Organocatalytic Name Reactions Enabled by NHCs
Abstract
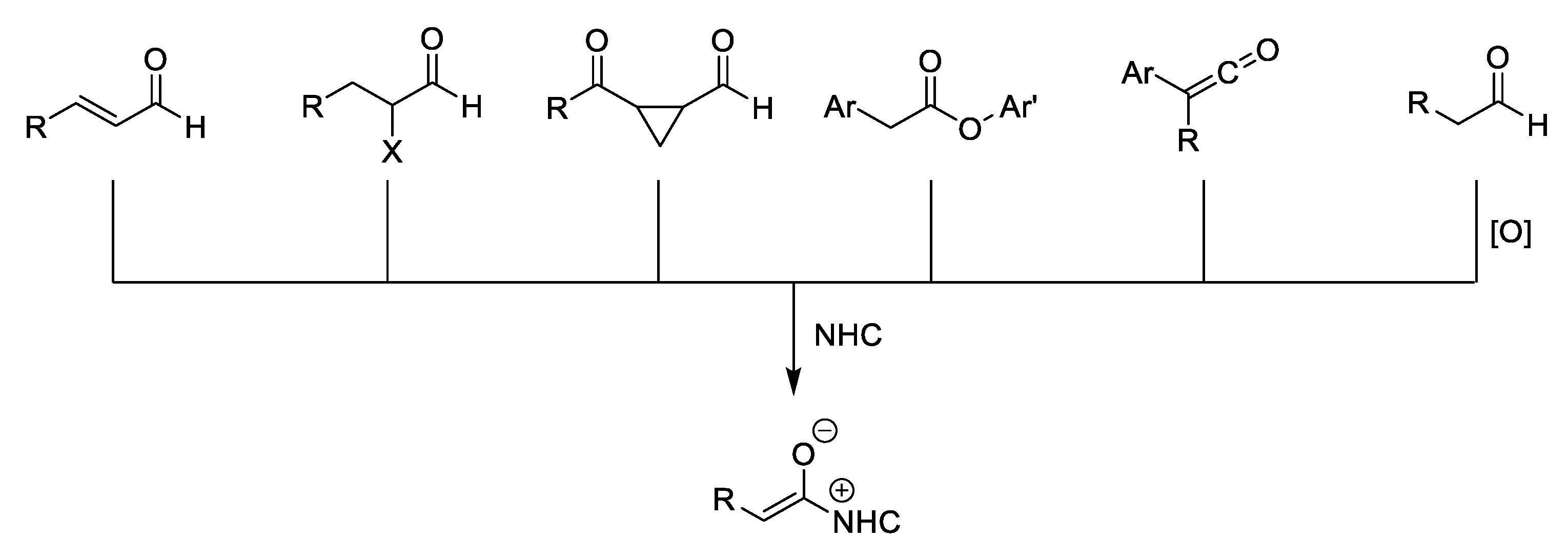
:1. Introduction
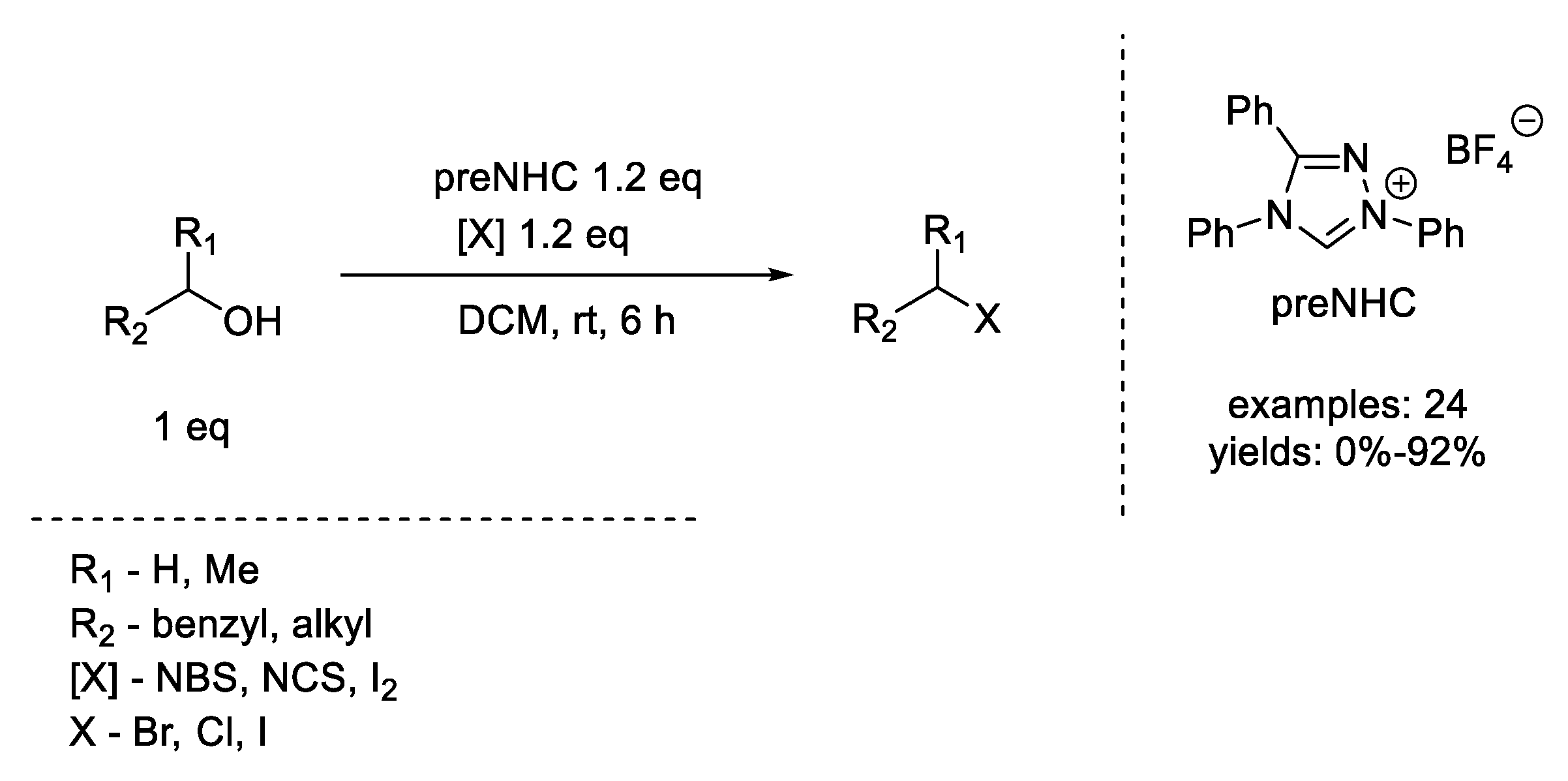
2. Appel Reaction
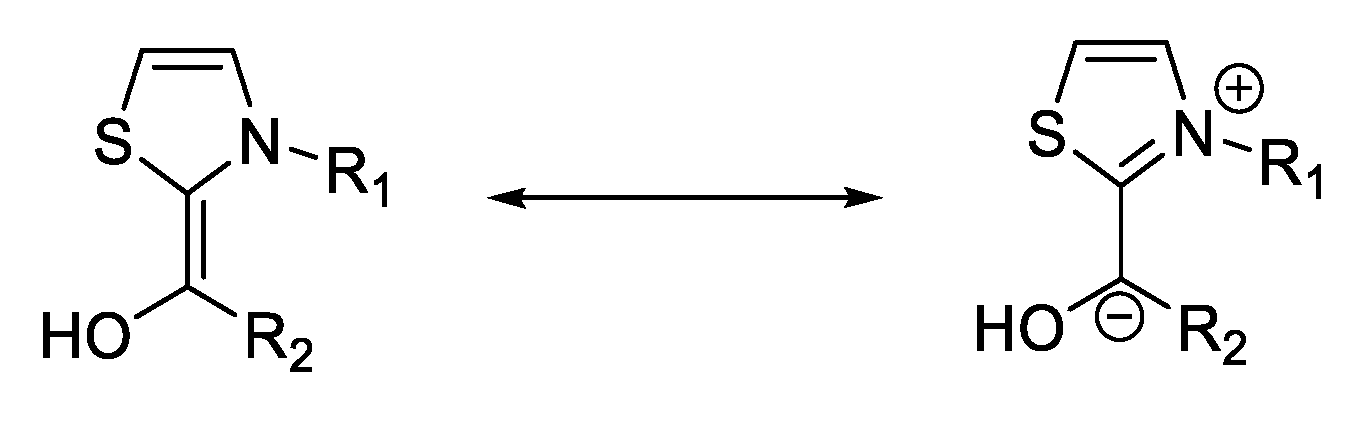
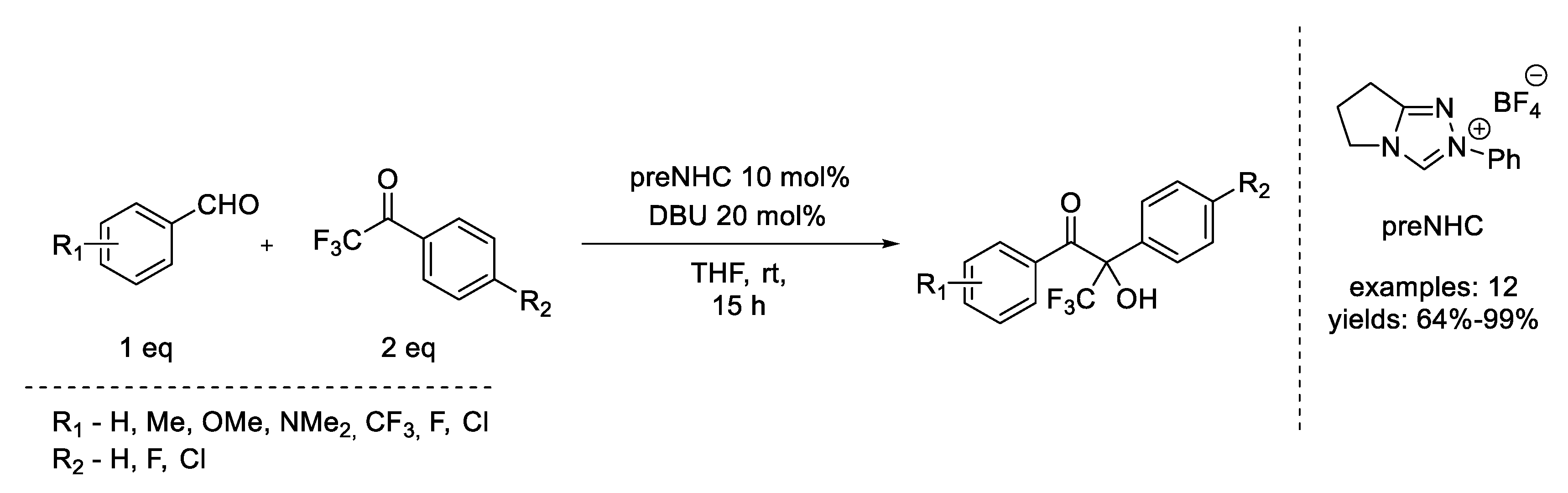
3. Benzoin Condensation
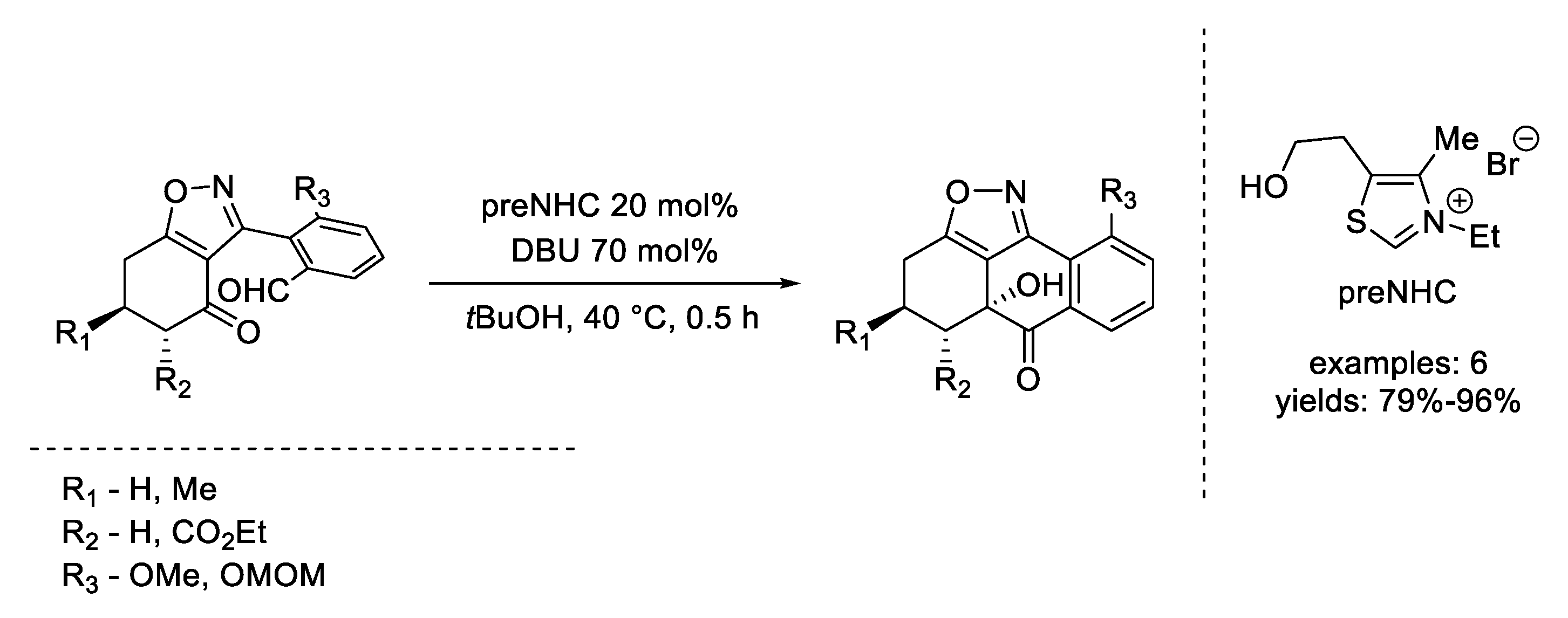
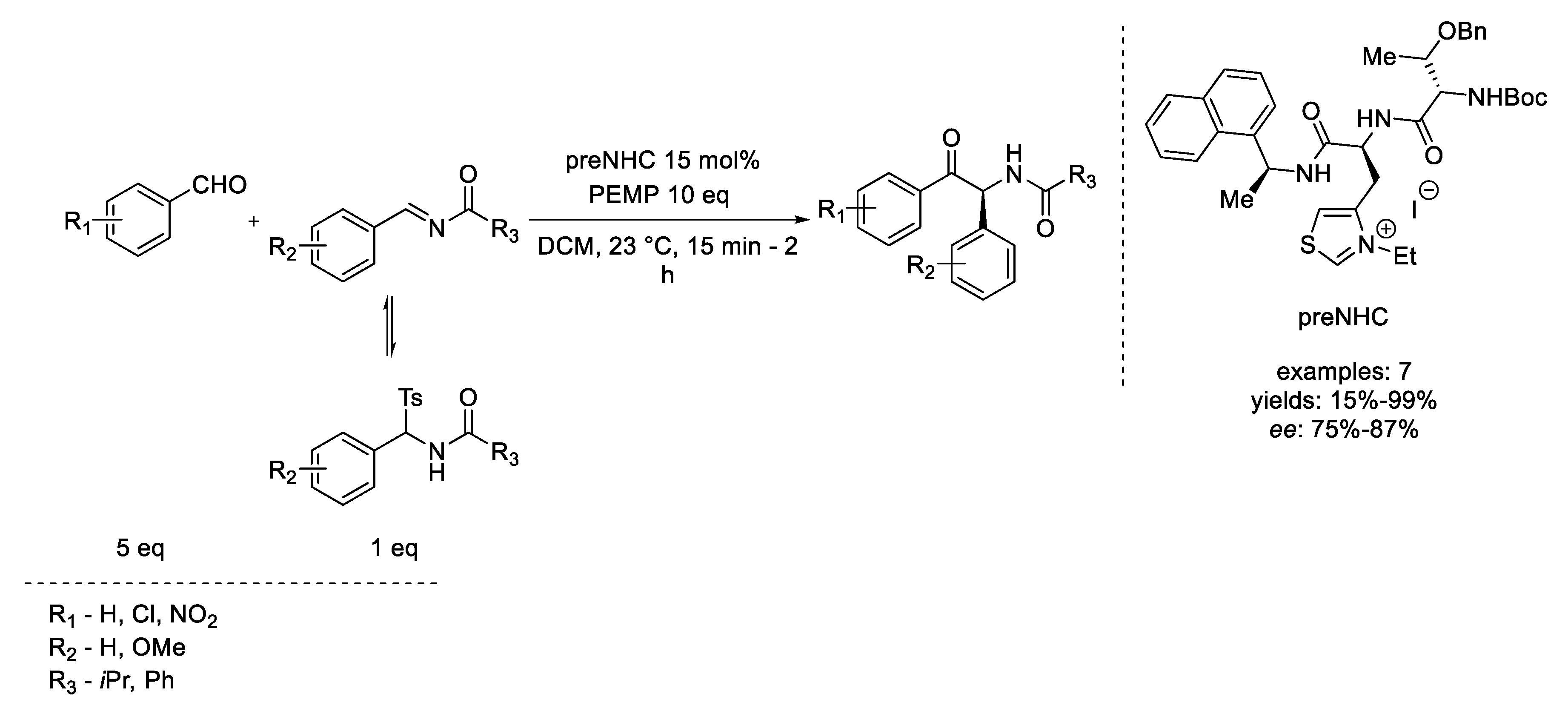
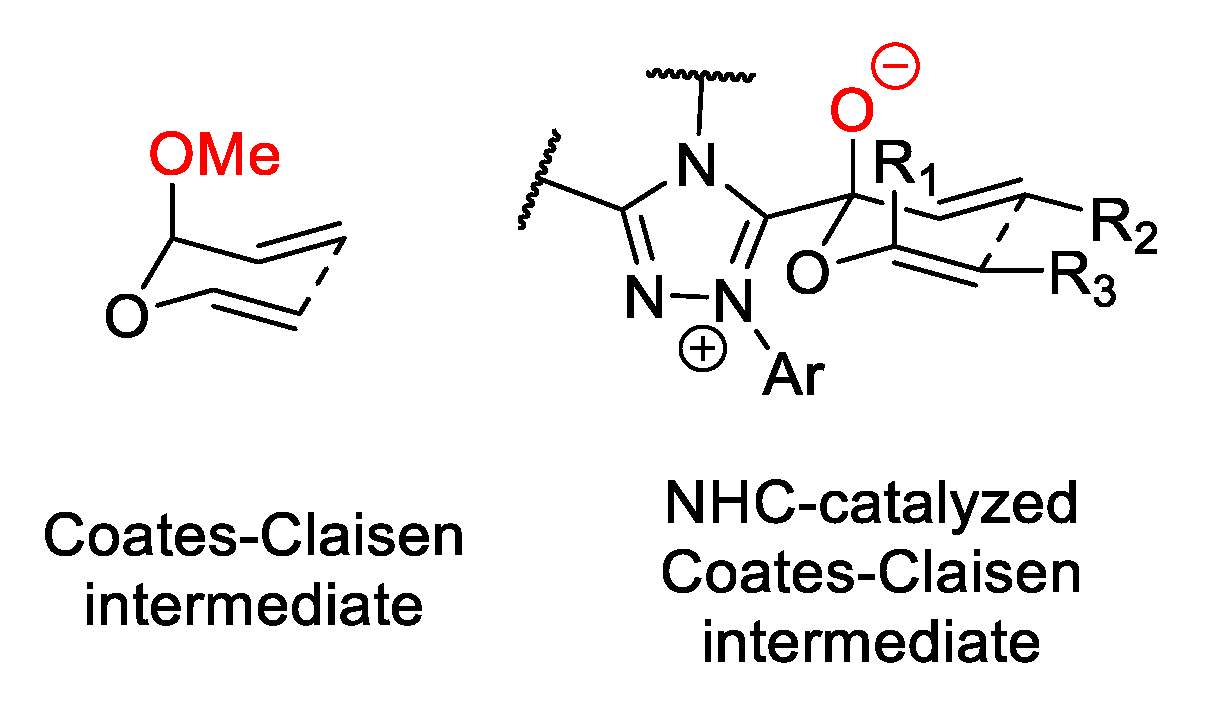
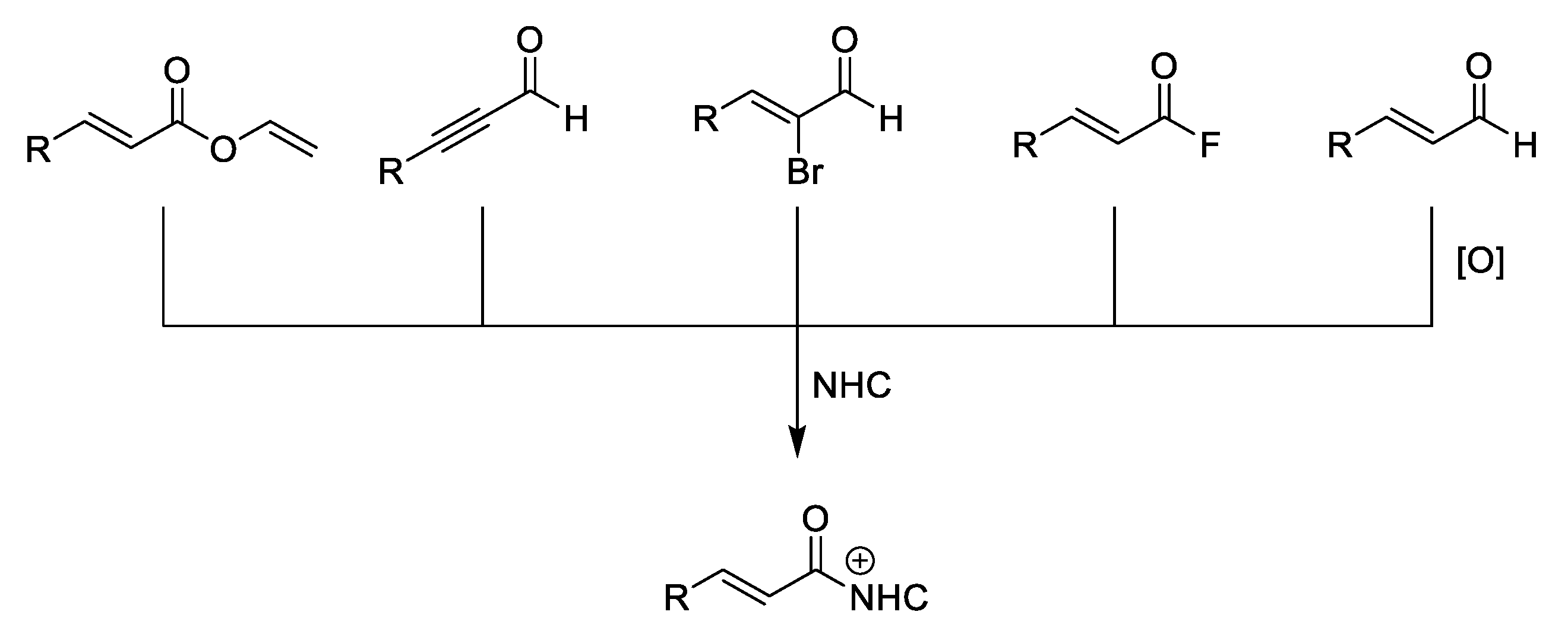
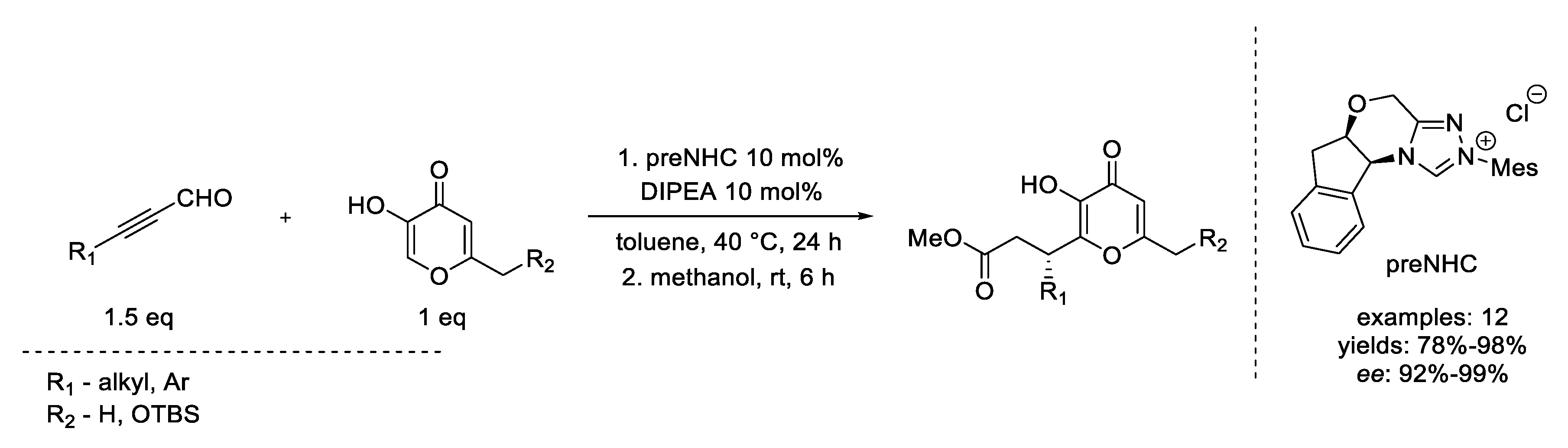
4. Coates–Claisen and Ireland–Coates–Claisen Rearrangements
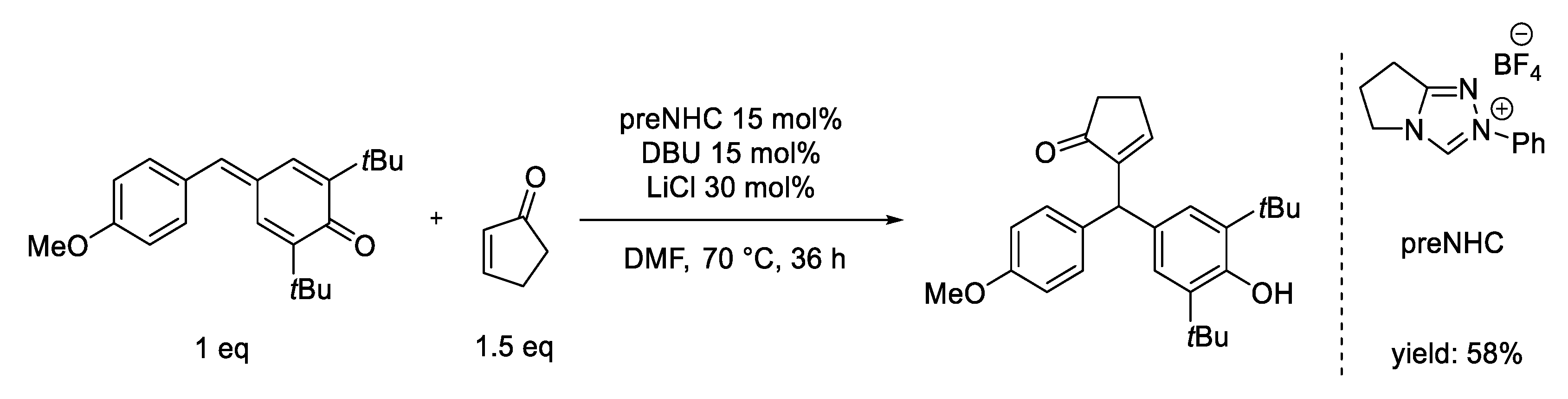
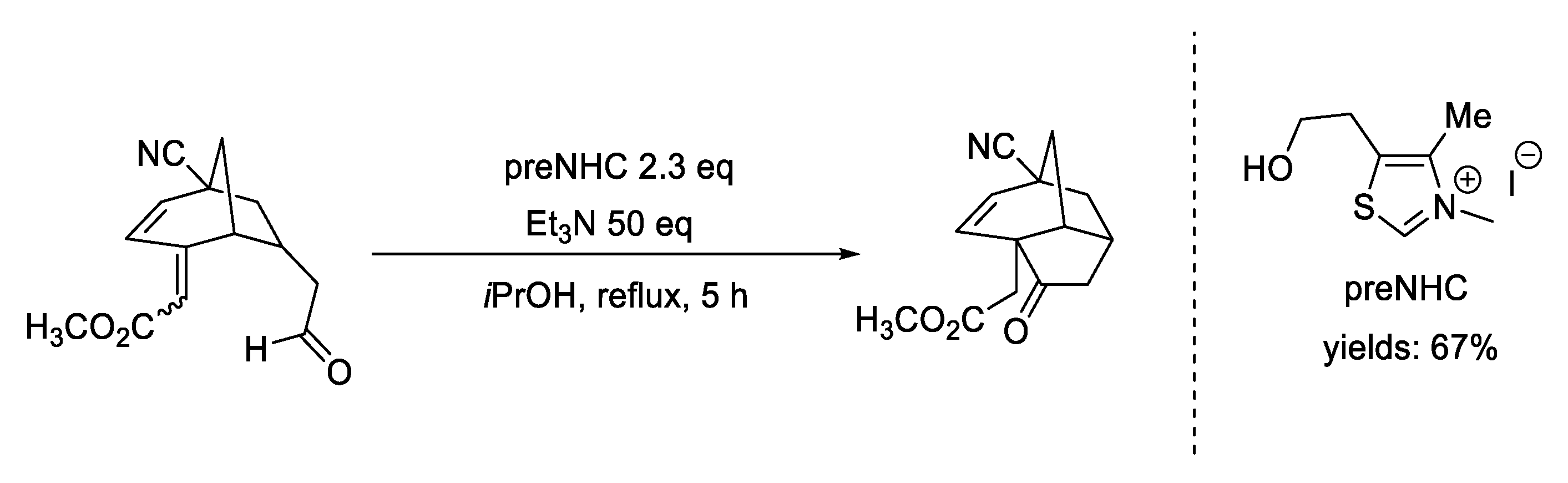
5. oxy-Cope Rearrangement
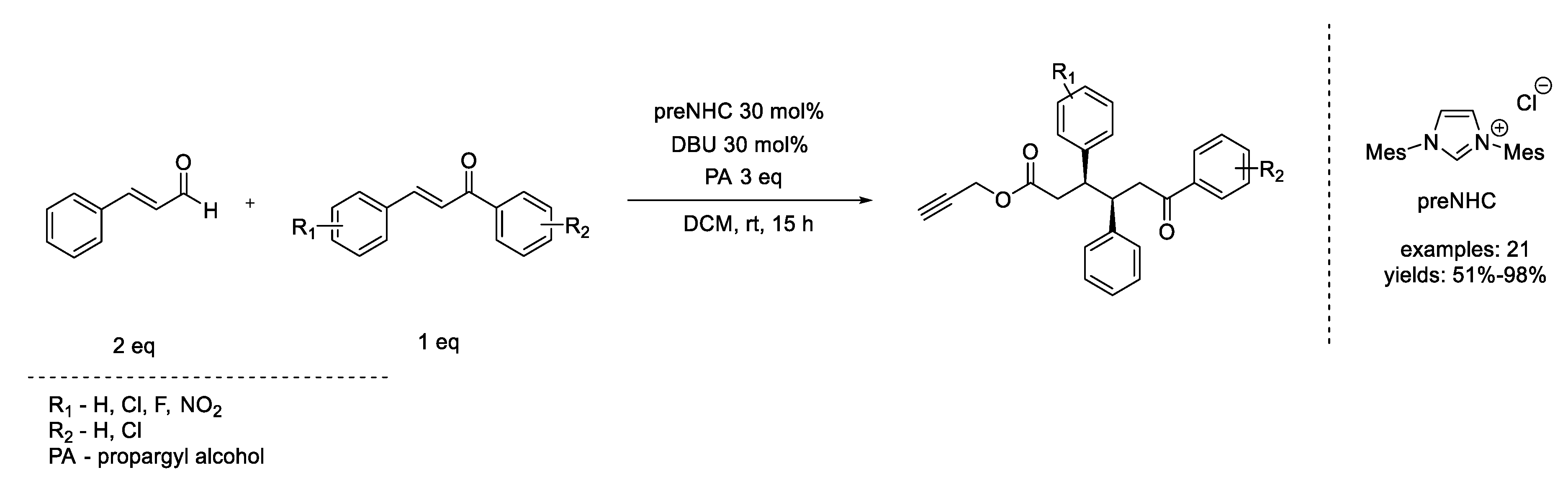
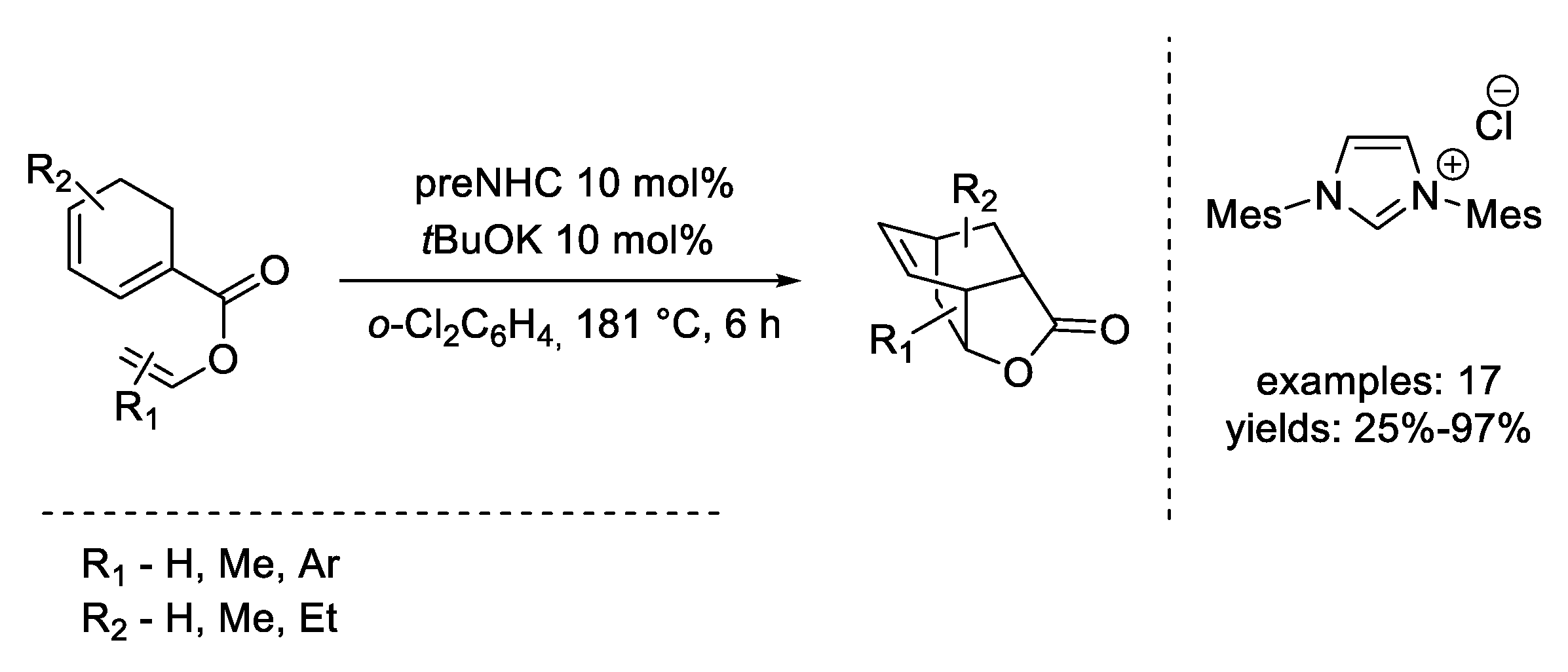
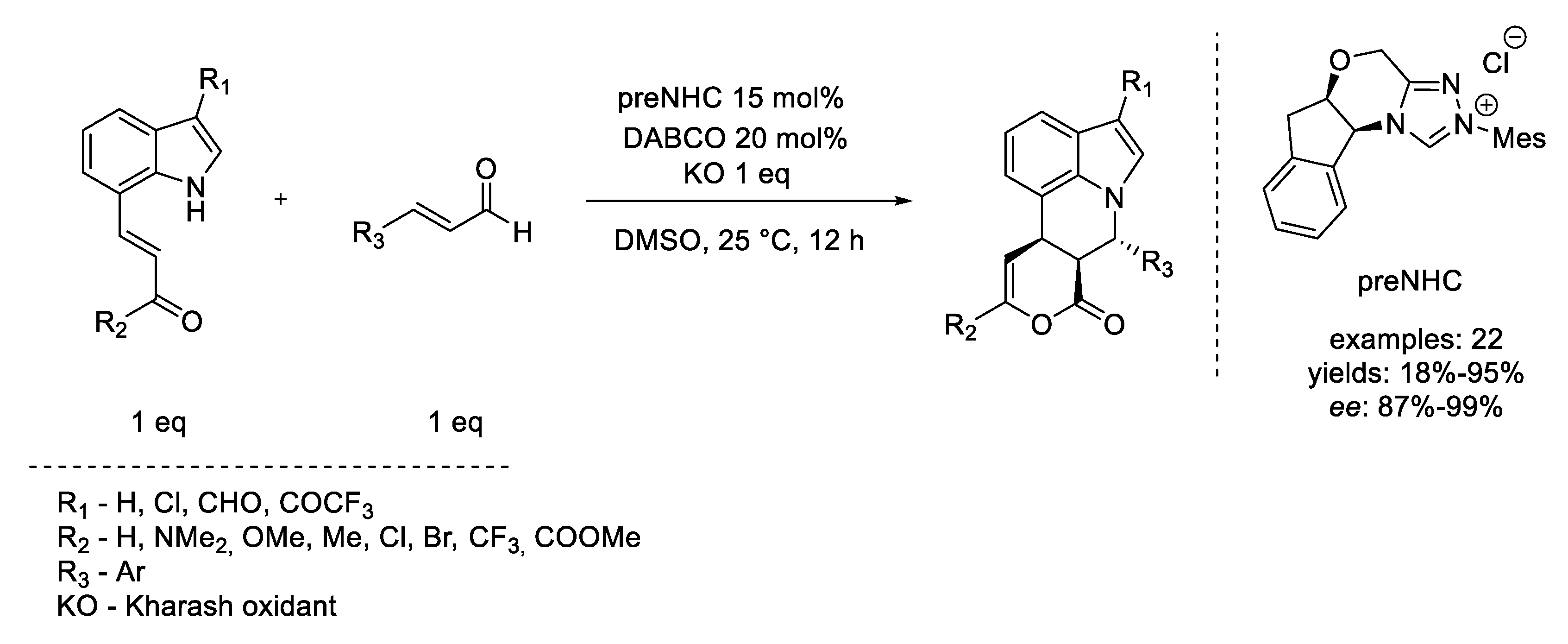
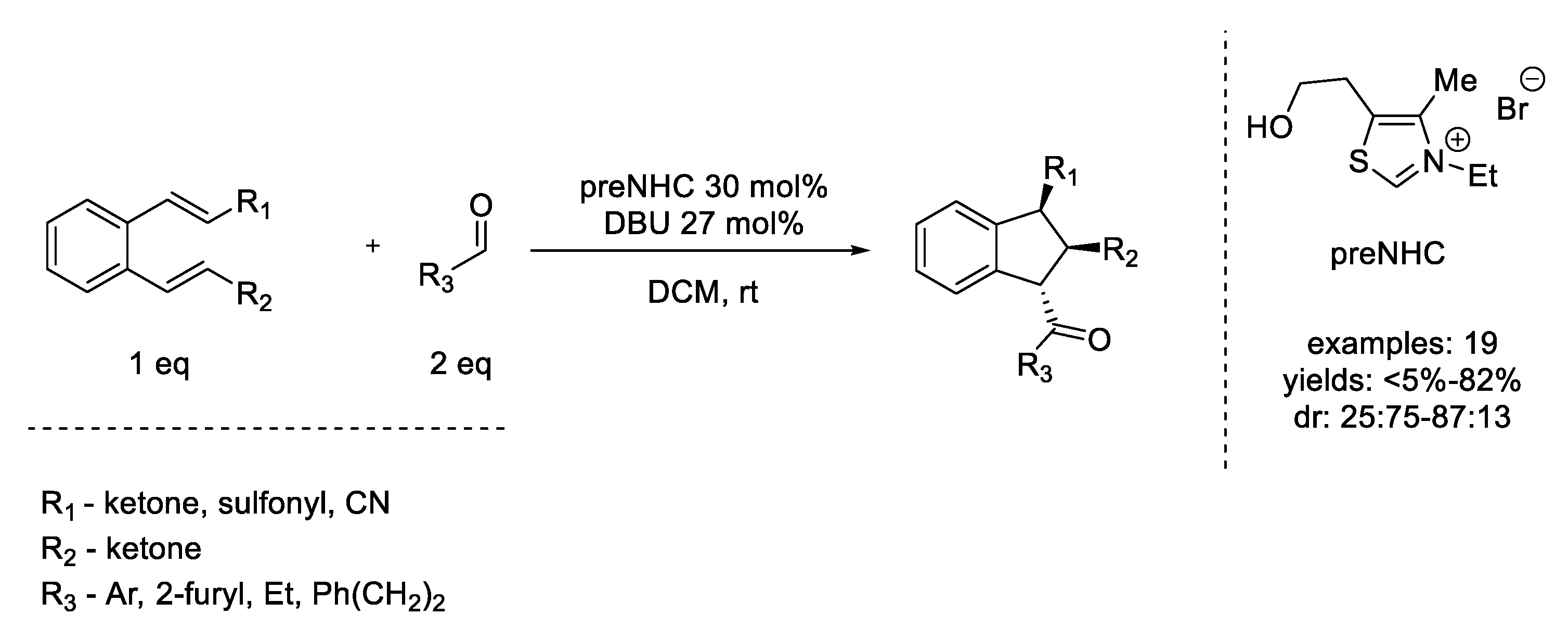
6. Diels–Alder Reaction
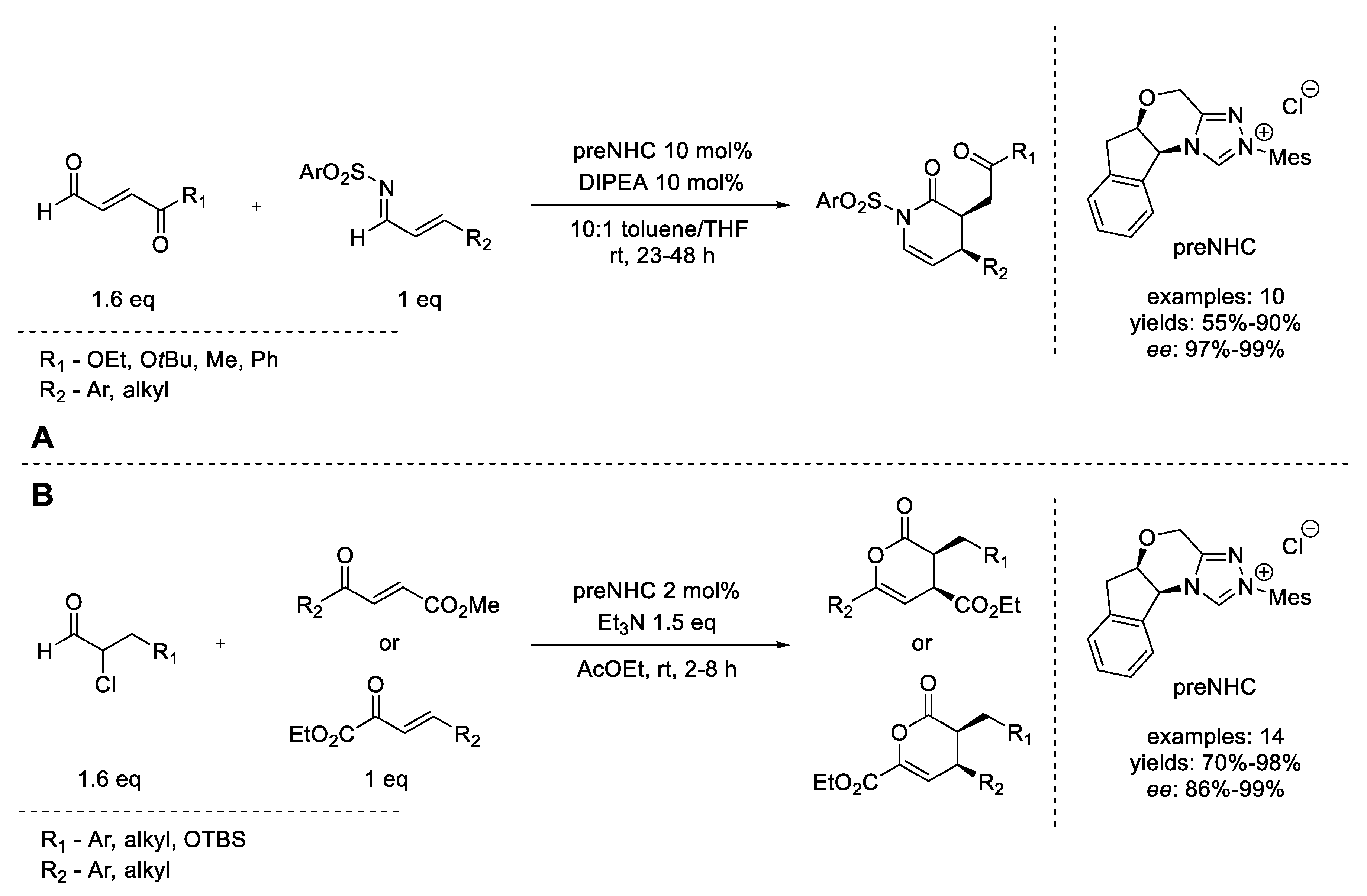
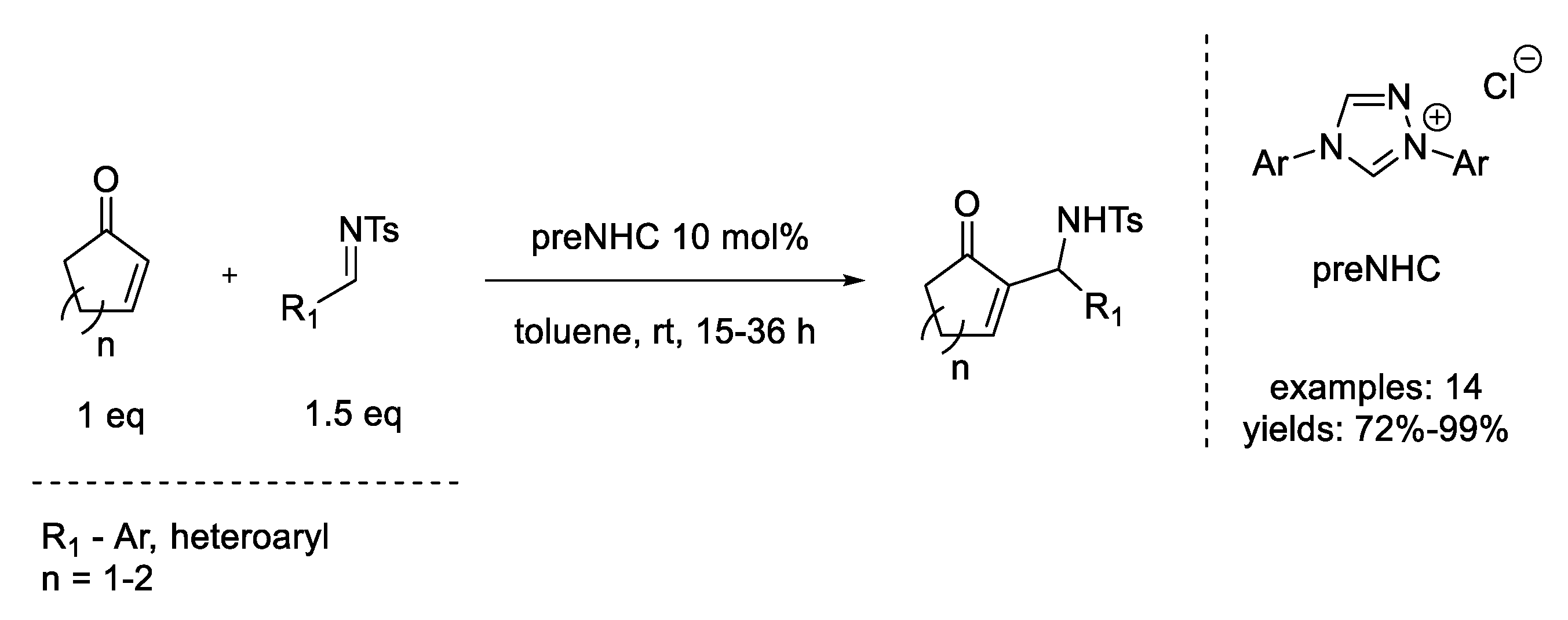
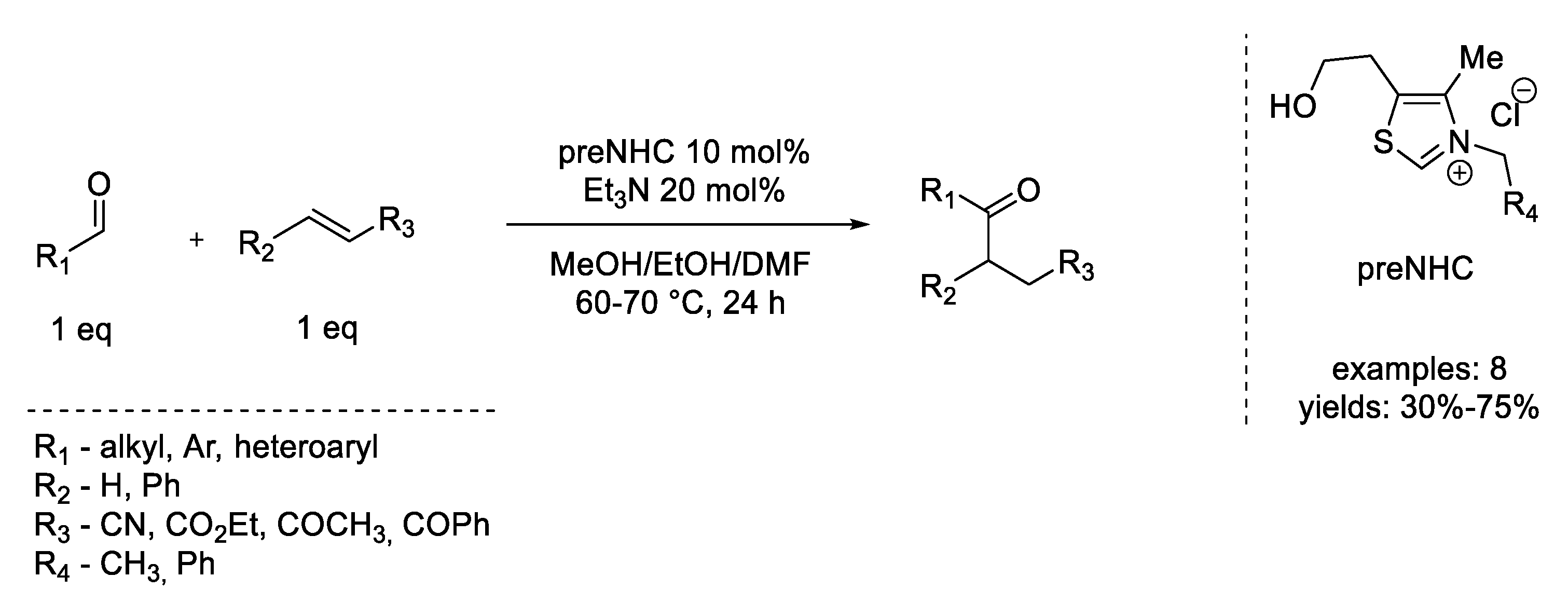
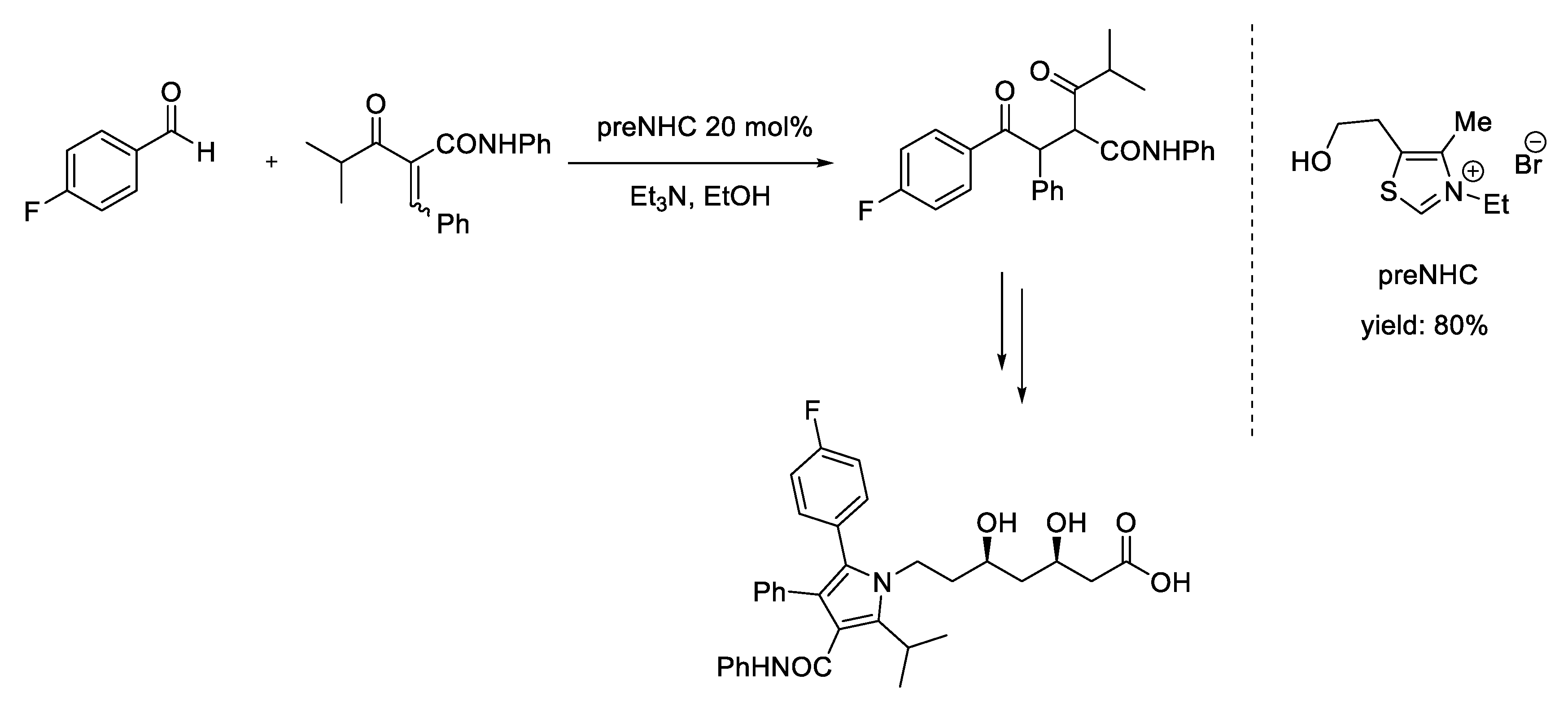
7. Michael Addition
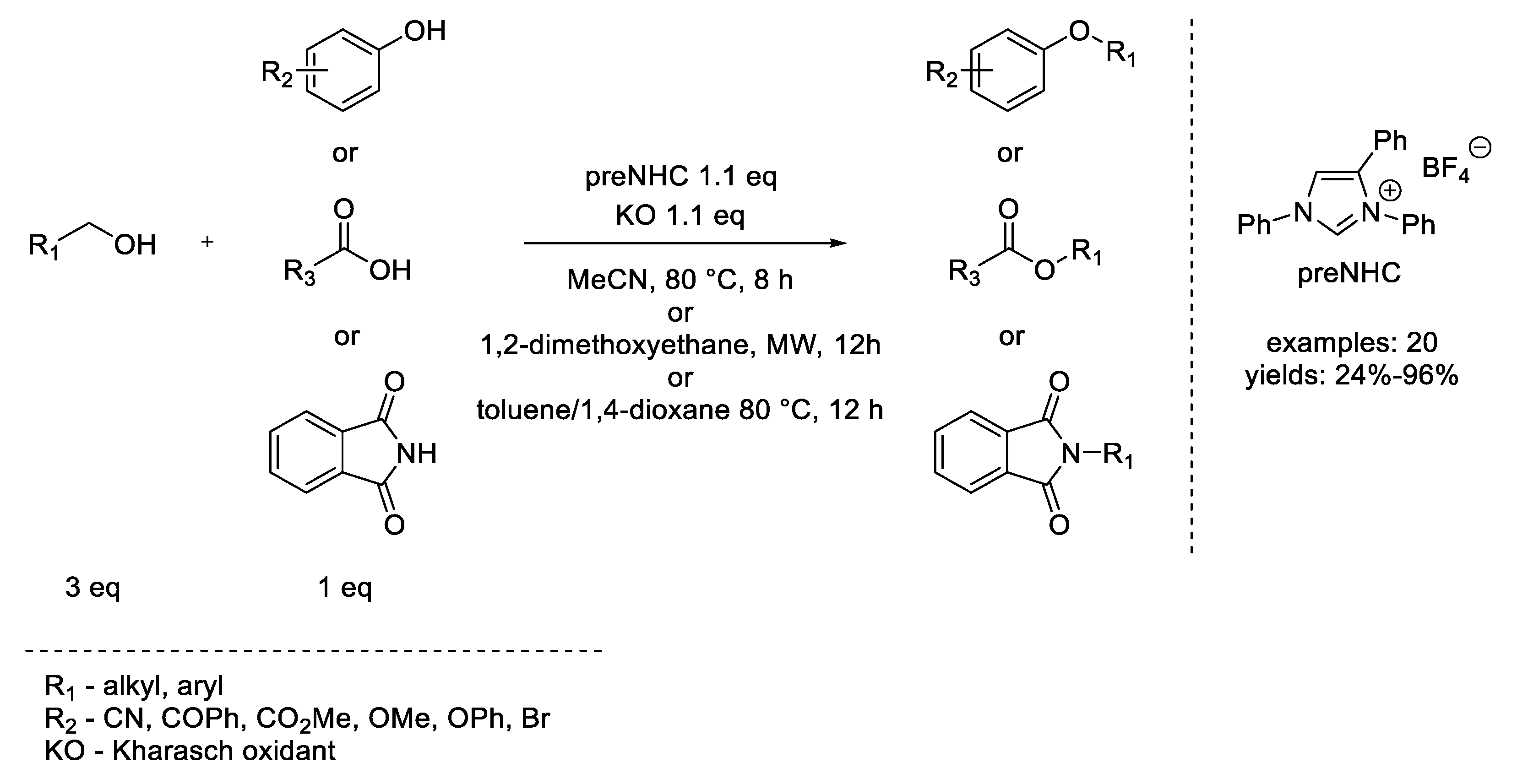
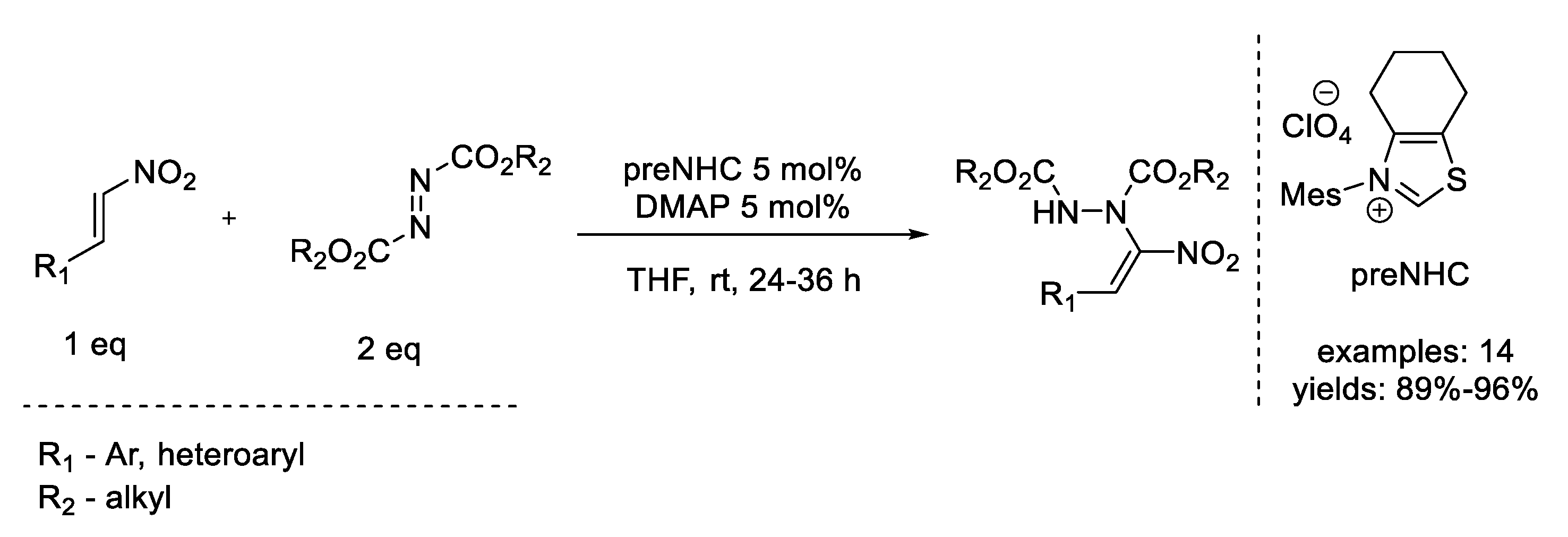
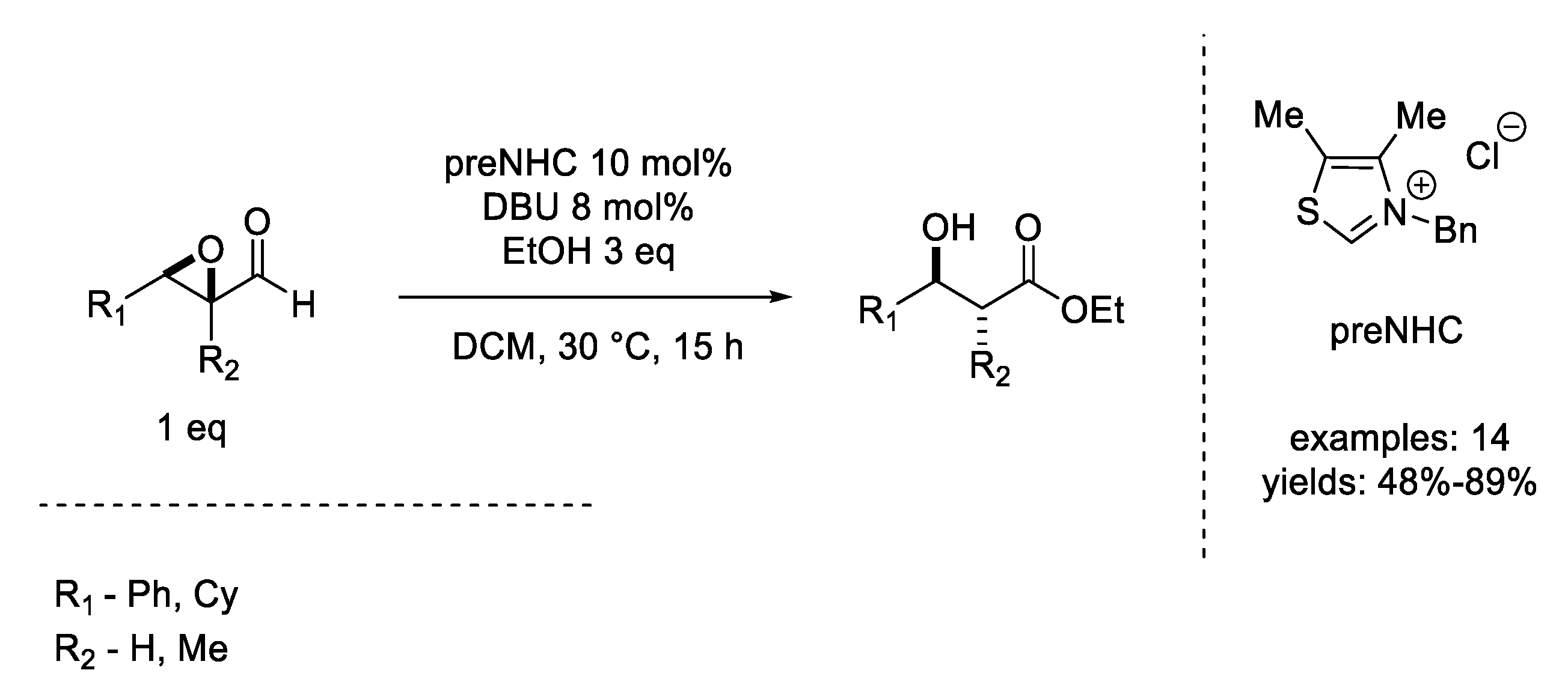
8. Mitsunobu Reaction
9. Morita–Baylis–Hillman Reaction
10. Rauhut–Currier Reaction
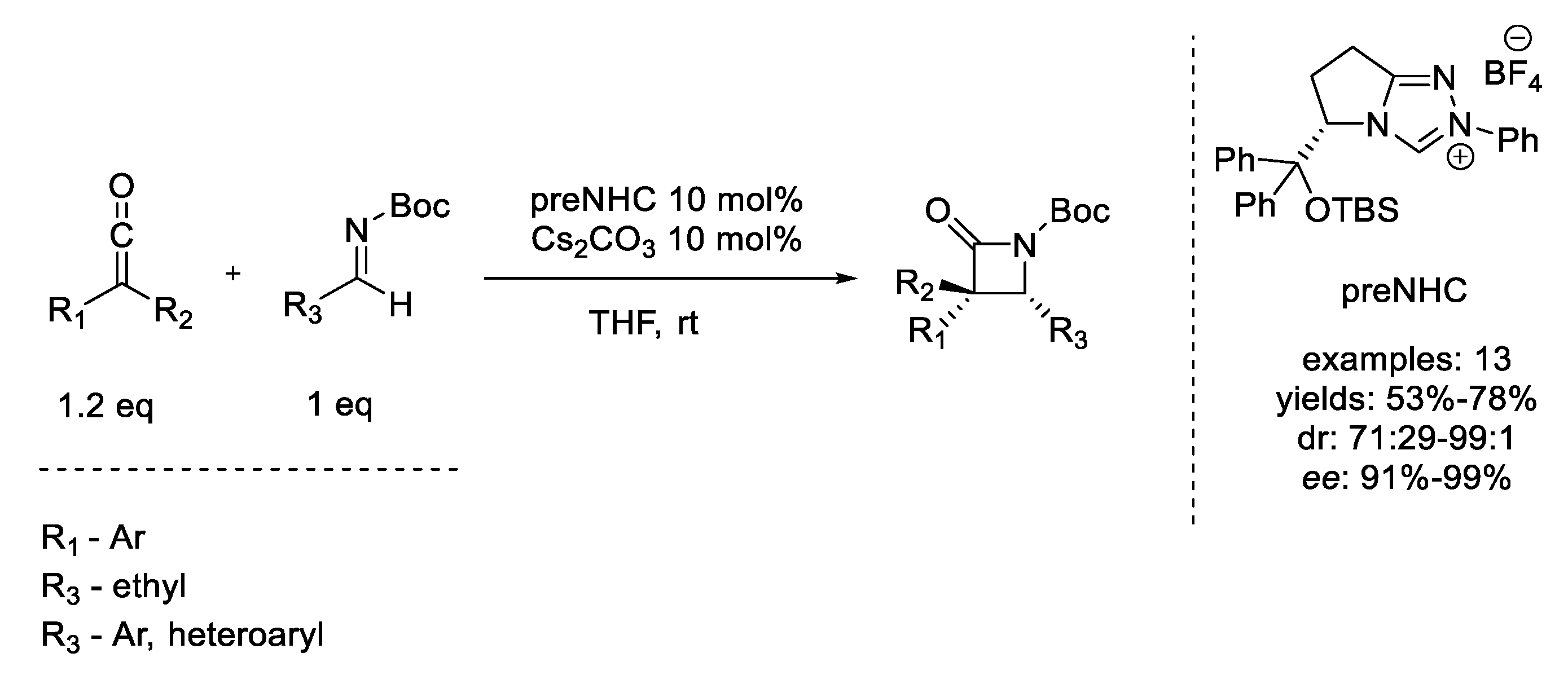
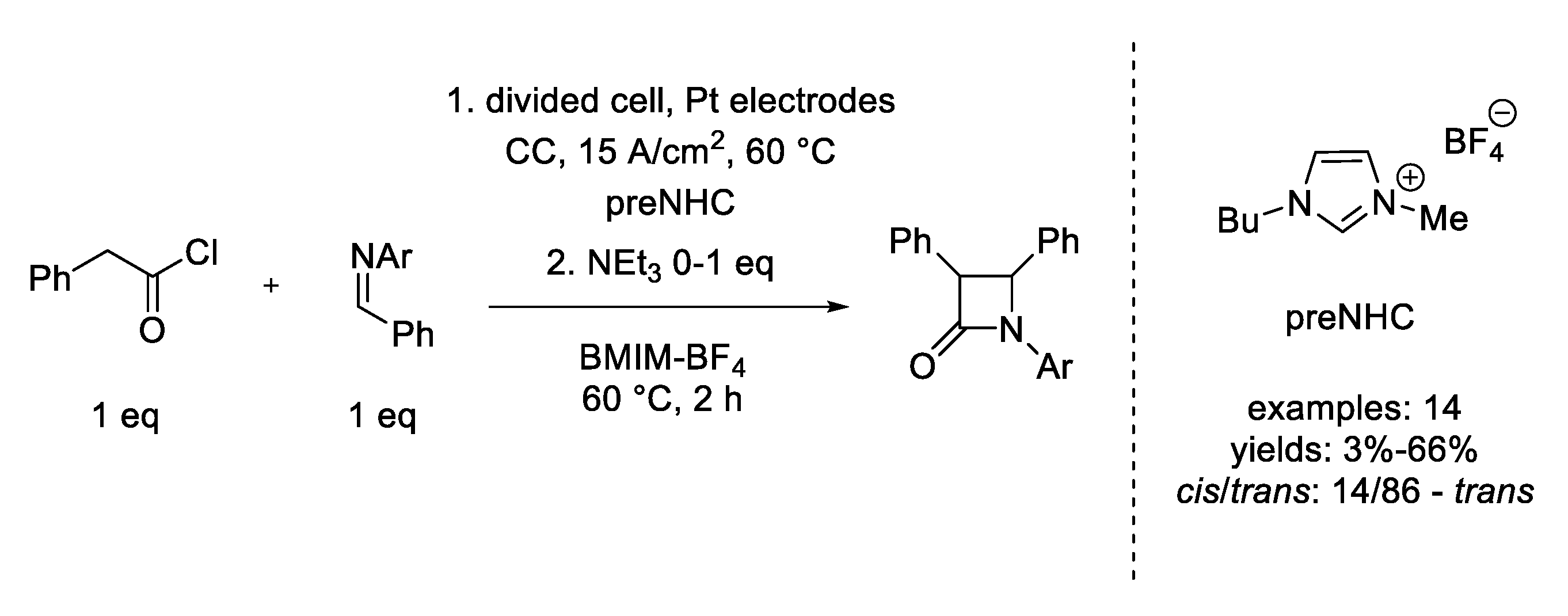
11. Staudinger Cycloaddition
12. Stetter Reaction
13. Wallach Reaction
14. Summary
Author Contributions
Funding
Conflicts of Interest
Acronyms
| CC | constant current |
| DBU | 1,8-diazabicycloundec-7-ene |
| DCM | dichloromethane |
| DIPEA | diisopropylamine |
| DMSO | dimethylsulfoxide |
| KHMDS | potassium hexamethyldisilazide |
| KO | Kharasch oxidant (3,3’,5,5’-tetra-tert-butyldiphenoquinone) |
| NHC | N-heterocyclic carbene |
| PEMP | 1,2,2,6,6-pentamethylpiperidine |
| preNHC | NHC precatalyst |
| THFTMS | TetrahydrofuranTrimethylsilyl group |
References
- Flanigan, D.M.; Romanov-Michailidis, F.; White, N.A.; Rovis, T. Organocatalytic Reactions Enabled by N-Heterocyclic Carbenes. Chem. Rev. 2015, 115, 9307–9387. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Douglas, J.; Churchill, G.; Smith, A.D. NHCs in asymmetric organocatalysis: Recent advances in azolium enolate generation and reactivity. Synthesis 2012, 44, 2295–2309. [Google Scholar]
- Bugaut, X.; Glorius, F. Organocatalytic umpolung: N-heterocyclic carbenes and beyond. Chem. Soc. Rev. 2012, 41, 3511–3522. [Google Scholar] [CrossRef] [PubMed]
- Izquierdo, J.; Hutson, G.E.; Cohen, D.T.; Scheidt, K.A. A continuum of progress: Applications of N-hetereocyclic carbene catalysis in total synthesis. Angew. Chem.-Int. Ed. 2012, 51, 11686–11698. [Google Scholar] [CrossRef] [Green Version]
- Nair, V.; Menon, R.S.; Biju, A.T.; Sinu, C.R.; Paul, R.R.; Jose, A.; Sreekumar, V. Employing homoenolates generated by NHC catalysis in carbon-carbon bond-forming reactions: State of the art. Chem. Soc. Rev. 2011, 40, 5336–5346. [Google Scholar] [CrossRef]
- Nguyen, X.B.; Nakano, Y.; Lupton, D.W. Polarity Inversion Catalysis by the 1,4-Addition of N-Heterocyclic Carbenes. Aust. J. Chem. 2020, 73, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Biju, A.T. N-Heterocyclic Carbenes in Organocatalysis; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2018; Volume 2019, ISBN 9783527809042. [Google Scholar]
- Vora, H.U.; Wheeler, P.; Rovis, T. Exploiting acyl and enol azolium intermediates via N-heterocyclic carbene-catalyzed reactions of α-reducible aldehydes. Adv. Synth. Catal. 2012, 354, 1617–1639. [Google Scholar] [CrossRef]
- Albanese, D.C.M.; Gaggero, N. An overview on the N-heterocyclic carbene-catalyzed Aza-Benzoin condensation reaction. Catalysts 2018, 8, 181. [Google Scholar] [CrossRef] [Green Version]
- Hussein, M.A.; Nguyen, T.V. Promotion of Appel-type reactions by N-heterocyclic carbenes. Chem. Commun. 2019, 55, 7962–7965. [Google Scholar] [CrossRef]
- Glorius, F. N-heterocyclic carbenes in catalysis—An introduction. Top. Organomet. Chem. 2006, 21, 1–20. [Google Scholar]
- Kantchev, E.A.B.; O’Brien, C.J.; Organ, M.G. Palladium complexes of N-heterocyclic carbenes as catalysts for cross-coupling reactions—A synthetic chemist’s perspective. Angew. Chem.-Int. Ed. 2007, 46, 2768–2813. [Google Scholar] [CrossRef] [PubMed]
- Appel, R. Tertiary Phosphane/Tetrachloromethane, a Versatile Reagent for Chlorination, Dehydration, and P-N Linkage. Angew. Chem. Int. Ed. Engl. 1975, 14, 801–811. [Google Scholar] [CrossRef]
- Wöhler, F.; Liebig, J. Untersuchungen über das Radikal der Benzoesäure. Ann. Pharm. 1832, 3, 249–282. [Google Scholar] [CrossRef]
- Zinin, N. Beiträge zur Kenntniss einiger Verbindungen aus der Benzoylreihe. Ann. Pharm. 1839, 31, 329–332. [Google Scholar] [CrossRef] [Green Version]
- Zinin, N. Ueber einige Zersetzungsprodukte des Bittermandelöls. Ann. Chem. Pharm. 1840, 34, 186–192. [Google Scholar] [CrossRef] [Green Version]
- Lapworth, A. CXXII.-Reactions involving the addition of hydrogen cyanide to carbon compounds. Part II. Cyanohydrins regarded as complex acids. J. Chem. Soc. Trans. 1904, 85, 1206–1214. [Google Scholar] [CrossRef] [Green Version]
- Ukai, T.; Tanaka, R.; Dokawa, T.A. A New Catalyst for Acyloin Condensation. J. Pharm. Soc. Jpn. 1943, 63, 296–300. [Google Scholar]
- Breslow, R. On the Mechanism of Thiamine Action. IV. Evidence from Studies on Model Systems. J. Am. Chem. Soc. 1958, 80, 3719–3726. [Google Scholar] [CrossRef]
- Li, C.Y.; Kuo, Y.Y.; Tsai, J.H.; Yap, G.P.A.; Ong, T.G. Amine-linked N-heterocyclic carbenes: The importance of an pendant free-amine auxiliary in assisting the catalytic reaction. Chem.-An Asian J. 2011, 6, 1520–1524. [Google Scholar] [CrossRef]
- Iwamoto, K.I.; Hamaya, M.; Hashimoto, N.; Kimura, H.; Suzuki, Y.; Sato, M. Benzoin reaction in water as an aqueous medium catalyzed by benzimidazolium salt. Tetrahedron Lett. 2006, 47, 7175–7177. [Google Scholar] [CrossRef]
- Baragwanath, L.; Rose, C.A.; Zeitler, K.; Connon, S.J. Highly enantioselective benzoin condensation reactions involving a bifunctional protic pentafluorophenyl-substituted triazolium precatalyst. J. Org. Chem. 2009, 74, 9214–9217. [Google Scholar] [CrossRef]
- Stetter, H.; Kuhlmann, H. Acyloin Condensation By Thiazolium Ion Catalysis: Butyroin. Org. Synth. 1984, 62, 170. [Google Scholar]
- Iwamoto, K.I.; Kimura, H.; Oike, M.; Sato, M. Methylene-bridged bis(benzimidazolium) salt as a highly efficient catalyst for the benzoin reaction in aqueous media. Org. Biomol. Chem. 2008, 6, 912–915. [Google Scholar] [CrossRef] [PubMed]
- Miyashita, A.; Suzuki, Y.; Iwamoto, K.; Higashino, T. Catalytic Action of Azolium Salts. VI. Preparation of Benzoins and Acyloins by Condensation of Aldehydes Catalyzed by Azolium Salts. Chem. Pharm. Bull. 1994, 42, 2633–2635. [Google Scholar] [CrossRef] [Green Version]
- Teles, J.H.; Melder, J.-P.; Ebel, K.; Schneider, R.; Gehrer, E.; Harder, W.; Brode, S.; Enders, D.; Breuer, K.; Raabe, G. The Chemistry of Stable Carbenes. Helv. Chim. Acta 1996, 79, 61–83. [Google Scholar]
- Sheehan, J.C.; Hunneman, D.H. Homogeneous Asymmetric Catalysis. J. Am. Chem. Soc. 1966, 88, 3666–3667. [Google Scholar] [CrossRef]
- Yano, Y.; Tamura, Y.; Tagaki, W. Structural Aspects of Catalytic Activities of Thiazolium Salts in Benzoin Condensation Reaction in Methanol. Bull. Chem. Soc. Jpn. 1980, 53, 740–744. [Google Scholar] [CrossRef] [Green Version]
- Knight, R.L.; Leeper, F.J. Synthesis of and asymmetric induction by chiral bicyclic thiazolium salts. Tetrahedron Lett. 1997, 38, 3611–3614. [Google Scholar] [CrossRef]
- Gerhard, A.U.; Leeper, F.J. Synthesis of and asymmetric induction by chiral polycyclic thiazolium salts. Tetrahedron Lett. 1997, 38, 3615–3618. [Google Scholar] [CrossRef]
- Dvorak, C.A.; Rawal, V.H. Catalysis of benzoin condensation by conformationally-restricted chiral bicyclic thiazolium salts. Tetrahedron Lett. 1998, 39, 2925–2928. [Google Scholar] [CrossRef]
- Sheehan, J.C.; Hara, T. Asymmetric Thiazolium Salt Catalysis of the Benzoin Condensation. J. Org. Chem. 1974, 39, 1196–1199. [Google Scholar] [CrossRef]
- Martí, J.; Castells, J.; López-Calahorra, F. Introduction to a rational design of chiral thiazolium salts. Tetrahedron Lett. 1993, 34, 521–524. [Google Scholar] [CrossRef]
- Knight, R.L.; Leeper, F.J. Comparison of chiral thiazolium and triazolium salts as asymmetric catalysts for the benzoin condensation. J. Chem. Soc.-Perkin Trans. 1998, 1, 1891–1893. [Google Scholar] [CrossRef]
- Orlandi, S.; Caporale, M.; Benaglia, M.; Annunziata, R. Synthesis of new enantiomerically pure C1- and C2-symmetric N-alkyl-benzimidazolium and thiazolium salts. Tetrahedron Asymmetry 2003, 14, 3827–3830. [Google Scholar] [CrossRef]
- Tachibana, Y.; Kihara, N.; Takata, T. Asymmetric Benzoin Condensation Catalyzed by Chiral Rotaxanes Tethering a Thiazolium Salt Moiety via the Cooperation of the Component: Can Rotaxane Be an Effective Reaction Field? J. Am. Chem. Soc. 2004, 126, 3438–3439. [Google Scholar] [CrossRef]
- Pesch, J.; Harms, K.; Bach, T. Preparation of axially chiral N,N′-diarylimidazolium and N-arylthiazolium salts and evaluation of their catalytic potential in the benzoin and in the intramolecular stetter reactions. European J. Org. Chem. 2004, 2025–2035. [Google Scholar] [CrossRef]
- Enders, D.; Breuer, K.; Teles, J.H. A novel asymmetric benzoin reaction catalyzed by a chiral triazolium salt: Preliminary communication. Helv. Chim. Acta 1996, 79, 1217–1221. [Google Scholar] [CrossRef]
- Enders, D.; Kallfass, U. An efficient nucleophilic carbene catalyst for the asymmetric benzoin condensation. Angew. Chem.-Int. Ed. 2002, 41, 1743–1745. [Google Scholar] [CrossRef]
- Ma, Y.; Wei, S.; Wu, J.; Yang, F.; Liu, B.; Lan, J.; Yang, S.; You, J. From mono-triazolium salt to bis-triazolium salt: Improvement of the asymmetric intermolecular benzoin condensation. Adv. Synth. Catal. 2008, 350, 2645–2651. [Google Scholar] [CrossRef]
- Enders, D.; Han, J. Synthesis of enantiopure triazolium salts from pyroglutamic acid and their evaluation in the benzoin condensation. Tetrahedron Asymmetry 2008, 19, 1367–1371. [Google Scholar] [CrossRef]
- Brand, J.P.; Siles, J.I.O.; Waser, J. Synthesis of Chiral Bifunctional (Thio)urea N-Heterocyclic Carbenes. Synlett 2010, 6, 881–884. [Google Scholar]
- Soeta, T.; Tabatake, Y.; Inomata, K.; Ukaji, Y. Asymmetric benzoin condensation promoted by chiral triazolium precatalyst bearing a pyridine moiety. Tetrahedron 2012, 68, 894–899. [Google Scholar] [CrossRef]
- Rafiński, Z.; Kozakiewicz, A.; Rafińska, K. Highly efficient synthesis of spirocyclic (1R)-camphor-derived triazolium salts: Application in the catalytic asymmetric benzoin condensation. Tetrahedron 2014, 70, 5739–5745. [Google Scholar] [CrossRef]
- Rafiński, Z. Enantioselective benzoin condensation catalyzed by spirocyclic terpene-based N-heterocyclic carbenes. Tetrahedron 2016, 72, 1860–1867. [Google Scholar] [CrossRef]
- Hachisu, Y.; Bode, J.W.; Suzuki, K. Catalytic intramolecular crossed aldehyde-ketone benzoin reactions: A novel synthesis of functionalized preanthraquinones. J. Am. Chem. Soc. 2003, 125, 8432–8433. [Google Scholar] [CrossRef]
- Enders, D.; Niemeier, O. Thiazol-2-ylidene catalysis in intramolecular crossed aldehyde-ketone benzoin reactions. Synlett 2004, 12, 2111–2114. [Google Scholar] [CrossRef]
- Hachisu, Y.; Bode, J.W.; Suzuki, K. Thiazolium ylide-catalyzed intramolecular aldehyde-ketone benzoin-forming reactions: Substrate scope. Adv. Synth. Catal. 2004, 346, 1097–1100. [Google Scholar] [CrossRef]
- Cookson, R.C.; Lane, R.M. Conversion of dialdehydes into cyclic α-ketols by thiazolium salts: Synthesis of cyclic 2-hydroxy-2-enones. J. Chem. Soc. Chem. Commun. 1976, 804–805. [Google Scholar] [CrossRef]
- Mennen, S.M.; Miller, S.J. Development of a bio-inspired acyl-anion equivalent macrocyclization and synthesis of a trans-resorcylide precursor. J. Org. Chem. 2007, 72, 5260–5269. [Google Scholar] [CrossRef]
- Enders, D.; Niemeier, O.; Balensiefer, T. Asymmetric intramolecular crossed-benzoin reactions by N-heterocyclic carbene catalysis. Angew. Chem.-Int. Ed. 2006, 45, 1463–1467. [Google Scholar] [CrossRef]
- Enders, D.; Niemeier, O.; Raabe, G. Asymmetric synthesis of chromanones via N-heterocyclic carbene catalyzed intramolecular crossed-benzoin reactions. Synlett 2006, 2431–2434. [Google Scholar] [CrossRef]
- Takikawa, H.; Hachisu, Y.; Bode, J.W.; Suzuki, K. Catalytic enantioselective crossed aldehyde-ketone benzoin cyclization. Angew. Chem.-Int. Ed. 2006, 45, 3492–3494. [Google Scholar] [CrossRef]
- Takikawa, H.; Suzuki, K. Modified Chiral Triazolium Salts for Enantioselective Benzoin Cyclization of Enolizable Keto-Aldehydes: Synthesis of (+)-Sappanone B. Org. Lett. 2007, 9, 2713–2716. [Google Scholar] [CrossRef] [PubMed]
- Ema, T.; Oue, Y.; Akihara, K.; Miyazaki, Y.; Sakai, T. Stereoselective synthesis of bicyclic tertiary alcohols with quaternary stereocenters via intramolecular crossed benzoin reactions catalyzed by N-heterocyclic carbenes. Org. Lett. 2009, 11, 4866–4869. [Google Scholar] [CrossRef]
- Ema, T.; Akihara, K.; Obayashi, R.; Sakai, T. Construction of contiguous tetrasubstituted carbon stereocenters by intramolecular crossed benzoin reactions catalyzed by N-heterocyclic carbene (NHC) organocatalyst. Adv. Synth. Catal. 2012, 354, 3283–3290. [Google Scholar] [CrossRef]
- Enders, D.; Henseler, A. A direct intermolecular cross-benzoin type reaction: N-heterocyclic carbene-catalyzed coupling of aromatic aldehydes with trifluoromethyl ketones. Adv. Synth. Catal. 2009, 351, 1749–1752. [Google Scholar] [CrossRef]
- Rose, C.A.; Gundala, S.; Fagan, C.L.; Franz, J.F.; Connon, S.J.; Zeitler, K. NHC-catalysed, chemoselective crossed-acyloin reactions. Chem. Sci. 2012, 3, 735–740. [Google Scholar] [CrossRef]
- Thai, K.; Langdon, S.M.; Bilodeau, F.; Gravel, M. Highly chemo- and enantioselective cross-benzoin reaction of aliphatic aldehydes and α-ketoesters. Org. Lett. 2013, 15, 2214–2217. [Google Scholar] [CrossRef]
- Stetter, H.; Dämbkes, G. Über die präparative Nutzung der Thiazoliumsalz-katalysierten Acyloin- und Benzoin-Bildung; II 1. Herstellung unsymmetrischer Acyloine und α-Diketone. Synthesis 1977, 1977, 403–404. [Google Scholar] [CrossRef]
- Stetter, H.; Dämbkes, G. Über die präparative Nutzung der 1,3-Thiazoliumsalz-katalysierten Acyloin- und Benzoin-Bildung; III 1. Eine neue Methode zur Herstellung von substituierten Enol-trimethylsilyl-ethern des 1,2-Cyclopentandions. Synthesis 1980, 1980, 309–310. [Google Scholar] [CrossRef]
- Piel, I.; Pawelczyk, M.D.; Hirano, K.; Fröhlich, R.; Glorius, F. A family of thiazolium salt derived N-heterocyclic carbenes (NHCs) for organocatalysis: Synthesis, investigation and application in cross-benzoin condensation. Eur. J. Org. Chem. 2011, 5475–5484. [Google Scholar] [CrossRef]
- O’Toole, S.E.; Rose, C.A.; Gundala, S.; Zeitler, K.; Connon, S.J. Highly chemoselective direct crossed aliphatic-aromatic acyloin condensations with triazolium-derived carbene catalysts. J. Org. Chem. 2011, 76, 347–357. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Langdon, S.M.; Legault, C.Y.; Gravel, M. Origin of chemoselectivity in N-heterocyclic carbene catalyzed cross-benzoin reactions: DFT and experimental insights. J. Org. Chem. 2015, 80, 3597–3610. [Google Scholar] [CrossRef] [PubMed]
- Langdon, S.M.; Wilde, M.M.D.; Thai, K.; Gravel, M. Chemoselective N-heterocyclic carbene-catalyzed cross-benzoin reactions: Importance of the fused ring in triazolium salts. J. Am. Chem. Soc. 2014, 136, 7539–7542. [Google Scholar] [CrossRef] [PubMed]
- Jin, M.Y.; Kim, S.M.; Han, H.; Ryu, D.H.; Yang, J.W. Switching regioselectivity in crossed acyloin condensations between aromatic aldehydes and acetaldehyde by altering N-heterocyclic carbene catalysts. Org. Lett. 2011, 13, 880–883. [Google Scholar] [CrossRef]
- Kuhl, N.; Glorius, F. Direct and efficient N-heterocyclic carbene-catalyzed hydroxymethylation of aldehydes. Chem. Commun. 2011, 47, 573–575. [Google Scholar] [CrossRef]
- Haghshenas, P.; Gravel, M. Chemo- and Diastereoselective N-Heterocyclic Carbene-Catalyzed Cross-Benzoin Reactions Using N-Boc-α-amino Aldehydes. Org. Lett. 2016, 18, 4518–4521. [Google Scholar] [CrossRef]
- Haghshenas, P.; Quail, J.W.; Gravel, M. Substrate-Controlled Diastereoselectivity Reversal in NHC-Catalyzed Cross-Benzoin Reactions Using N-Boc-N-Bn-Protected α-Amino Aldehydes. J. Org. Chem. 2016, 81, 12075–12083. [Google Scholar] [CrossRef]
- Castells, J.; López-Calahorra, F.; Bassedas, M.; Urrios, P. A New Thiazolium Salt-Catalyzed Synthesis of α-Aminoketones from Aldehydes and Iminium Salts. Synthesis 1988, 1988, 314–315. [Google Scholar] [CrossRef]
- Murry, J.A.; Frantz, D.E.; Soheili, A.; Tillyer, R.; Grabowski, E.J.J.; Reider, P.J. Synthesis of α-amido ketones via organic catalysis: Thiazolium-catalyzed cross-coupling of aldehydes with acylimines. J. Am. Chem. Soc. 2001, 123, 9696–9697. [Google Scholar] [CrossRef]
- Mattson, A.E.; Scheidt, K.A. Catalytic additions of acylsilanes to imines: An acyl anion strategy for the direct synthesis of α-amino ketones. Org. Lett. 2004, 6, 4363–4366. [Google Scholar] [CrossRef] [PubMed]
- Wilde, M.M.D.; Gravel, M. Bis(amino)cyclopropenylidene (BAC) Catalyzed Aza-Benzoin Reaction. Org. Lett. 2014, 16, 5308–5311. [Google Scholar] [CrossRef] [PubMed]
- Li, G.Q.; Dai, L.X.; You, S.L. Thiazolium-derived N-heterocyclic carbene-catalyzed cross-coupling of aldehydes with unactivated imines. Chem. Commun. 2007, 852–854. [Google Scholar] [CrossRef] [PubMed]
- Dirocco, D.A.; Rovis, T. Catalytic asymmetric acylation of tertiary amines mediated by a dual catalysis mode: N-heterocyclic carbene and photoredox catalysis. J. Am. Chem. Soc. 2012, 134, 8094–8097. [Google Scholar] [CrossRef] [Green Version]
- Dirocco, D.A.; Rovis, T. Catalytic asymmetric cross-aza-benzoin reactions of aliphatic aldehydes with N-Boc-protected imines. Angew. Chem.-Int. Ed. 2012, 51, 5904–5906. [Google Scholar] [CrossRef] [Green Version]
- Enders, D.; Henseler, A.; Lowins, S. N-heterocyclic carbene catalyzed nucleophilic acylation of trifluoromethyl ketimines. Synthesis 2009, 4125–4128. [Google Scholar] [CrossRef]
- Sun, L.H.; Liang, Z.Q.; Jia, W.Q.; Ye, S. Enantioselective N-heterocyclic carbene catalyzed aza-benzoin reaction of enals with activated ketimines. Angew. Chem.-Int. Ed. 2013, 52, 5803–5806. [Google Scholar] [CrossRef]
- Xu, J.; Mou, C.; Zhu, T.; Song, B.A.; Chi, Y.R. N-heterocyclic carbene-catalyzed chemoselective cross-aza-benzoin reaction of enals with isatin-derived ketimines: Access to chiral quaternary aminooxindoles. Org. Lett. 2014, 16, 3272–3275. [Google Scholar] [CrossRef]
- Claisen, L. Über Umlagerung von Phenol-allyläthern in C-Allyl-phenole. Ber. Dtsch. Chem. Ges. 1912, 45, 3157–3166. [Google Scholar] [CrossRef] [Green Version]
- Claisen, L.; Tietze, E. Über den Mechanismus der Umlagerung der Phenol-allyläther. Ber. Dtsch. Chem. Ges. 1925, 58, 275–281. [Google Scholar] [CrossRef]
- Claisen, L.; Tietze, E. Über den Mechanismus der Umlagerung der Phenol-allyläther. (2. Mitteilung). Ber. Dtsch. Chem. Ges. 1926, 59, 2344–2351. [Google Scholar] [CrossRef]
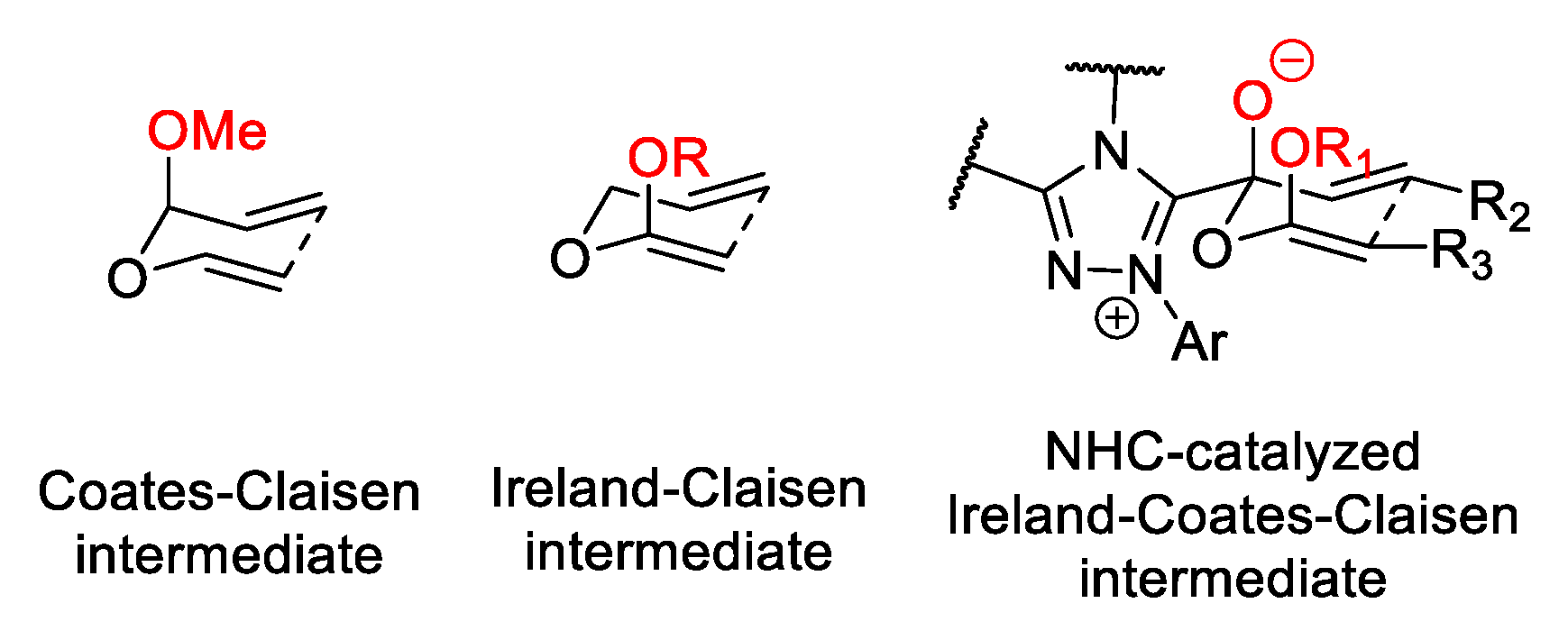
- Coates, R.M.; Rogers, B.D.; Hobbs, S.J.; Peck, D.R.; Curran, D.P. Synthesis and Claisen Rearrangement of Alkoxyallyl Enol Ethers. Evidence for a Dipolar Transition State. J. Am. Chem. Soc. 1987, 109, 1160–1170. [Google Scholar] [CrossRef]
- Ryan, S.J.; Candish, L.; Lupton, D.W. N-heterocyclic carbene-catalyzed generation of α,β-unsaturated acyl imidazoliums: Synthesis of dihydropyranones by their reaction with enolates. J. Am. Chem. Soc. 2009, 131, 14176–14177. [Google Scholar] [CrossRef] [PubMed]
- Candish, L.; Lupton, D.W. The Total Synthesis of (−)-7-Deoxyloganin via N-Heterocyclic Carbene Catalyzed Rearrangement of α,β-Unsaturated Enol Esters. Org. Lett. 2010, 12, 4836–4839. [Google Scholar] [CrossRef]
- Ryan, S.J.; Stasch, A.; Paddon-Row, M.N.; Lupton, D.W. Synthetic and Quantum Mechanical Studies into the N- Heterocyclic Carbene Catalyzed (4 + 2) Cycloaddition. J. Org. Chem. 2012, 77, 1113–1124. [Google Scholar] [CrossRef]
- Zeitler, K. Stereoselective synthesis of (E)-α,β-unsaturated esters via carbene-catalyzed redox esterification. Org. Lett. 2006, 8, 637–640. [Google Scholar] [CrossRef]
- Zhu, Z.Q.; Xiao, J.C. N-heterocyclic carbene-catalyzed reaction of alkynyl aldehydes with 1,3-keto esters or 1,3-diketones. Adv. Synth. Catal. 2010, 352, 2455–2458. [Google Scholar] [CrossRef]
- Kaeobamrung, J.; Mahatthananchai, J.; Zheng, P.; Bode, J.W. An enantioselective Claisen rearrangement catalyzed by N-heterocyclic carbenes. J. Am. Chem. Soc. 2010, 132, 8810–8812. [Google Scholar] [CrossRef] [Green Version]
- Zhao, C.; Guo, D.; Munkerup, K.; Huang, K.-W.; Li, F.; Wang, J. Enantioselective [3+3] atroposelective annulation catalyzed by N-heterocyclic carbenes. Nat. Commun. 2018, 9, 611. [Google Scholar] [CrossRef] [Green Version]
- Yao, C.; Wang, D.; Lu, J.; Li, T.; Jiao, W.; Yu, C. N-Heterocyclic Carbene Catalyzed Reactions of α-Bromo-α,β-unsaturated Aldehydes/α,β-Dibromoaldehydes with 1,3-Dinucleophilic Reagents. Chem.-A Eur. J. 2012, 18, 1914–1917. [Google Scholar] [CrossRef]
- Zhang, B.; Feng, P.; Cui, Y.; Jiao, N. NHC-catalyzed C-O or C-N bond formation: Efficient approaches to α,β-unsaturated esters and amides. Chem. Commun. 2012, 48, 7280–7282. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.B.; Zou, X.L.; Du, G.F.; Liu, Z.Y.; Dai, B. Nucleophilic carbene-catalyzed redox-esterification reaction of α-halo-α,β-unsaturated aldehyde. Tetrahedron 2012, 68, 6498–6503. [Google Scholar] [CrossRef]
- Lang, M.; Wang, J. N-Heterocyclic Carbene-Catalyzed Enantioselective β-Amination of α-Bromoenals Enabled by a Proton-Shuttling Strategy. Eur. J. Org. Chem. 2018, 2018, 2958–2962. [Google Scholar] [CrossRef]
- Ryan, S.J.; Schimler, S.D.; Bland, D.C.; Sanford, M.S. Acyl azolium fluorides for room temperature nucleophilic aromatic fluorination of chloro- and nitroarenes. Org. Lett. 2015, 17, 1866–1869. [Google Scholar] [CrossRef] [PubMed]
- Gillard, R.M.; Fernando, J.E.M.; Lupton, D.W. Enantioselective N-Heterocyclic Carbene Catalysis via the Dienyl Acyl Azolium. Angew. Chem.-Int. Ed. 2018, 57, 4712–4716. [Google Scholar] [CrossRef] [PubMed]
- Knappke, C.E.I.; Imami, A.; Jacobi von Wangelin, A. Oxidative N-Heterocyclic Carbene Catalysis. ChemCatChem 2012, 4, 937–941. [Google Scholar] [CrossRef]
- De Sarkar, S.; Biswas, A.; Samanta, R.C.; Studer, A. Catalysis with N-heterocyclic carbenes under oxidative conditions. Chem.-A Eur. J. 2013, 19, 4664–4678. [Google Scholar] [CrossRef]
- Zhang, C.; Hooper, J.F.; Lupton, D.W. N-Heterocyclic Carbene Catalysis via the α,β-Unsaturated Acyl Azolium. ACS Catal. 2017, 7, 2583–2596. [Google Scholar] [CrossRef]
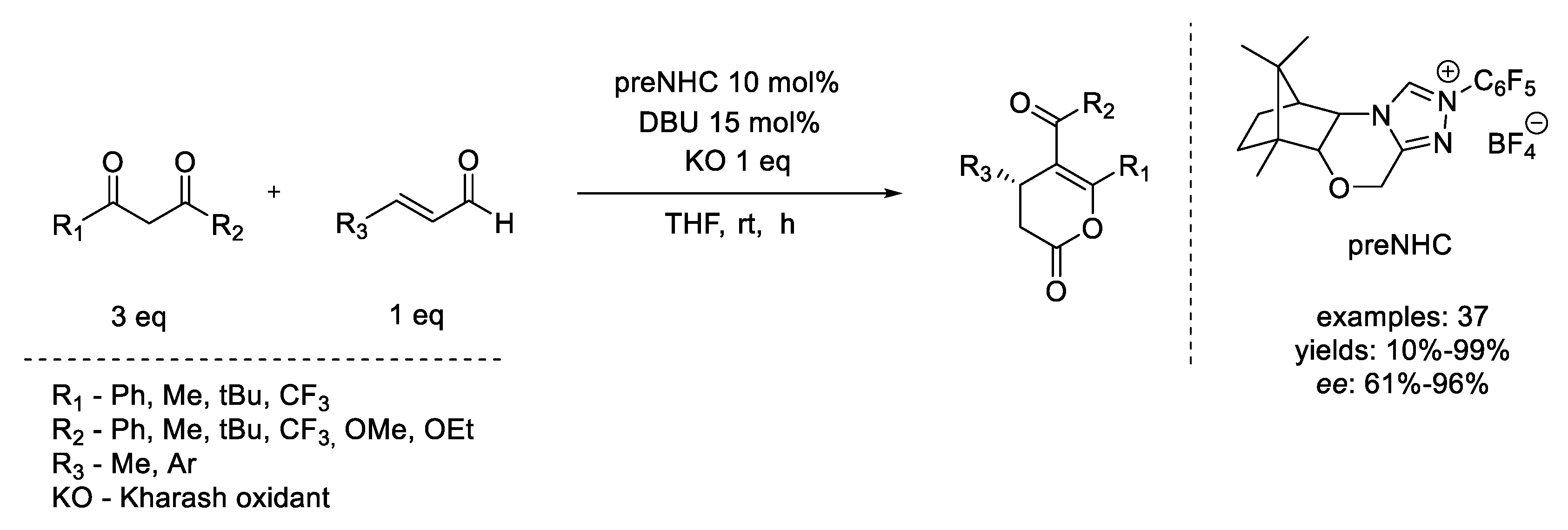
- Dzieszkowski, K.; Rafiński, Z. N-Heterocyclic Carbene Catalysis under Oxidizing Conditions. Catalysts 2018, 8, 549. [Google Scholar] [CrossRef] [Green Version]
- Ta, L.; Axelsson, A.; Sundén, H. Attractive aerobic access to the α,β-unsaturated acyl azolium intermediate: Oxidative NHC catalysis via multistep electron transfer. Green Chem. 2016, 18, 686–690. [Google Scholar] [CrossRef] [Green Version]
- Ta, L.; Axelsson, A.; Sundén, H. N-Acylation of Oxazolidinones via Aerobic Oxidative NHC Catalysis. J. Org. Chem. 2018, 83, 12261–12268. [Google Scholar] [CrossRef] [PubMed]
- Ta, L.; Sundén, H. Oxidative organocatalytic chemoselective: N-acylation of heterocycles with aromatic and conjugated aldehydes. Chem. Commun. 2018, 54, 531–534. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mahatthananchai, J.; Kaeobamrung, J.; Bode, J.W. Chiral N-heterocyclic carbene-catalyzed annulations of enals and ynals with stable enols: A highly enantioselective Coates-Claisen rearrangement. ACS Catal. 2012, 2, 494–503. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, G.T.; Gu, Q.; You, S.L. Enantioselective annulation of enals with 2-naphthols by triazolium salts derived from L-phenylalanine. Chem. Sci. 2015, 6, 4273–4278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wanner, B.; Mahatthananchai, J.; Bode, J.W. Enantioselective synthesis of dihydropyridinones via NHC-Catalyzed Aza-Claisen reaction. Org. Lett. 2011, 13, 5378–5381. [Google Scholar] [CrossRef] [PubMed]
- Dzieszkowski, K.; Barańska, I.; Rafiński, Z. Construction of Dihydropyrido[2,3-d]pyrimidine Scaffolds via Aza-Claisen Rearrangement Catalyzed by N-Heterocyclic Carbenes. J. Org. Chem. 2020, 85, 6645–6662. [Google Scholar] [CrossRef]
- Barik, S.; Shee, S.; Ghosh, A.; Biju, A.T. Catalytic, Enantioselective C2-Functionalization of 3-Aminobenzofurans Using N-Heterocyclic Carbenes. Org. Lett. 2020, 22, 3865–3869. [Google Scholar] [CrossRef]
- Lyngvi, E.; Bode, J.W.; Schoenebeck, F. A computational study of the origin of stereoinduction in NHC-catalyzed annulation reactions of α,β-unsaturated acyl azoliums. Chem. Sci. 2012, 3, 2346–2350. [Google Scholar] [CrossRef]
- Mahatthananchai, J.; Zheng, P.; Bode, J.W. α,β-Unsaturated acyl azoliums from N-heterocyclic carbene catalyzed reactions: Observation and mechanistic investigation. Angew. Chem.-Int. Ed. 2011, 50, 1673–1677. [Google Scholar] [CrossRef] [Green Version]
- Mahatthananchai, J.; Bode, J.W. The effect of the N-mesityl group in NHC-catalyzed reactions. Chem. Sci. 2012, 3, 192–197. [Google Scholar] [CrossRef] [Green Version]
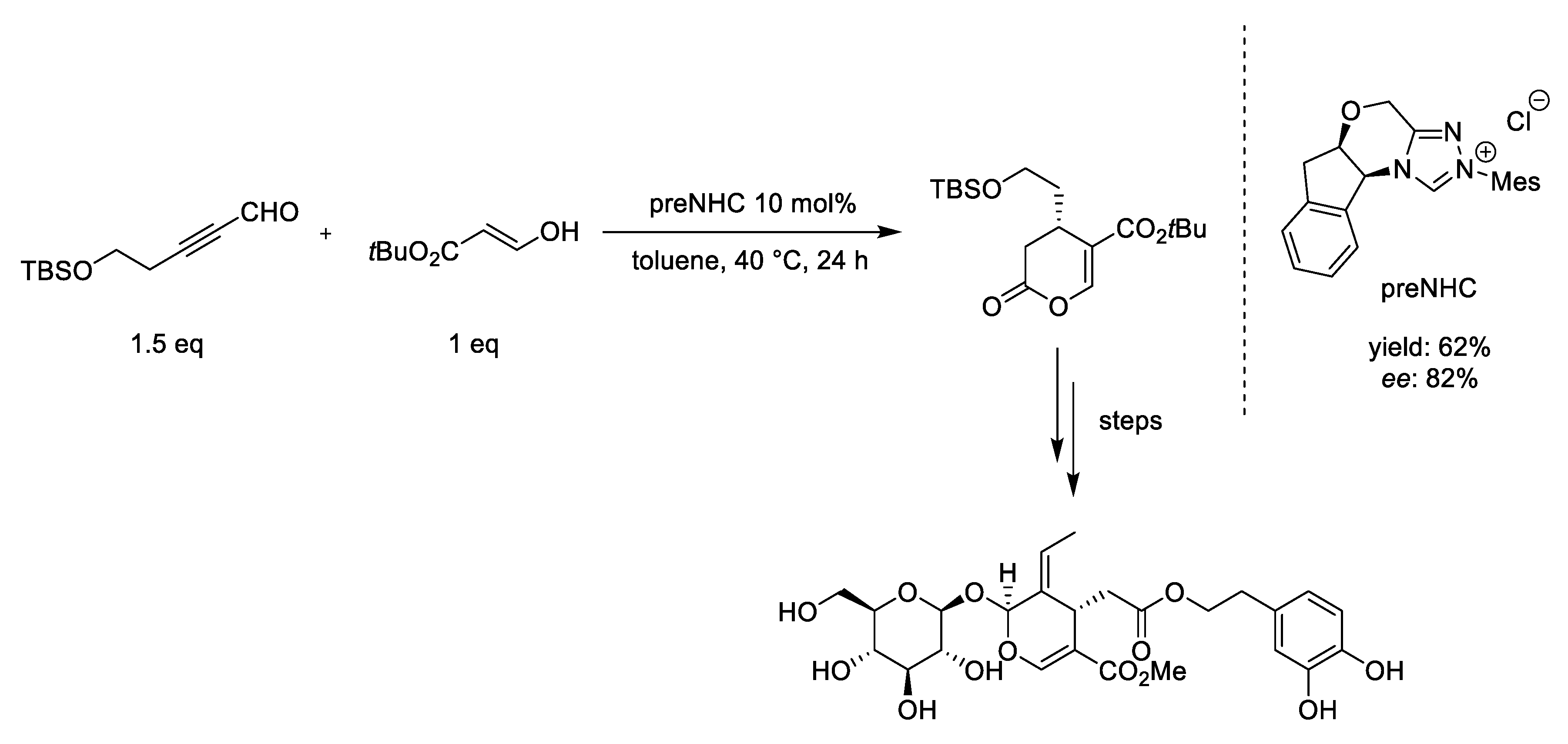
- Vedachalam, S.; Murugesh, N.; Chakraborty, P.; Karvembu, R.; Liu, X.W. NHC catalyzed enantioselective Coates-Claisen rearrangement: A rapid access to the dihydropyran core for oleuropein based secoiridoids. New J. Chem. 2018, 42, 1832–1839. [Google Scholar] [CrossRef]
- Ireland, R.E.; Mueller, R.H. Claisen rearrangement of allyl esters. J. Am. Chem. Soc. 1972, 94, 5897–5898. [Google Scholar] [CrossRef]
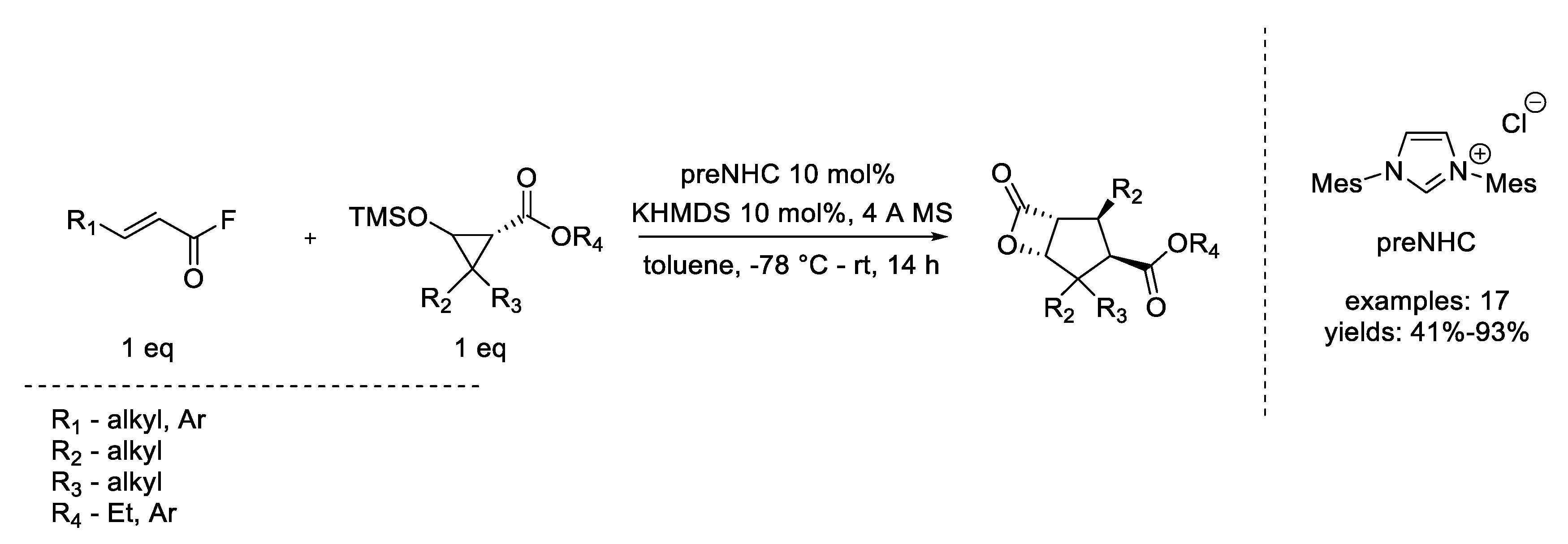
- Candish, L.; Lupton, D.W. N-heterocyclic carbene-catalyzed Ireland-Coates-Claisen rearrangement: Synthesis of functionalized β-lactones. J. Am. Chem. Soc. 2013, 135, 58–61. [Google Scholar] [CrossRef] [PubMed]
- Cope, A.C.; Hardy, E.M. The Introduction of Substituted Vinyl Groups. V. A Rearrangement Involving the Migration of an Allyl Group in a Three-Carbon System1. J. Am. Chem. Soc. 1940, 62, 441–444. [Google Scholar] [CrossRef]
- Berson, J.A.; Jones, M. A Synthesis of Ketones by the Thermal Isomerization of 3-Hydroxy-1,5-hexadienes. The Oxy-Cope Rearrangement. J. Am. Chem. Soc. 1964, 86, 5019–5020. [Google Scholar] [CrossRef]
- Padarti, A.; Han, H. Rationally Designed Chiral Synthons Enabling Asymmetric Z- and E-Selective Vinylogous Aldol Reactions of Aldehydes. Org. Lett. 2018, 20, 1448–1452. [Google Scholar] [CrossRef]
- Reid, J.P.; McAdam, C.A.; Johnston, A.J.S.; Grayson, M.N.; Goodman, J.M.; Cook, M.J. Base-Mediated Cascade Rearrangements of Aryl-Substituted Diallyl Ethers. J. Org. Chem. 2015, 80, 1472–1498. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.; Cao, C.-G.; Sun, H.-B.; Zhang, X.; Niu, D. Catalytic Asymmetric Umpolung Allylation of Imines. J. Am. Chem. Soc. 2016, 138, 13103–13106. [Google Scholar] [CrossRef]
- Ogawa, Y.; Ueno, T.; Karikomi, M.; Seki, K.; Haga, K.; Uyehara, T. Synthesis of 2-acetoxy[5]helicene by sequential double aromatic oxy-Cope rearrangement. Tetrahedron Lett. 2002, 43, 7827–7829. [Google Scholar] [CrossRef]
- Struble, J.R.; Kaeobamrung, J.; Bode, J.W. Synthesis of an N-Mesityl Substituted Chiral Imidazolium Salt for NHC-Catalyzed Reactions. Org. Lett. 2008, 10, 957–960. [Google Scholar] [CrossRef]
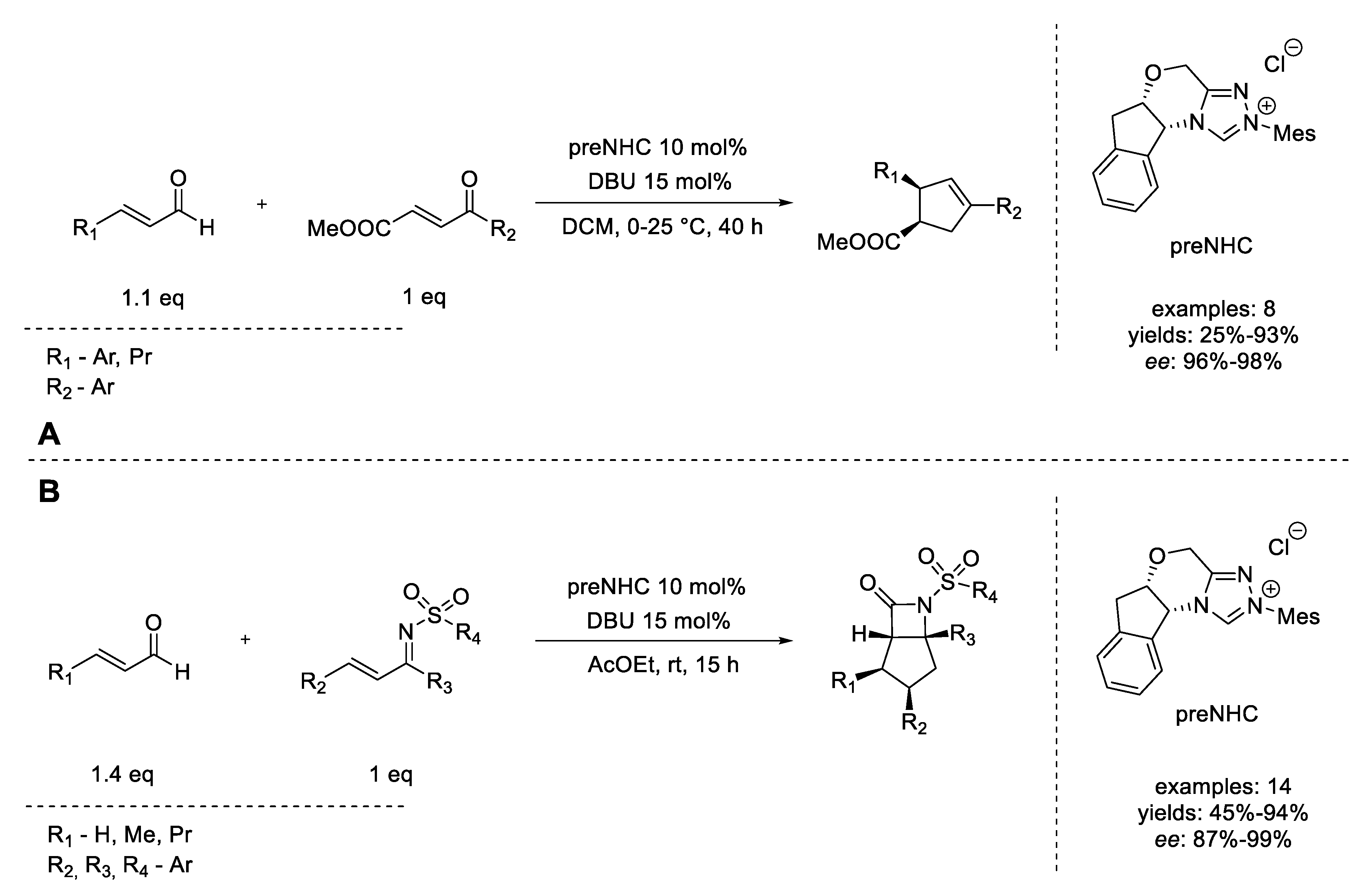
- Chiang, P.-C.; Kaeobamrung, J.; Bode, J.W. Enantioselective, Cyclopentene-Forming Annulations via NHC-Catalyzed Benzoin−Oxy-Cope Reactions. J. Am. Chem. Soc. 2007, 129, 3520–3521. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Bode, J.W. Enantioselective, NHC-Catalyzed Bicyclo-β-Lactam Formation via Direct Annulations of Enals and Unsaturated N-Sulfonyl Ketimines. J. Am. Chem. Soc. 2008, 130, 418–419. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Huang, Y.; Chen, R. N-Heterocyclic carbene-catalyzed (NHC) three-component domino reactions: Highly stereoselective synthesis of functionalized acyclic ε-ketoesters. Org. Biomol. Chem. 2011, 9, 1791–1798. [Google Scholar] [CrossRef] [PubMed]
- Stork, G.; Raucher, S. Chiral synthesis of prostaglandins from carbohydrates. Synthesis of (+)-15-(S)-prostaglandin A2. J. Am. Chem. Soc. 1976, 98, 1583–1584. [Google Scholar] [CrossRef] [PubMed]
- Nicolaou, K.C.; Chakraborty, T.K.; Ogawa, Y.; Daines, R.A.; Simpkins, N.S.; Furst, G.T. Chemistry of amphotericin B. Degradation studies and preparation of amphoteronolide B. J. Am. Chem. Soc. 1988, 110, 4660–4672. [Google Scholar] [CrossRef]
- Crimmins, M.T.; Jung, D.K.; Gray, J.L. Synthetic studies on the ginkgolides: Total synthesis of (±)-bilobalide. J. Am. Chem. Soc. 1993, 115, 3146–3155. [Google Scholar] [CrossRef]
- Corey, E.J.; Su, W.G. Total synthesis of a C15 ginkgolide, (±)-bilobalide. J. Am. Chem. Soc. 1987, 109, 7534–7536. [Google Scholar] [CrossRef]
- Diels, O.; Alder, K. Synthesen in der hydroaromatischen Reihe. XXIV “Dien-Synthesen” stickstoffhaltiger Heteroringe. Justus Liebigs Ann. Chem. 1928, 460, 98–122. [Google Scholar] [CrossRef]
- Kowalczyk, M.; Lupton, D.W. Cascade olefin isomerization/intramolecular Diels-Alder reaction catalyzed by N-heterocyclic carbenes. Angew. Chem.-Int. Ed. 2014, 53, 5314–5317. [Google Scholar] [CrossRef]
- He, M.; Struble, J.R.; Bode, J.W. Highly Enantioselective Azadiene Diels-Alder Reactions Catalyzed by Chiral. J. Am. Chem. Soc. 2006, 128, 8418–8420. [Google Scholar] [CrossRef]
- He, M.; Uc, G.J.; Bode, J.W. Chiral N-heterocyclic carbene catalyzed, enantioselective oxodiene Diels-Alder reactions with low catalyst loadings. J. Am. Chem. Soc. 2006, 128, 15088–15089. [Google Scholar] [CrossRef] [PubMed]
- Jian, T.Y.; Sun, L.H.; Ye, S. Highly enantioselective [4 + 2] cyclization of chloroaldehydes and 1-azadienes catalyzed by N-heterocyclic carbenes. Chem. Commun. 2012, 48, 10907–10909. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Wang, F.; Chua, P.J.; Lv, Y.; Zhong, L.J.; Zhong, G. N-heterocyclic carbene (NHC)-catalyzed highly diastereo- and enantioselective oxo-Diels-Alder reactions for synthesis of fused pyrano[2,3-b]indoles. Org. Lett. 2012, 14, 2894–2897. [Google Scholar] [CrossRef] [PubMed]
- Rong, Z.Q.; Wang, M.; Chow, C.H.E.; Zhao, Y. A Catalyst-Enabled Diastereodivergent Aza-Diels–Alder Reaction: Complementarity of N-Heterocyclic Carbenes and Chiral Amines. Chem.-A Eur. J. 2016, 22, 9483–9487. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.L.; He, L.; Shao, P.L.; Ye, S. [4+2] Cycloaddition of ketenes with N-benzoyldiazenes catalyzed by N-heterocyclic carbenes. Angew. Chem.-Int. Ed. 2009, 48, 192–195. [Google Scholar] [CrossRef] [PubMed]
- Jian, T.Y.; Shao, P.L.; Ye, S. Enantioselective [4+2] cycloaddition of ketenes and 1-azadienes catalyzed by N-heterocyclic carbenes. Chem. Commun. 2011, 47, 2381–2383. [Google Scholar] [CrossRef]
- Kaeobamrung, J.; Kozlowski, M.C.; Bode, J.W. Chiral N-heterocyclic carbene-catalyzed generation of ester enolate equivalents from a,β-unsaturated aldehydes for enantioselective Diels-Alder reactions. Proc. Natl. Acad. Sci. USA 2010, 107, 20661–20665. [Google Scholar] [CrossRef] [Green Version]
- Lv, H.; Mo, J.; Fang, X.; Chi, Y.R. Formal Diels-Alder reactions of chalcones and formylcyclopropanes catalyzed by chiral N-heterocyclic carbenes. Org. Lett. 2011, 13, 5366–5369. [Google Scholar] [CrossRef]
- Hao, L.; Du, Y.; Lv, H.; Chen, X.; Jiang, H.; Shao, Y.; Chi, Y.R. Enantioselective Activation of Stable Carboxylate Esters as Enolate Equivalents via N-Heterocyclic Carbene Catalysts. Org. Lett. 2012, 14, 2154–2157. [Google Scholar] [CrossRef]
- Zhao, X.; Ruhl, K.E.; Rovis, T. N-Heterocyclic-carbene-catalyzed asymmetric oxidative hetero-Diels-Alder reactions with simple aliphatic aldehydes. Angew. Chem.-Int. Ed. 2012, 51, 12330–12333. [Google Scholar] [CrossRef] [Green Version]
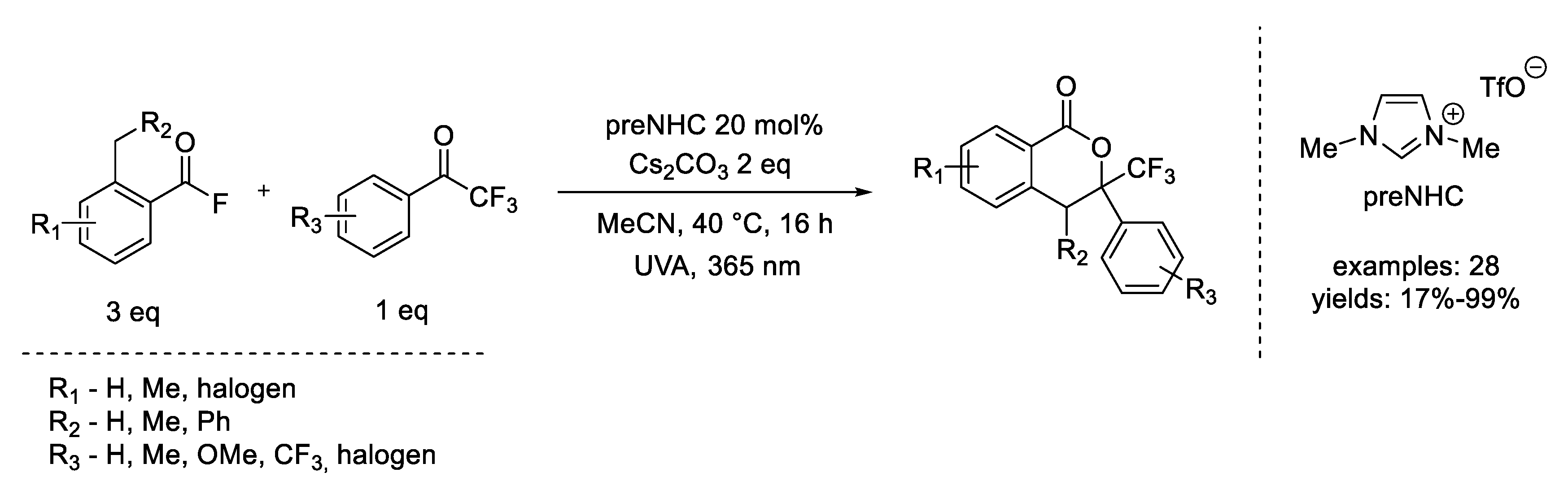
- Mavroskoufis, A.; Rajes, K.; Golz, P.; Agrawal, A.; Ruß, V.; Götze, J.P.; Hopkinson, M.N. N-Heterocyclic Carbene Catalyzed Photoenolization/Diels–Alder Reaction of Acid Fluorides. Angew. Chem.-Int. Ed. 2020, 59, 3190–3194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Michael, A. Ueber die Addition von Natriumacetessig- und Natriummalonsäureäthern zu den Aethern ungesättigter Säuren. J. Prakt. Chem. 1887, 35, 349–356. [Google Scholar] [CrossRef] [Green Version]
- Pfau, M.; Revial, G.; Guingant, A.; d’Angelo, J. Enantioselective synthesis of quaternary carbon centers through Michael-type alkylation of chiral imines. J. Am. Chem. Soc. 1985, 107, 273–274. [Google Scholar] [CrossRef]
- Saito, S.; Hirohara, Y.; Narahara, O.; Moriwake, T. Rotamer distribution control and double Michael addition for cyclopentane annulation with superb selectivity. J. Am. Chem. Soc. 1989, 111, 4533–4535. [Google Scholar] [CrossRef]
- Rubio, A.; Ezquerra, J.; Escribano, A.; Remuiñán, M.J.; Vaquero, J.J. New enantioselective approach to the total synthesis of (−)-α-Kainic Acid. Tetrahedron Lett. 1998, 39, 2171–2174. [Google Scholar] [CrossRef]
- Halland, N.; Aburel, P.S.; Jørgensen, K.A. Highly Enantio- and Diastereoselective Organocatalytic Asymmetric Domino Michael–Aldol Reaction of β-Ketoesters and α,β-Unsaturated Ketones. Angew. Chem. Int. Ed. 2004, 43, 1272–1277. [Google Scholar]
- Adderley, N.J.; Buchanan, D.J.; Dixon, D.J.; Lainé, D.I. Highly Diastereoselective Oxy-Michael Additions of Enantiopure δ-Lactol Anions to Nitroalkenes: Asymmetric Synthesis of 1,2-Amino Alcohols. Angew. Chem. Int. Ed. 2003, 42, 4241–4244. [Google Scholar] [CrossRef]
- Sarkar, D.; Bhattarai, R.; Headley, A.D.; Ni, B. A Novel Recyclable Organocatalytic System for the Highly Asymmetric Michael Addition of Aldehydes to Nitroolefins in Water. Synthesis 2011, 12, 1993–1997. [Google Scholar] [CrossRef]
- D’Angelo, J.; Desmaële, D.; Dumas, F.; Guingant, A. The asymmetric Michael addition reactions using chiral imines. Tetrahedron: Asymmetry 1992, 3, 459–505. [Google Scholar] [CrossRef]
- Arduengo, A.J.; Harlow, R.L.; Kline, M. A stable crystalline carbene. J. Am. Chem. Soc. 1991, 113, 361–363. [Google Scholar] [CrossRef]
- Nair, V.; Sinu, C.R.; Babu, B.P.; Varghese, V.; Jose, A.; Suresh, E. Novel Nucleophilic Heterocyclic Carbene Mediated Stereoselective Conjugate Addition of Enals to Nitrostyrenes via Homoenolate. Org. Lett. 2009, 11, 5570–5573. [Google Scholar] [CrossRef] [PubMed]
- Hans, M.; Delaude, L.; Rodriguez, J.; Coquerel, Y. N-Heterocyclic Carbene Catalyzed Carba-, Sulfa-, and Phospha-Michael Additions with NHC·CO2 Adducts as Precatalysts. J. Org. Chem. 2014, 79, 2758–2764. [Google Scholar] [CrossRef] [PubMed]
- Rong, Z.-Q.; Jia, M.-Q.; You, S.-L. Enantioselective N-Heterocyclic Carbene-Catalyzed Michael Addition to α,β-Unsaturated Aldehydes by Redox Oxidation. Org. Lett. 2011, 13, 4080–4083. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.-Y.; Corey, E.J. Highly Enantioselective Michael Reactions Catalyzed by a Chiral Quaternary Ammonium Salt. Illustration by Asymmetric Syntheses of (S)-Ornithine and Chiral 2-Cyclohexenones. Org. Lett. 2000, 2, 1097–1100. [Google Scholar] [CrossRef]
- Wadamoto, M.; Phillips, E.M.; Reynolds, T.E.; Scheidt, K.A. Enantioselective Synthesis of α,α-Disubstituted Cyclopentenes by an N-Heterocyclic Carbene-Catalyzed Desymmetrization of 1,3-Diketones. J. Am. Chem. Soc. 2007, 129, 10098–10099. [Google Scholar] [CrossRef] [Green Version]
- DeSarkar, S.; Studer, A. NHC-Catalyzed Michael Addition to α,β-Unsaturated Aldehydes by Redox Activation. Angew. Chem. Int. Ed. 2010, 49, 9266–9269. [Google Scholar] [CrossRef]
- Lathrop, S.P.; Rovis, T. Asymmetric Synthesis of Functionalized Cyclopentanones via a Multicatalytic Secondary Amine/N-Heterocyclic Carbene Catalyzed Cascade Sequence. J. Am. Chem. Soc. 2009, 131, 13628–13630. [Google Scholar] [CrossRef] [Green Version]
- Ozboya, K.E.; Rovis, T. Enamine/carbene cascade catalysis in the diastereo- and enantioselective synthesis of functionalized cyclopentanones. Chem. Sci. 2011, 2, 1835–1838. [Google Scholar] [CrossRef] [Green Version]
- Enders, D.; Grossmann, A.; Huang, H.; Raabe, G. Dual Secondary Amine/N-Heterocyclic Carbene Catalysis in the Asymmetric Michael/Cross-Benzoin Cascade Reaction of β-Oxo-Sulfones with Enals. Eur. J. Org. Chem. 2011, 2011, 4298–4301. [Google Scholar] [CrossRef]
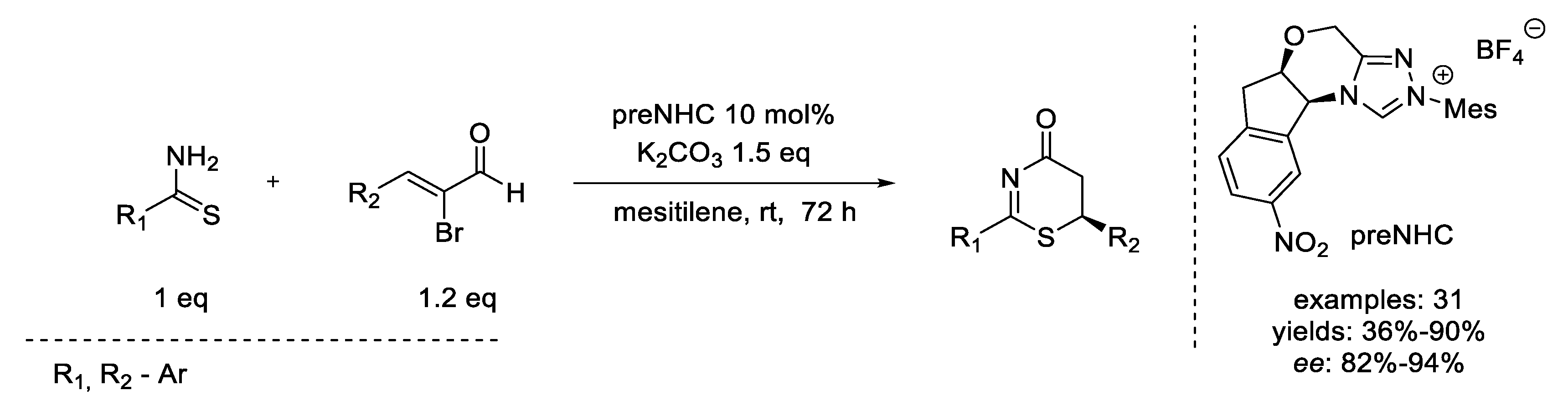
- Ghosh, A.; Barik, S.; Biju, A.T. NHC-Catalyzed [3 + 3] Annulation of Thioamides and Modified Enals for the Enantioselective Synthesis of Functionalized Thiazinones. Org. Lett. 2019, 21, 8598–8602. [Google Scholar] [CrossRef] [Green Version]
- Fang, C.; Cao, J.; Sun, K.; Zhu, J.; Lu, T.; Du, D. Direct and Enantioselective Synthesis of N−H-Free 1,5-Benzodiazepin-2-ones by an N-Heterocyclic Carbene Catalyzed [3+4] Annulation Reaction. Chem.-A Eur. J. 2018, 24, 2103–2108. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, X.-Q.; Zheng, C.; You, S.-L. Highly enantioselective intramolecular Michael reactions by D-camphor-derived triazolium salts. Chem. Commun. 2009, 5823–5825. [Google Scholar] [CrossRef] [PubMed]
- Phillips, E.M.; Wadamoto, M.; Chan, A.; Scheidt, K.A. A Highly Enantioselective Intramolecular Michael Reaction Catalyzed by N-Heterocyclic Carbenes. Angew. Chem. Int. Ed. 2007, 46, 3107–3110. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; Shee, S.; Poisson, T.; Besset, T.; Biju, A.T. Enantioselective N-Heterocyclic Carbene-Catalyzed Cascade Reaction for the Synthesis of Pyrroloquinolines via N–H Functionalization of Indoles. Org. Lett. 2018, 20, 6998–7002. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rapson, W.S.; Robinson, R. 307. Experiments on the synthesis of substances related to the sterols. Part II. A new general method for the synthesis of substituted cyclohexenones. J. Chem. Soc. 1935, 37, 1285. [Google Scholar] [CrossRef]
- Tong, Y.; Mao, J.; Wu, S.; Zhao, Y.; Cheng, Y. Changing the Reaction Pathway by NHC/Brønsted Base Cooperative Catalysis: Highly Stereoselective Synthesis of Multifunctional Benzo[a]fluoren-11-ones from the Dimerization of 2-(Aroylvinyl)arylaldehydes. J. Org. Chem. 2014, 79, 2075–2081. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Larios, E.; Holmes, J.M.; Daschner, C.L.; Gravel, M. NHC-Catalyzed Spiro Bis-Indane Formation via Domino Stetter−Aldol−Michael and Stetter−Aldol−Aldol Reactions. Org. Lett. 2010, 12, 5772–5775. [Google Scholar] [CrossRef]
- Sánchez-Larios, E.; Holmes, J.M.; Daschner, C.L.; Gravel, M. Synthesis of Spiro Bis-Indanes via Domino Stetter-Aldol-Michael and Stetter-Aldol-Aldol Reactions: Scope and Limitations. Synthesis 2011, 2011, 1896–1904. [Google Scholar]
- Ye, W.; Cai, G.; Zhuang, Z.; Jia, X.; Zhai, H. One-Step Assembly of Functionalized γ-Butyrolactones from Benzoins or Benzaldehydes via an N-Heterocyclic Carbene-Mediated Tandem Reaction. Org. Lett. 2005, 7, 3769–3771. [Google Scholar] [CrossRef]
- Phillips, E.M.; Wadamoto, M.; Roth, H.S.; Ott, A.W.; Scheidt, K.A. NHC-Catalyzed Reactions of Aryloxyacetaldehydes: A Domino Elimination/Conjugate Addition/Acylation Process for the Synthesis of Substituted Coumarins. Org. Lett. 2009, 11, 105–108. [Google Scholar] [CrossRef] [Green Version]
- Shu, T.; Ni, Q.; Song, X.; Zhao, K.; Wu, T.; Puttreddy, R.; Rissanen, K.; Enders, D. Asymmetric synthesis of cyclopentanes bearing four contiguous stereocenters via an NHC-catalyzed Michael/Michael/esterification domino reaction. Chem. Commun. 2016, 52, 2609–2611. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shu, T.; Li, S.; Chen, X.-Y.; Liu, Q.; von Essen, C.; Rissanen, K.; Enders, D. Asymmetric synthesis of functionalized tetrahydrofluorenones via an NHC-catalyzed homoenolate Michael addition. Chem. Commun. 2018, 54, 7661–7664. [Google Scholar] [CrossRef] [PubMed]
- Mitsunobu, O.; Obata, T.; Mukaiyama, T. Preparation of Esters of Phosphoric Acid via Quaternary Phosphonium Salts. J. Org. Chem. 1965, 30, 1071–1073. [Google Scholar] [CrossRef]
- Mitsunobu, O.; Yamada, M. Preparation of Esters of Phosphoric Acid via Quaternary Phosphonium Salts. Bull. Chem. Soc. Jpn. 1967, 40, 2380–2382. [Google Scholar] [CrossRef] [Green Version]
- Mitsunobu, O.; Yamada, M.; Mukaiyama, T. Preparation of Esters of Phosphoric Acid by the Reaction of Trivalent Phosphorus Compounds with Diethyl Azodicarboxylate in the Presence of Alcohols. Bull. Chem. Soc. Jpn. 1967, 40, 935–939. [Google Scholar] [CrossRef] [Green Version]
- Kato, T.; Matsuoka, S.I.; Suzuki, M. N-Heterocyclic carbene-mediated redox condensation of alcohols. Chem. Commun. 2016, 52, 8569–8572. [Google Scholar] [CrossRef]
- Kharasch, M.S.; Joshi, B.S. Reactions of Hindered Phenols. II. Base-Catalyzed Oxidations of Hindered Phenols. J. Org. Chem. 1957, 22, 1439–1443. [Google Scholar] [CrossRef]
- Morita, K.; Suzuki, Z.; Hirose, H. A Tertiary Phosphine-catalyzed with Reaction Aldehydes of Acrylic Compounds. Bull. Chem. Soc. Jpn. 1968, 41, 2815. [Google Scholar] [CrossRef] [Green Version]
- Baylis, A.B.; Hillman, M.E.D. German Patent 2155113, 1972.
- He, L.; Jian, T.; Ye, S. N-Heterocyclic Carbene Catalyzed Aza-Morita-Baylis-Hillman Reaction of Cyclic Enones with N-Tosylarylimines. J. Org. Chem. 2007, 72, 7466–7468. [Google Scholar] [CrossRef]
- He, L.; Zhang, Y.R.; Huang, X.L.; Ye, S. Chiral bifunctional N-heterocyclic carbenes: Synthesis and application in the aza-Morita-Baylis-Hillman reaction. Synthesis 2008, 17, 2825–2829. [Google Scholar]
- Chen, X.Y.; Xia, F.; Ye, S. Catalytic MBH reaction of β-substituted nitroalkenes with azodicarboxylates. Org. Biomol. Chem. 2013, 11, 5722–5726. [Google Scholar] [CrossRef] [PubMed]
- Dadwal, M.; Mohan, R.; Panda, D.; Mobin, S.M.; Namboothiri, I.N.N. The Morita-Baylis-Hillman adducts of β-aryl nitroethylenes with other activated alkenes: Synthesis and anticancer activity studies. Chem. Commun. 2006, 338–340. [Google Scholar] [CrossRef]
- Rauhut, M.M.; Currier, H. Preparation of Dialkyl-2-Methylene Glutamates. U.S. Patent 3074999, 22 January 1963. [Google Scholar]
- Atienza, R.L.; Scheidt, K.A. N-heterocyclic carbene-promoted rauhutcurrier reactions between vinyl sulfones and α,β-unsaturated aldehydes. Aust. J. Chem. 2011, 64, 1158–1164. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Zhou, L.; Maiti, R.; Mou, C.; Pan, L.; Chi, Y.R. Sulfinate and Carbene Co-catalyzed Rauhut–Currier Reaction for Enantioselective Access to Azepino[1,2-a]indoles. Angew. Chem.-Int. Ed. 2019, 58, 477–481. [Google Scholar] [CrossRef]
- Goswami, P.; Sharma, S.; Singh, G.; Vijaya Anand, R. Bis(amino)cyclopropenylidene catalyzed Rauhut-Currier reaction between α,β-unsaturated carbonyl compounds and para-quinone methides. J. Org. Chem. 2018, 83, 4213–4220. [Google Scholar] [CrossRef]
- Bae, S.; Zhang, C.; Gillard, R.M.; Lupton, D.W. Enantioselective N-Heterocyclic Carbene Catalyzed Bis(enoate) Rauhut–Currier Reaction. Angew. Chem.-Int. Ed. 2019, 58, 13370–13374. [Google Scholar] [CrossRef]
- Staudinger, H. Mittheilungen aus dem chemischen Institut der Universitit. Liebigs Ann. Chem. 1907, 356, 51–123. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.R.; He, L.; Wu, X.; Shao, P.L.; Ye, S. Chiral N-heterocyclic carbene catalyzed staudinger reaction of ketenes with imines: Highly enantioselective synthesis of N-boc β-lactams. Org. Lett. 2008, 10, 277–280. [Google Scholar] [CrossRef]
- Hans, M.; Wouters, J.; Demonceau, A.; Delaude, L. Mechanistic insight into the Staudinger reaction catalyzed by N-heterocyclic carbenes. Chem.-A Eur. J. 2013, 19, 9668–9676. [Google Scholar] [CrossRef]
- Wang, X.; Shao, P.; Lv, H.; Ye, S. Enantioselective Synthesis of β-Trifluoromethyl-β-lactones via NHC-Catalyzed Ketene−Ketone Cycloaddition Reactions. Org. Lett. 2009, 11, 4029–4031. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Lv, H.; Zhang, Y.R.; Ye, S. Formal cycloaddition of disubstituted ketenes with 2-oxoaldehydes catalyzed by chiral N-heterocyclic carbenes. J. Org. Chem. 2008, 73, 8101–8103. [Google Scholar] [CrossRef] [PubMed]
- Douglas, J.; Taylor, J.E.; Churchill, G.; Slawin, A.M.Z.; Smith, A.D. NHC-promoted asymmetric β-lactone formation from arylalkylketenes and electron-deficient benzaldehydes or pyridinecarboxaldehydes. J. Org. Chem. 2013, 78, 3925–3938. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.N.; Zhang, Y.Y.; Ye, S. Enantioselective synthesis of spirocyclic oxindole-β-lactones via N-heterocyclic carbene-catalyzed cycloaddition of ketenes and isatins. Adv. Synth. Catal. 2010, 352, 1892–1895. [Google Scholar] [CrossRef]
- Huang, X.L.; Chen, X.Y.; Ye, S. Enantioselective synthesis of Aza-β-lactams via NHC-catalyzed [2 + 2] cycloaddition of ketenes with diazenedicarboxylates. J. Org. Chem. 2009, 74, 7585–7587. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Huang, X.L.; Ye, S. Enantioselective formal [2+2] cycloaddition of ketenes with nitroso compounds catalyzed by N-heterocyclic carbenes. Org. Biomol. Chem. 2010, 8, 5007–5011. [Google Scholar] [CrossRef]
- Jian, T.Y.; He, L.; Tang, C.; Ye, S. N-heterocyclic carbene catalysis: Enantioselective formal [2+2] cycloaddition of ketenes and N-sulfinylanilines. Angew. Chem.-Int. Ed. 2011, 50, 9104–9107. [Google Scholar] [CrossRef]
- Feroci, M.; Chiarotto, I.; Orsini, M.; Inesi, A. Electrogenerated NHC as an organocatalyst in the Staudinger reaction. Chem. Commun. 2010, 46, 4121–4123. [Google Scholar] [CrossRef]
- Chiarotto, I.; Mattiello, L.; Pandolfi, F.; Rocco, D.; Feroci, M. NHC in imidazolium acetate ionic liquids: Actual or potential presence? Front. Chem. 2018, 6, 355. [Google Scholar] [CrossRef]
- Feroci, M.; Chiarotto, I.; Orsini, M.; Pelagalli, R.; Inesi, A. Umpolung reactions in an ionic liquid catalyzed by electrogenerated N-heterocyclic carbenes. Synthesis of saturated esters from activated α,β-unsaturated aldehydes. Chem. Commun. 2012, 48, 5361–5363. [Google Scholar] [CrossRef]
- Stetter, H.; Kuhlmann, H. Addition of Aliphatic Aldehydes to Activated Double Bonds. Angew. Chem.-Int. Ed. 1974, 13, 539. [Google Scholar] [CrossRef]
- Enders, D.; Han, J.; Henseler, A. Asymmetric intermolecular Stetter reactions catalyzed by a novel triazolium derived N-heterocyclic carbene. Chem. Commun. 2008, 3989–3991. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Perreault, S.; Rovis, T. Catalytic asymmetric intermolecular Stetter reaction of glyoxamides with alkylidenemalonates. J. Am. Chem. Soc. 2008, 130, 14066–14067. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jousseaume, T.; Wurz, N.E.; Glorius, F. Highly enantioselective synthesis of α-amino acid derivatives by an NHC-catalyzed intermolecular Stetter reaction. Angew. Chem.-Int. Ed. 2011, 50, 1410–1414. [Google Scholar] [CrossRef] [PubMed]
- Wurz, N.E.; Daniliuc, C.G.; Glorius, F. Highly enantioselective intermolecular Stetter reaction of simple acrylates: Synthesis of α-chiral γ-Ketoesters. Chem.-A Eur. J. 2012, 18, 16297–16301. [Google Scholar] [CrossRef]
- Fang, X.; Chen, X.; Lv, H.; Chi, Y.R. Enantioselective Stetter reactions of enals and modified chalcones catalyzed by N-heterocyclic carbenes. Angew. Chem.-Int. Ed. 2011, 50, 11782–11785. [Google Scholar] [CrossRef]
- Patra, A.; Bhunia, A.; Biju, A.T. Facile synthesis of γ-ketophosphonates by an intermolecular Stetter reaction onto vinylphosphonates. Org. Lett. 2014, 16, 4798–4801. [Google Scholar] [CrossRef]
- Dirocco, D.A.; Oberg, K.M.; Dalton, D.M.; Rovis, T. Catalytic Asymmetric Intermolecular Stetter Reaction of Heterocyclic Aldehydes with Nitroalkenes: Backbone Fluorination Improves Selectivity. J. Am. Chem. Soc. 2009, 131, 10872–10874. [Google Scholar] [CrossRef] [Green Version]
- Gacem, B.; Jenner, G. Effect of pressure on stetter reactions: Synthesis of hindered aliphatic acyloins and γ-ketonitriles. High Press. Res. 2004, 24, 233–236. [Google Scholar] [CrossRef]
- Fernando, J.E.M.; Nakano, Y.; Zhang, C.; Lupton, D.W. Enantioselective N-Heterocyclic Carbene Catalysis that Exploits Imine Umpolung. Angew. Chem.-Int. Ed. 2019, 58, 4007–4011. [Google Scholar] [CrossRef]
- Mattson, A.E.; Bharadwaj, A.R.; Scheidt, K.A. The Thiazolium-Catalyzed Sila-Stetter Reaction: Conjugate Addition of Acylsilanes to Unsaturated Esters and Ketones. J. Am. Chem. Soc. 2004, 126, 2314–2315. [Google Scholar] [CrossRef] [PubMed]
- Trost, B.M.; Shuey, C.D.; Dininno, F.; McElvain, S.M. A Stereocontrolled Total Synthesis of (i)-Hirsutic Acid C. J. Am. Chem. Soc. 1979, 101, 1908. [Google Scholar] [CrossRef]
- Paal, C. Ueber die Derivate des Acetophenonacetessigesters und des Acetonylacetessigesters. Ber. Dtsch. Chem. Ges. 1884, 17, 2756–2767. [Google Scholar] [CrossRef] [Green Version]
- Knorr, L. Synthese von Furfuranderivaten aus dem Diacetbernsteinsäureester. Ber. Dtsch. Chem. Ges. 1884, 17, 2863–2870. [Google Scholar] [CrossRef] [Green Version]
- Braun, R.U.; Zeitler, K.; Müller, T.J.J. A novel one-pot pyrrole synthesis via a coupling-isomerization-Stetter-Paal-Knorr sequence. Org. Lett. 2001, 3, 3297–3300. [Google Scholar] [CrossRef]
- Bharadwaj, A.R.; Scheidt, K.A. Catalytic multicomponent synthesis of highly substituted pyrroles utilizing a one-pot sila-Stetter/Paal-Knorr strategy. Org. Lett. 2004, 6, 2465–2468. [Google Scholar] [CrossRef]
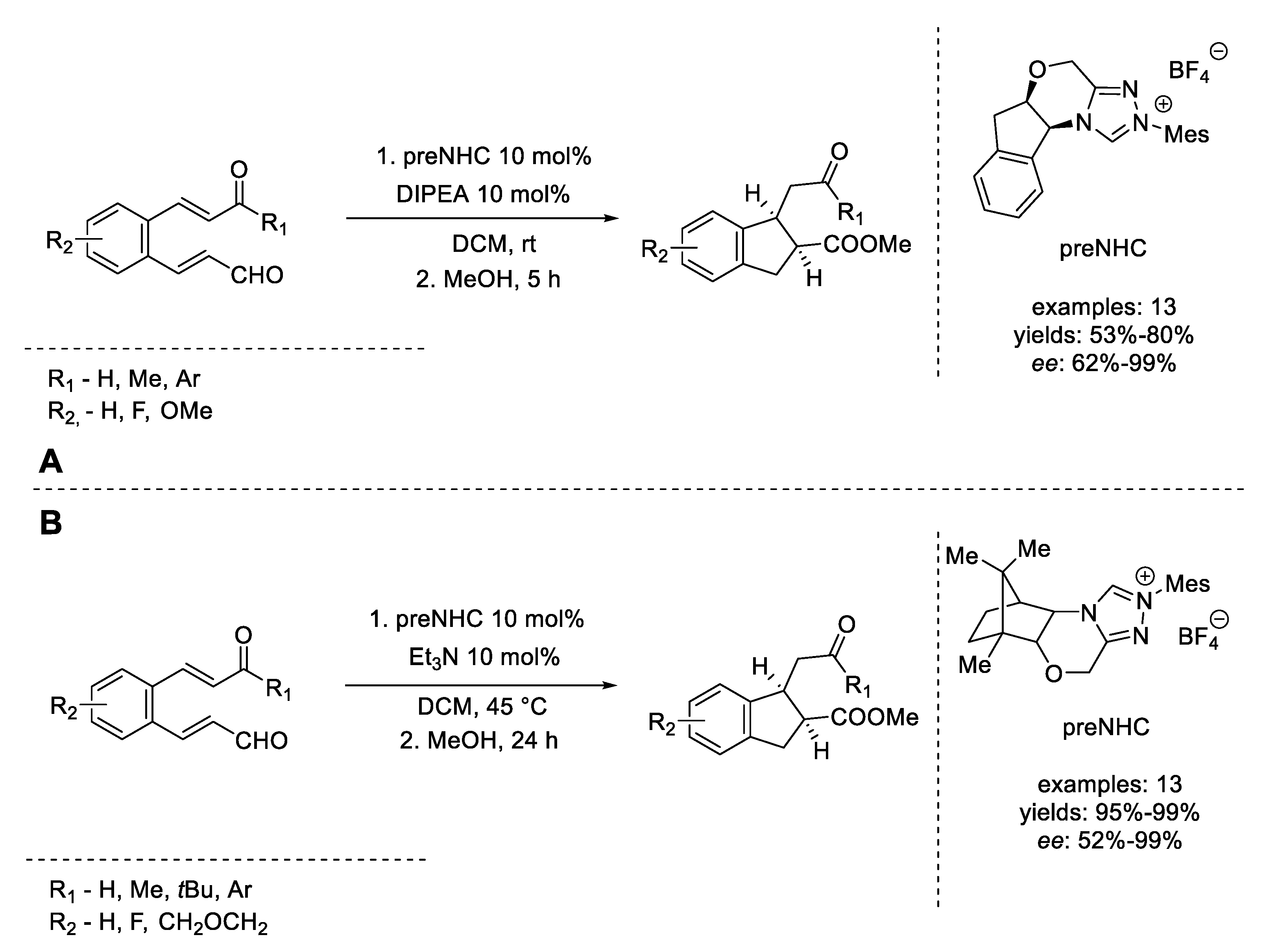
- Sánchez-Larios, E.; Gravel, M. Diastereoselective synthesis of indanes via a domino Stetter-Michael reaction. J. Org. Chem. 2009, 74, 7536–7539. [Google Scholar] [CrossRef]
- Stetter, H.; Kuhlmann, H. Addition von Aldehyden an aktivierte Doppelbindungen; VII1. Eine neue einfache Synthese von cis-Jasmon und Dihydrojasmon. Synthesis 1975, 6, 379–380. [Google Scholar] [CrossRef]
- Galopin, C.C. A short and efficient synthesis of (±)-trans-sabinene hydrate. Tetrahedron Lett. 2001, 42, 5589–5591. [Google Scholar] [CrossRef]
- Randl, S.; Blechert, S. Concise Enantioselective Synthesis of 3,5-Dialkyl-Substituted Indolizidine Alkaloids via Sequential Cross-Metathesis-Double-Reductive Cyclization. J. Org. Chem. 2003, 68, 8879–8882. [Google Scholar] [CrossRef]
- Baumann, K.L.; Butler, D.E.; Deering, C.F.; Mennen, K.E.; Millar, A.; Nanninga, T.N.; Palmer, C.W.; Roth, B.D. The convergent synthesis of CI-981, an optically active, highly potent, tissue selective inhibitor of HMG-CoA reductase. Tetrahedron Lett. 1992, 33, 2283–2284. [Google Scholar] [CrossRef]
- O’Sullivan, S.; O’Neill, J. Preparation of an Atorvastatin Intermediate. U.S. Patent US20090221839A1, 3 September 2009. [Google Scholar]
- Wallach, O. Ueber die Einwirkung von Cyankalium auf Chloral; eine neue Darstellungsweise der Dichloressigsäure. Ber. Dtsch. Chem. Ges. 1873, 6, 114–119. [Google Scholar] [CrossRef]
- Corey, E.J.; Gilman, N.W.; Ganem, B.E. New methods for the oxidation of aldehydes to carboxylic acids and esters. J. Am. Chem. Soc. 1968, 90, 5616–5617. [Google Scholar] [CrossRef]
- Amatore, M.; Beeson, T.D.; Brown, S.P.; MacMillan, D.W.C. Enantioselective Linchpin Catalysis by SOMO Catalysis: An Approach to the Asymmetric α-Chlorination of Aldehydes and Terminal Epoxide Formation. Angew. Chemie Int. Ed. 2009, 48, 5121–5124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burns, N.Z.; Baran, P.S.; Hoffmann, R.W. Redox Economy in Organic Synthesis. Angew. Chemie Int. Ed. 2009, 48, 2854–2867. [Google Scholar] [CrossRef] [PubMed]
- Castells, J.; Llitjos, H.; Moreno-Mañas, M. Nitrobenzene aldehyde oxidations catalyzed by the conjugate bases of thiazolium ions. Tetrahedron Lett. 1977, 18, 205–206. [Google Scholar] [CrossRef]
- Chow, K.Y.-K.; Bode, J.W. Catalytic Generation of Activated Carboxylates: Direct, Stereoselective Synthesis of β-Hydroxyesters from Epoxyaldehydes. J. Am. Chem. Soc. 2004, 126, 8126–8127. [Google Scholar] [CrossRef]
- Reynolds, N.T.; Read de Alaniz, J.; Rovis, T. Conversion of α-Haloaldehydes into Acylating Agents by an Internal Redox Reaction Catalyzed by Nucleophilic Carbenes. J. Am. Chem. Soc. 2004, 126, 9518–9519. [Google Scholar] [CrossRef]
- Reynolds, N.T.; Rovis, T. Enantioselective Protonation of Catalytically Generated Chiral Enolates as an Approach to the Synthesis of α-Chloroesters. J. Am. Chem. Soc. 2005, 127, 16406–16407. [Google Scholar] [CrossRef]
- Chan, A.; Scheidt, K.A. Conversion of α,β-Unsaturated Aldehydes into Saturated Esters: An Umpolung Reaction Catalyzed by Nucleophilic Carbenes. Org. Lett. 2005, 7, 905–908. [Google Scholar] [CrossRef]
- Zhao, G.-L.; Córdova, A. A one-pot combination of amine and heterocyclic carbene catalysis: Direct asymmetric synthesis of β-hydroxy and β-malonate esters from α,β-unsaturated aldehydes. Tetrahedron Lett. 2007, 48, 5976–5980. [Google Scholar] [CrossRef]
- Jiang, H.; Gschwend, B.; Albrecht, Ł.; Anker Jørgensen, K. Organocatalytic Preparation of Simple β-Hydroxy and β-Amino Esters: Low Catalyst Loadings and Gram-Scale Synthesis. Org. Lett. 2010, 12, 5052–5055. [Google Scholar] [CrossRef]
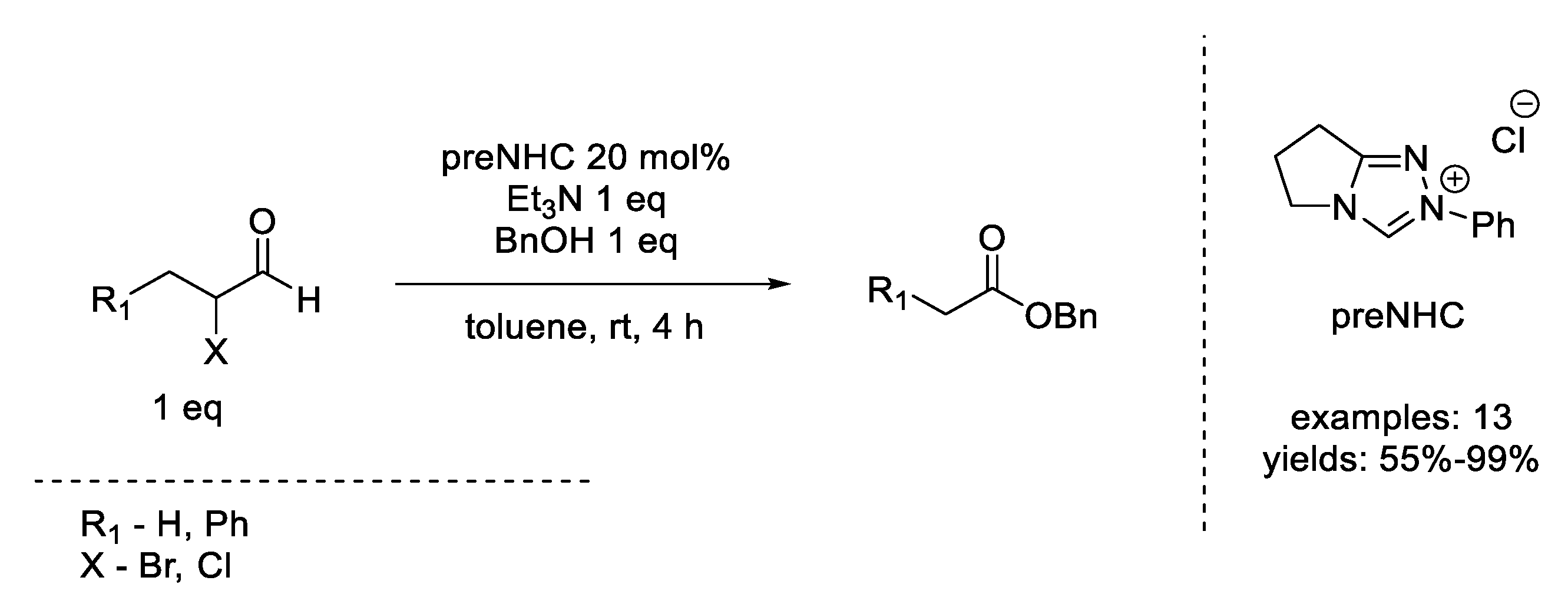
- Sohn, S.S.; Bode, J.W. Catalytic Generation of Activated Carboxylates from Enals: A Product-Determining Role for the Base. Org. Lett. 2005, 7, 3873–3876. [Google Scholar] [CrossRef] [PubMed]
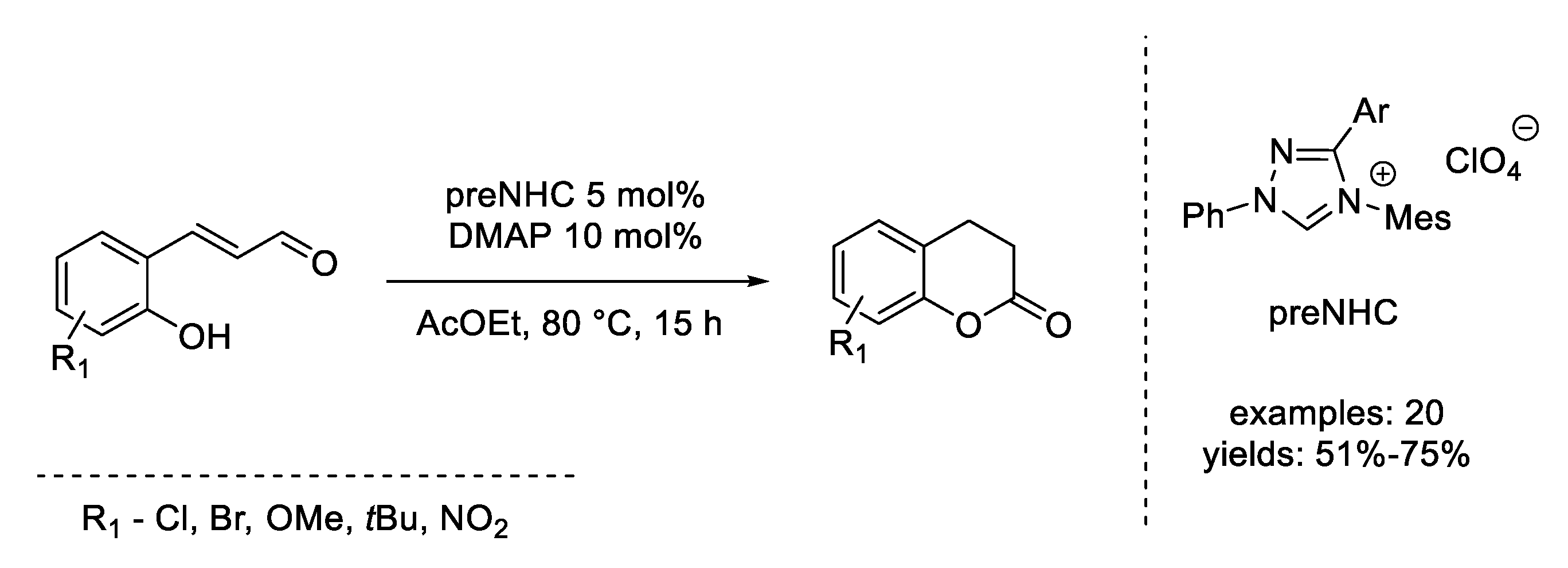
- Zeitler, K.; Rose, C.A. An Efficient Carbene-Catalyzed Access to 3,4-Dihydrocoumarins. J. Org. Chem. 2009, 74, 1759–1762. [Google Scholar] [CrossRef] [PubMed]



© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dzieszkowski, K.; Barańska, I.; Mroczyńska, K.; Słotwiński, M.; Rafiński, Z. Organocatalytic Name Reactions Enabled by NHCs. Materials 2020, 13, 3574. https://doi.org/10.3390/ma13163574
Dzieszkowski K, Barańska I, Mroczyńska K, Słotwiński M, Rafiński Z. Organocatalytic Name Reactions Enabled by NHCs. Materials. 2020; 13(16):3574. https://doi.org/10.3390/ma13163574
Chicago/Turabian StyleDzieszkowski, Krzysztof, Izabela Barańska, Karina Mroczyńska, Michał Słotwiński, and Zbigniew Rafiński. 2020. "Organocatalytic Name Reactions Enabled by NHCs" Materials 13, no. 16: 3574. https://doi.org/10.3390/ma13163574
APA StyleDzieszkowski, K., Barańska, I., Mroczyńska, K., Słotwiński, M., & Rafiński, Z. (2020). Organocatalytic Name Reactions Enabled by NHCs. Materials, 13(16), 3574. https://doi.org/10.3390/ma13163574






