Influence of Controlled Cooling on Crystallinity of Poly(L-Lactic Acid) Scaffolds after Hydrolytic Degradation
Abstract
:1. Introduction
1.1. Justification
1.2. Related Work
1.3. Objective
2. Materials and Methods
2.1. Polymer
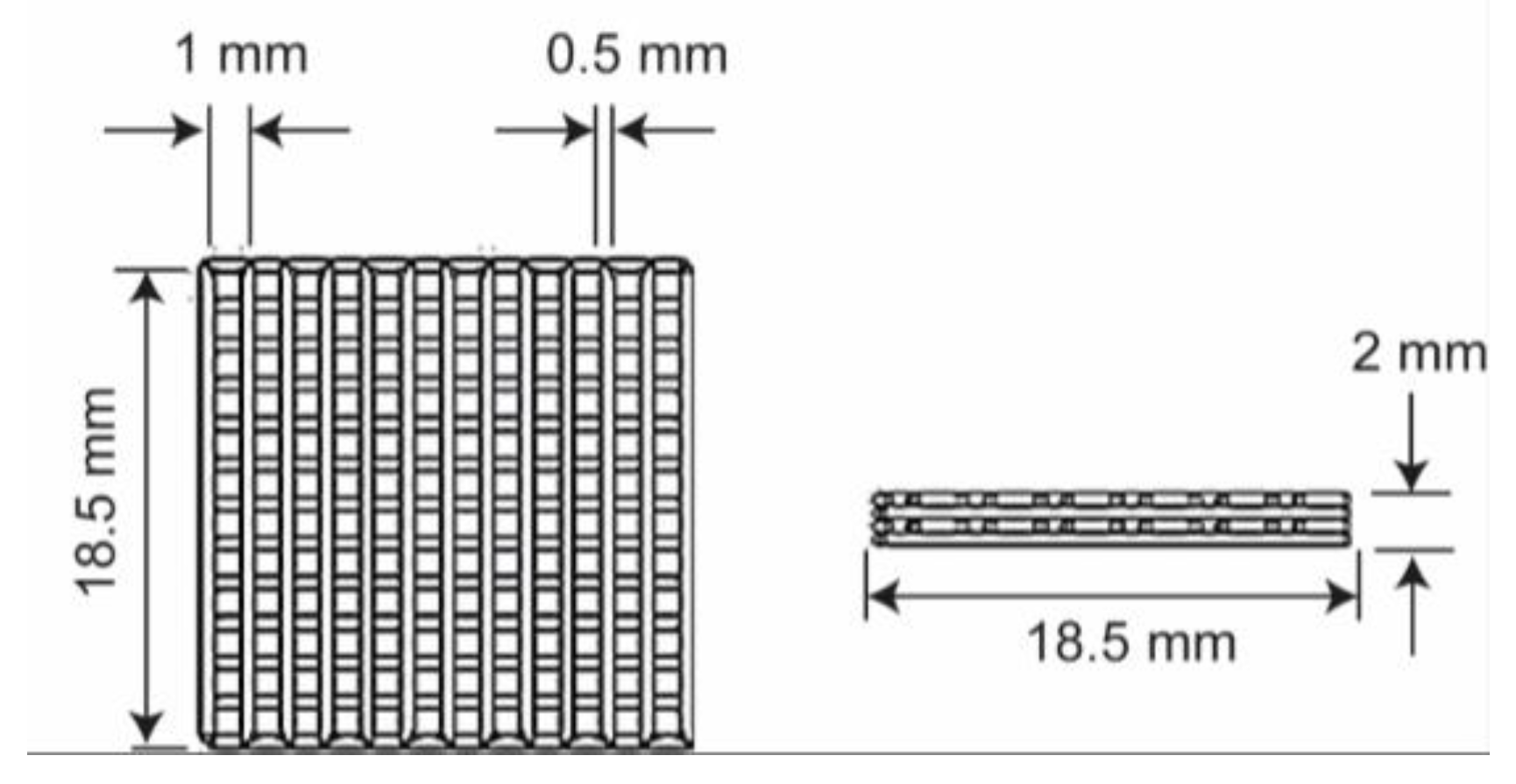
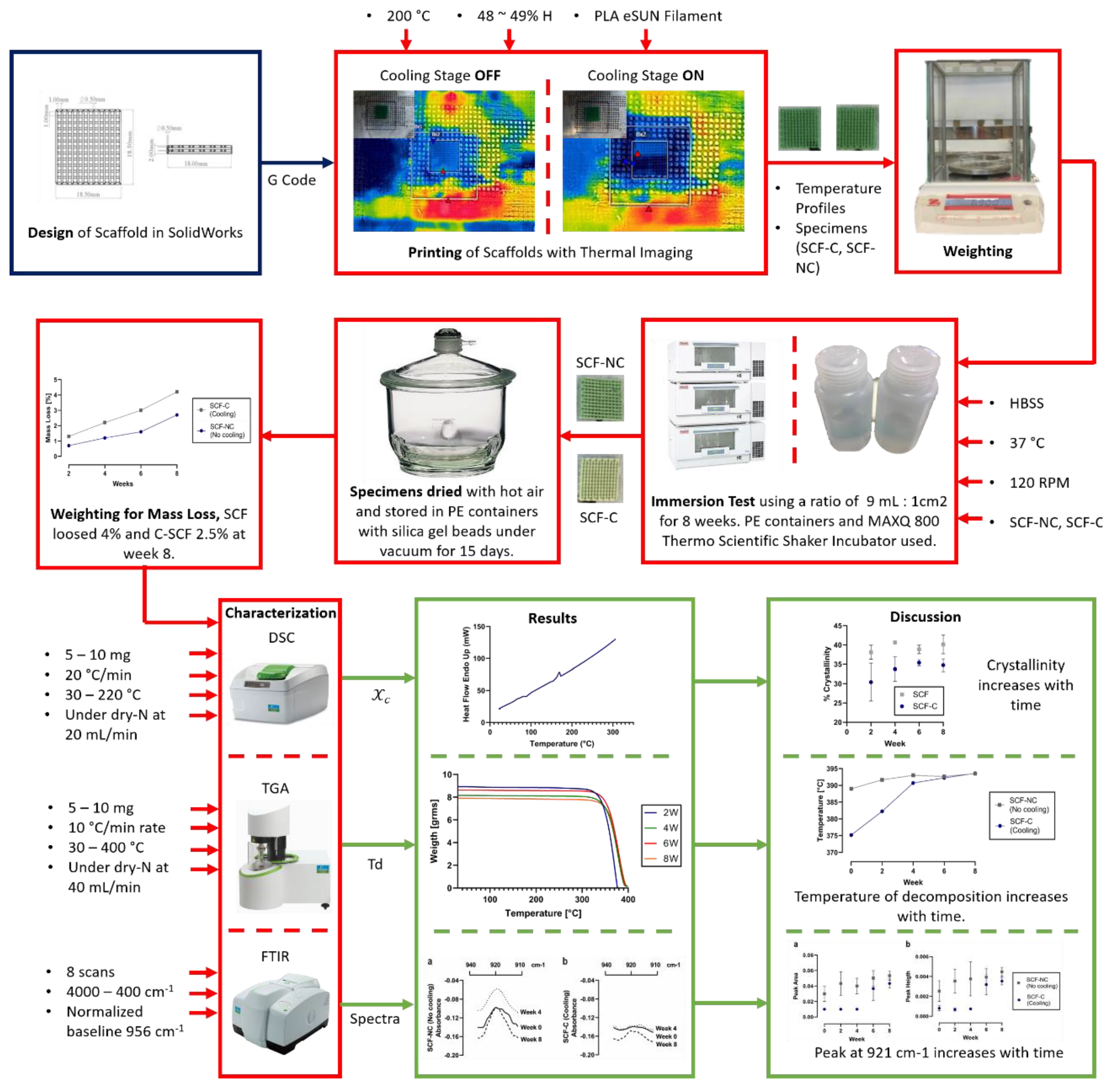
2.2. Manufacturing Process
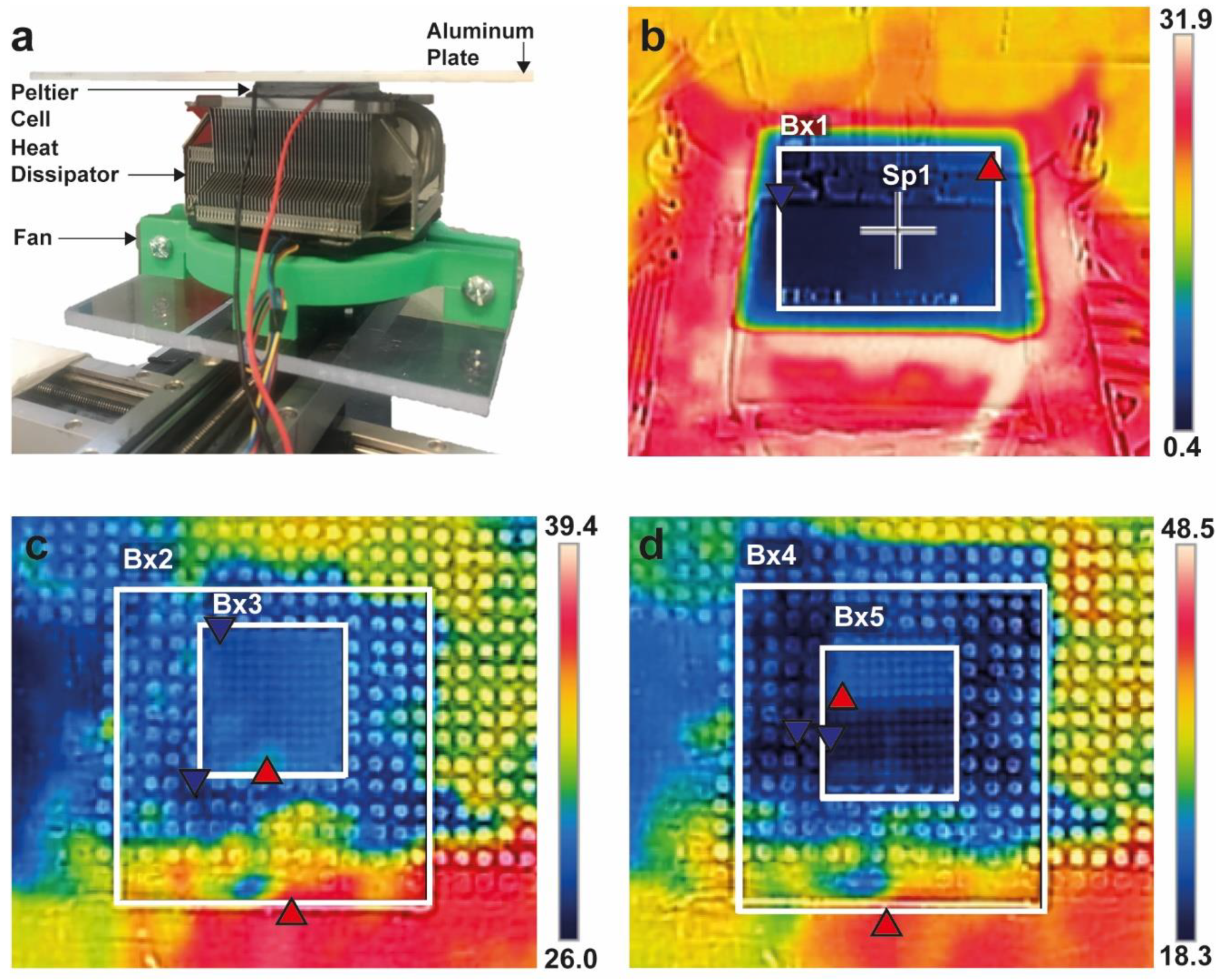
2.3. Thermal Analysis of the Printing Process
2.4. Immersion Test
2.5. Crystallinity Measurements
3. Results
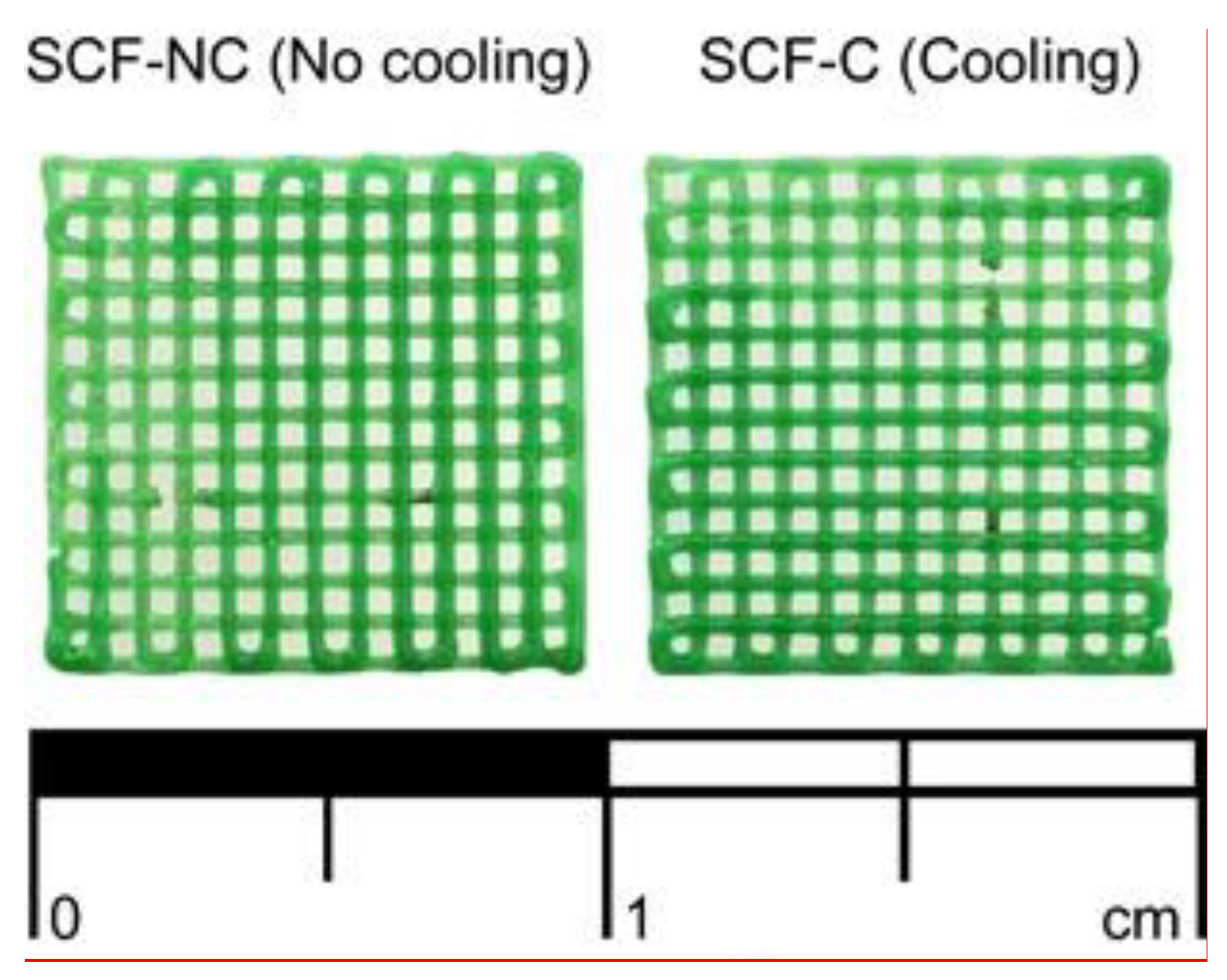
3.1. Manufacturing Process
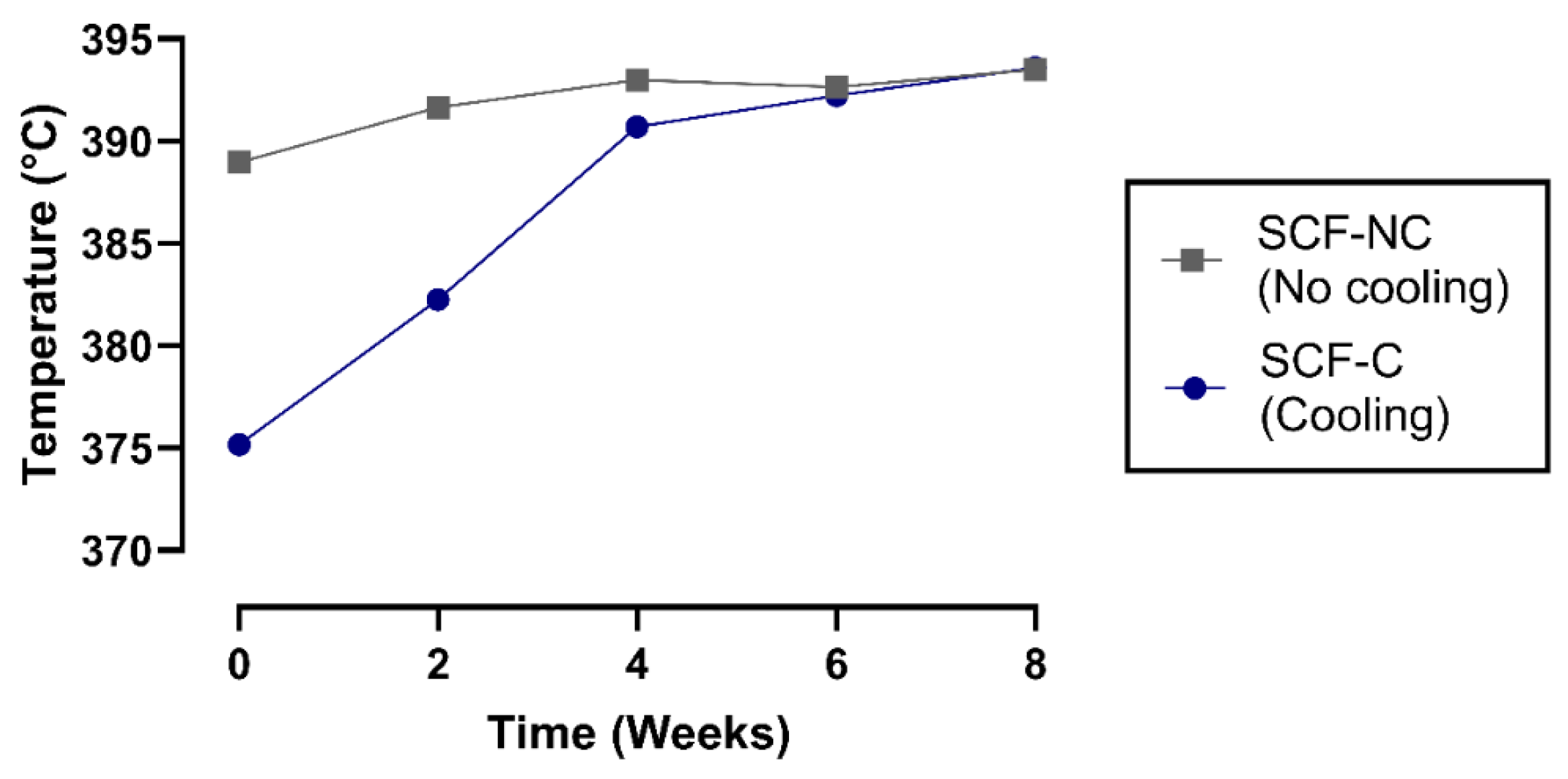
3.2. Thermal Analysis of Manufacturing Process
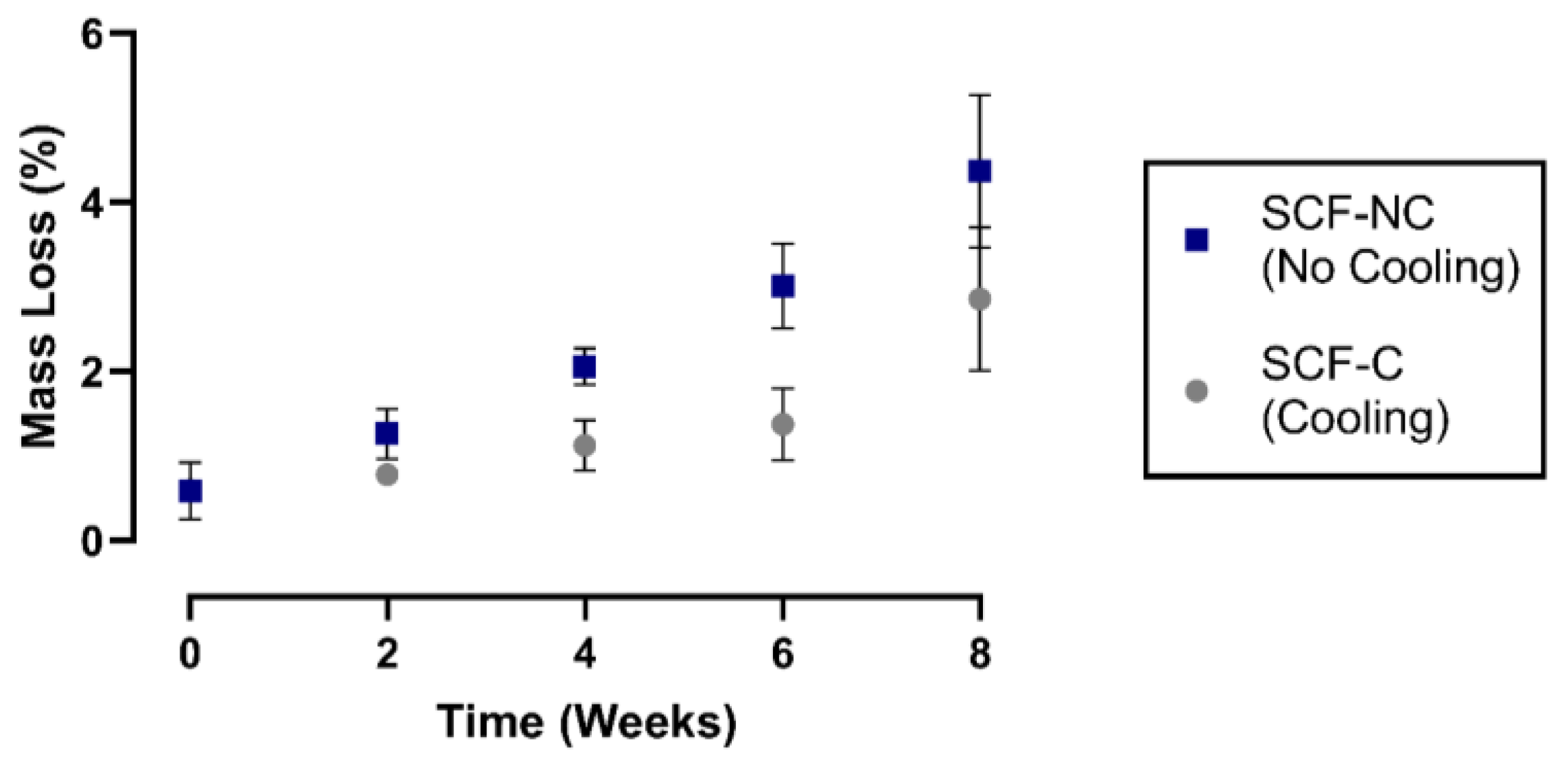
3.3. Mass Loss of Scaffolds after the Immersion Test
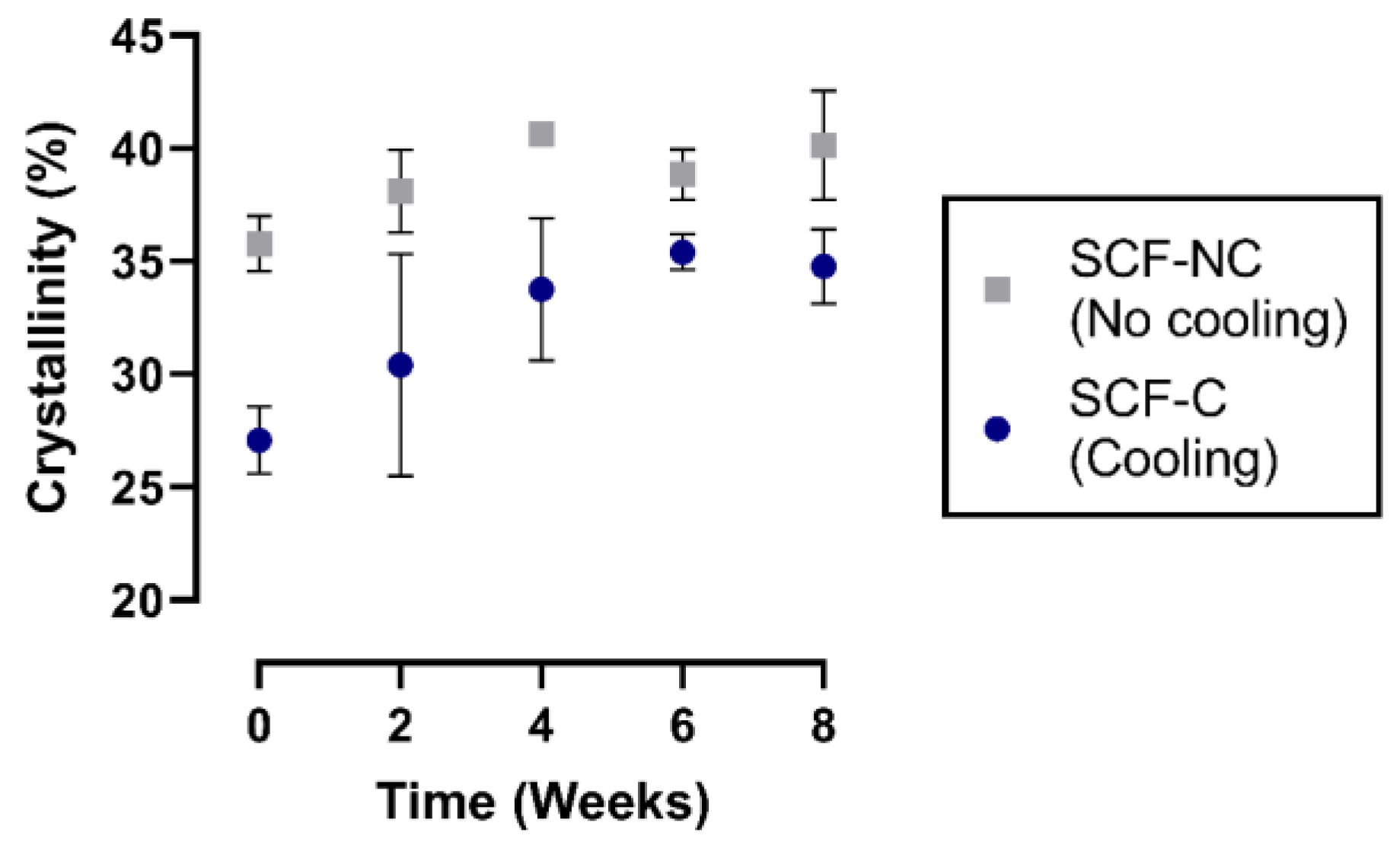
3.4. Crystallinity Measurements
4. Discussion
4.1. Thermal Analysis of Manufacturing Process
4.2. Mass Loss of Scaffolds after Immersion Test
4.3. Crystallinity Measurements after Immersion
4.4. Relationship between 3D Printing Process Parameters and PLA Scaffold Degradation
5. Conclusions and Future Work
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Nomenclature
| Symbol | Description |
| PLA | Polylactic acid |
| PLLA | Poly (L-lactide acid) |
| FDM | Fused Deposition Modeling |
| HBSS | Hank´s Base Solution |
| AM | Additive Manufacturing |
| CAD | Computer Aided Design |
| PEEK | Poly-Ether-Ether-Ketone |
| PID | Proportional-Integrative-Derivative control |
| SCF-NC | Scaffolds printed with No-Cooling |
| SCF-C | Scaffolds printed with Cooling |
| PE | Polyethylene |
| TGA | Thermo Gravimetric Analysis |
| FTIR | Fourier Transformed Infrared spectroscopy |
| DSC | Differential Scanning Calorimetry |
| Crystallinity | |
| Tg | Glass transition temperature |
| Tm | Melting temperature |
| ΔH | Heat of fusion |
| ΔHo | Theoretical heat of fusion for 100% crystalline PLA |
| ΔHS | Heat of fusion for sample |
References
- Rodriguez, C.A.; Lara-Padilla, H.; Dean, D. Bioceramics for musculoskeletal regenerative medicine: Materials and manufacturing process compatibility for synthetic bone grafts and medical devices. In 3D Printing and Biofabrication; Ovsianikov, A., Yoo, J., Mironov, V., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 161–193. ISBN 978-3-319-45443-6. [Google Scholar]
- Parvizi, J.; Kim, G.K. Chapter 32—Bone grafting. In High Yield Orthopaedics; Parvizi, J., Kim, G.K., Eds.; W.B. Saunders: Philadelphia, PA, USA, 2010; pp. 64–65. ISBN 978-1-4160-0236-9. [Google Scholar]
- Research and Markets. Bone Graft and Substitutes Market: Global Industry Trends, Share, Size, Growth, Opportunity and Forecast 2019–2024. Available online: https://www.researchandmarkets.com/reports/4828749/bone-graft-and-substitutes-market-global (accessed on 18 February 2020).
- Reichert, J.C.; Wullschleger, M.E.; Cipitria, A.; Lienau, J.; Cheng, T.K.; Schütz, M.A.; Duda, G.N.; Nöth, U.; Eulert, J.; Hutmacher, D.W. Custom-made composite scaffolds for segmental defect repair in long bones. Int. Orthop. (SICOT) 2011, 35, 1229–1236. [Google Scholar] [CrossRef] [Green Version]
- Iaquinta, M.R.; Mazzoni, E.; Manfrini, M.; D’Agostino, A.; Trevisiol, L.; Nocini, R.; Trombelli, L.; Barbanti-Brodano, G.; Martini, F.; Tognon, M. Innovative biomaterials for bone regrowth. Int. J. Mol. Sci. 2019, 20, 618. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gorrasi, G.; Pantani, R. Effect of PLA grades and morphologies on hydrolytic degradation at composting temperature: Assessment of structural modification and kinetic parameters. Polymer Degrad. Stab. 2013, 98, 1006–1014. [Google Scholar] [CrossRef]
- Lam, C.X.; Teoh, S.H.; Hutmacher, D.W. Comparison of the degradation of polycaprolactone and polycaprolactone-(β-tricalcium phosphate) scaffolds in alkaline medium. Polymer Int. 2007, 56, 718–728. [Google Scholar] [CrossRef]
- Orciani, M.; Fini, M.; Di Primio, R.; Mattioli-Belmonte, M. Biofabrication and bone tissue regeneration: Cell source, approaches, and challenges. Front. Bioeng. Biotechnol. 2017, 5. [Google Scholar] [CrossRef] [Green Version]
- Farzamfar, S.; Naseri-Nosar, M.; Sahrapeyma, H.; Ehterami, A.; Goodarzi, A.; Rahmati, M.; Lakalayeh, G.A.; Ghorbani, S.; Vaez, A.; Salehi, M. Tetracycline hydrochloride-containing poly (ε-caprolactone)/poly lactic acid scaffold for bone tissue engineering application: In vitro and in vivo study. Int. J. Polymer. Mater. Polymer. Biomater. 2019, 68, 472–479. [Google Scholar] [CrossRef]
- Tan, G.Z.; Zhou, Y. Tunable 3D nanofiber architecture of polycaprolactone by divergence electrospinning for potential tissue engineering applications. Nano Micro Lett. 2018, 10, 73. [Google Scholar] [CrossRef] [Green Version]
- Lara-Padilla, H.; Mendoza-Buenrostro, C.; Cardenas, D.; Rodriguez-Garcia, A.; Rodriguez, C.A. Influence of controlled cooling in bimodal scaffold fabrication using polymers with different melting temperatures. Materials 2017, 10, 640. [Google Scholar] [CrossRef] [Green Version]
- Shin, H.; Jo, S.; Mikos, A.G. Biomimetic materials for tissue engineering. Biomaterials 2003, 24, 4353–4364. [Google Scholar] [CrossRef]
- Avérous, L.; Pollet, E. Biodegradable polymers. In Environmental Silicate Nano-Biocomposites; Avérous, L., Pollet, E., Eds.; Green Energy and Technology; Springer London: London, UK, 2012; pp. 13–39. ISBN 978-1-4471-4101-3. [Google Scholar]
- Grémare, A.; Guduric, V.; Bareille, R.; Heroguez, V.; Latour, S.; L’heureux, N.; Fricain, J.-C.; Catros, S.; Nihouannen, D.L. Characterization of printed PLA scaffolds for bone tissue engineering. J. Biomed. Mater. Res. Part A 2018, 106, 887–894. [Google Scholar] [CrossRef]
- Teixeira, B.; Maia-Pinto, M.O.C.; Calasans-Maia, M.D.; Kelly, D.J.; Thiré, R.M.S.M. Structural evaluation of PLA scaffolds obtained by 3D printing via fused deposition modeling (FDM) technique for applications in tissue engineering. Front. Bioeng. Biotechnol. 2016, 4. [Google Scholar] [CrossRef]
- Maia-Pinto, M.O.C.; Maia, M.C.; Thiré, R.M.S.M. Estudo da biocompatibilidade in vivo de arcabouço de poli(ácido lático) (PLA) fabricados por impressão 3d para aplicações em engenharia tecidual. In A Produção do Conhecimento nas Ciências da Saúde 5; Atena Editora: Sao Paulo, Brazil, 2019; pp. 118–125. ISBN 978-85-7247-302-6. [Google Scholar]
- Medtronic. ProGripTM Laparoscopic Self-Fixating Mesh. Available online: https://www.medtronic.com/covidien/en-us/products/hernia-repair/progrip-laparoscopic-self-fixating-mesh.html (accessed on 9 June 2020).
- Campanelli, G. The Art of Hernia Surgery: A Step-by-Step Guide; Springer: New York, NY, USA, 2018; ISBN 978-3-319-72626-7. [Google Scholar]
- Cutright, D.E.; Hunsuck, E.E. Tissue reaction to the biodegradable polylactic acid suture. Oral Surg. Oral Med. Oral Pathol. 1971, 31, 134–139. [Google Scholar] [CrossRef]
- Zielhuis, S.W.; Nijsen, J.F.W.; Seppenwoolde, J.-H.; Bakker, C.J.G.; Krijger, G.C.; Dullens, H.F.J.; Zonnenberg, B.A.; van Rijk, P.P.; Hennink, W.E.; van het Schip, A.D. Long-term toxicity of holmium-loaded poly(L-lactic acid) microspheres in rats. Biomaterials 2007, 28, 4591–4599. [Google Scholar] [CrossRef]
- Schwach, G.; Vert, M. In vitro and in vivo degradation of lactic acid-based interference screws used in cruciate ligament reconstruction. Int. J. Biol. Macromol. 1999, 25, 283–291. [Google Scholar] [CrossRef]
- Tschakaloff, A.; Losken, H.W.; von Oepen, R.; Michaeli, W.; Moritz, O.; Mooney, M.P.; Losken, A. Degradation kinetics of biodegradable dl-polylactic acid biodegradable implants depending on the site of implantation. Int. J. Oral Maxillofac. Surg. 1994, 23, 443–445. [Google Scholar] [CrossRef]
- Liao, S.S.; Cui, F.Z.; Zhang, W.; Feng, Q.L. Hierarchically biomimetic bone scaffold materials: Nano-HA/collagen/PLA composite. J. Biomed. Mater. Res. Part B Appl. Biomater. 2004, 69, 158–165. [Google Scholar] [CrossRef]
- Schliephake, H.; Weich, H.A.; Dullin, C.; Gruber, R.; Frahse, S. Mandibular bone repair by implantation of rhBMP-2 in a slow release carrier of polylactic acid—An experimental study in rats. Biomaterials 2008, 29, 103–110. [Google Scholar] [CrossRef] [Green Version]
- Sabir, M.I.; Xu, X.; Li, L. A review on biodegradable polymeric materials for bone tissue engineering applications. J. Mater. Sci. 2009, 44, 5713–5724. [Google Scholar] [CrossRef]
- Harris, M.; Potgieter, J.; Archer, R.; Arif, K.M. Effect of material and process specific factors on the strength of printed parts in fused filament fabrication: A review of recent developments. Materials 2019, 12, 1664. [Google Scholar] [CrossRef] [Green Version]
- Kotsilkova, R.; Angelova, P.; Batakliev, T.; Angelov, V.; Di Maio, R.; Silvestre, C. Study on aging and recover of poly (lactic) acid composite films with graphene and carbon nanotubes produced by solution blending and extrusion. Coatings 2019, 9, 359. [Google Scholar] [CrossRef] [Green Version]
- Jain, S.; Fuoco, T.; Yassin, M.A.; Mustafa, K.; Finne-Wistrand, A. Printability and critical insight into polymer properties during direct-extrusion based 3D printing of medical grade polylactide and copolyesters. Biomacromolecules 2020, 21, 388–396. [Google Scholar] [CrossRef]
- Zhang, W.; Ullah, I.; Shi, L.; Zhang, Y.; Ou, H.; Zhou, J.; Ullah, M.W.; Zhang, X.; Li, W. Fabrication and characterization of porous polycaprolactone scaffold via extrusion-based cryogenic 3D printing for tissue engineering. Mater. Design 2019, 180, 107946. [Google Scholar] [CrossRef]
- Ma, Y.; Dai, H.; Huang, X.; Long, Y. 3D printing of bioglass-reinforced β-TCP porous bioceramic scaffolds. J. Mater. Sci. 2019, 54, 10437–10446. [Google Scholar] [CrossRef]
- Cengiz, I.F.; Pereira, H.; Espregueira-Mendes, J.; Kwon, I.K.; Reis, R.L.; Oliveira, J.M. Suturable regenerated silk fibroin scaffold reinforced with 3D-printed polycaprolactone mesh: Biomechanical performance and subcutaneous implantation. J. Mater. Sci. Mater. Med. 2019, 30, 63. [Google Scholar] [CrossRef]
- Li, X.; Wang, Y.; Wang, Z.; Qi, Y.; Li, L.; Zhang, P.; Chen, X.; Huang, Y. Composite PLA/PEG/nHA/dexamethasone scaffold prepared by 3D printing for bone regeneration. Macromol. Biosci. 2018, 18, 1800068. [Google Scholar] [CrossRef]
- Yang, C.; Tian, X.; Li, D.; Cao, Y.; Zhao, F.; Shi, C. Influence of thermal processing conditions in 3D printing on the crystallinity and mechanical properties of PEEK material. J. Mater. Process. Technol. 2017, 248, 1–7. [Google Scholar] [CrossRef]
- Harris, M.; Potgieter, J.; Archer, R.; Arif, K.M. In-process thermal treatment of polylactic acid in fused deposition modelling. Mater. Manuf. Process. 2019, 34, 701–713. [Google Scholar] [CrossRef]
- Ferreira, J.; Gloria, A.; Cometa, S.; Coelho, J.F.J.; Domingos, M. Effect of in vitro enzymatic degradation on 3D printed poly(ε-caprolactone) scaffolds: Morphological, chemical and mechanical properties. JABFM 2017, 15. [Google Scholar] [CrossRef] [Green Version]
- Benwood, C.; Anstey, A.; Andrzejewski, J.; Misra, M.; Mohanty, A.K. Improving the impact strength and heat resistance of 3D printed models: Structure, property, and processing correlationships during fused deposition modeling (FDM) of poly(lactic acid). ACS Omega 2018, 3, 4400–4411. [Google Scholar] [CrossRef]
- Mendoza-Buenrostro, C.; Lara, H.; Rodriguez, C. Hybrid fabrication of a 3D printed geometry embedded with PCL nanofibers for tissue engineering applications. Procedia Eng. 2015, 110, 128–134. [Google Scholar] [CrossRef] [Green Version]
- eSun. Safety Data Sheet PLA Filament. Available online: http://www.esun3d.net/UploadFiles/Download/MSDS_Esun_PLA_PLUS_filament.pdf (accessed on 8 June 2020).
- Vasanthan, N.; Ly, O. Effect of microstructure on hydrolytic degradation studies of poly (L-lactic acid) by FTIR spectroscopy and differential scanning calorimetry. Polymer Degrad. Stab. 2009, 94, 1364–1372. [Google Scholar] [CrossRef]
- Meaurio, E.; López-Rodríguez, N.; Sarasua, J.R. Infrared spectrum of poly(l-lactide): Application to crystallinity studies. Macromolecules 2006, 39, 9291–9301. [Google Scholar] [CrossRef]
- Carrasco, F.; Pagès, P.; Gámez-Pérez, J.; Santana, O.O.; Maspoch, M.L. Processing of poly(lactic acid): Characterization of chemical structure, thermal stability and mechanical properties. Polymer Degrad. Stab. 2010, 95, 116–125. [Google Scholar] [CrossRef]
- Tsuji, H.; Tezuka, Y.; Yamada, K. Alkaline and enzymatic degradation of l-lactide copolymers. II. crystallized films of poly(L-lactide-co-D-lactide) and poly(L-lactide) with similar crystallinities. J. Polymer Sci. Part B Polymer Phys. 2005. [Google Scholar] [CrossRef]
- Iannace, S.; Maffezzoli, A.; Leo, G.; Nicolais, L. Influence of crystal and amorphous phase morphology on hydrolytic degradation of PLLA subjected to different processing conditions. Polymer 2001. [Google Scholar] [CrossRef]



| Material | Technique | Characterization Parameters | Ref |
|---|---|---|---|
| Poly(L-lactide) (PLLA) poly(L-lactide-co-ε-caprolactone) (PCLA) poly(L-lactide-co-glycolide) (PLGA) poly(D,L-lactide-co-glycolide) (PDLGA) | Direct extrusion-based 3D printing |
| [28] |
| Polycaprolactone (PCL) | Extrusion based cryogenic 3D printing (ECP) (−20 °C) and subsequent freeze-drying approaches |
| [29] |
| Na2O–CaO–MgO–P2O5 Bioglass reinforced β-TCP | Inkjet 3D printing technology |
| [30] |
| Polycaprolactone (PCL) | 3D-Bioplotter |
| [31] |
| Polylactic acid (PLA)/ Polyethylene glycol (PEG)/ (nano hydroxyapatite (nHA)/Dexamethasone (Dex) | Fused deposition modeling (FDM) process |
| [32] |
| Poly-Ether-Ether-Ketone (PEEK) | Temperature control system into fused deposition modeling (FDM) process |
| [33] |
| Polylactic acid (PLA) | Continuous heat transfer during fused deposition modeling (FDM) process |
| [34] |
| Polylactic acid (PLA) | Fused deposition modeling (FDM) process |
| [14] |
| Polycaprolactone (PCL) | BioExtruder Extrusion-based additive manufacturing (AM) |
| [35] |
| Polylactic acid (PLA) | Fused deposition modeling (FDM) process |
| [36] |
| Property | Value |
|---|---|
| Density (kg/m3) | 1.20–1.25 |
| Melting Point (°C) | 190–220 |
| Tensile Yield Strength (MPa) | 65.63 |
| Flexural Strength (MPa) | 65.02 |
| Flexural Modulus (MPa) | 2504.4 |
| Glass Transition Temperature (°C) | 56–60 * |
| Crystallization Temperature (°C) | 130–173 ** |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vazquez-Armendariz, J.; Tejeda-Alejandre, R.; Rodriguez-Garcia, A.; Vega-Cantu, Y.I.; Mendoza-Buenrostro, C.; Rodriguez, C.A. Influence of Controlled Cooling on Crystallinity of Poly(L-Lactic Acid) Scaffolds after Hydrolytic Degradation. Materials 2020, 13, 2943. https://doi.org/10.3390/ma13132943
Vazquez-Armendariz J, Tejeda-Alejandre R, Rodriguez-Garcia A, Vega-Cantu YI, Mendoza-Buenrostro C, Rodriguez CA. Influence of Controlled Cooling on Crystallinity of Poly(L-Lactic Acid) Scaffolds after Hydrolytic Degradation. Materials. 2020; 13(13):2943. https://doi.org/10.3390/ma13132943
Chicago/Turabian StyleVazquez-Armendariz, Javier, Raquel Tejeda-Alejandre, Aida Rodriguez-Garcia, Yadira I. Vega-Cantu, Christian Mendoza-Buenrostro, and Ciro A. Rodriguez. 2020. "Influence of Controlled Cooling on Crystallinity of Poly(L-Lactic Acid) Scaffolds after Hydrolytic Degradation" Materials 13, no. 13: 2943. https://doi.org/10.3390/ma13132943






