Effect of GelMA Hydrogel Coatings on Corrosion Resistance and Biocompatibility of MAO-Coated Mg Alloys
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples and Coating Preparation Procedures
2.2. Characterization of Coatings and Electrochemical Test
2.2.1. Surface Analysis
2.2.2. Electrochemical Corrosion Test
2.3. The Evaluation of Cytocompatibility In Vitro
2.3.1. Preparation of Extracts
2.3.2. Cell Culture
2.3.3. Cell Viability and Proliferation Assay
3. Results
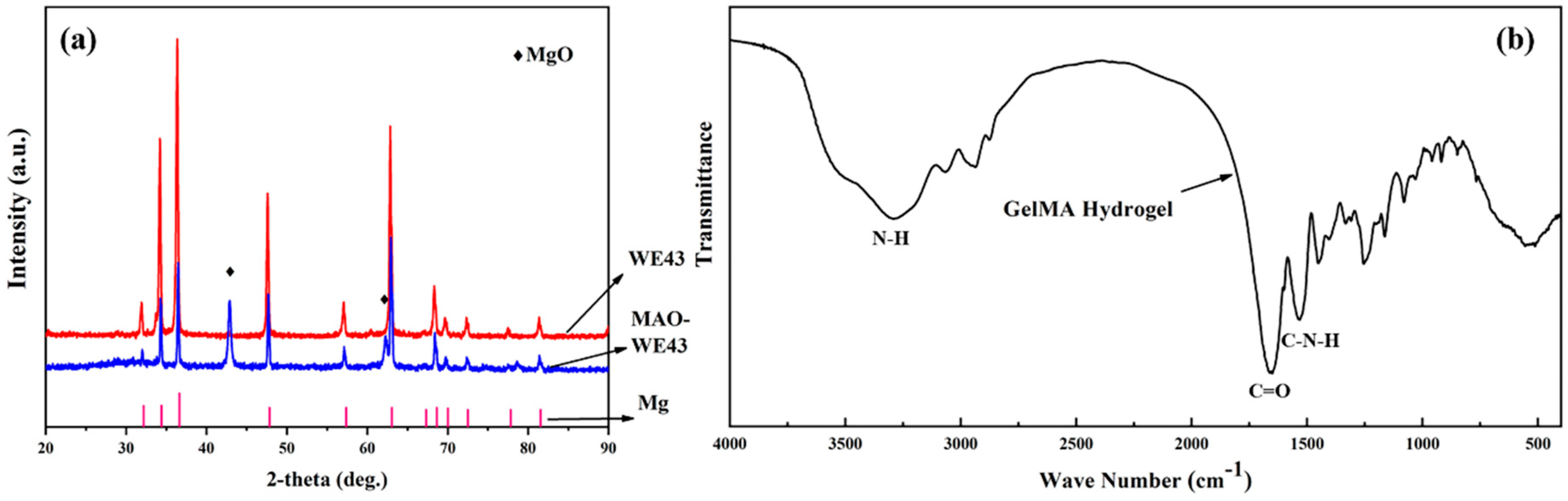
3.1. XRD Results and FTIR Spectra
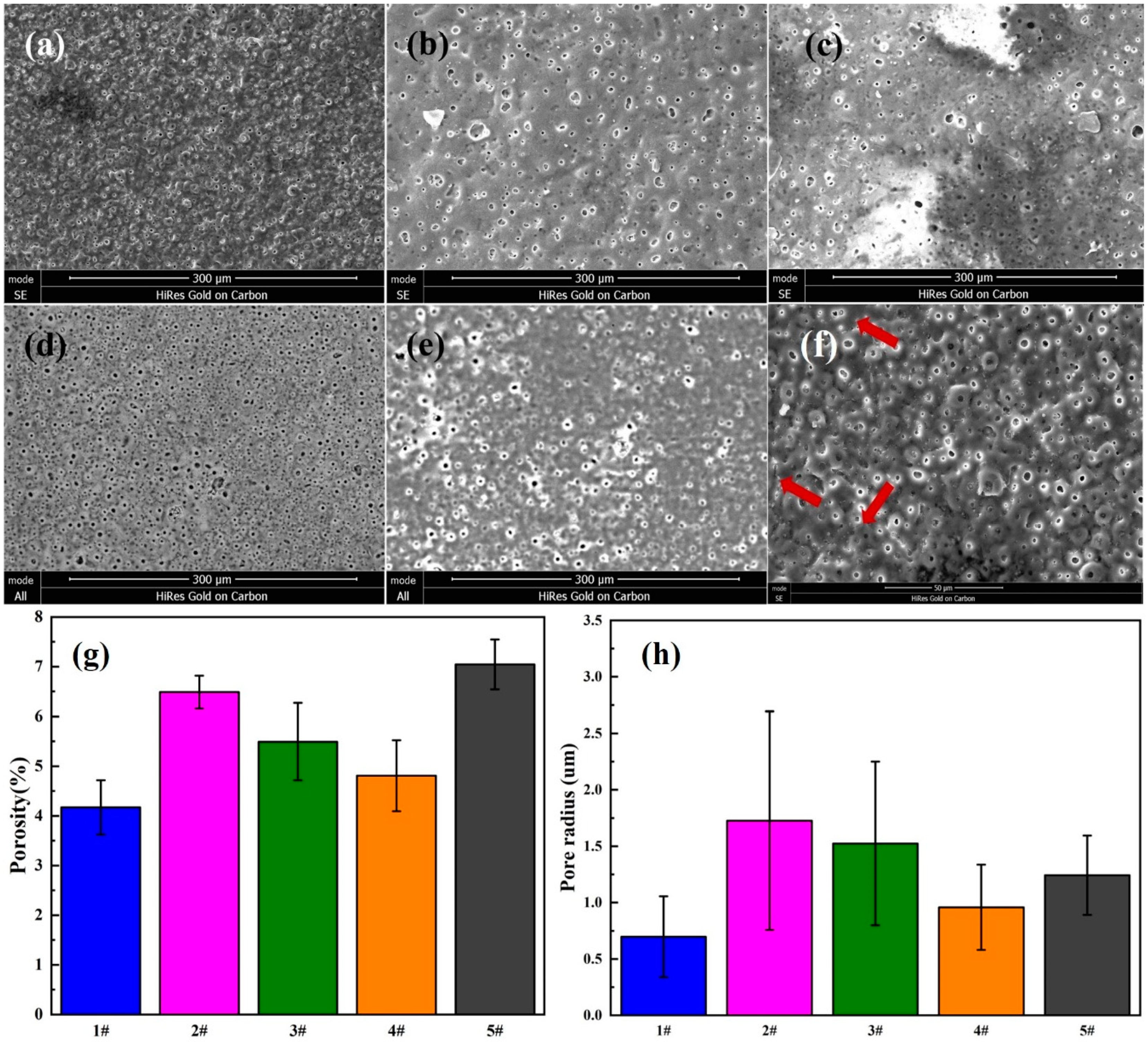
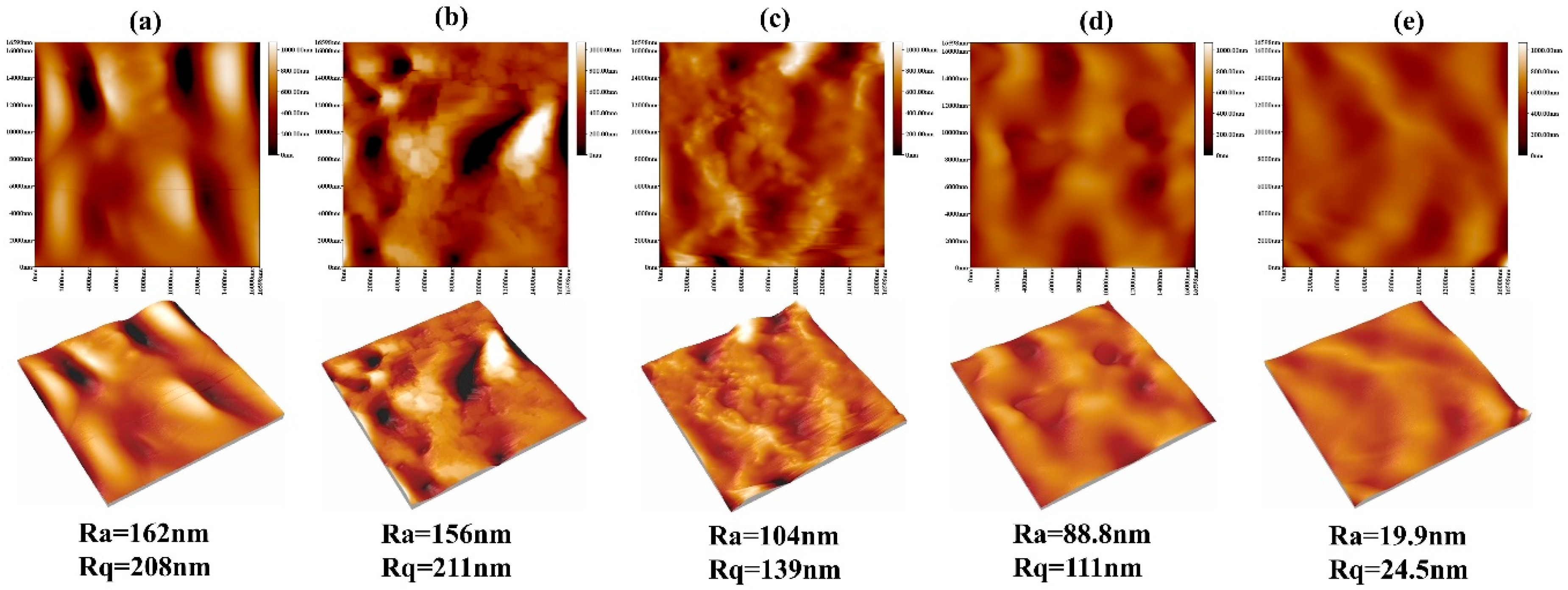
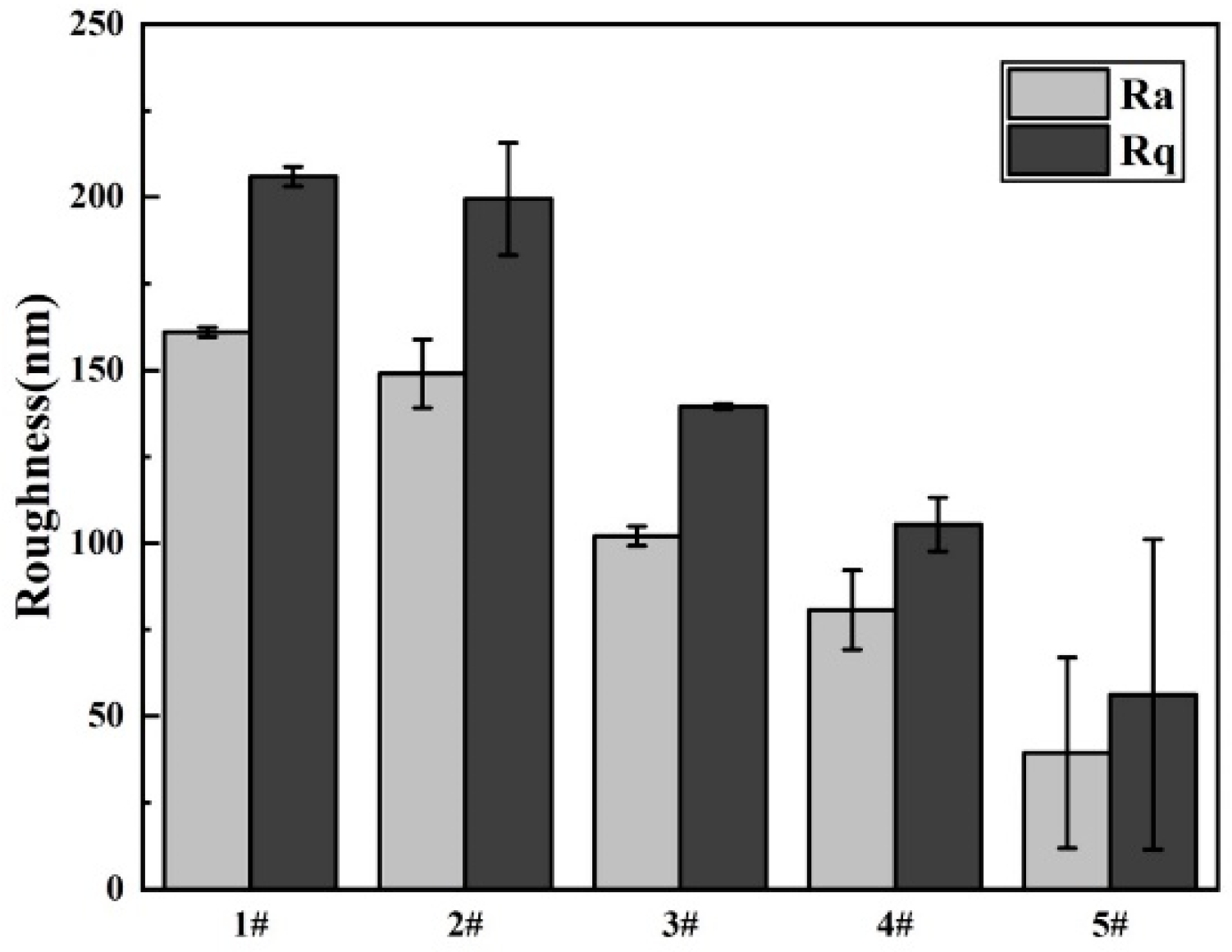
3.2. The Morphologies of the Coatings
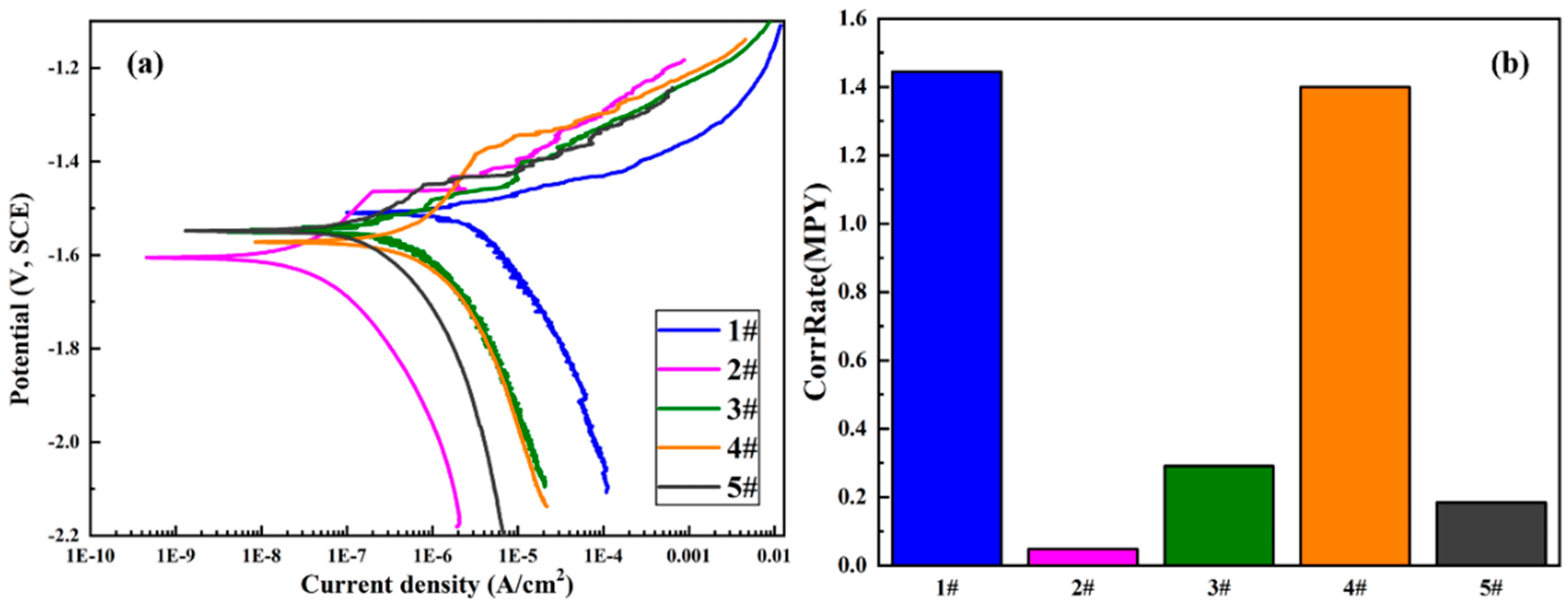
3.3. Electrochemical Corrosion
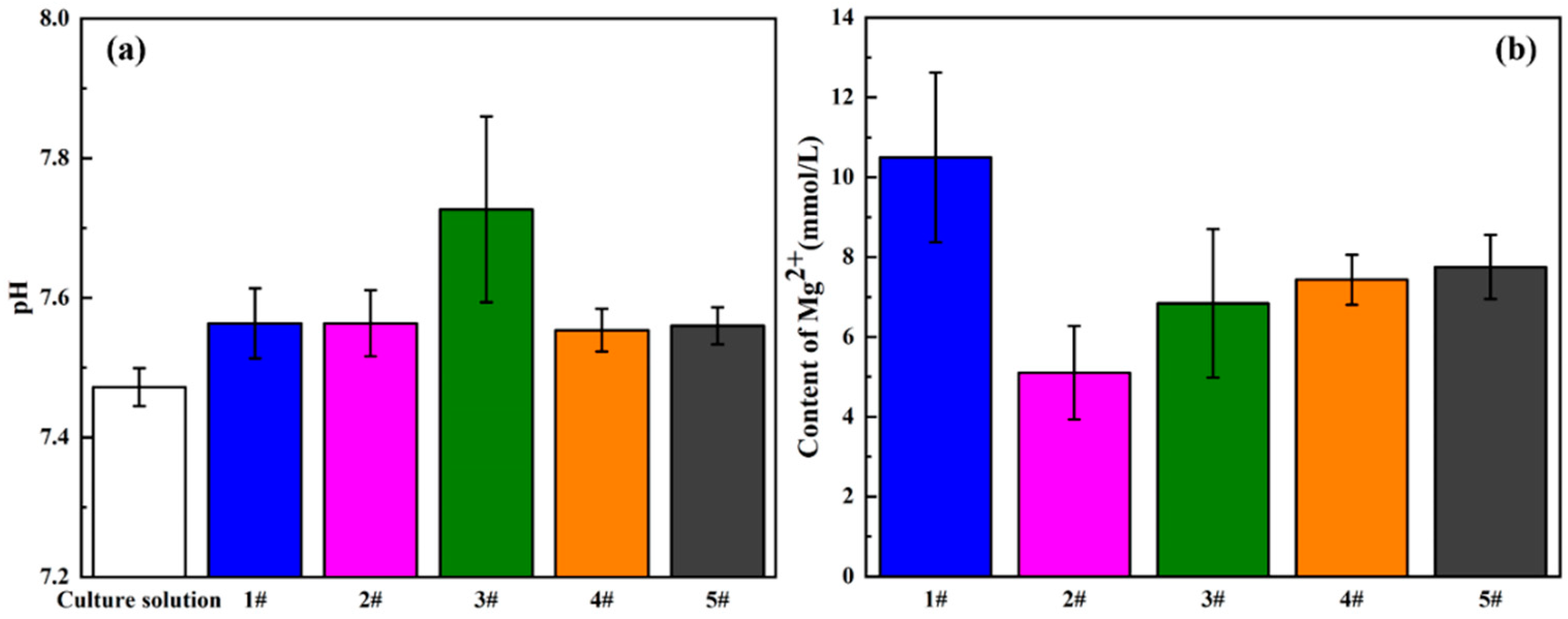
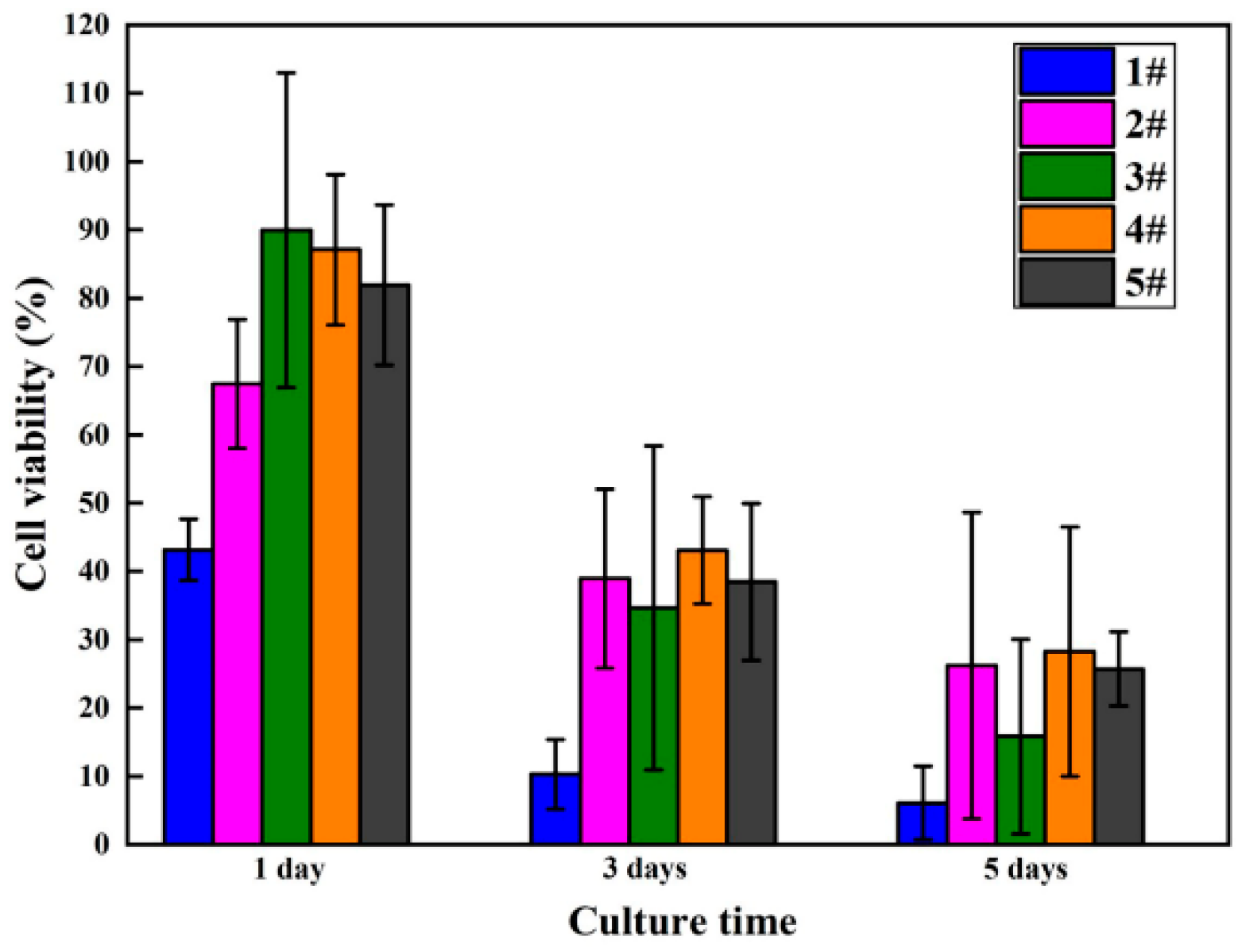
3.4. In Vitro Biocompatibility
4. Discussion
4.1. Corrosion Resistance
4.2. In Vitro Biocompatibility
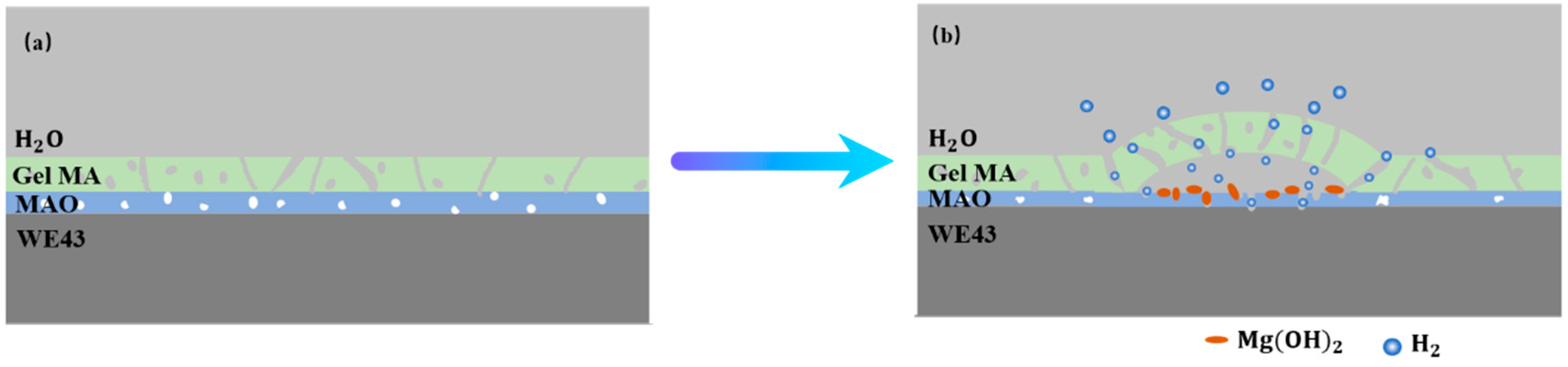
4.3. The Balance Mechanism of Corrosion and Coating Protection
5. Conclusions
- The GelMA hydrogel coating plays a role in the sealing of the MAO-coated magnesium alloys, which effectively prevents the entry of corrosive ions, and has more corrosion resistance than the ones without GelMA hydrogel coating.
- GelMA hydrogel coatings can effectively control the Mg2+ content of the extract, promoting the cell proliferation and growth, and the good cytocompatibility of the GelMA/MAO-coated magnesium alloys is expected to be a promising bone tissue engineering material.
- There is no linear relationship between coating thickness and biocompatibility results and electrochemical test results consisting of corrosion potential and corrosion rate due to the balance mechanism between corrosion of magnesium alloy and the protection of GelMA hydrogel coatings. Further study may be focused on optimizing the processing of GelMA hydrogel coatings.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kiani, F.; Wen, C.; Li, Y. Prospects and strategies for magnesium alloys as biodegradable implants from crystalline to bulk metallic glasses and composites—A review. Acta Biomater. 2020, 103, 1–23. [Google Scholar] [CrossRef]
- Kim, B.J.; Piao, Y.; Wufuer, M.; Son, W.C.; Choi, T.H. Biocompatibility and efficiency of biodegradable magnesium-based plates and screws in the facial fracture model of beagles. J. Oral. Maxillofac. Surg. 2018, 76, 1055.e1–1055.e9. [Google Scholar] [CrossRef]
- Yang, Y.; He, C.; Dianyu, E.; Yang, W.; Qi, F.; Xie, D.; Shen, L.; Peng, S.; Shuai, C. Mg bone implant: Features, developments and perspectives. Mater. Des. 2020, 185, 108259. [Google Scholar] [CrossRef]
- Askari, E.; Cengiz, I.F.; Alves, J.L.; Henriques, B.; Flores, P.; Fredel, M.C.; Reis, R.L.; Oliveira, J.M.; Silva, F.S.; Mesquita-Guimaraes, J. Micro-CT based finite element modelling and experimental characterization of the compressive mechanical properties of 3-D zirconia scaffolds for bone tissue engineering. J. Mech. Behav. Biomed. Mater. 2019, 102, 103516. [Google Scholar] [CrossRef]
- Li, G.; Zhang, L.; Wang, L.; Yuan, G.; Dai, K.; Pei, J.; Hao, Y. Dual modulation of bone formation and resorption with zoledronic acid-loaded biodegradable magnesium alloy implants improves osteoporotic fracture healing: An in vitro and in vivo study. Acta Biomater. 2018, 65, 486–500. [Google Scholar] [CrossRef] [PubMed]
- Witte, F.; Kaese, V.; Haferkamp, H.; Switzer, E.; Meyer-Lindenberg, A.; Wirth, C.J.; Windhagen, H. In vivo corrosion of four magnesium alloys and the associated bone response. Biomaterials 2005, 26, 3557–3563. [Google Scholar] [CrossRef] [PubMed]
- Jirjees, O.M. Materials evolution of bone plates for internal fixation of bone fractures: A review. Int. J. Adv. Res. Med. 2019, 1, 100–108. [Google Scholar] [CrossRef]
- Esmaily, M.; Svensson, J.E.; Fajardo, S.; Birbilis, N.; Frankel, G.S.; Virtanen, S.; Arrabal, R.; Thomas, S.; Johansson, L.G. Fundamentals and advances in magnesium alloy corrosion. Prog. Mater. Sci. 2017, 89, 92–193. [Google Scholar] [CrossRef]
- Zeng, Z.; Stanford, N.; Davies, C.H.J.; Nie, J.-F.; Birbilis, N. Magnesium extrusion alloys: A review of developments and prospects. Int. Mater. Rev. 2018, 64, 27–62. [Google Scholar] [CrossRef]
- Park, M.; Lee, J.E.; Park, C.G.; Lee, S.H.; Seok, H.K.; Choy, Y.B. Polycaprolactone coating with varying thicknesses for controlled corrosion of magnesium. J. Coat. Technol. Res. 2013, 10, 695–706. [Google Scholar] [CrossRef]
- Wang, P.; Liu, J.; Shen, S.; Li, Q.; Luo, X.; Xiong, P.; Gao, S.; Yan, J.; Cheng, Y.; Xi, T. In vitro and in vivo studies on two-step alkali-fluoride-treated Mg–Zn–Y–Nd alloy for vascular stent application: Enhancement in corrosion resistance and biocompatibility. ACS Biomater. Sci. Eng. 2019, 5, 3279–3292. [Google Scholar] [CrossRef]
- Wu, W.; Wang, Z.; Zang, S.; Yu, X.; Yang, H.; Chang, S. research progress on surface treatments of biodegradable Mg alloys: A review. ACS Omega 2020, 5, 941–947. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Pei, J.; Wang, H.; Shi, Y.; Niu, J.; Yuan, F.; Huang, H.; Zhang, H.; Yuan, G. Facile preparation of poly(lactic acid)/brushite bilayer coating on biodegradable magnesium alloys with multiple functionalities for orthopedic application. ACS Appl. Mater. Interfaces 2017, 9, 9437–9448. [Google Scholar] [CrossRef] [PubMed]
- Mousa, H.M.; Abdal-hay, A.; Bartnikowski, M.; Mohamed, I.M.A.; Yasin, A.S.; Ivanovski, S.; Park, C.H.; Kim, C.S. A multifunctional zinc oxide/poly(lactic acid) nanocomposite layer coated on magnesium alloys for controlled degradation and antibacterial function. ACS Biomater. Sci. Eng. 2018, 4, 2169–2180. [Google Scholar] [CrossRef]
- Narayanan, T.S.N.S.; Park, I.S.; Lee, M.H. Strategies to improve the corrosion resistance of microarc oxidation (MAO) coated magnesium alloys for degradable implants: Prospects and challenges. Prog. Mater. Sci. 2014, 60, 1–71. [Google Scholar] [CrossRef]
- Wang, C.; Jiang, B.; Liu, M.; Ge, Y. Corrosion characterization of micro-arc oxidization composite electrophoretic coating on AZ31B magnesium alloy. J. Alloys Compd. 2015, 621, 53–61. [Google Scholar] [CrossRef]
- Hornberger, H.; Virtanen, S.; Boccaccini, A.R. Biomedical coatings on magnesium alloys—A review. Acta Biomater. 2012, 8, 2442–2455. [Google Scholar] [CrossRef]
- Gu, X.N.; Li, N.; Zhou, W.R.; Zheng, Y.F.; Zhao, X.; Cai, Q.Z.; Ruan, L. Corrosion resistance and surface biocompatibility of a microarc oxidation coating on a Mg-Ca alloy. Acta Biomater. 2011, 7, 1880–1889. [Google Scholar] [CrossRef]
- Zeng, R.C.; Qi, W.C.; Song, Y.W.; He, Q.K.; Cui, H.Z.; Han, E.H. In vitro degradation of MAO/PLA coating on Mg-1.21Li-1.12Ca-1.0Y alloy. Front. Mater. Sci. 2014, 8, 343–353. [Google Scholar] [CrossRef]
- Zhang, X.P.; Zhao, Z.P.; Wu, F.M.; Wang, Y.L.; Wu, J. Corrosion and wear resistance of AZ91D magnesium alloy with and without microarc oxidation coating in Hank’s solution. J. Mater. Sci. 2007, 42, 8523–8528. [Google Scholar] [CrossRef]
- Liu, F.; Shan, D.; Song, Y.; Han, E.H.; Ke, W. Corrosion behavior of the composite ceramic coating containing zirconium oxides on AM30 magnesium alloy by plasma electrolytic oxidation. Corros. Sci. 2011, 53, 3845–3852. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, J.; Chen, C.F.; Gu, Y. Advances in microarc oxidation coated AZ31 Mg alloys for biomedical applications. Corros. Sci. 2015, 91, 7–28. [Google Scholar] [CrossRef]
- Fischerauer, S.F.; Kraus, T.; Wu, X.; Tangl, S.; Sorantin, E.; Hänzi, A.C.; Löffler, J.F.; Uggowitzer, P.J.; Weinberg, A.M. In vivo degradation performance of micro-arc-oxidized magnesium implants: A micro-CT study in rats. Acta Biomater. 2013, 9, 5411–5420. [Google Scholar] [CrossRef] [PubMed]
- Bakhsheshi-Rad, H.R.; Hamzah, E.; Ebrahimi-Kahrizsangi, R.; Daroonparvar, M.; Medraj, M. Fabrication and characterization of hydrophobic microarc oxidation/poly-lactic acid duplex coating on biodegradable Mg-Ca alloy for corrosion protection. Vacuum 2016, 125, 185–188. [Google Scholar] [CrossRef]
- Xuan, L.I.; Shi, C.; Bai, J.; Guo, C.; Xue, F.; Lin, P.H.; Chu, C.L. Degradation behaviors of surface modified magnesium alloy wires in different simulated physiological environments. Front. Mater. Sci. 2014, 8, 281–294. [Google Scholar]
- Lu, P.; Fan, H.; Liu, Y.; Cao, L.; Wu, X.; Xu, X. Controllable biodegradability, drug release behavior and hemocompatibility of PTX-eluting magnesium stents. Colloids Surf. B Biointerfaces 2011, 83, 23–28. [Google Scholar] [CrossRef]
- Li, C.Y.; Yu, C.; Zeng, R.C.; Zhang, B.C.; Cui, L.Y.; Wan, J.; Xia, Y. In vitro corrosion resistance of a Ta2O5 nanofilm on MAO coated magnesium alloy AZ31 by atomic layer deposition. Bioact. Mater. 2020, 5, 34–43. [Google Scholar] [CrossRef]
- Zeng, R.-C.; Cui, L.-Y.; Jiang, K.; Liu, R.; Zhao, B.-D.; Zheng, Y.-F. In Vitro Corrosion and Cytocompatibility of a Microarc Oxidation Coating and Poly(l-lactic acid) Composite Coating on Mg–1Li–1Ca Alloy for Orthopedic Implants. ACS Appl. Mater. Interfaces 2016, 8, 10014–10028. [Google Scholar] [CrossRef]
- Yu, C.; Cui, L.-Y.; Zhou, Y.-F.; Han, Z.-Z.; Chen, X.-B.; Zeng, R.-C.; Zou, Y.-H.; Li, S.-Q.; Zhang, F.; Han, E.-H.; et al. Self-degradation of micro-arc oxidation/chitosan composite coating on Mg-4Li-1Ca alloy. Surf. Coat. Technol. 2018, 344, 1–11. [Google Scholar] [CrossRef]
- Rahali, K.; Messaoud, G.B.; Kahn, C.; Sanchez-Gonzalez, L.; Kaci, M.; Cleymand, F.; Fleutot, S.; Linder, M.; Desobry, S.; Arab-Tehrany, E. Synthesis and characterization of nanofunctionalized gelatin methacrylate hydrogels. Int. J. Mol. Sci. 2017, 18, 2675. [Google Scholar] [CrossRef]
- Jiang, T.; Zhao, J.; Yu, S.; Mao, Z.; Gao, C.; Zhu, Y.; Mao, C.; Zheng, L. Untangling the response of bone tumor cells and bone forming cells to matrix stiffness and adhesion ligand density by means of hydrogels. Biomaterials 2019, 188, 130–143. [Google Scholar] [CrossRef] [PubMed]
- Ying, G.L.; Jiang, N.; Maharjan, S.; Yin, Y.X.; Chai, R.R.; Cao, X.; Yang, J.Z.; Miri, A.K.; Hassan, S.; Zhang, Y.S. Aqueous two-phase emulsion bioink-enabled 3D bioprinting of porous hydrogels. Adv. Mater. 2018, 30, e1805460. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Yan, M.; Wang, Y.; Fu, J.; Suo, H. 3D bioprinting of low-concentration cell-laden gelatin methacrylate (gelma) bioinks with a two-step cross-linking strategy. ACS Appl. Mater. Interfaces 2018, 10, 6849–6857. [Google Scholar] [CrossRef] [PubMed]
- Tan, G.; Zhou, L.; Ning, C.; Tan, Y.; Ni, G.; Liao, J.; Yu, P.; Chen, X. Biomimetically-mineralized composite coatings on titanium functionalized with gelatin methacrylate hydrogels. Appl. Surf. Sci. 2013, 279, 293–299. [Google Scholar] [CrossRef]
- Ramon-Azcon, J.; Ahadian, S.; Obregon, R.; Camci-Unal, G.; Ostrovidov, S.; Hosseini, V.; Kaji, H.; Ino, K.; Shiku, H.; Khademhosseini, A.; et al. Gelatin methacrylate as a promising hydrogel for 3D microscale organization and proliferation of dielectrophoretically patterned cells. Lab Chip 2012, 12, 2959–2969. [Google Scholar] [CrossRef]
- Guillen, G.R.; Pan, Y.; Li, M.; Hoek, E.M.V. Preparation and Characterization of Membranes Formed by Nonsolvent Induced Phase Separation: A Review. Ind. Eng. Chem. Res. 2011, 50, 3798–3817. [Google Scholar] [CrossRef]
- Lin, X.; Tan, L.; Zhang, Q.; Yang, K.; Hu, Z.; Qiu, J.; Cai, Y. The in vitro degradation process and biocompatibility of a ZK60 magnesium alloy with a forsterite-containing micro-arc oxidation coating. Acta Biomater. 2013, 9, 8631–8642. [Google Scholar] [CrossRef]
- Shang, W.; Wu, F.; Wang, Y.; Baboukani, A.R.; Jiang, J. Corrosion Resistance of Micro-Arc Oxidation/Graphene Oxide Composite Coatings on Magnesium Alloys. ACS Omega 2020, 5, 7262–7270. [Google Scholar] [CrossRef]


| Sample | Processing | The Number of Dip-Coating |
|---|---|---|
| 1#, MAO-WE43 | MAO | 0 |
| 2#, 1GelMA/MAO-WE43 | MAO + dip-coating | 1 |
| 3#, 5GelMA/MAO-WE43 | MAO + dip-coating | 5 |
| 4#, 10GelMA/MAO-WE43 | MAO + dip-coating | 10 |
| 5#, 15GelMA/MAO-WE43 | MAO + dip-coating | 15 |
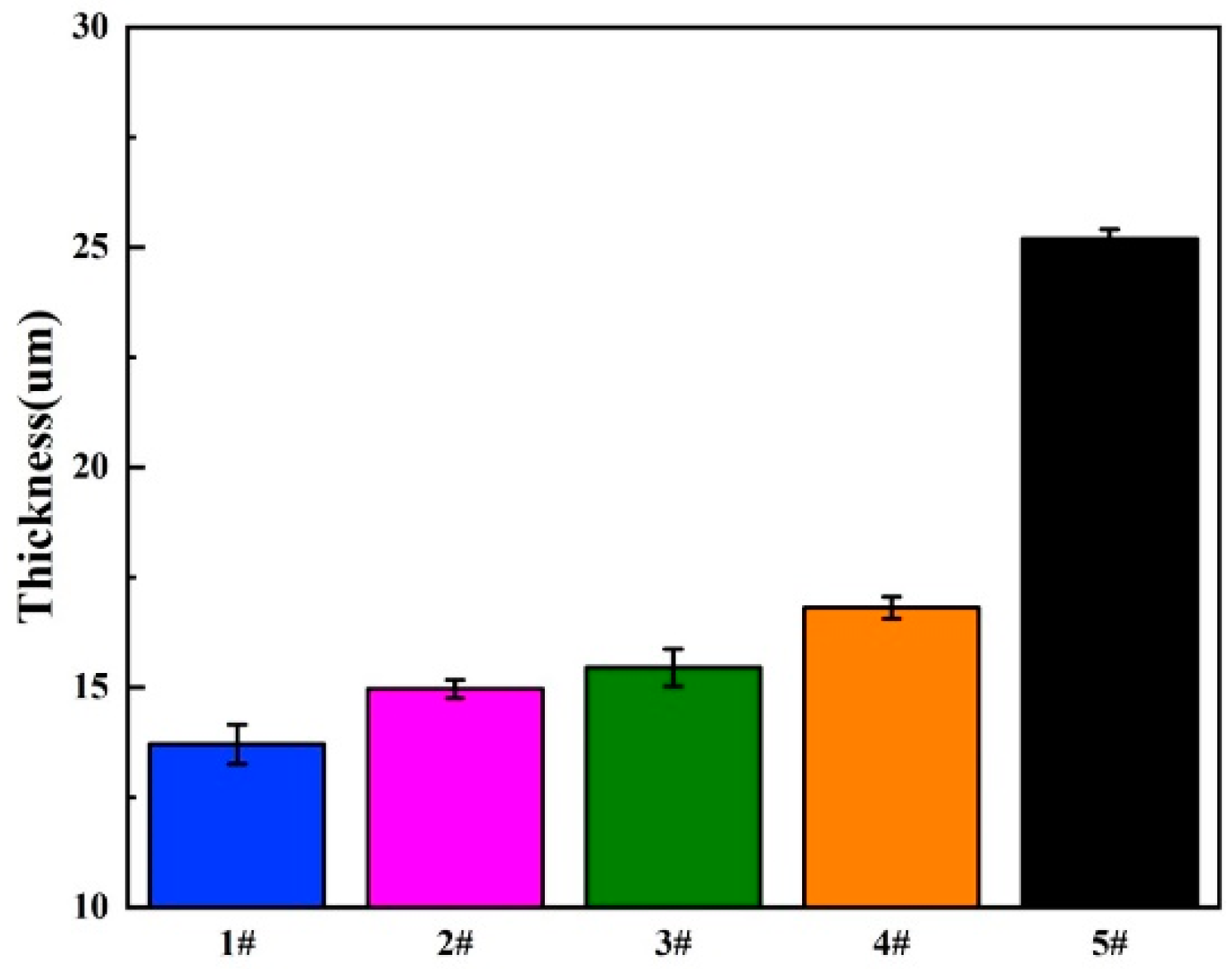
| Sample | The Number of Dip Coating | Thickness (um) |
|---|---|---|
| 1# | 0 | 13.7 |
| 2# | 1 | 14.9 |
| 3# | 5 | 15.4 |
| 4# | 10 | 16.8 |
| 5# | 15 | 25.2 |
| Sample | Processing Mode | Beta A (mV) | Beta C (mV) | Icorr (A/cm2) | Ecorr (V) | CorrRate (MPY) | R (omega) |
|---|---|---|---|---|---|---|---|
| 1# | MAO | 49.402 | 148.65 | 1.65 × 10−6 | 1.5079 | 1.44470 | 9.8 × 106 |
| 2# | MAO + 1GelMA | 191.63 | 189.42 | 5.40 × 10−8 | 1.6057 | 0.047302 | 7.7 × 108 |
| 3# | MAO + 5GelMA | 80.638 | 146.38 | 3.33 × 10−7 | 1.5437 | 0.29139 | 6.8 × 107 |
| 4# | MAO + 10GelMA | 408.37 | 337.01 | 1.60 × 10−6 | 1.5718 | 1.40000 | 5.0 × 107 |
| 5# | MAO + 15GelMA | 110.14 | 197.61 | 2.10 × 10−7 | 1.5480 | 0.18401 | 1.5 × 108 |
| Sample | 1 Day | 3 Days | 5 Days | |||
|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | |
| 1# | 1.647 | 0.144 | 1.372 | 0.551 | 2.222 | 1.726 |
| 2# | 2.430 | 0.304 | 4.472 | 1.418 | 8.705 | 7.234 |
| 3# | 3.155 | 0.742 | 4.005 | 2.570 | 5.355 | 4.594 |
| 4# | 3.063 | 0.355 | 4.922 | 0.850 | 9.355 | 5.887 |
| 5# | 2.897 | 0.379 | 4.422 | 1.242 | 8.538 | 1.754 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Weng, W.; Wu, W.; Yu, X.; Sun, M.; Lin, Z.; Ibrahim, M.; Yang, H. Effect of GelMA Hydrogel Coatings on Corrosion Resistance and Biocompatibility of MAO-Coated Mg Alloys. Materials 2020, 13, 3834. https://doi.org/10.3390/ma13173834
Weng W, Wu W, Yu X, Sun M, Lin Z, Ibrahim M, Yang H. Effect of GelMA Hydrogel Coatings on Corrosion Resistance and Biocompatibility of MAO-Coated Mg Alloys. Materials. 2020; 13(17):3834. https://doi.org/10.3390/ma13173834
Chicago/Turabian StyleWeng, Wenxian, Weiwei Wu, Xiaoming Yu, Mingyue Sun, Zhensheng Lin, Muhammad Ibrahim, and Huazhe Yang. 2020. "Effect of GelMA Hydrogel Coatings on Corrosion Resistance and Biocompatibility of MAO-Coated Mg Alloys" Materials 13, no. 17: 3834. https://doi.org/10.3390/ma13173834
APA StyleWeng, W., Wu, W., Yu, X., Sun, M., Lin, Z., Ibrahim, M., & Yang, H. (2020). Effect of GelMA Hydrogel Coatings on Corrosion Resistance and Biocompatibility of MAO-Coated Mg Alloys. Materials, 13(17), 3834. https://doi.org/10.3390/ma13173834





