Hybrid Fluorescent Poly(silsesquioxanes) with Amide- and Triazole-Containing Side Groups for Light Harvesting and Cation Sensing
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Instrumentation
2.2.1. Nuclear Magnetic Resonance (NMR)
2.2.2. Thermal Analysis
2.2.3. Fourier-Transform Infrared Spectroscopy (FTIR)
2.2.4. Wide Angle X-Ray Scattering (WAXS)
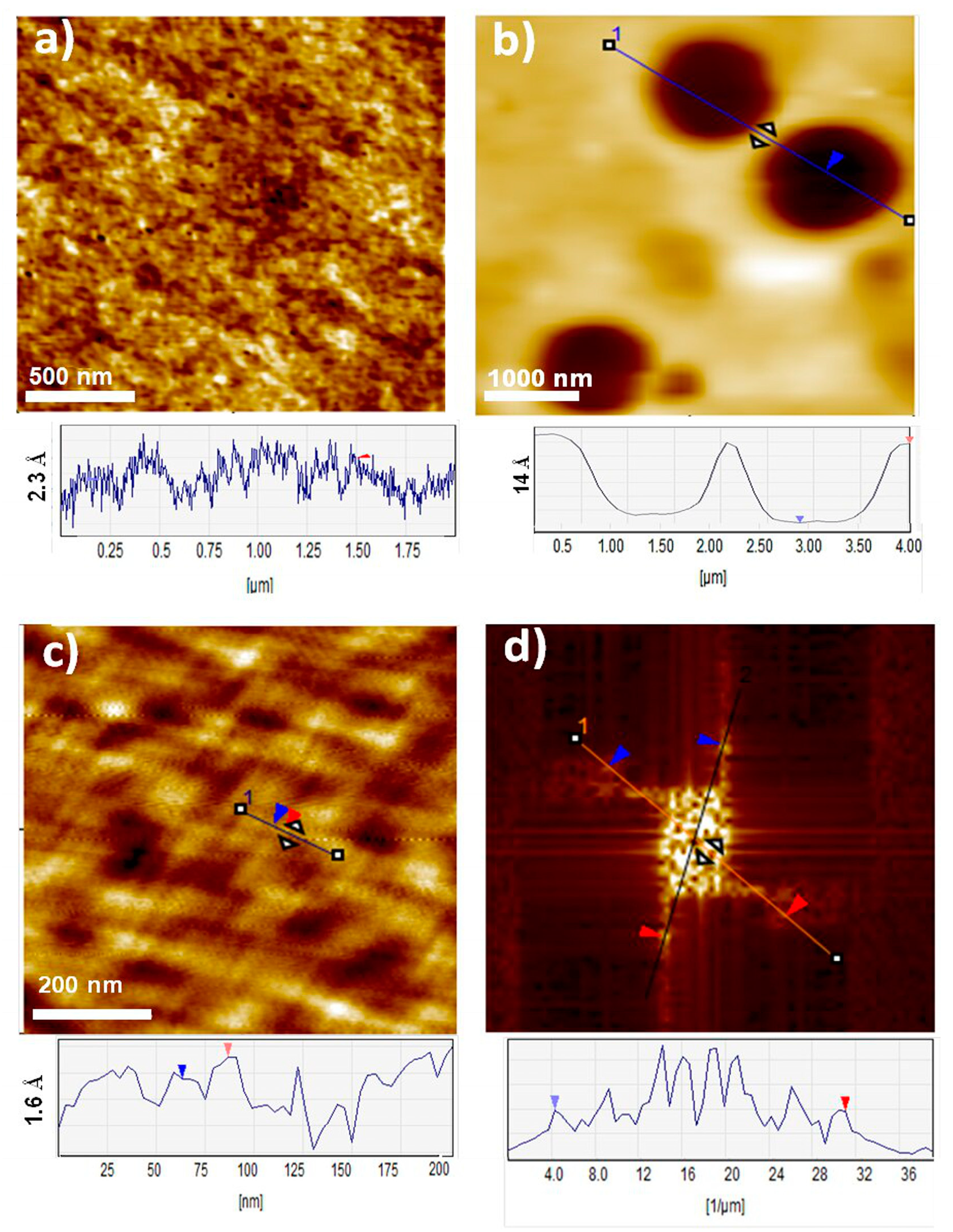
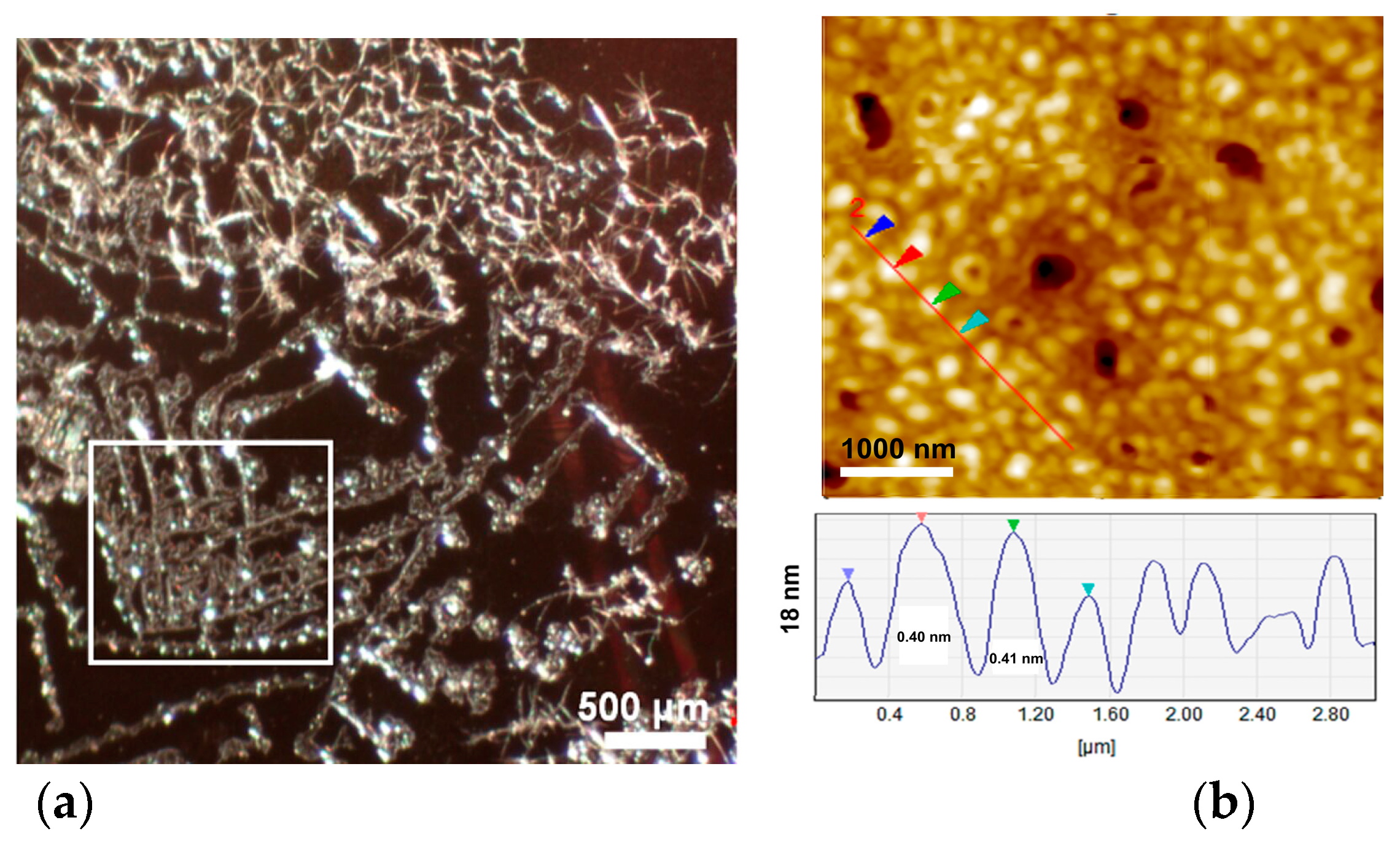
2.2.5. Atomic Force Microscopy (AFM)
2.2.6. Molecular Modelling
2.2.7. Reflected Light Microscopy
2.2.8. Ultraviolet–Visible Spectroscopy (UV)
2.2.9. Fluorescence Spectroscopy (FL)
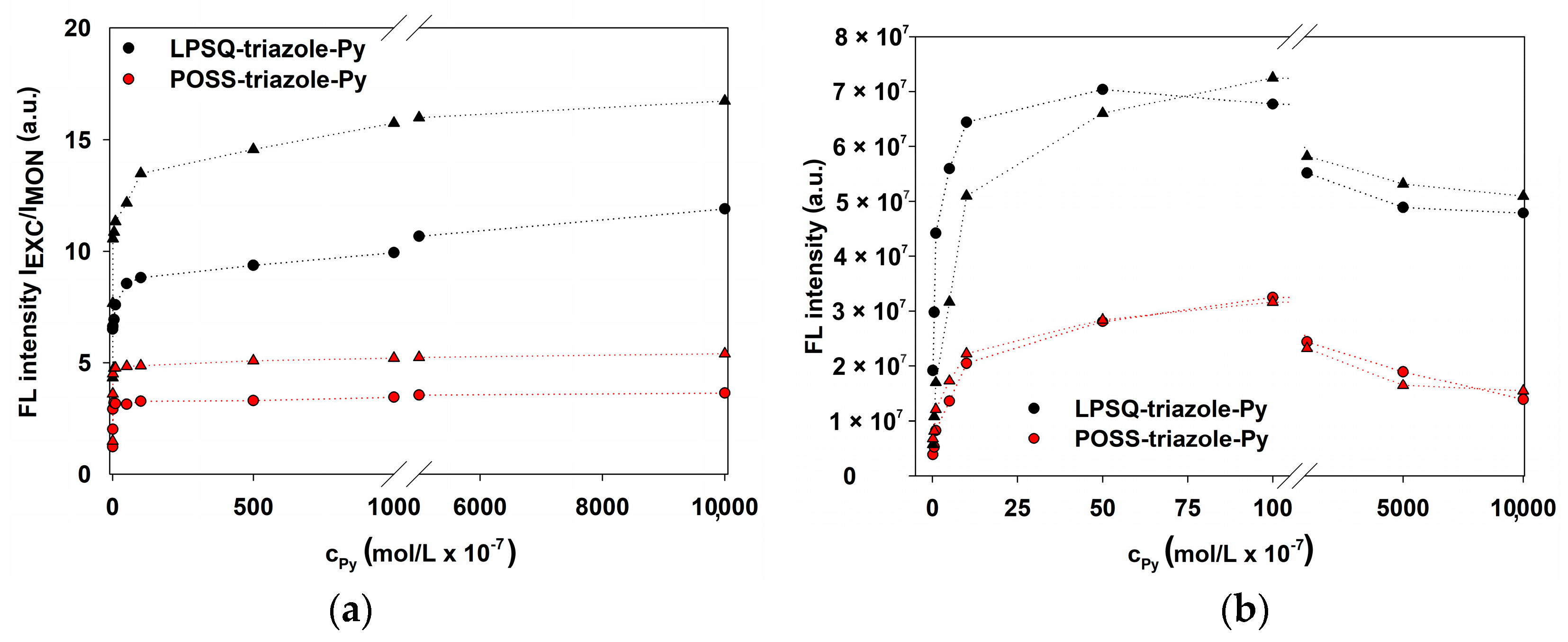
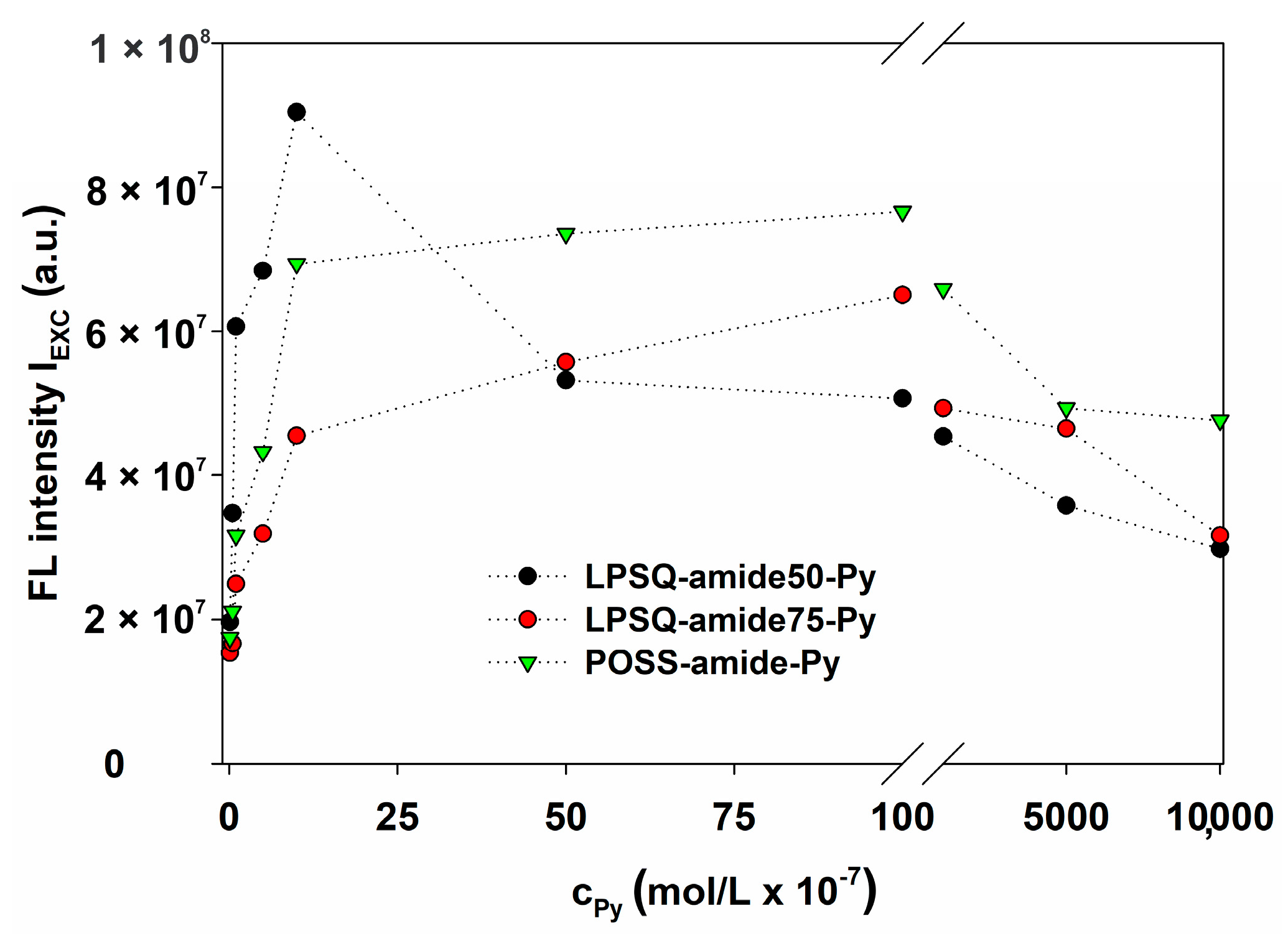
2.2.10. Fluorescence Energy Transfer
2.2.11. Detection of Metal Cations
3. Results and Discussion
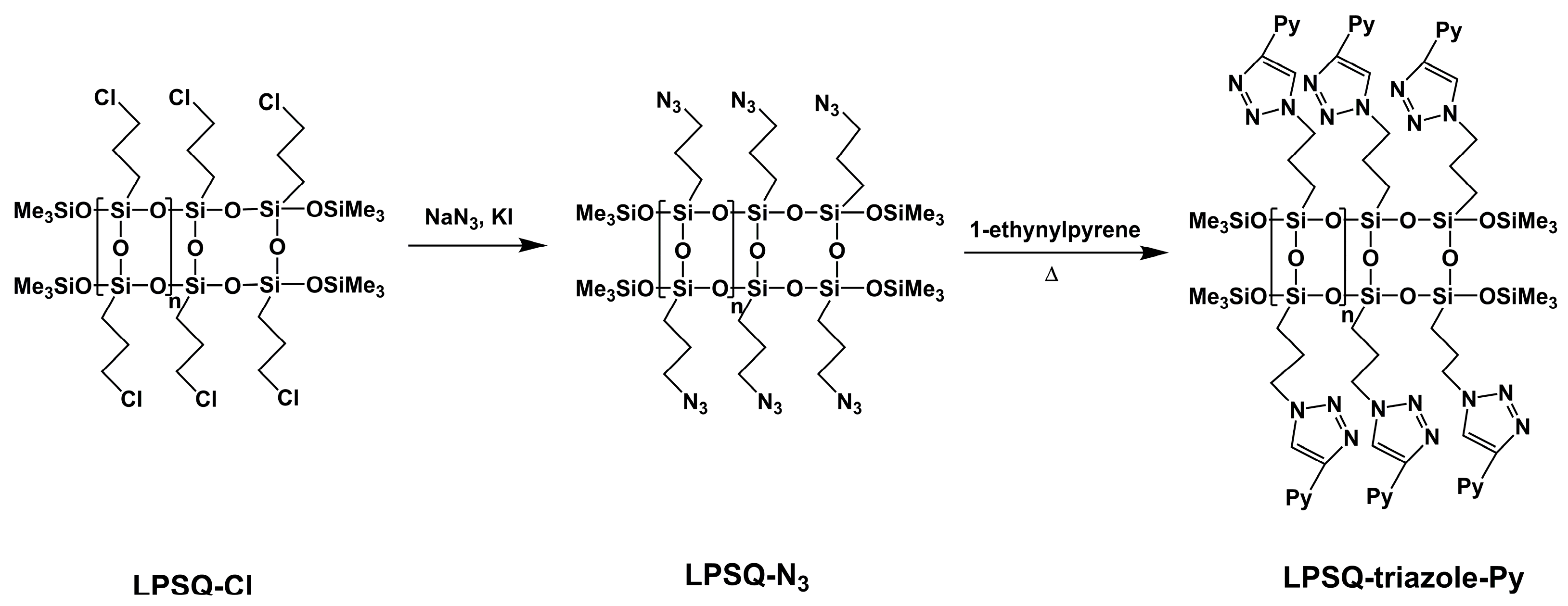
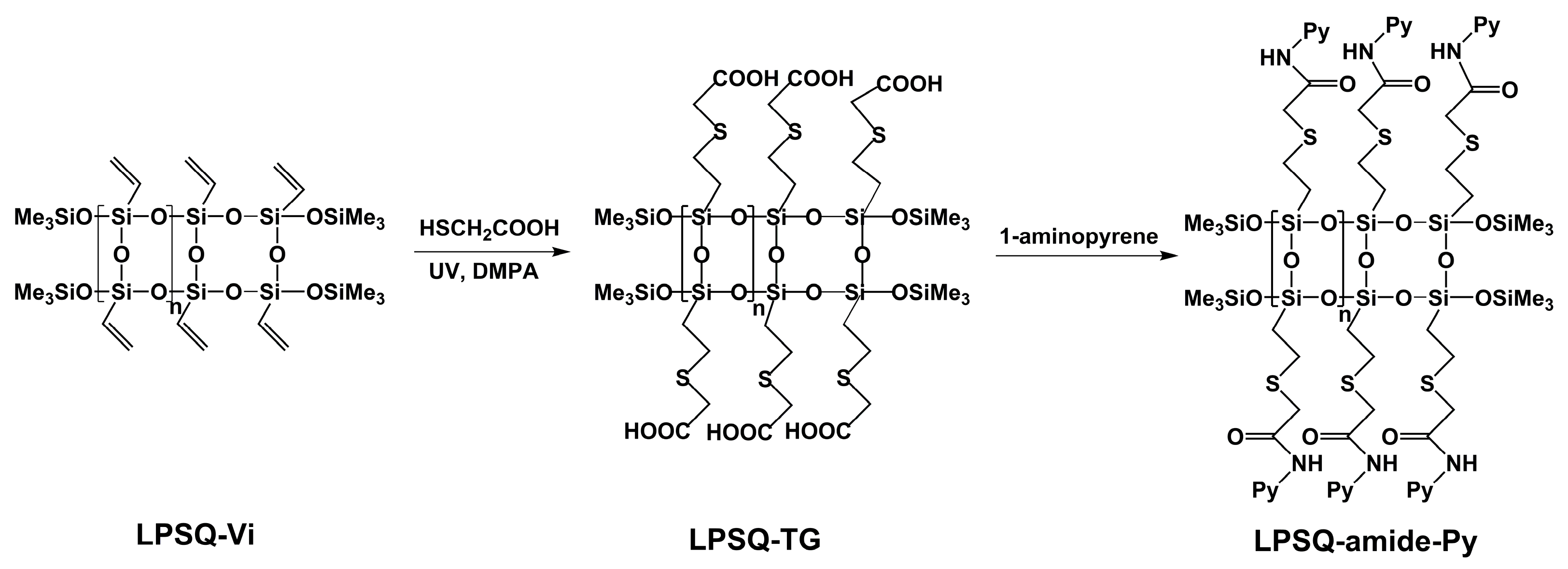
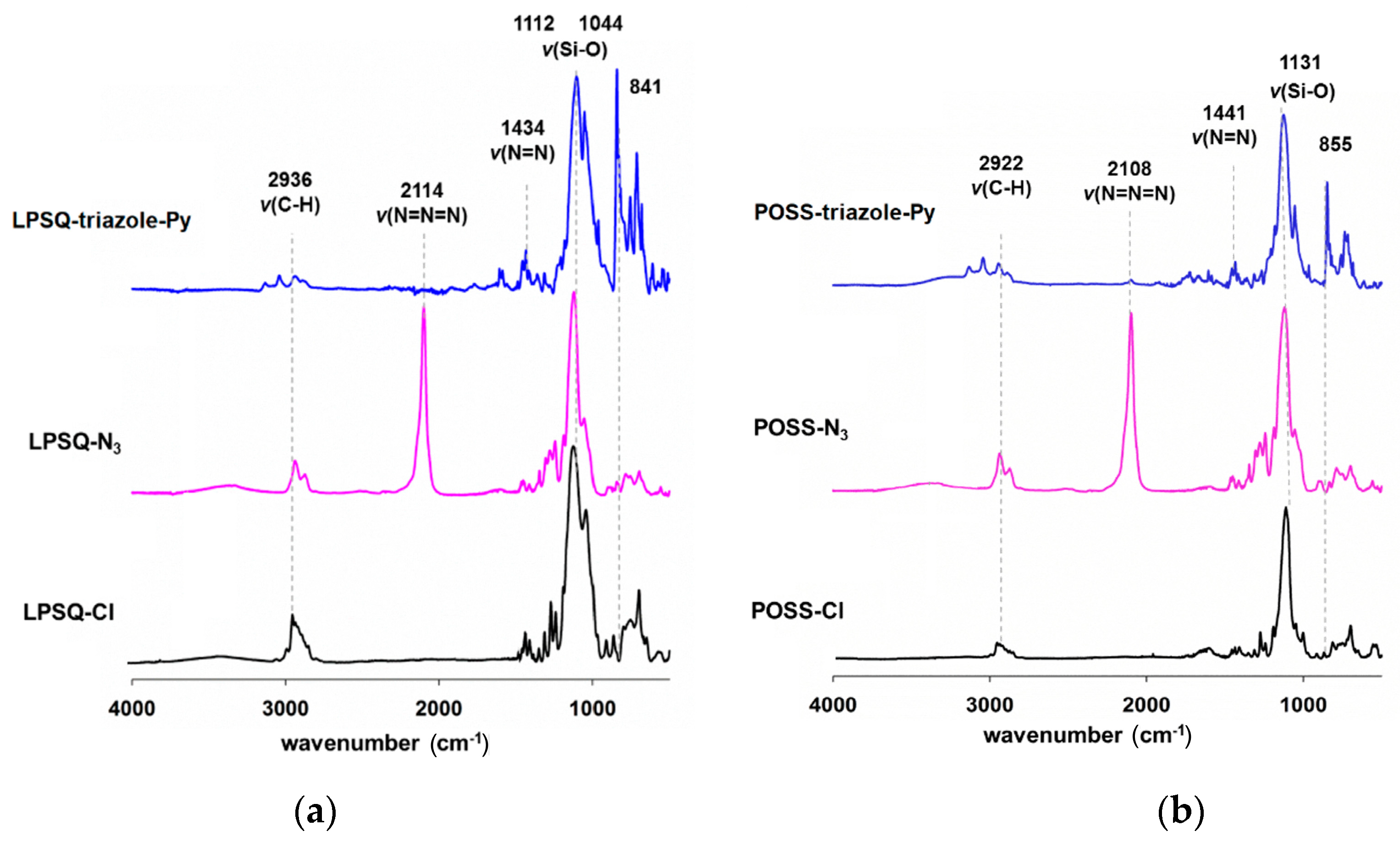
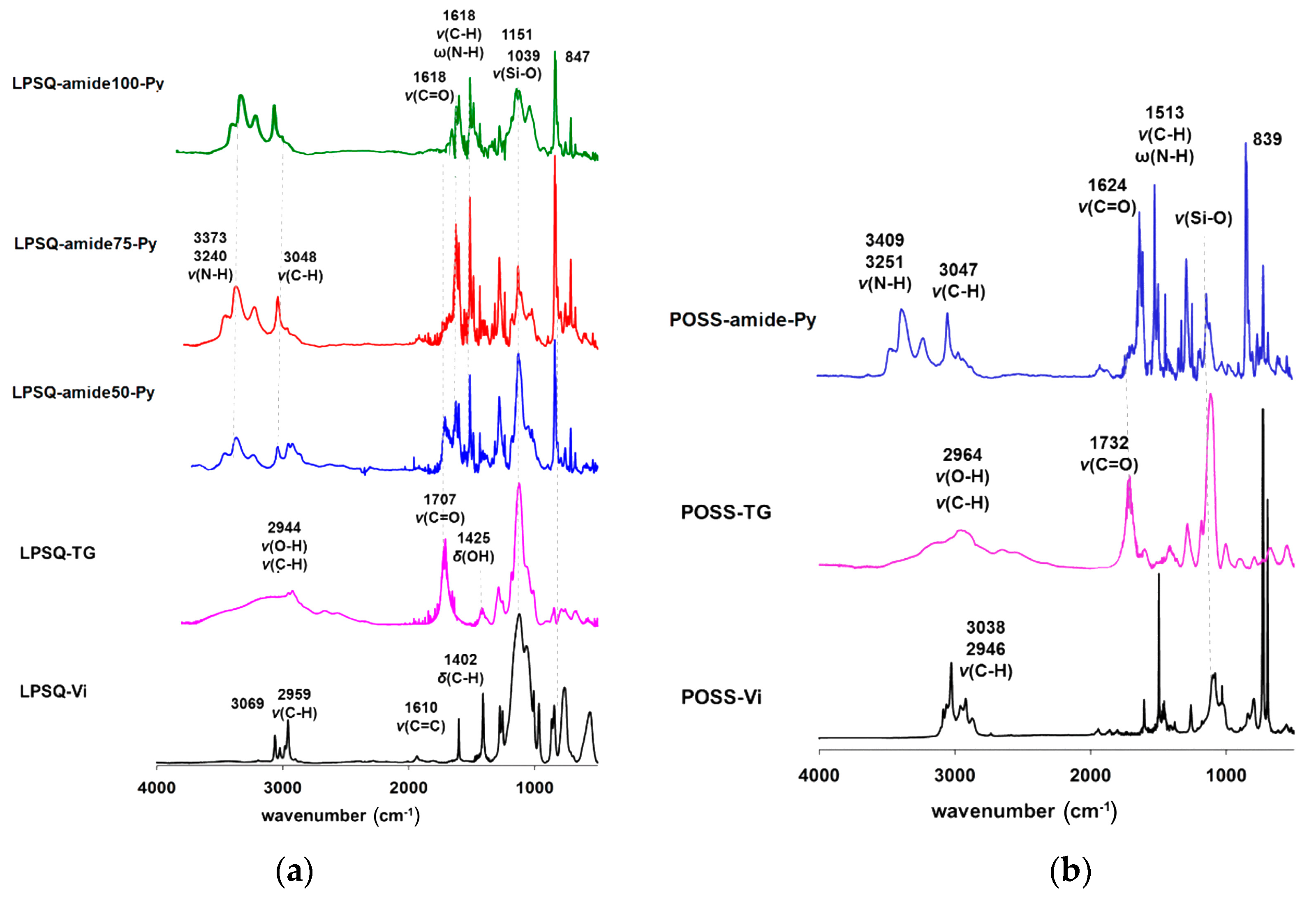
3.1. Synthesis, Structure and Thermal Properties of Fluorescent LPSQ
3.2. Organization of LPSQ and POSS Materials in Solid State
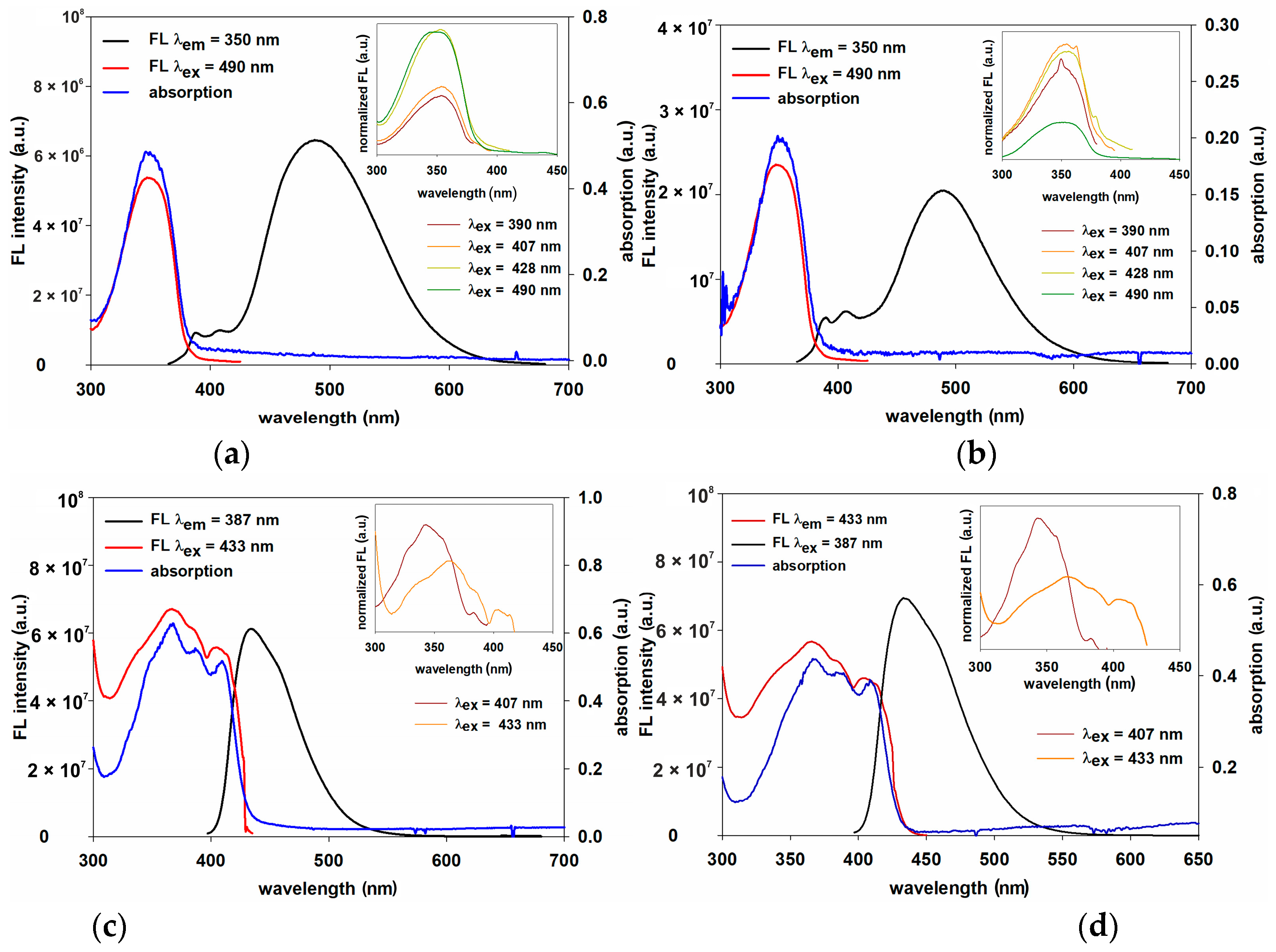
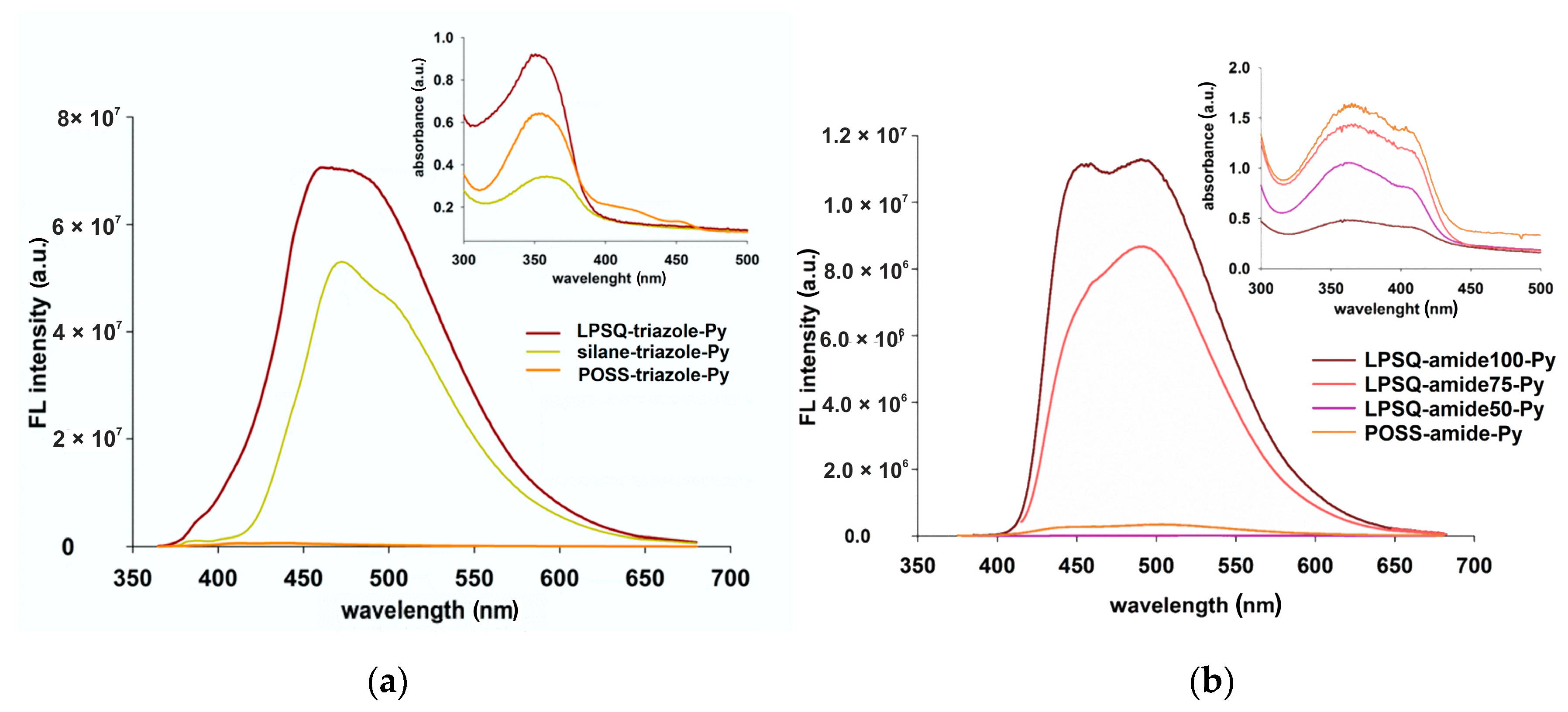
3.3. Optical and Photophysical Properties—Comparison of LPSQ/POSS-Triazole and LPSQ-POSS-Amide
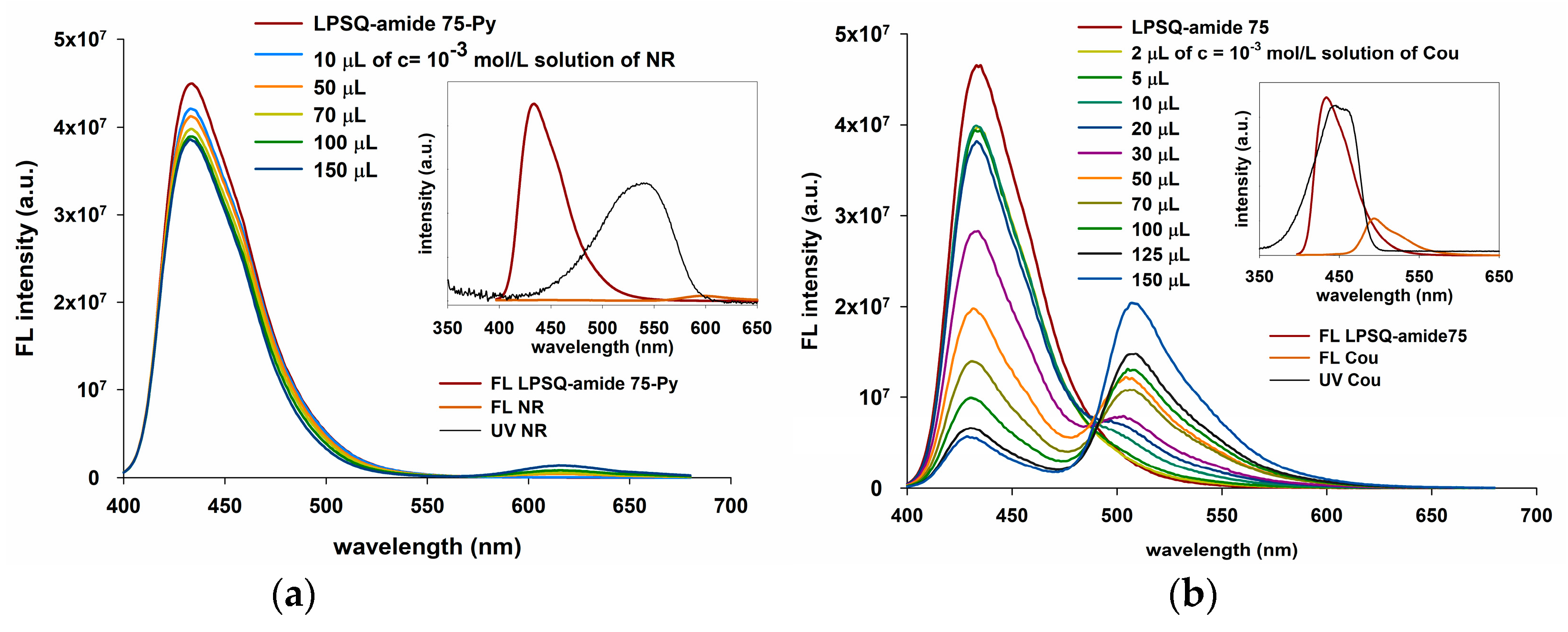
3.4. Energy Transfer with LPSQ-Triazole-Py and LPSQ-Amide-Py
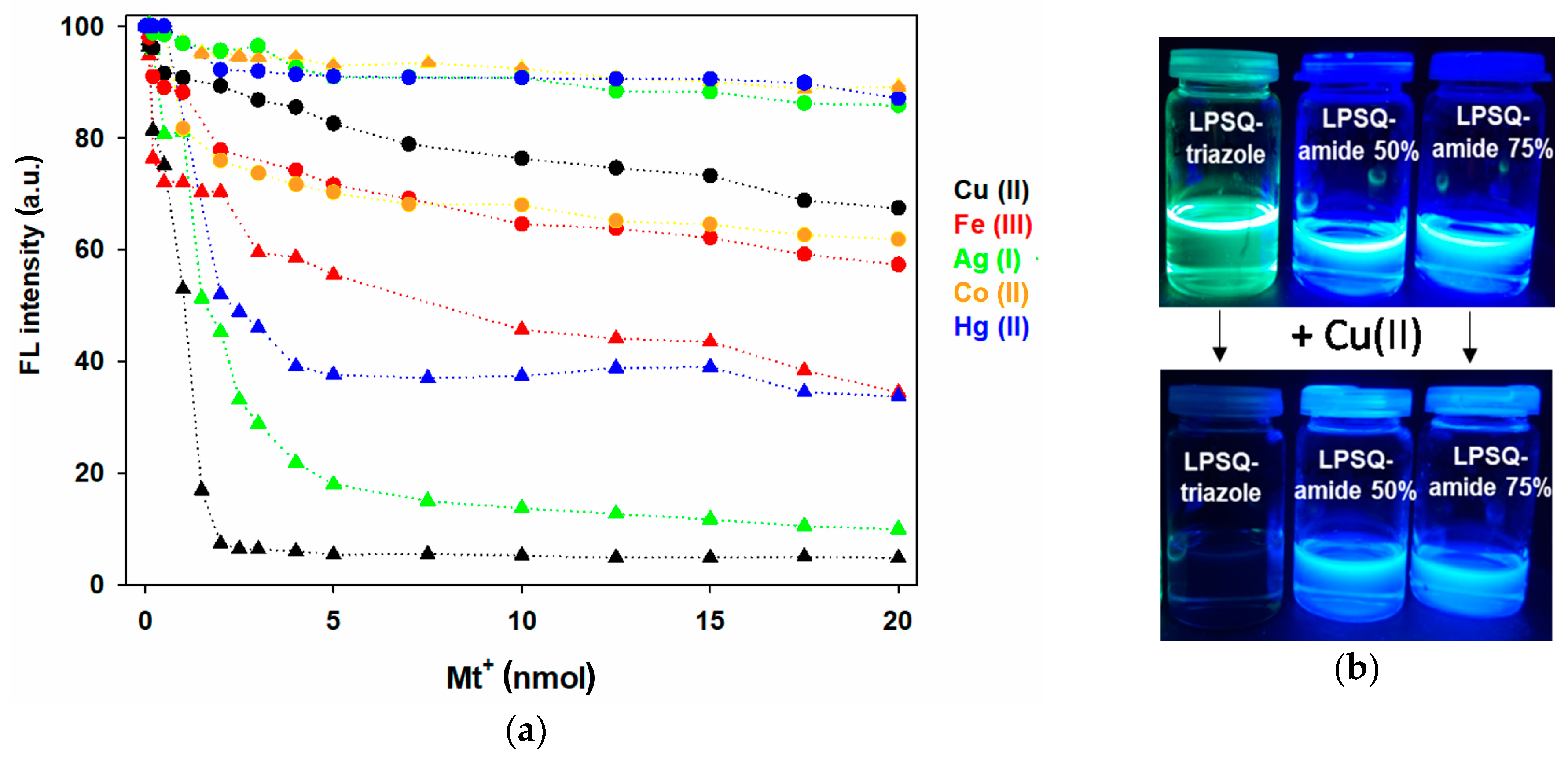
3.5. Cation Sensing Ability of the Hybrid Silsesquioxanes
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Cho, S.; Hwang, N.; Lee, S.E.; Kim, Y.K.; Yoon, S.S. Highly efficient blue prganic light-emitting diodes based on diarylamine-substituted pyrene derivatives. J. Nanosci. Nanotechnol. 2019, 19, 1056–1060. [Google Scholar] [CrossRef] [PubMed]
- Cai, G.; Xue, P.; Chen, Z.; Li, T.; Liu, K.; Ma, W.; Lian, J.; Zeng, P.; Wang, Y.; Han, R.P.S.; et al. High-performance mid-bandgap fused-pyrene electron acceptor. Chem. Mater. 2019, 31, 6484–6490. [Google Scholar] [CrossRef]
- Feng, X.; Hu, J.-Y.; Redshaw, C.; Yamato, T. Functionalization of pyrene to prepare luminescent materials-typical examples of synthetic methodology. Chem. Eur. J. 2016, 22, 11898–11916. [Google Scholar] [CrossRef] [PubMed]
- Winnik, F.M. Photophysics of preassociated pyrenes in aqueous polymer solutions and in other organized media. Chem. Rev. 1993, 93, 587–614. [Google Scholar] [CrossRef]
- Figueira-Duarte, T.M.; Müllen, K. Pyrene-based materials for organic electronics. Chem. Rev. 2011, 111, 7260–7314. [Google Scholar] [CrossRef]
- Armaroli, N.; Balzani, V. The future of energy supply: Challenges and opportunities. Angew. Chem. Int. Ed. 2007, 46, 52–62. [Google Scholar] [CrossRef]
- Kundu, S.; Patra, A. Nanoscale strategies for light harvesting. Chem. Rev. 2017, 117, 712–757. [Google Scholar] [CrossRef]
- Sahoo, H. Förster resonance energy transfer—A spectroscopic nanoruler: Principle and applications. J. Photochem. Photobiol. C Photochem. Rev. 2011, 12, 20–30. [Google Scholar] [CrossRef]
- Ferguson, J. Absorption and fluorescence spectra of crystalline pyrene. J. Chem. Phys. 1958, 28, 765. [Google Scholar] [CrossRef]
- Vassiliev, S.; Bruce, D. Toward understanding molecular mechanisms of light harvesting and charge separation in photosystem II. Photosynth. Res. 2008, 97, 75–89. [Google Scholar] [CrossRef]
- Wu, J.; Liu, W.; Ge, J.; Zhang, H.; Wang, P. New sensing mechanisms for design of fluorescent chemosensors emerging in recent years. Chem. Soc. Rev. 2011, 40, 3483–3495. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Sedgwick, A.C.; Gunnlaugsson, T.; Akkaya, E.U.; Yoon, J.; James, T. Fluorescent chemosensors: The past, present and future. Chem. Soc. Rev. 2017, 46, 7105–7123. [Google Scholar] [CrossRef] [PubMed]
- Jeong, Y.; Yoon, J. Recent progress on fluorescent chemosensors for metal ions. Inorg. Chim. Acta 2012, 381, 2–14. [Google Scholar] [CrossRef]
- Feng, J.; Tian, K.; Hu, D.; Wang, S.; Zeng, Y.; Li, Y.; Yang, Y. A triarylboron-based fluorescent thermometer: Sensitive over a wide temperature range. Angew. Chem. Int. Ed. 2011, 50, 8072–8076. [Google Scholar] [CrossRef]
- Templer, R.H.; Castle, S.J.; Curran, A.R.; Rumbles, G.; Klug, D.R. Sensing isothermal changes in the lateral pressure in model membranes using di-pyrenyl phosphatidylcholine. Faraday Discuss. 1998, 111, 41–53. [Google Scholar] [CrossRef]
- Faggi, E.; Serra-Vinardell, J.; Pandey, M.D.; Casas, J.; Fabriàs, G.; Luis, S.V.; Alfonso, I. Pseudopeptidic fluorescent on-off pH sensor based on pyrene excimer emission: Imaging of acidic cellular organelles. Sens. Actuators B Chem. 2016, 234, 633–640. [Google Scholar] [CrossRef]
- Fujiwara, Y.; Amao, Y. Optical oxygen sensor based on controlling the excimer formation of pyrene-1-butylic acid chemisorption layer onto nano-porous anodic oxidized aluminium plate by myristic acid. Sens. Actuators B Chem. 2003, 89, 58–61. [Google Scholar] [CrossRef]
- Kathiravan, A.; Gowri, A.; Khamrang, T.; Kumar, M.D.; Dhenadhayalan, N.; Lin, K.C.; Velusamy, M.; Jaccob, M. Pyrene-based chemosensor for picric acid—Fundamentals to smartphone device design. Anal. Chem. 2019, 91, 13244–13250. [Google Scholar] [CrossRef]
- Zang, L.; Liang, C.; Wang, Y.; Bu, W.; Sun, H.; Jiang, S. A highly specific pyrene-based fluorescent probe for hypochlorite and its application in cell imaging. Sens. Actuators B Chem. 2015, 211, 164–169. [Google Scholar] [CrossRef]
- Guo, Y.; Wang, L.; Zhuo, J.; Xu, B.; Li, X.; Zhang, J.; Zhang, Z.; Chi, H.; Dong, Y.; Lu, G. A pyrene-based dual chemosensor for colorimetric detection of Cu2+ and fluorescent detection of Fe3+. Tetrahedron Lett. 2017, 58, 3951–3956. [Google Scholar] [CrossRef]
- Shen, T.; Jiang, N.; Zhang, X.; Lang, X.H.; Sun, J.Z.; Zhao, H. Probing Side Chain Dynamics of Branched Macromolecules by Pyrene Excimer Fluorescence. Polymers 2019, 11, 1366. [Google Scholar] [CrossRef] [PubMed]
- Mitra, K.; Singh, S.; Hira, S.K.; Patel, V.K.; Singh, D.; Vishwakarma, S.; Singh, R.; Kumari, A.; Manna, P.P.; Ray, B. A Fluorescent Thermometer Based on a Pyrene-Labeled Thermoresponsive Polymer. ACS Biomater. Sci. Eng. 2016, 2, 1630–1640. [Google Scholar] [CrossRef]
- Farhangi, S.; Duhamel, J. Probing side chain dynamics of branched macromolecules by pyrene excimer fluorescence. Macromolecules 2016, 49, 353–361. [Google Scholar] [CrossRef]
- Ihara, E.; Okada, R.; Sogai, T.; Asano, T.; Kida, M.; Inoue, K.; Itoh, T.; Shimomoto, T.; Ishibashi, Y.; Asahi, T. Pd-mediated polymerization of diazoacetates with aromatic ester group: Synthesis and photophysical property of poly(1-pyrenylmethoxycarbonylmethylene). Polym. Chem. 2013, 5, 1020–1023. [Google Scholar] [CrossRef]
- Pietsch, C.; Vollrath, A.; Hoogenboom, R.; Schubert, U.S. A Fluorescent Thermometer Based on a Pyrene-Labeled Thermoresponsive Polymer. Sensors 2010, 10, 7979–7990. [Google Scholar] [CrossRef]
- Senthamizhan, A.; Celebioglu, A.; Bayir, S.; Gorur, M.; Doganci, E.; Yilmaz, F.; Uyar, T. Highly Fluorescent Pyrene-Functional Polystyrene Copolymer Nanofibers for Enhanced Sensing Performance of TNT. ACS Appl. Mater. Interfaces 2015, 7, 21038–21046. [Google Scholar] [CrossRef]
- Lo, M.Y.; Zhen, C.; Lauters, M.I.; Jabbour, G.E.; Sellinger, A. Organic−Inorganic Hybrids Based on Pyrene Functionalized Octavinylsilsesquioxane Cores for Application in OLEDs. J. Am. Chem. Soc. 2007, 129, 5808–5809. [Google Scholar] [CrossRef]
- Sellinger, A.; Tamaki, R.; Laine, R.M.; Ueno, K.; Tanabe, H.; Williams, E.; Jabbour, G.E. Heck coupling of haloaromatics with octavinylsilsesquioxane: Solution processable nanocomposites for application in electroluminescent devices. Chem. Commun. 2005, 3700–3702. [Google Scholar] [CrossRef]
- Dong, Y.; Yang, Z.; Ren, Z.; Yan, S. Synthesis and the aggregation induced enhanced emission effect of pyrene based polysiloxanes. Polym. Chem. 2015, 6, 7827–7832. [Google Scholar] [CrossRef]
- Nowacka, M.; Kowalewska, A.; Plażuk, D.; Makowski, T. Hybrid polysilsesquioxanes for fluorescence resonance energy transfer. Dyes Pigment. 2019, 170, 107622. [Google Scholar] [CrossRef]
- Kowalewska, A.; Nowacka, M. Synthesis of ladder silsesquioxanes by in situ polycondensation of cyclic tetravinylsiloxanetetraols. Silicon 2015, 7, 133–146. [Google Scholar] [CrossRef]
- Kowalewska, A.; Nowacka, M.; Tracz, A.; Makowski, T. Supramolecular self-assembly of linear oligosilsesquioxanes on mica—AFM surface imaging and hydrophilicity studies. Soft Matter 2015, 11, 4818–4829. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, P.S.G.; He, C.; Shang, C.T. Synthesis, characterization, and curing kinetics of novel ladder-like polysilsesquioxanes containing side-chain maleimide groups. J. Polym. Sci. Polym. Chem. 2004, 42, 4036–4046. [Google Scholar] [CrossRef]
- Dittmar, U.; Hendan, B.J.; Florke, U.; Marsmann, H.C. Funktionalisierte octa-(propylsilsesquioxane) (3-XC3H6)8 (Si8O12) modellverbindungen für oberflächenmodifizierte kieselgele. J. Organomet. Chem. 1995, 489, 185–194. [Google Scholar] [CrossRef]
- Slater, M.D.; Frechet, J.M.J.; Svec, F. In-column preparation of a brush-type chiral stationary phase using click chemistry and a silica monolith. J. Sep. Sci. 2009, 32, 21–28. [Google Scholar] [CrossRef]
- Armarego, W.L.F.; Chai, C.L.L. Purification of Laboratory Chemicals, 5th ed.; Elsevier Science: Bodmin, UK, 2003. [Google Scholar]
- Hypercube, Inc. HyperChem (TM); Version Professional 7.51; Hypercube, Inc.: Gainesville, FL, USA, 2019. [Google Scholar]
- Chi, H.; Mya, K.Y.; Lin, T.; He, C.; Wang, F.; Chin, W.S. Thermally stable azobenzenedyes through hybridization with POSS. New J. Chem. 2013, 37, 735–742. [Google Scholar] [CrossRef]
- Dong, F.; Ha, C.S. Multifunctional materials based on polysilsesquioxanes. Macromol. Res. 2012, 20, 335–343. [Google Scholar] [CrossRef]
- Nowacka, M.; Fischer, C.; Kowalewska, A.; Hebda, M.; Hodor, K. Thermally induced phenomena leading to degradation of poly (silsesquioxane) materials. Eur. Polym. J. 2017, 86, 17–28. [Google Scholar] [CrossRef]
- Ervithayasuporn, V.; Wang, X.; Kawakami, Y. Synthesis and characterization of highly pure azido-functionalized polyhedral oligomeric silsesquioxanes (POSS). Chem. Commun. 2009, 34, 5130–5132. [Google Scholar] [CrossRef]
- Mittal, K.L. Polyimides and Other High Temperature Polymers: Synthesis, Characterization and Applications; CRC Press: Boca Raton, FL, USA, 2001; Volume 1. [Google Scholar]
- Bräse, S.; Gil, C.; Knepper, K.; Zimmermann, V. Organic azides: An exploding diversity of a unique class of compounds. Angew. Chem. Int. Ed. 2005, 44, 5188–5240. [Google Scholar] [CrossRef]
- L’abbe, G. Decomposition and addition reactions of organic azides. Chem. Rev. 1969, 69, 345–363. [Google Scholar] [CrossRef]
- Trastoy, B.; Pérez-Ojeda, M.E.; Sastre, R.; Chiara, J.L. Octakis (3-azidopropyl) octasilsesquioxane: A versatile nanobuilding block for the efficient preparation of highly functionalized cube-octameric polyhedral oligosilsesquioxane frameworks through click assembly. Chem. Eur. J. 2010, 16, 3833–3841. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, D.S.; Maraldi, V.A.; Dias, F.N.L.; do Carmo, D.R. Reactivity of a silsesquioxane organofunctionalized with 4-Amino-5-Phenyl-4H-[1,2,4]-Triazole-3-thiol: Complementary characterization and an application to chronoamperometric detection of L-dopamine. Silicon 2019, 11, 1131–1142. [Google Scholar] [CrossRef]
- Cordes, D.B.; Lickiss, P.D.; Rataboul, F. Recent developments in the chemistry of cubic polyhedral oligosilsesquioxanes. Chem. Rev. 2010, 110, 2081–2173. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.H.; Thomas, P.; Hume, P.; Jin, J. Effective conversion of amide to carboxylic acid on Polymers of Intrinsic Microporosity (PIM-1) with nitrous acid. Membranes 2018, 8, 20. [Google Scholar] [CrossRef]
- Ervithayasuporn, V.; Chimjarn, S. Synthesis and isolation of methacrylate and acrylate-functionalized polyhedral oligomeric silsesquioxanes (T8, T10, and T12) and characterization of the relationship between their chemical structures and physical properties. Inorg. Chem. 2013, 52, 13108–13112. [Google Scholar] [CrossRef]
- Kowalewska, A.; Nowacka, M.; Włodarska, M.; Zgardzińska, B.; Zaleski, R.; Oszajca, M.; Krajenta, J.; Kaźmierski, S. Solid-state dynamics and single-crystal to single-crystal structural transformations in octakis (3-chloropropyl) octasilsesquioxane and octavinyloctasilsesquioxane. Phys. Chem. Chem. Phys. 2017, 19, 27516–27529. [Google Scholar] [CrossRef]
- Chimjarn, S.; Kunthom, R.; Chancharone, P.; Sodkhomkhum, R.; Sangtrirutnugul, P.; Ervithayasuporn, V. Synthesis of aromatic functionalized cage-rearranged silsesquioxanes (T8, T10, and T12) via nucleophilic substitution reactions. Dalton Trans. 2015, 44, 916–919. [Google Scholar] [CrossRef]
- Kopesky, E.T.; Boyes, S.G.; Treat, N.; Cohen, R.E.; McKinley, G.H. Thermorheological properties near the glass transition of oligomeric poly (methyl methacrylate) blended with acrylic polyhedral oligomeric silsesquioxane nanocages. Rheol. Acta 2006, 45, 971–981. [Google Scholar] [CrossRef][Green Version]
- Cheng, C.-C.; Chang, F.-C.; Dai, S.A.; Lin, Y.-L.; Lee, D.-J. Bio-complementary supramolecular polymers with effective self-healing functionality. RSC Adv. 2015, 5, 90466–90472. [Google Scholar] [CrossRef]
- Kowalewska, A.; Fortuniak, W.; Handke, B. New hybrid silsesquioxane materials with sterically hindered carbosilane side groups. J. Organomet. Chem. 2009, 694, 1345–1353. [Google Scholar] [CrossRef]
- Nowacka, M.; Kowalewska, A.; Makowski, T. Structural studies on ladder phenylsilsesquioxane oligomers formed by polycondensation of cyclotetrasiloxanetetraols. Polymer 2016, 87, 81–89. [Google Scholar] [CrossRef]
- Unno, M.; Akiko, S.; Keisuke, T.; Hideyuki, M. Synthesis of Ladder and Cage Silsesquioxanes from 1,2,3,4-Tetrahydroxycyclotetrasiloxane. Bull. Chem. Soc. Jpn. 2000, 73, 215–220. [Google Scholar]
- Ge, Y.; Wen, Y.; Liu, H.; Lu, T.; Yu, Y.; Zhang, X.; Li, B.; Zhang, S.-T.; Li, W.; Yang, B. A key stacking factor for the effective formation of pyrene excimer in crystals: Degree of π–π overlap. J. Mater. Chem. C 2020, 8, 11830–11838. [Google Scholar] [CrossRef]
- Makal, A.; Krzeszczakowska, J.; Gajda, R. Pressure-dependent structural and luminescence properties of 1-(Pyren-1-yl) but-2-yn-1-one. Molecules 2019, 24, 1107. [Google Scholar] [CrossRef]
- Ishida, Y.; Tada, Y.; Hirai, T.; Goseki, R.; Kakimoto, M.; Yoshida, H.; Hayakawa, T. Directed self-assembly of cage silsesquioxane containing block copolymers via graphoepitaxy techniques. J. Photopolym. Sci. Technol. 2010, 23, 155–159. [Google Scholar] [CrossRef][Green Version]
- Stark, R.W.; Heckl, W.H. Fourier transformed atomic force microscopy: Tapping mode atomic force microscopy beyond the Hookian approximation. Surf. Sci. 2000, 457, 219–228. [Google Scholar] [CrossRef]
- Bowen, W.R.; Doneva, T.A. Artefacts in AFM studies of membranes: Correcting pore images using fast fourier transform filtering. J. Membr. Sci. 2000, 171, 141–147. [Google Scholar] [CrossRef]
- Silva, N.J.; Machado, F.B.C.; Lischka, H.; Aquino, A.J.A. π–π stacking between polyaromatic hydrocarbon sheets beyond dispersion interactions. Phys. Chem. Chem. Phys. 2016, 18, 22300–22310. [Google Scholar] [CrossRef]
- Duhamel, J. New insights in the study of pyrene excimer fluorescence to characterize macromolecules and their supramolecular assemblies in solution. Langmuir 2012, 28, 6527–6538. [Google Scholar] [CrossRef]
- Birks, J.B. Excimers. Rep. Prog. Phys. 1975, 38, 903–974. [Google Scholar] [CrossRef]
- Fujii, A.; Sekiguchi, Y.; Matsumura, H.; Inoue, T.; Chung, W.S.; Hirota, S.; Matsuo, T. Excimer emission properties on pyrene-labeled protein surface: Correlation between emission spectra, ring stacking modes, and flexibilities of pyrene probes. Bioconjug. Chem. 2015, 26, 537–548. [Google Scholar] [CrossRef] [PubMed]
- Niko, Y.; Hiroshige, Y.; Kawauchi, S.; Konishi, G. Additional insights into luminescence process of polycyclic aromatic hydrocarbons with carbonyl groups: Photophysical properties of secondary N-Alkyl and tertiary N, N-Dialkyl carboxamides of naphthalene, anthracene, and pyrene. J. Org. Chem. 2012, 77, 3986–3996. [Google Scholar] [CrossRef] [PubMed]
- Wrona-Piotrowicz, A.; Zakrzewski, J.; Métivier, R.; Brosseau, A.; Makal, A.; Woźniak, K. Efficient synthesis of pyrene-1-carbothioamides and carboxamides. Tunable solid-state fluorescence of pyrene-1-carboxamides. RSC Adv. 2014, 4, 56003–56012. [Google Scholar] [CrossRef]
- Lekha, P.K.; Ghosh, T.; Prasad, E. Utilizing dendritic scaffold for feasible formation of naphthalene excimer. J. Chem. Sci. 2011, 123, 919–926. [Google Scholar] [CrossRef]
- Grigalevicius, S.; Forster, M.; Ellinger, S.; Landfester, K.; Scherf, U. Excitation energy transfer from semi-conducting polymer nanoparticles to surface-bound fluorescent dyes. Macromol. Rapid Commun. 2006, 27, 200–202. [Google Scholar] [CrossRef]
- Bhattacharyya, S.; Jana, B.; Patra, A. Multichromophoric organic molecules encapsulated in polymer nanoparticles for artificial light harvesting. ChemPhysChem 2015, 16, 796–804. [Google Scholar] [CrossRef]
- Chai, J.; Wu, Y.; Yang, B.; Liu, B. The photochromism, light harvesting and self-assembly activity of a multi-function schiff-base compound based on the AIE effect. J. Mater. Chem. C 2018, 6, 4057–4064. [Google Scholar] [CrossRef]
- Dietz, E.M. Chlorophyll and hemoglobin—Two natural pyrrole pigments. J. Chem. Educ. 1935, 12, 208. [Google Scholar] [CrossRef]
- Sadiq, S.; Ghazala, Z.; Chowdhury, A.; Busselberg, D. Metal toxicity at the synapse: Presynaptic, postsynaptic, and long-term effects. J. Toxicol. 2011, 2012, 132671. [Google Scholar] [CrossRef]
- Thirupathi, P.; Lee, K.H. A ratiometric fluorescent detection of Zn (II) in aqueous solutions using pyrene-appended histidine. Bioorg. Med. Chem. Lett. 2013, 23, 6811–6815. [Google Scholar] [CrossRef] [PubMed]
- Raj, T.; Saluja, P.; Singh, N. A new class of pyrene based multifunctional chemosensors for differential sensing of metals in different media: Selective recognition of Zn2+ in organic and Fe3+ in aqueous medium. Sens. Actuators B 2015, 206, 98–106. [Google Scholar] [CrossRef]
- Chung, P.K.; Liu, S.R.; Wang, H.F.; Wu, S.P.J. A Pyrene-based highly selective turn-on fluorescent chemosensor for iron (III) ions and its application in living cell imaging. J. Fluoresc. 2013, 23, 1139–111145. [Google Scholar] [CrossRef] [PubMed]
- Manandhar, E.; Wallace, K.J. Host–guest chemistry of pyrene-based molecular receptors. Inorg. Chim. Acta 2012, 381, 15–43. [Google Scholar] [CrossRef]
- Formica, M.; Fusi, V.; Giorgi, L.; Micheloni, M. New fluorescent chemosensors for metal ions in solution. Coord. Chem. Rev. 2012, 256, 170–192. [Google Scholar] [CrossRef]
- Parker, D.; Williams, J.A.G. Luminescence behaviour of cadmium, lead, zinc, copper, nickel and lanthanide complexes of octadentate macrocyclic ligands bearing naphthyl chromophores. J. Chem. Soc. Perkin Trans. 1995, 2, 1305–1314. [Google Scholar] [CrossRef]
- Bergonzia, R.; Fabbrizzi, L.; Licchellia, M.; Manganoa, C. Molecular switches of fluorescence operating through metal centred redox couples. Coord. Chem. Rev. 1998, 170, 31–46. [Google Scholar] [CrossRef]
- Fages, F.; Bodenant, B.; Weil, T. Fluorescent, siderophore-based chelators. design and synthesis of a trispyrenyl trishydroxamate ligand, an intramolecular excimer-forming sensing molecule which responds to iron (III) and gallium (III) metal cations. J. Org. Chem. 1996, 61, 3956–3961. [Google Scholar] [CrossRef]
- Maity, D.; Raj, A.; Karthigeyan, D.; Kundu, T.K.; Govindaraju, T. Reaction-based probes for Co (II) and Cu (I) with dual output modes: Fluorescence live cell imaging. RSC Adv. 2013, 3, 16788–16794. [Google Scholar] [CrossRef]
- Reimann, C.; Koller, F.; Kashulina, G.; Englmaier, H.P. Influence of extreme pollution on the inorganic chemical composition of some plants. Environ. Pollut. 2001, 115, 239–252. [Google Scholar] [CrossRef]
- Zhu, M.; Wang, W.; Liu, J.; Na, R.; Li, Z.; Wang, Y. A novel pyrene-based fluorescent probe for the rapid and efficient detection of Co2+ in HeLa cells and natural water samples. J. Mol. Liq. 2020, 303, 112680. [Google Scholar] [CrossRef]
- Mohandoss, S.; Stalin, T. A new fluorescent PET sensor probe for Co2+ ion detection: Computational, logic device and living cell imaging applications. RSC Adv. 2017, 7, 16581–16593. [Google Scholar] [CrossRef]
- Shiraishi, Y.; Ishizumi, K.; Nishimura, G.; Hirai, T. Effects of metal cation coordination on fluorescence properties of a diethylenetriamine bearing two end pyrene fragments. J. Phys. Chem. B 2007, 111, 8812–8822. [Google Scholar] [CrossRef] [PubMed]
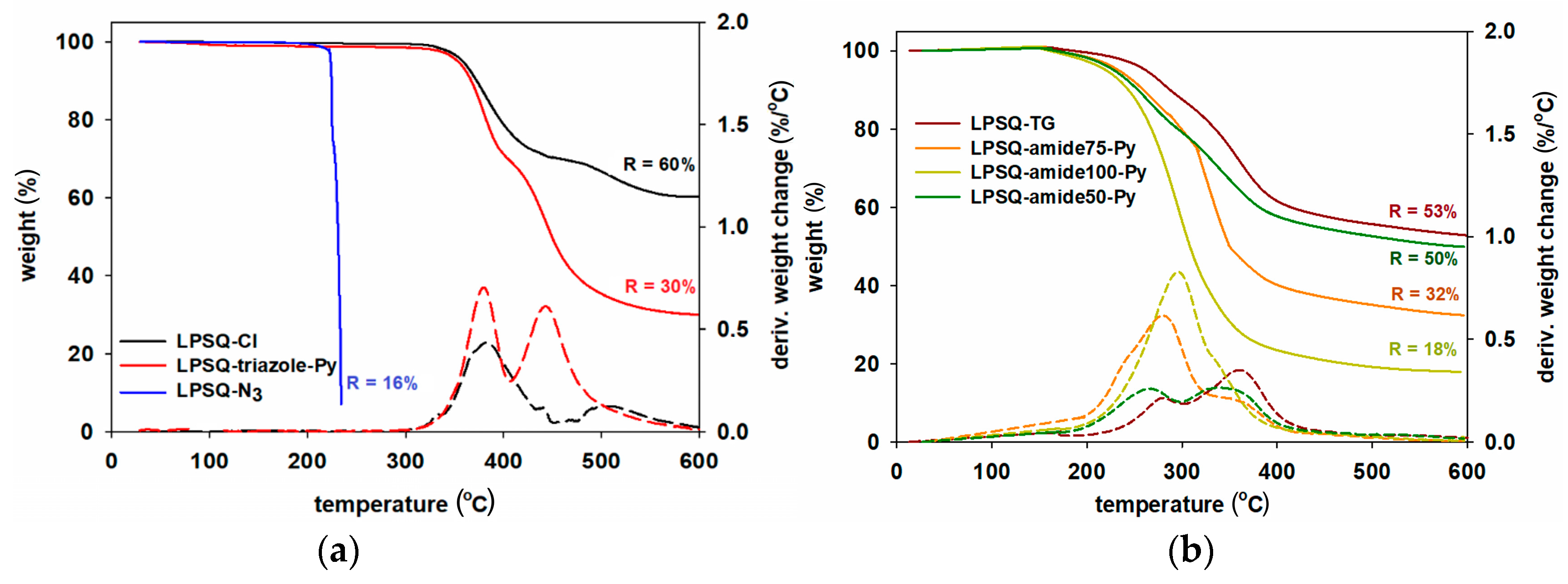

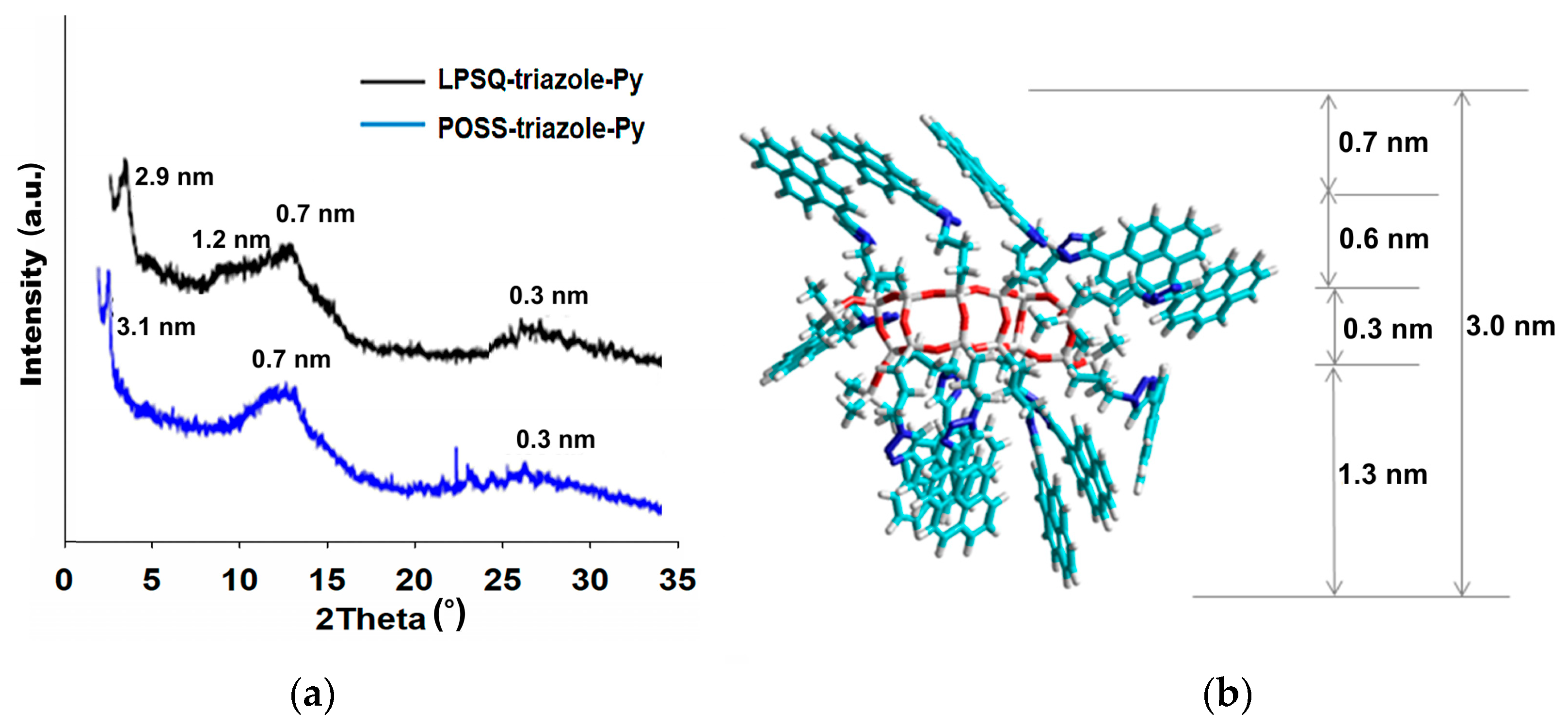
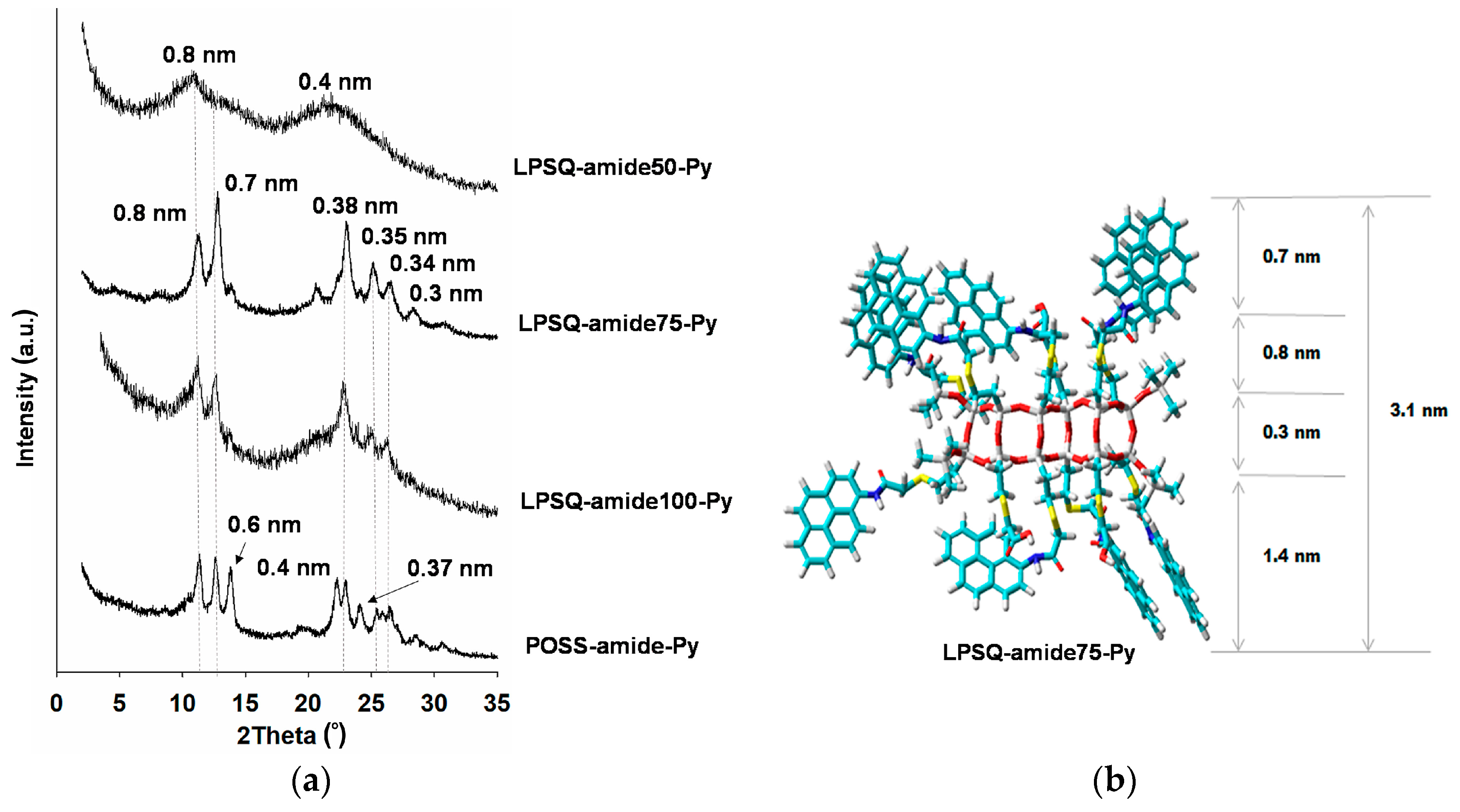


| Sample | Td1 (°C) | Td2 (°C) | Rate (%∙min/°C) | T5% (°C) | Residue (%) | Residue Calculated for SiO2 (%) | |
|---|---|---|---|---|---|---|---|
| V1 | V2 | ||||||
| LPSQ-Cl | 382.2 | 509.6 | 2.90 | 1.09 | 358 | 60.2 | 46.2 |
| LPSQ-N3 | – | – | – | – | 223 | 16.2 | 44.1 |
| LPSQ-triazole-Py | 379.7 | 443.0 | 2.86 | 3.78 | 353 | 30.0 | 16.5 |
| POSS-Cl | 358.2 | 502.1 | 5.56 | 0.63 | 358 | 37.3 | 46.3 |
| POSS-N3 | – | – | – | – | 235 | 12.7 | 44.1 |
| POSS-triazole-Py | 376.7 | 434.7 | 2.23 | 1.73 | 352 | 57.5 | 16.6 |
| silane-triazole-Py | 365.3 | 403.0 | 7.35 | 1.11 | 213 | 10.2 | 16.5 |
| Sample | Td1 (°C) | Td2 (°C) | Rate (%∙min/°C) | T5% (°C) | Residue (%) | Residue Calculated for SiO3/2 (%) | |
|---|---|---|---|---|---|---|---|
| Vd1 | Vd2 | ||||||
| LPSQ-TG | 279.5 | 362.6 | 1.99 | 3.12 | 262 | 53.1 | 32.0 |
| LPSQ-amide50-Py | 264.8 | 386.9 | 1.89 | 2.53 | 229 | 49.8 | 19.2 |
| LPSQ-amide75-Py | 279.8 | - | 6.36 | - | 233 | 31.9 | 16.4 |
| LPSQ-amide100-Py | 298.6 | 467.0 | 7.52 | 0.25 | 220 | 18.2 | 14.3 |
| POSS-TG | 380.0 | - | 1.72 | - | 190 | 47.1 | 27.8 |
| POSS-amide-Py | 303.4 | - | 7.54 | - | 225 | 20.0 | 13.4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nowacka, M.; Makowski, T.; Kowalewska, A. Hybrid Fluorescent Poly(silsesquioxanes) with Amide- and Triazole-Containing Side Groups for Light Harvesting and Cation Sensing. Materials 2020, 13, 4491. https://doi.org/10.3390/ma13204491
Nowacka M, Makowski T, Kowalewska A. Hybrid Fluorescent Poly(silsesquioxanes) with Amide- and Triazole-Containing Side Groups for Light Harvesting and Cation Sensing. Materials. 2020; 13(20):4491. https://doi.org/10.3390/ma13204491
Chicago/Turabian StyleNowacka, Maria, Tomasz Makowski, and Anna Kowalewska. 2020. "Hybrid Fluorescent Poly(silsesquioxanes) with Amide- and Triazole-Containing Side Groups for Light Harvesting and Cation Sensing" Materials 13, no. 20: 4491. https://doi.org/10.3390/ma13204491
APA StyleNowacka, M., Makowski, T., & Kowalewska, A. (2020). Hybrid Fluorescent Poly(silsesquioxanes) with Amide- and Triazole-Containing Side Groups for Light Harvesting and Cation Sensing. Materials, 13(20), 4491. https://doi.org/10.3390/ma13204491





