Diffusion Reflection Measurements of Antibodies Conjugated to Gold Nanoparticles as a Method to Identify Cutaneous Squamous Cell Carcinoma Borders
Abstract
1. Introduction
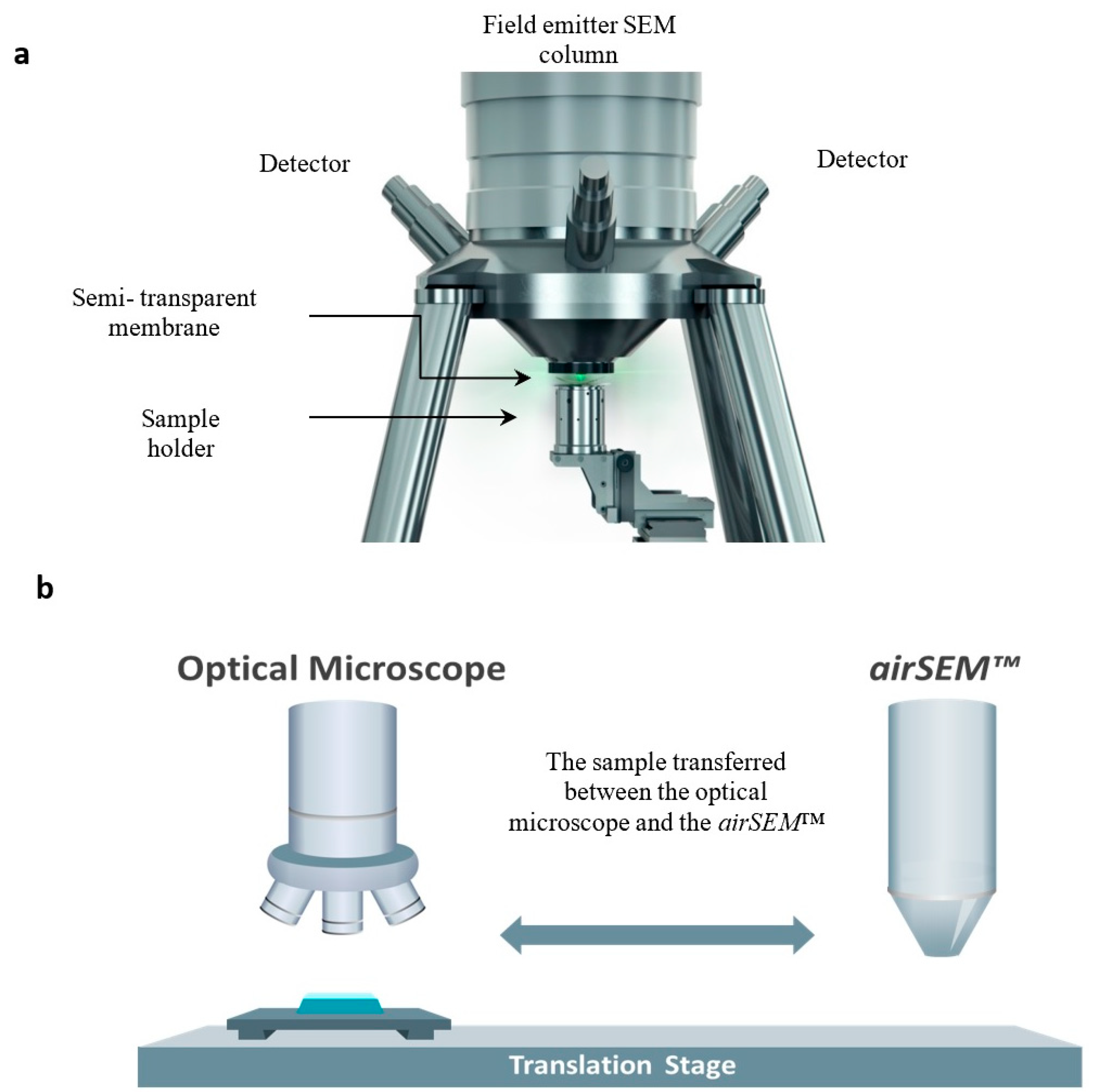
2. Materials and Methods
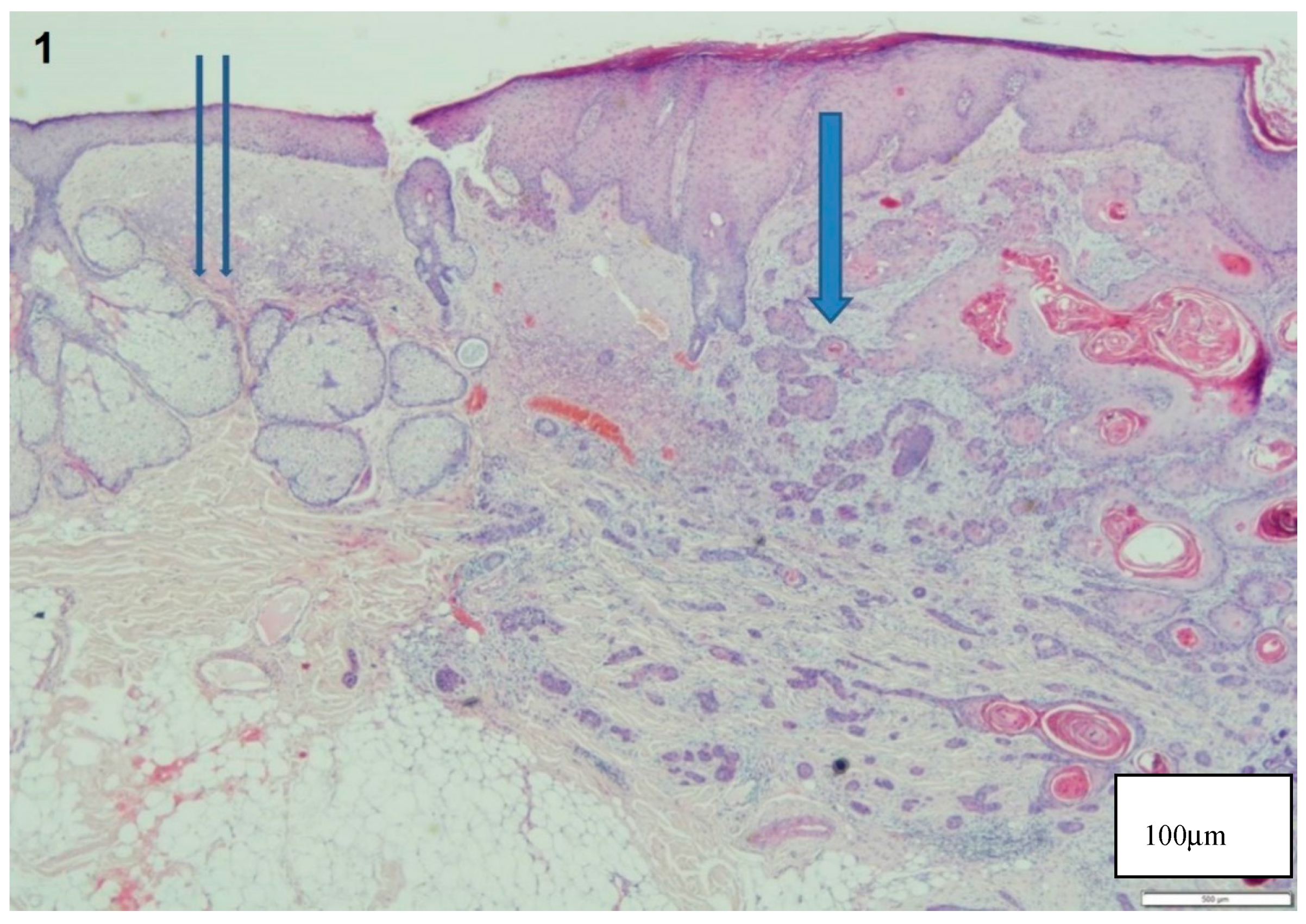
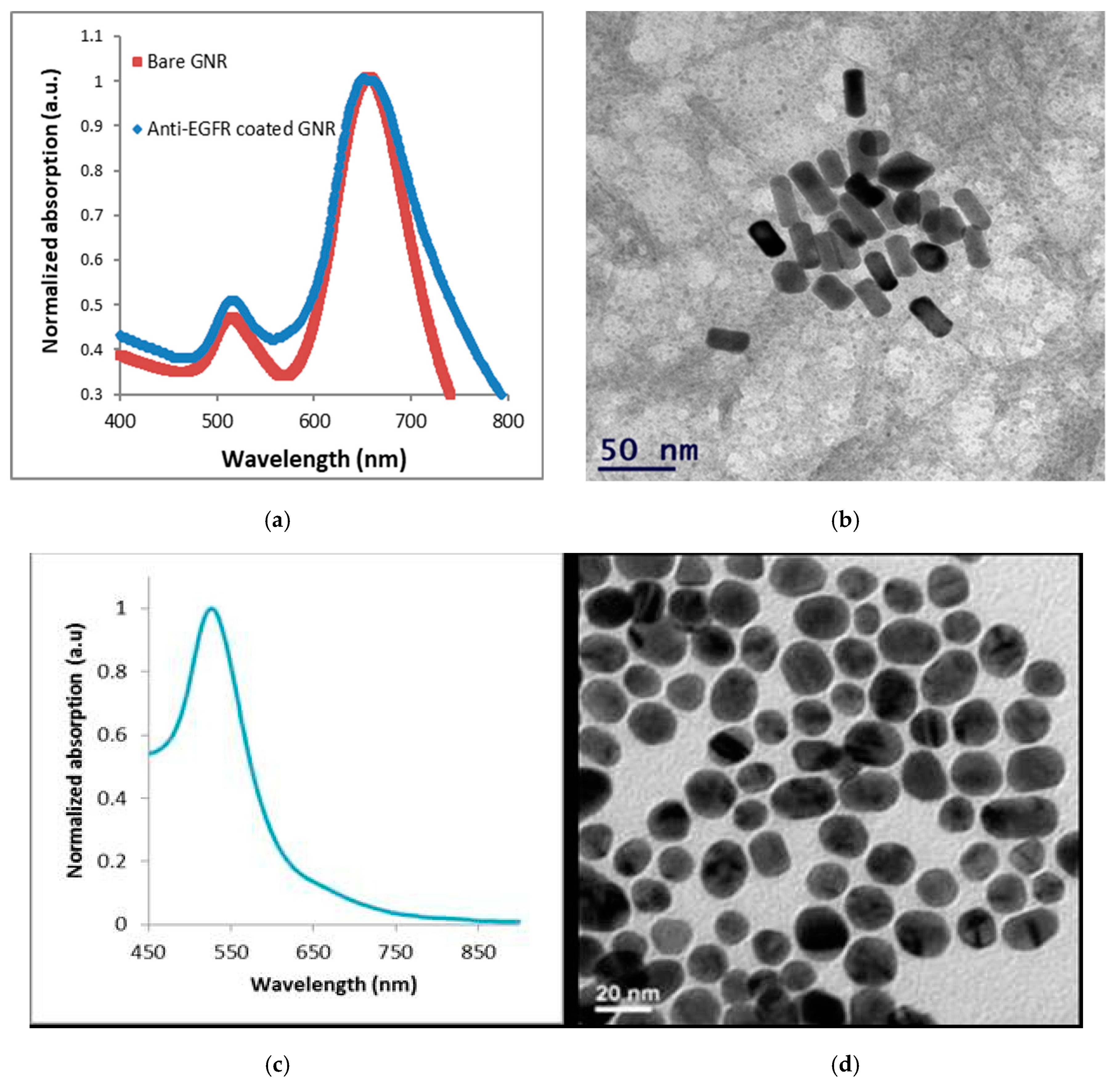
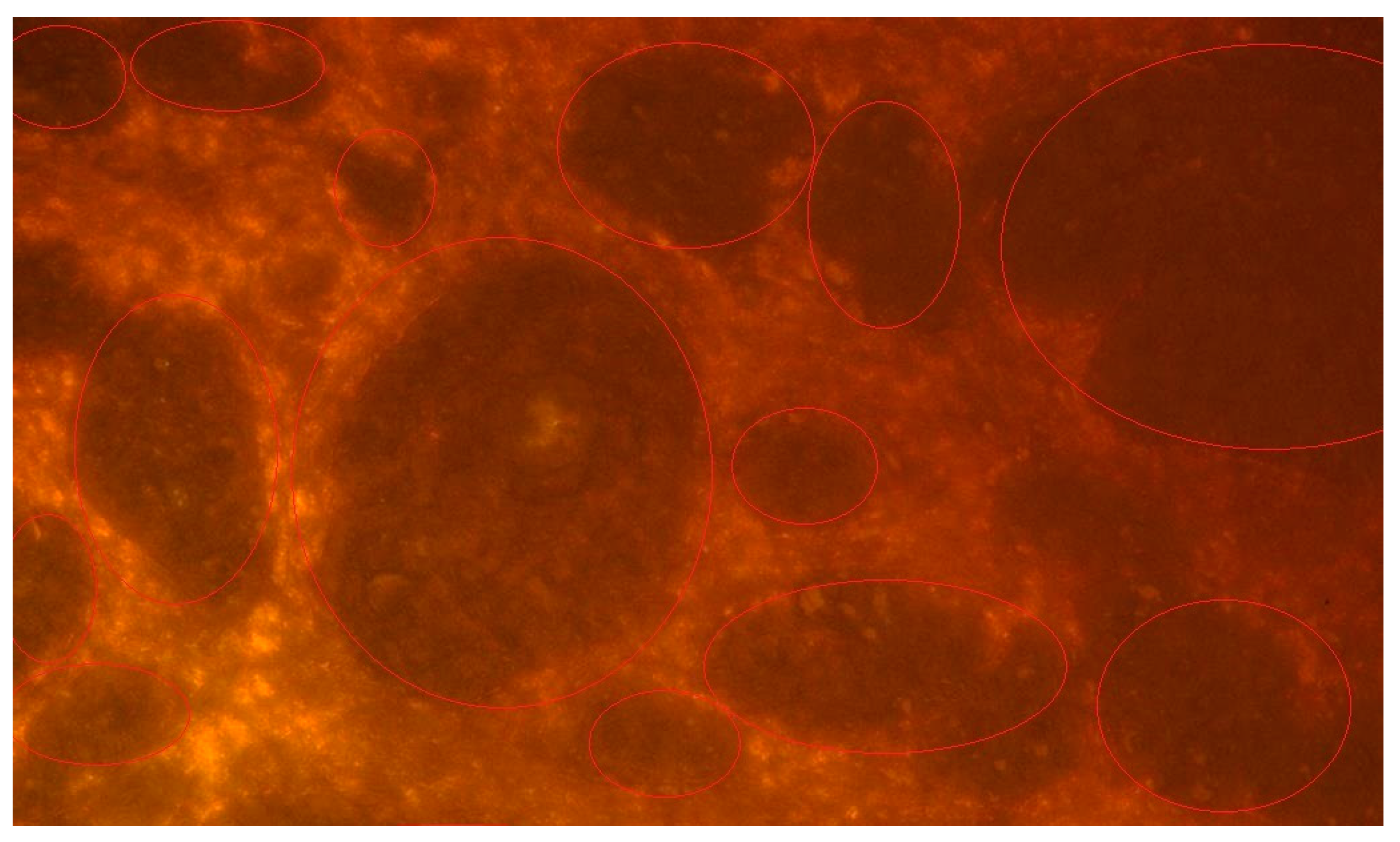
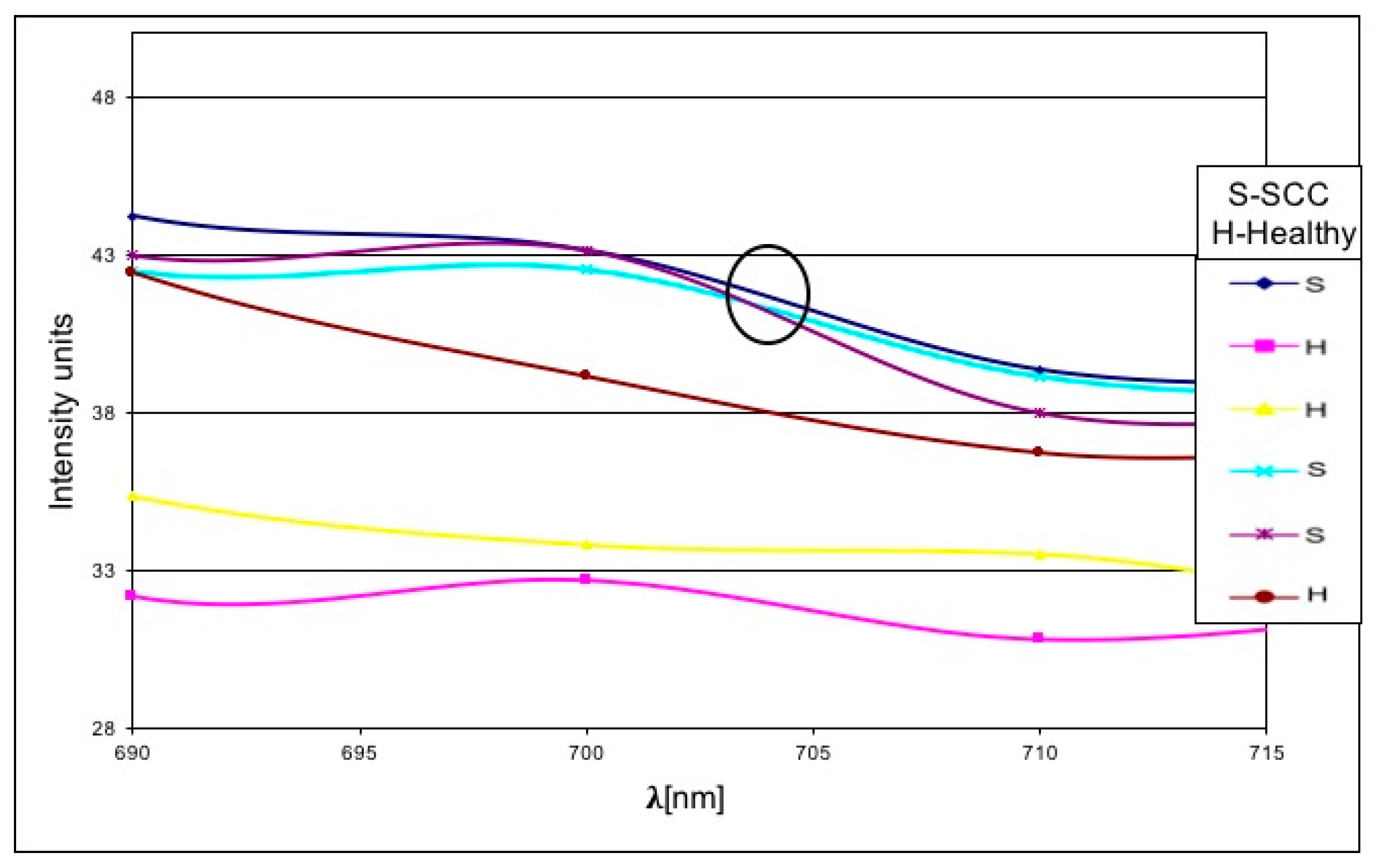
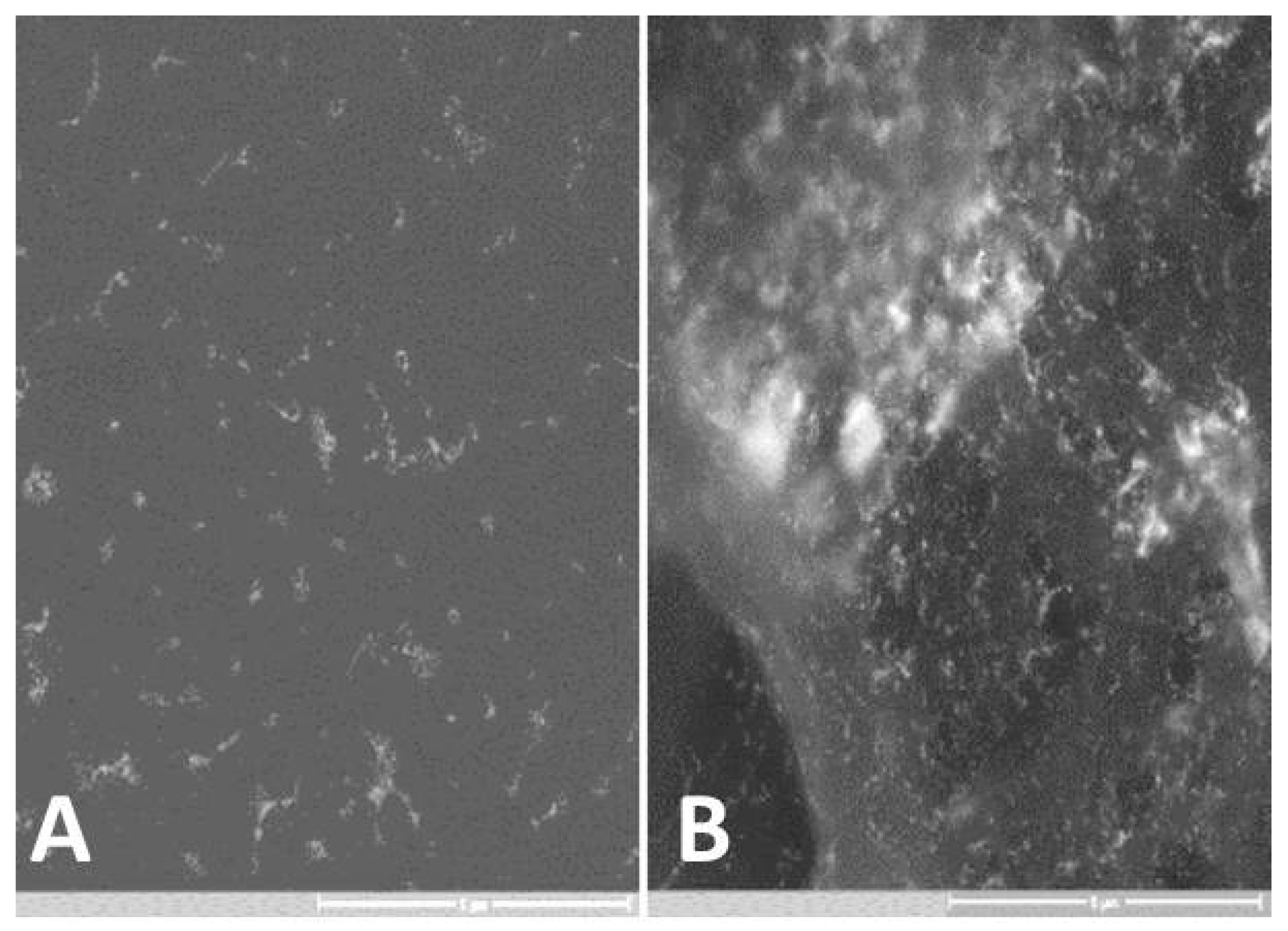
3. Results
4. Discussion
5. Conclusions
6. Summary Points
- Squamous cell carcinoma (SCC) is the second most frequent type of skin cancer.
- SCC is considered to be biologically aggressive, with a 12% rate of metastasis, especially to the lymph nodes.
- The main factor determining survival is completeness of the surgical resection.
- SCC is characterized by overexpression of epidermal growth factor receptor (EGFR).
- GNPs are a promising agent for the diagnosis and treatment of carcinoma, and, in this case, were conjugated to EGFR.
- This study investigated the ability to detect residual SCC stained with gold nanoparticles using diffusion reflectance.
- This study verifies the use of nanotechnology in identifying SCC tumor margins.
- The noninvasive, nonionizing direct diffusion reflection scanning has high sensitivity for distinguishing cancerous tissue histologically.
- Diffusion reflection is a promising tool for the intraoperative identification of residual disease in SCC excision borders.
Author Contributions
Funding
Conflicts of Interest
References
- Parkin, D.M.; Pisani, P.; Ferlay, J. Global cancer statistics. CA Cancer J. Clin. 1999, 49, 33–64. [Google Scholar] [CrossRef] [PubMed]
- Uribe, P.; Gonzalez, S. Epidermal growth factor receptor (EGFR) and squamous cell carcinoma of the skin: Molecular bases for EGFR-targeted therapy. Pathol. Res. Pract. 2011, 207, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Aubry, F.; MacGibbon, B. Risk factors of squamous cell carcinoma of the skin. A case-control study in the Montreal region. Cancer 1985, 55, 907–911. [Google Scholar] [CrossRef]
- Vitasa, B.C.; Taylor, H.R.; Strickland, P.T.; Rosenthal, F.S.; West, S.; Abbey, H.; Ng, S.K.; Munoz, B.; Emmett, E.A. Association of nonmelanoma skin cancer and actinic keratosis with cumulative solar ultraviolet exposure in Maryland watermen. Cancer 1990, 65, 2811–2817. [Google Scholar] [CrossRef]
- Jesse, R.H.; Sugarbaker, E.V. Squamous cell carcinoma of the oropharynx: Why we fail. Am. J. Surg. 1976, 132, 435–438. [Google Scholar] [CrossRef]
- Kowalski, L.P.; Magrin, J.; Waksman, G.; Santo, G.F.; Lopes, M.E.; de Paula, R.P.; Pereira, R.N.; Torloni, H. Supraomohyoid neck dissection in the treatment of head and neck tumors. Survival results in 212 cases. Arch. Otolaryngol. Head Neck Surg. 1993, 119, 958–963. [Google Scholar] [CrossRef]
- DiNardo, L.J.; Lin, J.; Karageorge, L.S.; Powers, C.N. Accuracy, utility, and cost of frozen section margins in head and neck cancer surgery. Laryngoscope 2000, 110, 1773–1776. [Google Scholar] [CrossRef]
- Davidson, T.M.; Haghighi, P.; Astarita, R.; Baird, S.; Seagren, S. MOHS for head and neck mucosal cancer: Report on 111 patients. Laryngoscope 1988, 98, 1078–1083. [Google Scholar] [CrossRef]
- Chen, T.Y.; Emrich, L.J.; Driscoll, D.L. The clinical significance of pathological findings in surgically resected margins of the primary tumor in head and neck carcinoma. Int. J. Radiat. Oncol. Biol. Phys. 1987, 13, 833–837. [Google Scholar] [CrossRef]
- Cooper, J.S.; Pajak, T.F.; Forastiere, A.A.; Jacobs, J.; Campbell, B.H.; Saxman, S.B.; Kish, J.A.; Kim, H.E.; Cmelak, A.J.; Rotman, M.; et al. Radiation Therapy Oncology Group 9501/Intergroup. Postoperative concurrent radiotherapy and chemotherapy for high-risk squamous-cell carcinoma of the head and neck. N. Engl. J. Med. 2004, 350, 1937–1944. [Google Scholar] [CrossRef]
- Bernier, J.; Cooper, J.S.; Pajak, T.F.; van Glabbeke, M.; Bourhis, J.; Forastiere, A.; Ozsahin, E.M.; Jacobs, J.R.; Jassem, J.; Ang, K.K.; et al. Defining risk levels in locally advanced head and neck cancers: A comparative analysis of concurrent postoperative radiation plus chemotherapy trials of the EORTC (#22931) and RTOG (#9501). Head Neck 2005, 27, 843–850. [Google Scholar] [PubMed]
- Serewko, M.M.; Popa, C.; Dahler, A.L.; Smith, L.; Strutton, G.M.; Coman, W.; Dicker, A.J.; Saunders, N.A. Alterations in gene expression and activity during squamous cell carcinoma development. Cancer Res. 2002, 62, 3759–3765. [Google Scholar] [PubMed]
- Jost, M.; Kari, C.; Rodeck, U. The EGF receptor-an essential regulator of multiple epidermal functions. Eur. J. Dermatol. 2000, 10, 505–510. [Google Scholar] [PubMed]
- Ullrich, A.; Coussens, L.; Hayflick, J.S.; Dull, T.J.; Gray, A.; Tam, A.W.; Lee, J.; Yarden, Y.; Libermann, T.A.; Schlessinger, J.; et al. Human epidermal growth factor receptor cDNA sequence and aberrant expression of the amplified gene in A431 epidermoid carcinoma cells. Nature 1984, 309, 418–425. [Google Scholar] [CrossRef] [PubMed]
- Maurizi, M.; Almadori, G.; Ferrandina, G.; Distefano, M.; Romanini, M.E.; Cadoni, G.; Benedetti-Panici, P.; Paludetti, G.; Scambia, G.; Mancuso, S. Prognostic significance of epidermal growth factor receptor in laryngeal squamous cell carcinoma. Br. J. Cancer 1996, 74, 1253–1257. [Google Scholar] [CrossRef]
- Mukaida, H.; Toi, M.; Hirai, T.; Yamashita, Y.; Toge, T. Clinical significance of the expression of epidermal growth factor and its receptor in esophageal cancer. Cancer 1991, 68, 142–148. [Google Scholar] [CrossRef]
- Ang, K.K.; Berkey, B.A.; Tu, X.; Zhang, H.Z.; Katz, R.; Hammond, E.H.; Fu, K.K.; Milas, L. Impact of epidermal growth factor receptor expression on survival and pattern of relapse in patients with advanced head and neck carcinoma. Cancer Res. 2002, 62, 7350–7356. [Google Scholar]
- Hansen, A.R.; Siu, L.L. Epidermal growth factor receptor targeting in head and neck cancer: Have we been just skimming the surface? J. Clin. Oncol. 2013, 31, 2013–2015. [Google Scholar] [CrossRef]
- Cempel, D.; Nguyen, M.T.; Ishida, Y.; Tsukamoto, H.; Shirai, H.; Wang, Y.; Wu, K.C.W.; Yonezwa, T. Au Nanoparticles Prepared Using a Coated Electrode in Plasma-in-Liquid Process: Effect of the Solution pH. J. Nanosci. Nanotechnol. 2016, 16, 9257–9262. [Google Scholar] [CrossRef]
- Liao, Y.T.; Chen, J.E.; Isida, Y.; Yonezawa, T.; Chang, W.C.; Alshehri, S.M.; Yamauchi, Y.; Wu, K.C.W. De Novo Synthesis of Gold-Nanoparticle-Embedded,Nitrogen-Doped Nanoporous Carbon Nanoparticles (Au@NC) with Enhanced Reduction Ability. ChemCatChem 2016, 8, 506–509. [Google Scholar]
- Shirai, T.; Huang, Y.Y.; Yonezawa, T.; Tokunaga, T.; Chang, W.C.; Alshehri, S.M.; Jiang, B.; Yamauchi, Y.; Wu, K.C.W. Hard-Templating Synthesis of Macroporous Platinum Microballs (MPtM). Mater. Lett. 2016, 164, 488–492. [Google Scholar] [CrossRef]
- Hilderbrand, S.A.; Weissleder, R. Near-infrared fluorescence: Application to in vivo molecular imaging. Curr. Opin. Chem. Biol. 2010, 14, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Copland, J.A.; Eghtedari, M.; Popov, V.L.; Kotov, N.; Mamedova, N.; Motamedi, M.; Oraevsky, A.A. Bioconjugated gold nanoparticles as a molecular based contrast agent: Implications for imaging of deep tumors using optoacoustic tomography. Mol. Imaging Biol. 2004, 6, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Popovtzer, R.; Agrawal, A.; Kotov, N.A.; Popovtzer, A.; Balter, J.; Carey, T.E.; Kopelman, R. Targeted gold nanoparticles enable molecular ct imaging of cancer. Nano Lett. 2008, 8, 4593–4596. [Google Scholar] [CrossRef]
- Ankri, R.; Melzer, S.; Tarnok, A.; Fixler, D. Detection of gold nanorods uptake by macrophages using scattering analyses combined with diffusion reflection measurements as a potential tool for in vivo atherosclerosis tracking. Int. J. Nanomed. 2015, 10, 4437–4446. [Google Scholar]
- Fixler, D.; Ankri, R.; Kaplan, I.; Novikov, I.; Hirshberg, A. Diffusion reflection: A novel method for detection of oral cancer. J. Dent. Res. 2014, 93, 602–606. [Google Scholar] [CrossRef]
- Ankri, R.; Leshem-Lev, D.; Fixler, D.; Popovtzer, R.; Motiei, M.; Kornowski, R.; Hochhauser, E.; Lev, I. Gold nanorods as absorption contrast agents for the noninvasive detection of arterial vascular disorders based on diffusion reflection measurements. Nano Lett. 2014, 14, 2681–2687. [Google Scholar] [CrossRef]
- Ankri, R.; Fixler, D. Gold nanorods based diffusion reflection measurements: Current status and perspectives for clinical applications. Nanophotonics 2017, 6, 1031–1042. [Google Scholar] [CrossRef]
- Reif, R.; A’Amar, O.; Bigio, I.J. Analytical model of light reflectance for extraction of the optical properties in small volumes of turbid media. Appl. Opt. 2007, 46, 7317–7328. [Google Scholar] [CrossRef]
- Jain, P.K.; Lee, K.S.; El-Sayed, I.H.; El-Sayed, M.A. Calculated absorption and scattering properties of gold nanoparticles of different size, shape, and composition: Applications in biological imaging and biomedicine. J. Phys. Chem. B 2006, 110, 7238–7248. [Google Scholar] [CrossRef]
- Chen, J.; Saeki, F.; Wiley, B.J.; Cang, H.; Cobb, M.J.; Li, Z.Y.; Au, L.; Zhang, H.; Kimmey, M.B.; Li, X.; et al. Gold nanocages: Bioconjugation and their potential use as optical imaging contrast agents. Nano Lett. 2005, 5, 473–477. [Google Scholar] [CrossRef] [PubMed]
- Eghtedari, M.; Liopo, A.V.; Copland, J.A.; Oraevsky, A.A.; Motamedi, M. Engineering of hetero-functional gold nanorods for the in vivo molecular targeting of breast cancer cells. Nano Lett. 2008, 9, 287–291. [Google Scholar] [CrossRef]
- Huang, X.; El-Sayed, I.H.; Qian, W.; El-Sayed, M.A. Cancer cell imaging and photothermal therapy in the near-infrared region by using gold nanorods. J. Am. Chem. Soc. 2006, 128, 2115–2120. [Google Scholar] [CrossRef]
- Enustun, B.V.; Turkevich, J. Coagulation of Colloidal Gold. J. Am. Chem. Soc. 1963, 85, 3317–3328. [Google Scholar] [CrossRef]
- Nikoobakht, B.; El-Sayed, M.A. Preparation and Growth Mechanism of Gold Nanorods (NRs) Using Seed-Mediated Growth Method. Chem. Mater. 2003, 15, 1957–1962. [Google Scholar] [CrossRef]
- Ai, H.; Fang, M.; Jones, S.A.; Lvov, Y.M. Electrostatic layer-by-layer nanoassembly on biological microtemplates: Platelets. Biomacromolecules 2002, 3, 560–564. [Google Scholar] [CrossRef] [PubMed]
- Daga, M.; Dianzani, C.; Ferrara, B.; Nardozza, V.; Cavalli, R.; Barrera, G.; Pizzimenti, S. Latest news on nanotechnology for melanoma therapy and diagnosis. J. Nanotechnol. Nanomed. 2016, 1, 1002–1015. [Google Scholar]
- Naves, L.B.; Dhand, C.; Venugopal, J.R.; Rajamani, L.; Ramakrishna, S.; Almeida, L. Nanotechnology for the treatment of melanoma skin cancer. Prog. Biomater. 2017, 6, 13–26. [Google Scholar] [CrossRef]
- Muldoon, T.J.; Anandasabapathy, S.; Maru, D.; Richards-Kortum, R. High-resolution imaging in Barrett’s esophagus: A novel, low-cost endoscopic microscope. Gastrointest. Endosc. 2008, 68, 737–744. [Google Scholar] [CrossRef][Green Version]
- Gareau, D.S.; Abeytunge, S.; Rajadhyaksha, M. Line-scanning reflectance confocal microscopy of human skin: Comparison of full-pupil and divided-pupil configurations. Opt. Lett. 2009, 34, 3235–3237. [Google Scholar] [CrossRef]
- Gleysteen, J.P.; Newman, J.R.; Chhieng, D.; Frost, A.; Zinn, K.R.; Rosenthal, E.L. Fluorescent labeled anti-EGFR antibody for identification of regional and distant metastasis in a preclinical xenograft model. Head Neck 2008, 30, 782–789. [Google Scholar] [CrossRef] [PubMed]
- Heath, C.H.; Deep, N.L.; Beck, L.N.; Day, K.E.; Sweeny, L.; Zinn, K.R.; Huang, C.C.; Rosenthal, E.L. Use of panitumumab-IRDye800 to image cutaneous head and neck cancer in mice. Otolaryngol. Head Neck Surg. 2013, 148, 982–990. [Google Scholar] [CrossRef] [PubMed]
- Orthaber, K.; Pristovnik, M.; Skok, K.; Perić, B.; Maver, U. Skin cancer and its treatment: Novel treatment approaches with emphasis on nanotechnology. J. Nanomater. 2017, 2017, 2606271. [Google Scholar] [CrossRef]
- Bernardi, R.J.; Lowery, A.R.; Thompson, P.A.; Blaney, S.M.; West, J.L. Immunonanoshells for targeted photothermal ablation in medulloblastoma and glioma: An in vitro evaluation using human cell lines. J. Neurooncol. 2008, 86, 165–172. [Google Scholar] [CrossRef]
- Setua, S.; Ouberai, M.; Piccirillo, S.G.; Watts, C.; Welland, M. Cisplatin-tethered gold nanospheres for multimodal chemo-radiotherapy of glioblastoma. Nanoscale 2014, 6, 10865–10873. [Google Scholar] [CrossRef]
- Agemy, L.; Friedmann-Morvisnski, D.; Kotamraju, V.R.; Roth, L.; Sugahara, K.N.; Girard, O.M.; Mattrey, R.F.; Verma, I.M.; Ruoslahti, E. Targeted nanoparticle enhanced proapoptotic peptide as potential therapy for glioblastoma. Proc. Natl. Acad. Sci. USA 2011, 108, 17450–17455. [Google Scholar] [CrossRef]
- Kim, W.; Na, K.Y.; Lee, K.H.; Lee, H.W.; Lee, J.K.; Kim, K.T. Selective uptake of epidermal growth factor-conjugated gold nanoparticle (EGF-GNP) facilitates non-thermal plasma (NTP)-mediated cell death. Sci. Rep. 2017, 7, 10971. [Google Scholar] [CrossRef]
- Yang, Y.; Celmer, E.J.; Koutcher, J.A.; Alfano, R.R. UV reflectance spectroscopy probes DNA and protein changes in human breast tissues. J. Clin. Laser Med. Surg. 2001, 19, 35–39. [Google Scholar] [CrossRef]
- Bigio, I.J.; Bown, S.G.; Briggs, G.; Kelley, C.; Lakhani, S.; Pickard, D.; Ripley, P.M.; Rose, I.G.; Saunders, C. Diagnosis of breast cancer using elastic-scattering spectroscopy: Preliminary clinical results. J. Biomed. Opt. 2000, 5, 221–228. [Google Scholar] [CrossRef]
- Zhu, C.; Palmer, G.M.; Breslin, T.M.; Harter, J.; Ramanujam, N. Diagnosis of breast cancer using diffuse reflectance spectroscopy: Comparison of a Monte Carlo versus partial least squares analysis based feature extraction technique. Lasers Surg. Med. 2006, 38, 714–724. [Google Scholar] [CrossRef]
- Stanton, P.; Richards, S.; Reeves, J.; Nikolic, M.; Edington, K.; Clark, L.; Robertson, G.; Souter, D.; Mitchell, R.; Hendler, F.J.; et al. Epidermal growth factor receptor expression by human squamous cell carcinomas of the head and neck, cell lines and xenografts. Br. J. Cancer 1994, 70, 427–433. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hirshberg, A.; Allon, I.; Novikov, I.; Ankri, R.; Ashkenazy, A.; Fixler, D. Gold nanorods reflectance discriminate benign from malignant oral lesions. Nanomedicine 2017, 13, 1333–1339. [Google Scholar] [CrossRef] [PubMed]
- Barnoy, E.A.; Fixler, D.; Popovtzer, R.; Nayhoz, T.; Ray, K. An ultra-sensitive dual-mode imaging system using metal-enhanced fluorescence in solid phantoms. Nano Res. 2015, 8, 3912–3921. [Google Scholar] [CrossRef] [PubMed]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olshinka, A.; Ad-El, D.; Didkovski, E.; Weiss, S.; Ankri, R.; Goldenberg-Cohen, N.; Fixler, D. Diffusion Reflection Measurements of Antibodies Conjugated to Gold Nanoparticles as a Method to Identify Cutaneous Squamous Cell Carcinoma Borders. Materials 2020, 13, 447. https://doi.org/10.3390/ma13020447
Olshinka A, Ad-El D, Didkovski E, Weiss S, Ankri R, Goldenberg-Cohen N, Fixler D. Diffusion Reflection Measurements of Antibodies Conjugated to Gold Nanoparticles as a Method to Identify Cutaneous Squamous Cell Carcinoma Borders. Materials. 2020; 13(2):447. https://doi.org/10.3390/ma13020447
Chicago/Turabian StyleOlshinka, Asaf, Dean Ad-El, Elena Didkovski, Shirel Weiss, Rinat Ankri, Nitza Goldenberg-Cohen, and Dror Fixler. 2020. "Diffusion Reflection Measurements of Antibodies Conjugated to Gold Nanoparticles as a Method to Identify Cutaneous Squamous Cell Carcinoma Borders" Materials 13, no. 2: 447. https://doi.org/10.3390/ma13020447
APA StyleOlshinka, A., Ad-El, D., Didkovski, E., Weiss, S., Ankri, R., Goldenberg-Cohen, N., & Fixler, D. (2020). Diffusion Reflection Measurements of Antibodies Conjugated to Gold Nanoparticles as a Method to Identify Cutaneous Squamous Cell Carcinoma Borders. Materials, 13(2), 447. https://doi.org/10.3390/ma13020447





