallites [21] (as was shown by GIXRD measurements);
- P5: CZTS (I-4) (251.6, 276.7, 285.3, 301.3, 334.9, 354.9, 371.3 cm−1), orthorhombic Cu3SnS4: (322.3 cm−1);
- P6: CZTS (I-4) (249.4, 260.7, 286.4, 306.4, 333.4, 337.0, 345.6, 354.3, 365.6, 374.0 cm−1), Cu–S out of range;
- P7: CZTS (I-4) (249.6, 261.0, 287.3, 299.8, 332.5, 335.5, 359.3, 373.4 cm−1);
- P8: CZTS (I-4) (249.8, 262.2, 286.3, 305.7, 333.0, 336.8, 345.9, 354.2, 365.5, 373.7 cm−1); Cu-S out of range;
- P9: CZTS (I-4) (249.9, 261.8, 286.5, 305.9, 333.1, 337.1, 347.2, 354.6, 365.7, 374.9 cm−1).
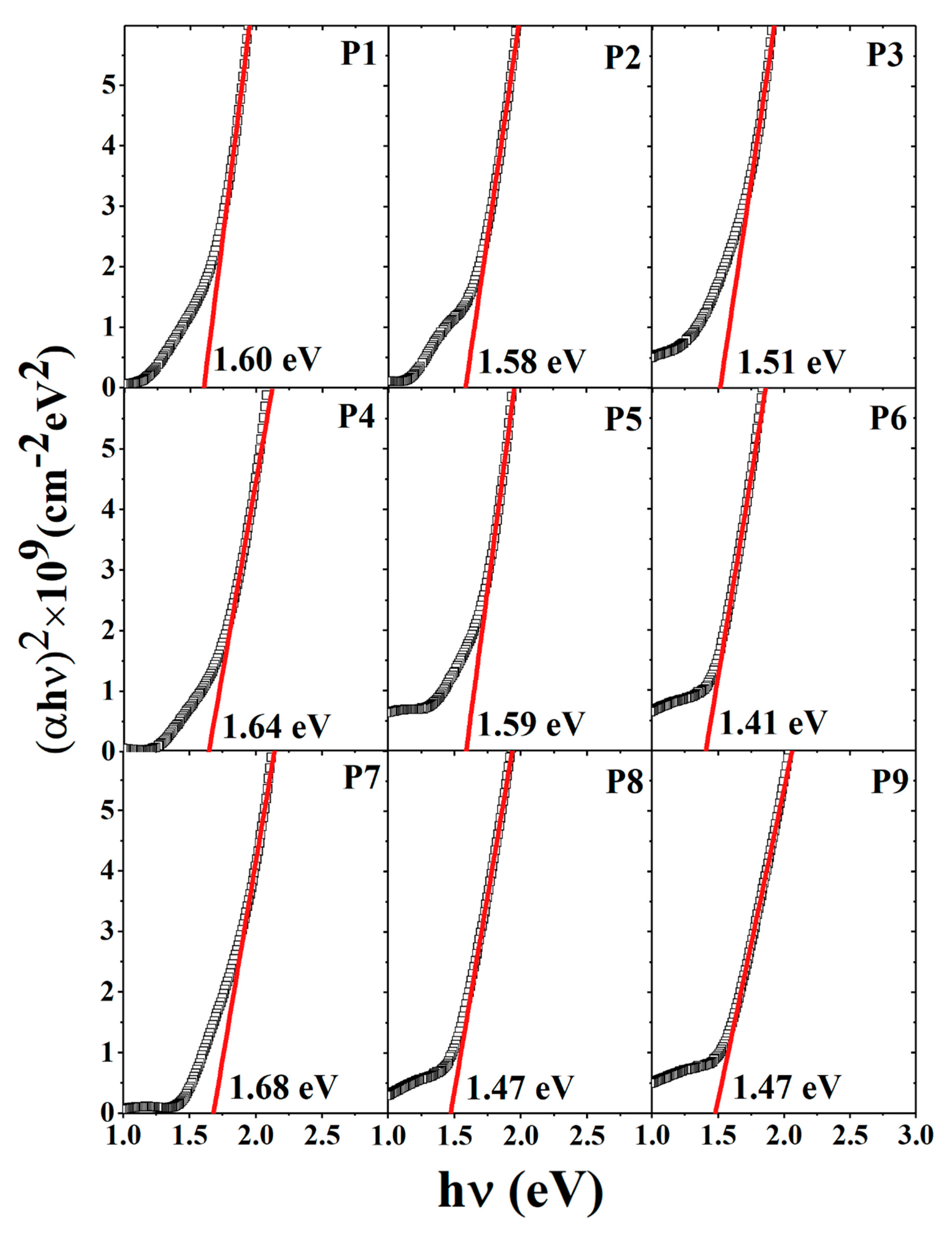
3.3. Optical Properties
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Service, R.F. SOLAR ENERGY: Is It Time to Shoot for the Sun? Science 2005, 309, 548–551. [Google Scholar] [CrossRef] [PubMed]
- Brabec, C.J. Organic photovoltaics: Technology and market. Sol. Energy Mater. Sol. Cells 2004, 83, 273–292. [Google Scholar] [CrossRef]
- Wadia, C.; Alivisatos, A.P.; Kammen, D.M. Materials Availability Expands the Opportunity for Large-Scale Photovoltaics Deployment. Environ. Sci. Technol. 2009, 43, 2072–2077. [Google Scholar] [CrossRef] [PubMed]
- Nelson, J. The Physics of Solar Cells; Imperial College Press: London, UK; World Scientific Publisher Co.: Singapore, 2003. [Google Scholar]
- Ito, K.; Nakazawa, T. Electrical and Optical Properties of Stannite-Type Quaternary Semiconductor Thin Films. Jpn. J. Appl. Phys. 1988, 27, 2094–2097. [Google Scholar] [CrossRef]
- Siebentritt, S.; Schorr, S. Kesterites-a challenging material for solar cells: Kesterites-a challenging material for solar cells. Prog. Photovolt. Res. Appl. 2012, 20, 512–519. [Google Scholar] [CrossRef]
- Schorr, S.; Hoebler, H.-J.; Tovar, M. A neutron diffraction study of the stannite-kesterite solid solution series. Eur. J. Miner. 2007, 19, 65–73. [Google Scholar] [CrossRef]
- Gurieva, G.; Rios, L.E.V.; Franz, A.; Whitfield, P.S.; Schorr, S. Intrinsic point defects in off-stoichiometric Cu2ZnSnSe4: A neutron diffraction study. J. Appl. Phys. 2018, 123, 161519. [Google Scholar] [CrossRef]
- Yoo, H.; Kim, J. Growth of Cu2ZnSnS4 thin films using sulfurization of stacked metallic films. Thin Solid Films 2010, 518, 6567–6572. [Google Scholar] [CrossRef]
- Kermadi, S.; Sali, S.; Ameur, F.A.; Zougar, L.; Boumaour, M.; Toumiat, A.; Melnik, N.N.; Hewak, D.; Duta, A. Effect of copper content and sulfurization process on optical, structural and electrical properties of ultrasonic spray pyrolysed Cu2ZnSnS4 thin films. Mater. Chem. Phys. 2016, 169, 96–104. [Google Scholar] [CrossRef]
- Liu, R.; Tan, M.; Zhang, X.; Chen, J.; Song, S.; Zhang, W. Impact of sol–gel precursor treatment with preheating temperature on properties of Cu2ZnSnS4 thin film and its photovoltaic solar cell. J. Alloys Compd. 2016, 655, 124–129. [Google Scholar] [CrossRef]
- Safdar, A.; Islam, M.; Akram, M.A.; Mujahid, M.; Khalid, Y.; Shah, S.I. Reaction Time and Film Thickness Effects on Phase Formation and Optical Properties of Solution Processed Cu2ZnSnS4 Thin Films. J. Mater. Eng. Perform. 2016, 25, 457–465. [Google Scholar] [CrossRef]
- Kaushik, D.K.; Rao, T.N.; Subrahmanyam, A. Studies on the disorder in DC magnetron sputtered Cu2ZnSnS4 (CZTS) thin films grown in sulfide plasma. Surf. Coat. Technol. 2017, 314, 85–91. [Google Scholar] [CrossRef]
- Katagiri, H.; Jimbo, K.; Tahara, M.; Araki, H.; Oishi, K. The Influence of the Composition Ratio on CZTS-based Thin Film Solar Cells. MRS Proc. 2009, 1165, 1165-M04-01. [Google Scholar] [CrossRef]
- Fairbrother, A.; Dimitrievska, M.; Sánchez, Y.; Izquierdo-Roca, V.; Pérez-Rodríguez, A.; Saucedo, E. Compositional paradigms in multinary compound systems for photovoltaic applications: A case study of kesterites. J. Mater. Chem. A 2015, 3, 9451–9455. [Google Scholar] [CrossRef]
- Davydova, A.; Rudisch, K.; Scragg, J.J.S. The Single Phase Region in Cu2ZnSnS4 Thin Films from Theory and Combinatorial Experiments. Chem. Mater. 2018, 30, 4624–4638. [Google Scholar] [CrossRef]
- Hemme, E.G.; Fairbrother, A.; Calvo-Barrio, L.; Saucedo, E.; Martil, I. Compositional Dependence of Chemical and Electrical Properties in Cu2ZnSnS4 Thin Films. IEEE J. Photovoltaics 2016, 6, 990–996. [Google Scholar] [CrossRef]
- Valakh, M.Y.; Kolomys, O.F.; Ponomaryov, S.S.; Yukhymchuk, V.O.; Babichuk, I.S.; Izquierdo-Roca, V.; Saucedo, E.; Perez-Rodriguez, A.; Morante, J.R.; Schorr, S.; et al. Raman scattering and disorder effect in Cu2ZnSnS4. Phys. Status Solidi RRL 2013, 7, 258–261. [Google Scholar] [CrossRef]
- Fontané, X.; Izquierdo-Roca, V.; Saucedo, E.; Schorr, S.; Yukhymchuk, V.O.; Valakh, M.Y.; Pérez-Rodríguez, A.; Morante, J.R. Vibrational properties of stannite and kesterite type compounds: Raman scattering analysis of Cu2(Fe,Zn)SnS4. J. Alloys Compd. 2012, 539, 190–194. [Google Scholar] [CrossRef]
- Fernandes, P.A.; Salomé, P.M.P.; Da Cunha, A.F. A study of ternary Cu2SnS3 and Cu3SnS4 thin films prepared by sulfurizing stacked metal precursors. J. Phys. D Appl. Phys. 2010, 43, 215403. [Google Scholar] [CrossRef]
- Camara, S.M.; Wang, L.; Zhang, X. Easy hydrothermal preparation of Cu2ZnSnS4 (CZTS) nanoparticles for solar cell application. Nanotechnology 2013, 24, 495401. [Google Scholar] [CrossRef]
- Himmrich, M.; Haeuseler, H. Far infrared studies on stannite and wurtzstannite type compounds. Spectrochim. Acta Part A Mol. Spectrosc. 1991, 47, 933–942. [Google Scholar] [CrossRef]
- Dimitrievska, M.; Fairbrother, A.; Fontané, X.; Jawhari, T.; Izquierdo-Roca, V.; Saucedo, E.; Pérez-Rodríguez, A. Multiwavelength excitation Raman scattering study of polycrystalline kesterite Cu2ZnSnS4 thin films. Appl. Phys. Lett. 2014, 104, 021901. [Google Scholar] [CrossRef]
- Fairbrother, A.; Fontané, X.; Izquierdo-Roca, V.; Espíndola-Rodríguez, M.; López-Marino, S.; Placidi, M.; Calvo-Barrio, L.; Pérez-Rodríguez, A.; Saucedo, E. On the formation mechanisms of Zn-rich Cu2ZnSnS4 films prepared by sulfurization of metallic stacks. Sol. Energy Mater. Sol. Cells 2013, 112, 97–105. [Google Scholar] [CrossRef]
- Liu, Q.; Zhao, Z.; Lin, Y.; Guo, P.; Li, S.; Pan, D.; Ji, X. Alloyed (ZnS) x (Cu2SnS3) 1−xand (CuInS2) x (Cu2SnS3)1−xnanocrystals with arbitrary composition and broad tunable band gaps. Chem. Commun. 2011, 47, 964–966. [Google Scholar] [CrossRef] [PubMed]
- Long, B.; Cheng, S.; Lai, Y.; Zhou, H.; Yu, J.; Zheng, Q. Effects of sulfurization temperature on phases and opto-electrical properties of Cu2ZnSnS4 films prepared by sol–gel deposition. Thin Solid Films 2014, 573, 117–121. [Google Scholar] [CrossRef]
- Fernandes, P.A.; Salomé, P.M.P.; Da Cunha, A.F. Study of polycrystalline Cu2ZnSnS4 films by Raman scattering. J. Alloys Compd. 2011, 509, 7600–7606. [Google Scholar] [CrossRef]
- Serrano, J.; Cantarero, A.; Cardona, M.; Garro, N.; Lauck, R.; Tallman, R.E.; Ritter, T.M.; Weinstein, B.A. Raman scattering inβ-ZnS. Phys. Rev. B 2004, 69, 014301. [Google Scholar] [CrossRef]
- Seol, J.-S.; Lee, S.-Y.; Lee, J.-C.; Nam, H.-D.; Kim, K.-H. Electrical and optical properties of CuZnSnS thin films prepared by rf magnetron sputtering process. Sol. Energy Mater. Sol. Cells 2003, 75, 155–162. [Google Scholar] [CrossRef]
- Liu, F.; Zhang, K.; Lai, Y.; Li, J.; Zhang, Z.; Liu, Y. Growth and Characterization of Cu2ZnSnS4 Thin Films by DC Reactive Magnetron Sputtering for Photovoltaic Applications. Electrochem. Solid-State Lett. 2010, 13, H379. [Google Scholar] [CrossRef]
- Jaffe, J.E.; Zunger, A. Theory of the band-gap anomaly inABC2chalcopyrite semiconductors. Phys. Rev. B 1984, 29, 1882–1906. [Google Scholar] [CrossRef]
- Sun, L.; He, J.; Kong, H.; Yue, F.; Yang, P.; Chu, J. Structure, composition and optical properties of Cu2ZnSnS4 thin films deposited by Pulsed Laser Deposition method. Sol. Energy Mater. Sol. Cells 2011, 95, 2907–2913. [Google Scholar] [CrossRef]
- Inamdar, A.I.; Lee, S.; Jeon, K.-Y.; Lee, C.H.; Pawar, S.M.; Kalubarme, R.S.; Park, C.J.; Im, H.; Jung, W.; Kim, H. Optimized fabrication of sputter deposited Cu2ZnSnS4 (CZTS) thin films. Sol. Energy 2013, 91, 196–203. [Google Scholar] [CrossRef]
- Tanaka, K.; Shinji, T.; Uchiki, H. Photoluminescence from Cu2ZnSnS4 thin films with different compositions fabricated by a sputtering-sulfurization method. Sol. Energy Mater. Sol. Cells 2014, 126, 143–148. [Google Scholar] [CrossRef]
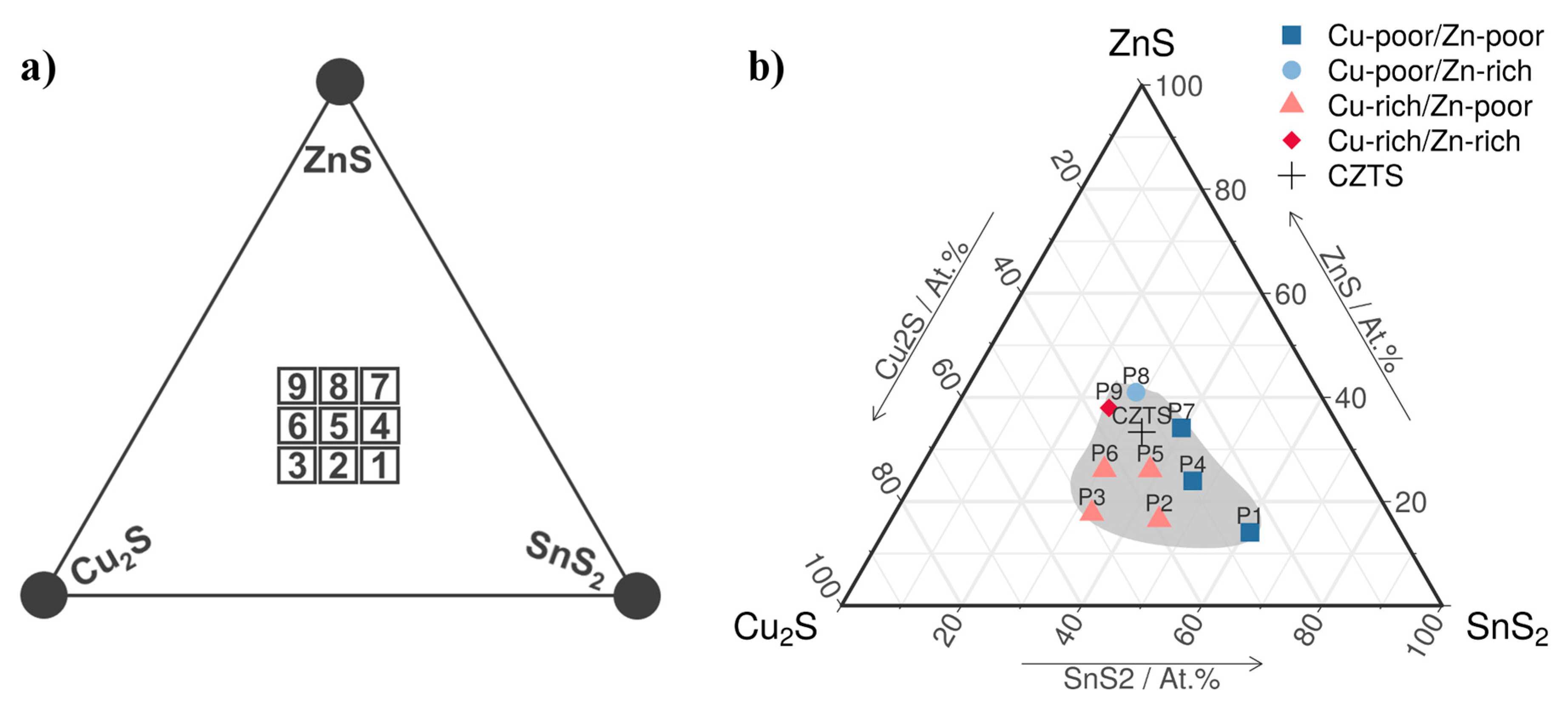
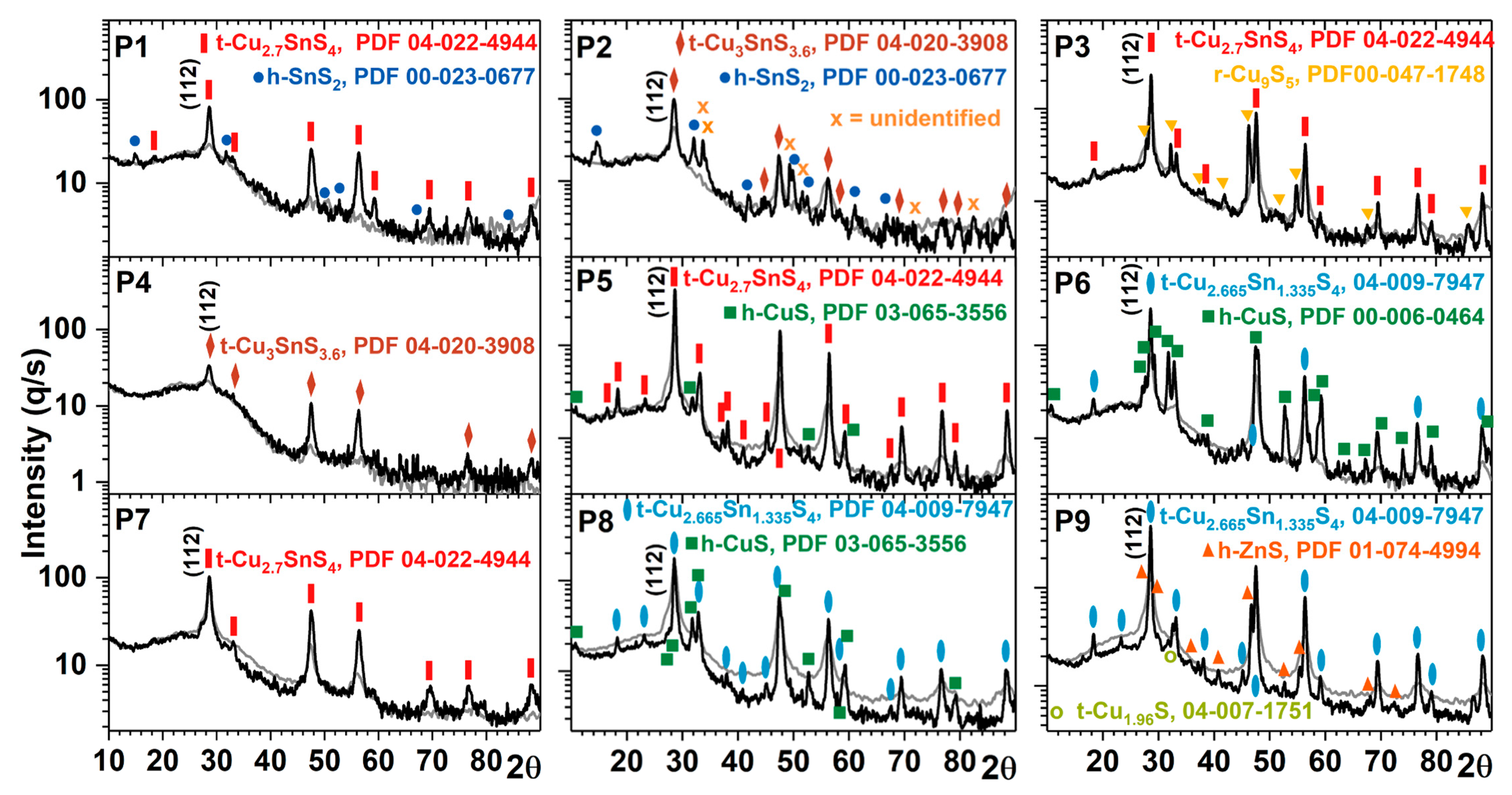
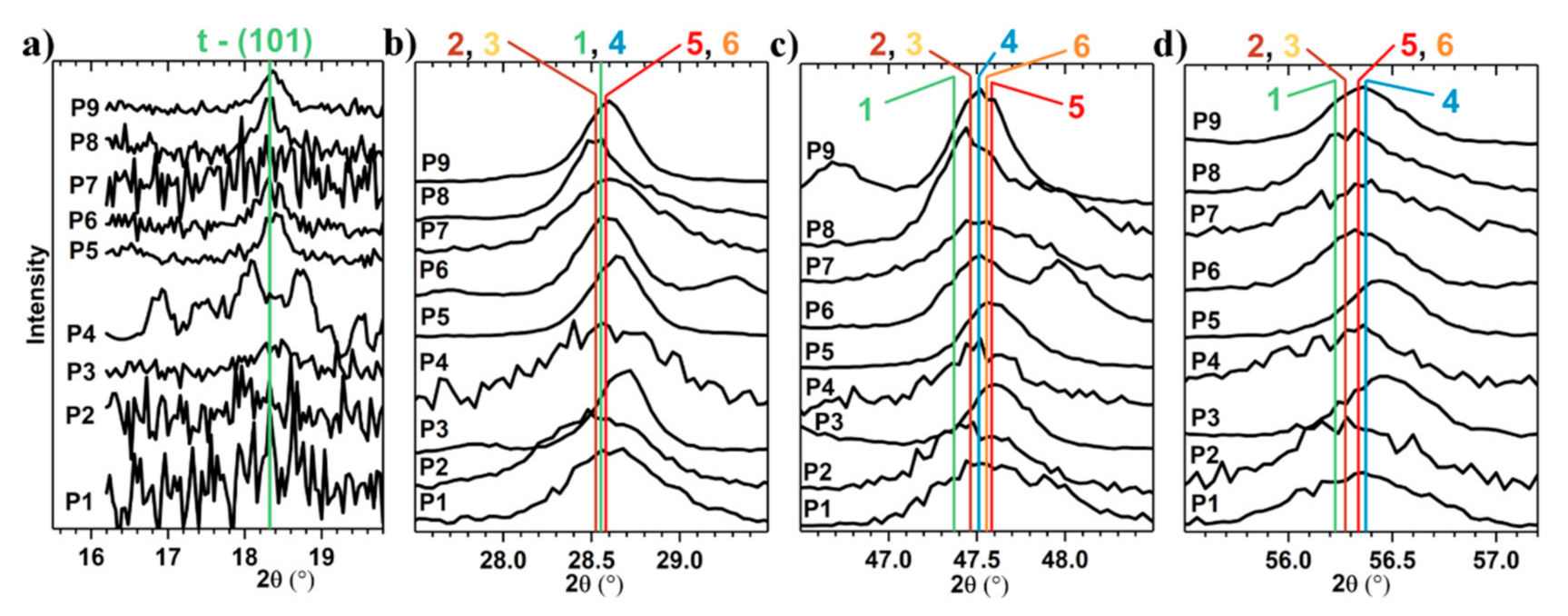
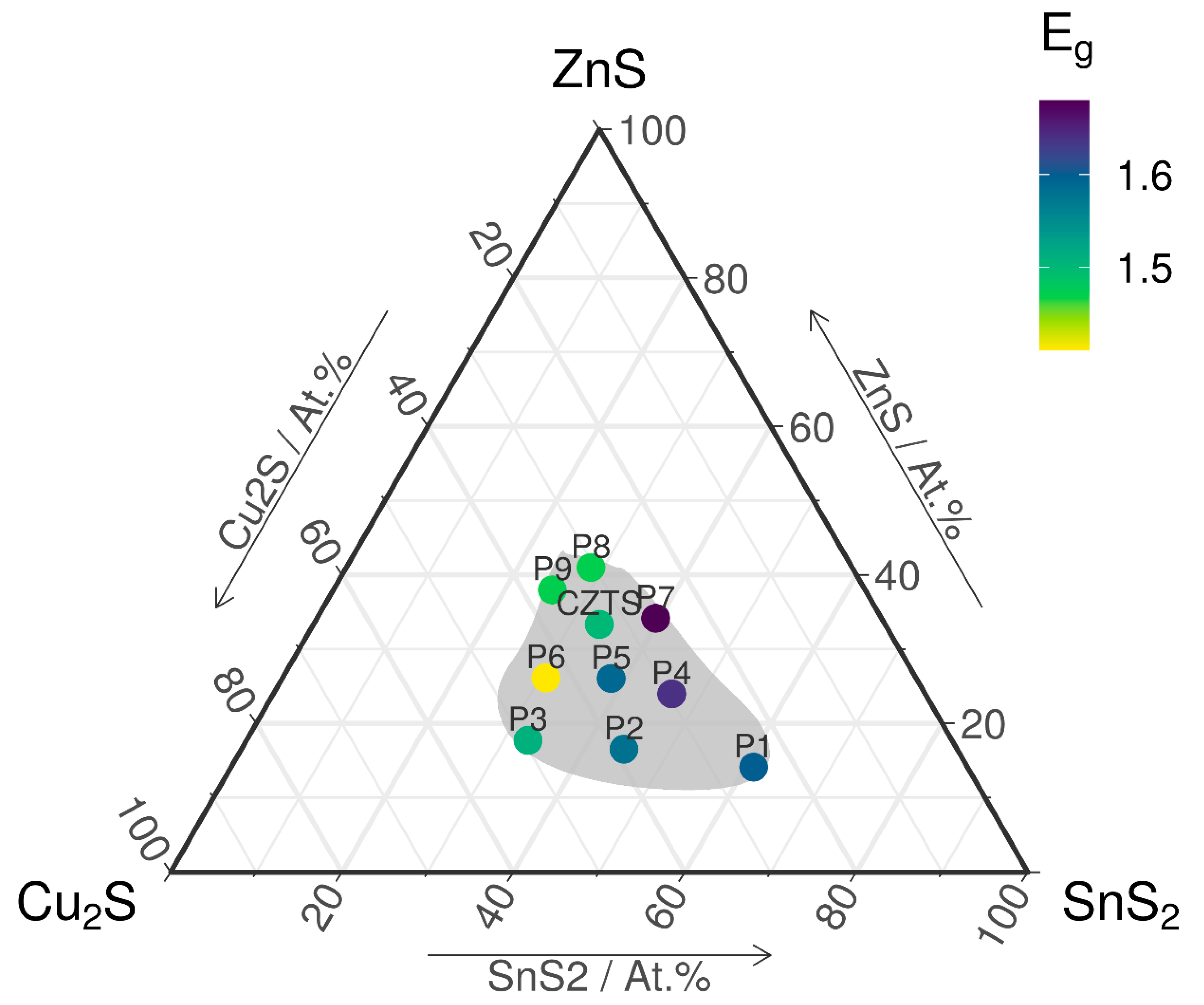
| P1 | P2 | P3 | P4 | P5 | P6 | P7 | P8 | P9 | CZTS | |
|---|---|---|---|---|---|---|---|---|---|---|
| pCu (at. %) | 20 −20 | 30 20 | 37 48 | 23 −8 | 27 8 | 30 20 | 22 −12 | 24 −4 | 29 16 | 25 |
| pZn (at. %) | 6 −52 | 7 −44 | 7 −44 | 10 −20 | 10 −20 | 9 −28 | 14 12 | 16 28 | 15 20 | 12.5 |
| pSn (at. %) | 24 92 | 17 36 | 12 −4 | 18 44 | 15 20 | 11 −14 | 16 28 | 12 −4 | 10 −20 | 12.5 |
| pS (at. %) | 50 0 | 46 −8 | 44 −12 | 49 −2 | 48 −4 | 50 0 | 48 −4 | 48 −4 | 46 −8 | 50 |
| −20 | −18 | −12 | −15 | −11 | 9 | −17 | −6 | −6 | 0 |
| pi (%) | pa (%) | r | pi (%) | pa (%) | r | pi (%) | pa (%) | r | |||
|---|---|---|---|---|---|---|---|---|---|---|---|
| P1 | 17 | 72 | 4.24 | P2 | 33 | 79 | 2.40 | P3 | 73 | 83 | 1.14 |
| P4 | 32 | 55 | 1.72 | P5 | 64 | 95 | 1.48 | P6 | 52 | 95 | 1.83 |
| P7 | 39 | 63 | 1.62 | P8 | 82 | 82 | 1.00 | P9 | 78 | 91 | 1.17 |
| Cc (%) | A | d (nm) | Crystalline Phases from GIXRD | |
|---|---|---|---|---|
| P1 | Cu: 20 | 420 | 17 | 65%: Cu2.7SnS4, tetragonal, I-42m (121), PDF 04-022-4944 7%: SnS2: hexagonal, P-3m1 (164), PDF 00-023-0677 28%: amorphous phase |
| Zn: 6 | ||||
| Sn: 24 | ||||
| S: 50 | ||||
| P2 | Cu: 30 | 800 | 11 | 67%: Cu3SnS3.6, PDF 04-020-3908, tetragonal, I-42m (121) 12%: SnS2: PDF 00-023-0677, hexagonal, P-3m1 (164) 21%: amorphous phase |
| Zn: 7 | ||||
| Sn: 17 | ||||
| S: 46 | ||||
| P3 | Cu: 37 | 1070 | 39 | 66%: Cu2.7SnS4, PDF 04-022-4944, tetragonal, I-42m (121) 17%: Cu9S5, PDF 00-047-1748, rhombohedral, R-3m (166) 17%: amorphous phase |
| Zn: 7 | ||||
| Sn: 12 | ||||
| S: 44 | ||||
| P4 | Cu: 23 | 250 | 8 | 55%: Cu3SnS3.6, PDF 04-020-3908, tetragonal, I-42m (121) 45%: amorphous phase |
| Zn: 10 | ||||
| Sn: 18 | ||||
| S: 49 | ||||
| P5 | Cu: 27 | 1950 | 37 | 93%: Cu2.7SnS4, PDF 04-022-4944, tetragonal, I-42m (121) 2% CuS: PDF 03-065-3556, hexagonal, P63/mmc (194) 5%: amorphous phase |
| Zn: 10 | ||||
| Sn: 15 | ||||
| S: 48 | ||||
| P6 | Cu: 30 | 1190 | 35 | 74%: Cu2.665Sn1.335S4, PDF 04-009-7947, tetragonal, I-42m (121) 21%: CuS, PDF 00-006-0464, hexagonal, P63/mmc (194) 5%: amorphous phase |
| Zn: 9 | ||||
| Sn: 11 | ||||
| S: 50 | ||||
| P7 | Cu: 22 | 780 | 12 | 63%: Cu2.7SnS4, PDF 04-022-4944, tetragonal, I-42m (121) 37%: amorphous phase |
| Zn: 14 | ||||
| Sn: 16 | ||||
| S: 48 | ||||
| P8 | Cu: 24 | 630 | 52 | 45%: Cu2.665Sn1.335S4, PDF 04-009-7947, tetragonal, I-42m (121) 37%: CuS, PDF 03-065-3556, hexagonal, P63/mmc (194) 18%: amorphous phase |
| Zn: 16 | ||||
| Sn: 12 | ||||
| S: 48 | ||||
| P9 | Cu: 29 | 2032 | 40 | 69%: Cu2.665Sn1.335S4, PDF 04-009-7947, tetragonal, I-42m (121) 19%: ZnS, PDF 01-074-4994, hexagonal, P-3m1 (156) 3%: Cu1.96S, PDF 04-007-1751, tetragonal, P43212 (96) 9%: amorphous phase |
| Zn: 15 | ||||
| Sn: 10 | ||||
| S: 46 |
| Compound | Structure | XRD, Cu Kα | Raman | |||
|---|---|---|---|---|---|---|
| 2θ (°), (hkl) | λexcitation (nm) | Peaks (cm−1) | ||||
| Cu2ZnSnS4 | Tetragonal, I-42m | 28.484, (112) 47.412, (204) 56.143, (312) | 514.5 [22] | 285 336 362 | ||
| Tetragonal, I-4 | 28.473, (112) 47.350, (204)/(220) 56.198, (116)/(132) | 632.8 [23,24] | 262.7 287.1 302.1 | 315.9 331.9 337.5 | 366.6 374.4 | |
| 514.5 [7,19] | 252 272 287 | 331 337 | 347 353 | |||
| Cu0.5Zn0.25Sn0.5S | Cubic, F-43m [25] | 28.525, (111) 47.446, (220) 56.296, (311) | - | - | ||
| Cu2.7SnS4 | Tetragonal, I-42m | 28.583, (112) 47.585, (204) 56.347, (312) | - | - | ||
| Cu2SnS3 | Tetragonal, I-42m | 28.566, (112) 47.507, (204)/(220) 56.337, (116)/(312) | 488 [26] | 297 336–337 351 | ||
| Cubic, F-43m | 28.470, (111) 47.350, (220) 56.180, (311) | 488 [26,27] | 267 303 355 | |||
| ZnS [28] | Cubic, F-43m | 28.582, (111) 47.555, (220) 56.337, (311) | 514.5 | 275 350 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sava, F.; Diagne, O.; Galca, A.-C.; Simandan, I.-D.; Matei, E.; Burdusel, M.; Becherescu, N.; Becherescu, V.; Mihai, C.; Velea, A. Secondary Crystalline Phases Influence on Optical Properties in Off-Stoichiometric Cu2S–ZnS–SnS2 Thin Films. Materials 2020, 13, 4624. https://doi.org/10.3390/ma13204624
Sava F, Diagne O, Galca A-C, Simandan I-D, Matei E, Burdusel M, Becherescu N, Becherescu V, Mihai C, Velea A. Secondary Crystalline Phases Influence on Optical Properties in Off-Stoichiometric Cu2S–ZnS–SnS2 Thin Films. Materials. 2020; 13(20):4624. https://doi.org/10.3390/ma13204624
Chicago/Turabian StyleSava, Florinel, Ousmane Diagne, Aurelian-Catalin Galca, Iosif-Daniel Simandan, Elena Matei, Mihail Burdusel, Nicu Becherescu, Virginia Becherescu, Claudia Mihai, and Alin Velea. 2020. "Secondary Crystalline Phases Influence on Optical Properties in Off-Stoichiometric Cu2S–ZnS–SnS2 Thin Films" Materials 13, no. 20: 4624. https://doi.org/10.3390/ma13204624
APA StyleSava, F., Diagne, O., Galca, A.-C., Simandan, I.-D., Matei, E., Burdusel, M., Becherescu, N., Becherescu, V., Mihai, C., & Velea, A. (2020). Secondary Crystalline Phases Influence on Optical Properties in Off-Stoichiometric Cu2S–ZnS–SnS2 Thin Films. Materials, 13(20), 4624. https://doi.org/10.3390/ma13204624







