Burst Strength of BIOLOX®delta Femoral Heads and Its Dependence on Low-Temperature Environmental Degradation
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples
2.2. Hydrothermal Ageing
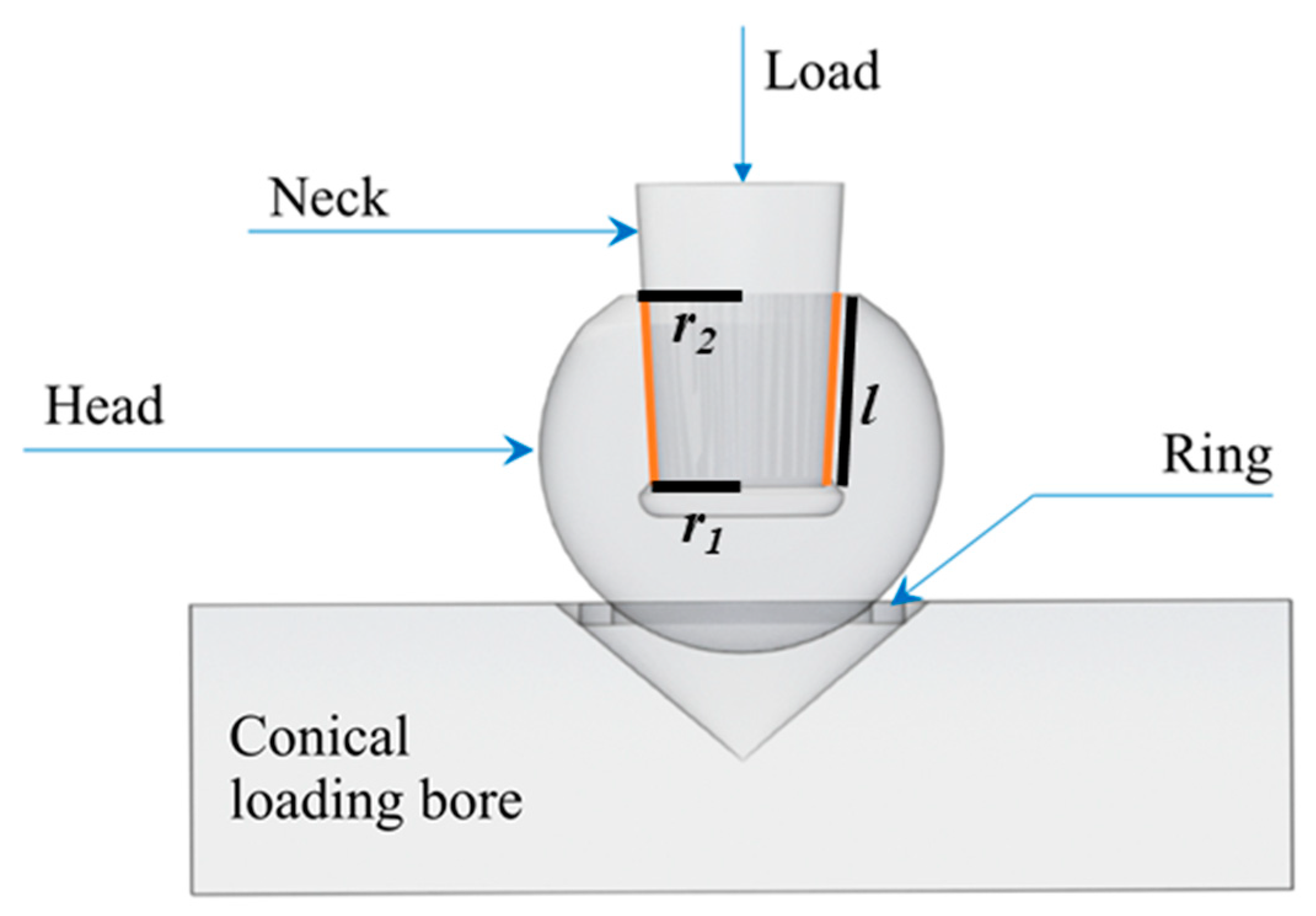
2.3. Burst-Strength Testing
- (1)
- Pristine heads (i.e., as received from the factory): two sets of n = 6 heads were simply unpackaged and then compression tested to failure using CoCr or Ti6Al4V trunnions.
- (2)
- Moderately loaded and hydrothermally aged heads: n = 6 heads were repetitively loaded and unloaded at 3 kN and ~1 kN (maximum and minimum load respectively), respectively, onto CoCr trunnions followed by 15 h of autoclave ageing at 132 °C in the unloaded condition. This load-unload and autoclave cycle was repeated 10 times for a total of 150 h. The heads were then compression tested to failure using CoCr trunnions.
- (3)
- Severely loaded and hydrothermally aged heads: n = 6 heads were repetitively loaded and unloaded at 20 kN and ~10 kN, respectively onto CoCr followed by 15 h of autoclave ageing at 132 °C in the unloaded condition. This load-unload and autoclave ageing cycle was then repeated 10 times for a total of 150 h. The test was further augmented by one final loading using CoCr trunnions with the inclusion of 0.5 mL of phosphate-buffered saline solution in the taper bore, followed by subjecting the assembled heads to a final 15 h autoclave cycle at 132 °C. These heads were then compression tested to failure using CoCr trunnions.
2.4. Sample Characterizations
2.4.1. Laser Microscopy
2.4.2. Scanning Electron Microscopy
2.4.3. Raman Spectroscopy
3. Results
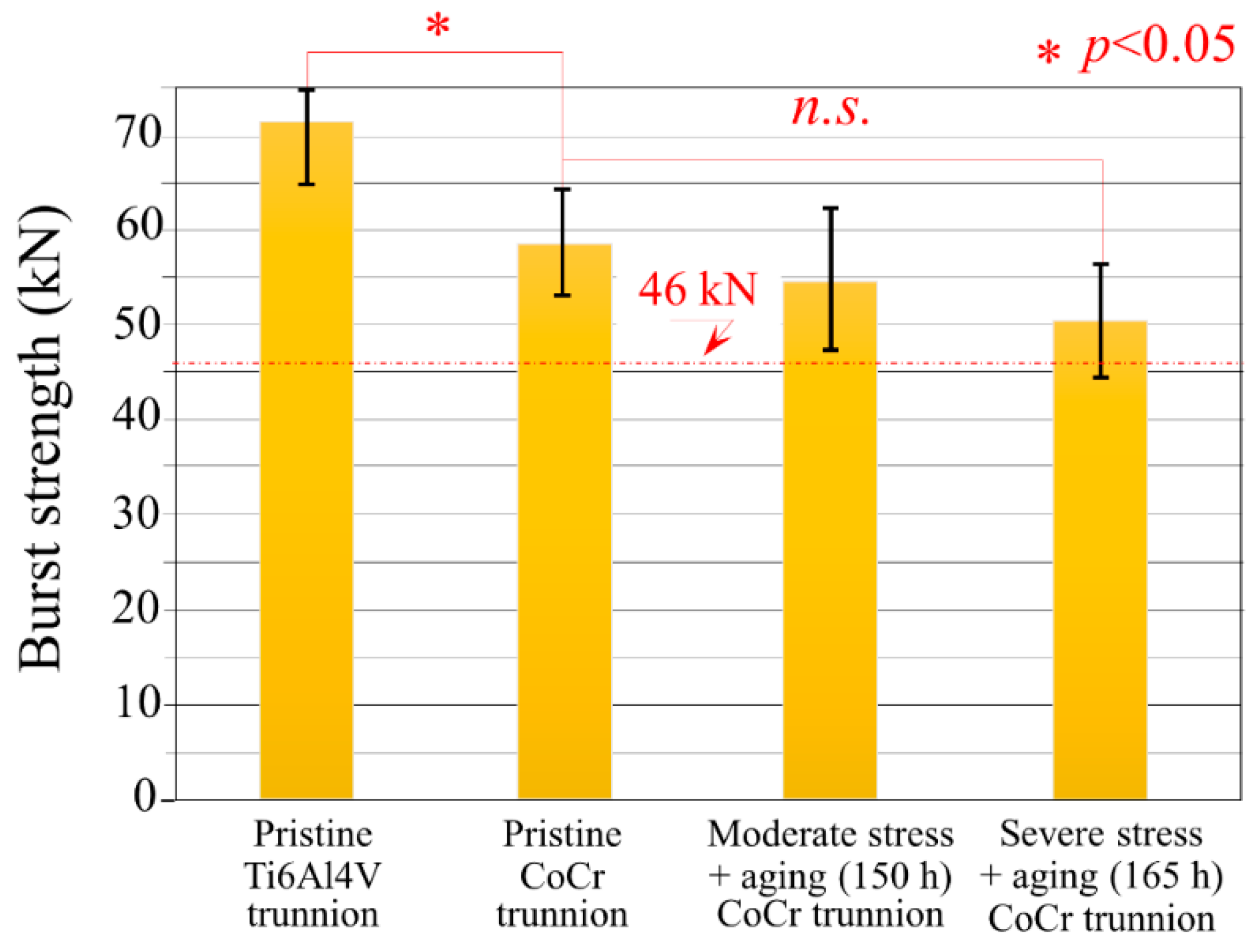
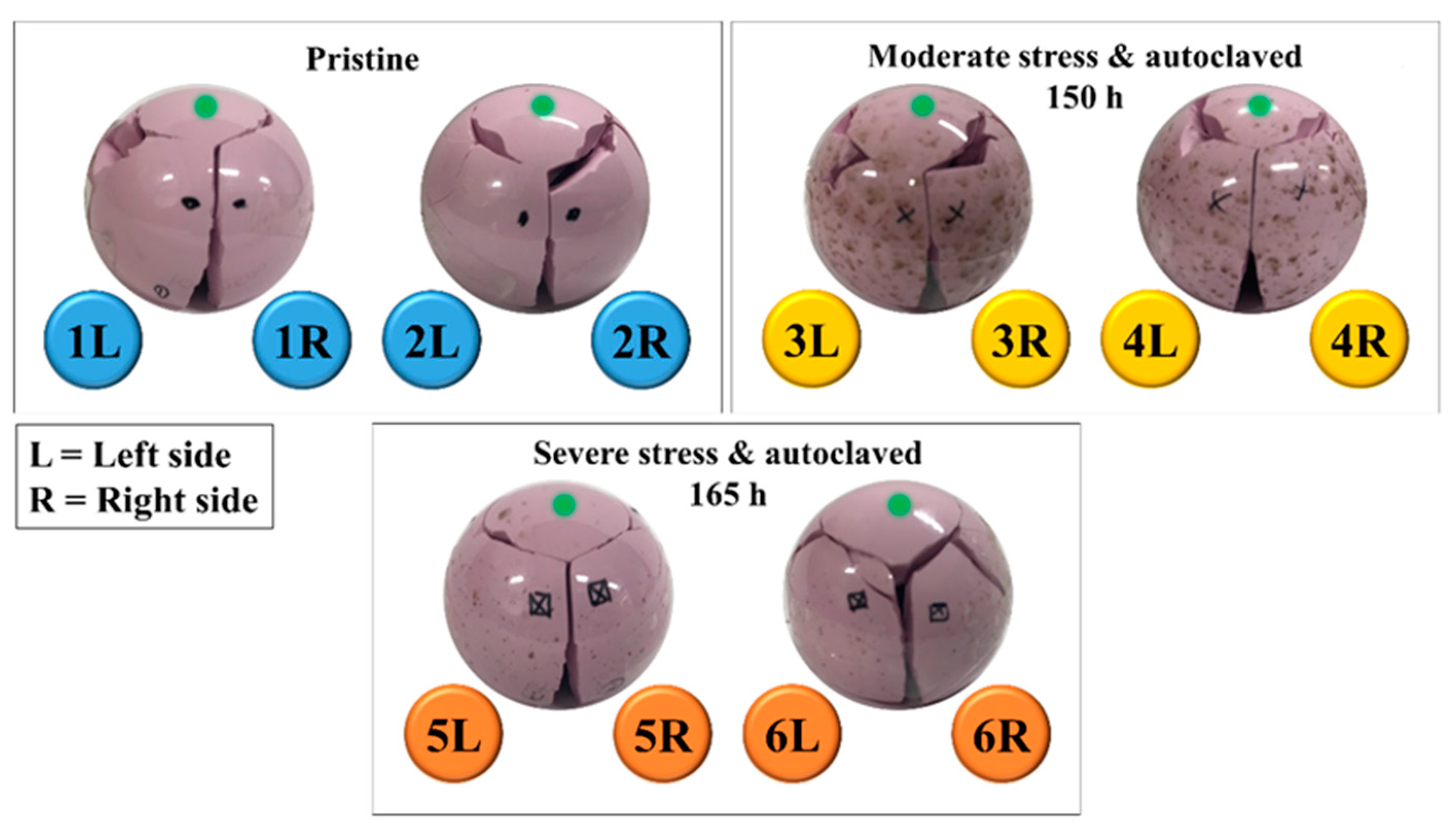
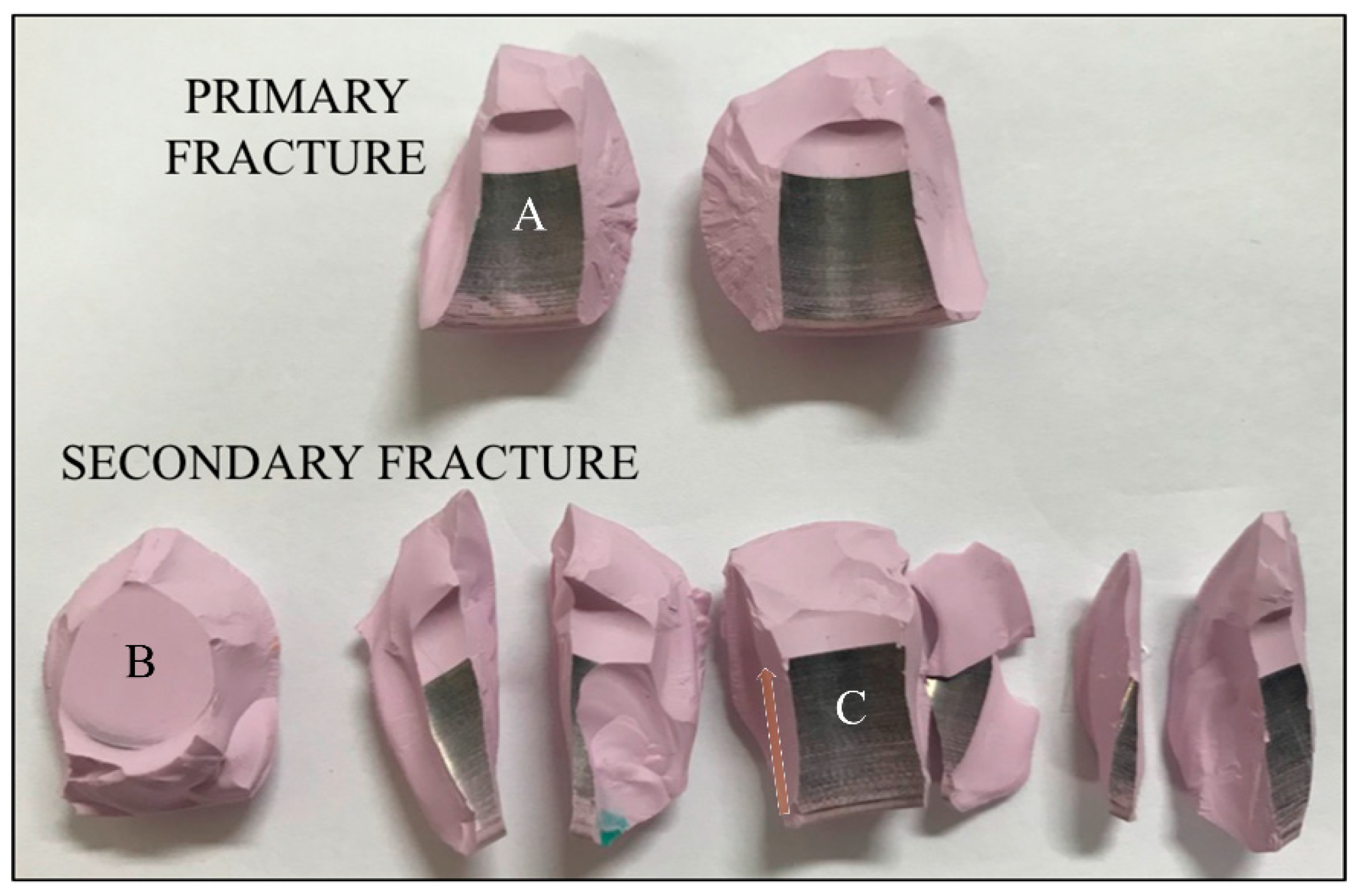
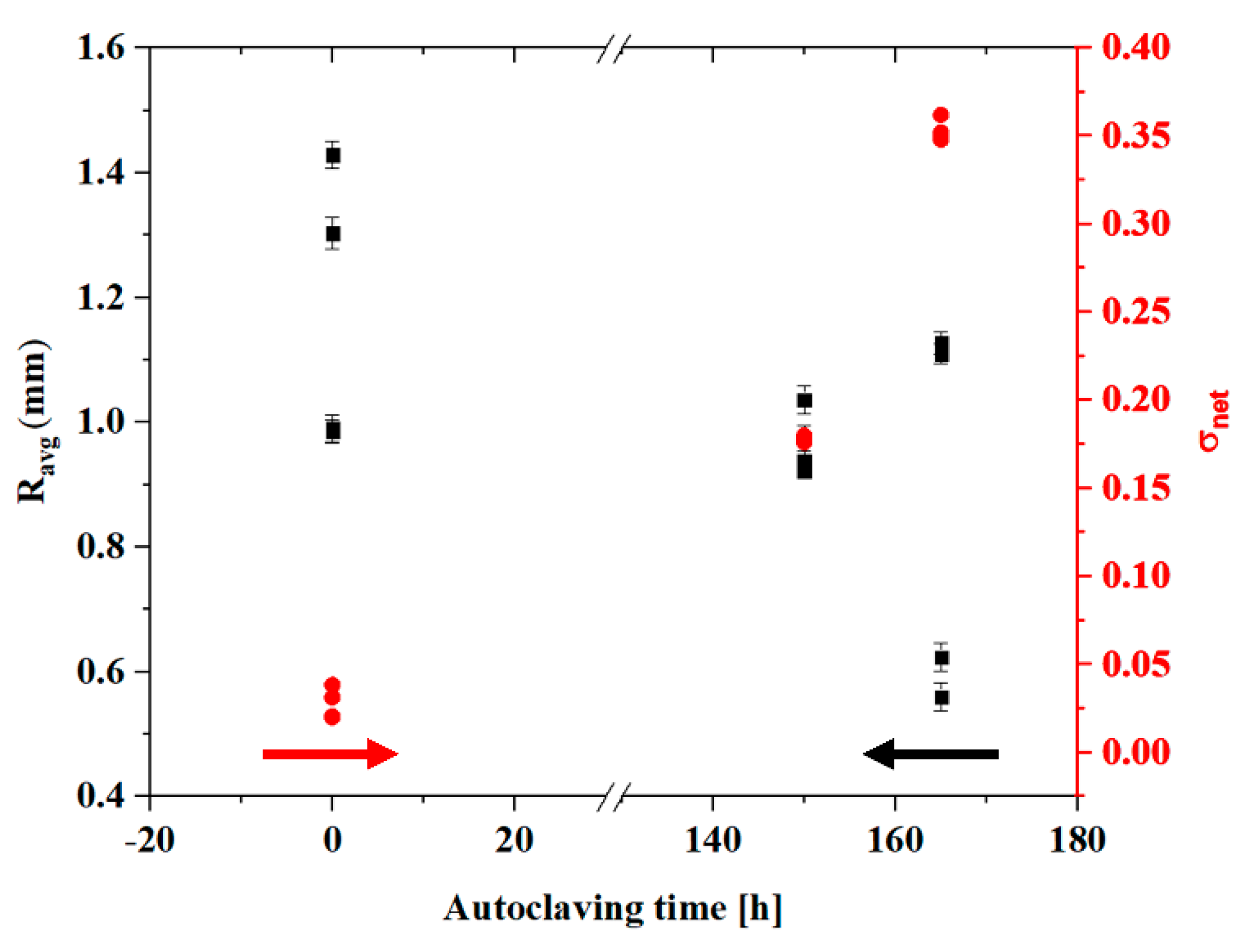
3.1. Burst-Strength Test Results and Post Fracture Head Reconstruction
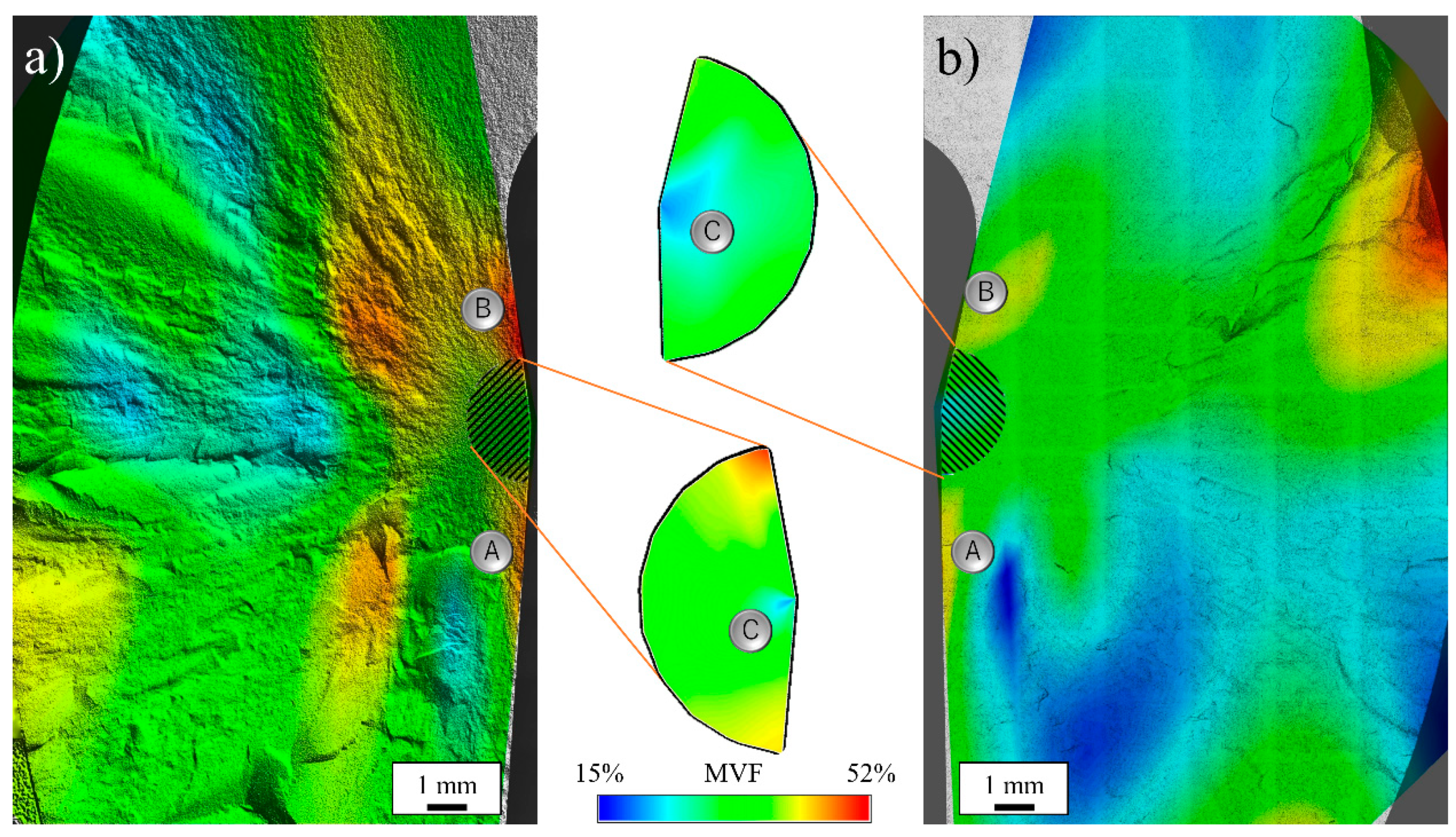
3.2. Surface Fracture Morphology
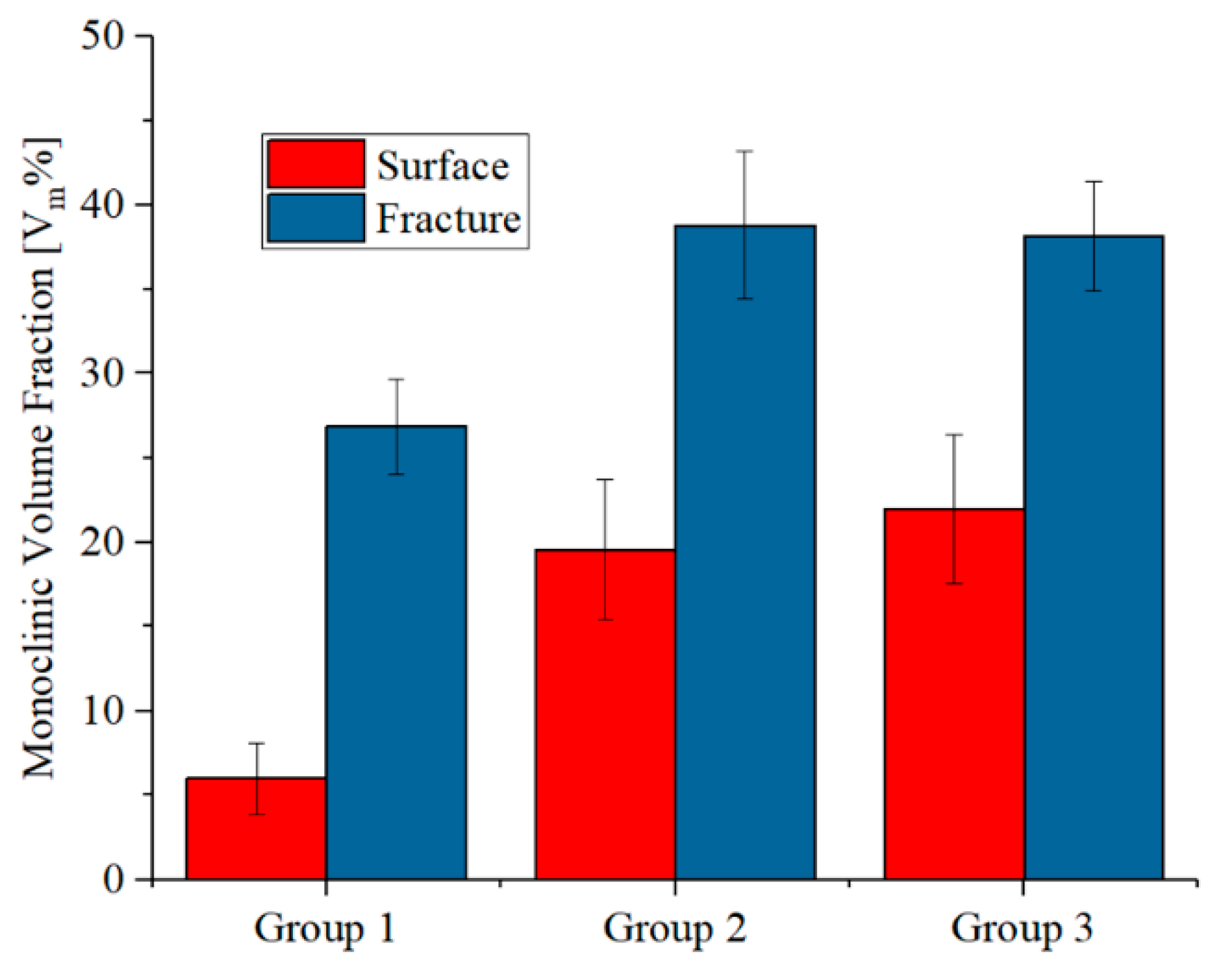
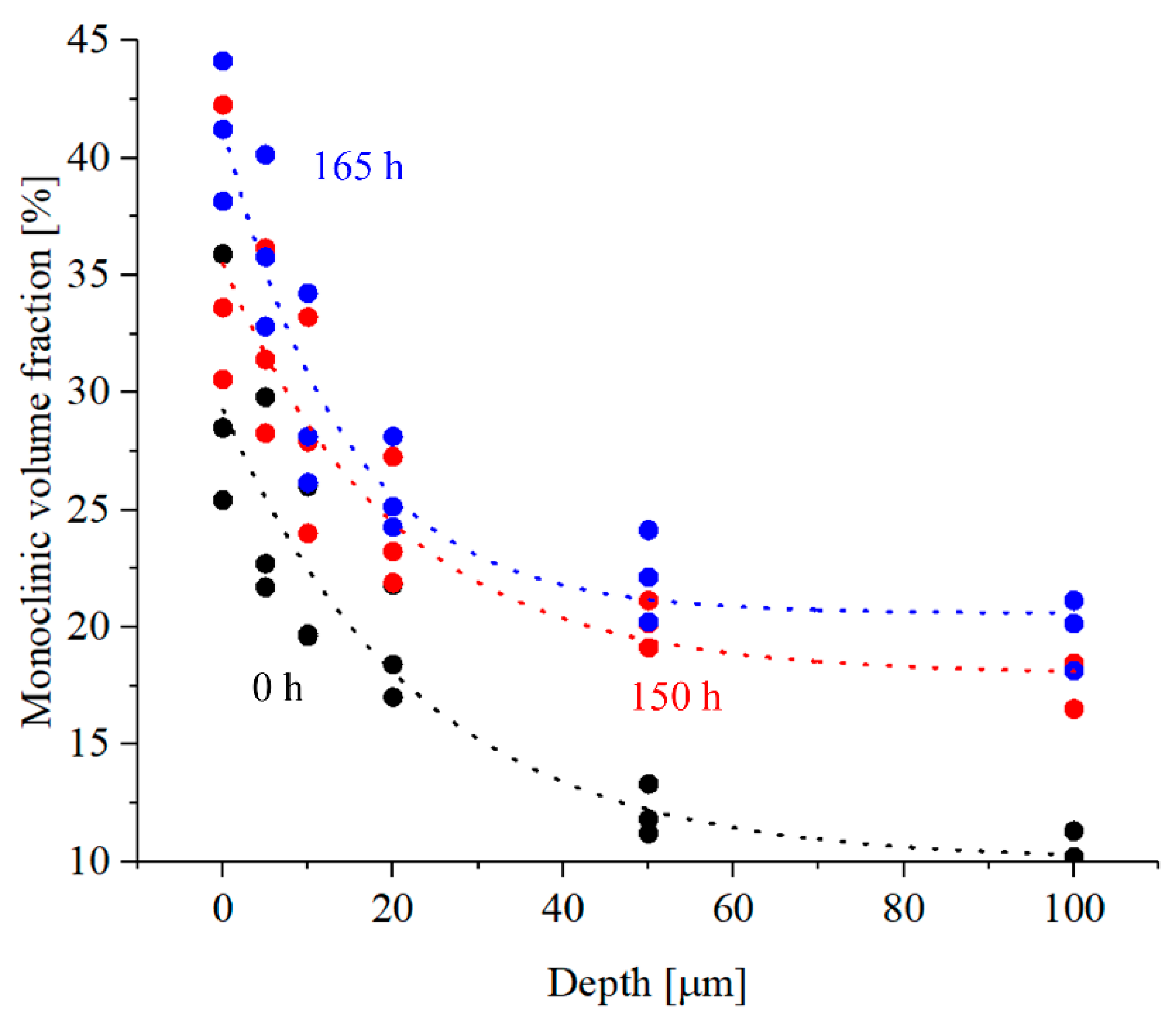
3.3. Zirconia Phase Transformation
3.4. Macroscopic Analysis
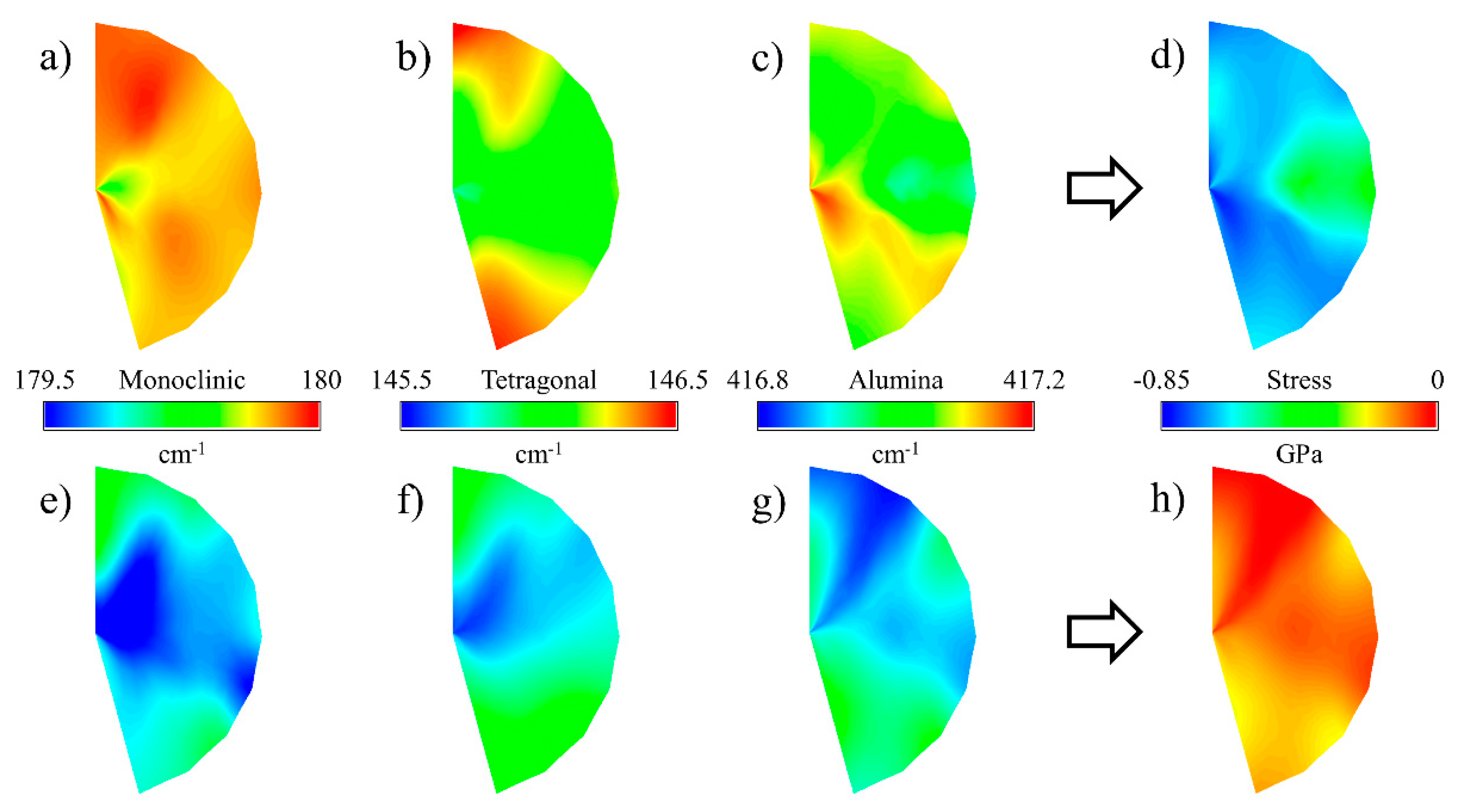
3.5. Microscopic Evaluations
3.6. Residual Stresses
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Affatato, S.; Ruggiero, A.; Merola, M. Advanced bio-materials in hip joint arthroplasty. A review on polymer and ceramics composites as alternative bearings. Compos. Part B Eng. 2015, 83, 276–283. [Google Scholar] [CrossRef]
- Chevalier, J.; Gremillard, L. Ceramics for medical applications: A picture for the next 20 years. J. Eur. Ceram. Soc. 2009, 29, 1245–1255. [Google Scholar] [CrossRef]
- Hannink, R.H.J.; Kelly, P.M.; Muddle, B.C. Transformation toughening in zirconia-containing ceramics. J. Am. Ceram. Soc. 2000, 83, 461–487. [Google Scholar] [CrossRef]
- Pezzotti, G.; Munisso, M.C.; Porporati, A.A.; Lessnau, K. On the role of oxygen vacancies and lattice strain in the tetragonal to monoclinic transformation in alumina/zirconia composites and improved environ-mental stability. Biomaterials 2010, 31, 6901–6908. [Google Scholar] [CrossRef] [PubMed]
- Pezzotti, G.; Yamada, K.; Sakakura, S.; Pitto, R.P. Raman spectro-scopic analysis of advanced ceramic composite for hip prosthesis. J. Am. Ceram. Soc. 2008, 91, 1199–1206. [Google Scholar] [CrossRef]
- Merkert, P. Next Generation Ceramic Bearings. In Bioceramics in Joint Arthroplasty; Steinkopff Verlag: Heidelberg, Germany, 2003; pp. 123–125. [Google Scholar]
- Insley, G.; Steicher, R. Next generation ceramics based on zirconia toughened alumina for hip joint prostheses. Key Eng. Mater. 2004, 254–256, 675–678. [Google Scholar] [CrossRef]
- Gutknecht, D.; Chevalier, J.; Garnier, V.; Fantozzi, G. Key role of processing to avoid low temperature ageing in alumina zirconia composites for orthopaedic application. J. Eur. Ceram. Soc. 2007, 27, 1547–1552. [Google Scholar] [CrossRef]
- Deville, S.; Chevalier, J.; Dauvergne, C.; Fantozzi, G.; Bartolomé, J.F.; Moya, J.S.; Torrecillas, R. Microstructural investigation of the ageing behavior of (3Y-TZP)–Al2O3 composites. J. Am. Ceram. Soc. 2005, 88, 1273–1280. [Google Scholar] [CrossRef]
- Pezzotti, G. Environmental phase stability of next generation ceramic composite for hip prostheses. Key Eng. Mater. 2006, 309–311, 1223–1226. [Google Scholar] [CrossRef]
- Kuntz, M. Live-time prediction of Biolox® delta. In Ceramics in Orthopaedics: Proceedings of the 12th BIOLOX® Symposium; Billau, K., Chang, J.D., Eds.; Steinkopff Verlag: Darmstadt, Germany, 2007; pp. 281–288. [Google Scholar]
- Chevalier, J.; Grandjean, S.; Kuntz, M.; Pezzotti, G. On the kinetics and impact of tetragonal to monoclinic transformation in an alumina/zirconia composite for arthroplasty applications. Biomaterials 2009, 30, 5279–5282. [Google Scholar] [CrossRef]
- Chevalier, J.; Cales, B.; Drouin, J.M. Low-temperature ageing of Y-TZP ceramics. J. Am. Ceram. Soc. 1999, 82, 2150–2154. [Google Scholar] [CrossRef]
- Sakona, A.; MacDonald, D.W.; Sharma, P.; Medel, F.J.; Kurtz, S.M. Retrieval analysis of historical zirconia femoral heads and contemporary alternatives: Oxinium and Biolox Delta. In Proceedings of the Transactions of the 56th Annual Meeting of the Orthopaedic Research Society, New Orleans, LA, USA, 6–9 March 2010; p. 2358. [Google Scholar]
- Clarke, I.C.; Green, D.D.; Williams, P.; Kubo, K.; Pezzotti, G.; Lombardi, A. Hip-simulator wear studies of an alumina-matrix composite (AMC) ceramic compared to retrieval studies of AMC balls with 1–7 years follow-up. Wear 2009, 267, 702–709. [Google Scholar] [CrossRef]
- Affatato, S.; Modena, E.; Toni, A.; Taddei, P. Retrieval analysis of three generations of Biolox® femoral heads: Spectroscopic and SEM characterization. J. Mech. Behav. Biomed. Mater. 2012, 13, 118–128. [Google Scholar] [CrossRef] [PubMed]
- Kurtz, S.M.; Kocagöz, S.; Arnholt, C.; Huet, R.; Ueno, M.; Walter, W.L. Advances in zirconia toughened alumina biomaterials for total joint replacement. J. Mech. Behav. Biomed. Mater. 2013, 31, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Boffelli, M.; Doimo, A.; Marin, E.; Puppulin, L.; Zhu, W.; Sugano, N.; Clarke, I.C.; Pezzotti, G. Chemically driven tetragonal-to-monoclinic transformation in retrieved ZTA femoral heads from dual mobility hip implants. J. Mech. Behav. Biomed. Mater. 2016, 56, 195–204. [Google Scholar] [CrossRef]
- Bal, B.S.; Zhu, W.; Zanocco, M.; Marin, E.; Sugano, N.; McEntire, B.J.; Pezzotti, G. Reconciling in vivo and in vitro kinetics of the polymorphic transformation in zirconia-toughened alumina for hip joints: I. Phenomenology. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 72, 252–258. [Google Scholar] [CrossRef]
- Pezzotti, G.; Affatato, S.; Rondinella, A.; Yorifuji, M.; Marin, E.; Zhu, W.; McEntire, B.; Bal, B.S.; Yamamoto, K. In vitro versus in vivo phase instability of zirconia-toughened alumina femoral heads: A critical comparative assessment. Materials 2017, 10, 466. [Google Scholar] [CrossRef]
- ASTM International Designation: F2345-03. Standard test methods for determination of static and cyclic fatigue strength of ceramic modular femoral heads. In Annual Book of ASTM Standards; ASTM International: West Conshohocken, PA, USA, 2004; pp. 1–8. [Google Scholar]
- Rondinella, A.; Affatato, S.; Marin, E.; Zhu, W.; McEntire, B.J.; Bal, B.S.; Tateiwa, T.; Yamamoto, K.; Valdré, G.; Pezzotti, G. In toto microscopic scanning of ZTA femoral head retrievals using CAD-assisted confocal Raman spectroscopy. Mater. Des. 2017, 116, 631–637. [Google Scholar] [CrossRef]
- Pezzotti, G.; Bal, B.S.; Zanocco, M.; Marin, E.; Sugano, N.; McEntire, B.J.; Zhu, W. Reconciling in vivo and in vitro kinetics of the polymorphic transformation in zirconia-toughened alumina for hip joints: III. Molecular scale mechanisms. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 71, 552–557. [Google Scholar] [CrossRef]
- Zhu, W.; Fujiwara, A.; Nishiike, N.; Nakashima, S.; Gu, H.; Marin, E.; Sugano, N.; Pezzotti, G. Mechanisms induced by transition metal contaminants and their effect on the hydrothermal stability of zirconia-containing bioceramics: An XPS study. Phys. Chem. Chem. Phys. 2018, 20, 28929–28940. [Google Scholar] [CrossRef]
- Rondinella, A.; Marin, E.; Boschetto, F.; Zanocco, M.; Zhu, W.; Affatato, S.; Yamamoto, K.; Tateiwa, T.; Pezzotti, G. Degradation phenomena occurring in the conical taper of a short-term retrieved ZTA femoral head: A case study. Mater. Des. 2018, 157, 362–370. [Google Scholar] [CrossRef]
- Piconi, C.; Maccauro, G.; Muratori, F. Alumina matrix composites in arthroplasty. Key Eng. Mater. 2005, 284, 979–982. [Google Scholar] [CrossRef]
- Dobbs, H.S. Quality improvement resulting from legal and regulatory developments. In Bioceramics in Joint Arthroplasty; Zippel, H., Dietrich, M., Eds.; Steinkopff Verlag: Heidelberg, Germany, 2003; pp. 205–208. [Google Scholar]
- ISO Standard 7206-10:2018 Implants for surgery–Partial and total hip-joint prostheses–Part 10: Determination of Resistance to Static Load of Modular Femoral Heads; ISO: Geneva, Switzerland, 2018.
- Porter, D.A.; Urban, R.M.; Jacobs, J.J.; Gilbert, J.L.; Rodriguez, J.A.; Cooper, H.J. Modern trunnions are more flexible: A mechanical analysis of THA taper designs. Clin. Orthop. Relat. Res. 2014, 472, 3963–3970. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, J.R.; Gilbert, J.L.; Jacobs, J.J.; Bauer, T.W.; Paprosky, W.; Leurgans, S. A multicenter retrieval study of the taper interfaces of modular hip prostheses. Clin. Orthop. Relat. Res. 2002, 401, 149–161. [Google Scholar] [CrossRef]
- Panagiotidou, A.; Meswania, J.; Hua, J.; Muirhead-Allwood, S.; Hart, A.; Blunn, G. Enhanced wear and corrosion in modular tapers in total hip replacement is associated with the contact area and surface topography. J. Orthop. Res. 2013, 31, 2032–2039. [Google Scholar] [CrossRef]
- Fallahnezhad, K.; Farhoudi, H.; Oskouei, R.H.; Taylor, M. Influence of geometry and materials on the axial and torsional strength of the head–neck taper junction in modular hip replacements: A finite element study. J. Mech. Behav. Biomed. Mater. 2016, 60, 118–126. [Google Scholar] [CrossRef]
- Smekal, A. Die Festigkeitseigenschaften spröder Körper. In Ergebnisse der Exakten Naturwissenschaften; Springer: Berlin/Heidelberg, Germany, 1936; Volume 15, pp. 106–188. [Google Scholar]
- Quinn, G.D. Guidelines for measuring fracture mirrors. Ceram. Trans. 2007, 199, 163. [Google Scholar]
- Bansal, G.K. On fracture mirror formation in glass and polycrystalline ceramics. Philos. Mag. 1977, 35, 935–944. [Google Scholar] [CrossRef]
- Zdaniewski, W.A. Fracture of polycrystalline TiB2-AlN composites in various environments. Acta Metall. 1989, 37, 2313–2320. [Google Scholar] [CrossRef]
- Lundberg, H.; Ha, N.; Hall, D.; Urban, R.; Levine, B.; Pourzal, R. Contact mechanics and plastic deformation at the local surface topography level after assembly of modular head-neck junctions in modern total hip replacement devices. In Modularity and Tapers in Total Joint Replacement Devices; ASTM International: West Conshohocken, PA, USA, 2015. [Google Scholar]
- Elkins, J.M.; Callaghan, J.J.; Brown, T.D. Stability and trunnion wear potential in large-diameter metal-on-metal total hips: A finite element analysis. Clin. Orthop. Relat. Res. 2014, 472, 529–542. [Google Scholar] [CrossRef]
- Dyrkacz, R.M.R.; Brandt, J.M.; Morrison, J.B.; O’Brien, S.T.; Ojo, O.A.; Turgeon, T.R.; Wyss, U.P. Finite element analysis of the head–neck taper interface of modular hip prostheses. Tribol. Int. 2015, 91, 206–213. [Google Scholar] [CrossRef]
- Katagiri, G.; Ishida, H.; Ishitani, A.; Masaki, T. Direct determination by a Raman microprobe of the transformation zone size in Y2O3 containing tetragonal ZrO2 polycrystals. Adv. Ceram. 1986, 24, 537–544. [Google Scholar]
- Masson, B.; Kuntz, M. Long-Term Stability of Ceramic Composite in Total Hip Arthroplasty. In Total Hip Arthroplasty; Springer: Berlin/Heidelberg, Germany, 2012; pp. 145–153. [Google Scholar]
- Corfield, V.; Khan, I.; Scott, R. Hydrothermal stability of ceramic femoral heads. In Bioceramics and Alternative Bearings in Joint Arthroplasty; Steinkopff: Darmstadt, Germany, 2007; pp. 59–64. [Google Scholar]
- Garino, J.P. The reliability of modern alumina bearings in total hip arthroplasty—Update to a 2006 report. In Seminars in Arthroplasty; WB Saunders: Philadelphia, PA, USA, 2013; pp. 193–201. [Google Scholar]
- Ceramtec. “Ceramic Ball Heads”, Company Brochure. Available online: https://www.ceramtec.com/files/mt_biolox_forte_delta_comparison_en.pdf (accessed on 6 September 2019).
- Khumrak, S.; Yakampor, T. Ceramic on ceramic bearings. Bangk. Med J. 2014, 4, 93–103. [Google Scholar] [CrossRef][Green Version]
- Clarke, I.C.; Pezzotti, G.; Sugano, N. Mechanical Properties That Influence Design and Performance of Ceramic Hip Bearings. In Materials in Hip Surgery; Saunders: Philadelphia, PA, USA, 2007. [Google Scholar]
- Upmann, C.; Eisele, M.; Pandorf, T. What is the Influence of Taper Contamination on the Burst Strength of Alumina Matrix Composite Ball Heads. Orthopaedic Proc. 2018, 100, 61. [Google Scholar]
- Rack, R.; Pfaff, H.G. Long-term performance of the alumina matrix composite Biolox delta. In Bioceramics in Joint Arthroplasty: 6 th International BIOLOX Symposium; Georg Thieme Verlag: Stuttgart, Germany, 2001; pp. 103–108. [Google Scholar]
- McEntire, B.J.; Enomoto, Y.; Zhu, W.; Boffelli, M.; Marin, E.; Pezzotti, G. Surface toughness of silicon nitride bioceramics: II, Comparison with commercial oxide materials. J. Mech. Behav. Biomed. Mater. 2016, 54, 346–359. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tateiwa, T.; Marin, E.; Rondinella, A.; Ciniglio, M.; Zhu, W.; Affatato, S.; Pezzotti, G.; Bock, R.M.; McEntire, B.J.; Bal, B.S.; et al. Burst Strength of BIOLOX®delta Femoral Heads and Its Dependence on Low-Temperature Environmental Degradation. Materials 2020, 13, 350. https://doi.org/10.3390/ma13020350
Tateiwa T, Marin E, Rondinella A, Ciniglio M, Zhu W, Affatato S, Pezzotti G, Bock RM, McEntire BJ, Bal BS, et al. Burst Strength of BIOLOX®delta Femoral Heads and Its Dependence on Low-Temperature Environmental Degradation. Materials. 2020; 13(2):350. https://doi.org/10.3390/ma13020350
Chicago/Turabian StyleTateiwa, Toshiyuki, Elia Marin, Alfredo Rondinella, Marco Ciniglio, Wenliang Zhu, Saverio Affatato, Giuseppe Pezzotti, Ryan M. Bock, Bryan J. McEntire, B. Sonny Bal, and et al. 2020. "Burst Strength of BIOLOX®delta Femoral Heads and Its Dependence on Low-Temperature Environmental Degradation" Materials 13, no. 2: 350. https://doi.org/10.3390/ma13020350
APA StyleTateiwa, T., Marin, E., Rondinella, A., Ciniglio, M., Zhu, W., Affatato, S., Pezzotti, G., Bock, R. M., McEntire, B. J., Bal, B. S., & Yamamoto, K. (2020). Burst Strength of BIOLOX®delta Femoral Heads and Its Dependence on Low-Temperature Environmental Degradation. Materials, 13(2), 350. https://doi.org/10.3390/ma13020350






