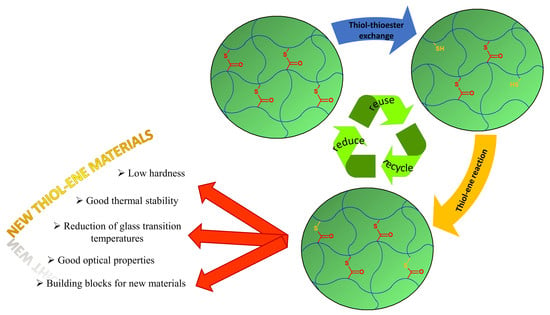
Cross-Linked Polythiomethacrylate Esters Based on Naphthalene—Synthesis, Properties and Reprocessing
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials in Experiments
2.2. Analytical Methods
3. Synthesis of Thiols
3.1. Synthesis of Naphthalene-1,5-Dithiol (1,5-NAF-SH)
3.1.1. Chlorosulfonation of Naphthalene [41]
3.1.2. Reduction of Naphthalene-1,5-Disulfonyl Dichloride
3.2. Synthesis of Naphthalene-1,4(1,5)-Di(Ylmethanethiol) (1,4(1,5)-NAF-CH2SH)
3.2.1. Reaction of Naphthalene with Paraformaldehyde
3.2.2. Reaction of 1,4(1,5)-Bis(Chloromethyl)Naphthalene with Thiourea
4. General Procedure for the Methacrylation of Thiols
5. Polymerization Reactions
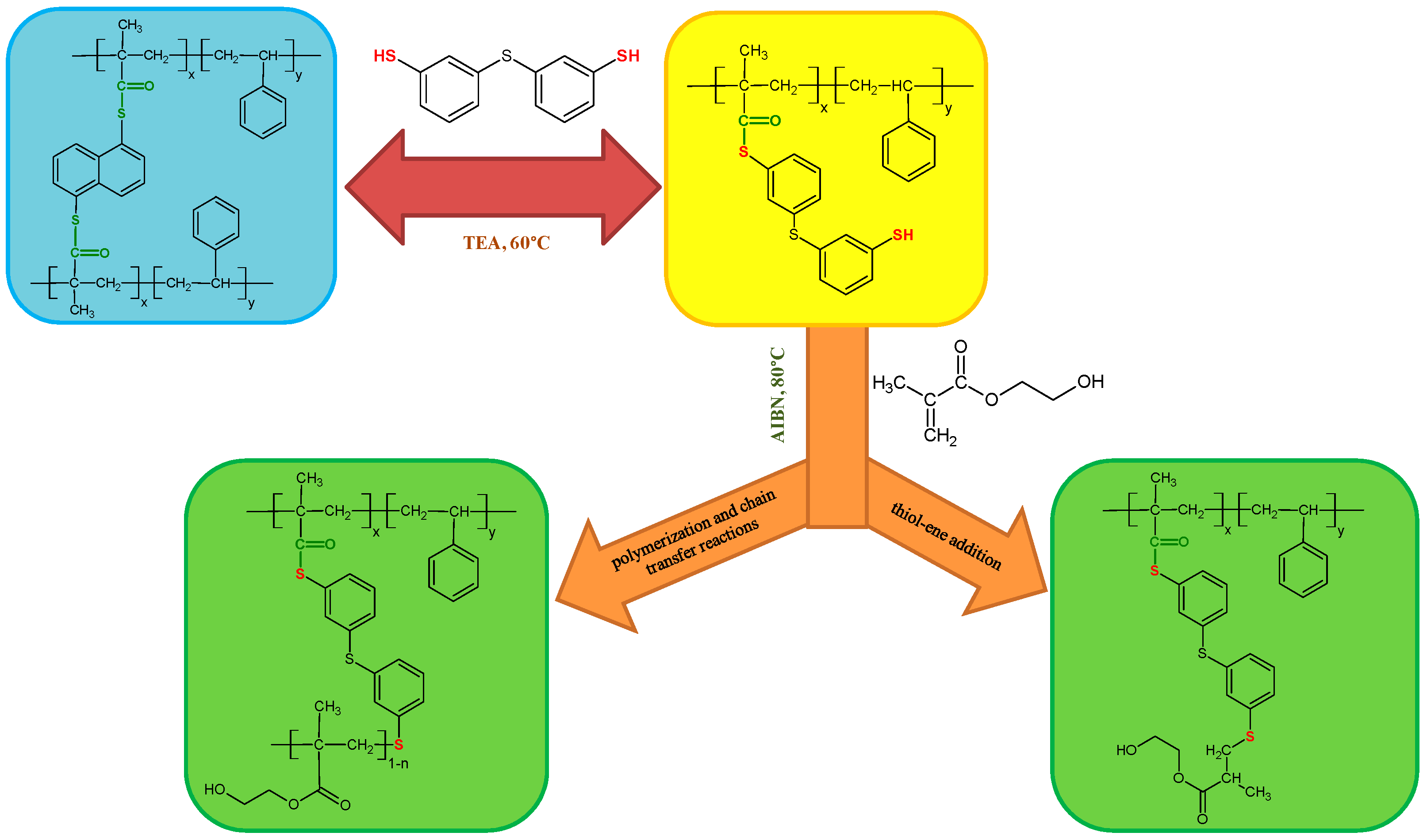
6. The Thiol-Thioester Exchange
7. Thiol-ene Reactions with 2-Hydroxyethyl Methacrylate
8. Results and Discussion
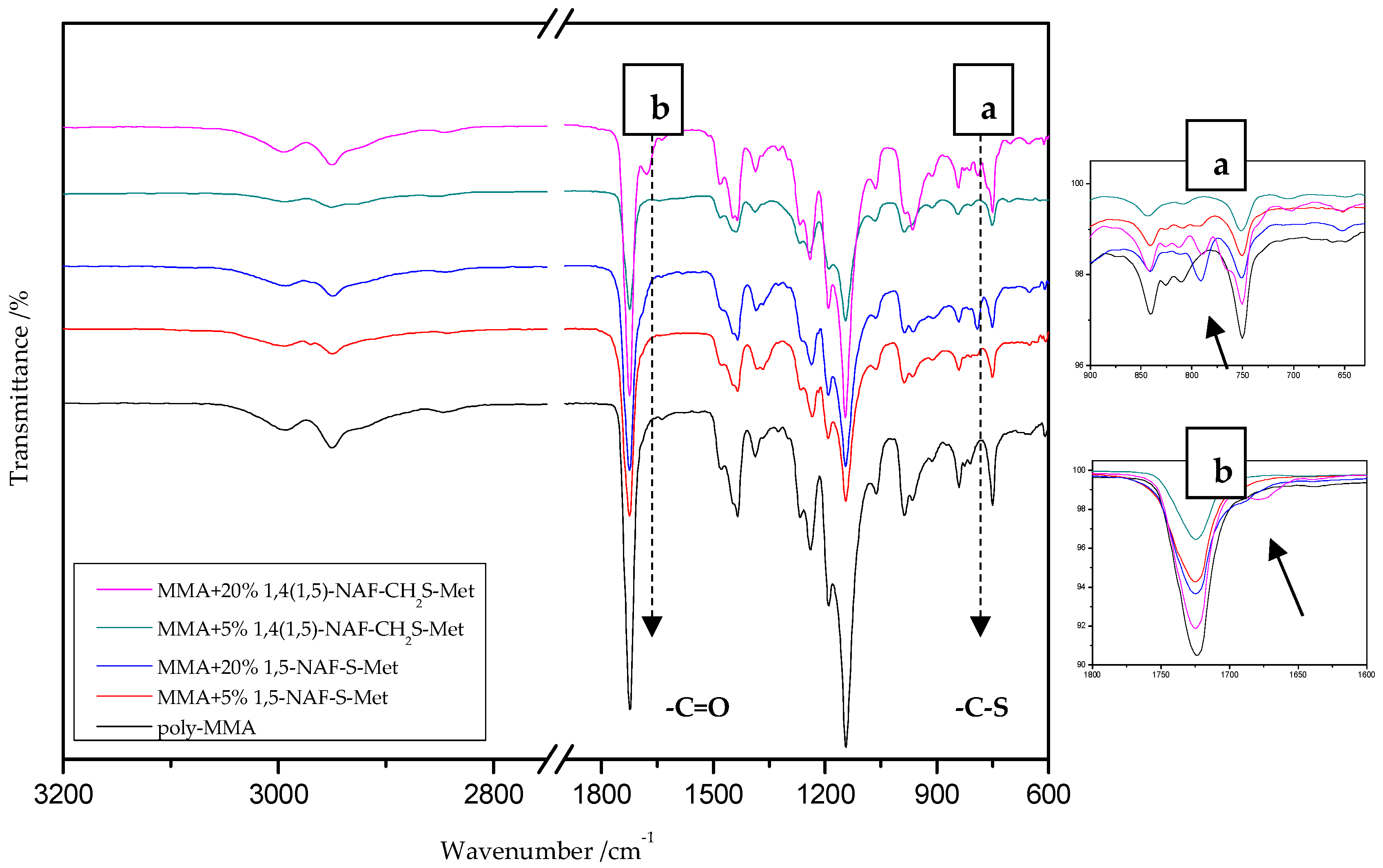
8.1. The ATR/FT-IR Spectra of Thiols and Thioesters
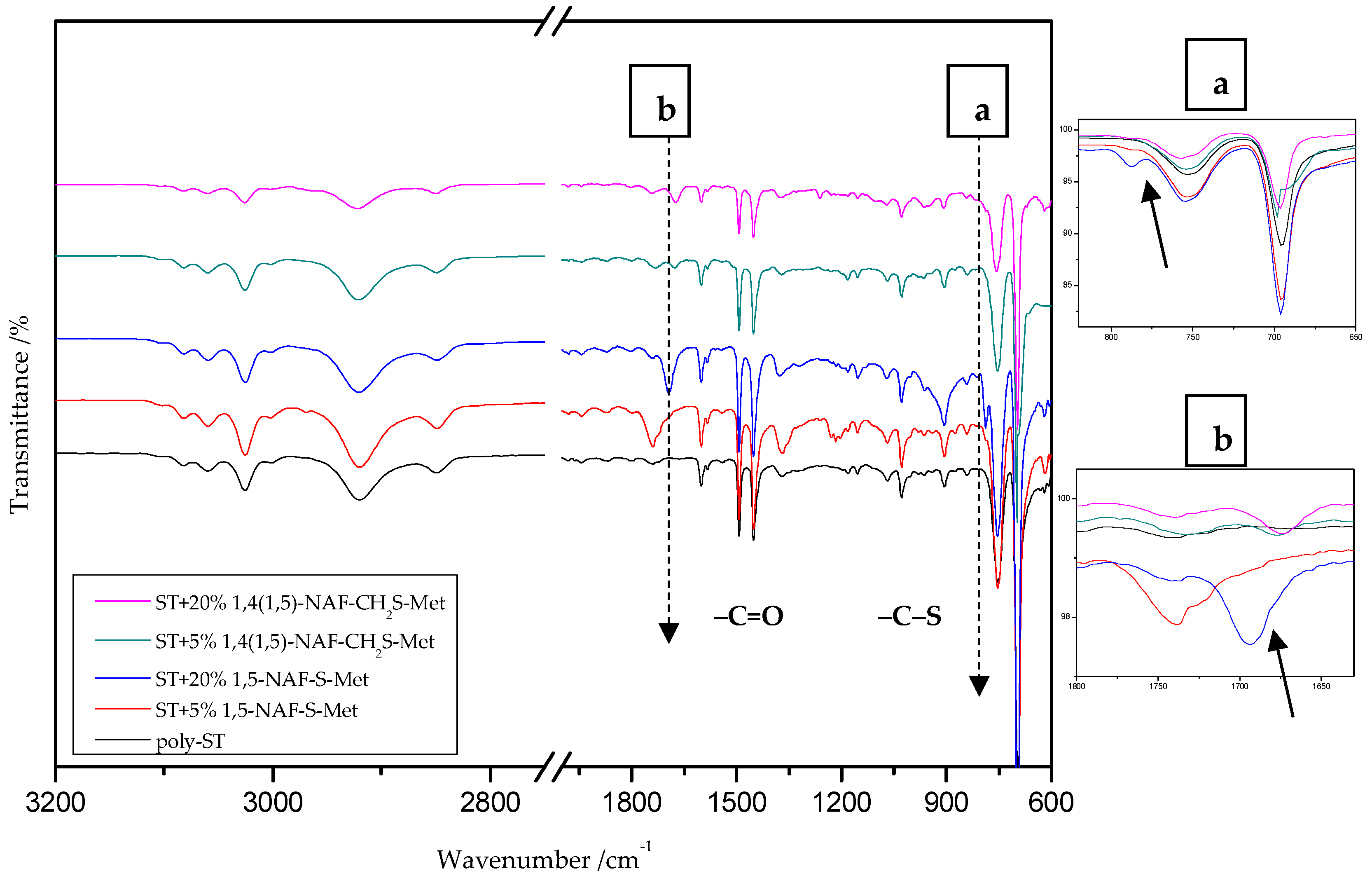
8.2. The ATR/FT-IR Spectra of MMA and ST Copolymers
8.3. Conversion of Unsaturated Bonds
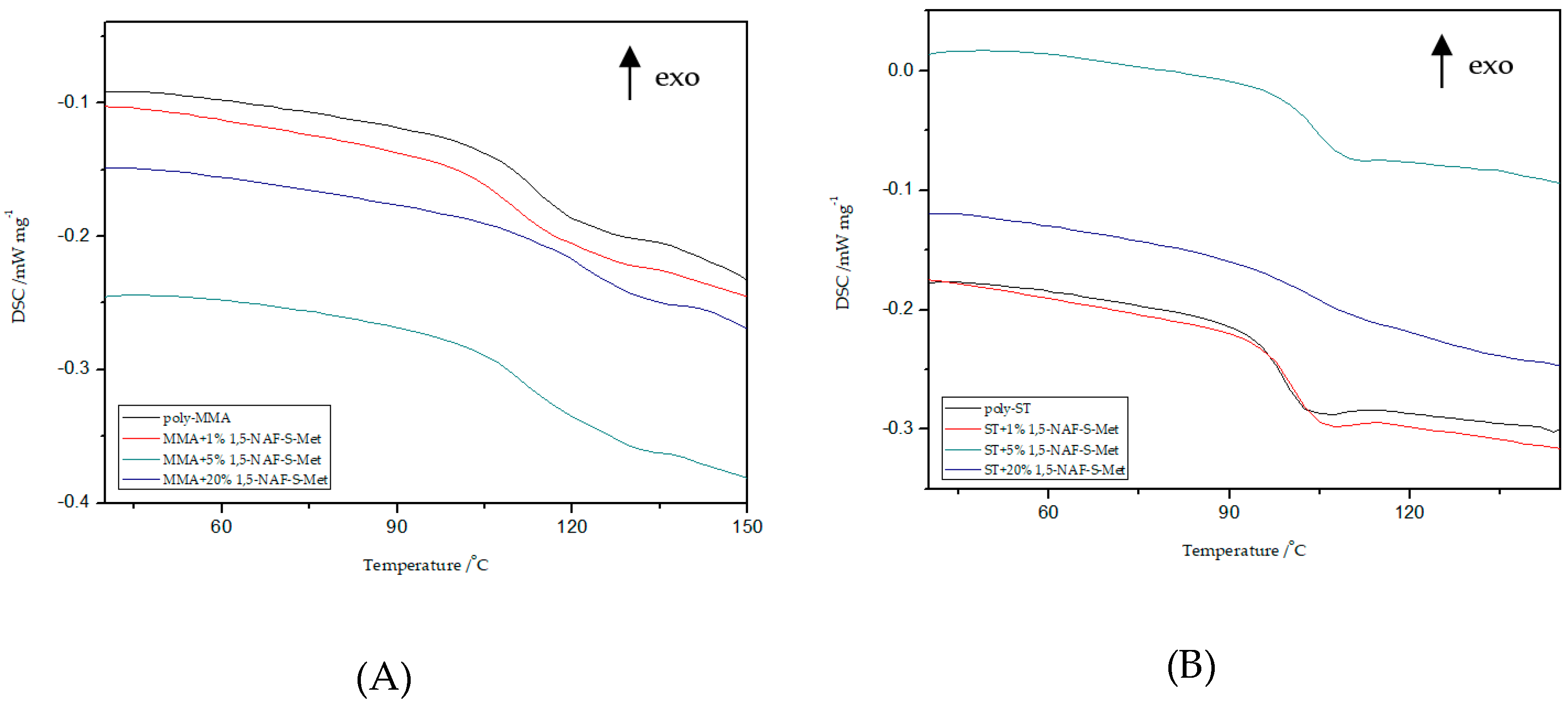
8.4. DSC Analysis
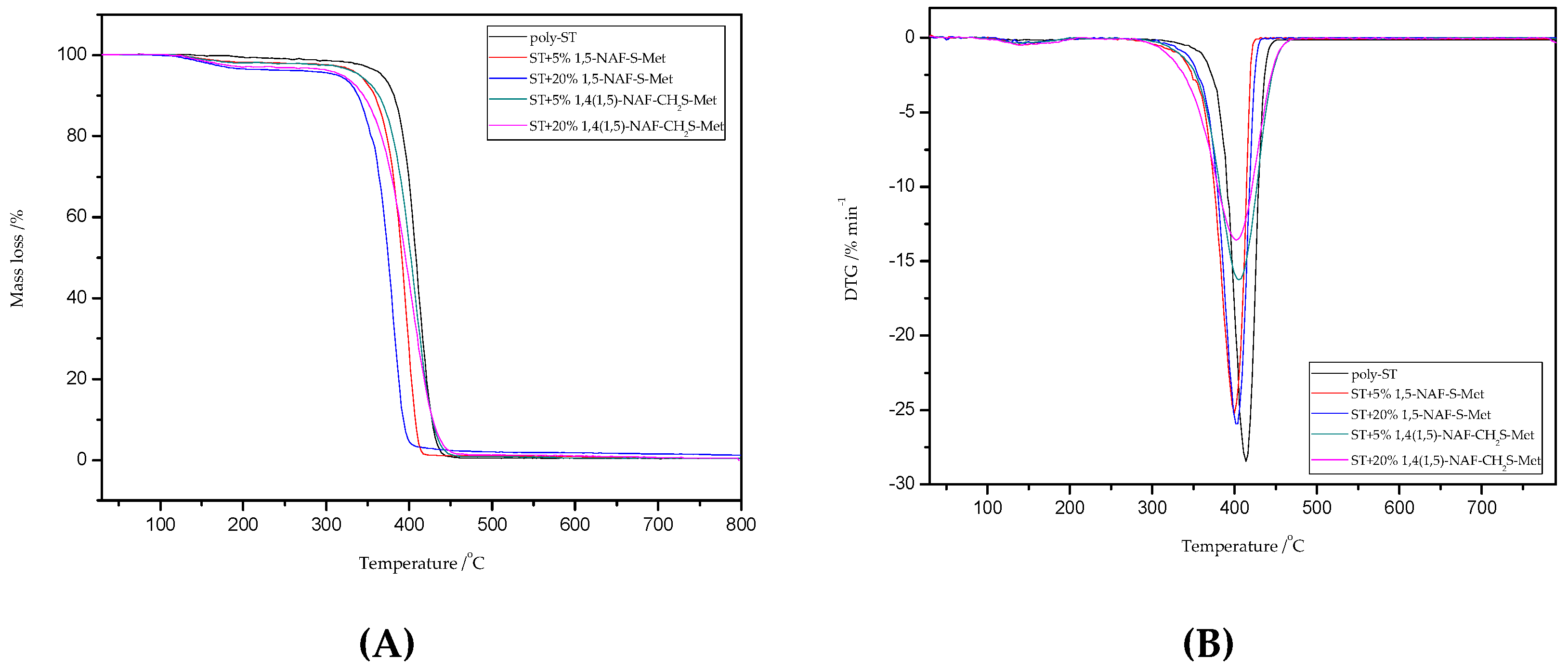
8.5. TG/DTG Analysis
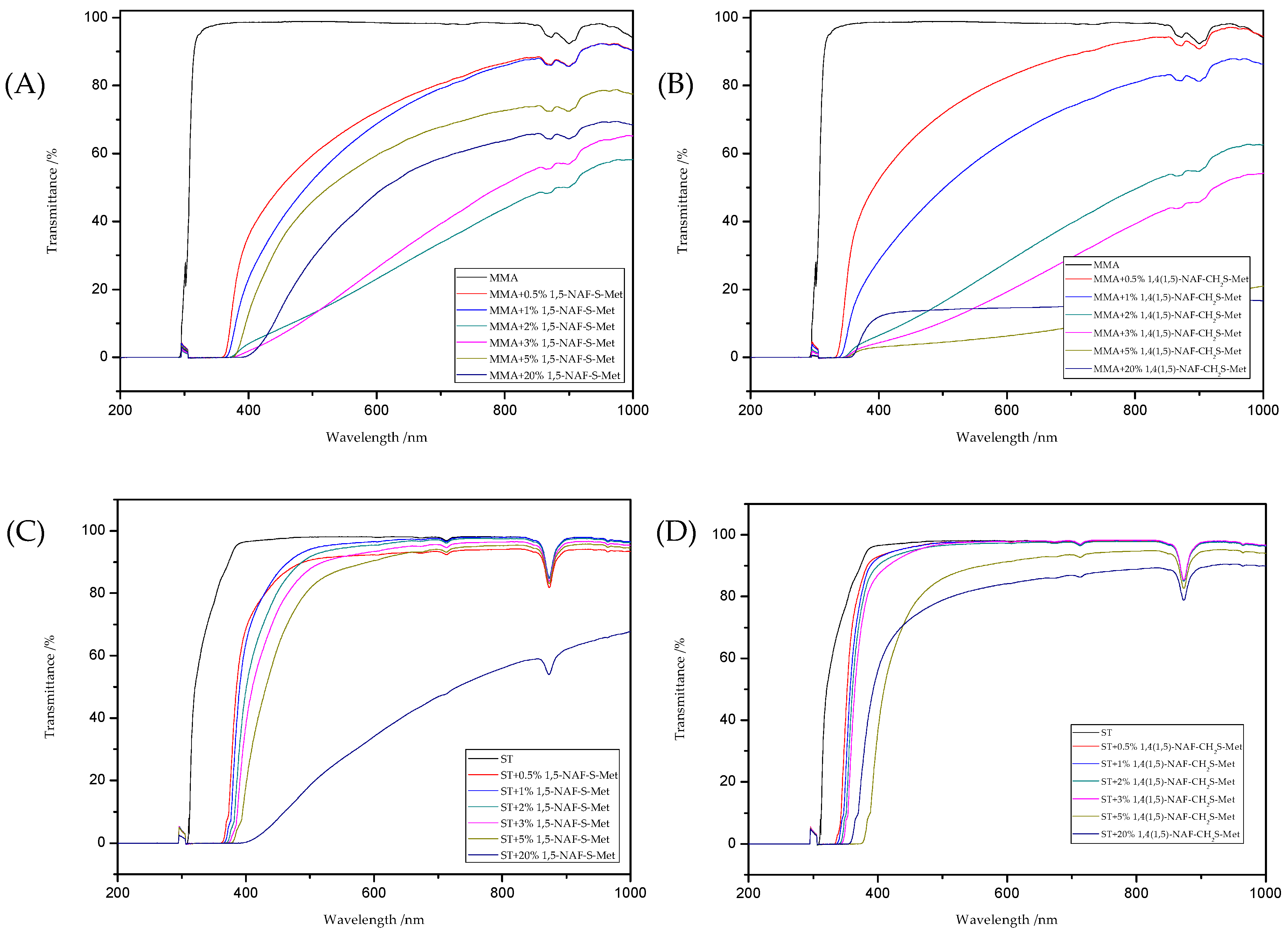
8.6. Optical Properties
8.7. Hardness Tests
8.8. The Swelling Studies of Cross-Linked Polymers
8.9. Thiol-Thioester Exchange and Thiol-Methacrylate Modification Reactions
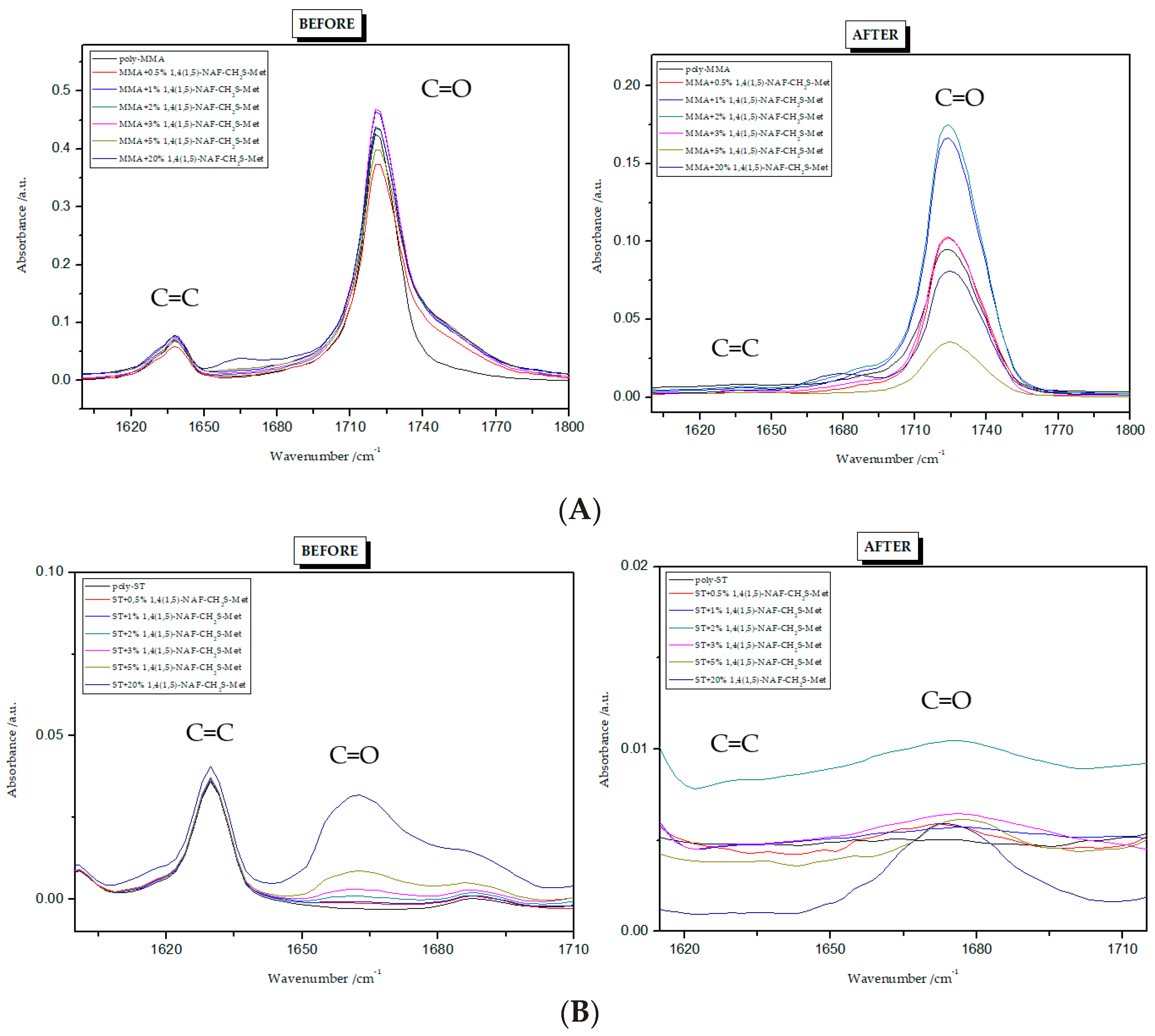
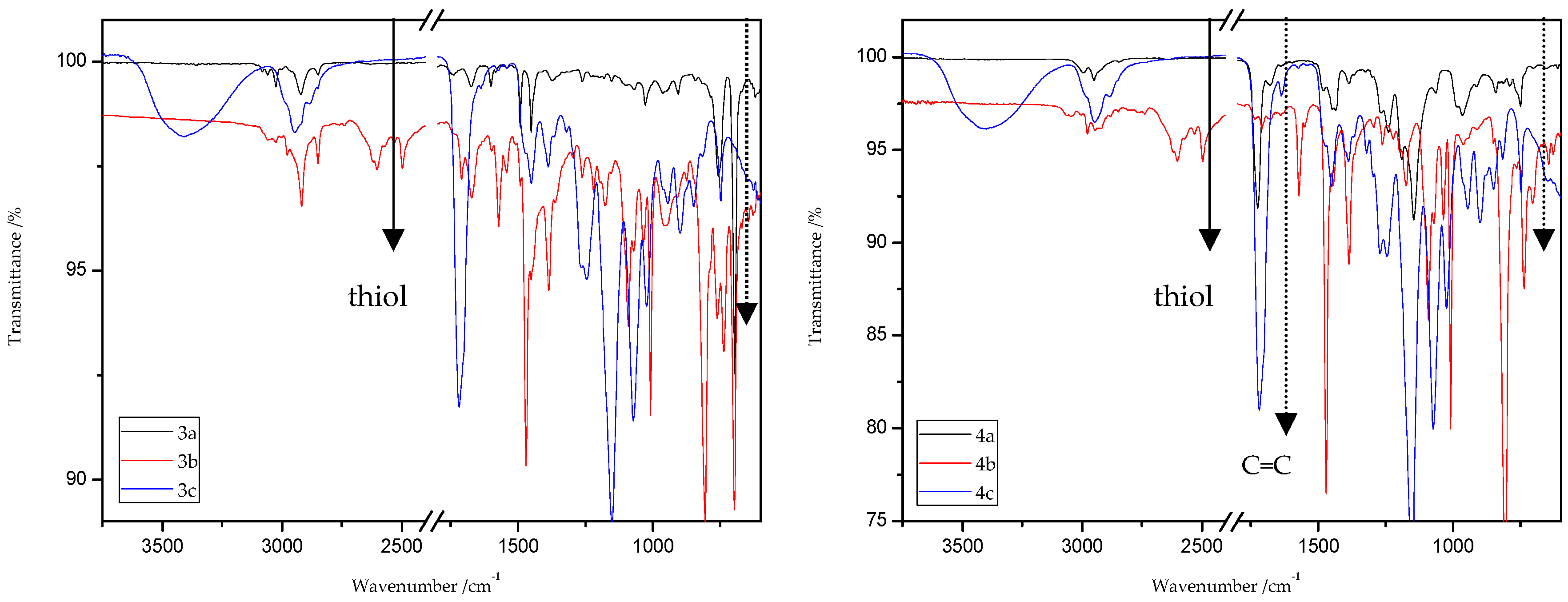
8.9.1. ATR/FT-IR Analysis
8.9.2. Studies of Hardness and Swelling of Materials After Thiol-Ene Reactions
8.9.3. DSC Study
9. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Naga, N.; Tanaka, H.; Moriyama, K. Synthesis of network polymers from multifunctional aromatic thiol compounds. J. Electrochem. Soc. 2019, 166, B3079–B3083. [Google Scholar] [CrossRef]
- Moad, G.; Solomon, D.H. The Chemistry of Free Radical Polymerization, 2nd ed.; Elsevier: Oxford, UK, 2006. [Google Scholar]
- Wu, J.; Li, P.; Ma, X.; Liang, Q.; Yuan, T.; Ma, G. Synthesis and characterization of polyacrylate composite with thiol-modified nanosilica as chain transfer agent. J. Appl. Polym. Sci. 2015, 132, 1–6. [Google Scholar] [CrossRef]
- Karasu, F.; Arsu, N.; Yagci, Y. 2-Mercapto thioxanthone as a chain transfer agent in free-radical polymerization: A versatile route to incorporate thioxanthone moieties into polymer chain-ends. J. Appl. Polym. Sci. 2007, 103, 3766–3770. [Google Scholar] [CrossRef]
- Ito, O.; Matsuda, M. Evaluation of Addition Rates of Thiyl Radicals to Vinyl Monomers by Flash Photolysis. 3. Polar Effect in Addition Reactions of Substituted Benzenethiyl Radicals. J. Am. Chem. Soc. 1981, 103, 5871–5874. [Google Scholar] [CrossRef]
- Cokbaglan, L.; Arsu, N.; Yagci, Y.; Jockusch, S.; Turro, N.J. 2-Mercaptothioxanthone As a Novel Photoinitiator for Free Radical Polymerization. Macromolecules 2003, 36, 2649–2653. [Google Scholar] [CrossRef]
- Henríquez, C.; Bueno, C.; Lissi, E.A.; Encinas, M.V. Thiols as chain transfer agents in free radical polymerization in aqueous solution. Polymer 2003, 44, 5559–5561. [Google Scholar] [CrossRef]
- Valdebenito, A.; Encinas, M.V. Thiophenols as chain transfer agents in the polymerization of vinyl monomers. Polymer 2005, 46, 10658–10662. [Google Scholar] [CrossRef]
- Krongauz, V.V.; Chawla, C.P. Revisiting aromatic thiols effects on radical photopolymerization. Polymer 2003, 44, 3871–3876. [Google Scholar] [CrossRef]
- Kolb, H.C.; Finn, M.G.; Sharpless, K.B. Click Chemistry: Diverse Chemical Function from a Few Good Reactions. Angew. Chem. Int. Ed. Engl. 2001, 40, 2004–2021. [Google Scholar] [CrossRef]
- Lowe, A.B. Thiol-ene “click” reactions and recent applications in polymer and materials synthesis: A first update. Polym. Chem. 2014, 5, 4820–4870. [Google Scholar] [CrossRef]
- Hoyle, C.E.; Bowman, C.N. Thiol-ene click chemistry. Angew. Chem. Int. Ed. 2010, 49, 1540–1573. [Google Scholar] [CrossRef] [PubMed]
- Northrop, B.H.; Coffey, R.N. Thiol−ene click chemistry: Computational and kinetic analysis of the influence of alkene functionality. J. Am. Chem. Soc. 2012, 134, 13804–13817. [Google Scholar] [CrossRef] [PubMed]
- David, R.L.A.; Kornfield, J.A. Facile, efficient routes to diverse protected thiols and to their deprotection and addition to create functional polymers by thiol-ene coupling. Macromolecules 2008, 41, 1151–1161. [Google Scholar] [CrossRef]
- Hoyle, C.E.; Lowe, A.B.; Bowman, C.N. Thiol-click chemistry: A multifaceted toolbox for small molecule and polymer synthesis. Chem. Soc. Rev. 2010, 39, 1355–1387. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Shard, A.; Andhare, N.H.; Richa; Sinha, A.K. Thiol-ene “click” reaction triggered by neutral ionic liquid: The “ambiphilic” character of [hmim]Br in the regioselective nucleophilic hydrothiolation. Angew. Chem. Int. Ed. 2015, 54, 828–832. [Google Scholar] [CrossRef]
- Sinha, A.K.; Equbal, D. Thiol−Ene Reaction: Synthetic Aspects and Mechanistic Studies of an Anti-Markovnikov-Selective Hydrothiolation of Olefins. Asian J. Org. Chem. 2019, 8, 32–47. [Google Scholar] [CrossRef]
- Tucker-Schwartz, A.K.; Farrell, R.A.; Garrell, R.L. Thiol-Ene click reaction as a general route to functional trialkoxysilanes for surface coating applications. J. Am. Chem. Soc. 2011, 133, 11026–11029. [Google Scholar] [CrossRef]
- Goddard-Borger, E.D.; Tropak, M.B.; Yonekawa, S.; Tysoe, C.; Mahuran, D.J.; Withers, S.G. Rapid Assembly of a Library of Lipophilic Iminosugars via the Thiol−Ene Reaction Yields Promising Pharmacological Chaperones for the Treatment of Gaucher Disease. J. Med. Chem. 2012, 55, 2737–2745. [Google Scholar] [CrossRef]
- Kanagasabapathy, S.; Sudalai, A.; Benicewicz, B.C. Montmorillonite K 10-catalyzed regioselective addition of thiols and thiobenzoic acids onto olefins: An efficient synthesis of dithiocarboxylic esters. Tetrahedron Lett. 2001, 42, 3791–3794. [Google Scholar] [CrossRef]
- Goldmann, A.S.; Walther, A.; Nebhani, L.; Joso, R.; Ernst, D.; Loos, K.; Barner-Kowollik, C.; Earner, L.; Müller, A.H.E. Surface modification of poly(divinylbenzene) microspheres via thiol-ene chemistry and alkyne-azide click reactions. Macromolecules 2009, 42, 3707–3714. [Google Scholar] [CrossRef]
- Hoyle, C.E.; Lee, T.Y.; Roper, T. Thiol-enes: Chemistry of the past with promise for the future. J. Polym. Sci. Part A Polym. Chem. 2004, 42, 5301–5338. [Google Scholar] [CrossRef]
- Vandenbergh, J.; Peeters, M.; Kretschmer, T.; Wagner, P.; Junkers, T. Cross-linked degradable poly(β-thioester) networks via amine-catalyzed thiol-ene click polymerization. Polymer 2014, 55, 3525–3532. [Google Scholar] [CrossRef]
- Gress, A.; Völkel, A.; Schlaad, H. Thio-click modification of poly[2 -(3-butenyl)-2-oxazoline]. Macromolecules 2007, 40, 7928–7933. [Google Scholar] [CrossRef]
- Chan, J.W.; Hoyle, C.E.; Lowe, A.B. Sequential phosphine-catalyzed, nucleophilic thiol ene/radical-mediated thiol-yne reactions and the facile orthogonal synthesis of polyfunctional materials. J. Am. Chem. Soc. 2009, 131, 5751–5753. [Google Scholar] [CrossRef]
- Mather, B.D.; Viswanathan, K.; Miller, K.M.; Long, T.E. Michael addition reactions in macromolecular design for emerging technologies. Prog. Polym. Sci. 2006, 31, 487–531. [Google Scholar] [CrossRef]
- Gimbert, C.; Moreno-Mañas, M.; Pérez, E.; Vallribera, A. Tributylphosphine, excellent organocatalyst for conjugate additions of non-nucleophilic N-containing compounds. Tetrahedron 2007, 63, 8305–8310. [Google Scholar] [CrossRef]
- Kloxin, C.J.; Bowman, C.N. Covalent adaptable networks: Smart, reconfigurable and responsive network systems. Chem. Soc. Rev. 2013, 42, 7161–7173. [Google Scholar] [CrossRef]
- Montarnal, D.; Capelot, M.; Tournilhac, F.; Leibler, L. Silica-like malleable materials from permanent organic networks. Science 2011, 334, 965–968. [Google Scholar] [CrossRef]
- Yu, K.; Taynton, P.; Zhang, W.; Dunn, M.L.; Qi, H.J. Reprocessing and recycling of thermosetting polymers based on bond exchange reactions. RSC Adv. 2014, 4, 10108–10117. [Google Scholar] [CrossRef]
- Lu, Y.X.; Tournilhac, F.; Leibler, L.; Guan, Z. Making insoluble polymer networks malleable via olefin metathesis. J. Am. Chem. Soc. 2012, 134, 8424–8427. [Google Scholar] [CrossRef]
- Rekondo, A.; Martin, R.; Ruiz De Luzuriaga, A.; Cabañero, G.; Grande, H.J.; Odriozola, I. Catalyst-free room-temperature self-healing elastomers based on aromatic disulfide metathesis. Mater. Horiz. 2014, 1, 237–240. [Google Scholar] [CrossRef]
- Denissen, W.; Winne, J.M.; Du Prez, F.E. Vitrimers: Permanent organic networks with glass-like fluidity. Chem. Sci. 2016, 7, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Taynton, P.; Yu, K.; Shoemaker, R.K.; Jin, Y.; Qi, H.J.; Zhang, W. Heat- or water-driven malleability in a highly recyclable covalent network polymer. Adv. Mater. 2014, 26, 3938–3942. [Google Scholar] [CrossRef] [PubMed]
- Cromwell, O.R.; Chung, J.; Guan, Z. Malleable and self-healing covalent polymer networks through tunable dynamic boronic ester bonds. J. Am. Chem. Soc. 2015, 137, 6492. [Google Scholar] [CrossRef]
- Worrell, B.T.; McBride, M.K.; Lyon, G.B.; Cox, L.M.; Wang, C.; Mavila, S.; Lim, C.H.; Coley, H.M.; Musgrave, C.B.; Ding, Y.; et al. Bistable and photoswitchable states of matter. Nat. Commun. 2018, 9, 2804. [Google Scholar] [CrossRef]
- McBride, M.K.; Worrell, B.T.; Brown, T.; Cox, L.M.; Sowan, N.; Wang, C.; Podgorski, M.; Martinez, A.M.; Bowman, C.N. Enabling Applications of Covalent Adaptable Networks. Annu. Rev. Chem. Biomol. Eng. 2019, 10, 175–198. [Google Scholar] [CrossRef]
- Wang, C.; Mavila, S.; Worrell, B.T.; Xi, W.; Goldman, T.M.; Bowman, C.N. Productive Exchange of Thiols and Thioesters to Form Dynamic Polythioester-Based Polymers. ACS Macro Lett. 2018, 7, 1312–1316. [Google Scholar] [CrossRef]
- Lekishvili, N.; Nadareishvili, L.; Zaikov, G.; Khananashvili, L. Polymers and Polymeric Materials for Fiber and Gradient Optics; VSP BV: Oud-Beijerland, The Netherlands, 2002. [Google Scholar]
- Goseki, R.; Ishizone, T. Encyclopedia of Polymeric Nanomaterials. Encycl. Polym. Nanomater. 2014, 1–11. [Google Scholar] [CrossRef]
- Ralph, M.; Swisher, R.D. Preparation of 1,5-Naphthalene Disulfonyl Chloride. U.S. Patent 2,827,487, 18 May 1958. [Google Scholar]
- Caesar, P.D. Naphthalene-1,5-disulfonyl chloride. Org. Synth. 1963, 4, 693–694. [Google Scholar] [CrossRef]
- Marvel, C.S.; Caesar, P.D. Polyarylene-Alkylene Sulfides. J. Am. Chem. Soc. 1951, 73, 1097–1099. [Google Scholar] [CrossRef]
- Lock, G.; Schneider, R. Uber die Chlormethylierung des Naphthalins, 11. Mitteil.: 1.4- und 1.5-Di-chlormethyl-naphthalin. Chem. Ber. 1951, 84, 636–641. [Google Scholar] [CrossRef]
- Murahaski, S.; Matsukowa, H. No Title. Bull. Inst. Phys. Chem. Res. 1942, 71, 509. [Google Scholar]
- Bagder, G.M.; Cook, J.W.; Crosbie, G.W. The chloromethylation of naphthalene and of tetralin. J. Chem. Soc. 1947, 1432–1434. [Google Scholar]
- Podkościelny, W.; Charmas, W. Linear Polythioesters. II. Products of interfacial polycondensation of 1,4-di (mercaptomethyl)-naphthalene, 1,5-di (mercaptomethyl)-naphthalene, and a mixture of 1,4- and 1,5-di(mercaptornethyl)-naphthalene with terephthaloyl and isophthaloyl chlorides. J. Polym. Sci. Polym. Chem. Ed. 1979, 17, 2429–2438. [Google Scholar] [CrossRef]
- Fila, K.; Goliszek, M.; Podkościelna, B.; Podgórski, M. Polymer side-chain modification in methacrylate and styrene copolymers through thiol-thioester dynamic exchange. Eur. Polym. J. (Under Review) 2020. [Google Scholar]
- Koo, S.P.S.; Stamenović, M.M.; Arun Prasath, R.; Inglis, A.J.; Prez, F.E.D.U.; Barner-Kowollik, C.; Van Camp, W.I.M.; Junkers, T. Limitations of radical thiol-ene reactions for polymer-polymer conjugation. J. Polym. Sci. Part A Polym. Chem. 2010, 48, 1699–1713. [Google Scholar] [CrossRef]
- Illy, N.; Robitzer, M.; Auvergne, R.; Caillol, S.; David, G.; Boutevin, B. Synthesis of water-soluble allyl-functionalized oligochitosan and its modification by thiol-ene addition in water. J. Polym. Sci. Part A Polym. Chem. 2014, 52, 39–48. [Google Scholar] [CrossRef]
- Way, D.V.; Barbosa, P.I.T.R.; Pinto, J.C. On some practical aspects of click thiol-ene reactions for polymer modification and conjugation with biomolecules. Glob. J. Pure Appl. Chem. Res. 2018, 6, 9–40. [Google Scholar] [CrossRef]
- Campos, L.M.; Killops, K.L.; Sakai, R.; Paulusse, J.M.J.; Damiron, D.; Drockenmuller, E.; Messmore, B.W.; Hawker, C.J. Development of thermal and photochemical strategies for thiol-ene click polymer functionalization. Macromolecules 2008, 41, 7063–7070. [Google Scholar] [CrossRef]
- Lee, T.Y.; Smith, Z.; Reddy, S.K.; Cramer, N.B.; Bowman, C.N. Thiol-allyl ether-methacrylate ternary systems. Polymerization mechanism. Macromolecules 2007, 40, 1466–1472. [Google Scholar] [CrossRef]
- Okamura, H.; Yamagaki, M.; Nakata, K. Analysis of network structures in thiol-ene UV curing system using reworkable resins. Polymers 2018, 11, 5. [Google Scholar] [CrossRef] [PubMed]
- Cramer, N.B.; Couch, C.L.; Schreck, K.M.; Carioscia, J.A.; Boulden, J.E.; Stansbury, J.W.; Bowman, C.N. Investigation of thiol-ene and thiol-ene-methacrylate based resins as dental restorative materials. Dent. Mater. 2010, 26, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Fila, K.; Grochowicz, M.; Podkościelna, B. Thermal and spectral analysis of copolymers with sulphur groups. J. Therm. Anal. Calorim. 2018, 133, 489–497. [Google Scholar] [CrossRef]
- Rieger, J. The glass transition temperature of polystyrene. J. Therm. Anal. Calorim. 1996, 46, 965–972. [Google Scholar] [CrossRef]
- Olson, B.G.; Peng, Z.L.; Srithawatpong, R.; McGervey, J.D.; Ishida, H.; Jamieson, A.M.; Manias, E.; Giannelis, E.P. Free volume in layered organosilicate-polystyrene nanocomposites. Mater. Sci. Forum 1997, 255–257, 336–338. [Google Scholar] [CrossRef]
- Gałka, P.; Kowalonek, J.; Kaczmarek, H. Thermogravimetric analysis of thermal stability of poly(methyl methacrylate) films modified with photoinitiators. J. Therm. Anal. Calorim. 2014, 115, 1387–1394. [Google Scholar] [CrossRef]
- Saladino, M.L.; Motaung, T.E.; Luyt, A.S.; Spinella, A.; Nasillo, G.; Caponetti, E. The effect of silica nanoparticles on the morphology, mechanical properties and thermal degradation kinetics of PMMA. Polym. Degrad. Stab. 2012, 97, 452–459. [Google Scholar] [CrossRef]
- Kashiwagi, T.; Brown, J.E.; Inaba, A.; Hatada, K.; Kitayama, T.; Masuda, E. Effects of Weak Linkages on the Thermal and Oxidative Degradation of Poly(methyl methacrylates). Macromolecules 1986, 19, 2160–2168. [Google Scholar] [CrossRef]
- Mansour, S.A. Study of thermal stabilization for polystyrene/carbon nanocomposites via TG/DSC techniques. J. Therm. Anal. Calorim. 2013, 112, 579–583. [Google Scholar] [CrossRef]
- You, N.H.; Higashihara, T.; Yasuo, S.; Ando, S.; Ueda, M. Synthesis of sulfur-containing poly(thioester)s with high refractive indices and high Abbe numbers. Polym. Chem. 2010, 1, 480–484. [Google Scholar] [CrossRef]
- Song, S.; Zhang, Z.; Liu, X.; Fu, Z.; Xu, J.; Fan, Z. Synthesis and characterization of functional polyethylene with regularly distributed thioester pendants via ring-opening metathesis polymerization. J. Polym. Sci. Part A Polym. Chem. 2017, 55, 4027–4036. [Google Scholar] [CrossRef]
- Aksakal, S.; Remzi Becer, C. Poly(thioacrylate)s: Expanding the monomer toolbox of functional polymers. Polym. Chem. 2016, 7, 7011–7018. [Google Scholar] [CrossRef]
- Rogulska, M.; Kultys, A.; Puszka, A. New thermoplastic poly(carbonate-urethane)s based on chain extenders with sulfur atoms. Chem. Pap. 2017, 71, 1195–1204. [Google Scholar] [CrossRef] [PubMed]
- Jha, G.S.; Seshadri, G.; Mohan, A.; Khandal, R.K. Sulfur containing optical plastics and its ophthalmic lenses applications. E-Polymers 2008, 8, 1–27. [Google Scholar] [CrossRef]
- Matsuda, T.; Funae, Y.; Yoshida, M.; Yamamoto, T.; Takaya, T. Optical material of high refractive index resin composed of sulfur-containing aromatic methacrylates. J. Appl. Polym. Sci. 2000, 76, 50–54. [Google Scholar] [CrossRef]
- Matsuda, T.; Funae, Y.; Yoshida, M.; Yamamoto, T. High index optical materials prepared by copolymerization of novel bifunctional thiomethacrylates. J. Macromol. Sci. Part A Pure Appl. Chem. 2000, 37, 239–257. [Google Scholar] [CrossRef]
- Cullity, B.D.; Stock, S.R. Elements of X-ray Diffraction, 3rd ed.; Prentice Hall: Upper Saddle River, NJ, USA, 2001. [Google Scholar]










| Thioester Methacrylate | Comonomer | Thioester Monomer—Comonomer Feed Ratios (w/w) | Amount of Sulfur Monomer /g | Amount of Comonomer/g | Yield of Polymerization /% | |
|---|---|---|---|---|---|---|
| MMA Polymers | ST Polymers | |||||
| 1,5-NAF-S-Met | MMA (or ST) | - | - | 2 | 97 | 99 |
| 1:200 | 0.01 | 2 | 97 | 98 | ||
| 1:100 | 0.02 | 2 | 96 | 97 | ||
| 1:50 | 0.04 | 2 | 96 | 96 | ||
| 1:33 | 0.06 | 2 | 95 | 97 | ||
| 1:20 | 0.1 | 2 | 97 | 98 | ||
| 1:5 | 0.4 | 2 | 97 | 98 | ||
| 1,4(1,5)-NAF-CH2S-Met | MMA (or ST) | - | - | 2 | 97 | 99 |
| 1:200 | 0.01 | 2 | 97 | 99 | ||
| 1:100 | 0.02 | 2 | 95 | 97 | ||
| 1:50 | 0.04 | 2 | 96 | 96 | ||
| 1:33 | 0.06 | 2 | 97 | 98 | ||
| 1:20 | 0.1 | 2 | 97 | 99 | ||
| 1:5 | 0.4 | 2 | 95 | 97 | ||
| Material | Simplified Designation of Sample |
|---|---|
| ST+1,5-NAF-S-Met | 1a |
| ST+1,5-NAF-S-Met+TBT | 1b |
| ST+1,5-NAF-S-Met+TBT+HEMA | 1c |
| MMA+1,5-NAF-S-Met | 2a |
| MMA+1,5-NAF-S-Met+TBT | 2b |
| MMA+1,5-NAF-S-Met+TBT+HEMA | 2c |
| ST+1,4(1,5)-NAF-CH2S-Met | 3a |
| ST+1,4(1,5)-NAF-CH2S-Met+TBT | 3b |
| ST+1,4(1,5)-NAF-CH2S-Met+TBT+HEMA | 3c |
| MMA+1,4(1,5)-NAF-CH2S-Met | 4a |
| MMA+1,4(1,5)-NAF-CH2S-Met+TBT | 4b |
| MMA+1,4(1,5)-NAF-CH2S-Met+TBT+HEMA | 4c |
| % w/w 1,5-NAF-S-Met (or 1,4(1,5)-NAF-CH2S-Met) | Degree of Conversion (DC)/% | |||
|---|---|---|---|---|
| ST+1,5-NAF-S-Met | MMA+1,5-NAF-S-Met | ST+1,4(1,5)-NAF-CH2S-Met | MMA+1,4(1,5)-NAF-CH2S-Met | |
| 0 | 100 (-) | 95 (3) | 100 (-) | 95 (3) |
| 0.5 | 100 (-) | 97 (2) | 100 (-) | 96 (2) |
| 1 | 100 (2) | 95 (3) | 99 (1) | 95 (2) |
| 2 | 99 (2) | 96 (2) | 99 (1) | 95 (3) |
| 3 | 99 (1) | 96 (2) | 99 (2) | 94 (3) |
| 5 | 99 (2) | 98 (2) | 99 (2) | 93 (2) |
| 20 | 98 (2) | 97 (3) | 98 (2) | 93 (2) |
| Copolymer | Temperature/ °C | R/% | |||||
|---|---|---|---|---|---|---|---|
| a T2 | b T5 | c T10 | d T50 | e Tf | f Tmax | ||
| poly-MMA | 155 | 240 | 272 | 355 | 435 | 180; 250; 367 | 0.40 |
| MMA + 5% 1,5-NAF-S-Met | 162 | 270 | 298 | 345 | 436 | 157; 337; 381 | 0.69 |
| MMA + 20% 1,5-NAF-S-Met | 161 | 236 | 275 | 335 | 450 | 171; 328; 391 | 1.17 |
| MMA + 5% 1,4(1,5)-NAF-CH2S-Met | 148 | 228 | 285 | 345 | 445 | 149; 346 | 0.90 |
| MMA + 20% 1,4(1,5)-NAF-CH2S-Met | 131 | 181 | 290 | 347 | 460 | 142; 337; 367 | 1.52 |
| poly-ST | 329 | 368 | 384 | 409 | 460 | 414 | 0.26 |
| ST + 5% 1,5-NAF-S-Met | 262 | 341 | 359 | 391 | 440 | 144; 397 | 0.60 |
| ST + 20% 1,5-NAF-S-Met | 158 | 314 | 338 | 375 | 434 | 139; 387 | 1.26 |
| ST + 5% 1,4(1,5)-NAF-CH2S-Met | 253 | 341 | 364 | 402 | 470 | 141; 405 | 0.22 |
| ST + 20% 1,4(1,5)-NAF-CH2S-Met | 168 | 321 | 345 | 396 | 470 | 139; 402 | 0.36 |
| % Thioester | Refractive Index | |||
|---|---|---|---|---|
| ST+1,5-NAF-S-Met | MMA+1,5-NAF-S-Met | ST+1,4(1,5)-NAF-CH2S-Met | MMA+1,4(1,5)-NAF-CH2S-Met | |
| 0 | 1.5440 | 1.4142 | 1.5440 | 1.4142 |
| 0.5 | 1.5448 | 1.4143 | 1.5442 | 1.4142 |
| 1 | 1.5452 | 1.4146 | 1.5445 | 1.4152 |
| 2 | 1.5460 | 1.4153 | 1.5452 | 1.4166 |
| 3 | 1.5470 | 1.4190 | 1.5458 | 1.4196 |
| 5 | 1.5485 | 1.4221 | 1.5470 | 1.4238 |
| 20 | 1.5580 | 1.4473 | 1.5540 | 1.4402 |
| Solvent | Swelling Coefficients, B /% | |||
|---|---|---|---|---|
| After 24 h in Solvent | After 15 Min in Air | After 2 h in Air | After 24 h in Air | |
| ST+20% 1,5-NAF-S-Met | ||||
| THF | 86.1 | 38.8 | 23.5 | 11.0 |
| MeOH | 0.9 | 0.2 | 0.1 | 0.05 |
| ACN | 5.3 | 4.2 | 3.0 | 1.7 |
| Acetone | 29.0 | 22.8 | 17.3 | 8.9 |
| DCM | 83.0 | 34.1 | 11.0 | 1.2 |
| MMA+20% 1,5-NAF-S-Met | ||||
| THF | 1.4 | 0.1 | 0 | 0 |
| MeOH | 3.4 | 2.1 | 1.5 | 0.6 |
| ACN | 16.3 | 12.1 | 9.3 | 3.3 |
| Acetone | 13.0 | 8.7 | 5.8 | 3.2 |
| DCM | 125.6 | 43.8 | 18.3 | 11.1 |
| ST+20% 1,4(1,5)-NAF-CH2S-Met | ||||
| THF | 152.0 | 113.0 | 29.6 | 16.6 |
| MeOH | 0.5 | 0.3 | 0.1 | 0.03 |
| ACN | 3.4 | 3.1 | 2.5 | 0.9 |
| Acetone | 31.8 | 27.0 | 21.2 | 11.7 |
| DCM | 171.4 | 33.7 | 1.9 | 0.4 |
| MMA+20% 1,4(1,5)-NAF-CH2S-Met | ||||
| THF | 1.7 | 0.6 | 0.5 | 0.1 |
| MeOH | 1.4 | 0.9 | 0.4 | 0 |
| ACN | 26.5 | 20.9 | 15.6 | 7.1 |
| Acetone | 11.6 | 6.8 | 3.9 | 2.2 |
| DCM | 149.2 | 30.0 | 7.5 | 1.1 |
| Solvent | Sample | |||
|---|---|---|---|---|
| 1c | 2c | 3c | 4c | |
| Swellability Coefficient, B/% | ||||
| THF | 19 | 43 | 25 | 70 |
| MeOH | 205 | 352 | 114 | 179 |
| ACN | 14 | 45 | 17 | 19 |
| Acetone | 44 | 84 | 41 | 50 |
| DCM | 25 | 55 | 16 | 19 |
| Distilled Water | 93 | 138 | 72 | 90 |
| Sample | DSC Data | ||
|---|---|---|---|
| Tg/°C | Td/°C | ΔHd/J g−1 | |
| 1a | 103 | 380 | 515 |
| 1b | 112 | 354 | 294 |
| 1c | 83 | 356; 413 | 406 |
| 2a | 122 | 261; 336; 398 | 823 |
| 2b | 135 | 302; 338; 353 | 340 |
| 2c | 84 | 349; 422 | 458 |
| 3a | 102 | 408 | 741 |
| 3b | - | 347; 367; 472 | 201 |
| 3c | 105 | 348; 376; 434 | 703 |
| 4a | 120 | 337; 358 | 764 |
| 4b | - | 338; 441 | 253 |
| 4c | 60 | 384; 433 | 432 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fila, K.; Podkościelna, B.; Podgórski, M. Cross-Linked Polythiomethacrylate Esters Based on Naphthalene—Synthesis, Properties and Reprocessing. Materials 2020, 13, 3021. https://doi.org/10.3390/ma13133021
Fila K, Podkościelna B, Podgórski M. Cross-Linked Polythiomethacrylate Esters Based on Naphthalene—Synthesis, Properties and Reprocessing. Materials. 2020; 13(13):3021. https://doi.org/10.3390/ma13133021
Chicago/Turabian StyleFila, Karolina, Beata Podkościelna, and Maciej Podgórski. 2020. "Cross-Linked Polythiomethacrylate Esters Based on Naphthalene—Synthesis, Properties and Reprocessing" Materials 13, no. 13: 3021. https://doi.org/10.3390/ma13133021
APA StyleFila, K., Podkościelna, B., & Podgórski, M. (2020). Cross-Linked Polythiomethacrylate Esters Based on Naphthalene—Synthesis, Properties and Reprocessing. Materials, 13(13), 3021. https://doi.org/10.3390/ma13133021