On the Dependence of Rheology of Hyaluronic Acid Solutions and Frictional Behavior of Articular Cartilage
Abstract
1. Introduction
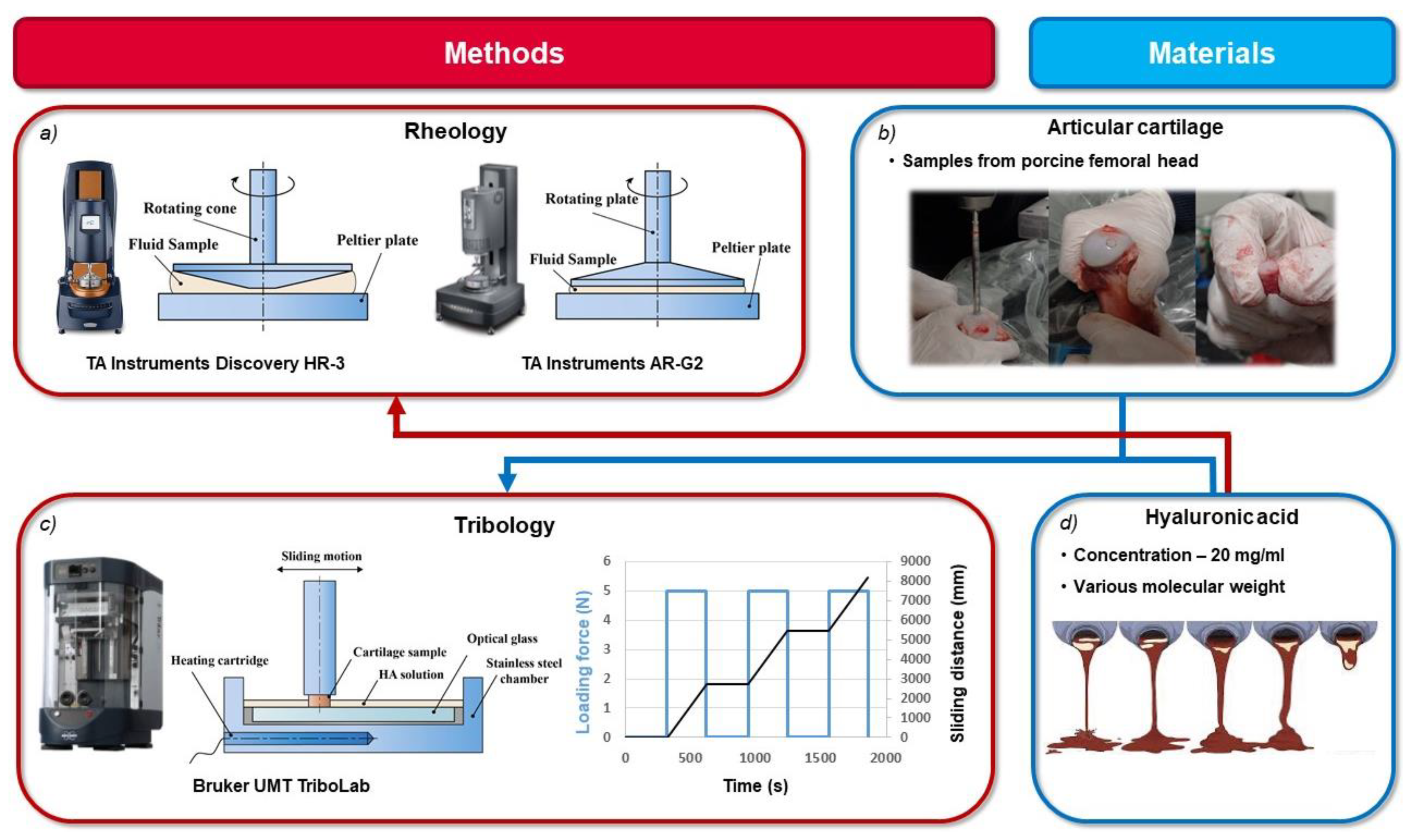
2. Materials and Methods
3. Results and Discussion
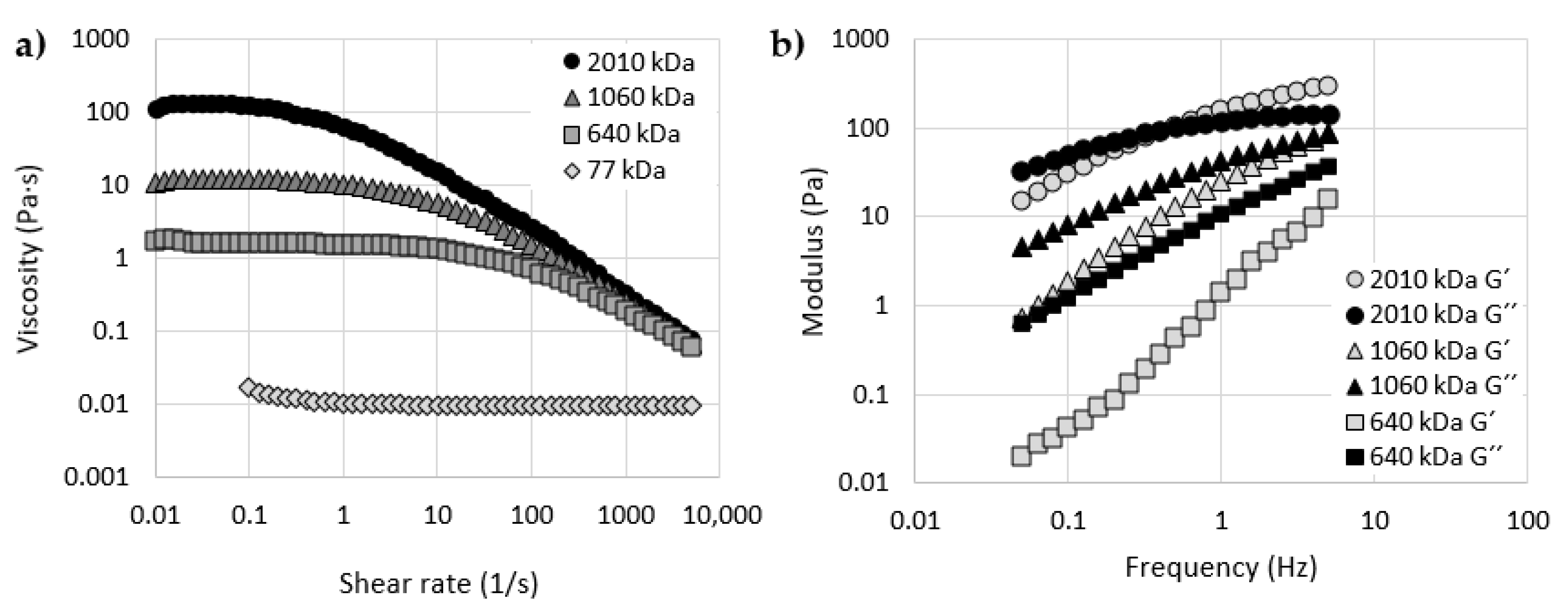
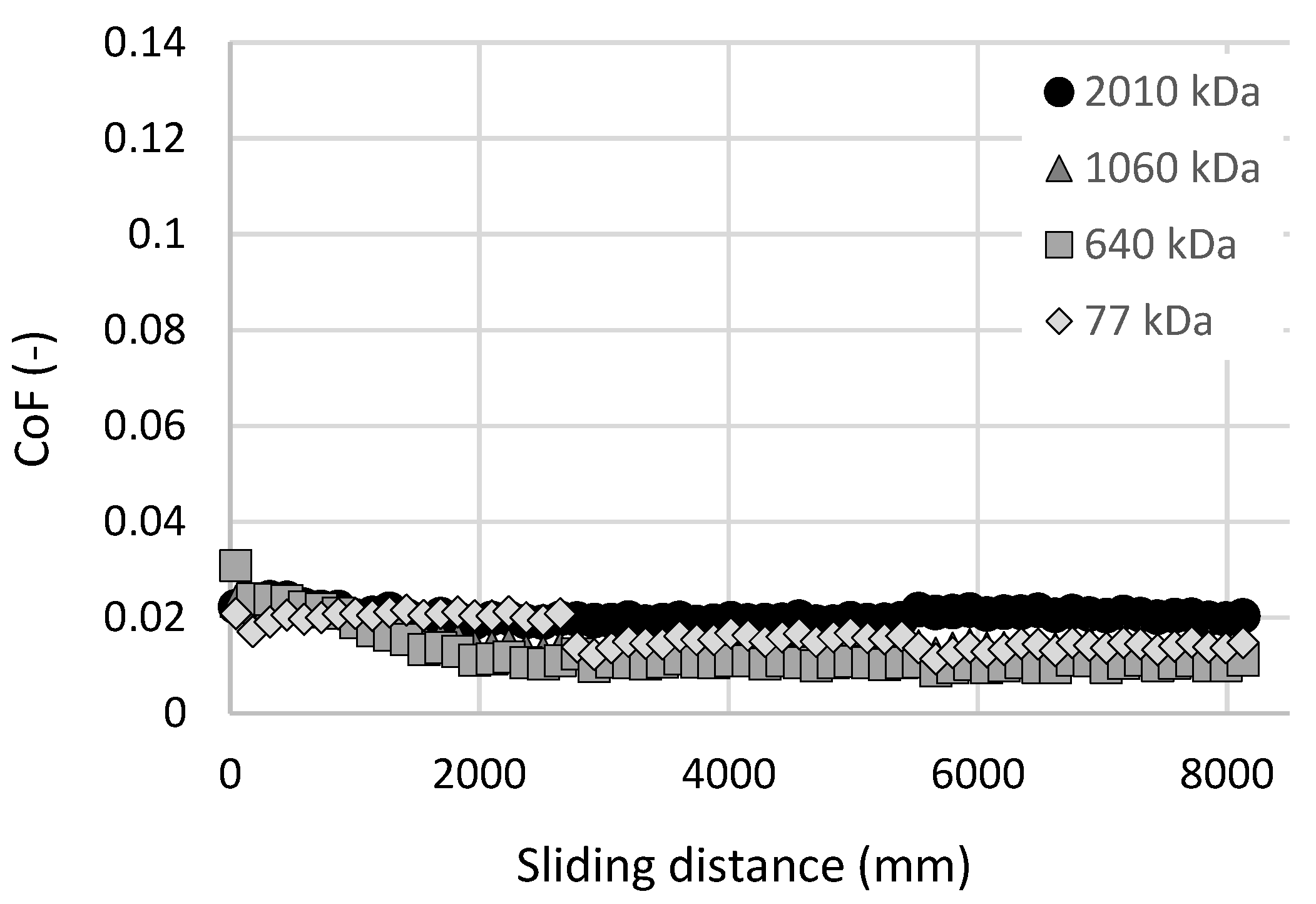
3.1. Rheology of HA
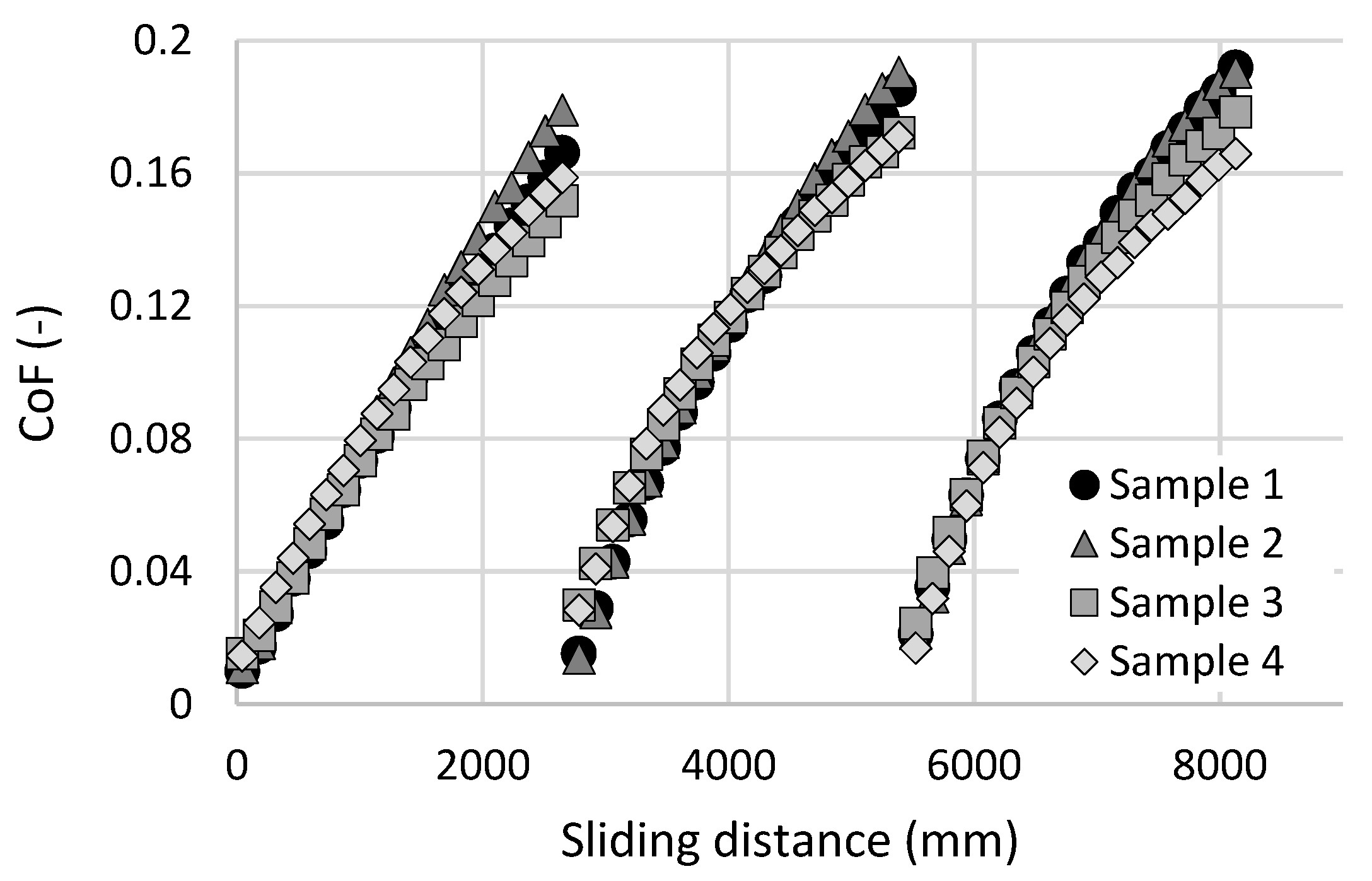
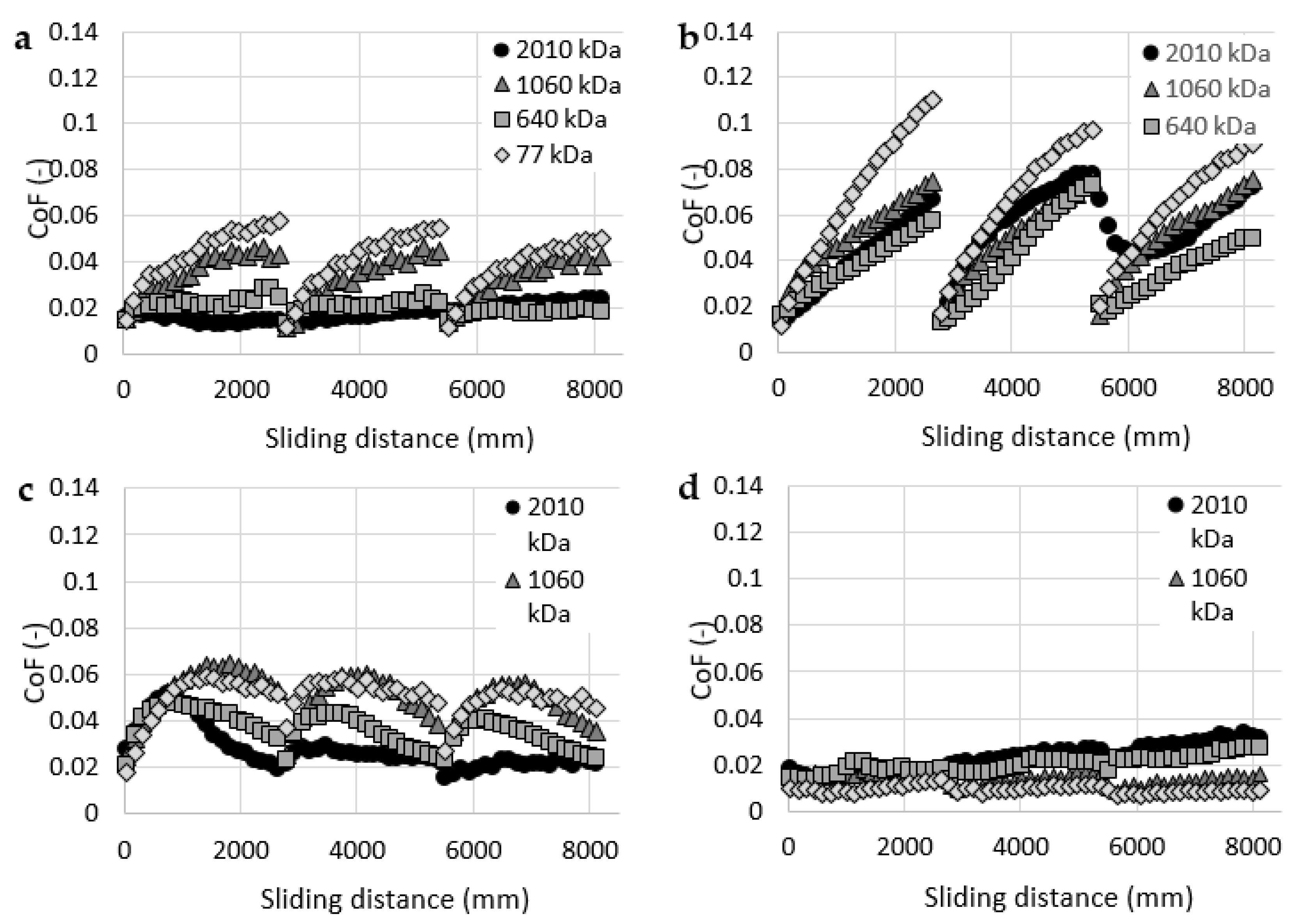
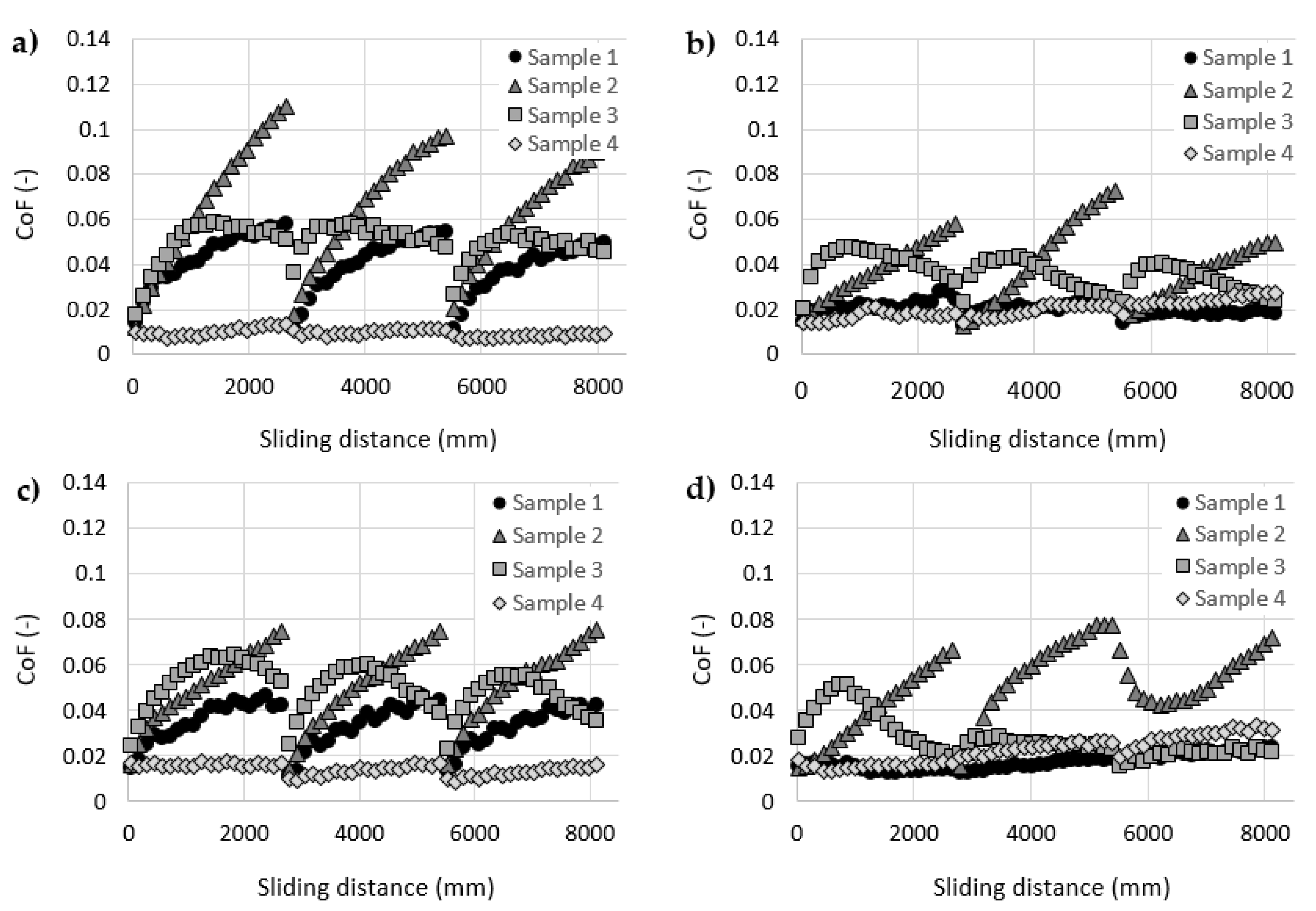
3.2. Cartilage Friction Analysis
4. Conclusions
- Rheological measurements showed a strong dependency between the molecular weight and the viscosity or viscoelastic properties of HA solutions. HA solutions with higher molecular weights exhibited higher viscosity, dynamic moduli and shear thinning ratio.
- The crossover point was measured only for one of the tested HA samples. Based on the obtained data, it can be assumed that a higher molecular weight of HA leads to lower values of crossover frequency.
- CoF measurements showed a substantial dispersion in the results, showing no clear dependency between the HA molecular weight and the friction in the cartilage-on-glass contact.
- Mechanical properties and overall conditions of individual cartilage samples can significantly affect the effectiveness of HA solutions during the reciprocating sliding motion. In most cases, each cartilage sample exhibited the highest and the lowest values of CoF during measurements with different HA solution.
- Unclear results may support the contradictory conclusions of medical studies whose results are strongly dependent on the individual patient’s conditions. The cartilage condition and composition of synovial fluid can significantly affect the effectiveness of viscosupplementation.
- Different results of rheological and frictional measurements might also show the insufficiency of rheological measurements in the assessment of viscosupplements effectiveness.
Author Contributions
Funding
Conflicts of Interest
References
- Lees, D.; Partington, P. Articular cartilage. Orthop. Trauma 2016, 30, 265–272. [Google Scholar] [CrossRef]
- Goldring, M.B. Chondrogenesis, chondrocyte differentiation, and articular cartilage metabolism in health and osteoarthritis. Ther. Adv. Musculoskelet. Dis. 2012, 4, 269–285. [Google Scholar] [CrossRef] [PubMed]
- Ateshian, G.A. The role of interstitial fluid pressurization in articular cartilage lubrication. J. Biomech. 2009, 42, 1163–1176. [Google Scholar] [CrossRef] [PubMed]
- Forsey, R.; Fisher, J.; Thompson, J.; Stone, M.; Bell, C.; Ingham, E. The effect of hyaluronic acid and phospholipid-based lubricants on friction within a human cartilage damage model. Biomaterials 2006, 27, 4581–4590. [Google Scholar] [CrossRef] [PubMed]
- Galandáková, A.; Ulrichová, J.; Langová, K.; Hanáková, A.; Vrbka, M.; Hartl, M.; Gallo, J. Characteristics of synovial fluid required for optimization of lubrication fluid for biotribological experiments. J. Biomed. Mater. Res. B Appl. Biomater. 2017, 105, 1422–1431. [Google Scholar] [CrossRef]
- Balazs, E.A.; Denlinger, J.L. Viscosupplementation: A new concept in the treatment of osteoarthritis. J. Rheumatol. Suppl. 1993, 39, 3–9. [Google Scholar]
- Ghosh, P.; Guidolin, D. Potential mechanism of action of intra-articular hyaluronan therapy in osteoarthritis: Are the effects molecular weight dependent? Semin. Arthritis Rheum. 2002, 32, 10–37. [Google Scholar] [CrossRef]
- Altman, R.D.; Manjoo, A.; Fierlinger, A.; Niazi, F.; Nicholls, M. The mechanism of action for hyaluronic acid treatment in the osteoarthritic knee: A systematic review. BMC Musculoskelet. Disord. 2015, 16, 321. [Google Scholar] [CrossRef]
- Watterson, J.R.; Esdaile, J.M. Viscosupplementation: Therapeutic Mechanisms and clinical potential in osteoarthritis of the knee. J. Am. Acad. Orthop. Surg. 2000, 8, 277–284. [Google Scholar] [CrossRef]
- Mazzucco, D.; Scott, R.; Spector, M. Composition of joint fluid in patients undergoing total knee replacement and revision arthroplasty: Correlation with flow properties. Biomaterials 2004, 25, 4433–4445. [Google Scholar] [CrossRef]
- Ghosh, P. The role of hyaluronic acid (hyaluronan) in health and disease: Interactions with cells, cartilage and components of synovial fluid. Clin. Exp. Rheumatol. 1994, 12, 75–82. [Google Scholar] [PubMed]
- Zhang, Z.; Barman, S.; Christopher, G.F. The role of protein content on the steady and oscillatory shear rheology of model synovial fluids. Soft Matter 2014, 10, 5965–5973. [Google Scholar] [CrossRef] [PubMed]
- Balazs, E.A. Viscoelastic properties of hyaluronan and its therapeutic use. In Chemistry and Biology of Hyaluronan, 1st ed.; Garg, G.G., Hales, C.A., Eds.; Elsevier Ltd.: Amsterdam, The Netherlands, 2004; Volume 1, pp. 415–455. [Google Scholar]
- Falcone, S.J.; Palmeri, D.M.; Berg, R.A. Rheological and cohesive properties of hyaluronic acid. J. Biomed. Mater. Res. A 2006, 76A, 721–728. [Google Scholar] [CrossRef]
- Bhuanantanondh, P.; Grecov, D.; Kwok, E. Rheological Study of viscosupplements and synovial fluid in patients with osteoarthritis. J. Med. Biol. Eng. 2010, 32, 12–16. [Google Scholar] [CrossRef]
- Altman, R.D. Status of hyaluronan supplementation therapy in osteoarthritis. Curr. Rheumatol. Rep. 2003, 5, 7–14. [Google Scholar] [CrossRef]
- Mathieu, P.; Conrozier, T.; Vignon, E.; Rozand, Y.; Rinaudo, M. Rheologic behavior of osteoarthritic synovial fluid after addition of hyaluronic acid: A pilot study. Clin. Orthop. Relat. Res. 2009, 467, 3002–3009. [Google Scholar] [CrossRef]
- Bhuanantanondh, P.; Grecov, D.; Kwok, E.; Guy, P. Rheology of osteoarthritic synovial fluid mixed with viscosupplements: A pilot study. Biomed. Eng. Lett. 2011, 1, 213–219. [Google Scholar] [CrossRef]
- Dowson, D. Modes of lubrication in human joints. Proc. Inst. Mech. Eng. 1996, 181, 45–54. [Google Scholar]
- McCutchen, C.W. The frictional properties of animal joints. Wear 1962, 5, 1–17. [Google Scholar] [CrossRef]
- Dowson, D.; Jin, Z.-M. Micro-elastohydrodynamic lubrication of synovial joints. Eng. Med. 1986, 15, 63–65. [Google Scholar] [CrossRef]
- Murakami, T. The lubrication in natural synovial joints and joint prostheses. JSME Int. J. Ser. III Vib. Control Eng. Eng. Ind. 1990, 33, 465–474. [Google Scholar] [CrossRef][Green Version]
- Murakami, T.; Higaki, H.; Sawae, Y.; Ohtsuki, N.; Moriyama, S.; Nakanishi, Y. Adaptive multimode lubrication in natural synovial joints and artificial joints. Proc. Inst. Mech. Eng. H 2006, 212, 23–35. [Google Scholar] [CrossRef]
- Ateshian, G.A. A theoretical formulation for boundary friction in articular cartilage. J. Biomech. Eng. 1997, 119, 81–86. [Google Scholar] [CrossRef]
- Ikeuchi, K. Origin and future of hydration lubrication. Proc. Inst. Mech. Eng. J. 2007, 221, 301–305. [Google Scholar] [CrossRef]
- Bell, C.J.; Ingham, E.; Fisher, J. Influence of hyaluronic acid on the time-dependent friction response of articular cartilage under different conditions. Proc. Inst. Mech. Eng. H 2006, 220, 23–31. [Google Scholar] [CrossRef]
- Murakami, T.; Yarimitsu, S.; Nakashima, K.; Sawae, Y.; Sakai, N. Influence of synovia constituents on tribological behaviors of articular cartilage. Friction 2013, 1, 150–162. [Google Scholar] [CrossRef]
- Seror, J.; Zhu, L.; Goldberg, R.; Day, A.J.; Klein, J. Supramolecular synergy in the boundary lubrication of synovial joints. Nat. Commun. 2015, 6, 6497. [Google Scholar] [CrossRef]
- Yarimitsu, S.; Sasaki, S.; Murakami, T.; Suzuki, A. Evaluation of lubrication properties of hydrogel artificial cartilage materials for joint prosthesis. Biosurf. Biotribol. 2016, 2, 40–47. [Google Scholar] [CrossRef]
- Murakami, T.; Nakashima, K.; Yarimitsu, S.; Sawae, Y.; Sakai, N. Effectiveness of adsorbed film and gel layer in hydration lubrication as adaptive multimode lubrication mechanism for articular cartilage. Proc. Inst. Mech. Eng. J. 2011, 225, 1174–1185. [Google Scholar] [CrossRef]
- Szarko, M.; Muldrew, K.; Bertram, J.E.A. Freeze-thaw treatment effects on the dynamic mechanical properties of articular cartilage. BMC Musculoskelet. Disord. 2010, 11, 281. [Google Scholar] [CrossRef]
- Fam, H.; Bryant, J.T.; Kontopoulou, M. Rheological properties of synovial fluids. Biorheology 2007, 44, 59–74. [Google Scholar]
- Mazzucco, D.; McKinley, G.; Scott, R.D.; Spector, M. Rheology of joint fluid in total knee arthroplasty patients. J. Orthop. Res. 2002, 20, 1157–1163. [Google Scholar] [CrossRef]
- Lapasin, R. Rheological Studies Dedicated to the Development of a Novel Injectable Polymeric Blend for Viscosupplementation Treatment. Chem. Biochem. Eng. Q. 2016, 29, 511–518. [Google Scholar] [CrossRef]
- Bonnevie, E.D.; Galesso, D.; Secchieri, C.; Bonassar, L.J.; Awad, H.A. Frictional characterization of injectable hyaluronic acids is more predictive of clinical outcomes than traditional rheological or viscoelastic characterization. PLoS ONE 2019, 14, e0216702. [Google Scholar] [CrossRef]
- Rainer, F.; Ribitsch, V. Viscoelastic properties of normal human synovia and their relation to biomechanics. Zeitschrift fur Rheumatologie 1985, 44, 114–119. [Google Scholar]
- Nicholls, M.; Manjoo, A.; Shaw, P.; Niazi, F.; Rosen, J. A Comparison Between Rheological Properties of Intra-articular Hyaluronic Acid Preparations and Reported Human Synovial Fluid. Adv. Ther. 2018, 35, 523–530. [Google Scholar] [CrossRef]
- Borzacchiello, A.; Mayol, L.; Schiavinato, A.; Ambrosio, L. Effect of hyaluronic acid amide derivative on equine synovial fluid viscoelasticity. J. Biomed. Mater. Res. A 2009, 92A, 1162–1170. [Google Scholar] [CrossRef]
- Balazs, E.A. The physical properties of synovial fluid and the special role of hyaluronic acid. In Disorders of the Knee, 1st ed.; Helfet, A.J., Ed.; JB Lippincott & Co.: Philadelphia, PA, USA, 1974; Volume 2, pp. 63–75. [Google Scholar]
- Finelli, I.; Chiessi, E.; Galesso, D.; Renier, D.; Paradossi, G. A new viscosupplement based on partially hydrophobic hyaluronic acid: A comparative study. Biorheology 2011, 48, 263–275. [Google Scholar] [CrossRef]
- Krishnan, R.; Kopacz, M.; Ateshian, G.A. Experimental verification of the role of interstitial fluid pressurization in cartilage lubrication. J. Orthop. Res. 2004, 22, 565–570. [Google Scholar] [CrossRef]
- Bonnevie, E.D.; Galesso, D.; Secchieri, C.; Cohen, I.; Bonassar, L.J.; Awad, H.A. Elastoviscous transitions of articular cartilage reveal a mechanism of synergy between lubricin and hyaluronic acid. PLoS ONE 2015, 10, e0143415. [Google Scholar] [CrossRef]
- Kwiecinski, J.J.; Dorosz, S.G.; Ludwig, T.E.; Abubacker, S.; Cowman, M.K.; Schmidt, T.A. The effect of molecular weight on hyaluronan’s cartilage boundary lubricating ability—Alone and in combination with proteoglycan 4. Osteoarthr. Cartil. 2011, 19, 1356–1362. [Google Scholar] [CrossRef]
- Appleyard, R.C.; Burkhardt, D.; Ghosh, P.; Read, R.; Cake, M.; Swain, M.V.; Murrell, G.A.C. Topographical analysis of the structural, biochemical and dynamic biomechanical properties of cartilage in an ovine model of osteoarthritis. Osteoarthr. Cartil. 2003, 11, 65–77. [Google Scholar] [CrossRef]
- Kiviranta, P.; Lammentausta, E.; Töyräs, J.; Kiviranta, I.; Jurvelin, J.S. Indentation diagnostics of cartilage degeneration. Osteoarthr. Cartil. 2008, 16, 796–804. [Google Scholar] [CrossRef]
- Richard, F.; Villars, M.; Thibaud, S. Viscoelastic modeling and quantitative experimental characterization of normal and osteoarthritic human articular cartilage using indentation. J. Mech. Behav. Biomed. Mater. 2013, 24, 41–52. [Google Scholar] [CrossRef]
- DuRaine, G.; Neu, C.P.; Chan, S.M.T.; Komvopoulos, K.; June, R.K.; Reddi, A.H. Regulation of the friction coefficient of articular cartilage by TGF-β1 and IL-1β. J. Orthop. Res. 2009, 27, 249–256. [Google Scholar] [CrossRef]
- Chan, S.M.T.; Neu, C.P.; Komvopoulos, K.; Reddi, A.H. The role of lubricant entrapment at biological interfaces: Reduction of friction and adhesion in articular cartilage. J. Biomech. 2011, 44, 2015–2020. [Google Scholar] [CrossRef] [PubMed]
- Nečas, D.; Vrbka, M.; Urban, F.; Křupka, I.; Hartl, M. The effect of lubricant constituents on lubrication mechanisms in hip joint replacements. J. Mech. Behav. Biomed. Mater. 2016, 55, 295–307. [Google Scholar] [CrossRef]
- Myant, C.W.; Cann, P. The effect of transient conditions on synovial fluid protein aggregation lubrication. J. Mech. Behav. Biomed. Mater. 2014, 34, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Rainforth, W.M. The effect of lubrication on the friction and wear of Biolox®delta. Acta Biomater. 2012, 8, 2348–2359. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, T.A.; Gastelum, N.S.; Nguyen, Q.T.; Schumacher, B.L.; Sah, R.L. Boundary lubrication of articular cartilage: Role of synovial fluid constituents. Arthritis Rheum. 2007, 56, 882–891. [Google Scholar] [CrossRef] [PubMed]
- Seror, J.; Sorkin, R.; Klein, J. Boundary lubrication by macromolecular layers and its relevance to synovial joints. Polym. Adv. Technol. 2014, 25, 468–477. [Google Scholar] [CrossRef]
- Klein, J. Hydration lubrication. Friction 2013, 1, 1–23. [Google Scholar] [CrossRef]
- Jahn, S.; Klein, J. Lubrication of articular cartilage. Physics Today 2018, 71, 48–54. [Google Scholar] [CrossRef]
- Maheu, E.; Rannou, F.; Reginster, J.-Y. Efficacy and safety of hyaluronic acid in the management of osteoarthritis: Evidence from real-life setting trials and surveys. Semin. Arthritis Rheum. 2016, 45, S28–S33. [Google Scholar] [CrossRef]
- Tıkız, C.; Ünlü, Z.; Şener, A.; Efe, M.; Tüzün, Ç. Comparison of the efficacy of lower and higher molecular weight viscosupplementation in the treatment of hip osteoarthritis. Clin. Rheum. 2005, 24, 244–250. [Google Scholar] [CrossRef]
- Bannuru, R.R.; Vaysbrot, E.E.; Sullivan, M.C.; McAlindon, T.E. Relative efficacy of hyaluronic acid in comparison with NSAIDs for knee osteoarthritis: A systematic review and meta-analysis. Semin. Arthritis Rheum. 2014, 43, 593–599. [Google Scholar] [CrossRef]
- Jevsevar, D.; Donnelly, P.; Brown, G.A.; Cummins, D.S. Viscosupplementation for osteoarthritis of the knee. J. Bone Jt. Surg. Am. 2015, 97, 2047–2060. [Google Scholar] [CrossRef]
- Rutjes, A.W.S.; Jüni, P.; da Costa, B.R.; Trelle, S.; Nüesch, E.; Reichenbach, S. Viscosupplementation for osteoarthritis of the knee. Ann. Intern. Med. 2012, 157, 180–191. [Google Scholar] [CrossRef]
- Arrich, J. Intra-articular hyaluronic acid for the treatment of osteoarthritis of the knee: Systematic review and meta-analysis. Can. Med. Assoc. J. 2005, 172, 1039–1043. [Google Scholar] [CrossRef]
- Bruyère, O.; Cooper, C.; Pelletier, J.-P.; Branco, J.; Luisa Brandi, M.; Guillemin, F.; Hochberg, M.C.; Kanis, J.A.; Kvien, T.K.; Martel-Pelletier, J.; et al. An algorithm recommendation for the management of knee osteoarthritis in Europe and internationally: A report from a task force of the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO). Semin. Arthritis Rheum. 2014, 44, 253–263. [Google Scholar] [CrossRef]
- McAlindon, T.E.; Bannuru, R.R.; Sullivan, M.C.; Arden, N.K.; Berenbaum, F.; Bierma-Zeinstra, S.M.; Hawker, G.A.; Henrotin, Y.; Hunter, D.J.; Kawaguchi, H.; et al. OARSI guidelines for the non-surgical management of knee osteoarthritis. Osteoarthr. Cartil. 2014, 22, 363–388. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.-S.; Xiong, D.-S.; Ma, R.-Y. A study on the friction properties of poly (vinyl alcohol) hydrogel as articular cartilage against titanium alloy. Wear 2007, 262, 1021–1025. [Google Scholar] [CrossRef]
- Katta, J.K.; Marcolongo, M.; Lowman, A.; Mansmann, K.A. Friction and wear behavior of poly (vinyl alcohol)/poly (vinyl pyrrolidone) hydrogels for articular cartilage replacement. J. Biomed. Mater. Res. A 2007, 83A, 471–479. [Google Scholar] [CrossRef]
- Murakami, T.; Sakai, N.; Yamaguchi, T.; Yarimitsu, S.; Nakashima, K.; Sawae, Y.; Suzuki, A. Evaluation of a superior lubrication mechanism with biphasic hydrogels for artificial cartilage. Tribol. Int. 2015, 89, 19–26. [Google Scholar] [CrossRef]
- Murakami, T.; Yarimitsu, S.; Nakashima, K.; Yamaguchi, T.; Sawae, Y.; Sakai, N.; Suzuki, A. Superior lubricity in articular cartilage and artificial hydrogel cartilage. Proc. Inst. Mech. Eng. J. 2014, 228, 1099–1111. [Google Scholar] [CrossRef]
- Murakami, T.; Yarimitsu, S.; Sakai, N.; Nakashima, K.; Yamaguchi, T.; Sawae, Y.; Suzuki, A. Superior lubrication mechanism in poly (vinyl alcohol) hybrid gel as artificial cartilage. Proc. Inst. Mech. Eng. J. 2017, 231, 1160–1170. [Google Scholar] [CrossRef]
- Nečas, D.; Vrbka, M.; Galandáková, A.; Křupka, I.; Hartl, M. On the observation of lubrication mechanisms within hip joint replacements. Part I: Hard-on-soft bearing pairs. J. Mech. Behav. Biomed. Mater. 2019, 89, 237–248. [Google Scholar]
- Čípek, P.; Rebenda, D.; Nečas, D.; Vrbka, M.; Křupka, I.; Hartl, M. Visualization of lubrication film in model of synovial joint. Tribol. Ind. 2019, 41, 387–393. [Google Scholar] [CrossRef]
| MW (kDa) | Zero Shear Viscosity (Pa·s) | 0.5 Hz | 2.5 Hz | Crossover Frequency (Hz) | |||
|---|---|---|---|---|---|---|---|
| G′ (Pa) | G″ (Pa) | G′ (Pa) | G″ (Pa) | ||||
| 2010 | 107 ± 1.7 | 113.9 | 101 ± 3.5 | 92.3 ± 4 | 220 ± 9.5 | 125 ± 6.3 | 0.4 |
| 1060 | 11.6 ± 0.4 | 17.8 | 13.5 ± 1.5 | 29 ± 2.5 | 55.8 ± 5.6 | 67.5 ± 5.3 | - |
| 640 | 1.67 ± 0.05 | 4.1 | 0.4 ± 0.04 | 5.8 ± 0.03 | 5.4 ± 0.3 | 22.2 ± 0.2 | - |
| 77 | 0.013 ± 3 × 10‒3 | 1.3 | - | - | - | - | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rebenda, D.; Vrbka, M.; Čípek, P.; Toropitsyn, E.; Nečas, D.; Pravda, M.; Hartl, M. On the Dependence of Rheology of Hyaluronic Acid Solutions and Frictional Behavior of Articular Cartilage. Materials 2020, 13, 2659. https://doi.org/10.3390/ma13112659
Rebenda D, Vrbka M, Čípek P, Toropitsyn E, Nečas D, Pravda M, Hartl M. On the Dependence of Rheology of Hyaluronic Acid Solutions and Frictional Behavior of Articular Cartilage. Materials. 2020; 13(11):2659. https://doi.org/10.3390/ma13112659
Chicago/Turabian StyleRebenda, David, Martin Vrbka, Pavel Čípek, Evgeniy Toropitsyn, David Nečas, Martin Pravda, and Martin Hartl. 2020. "On the Dependence of Rheology of Hyaluronic Acid Solutions and Frictional Behavior of Articular Cartilage" Materials 13, no. 11: 2659. https://doi.org/10.3390/ma13112659
APA StyleRebenda, D., Vrbka, M., Čípek, P., Toropitsyn, E., Nečas, D., Pravda, M., & Hartl, M. (2020). On the Dependence of Rheology of Hyaluronic Acid Solutions and Frictional Behavior of Articular Cartilage. Materials, 13(11), 2659. https://doi.org/10.3390/ma13112659





