Features of Structure and Properties of pHEMA-gr-PVP Block Copolymers, Obtained in the Presence of Fe2+
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis Technique of pHEMA-gr-PVP Copolymers
2.3. Measurements and Characterization
2.3.1. Standard Methods of Instrumental Research
2.3.2. PVP Grafting
2.3.3. The Molecular Weight between Cross-Links in the Polymer Network
2.3.4. Properties of Copolymers
3. Results and Discussion
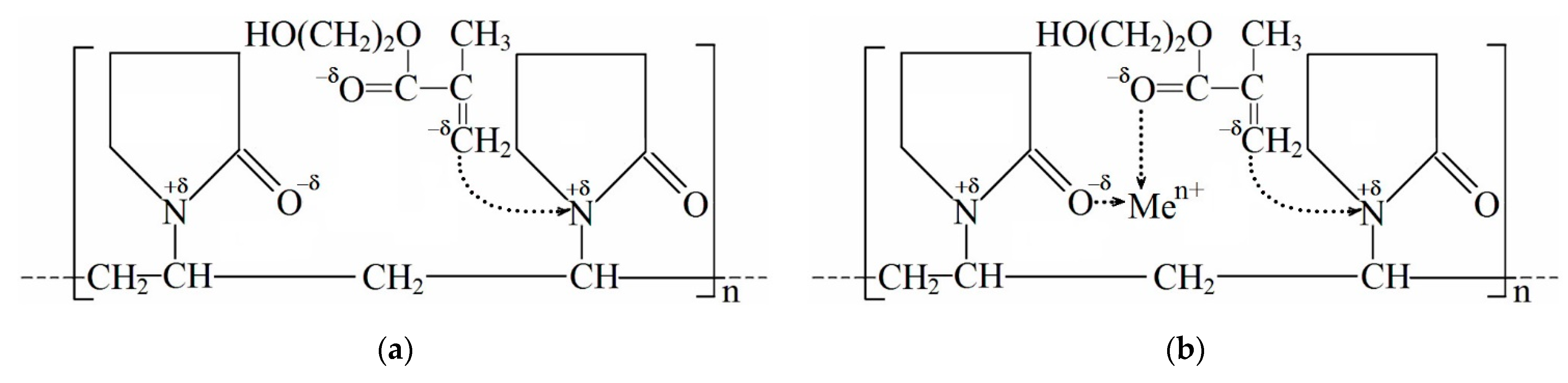
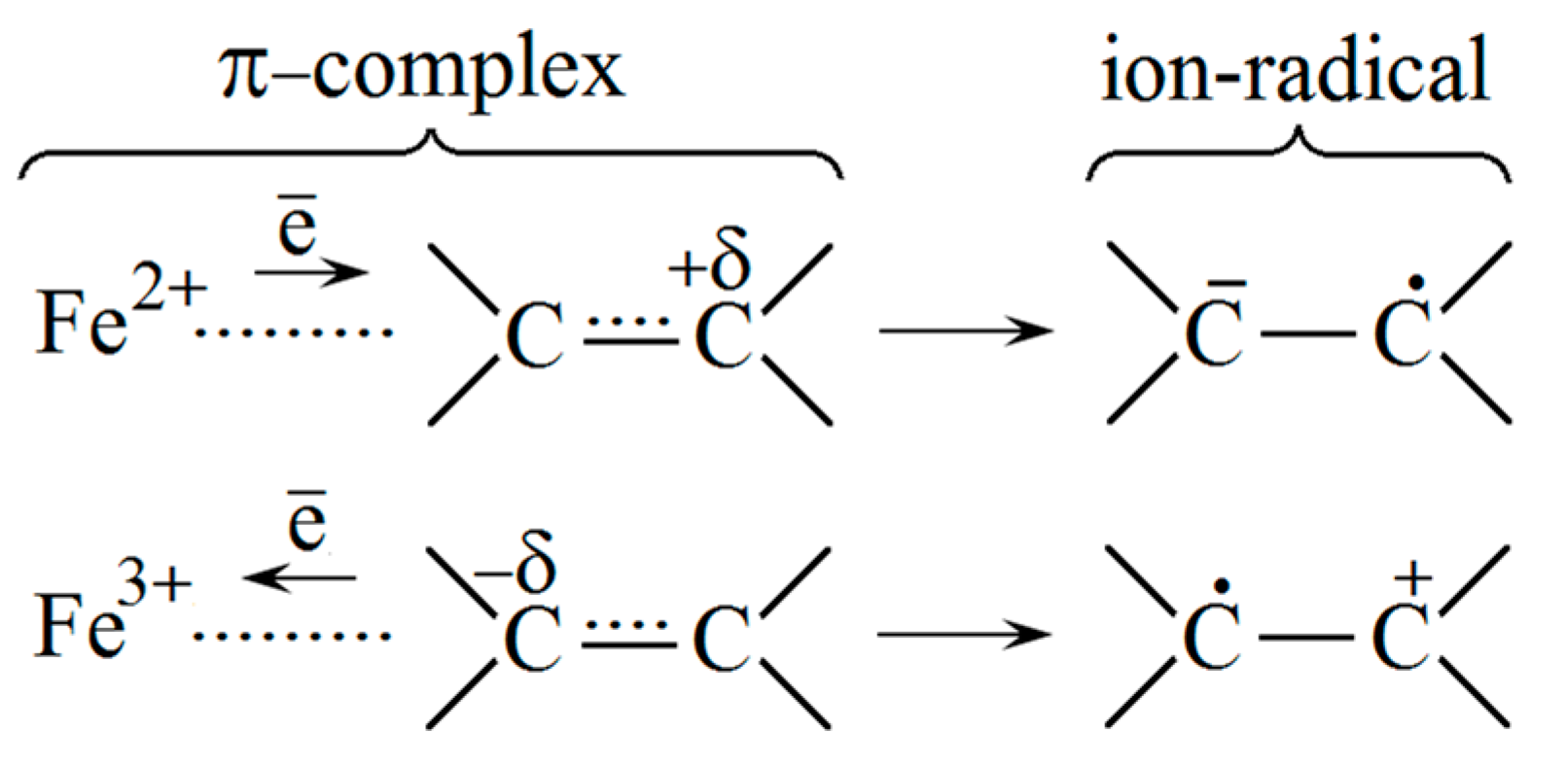
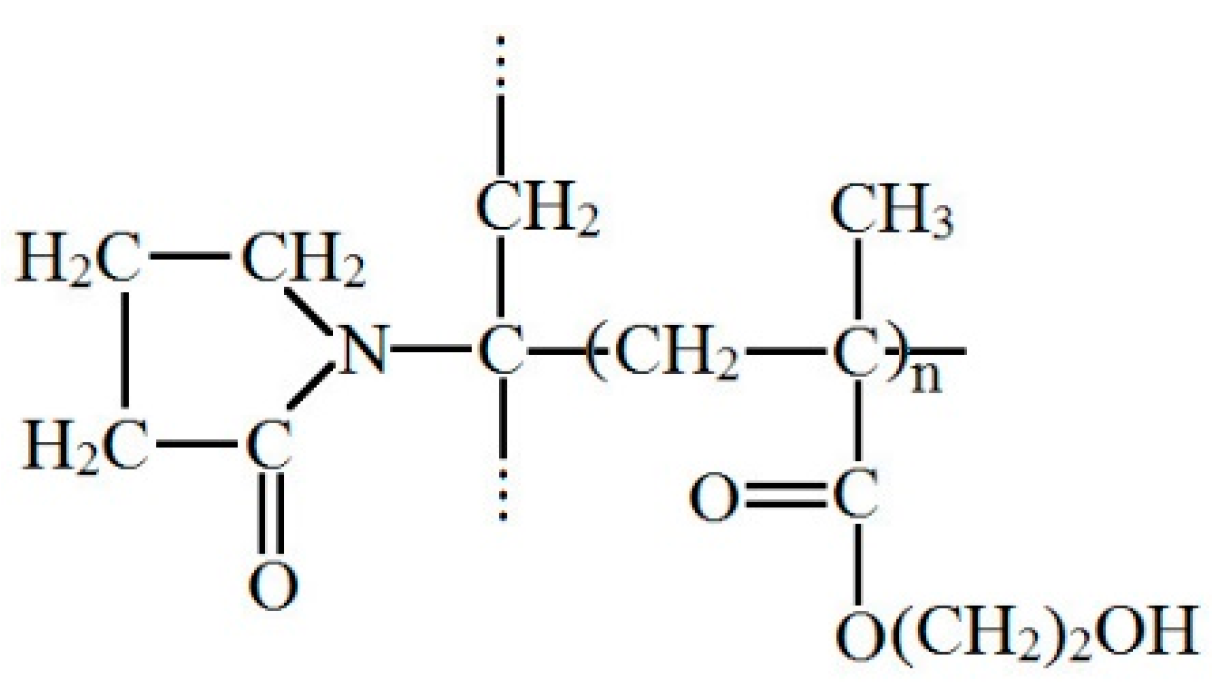
3.1. Study of the Structure of pHEMA-gr-PVP Copolymers
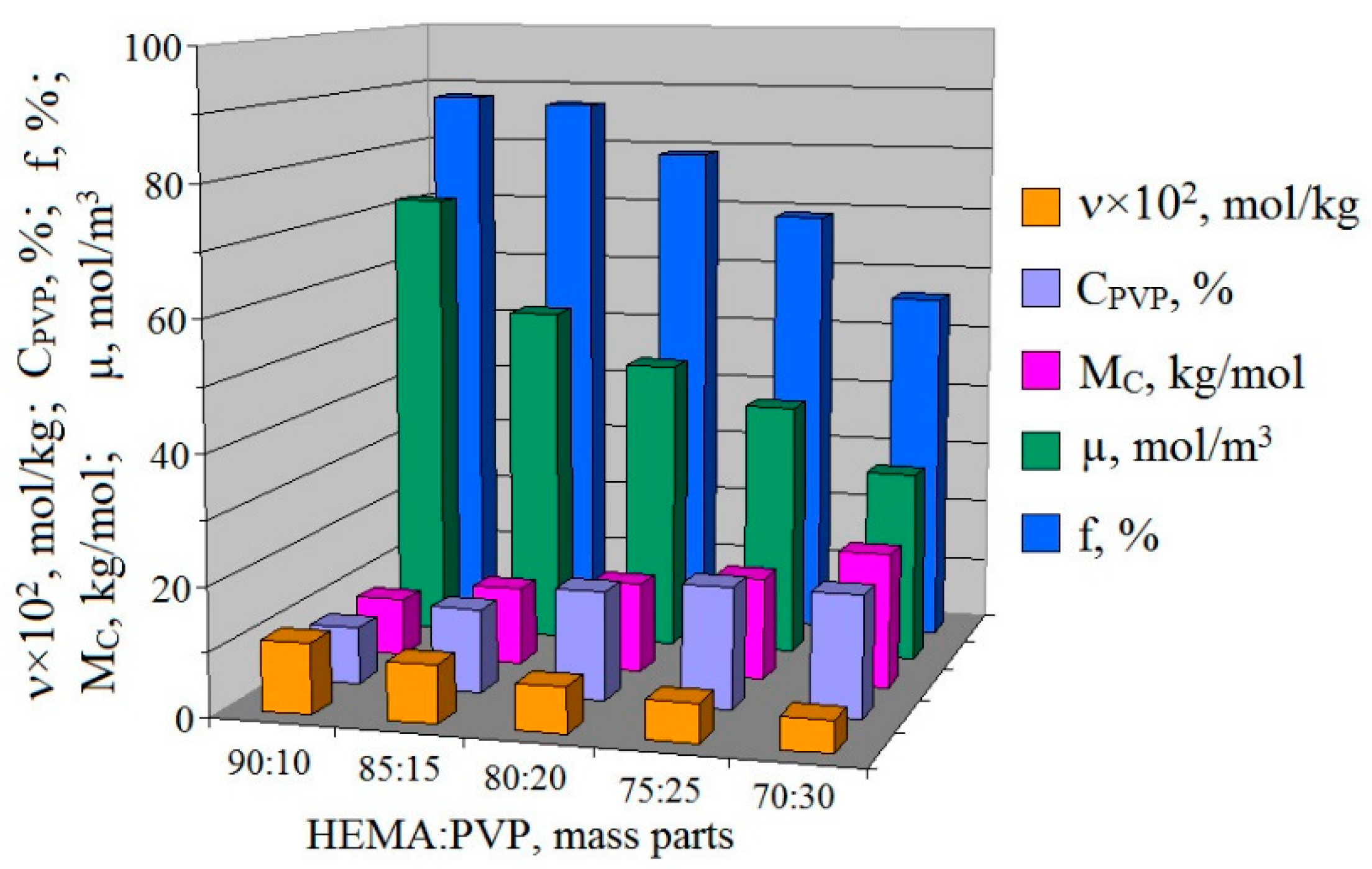
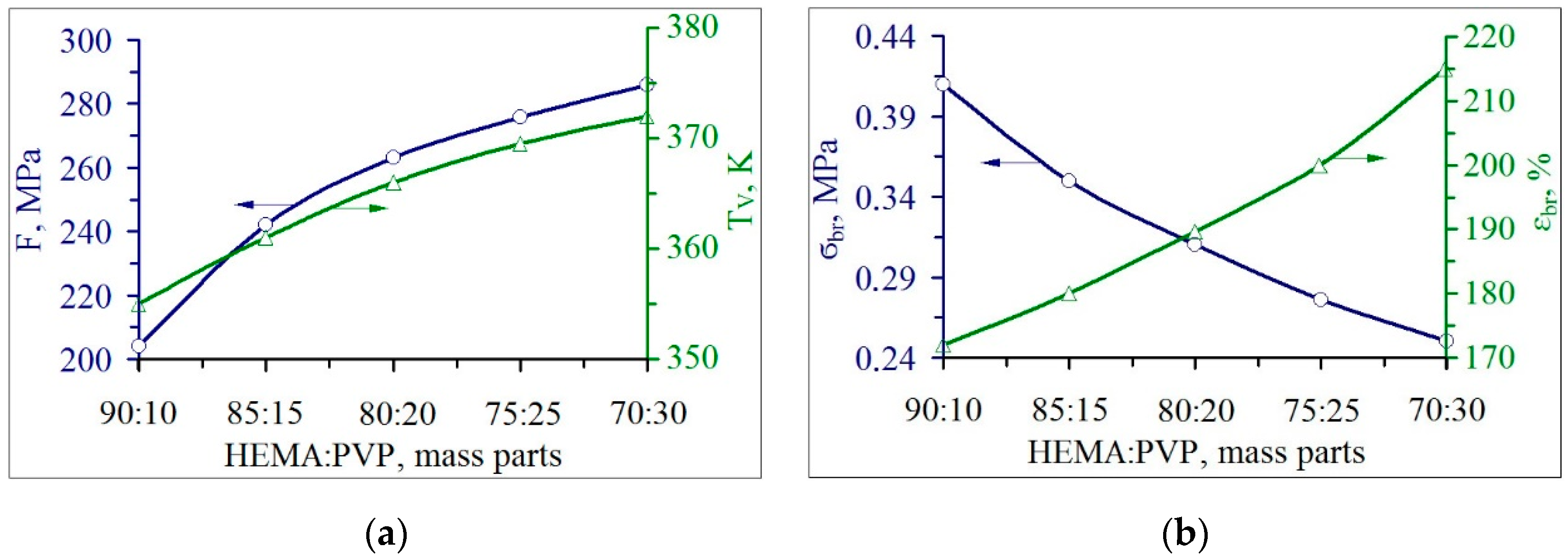
3.2. The Properties of pHEMA-gr-PVP Copolymers
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Khan, S.; Ullah, A.; Ullah, K.; Rehman, N. Insight into hydrogels. Des. Monomers Polym. 2016, 19, 456–478. [Google Scholar] [CrossRef]
- Ghobashy, M.M. Superabsorbent. In Hydrogels; Haider, S., Haider, A., Eds.; IntechOpen: London, UK, 2018; pp. 45–67. [Google Scholar] [CrossRef]
- Ahmed, E.M. Hydrogel: Preparation, characterization, and applications. J. Adv. Res. 2015, 6, 105–121. [Google Scholar] [CrossRef] [PubMed]
- Nadtoka, O.; Kutsevol, N.; Krysa, V.; Krysa, B. Hybrid polyacryamide hydrogels: Synthesis, properties and prospects of application. Mol. Cryst. Liq. Cryst. 2018, 672, 1–10. [Google Scholar] [CrossRef]
- Oyen, M.L. Mechanical characterisation of hydrogel materials. Int. Mater. Rev. 2013, 59, 44–59. [Google Scholar] [CrossRef]
- Caccavo, D.; Cascone, S.; Lamberti, G.; Barba, A.A. Hydrogels: Experimental characterization and mathematical modelling of their mechanical and diffusive behaviour. Chem. Soc. Rev. 2018, 47, 2357–2373. [Google Scholar] [CrossRef] [PubMed]
- Laftah, W.A.; Hashim, S.; Ibrahim, A.N. Polymer hydrogels: A Review. Polym. Plast. Technol. Eng. 2011, 50, 1475–1486. [Google Scholar] [CrossRef]
- Ganji, F.; Vasheghani-Farahani, S.; Vasheghani-Farahani, E. Theoretical description of hydrogel swelling: A review. Iran. Polym. J. 2010, 19, 375–398. [Google Scholar]
- Pal, K.; Banthia, A.K.; Majumdar, D.K. Polymeric hydrogels: Characterization and biomedical applications (review). Des. Monomers Polym. 2009, 12, 197–201. [Google Scholar] [CrossRef]
- Shoichet, M.S. Polymer scaffolds for biomaterials applications. Macromolecules 2010, 43, 581–591. [Google Scholar] [CrossRef]
- Nadtoka, O.; Kutsevol, N.; Linnik, O.; Nikiforov, M. Nanocomposite hydrogels containing silver nanoparticles as materials for wound dressings. In Nanophotonics, Nanooptics, Nanobiotechnology, and Their Applications; Fesenko, O., Yatsenko, L., Eds.; Springer: Cham, Switzerland, 2019; pp. 375–387. [Google Scholar] [CrossRef]
- Nawaz, S.; Khan, S.; Farooq, U.; Haider, M.S.; Ranjha, N.M.; Rasul, A.; Nawaz, A.; Arshad, N.; Hameed, R. Biocompatible hydrogels for the controlled delivery of anti-hypertensive agent: Development, characterization and in vitro evaluation. Des. Monomers Polym. 2018, 21, 18–32. [Google Scholar] [CrossRef]
- Hoffman, A.S. Hydrogels for biomedical applications. Adv. Drug Deliv. Rev. 2012, 64, 18–23. [Google Scholar] [CrossRef]
- Slaughter, B.V.; Khurshid, S.S.; Fisher, O.Z. Hydrogels in regenerative medicine. Adv. Mater. 2009, 21, 3307–3329. [Google Scholar] [CrossRef] [PubMed]
- Motealleh, A.; Kehr, N.S. Nanocomposite hydrogels and their applications in tissue engineering. Adv. Healthc. Mater. 2016, 6, 1600938. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Liu, M.; Zhang, Y.; Yin, J.; Pei, R. Nanocomposite hydrogels for tissue engineering applications. Nanoscale 2020, 12, 14976–14995. [Google Scholar] [CrossRef] [PubMed]
- Rafieian, S.; Mirzadeh, H.; Mahdavi, H.; Masoumi, M. Review on nanocomposite hydrogels and their biomedical applications. Sci. Eng. Compos. Mater. 2018, 26, 154–174. [Google Scholar] [CrossRef]
- Güven, O.; Sen, M. Preparation and characterization of poly(N-vinyl-2-pyrrolidone) hydrogels. Polymer 1991, 32, 2491–2495. [Google Scholar] [CrossRef]
- Suberlyak, O.; Grytsenko, O.; Hischak, K.; Hnatchuk, N. Effect of initiating system the structure and characteristics of hydrogels on the basis of polyvinylpyrrolidone copolymers. East. Eur. J. Enterp. Technol. 2013, 5/8, 59–63. [Google Scholar]
- Faragalla, M.M.; Hill, D.J.T.; Whittaker, A.K. The copolymerization ofN-vinyl-2-pyrrolidone with 2-hydoxyethyl methacrylate. Polym. Bull. 2002, 47, 421–427. [Google Scholar] [CrossRef]
- Grytsenko, O.; Pokhmurska, A.; Suberliak, S.; Kushnirchuk, M.; Panas, M.; Moravskyi, V.; Kovalchuk, R. Technological features in obtaining highly effective hydrogel dressings for medical purposes. East. Eur. J. Enterp. Technol. 2018, 6, 6–13. [Google Scholar] [CrossRef]
- Tang, Q.; Yu, J.-R.; Chen, L.; Zhu, J.; Hu, Z.-M. Preparation and properties of morphology controlled poly(2-hydroxyethyl methacrylate)/poly(N-vinyl pyrrolidone) double networks for biomedical use. Curr. Appl. Phys. 2010, 10, 766–770. [Google Scholar] [CrossRef]
- Yanez, F.; Concheiro, A.; Alvarez-Lorenzo, C. Macromolecule release and smoothness of semiinterpenetrating PVP–pHEMA networks for comfortable soft contact lenses. Eur. J. Pharm. Biopharm. 2008, 69, 1094–1103. [Google Scholar] [CrossRef] [PubMed]
- Suberlyak, O.; Skorokhoda, V.; Kozlova, N.; Melnyk, Y.; Semenyuk, N.; Chopyk, N. The polyvinylpyrrolidone graft copolymers and soft contact lenses on their basis. Sci. J. Sci. 2014, 5, 52–57. [Google Scholar] [CrossRef]
- Suberlyak, O.; Grytsenko, O.; Baran, N.; Yatsulchak, G.; Berezhnyy, B. Formation features of tubular products on the basis of composite hydrogels. Chem. Chem. Technol. 2020, 14, 312–317. [Google Scholar] [CrossRef]
- Jovašević, J.; Dimitrijević, S.; Filipović, J.; Tomić, S.; Mićić, M.; Suljovrujić, E. Swelling, mechanical and antimicrobial studies of Ag/P(HEMA/IA)/PVP semi-IPN hybrid hydrogels. Acta Phys. Pol. 2011, 120, 279–283. [Google Scholar] [CrossRef]
- Suberlyak, O.; Melnyk, Y.; Baran, N. High-hydrophilic membranes for dialysis and hemodialysis. Eng. Biomater. 2007, 63, 18–19. [Google Scholar]
- Ciardelli, G.; Cristallini, C.; Barbani, N.; Benedetti, G.; Crociani, A.; Travison, L.; Giusti, P. Bioartificial polymeric materials: -amylase, poly(2-hydroxyethyl methacrylate), poly(N-vinylpyrrolidone) system. Macromol. Chem. Phys. 2002, 203, 1666–1673. [Google Scholar] [CrossRef]
- Low, L.-M.; Seetharaman, S.; He, K.-Q.; Madou, M.J. Microactuators toward microvalves for responsive controlled drug delivery. Sens. Actuators B Chem. 2000, 67, 149–160. [Google Scholar] [CrossRef]
- Frutos, P.; Diez-Peña, E.; Frutos, G.; Barrales-Rienda, J. Release of gentamicin sulphate from a modified commercial bone cement. Effect of (2-hydroxyethyl methacrylate) comonomer and poly(N-vinyl-2-pyrrolidone) additive on release mechanism and kinetics. Biomaterials 2002, 23, 3787–3797. [Google Scholar] [CrossRef]
- Domingues, J.; Bonelli, N.; Giorgi, R.; Baglioni, P. Chemical semi-IPN hydrogels for the removal of adhesives from canvas paintings. Appl. Phys. A 2013, 114, 705–710. [Google Scholar] [CrossRef]
- Bashtyk, Y.; Fechan, A.; Grytsenko, O.; Hotra, Z.; Kremer, I.; Suberlyak, O.; Aksimentyeva, O.; Horbenko, Y.; Kotsarenko, M. Electrical elements of the optical systems based on hydrogel-electrochromic polymer composites. Mol. Cryst. Liq. Cryst. 2019, 672, 150–158. [Google Scholar] [CrossRef]
- Suberlyak, O.; Skorokhoda, V.; Grytsenko, O. Complex PVP–Men+–active catalyst of vinyl monomers polymerization. Mater. Polim. Ich Przetw. 2004, 1, 140–145. [Google Scholar]
- Suberlyak, O.; Grytsenko, O.; Kochubei, V. The role of FeSO4 in the obtaining of polyvinylpirolidone copolymers. Chem. Chem. Technol. 2015, 9, 429–434. [Google Scholar] [CrossRef]
- Montheard, J.; Chatzopoulos, M.; Chappard, D. 2-Hydroxyethyl Methacrylate (HEMA): Chemical properties and applications in biomedical fields. J. Macromol. Sci. C 1992, 32, 1–34. [Google Scholar] [CrossRef]
- Malesic, N.; Rusmirovic, J.; Jovasevic, J.; Perisic, M.; Dimitrijevic-Brankovic, S.; Filipovic, J.; Tomic, S. Antimicrobial hydrogels based on 2-hydroxyethyl methacrylate and itaconic acid containing silver(i) ion. Tehnika 2014, 69, 563–568. [Google Scholar] [CrossRef]
- Prasitsilp, M.; Siriwittayakorn, T.; Molloy, R.; Suebsanit, N.; Siriwittayakorn, P.; Veeranondha, S. Cytotoxicity study of homopolymers and copolymers of 2-hydroxyethyl methacrylate and some alkyl acrylates for potential use as temporary skin substitutes. J. Mater. Sci. Mater. Med. 2003, 14, 595–600. [Google Scholar] [CrossRef]
- Wang, J.; Wu, W. Swelling behaviors, tensile properties and thermodynamic studies of water sorption of 2-hydroxyethyl methacrylate/epoxy methacrylate copolymeric hydrogels. Eur. Polym. J. 2005, 41, 1143–1151. [Google Scholar] [CrossRef]
- Suberlyak, O.; Skorokhoda, V. Hydrogels based on polyvinylpyrrolidone copolymers. In Hydrogels; Haider, S., Haider, A., Eds.; IntechOpen: London, UK, 2018; pp. 136–214. [Google Scholar] [CrossRef]
- Çaykara, T. Solubility parameters of cross-Linked poly(N-vinyl-2-pyrrolidone-co-crotonic acid) copolymers prepared byg-ray-induced polymerization technique. J. Macromol. Sci. A 2004, 41, 971–979. [Google Scholar] [CrossRef]
- Teodorescu, M.; Bercea, M. Poly(vinylpyrrolidone)—A versatile polymer for biomedical and beyond medical applications. Polym. Plast. Technol. Eng. 2015, 54, 923–943. [Google Scholar] [CrossRef]
- Teodorescu, M.; Morariu, S.; Bercea, M.; Săcărescu, L. Viscoelastic and structural properties of poly(vinyl alcohol)/poly(vinylpyrrolidone) hydrogels. RSC Adv. 2016, 6, 39530–40625. [Google Scholar] [CrossRef]
- Rodriguez, G.; Gonzalez, G.; Silva, P. Synthesis and characterization of metallics nanoparticles stabilized with polyvinylpyrrolidone. Microsc. Microanal. 2005, 11, 1944–1945. [Google Scholar] [CrossRef]
- Grytsenko, O.M.; Suberlyak, O.V.; Moravskyi, V.S.; Gayduk, A.V. Investigation of nickel chemical precipitation kinetics. East. Eur. J. Enterp. Technol. 2016, 1, 26–31. [Google Scholar] [CrossRef][Green Version]
- Suberlyak, O.; Baran, N.; Melnyk, Y.; Grytsenko, O.; Yatsulchak, G. Influence of the molecular weight of polyvinylpyrrolidone on the physicomechanical properties of composite polyamide hydrogel membranes. Mater. Sci. 2020, 55, 758–764. [Google Scholar] [CrossRef]
- Suberlyak, O.V.; Krasins’kyi, V.V.; Shapoval, I.M.; Hrytsenko, O.M. Influence of the mechanism and parameters of hardening of modified novolac phenol-formaldehyde resins on the physicomechanical properties of the composite. Mater. Sci. 2011, 46, 669–678. [Google Scholar] [CrossRef]
- Krasinskyi, V.; Suberlyak, O.; Dulebová, L.; Antoniuk, V. Nanocomposites on the basis of thermoplastics and montmorillonite modified by polyvinylpyrrolidone. Key Eng. Mater. 2017, 756, 3–10. [Google Scholar] [CrossRef]
- Li, J.; Inukai, K.; Takahashi, Y.; Tsuruta, A.; Shin, W. Thin film coating with highly dispersible barium titanate-polyvinylpyrrolidone nanoparticles. Materials 2018, 11, 712. [Google Scholar] [CrossRef]
- Itoh, T.; Uchida, T.; Izu, N.; Shin, W. Effect of core-shell ceria/poly(vinylpyrrolidone) (PVP) nanoparticles incorporated in polymer films and their optical properties (2): Increasing the refractive index. Materials 2017, 10, 710. [Google Scholar] [CrossRef]
- Willersinn, J.; Schmidt, B.V.K.J. Self-Assembly of Double Hydrophilic Poly(2-ethyl-2-oxazoline)-b-poly(N-vinylpyrrolidone) Block Copolymers in Aqueous Solution. Polymers 2017, 9, 293. [Google Scholar] [CrossRef] [PubMed]
- Del Sorbo, G.R.; Truda, G.; Bifulco, A.; Passaro, J.; Petrone, G.; Vitolo, B.; Ausanio, G.; Vergara, A.; Marulo, F.; Branda, F. Non monotonous effects of noncovalently functionalized graphene addition on the structure and sound absorption properties of polyvinylpyrrolidone (1300 kDa) electrospun mats. Materials 2019, 12, 108. [Google Scholar] [CrossRef]
- Suberlyak, O.; Baran, N.; Melnyk, Y.; Yatsulchak, G. Formation of composite hydrogel membranes. Vopr. Khimii Khimicheskoi Tekhnologii 2018, 3, 121–126. [Google Scholar]
- Suberlyak, O.V.; Hrytsenko, O.M.; Hishchak, K.Y. Influence of the metal surface of powder filler om the structure and properties of composite materials based on the co-polymers of methacrylates with polyvinylpyrrolidone. Mater. Sci. 2016, 52, 155–164. [Google Scholar] [CrossRef]
- Krezović, B.; Dimitrijević, S.; Filipović, J.; Nikolić, R.; Tomić, S. Antimicrobial P(HEMA/IA)/PVP semi-interpenetrating network hydrogels. Polym. Bull. 2012, 70, 809–819. [Google Scholar] [CrossRef]
- Suberlyak, O.; Grytsenko, O.; Hischak, K.; Hnatchuk, N. Research of influence the nature of metal on mechanism of synthesis polyvinilpyrrolidone metal copolymers. Chem. Chem. Technol. 2013, 7, 289–294. [Google Scholar] [CrossRef]
- Roy, N.; Saha, N. PVP-based hydrogels: Synthesis, properties and applications. In Hydrogels: Synthesis, Characterization and Applications; Câmara, F., Ferreira, L., Eds.; Nova Science Publishers Inc.: Hauppauge, NY, USA, 2012; pp. 227–252. [Google Scholar]
- Pan, X.; Guo, X.; Choi, B.; Feng, A.; Wei, X.; Thang, S.H. A facile synthesis of pH stimuli biocompatible block copolymer poly(methacrylic acid)-block-poly(N-vinylpyrrolidone) utilizing switchable RAFT agents. Polym. Chem. 2019, 10, 2083–2090. [Google Scholar] [CrossRef]
- Muller, K. Identification and determination of polyvinylpyrrolidone (PVP) as well as determination of active substances in PVP-containing drug preparations. Pharm. Acta Helv. 1968, 43, 107–122. [Google Scholar]
- Babich, V.F.; Sivergin, Y.M.; Berlin, A.A.; Rabinovich, A.L. Correlation between the equilibrium modulus of high elasticity and the number of crosslinks in rigid crosslinked polymers. Polym. Mech. 1966, 2, 1–3. [Google Scholar] [CrossRef]
- Grytsenko, O.; Gajdos, I.; Spišák, E.; Krasinskyi, V.; Suberlyak, O. Novel Ni/pHEMA-gr-PVP composites obtained by polymerization with simultaneous metal deposition: Structure and properties. Materials 2019, 12, 1956. [Google Scholar] [CrossRef]
- Das, N. Preparation methods and properties of hydrogel: A review. Int. J. Pharm. Pharm. Sci. 2013, 5, 112–117. [Google Scholar]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grytsenko, O.; Dulebova, L.; Suberlyak, O.; Skorokhoda, V.; Spišák, E.; Gajdoš, I. Features of Structure and Properties of pHEMA-gr-PVP Block Copolymers, Obtained in the Presence of Fe2+. Materials 2020, 13, 4580. https://doi.org/10.3390/ma13204580
Grytsenko O, Dulebova L, Suberlyak O, Skorokhoda V, Spišák E, Gajdoš I. Features of Structure and Properties of pHEMA-gr-PVP Block Copolymers, Obtained in the Presence of Fe2+. Materials. 2020; 13(20):4580. https://doi.org/10.3390/ma13204580
Chicago/Turabian StyleGrytsenko, Oleksandr, Ludmila Dulebova, Oleh Suberlyak, Volodymyr Skorokhoda, Emil Spišák, and Ivan Gajdoš. 2020. "Features of Structure and Properties of pHEMA-gr-PVP Block Copolymers, Obtained in the Presence of Fe2+" Materials 13, no. 20: 4580. https://doi.org/10.3390/ma13204580
APA StyleGrytsenko, O., Dulebova, L., Suberlyak, O., Skorokhoda, V., Spišák, E., & Gajdoš, I. (2020). Features of Structure and Properties of pHEMA-gr-PVP Block Copolymers, Obtained in the Presence of Fe2+. Materials, 13(20), 4580. https://doi.org/10.3390/ma13204580






