Ultra-Thin ReS2 Nanosheets Grown on Carbon Black for Advanced Lithium-Ion Battery Anodes
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of ReS2@CB and ReS2 Powder
2.2. Structural Analysis of ReS2@CB, ReS2, and CB
2.3. Lithium Ion Battery Performance of ReS2@CB, ReS2 Powder, and CB Anode Structures
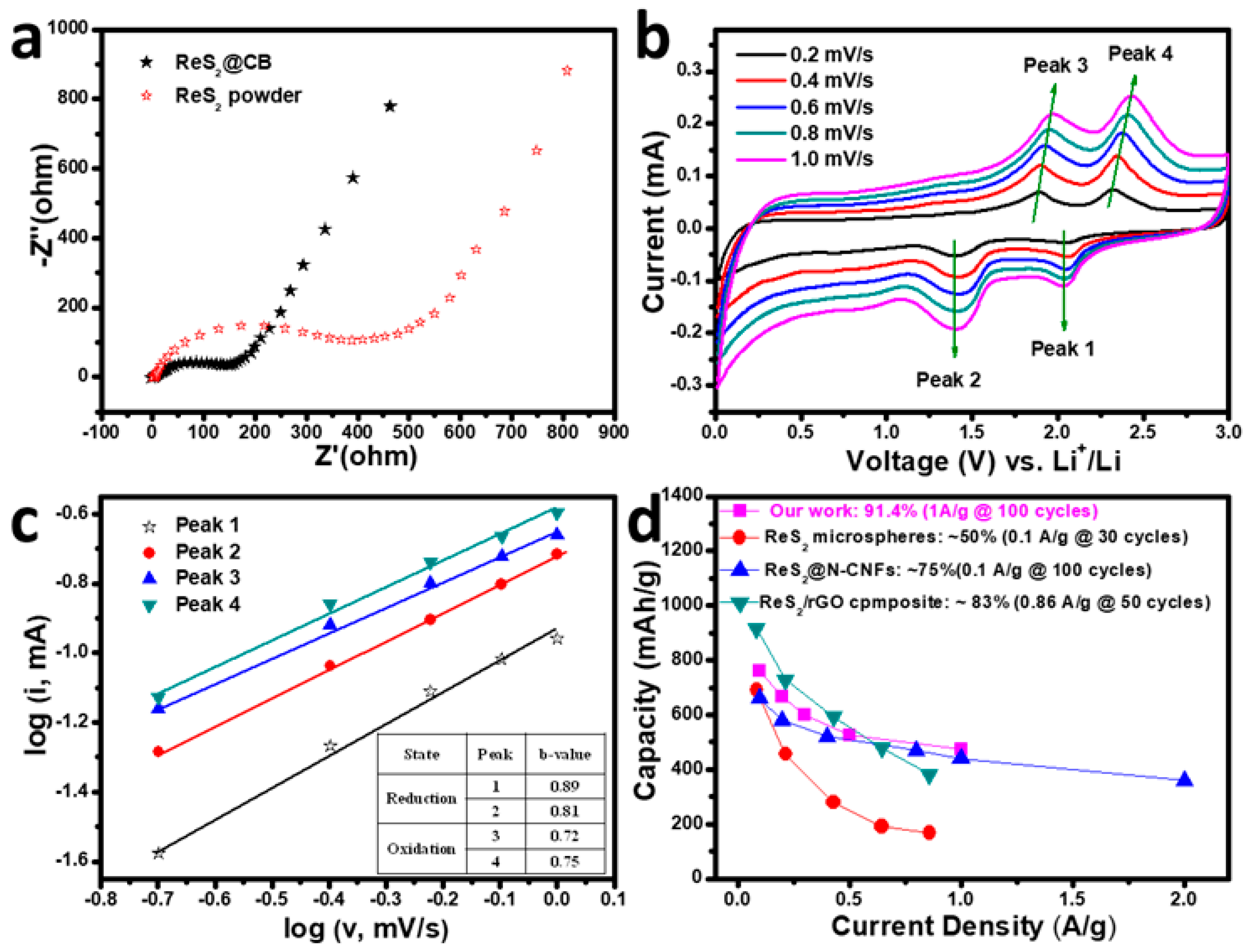
3. Results and Discussion
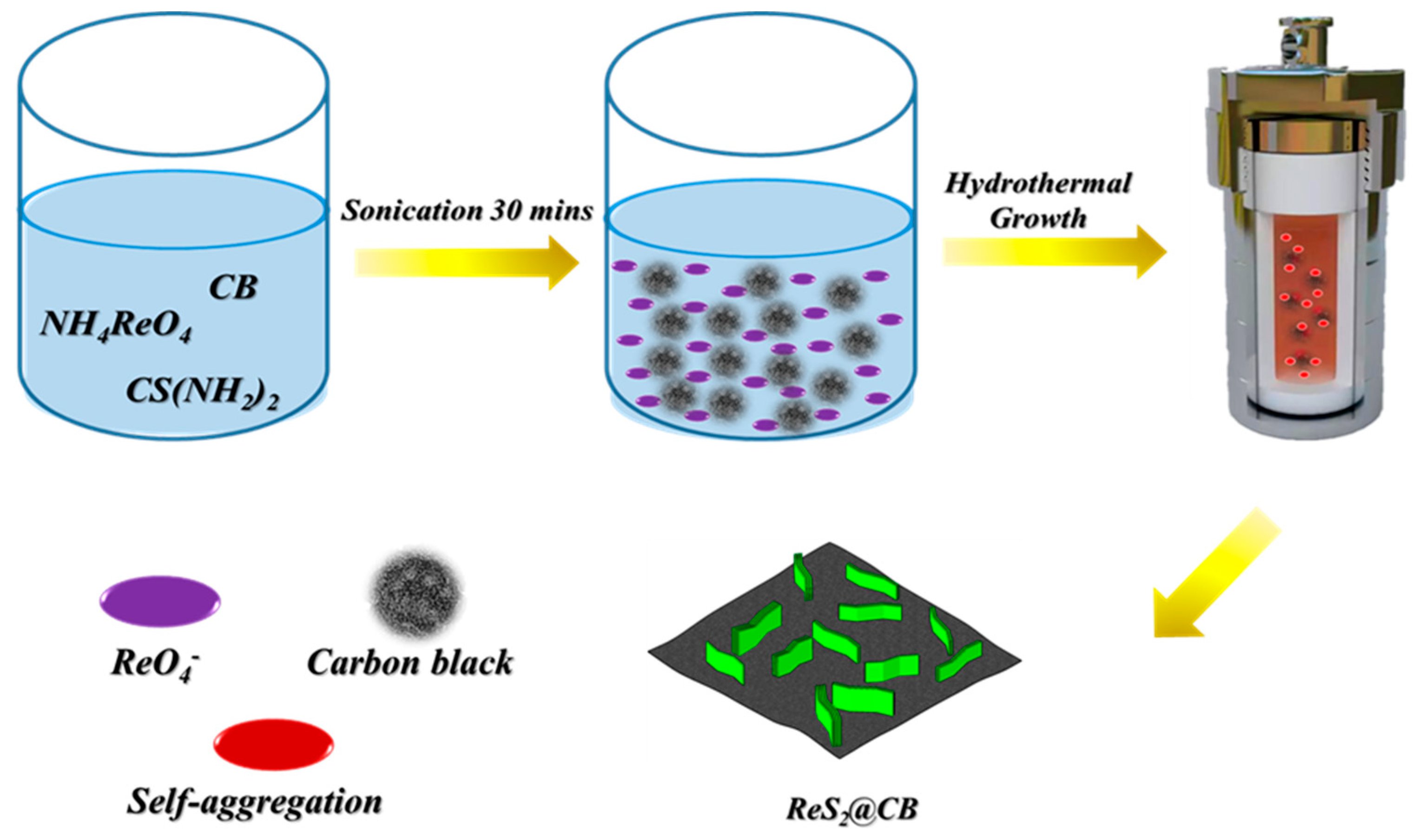
3.1. Schematic Synthesis Process of ReS2@CB and Morphological Properties of ReS2@CB, ReS2 Powder, and CB
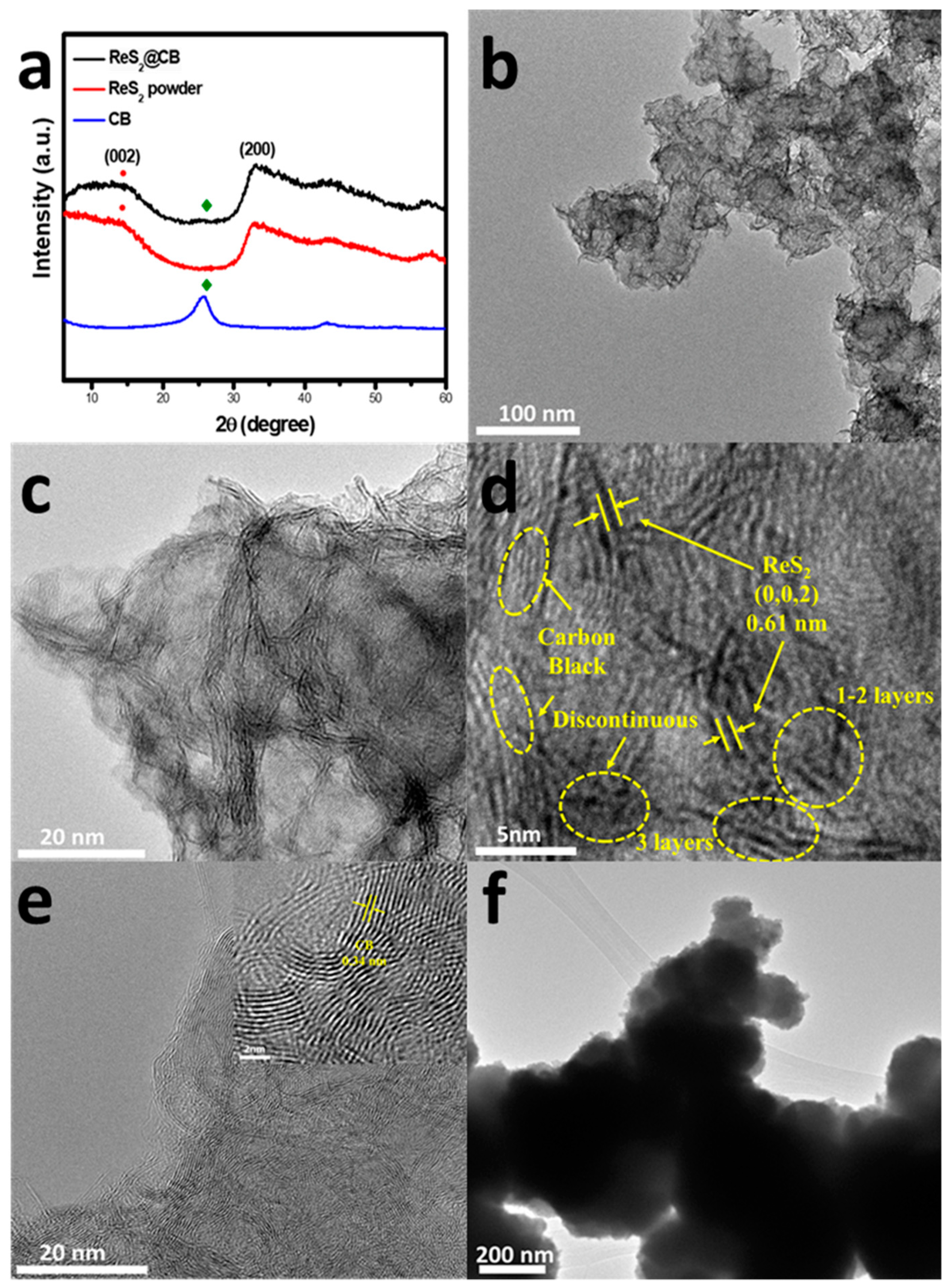
3.2. Structural Property of ReS2@CB, ReS2 Powder, and CB
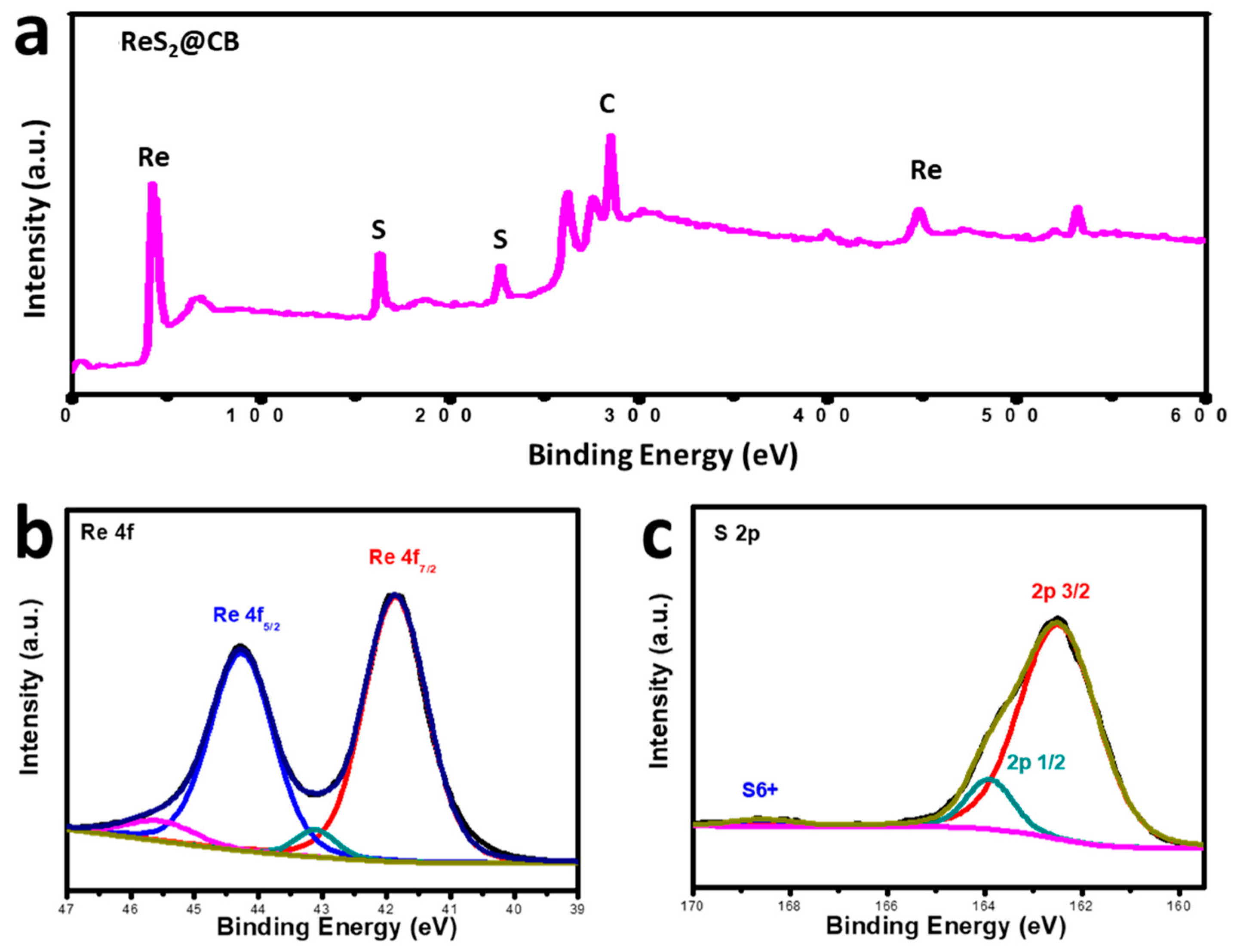
3.3. X-ray Photoelectron Spectroscopy (XPS) Analysis of ReS2@CB
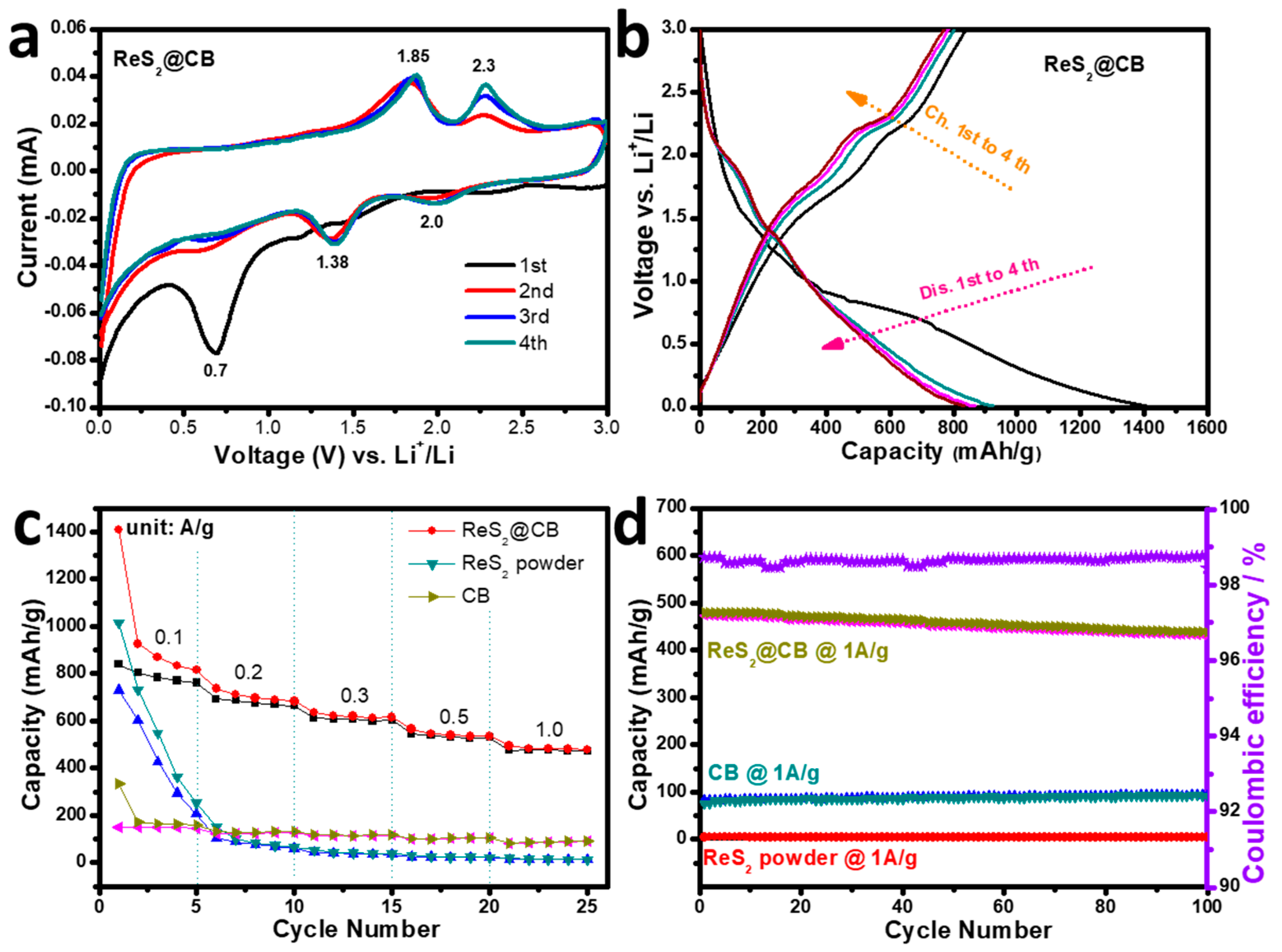
3.4. Electrochemical Performance of ReS2@CB, ReS2 Powder, and CB
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nitta, N.; Wu, F.; Lee, J.T.; Yushin, G. Li-ion battery materials: Present and future. Mater. Today 2015, 18, 252–264. [Google Scholar] [CrossRef]
- Kim, I.; Hyun, S.; Nam, S.; Lee, H.-J.; Kang, C. Fabrication of three-dimensional crystalline silicon-on-carbon nanotube nanocomposite anode by sputtering and laser annealing for high-performance lithium-ion battery. Jpn. J. Appl. Phys. 2018, 57, 05GB05. [Google Scholar] [CrossRef]
- Kang, C.; Lee, H.-J. Morphological control of three-dimensional carbon nanotube anode for high-capacity lithium-ion battery. Jpn. J. Appl. Phys. 2018, 57, 05GC05. [Google Scholar] [CrossRef]
- Oh, M.; Kim, I.; Lee, H.-J.; Hyun, S.; Kang, C. The role of thermal annealing on the microstructures of (Ti, Fe)-alloyed Si thin-film anodes for high-performance Li-ion batteries. RSC Adv. 2018, 8, 9168–9174. [Google Scholar] [CrossRef]
- Kang, C.; Cha, E.; Patel, M.D.; Wu, H.F.; Choi, W. Three-Dimensional Carbon Nanostructures for Advanced Lithium-Ion Batteries. C 2016, 2, 23. [Google Scholar] [CrossRef]
- Stephenson, T.; Li, Z.; Olsen, B.; Mitlin, D. Lithium ion battery applications of molybdenum disulfide (MoS2) nanocomposites. Energy Environ. Sci. 2013, 7, 209–231. [Google Scholar] [CrossRef]
- Zhou, L.; Yan, S.; Pan, L.; Wang, X.; Wang, Y.; Shi, Y. A scalable sulfuration of WS2 to improve cyclability and capability of lithium-ion batteries. Nano Res. 2016, 9, 857–865. [Google Scholar] [CrossRef]
- Wu, H.B.; Chen, J.S.; Hng, H.H.; Lou, X.W. (David). Nanostructured metal oxide-based materials as advanced anodes for lithium-ion batteries. Nanoscale 2012, 4, 2526–2542. [Google Scholar] [CrossRef]
- Jeżowski, P.; Crosnier, O.; Deunf, E.; Poizot, P.; Béguin, F.; Brousse, T. Safe and recyclable lithium-ion capacitors using sacrificial organic lithium salt. Nat. Mater. 2018, 17, 167–173. [Google Scholar] [CrossRef]
- Wang, R.; Wang, S.; Jin, D.; Zhang, Y.; Cai, Y.; Ma, J.; Zhang, L. Engineering layer structure of MoS2-graphene composites with robust and fast lithium storage for high-performance Li-ion capacitors. Energy Storage Mater. 2017, 9, 195–205. [Google Scholar] [CrossRef]
- Zhang, F.; Tang, Y.; Liu, H.; Ji, H.; Jiang, C.; Zhang, J.; Zhang, X.; Lee, C.-S. Uniform Incorporation of Flocculent Molybdenum Disulfide Nanostructure into Three-Dimensional Porous Graphene as an Anode for High-Performance Lithium Ion Batteries and Hybrid Supercapacitors. ACS Appl. Mater. Interfaces 2016, 8, 4691–4699. [Google Scholar] [CrossRef]
- Zhang, Q.; Tan, S.; Mendes, R.G.; Sun, Z.; Chen, Y.; Kong, X.; Xue, Y.; Rümmeli, M.H.; Wu, X.; Chen, S.; et al. Extremely Weak van der Waals Coupling in Vertical ReS2 Nanowalls for High-Current-Density Lithium-Ion Batteries. Adv. Mater. 2016, 28, 2616–2623. [Google Scholar] [CrossRef]
- Mao, M.; Cui, C.; Wu, M.; Zhang, M.; Gao, T.; Fan, X.; Chen, J.; Wang, T.; Ma, J.; Wang, C. Flexible ReS2 nanosheets/N-doped carbon nanofibers-based paper as a universal anode for alkali (Li, Na, K) ion battery. Nano Energy 2018, 45, 346–352. [Google Scholar] [CrossRef]
- Qi, F.; He, J.; Chen, Y.; Zheng, B.; Li, Q.; Wang, X.; Yu, B.; Lin, J.; Zhou, J.; Li, P.; et al. Few-layered ReS2 nanosheets grown on carbon nanotubes: A highly efficient anode for high-performance lithium-ion batteries. Chem. Eng. J. 2017, 315, 10–17. [Google Scholar] [CrossRef]
- Qi, F.; Chen, Y.; Zheng, B.; He, J.; Li, Q.; Wang, X.; Lin, J.; Zhou, J.; Yu, B.; Li, P.; et al. Hierarchical architecture of ReS2/rGO composites with enhanced electrochemical properties for lithium-ion batteries. Appl. Surf. Sci. 2017, 413, 123–128. [Google Scholar] [CrossRef]
- Qi, F.; Chen, Y.; Zheng, B.; He, J.; Li, Q.; Wang, X.; Yu, B.; Lin, J.; Zhou, J.; Li, P.; et al. 3D chrysanthemum-like ReS2 microspheres composed of curly few-layered nanosheets with enhanced electrochemical properties for lithium-ion batteries. J. Mater. Sci. 2017, 52, 3622–3629. [Google Scholar] [CrossRef]
- Liu, S.; Liu, Y.; Lei, W.; Zhou, X.; Xu, K.; Qiao, Q.; Zhang, W.-H. Few-layered ReS2 nanosheets vertically aligned on reduced graphene oxide for superior lithium and sodium storage. J. Mater. Chem. A 2018, 6, 20267–20276. [Google Scholar] [CrossRef]
- Teng, Y.; Zhao, H.; Zhang, Z.; Li, Z.; Xia, Q.; Zhang, Y.; Zhao, L.; Du, X.; Du, Z.; Lv, P.; et al. MoS2 Nanosheets Vertically Grown on Graphene Sheets for Lithium-Ion Battery Anodes. ACS Nano 2016, 10, 8526–8535. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, B.; Seo, D.H.; Han, Z.J.; Wong, J.I.; Ostrikov, K. (Ken); Zhang, H.; Yang, H.Y. MoS2-coated vertical graphene nanosheet for high-performance rechargeable lithium-ion batteries and hydrogen production. NPG Asia Mater. 2016, 8, e268. [Google Scholar] [CrossRef]
- Badam, R.; Joshi, P.; Vedarajan, R.; Natarajan, R.; Matsumi, N. Few-Layered MoS2/Acetylene Black Composite as an Efficient Anode Material for Lithium-Ion Batteries. Nanoscale Res. Lett. 2017, 12, 555. [Google Scholar] [CrossRef] [PubMed]
- Xue, N.; Diao, P. Composite of Few-Layered MoS2 Grown on Carbon Black: Tuning the Ratio of Terminal to Total Sulfur in MoS2 for Hydrogen Evolution Reaction. J. Phys. Chem. C 2017, 121, 14413–14425. [Google Scholar] [CrossRef]
- Zhang, L.; Fan, W.; Liu, T. A flexible free-standing defect-rich MoS2/graphene/carbon nanotube hybrid paper as a binder-free anode for high-performance lithium ion batteries. RSC Adv. 2015, 5, 43130–43140. [Google Scholar] [CrossRef]
- Yan, C.; Xue, X.; Zhang, W.; Li, X.; Liu, J.; Yang, S.; Hu, Y.; Chen, R.; Yan, Y.; Zhu, G.; et al. Well-designed Te/SnS2/Ag artificial nanoleaves for enabling and enhancing visible-light driven overall splitting of pure water. Nano Energy 2017, 39, 539–545. [Google Scholar] [CrossRef]
- Qian, Y.; Yang, M.; Zhang, F.; Du, J.; Li, K.; Lin, X.; Zhu, X.; Lu, Y.; Wang, W.; Kang, D.J. A stable and highly efficient visible-light-driven hydrogen evolution porous CdS/WO3/TiO2 photocatalysts. Mater. Charact. 2018, 142, 43–49. [Google Scholar] [CrossRef]
- Smith, A.J.; Chang, Y.-H.; Raidongia, K.; Chen, T.-Y.; Li, L.-J.; Huang, J. Molybdenum Sulfide Supported on Crumpled Graphene Balls for Electrocatalytic Hydrogen Production. Adv. Energy Mater. 2014, 4, 1400398. [Google Scholar] [CrossRef]
- Ji, C.; Yan, C.; Wang, Y.; Xiong, S.; Zhou, F.; Li, Y.; Sun, R.; Wong, C.-P. Thermal conductivity enhancement of CNT/MoS2/graphene−epoxy nanocomposites based on structural synergistic effects and interpenetrating network. Compos. Part B Eng. 2019, 163, 363–370. [Google Scholar] [CrossRef]
- Li, K.; Qian, Y.; Zhang, H.; Zhang, L.; Chai, Q.; Wang, Q.; Du, J.; Han, Y.; Wang, W.; Kang, D.J. Highly efficient oxygen evolution electrocatalysts based on nanosheet-shaped CuS in situ grown on carbon cloth. Ceram. Int. 2019, 45, 10664–10671. [Google Scholar] [CrossRef]
- George, C.; Morris, A.J.; Modarres, M.H.; De Volder, M. Structural Evolution of Electrochemically Lithiated MoS2 Nanosheets and the Role of Carbon Additive in Li-Ion Batteries. Chem. Mater. 2016, 28, 7304–7310. [Google Scholar] [CrossRef]
- Lin, D.; Liu, Y.; Cui, Y. Reviving the lithium metal anode for high-energy batteries. Nat. Nanotechnol. 2017, 12, 194–206. [Google Scholar] [CrossRef]
- Xiao, J.; Choi, D.; Cosimbescu, L.; Koech, P.; Liu, J.; Lemmon, J.P. Exfoliated MoS2 Nanocomposite as an Anode Material for Lithium Ion Batteries. Chem. Mater. 2010, 22, 4522–4524. [Google Scholar] [CrossRef]
- Wang, Z.; Wei, G.; Ozawa, K.; Cai, Y.; Cheng, Z.; Kimura, H. Nanoporous MoS2/C Composites for High Performance Lithium Ion Battery Anode Material. Electrochim. Acta 2017, 239, 74–83. [Google Scholar] [CrossRef]
- Li, J.; Hou, Y.; Gao, X.; Guan, D.; Xie, Y.; Chen, J.; Yuan, C. A three-dimensionally interconnected carbon nanotube/layered MoS2 nanohybrid network for lithium ion battery anode with superior rate capacity and long-cycle-life. Nano Energy 2015, 16, 10–18. [Google Scholar] [CrossRef]
- Xiang, Y.; Yang, Z.; Wang, S.; Hossain, M.S.A.; Yu, J.; Kumar, N.A.; Yamauchi, Y. Pseudocapacitive behavior of the Fe2O3 anode and its contribution to high reversible capacity in lithium ion batteries. Nanoscale 2018, 10, 18010–18018. [Google Scholar] [CrossRef]
- Wang, J.; Polleux, J.; Lim, J.; Dunn, B. Pseudocapacitive Contributions to Electrochemical Energy Storage in TiO2 (Anatase) Nanoparticles. J. Phys. Chem. C 2007, 111, 14925–14931. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yan, Y.; Song, K.-Y.; Cho, M.; Lee, T.H.; Kang, C.; Lee, H.-J. Ultra-Thin ReS2 Nanosheets Grown on Carbon Black for Advanced Lithium-Ion Battery Anodes. Materials 2019, 12, 1563. https://doi.org/10.3390/ma12091563
Yan Y, Song K-Y, Cho M, Lee TH, Kang C, Lee H-J. Ultra-Thin ReS2 Nanosheets Grown on Carbon Black for Advanced Lithium-Ion Battery Anodes. Materials. 2019; 12(9):1563. https://doi.org/10.3390/ma12091563
Chicago/Turabian StyleYan, Yaping, Kyeong-Youn Song, Minwoo Cho, Tae Hoon Lee, Chiwon Kang, and Hoo-Jeong Lee. 2019. "Ultra-Thin ReS2 Nanosheets Grown on Carbon Black for Advanced Lithium-Ion Battery Anodes" Materials 12, no. 9: 1563. https://doi.org/10.3390/ma12091563
APA StyleYan, Y., Song, K.-Y., Cho, M., Lee, T. H., Kang, C., & Lee, H.-J. (2019). Ultra-Thin ReS2 Nanosheets Grown on Carbon Black for Advanced Lithium-Ion Battery Anodes. Materials, 12(9), 1563. https://doi.org/10.3390/ma12091563



