Electrochemical Oxidation/Disinfection of Urine Wastewaters with Different Anode Materials
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Analytical Methods
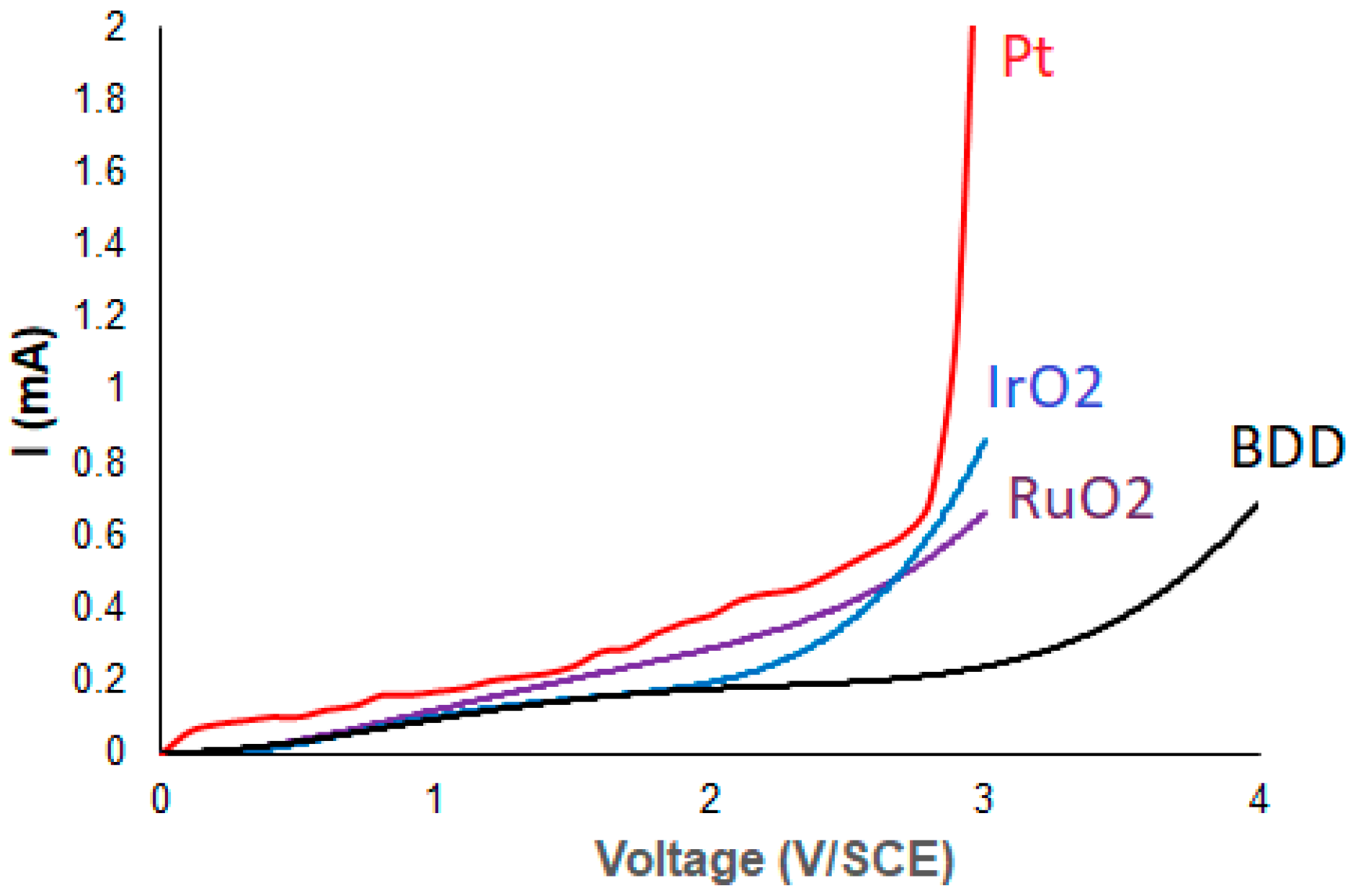
2.3. Voltammetry Experiments
2.4. Electrochemical Set Up
2.5. Experimental Procedures
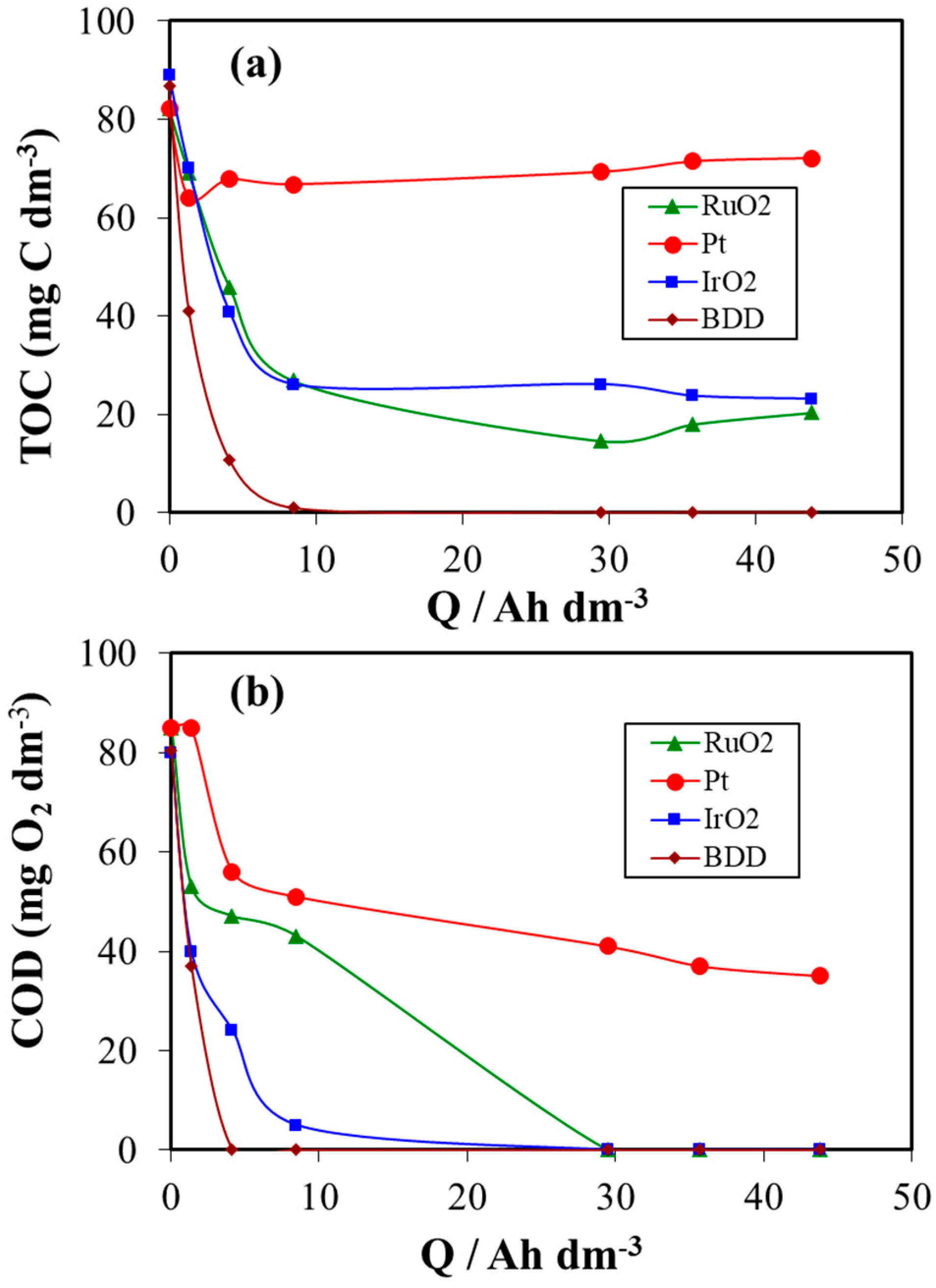
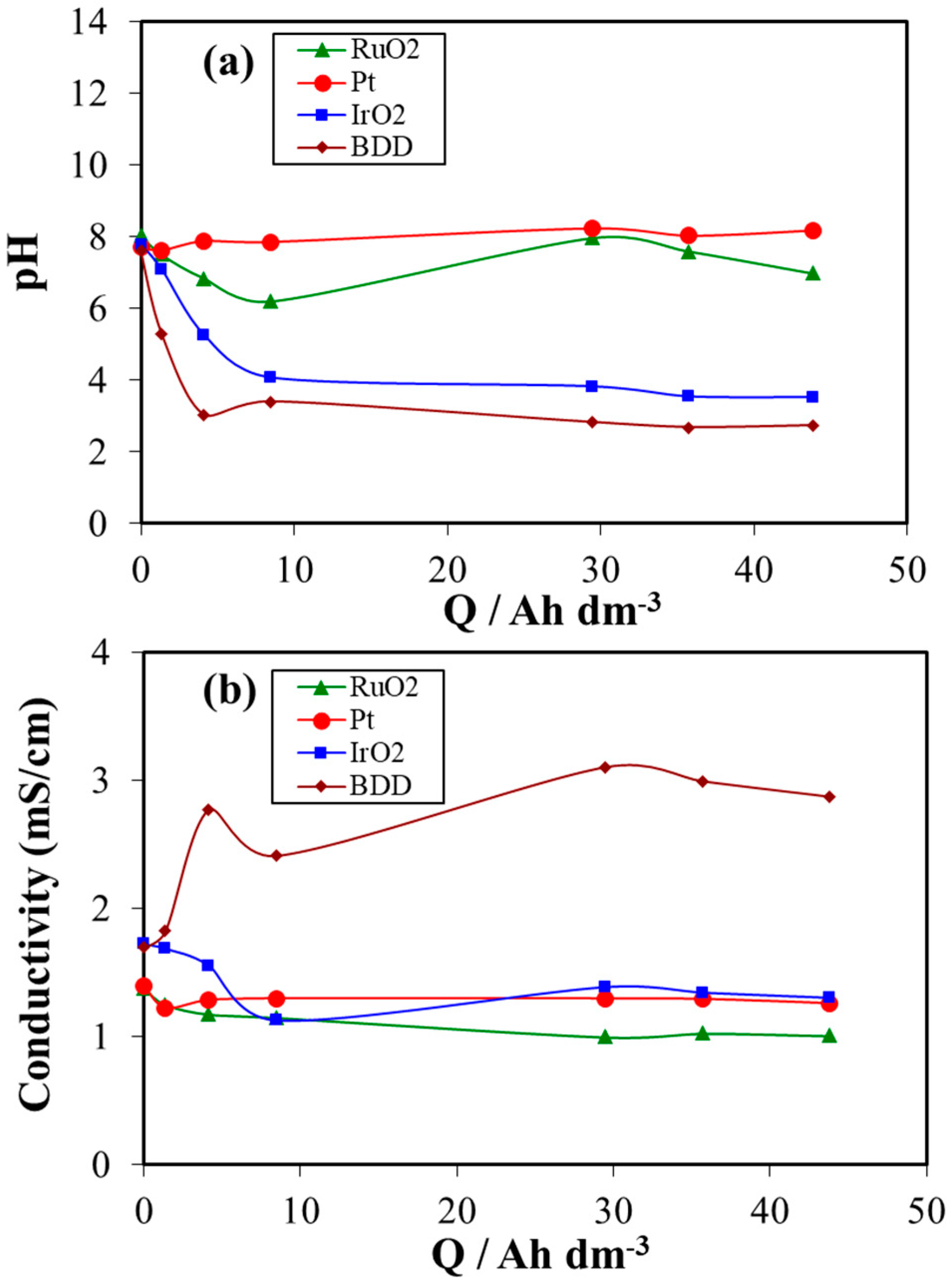
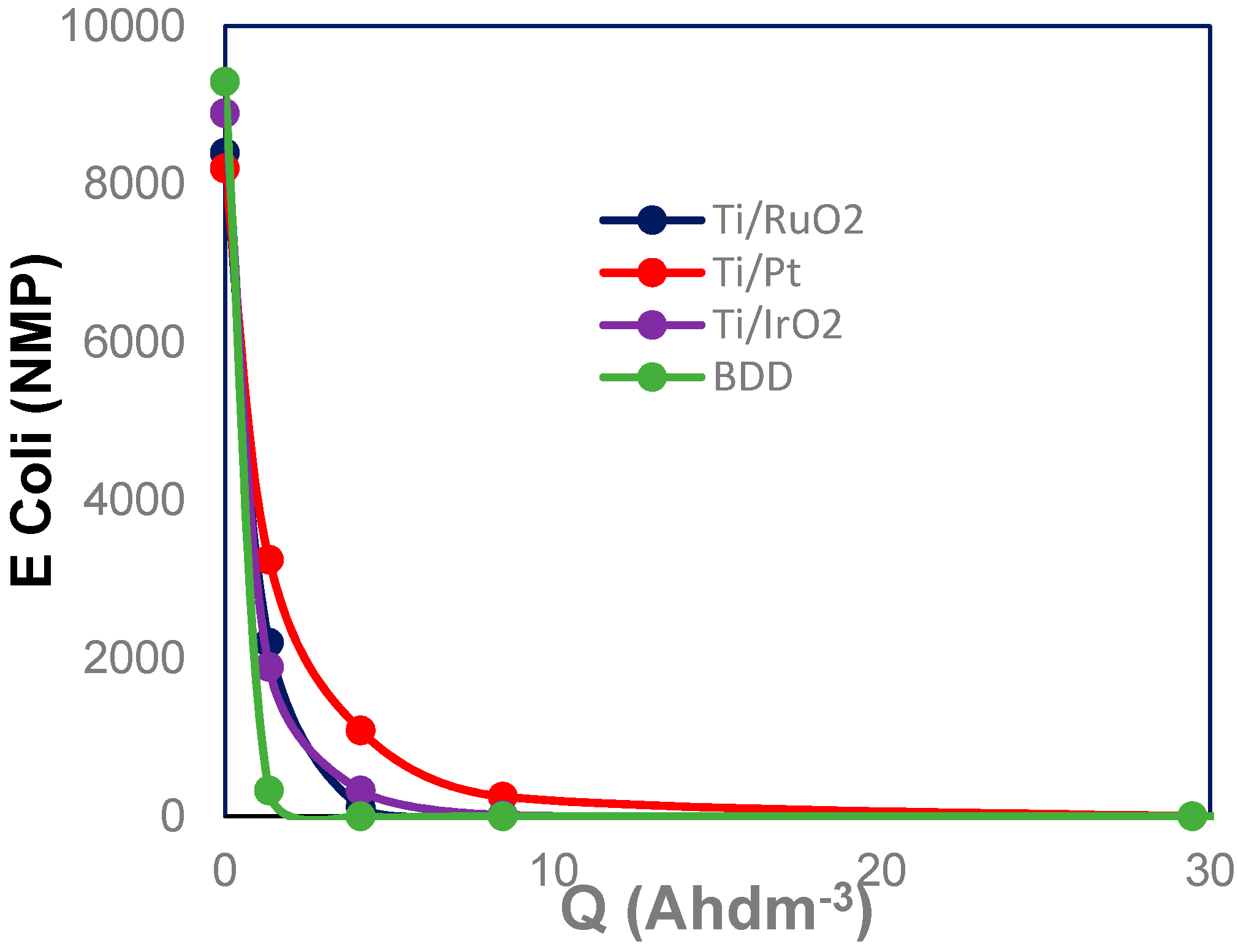
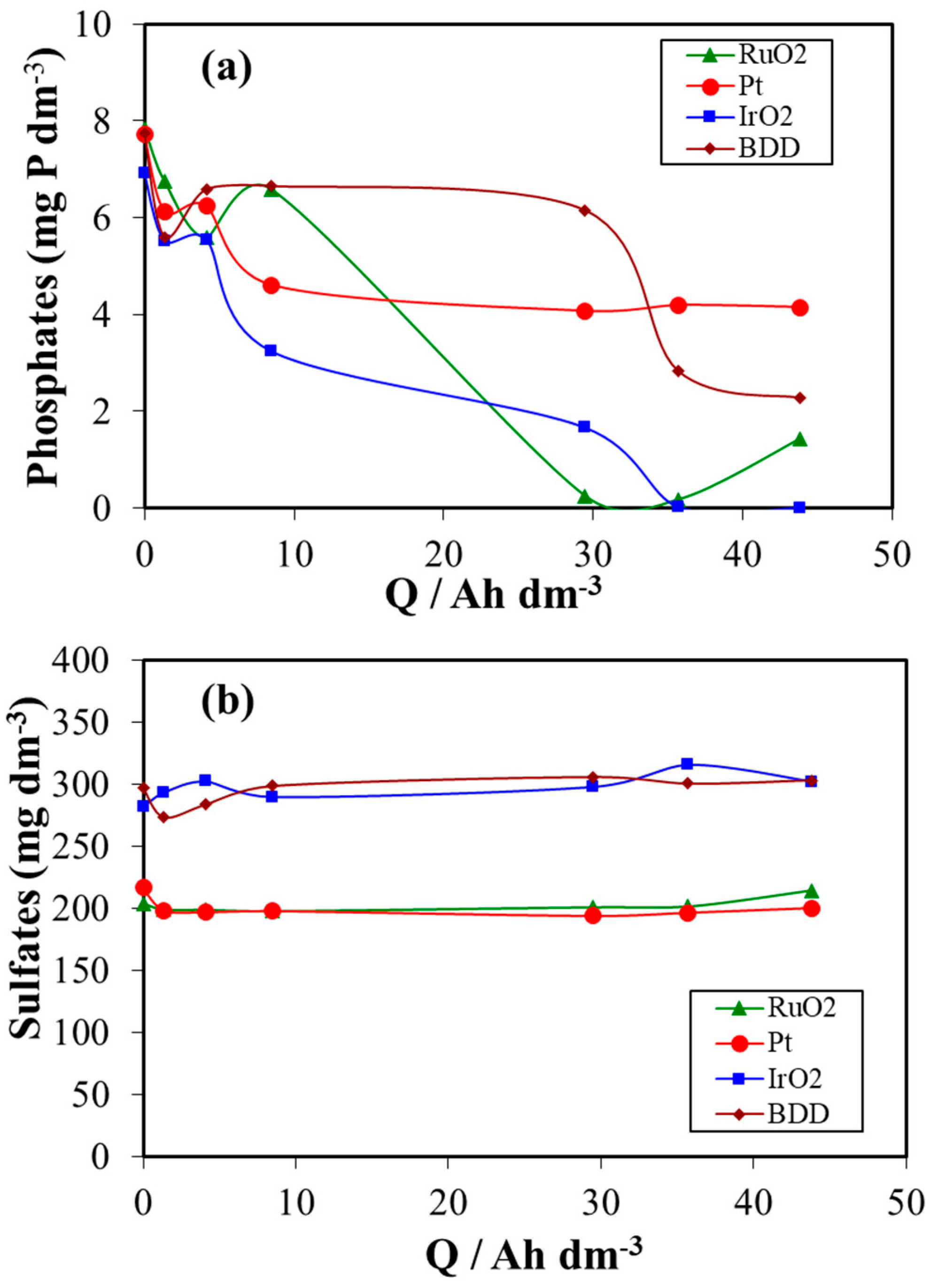
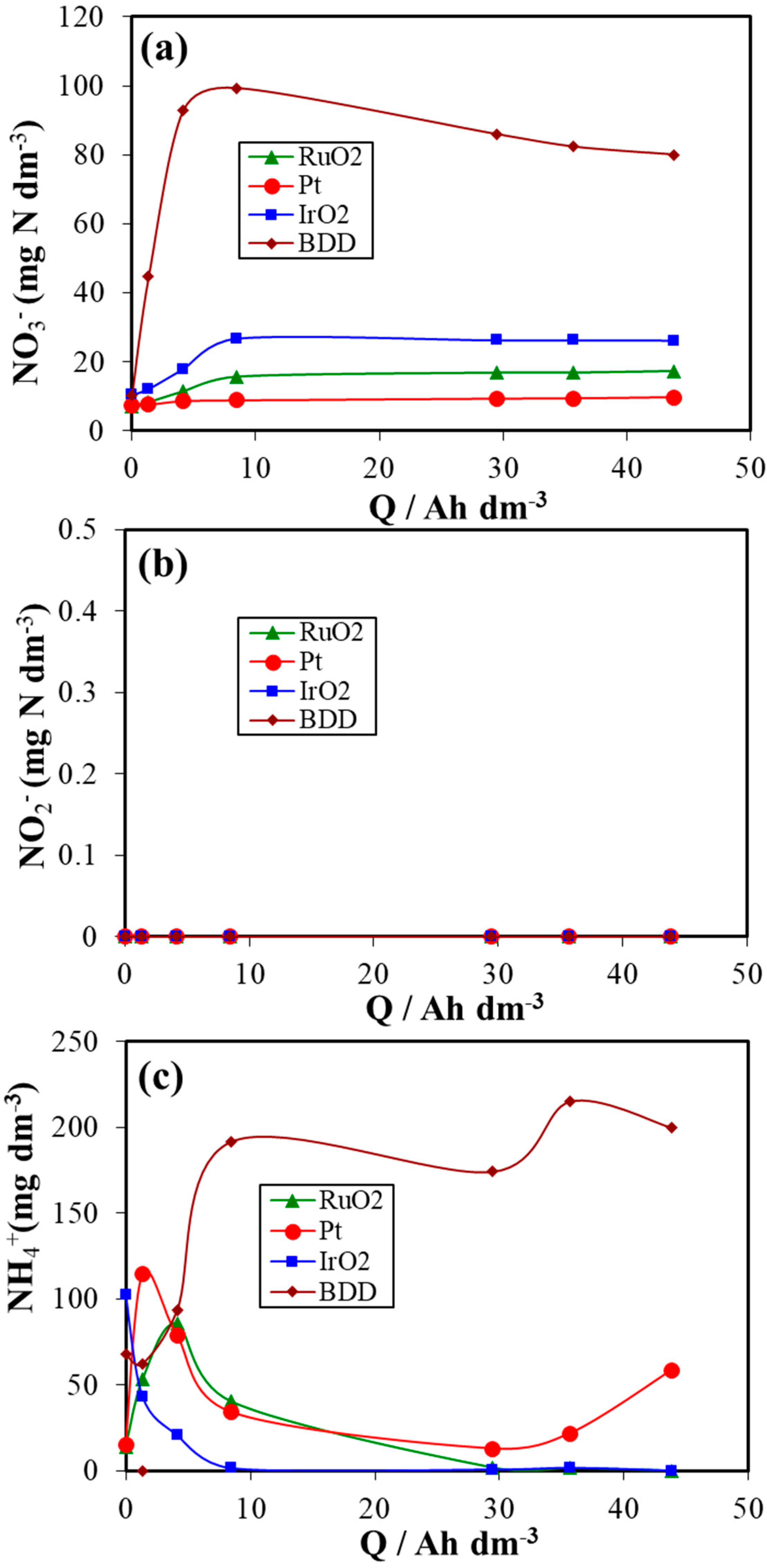
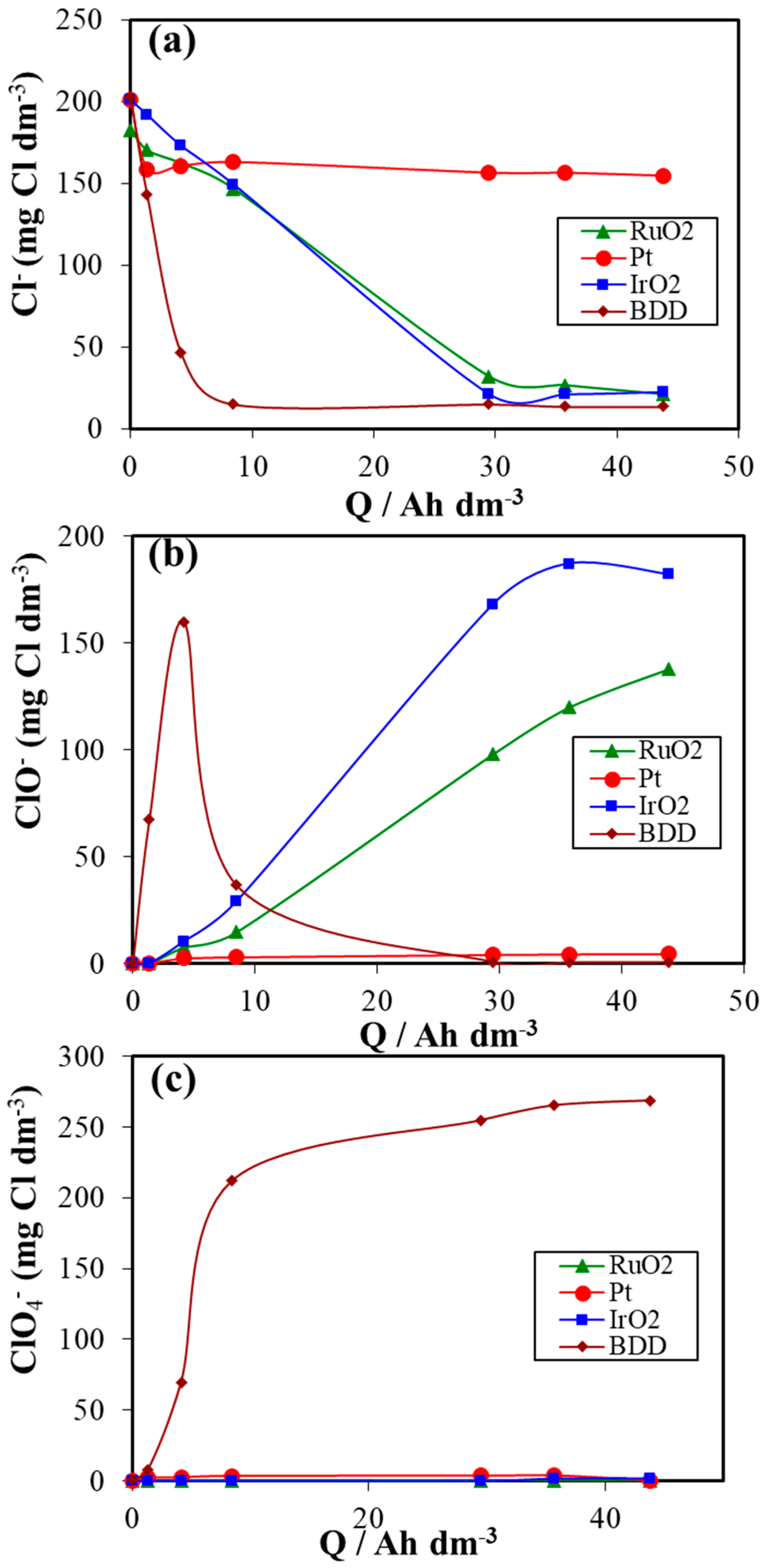
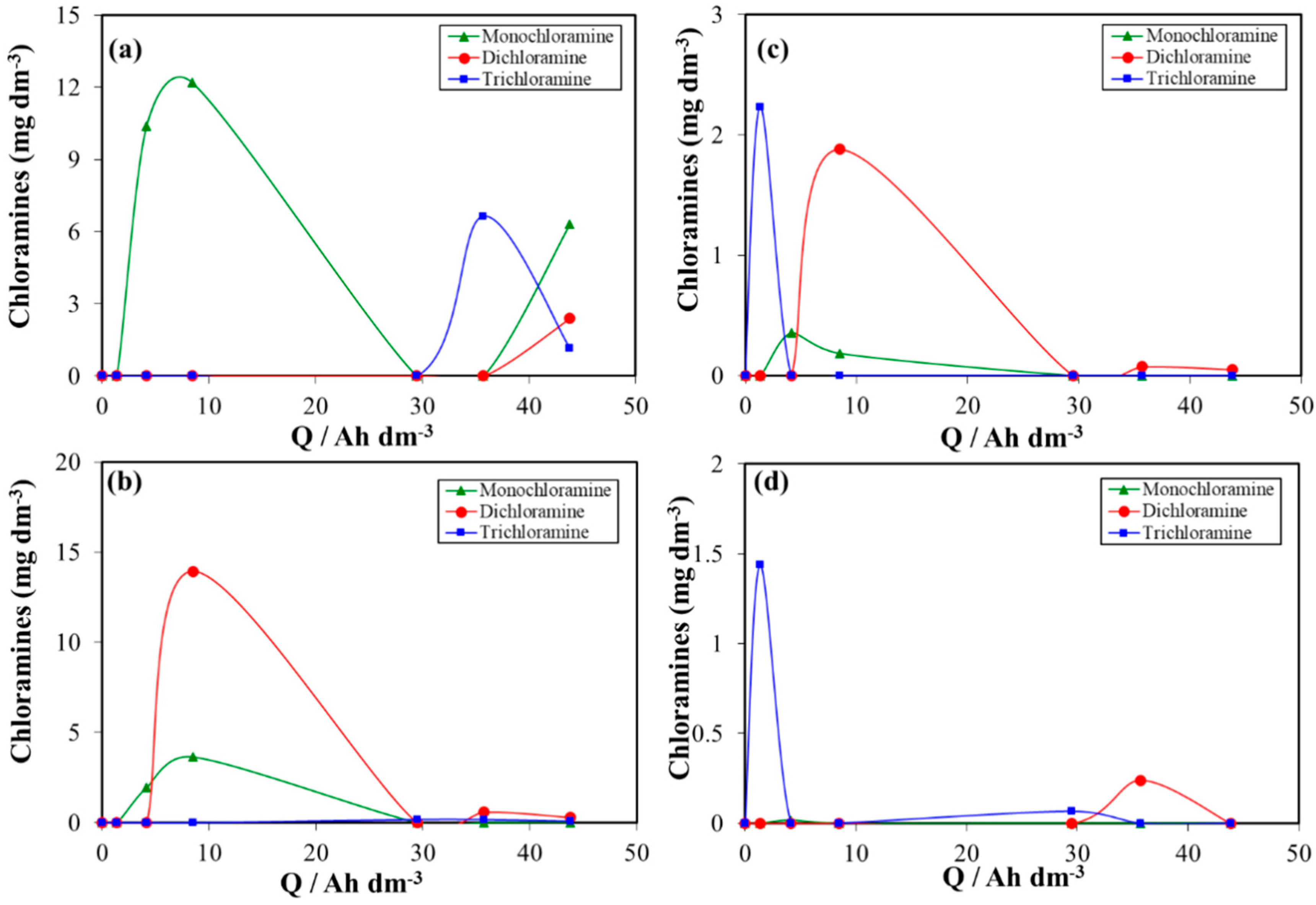
3. Results and Discussion
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Spångberg, J.; Tidåker, P.; Jönsson, H. Environmental Impact of Recycling Nutrients in Human Excreta to Agriculture Compared with Enhanced Wastewater Treatment. Sci. Total Environ. 2014, 493, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Heinonen-Tanski, H.; Sjöblom, A.; Fabritius, H.; Karinen, P. Pure Human Urine Is a Good Fertiliser for Cucumbers. Bioresour. Technol. 2007. [Google Scholar] [CrossRef]
- Jin, P.; Ren, W.; Liang, C.; Wang, X.; Zhang, L. The Study on the Separate Collection and Nutrients Recovery of Urine in Municipal Wastewater. Desalin. Water Treat. 2014. [Google Scholar] [CrossRef]
- Wilsenach, J.; Van Loosdrecht, M. Impact of Separate Urine Collection on Wastewater Treatment Systems. Water Sci. Technol. 2003, 48, 103–110. [Google Scholar] [CrossRef]
- Simha, P.; Ganesapillai, M. Ecological Sanitation and Nutrient Recovery from Human Urine: How Far Have We Come? A Review. Sustain. Environ. Res. 2017. [Google Scholar] [CrossRef]
- Rodrigo, M.A.; Cañizares, P.; Buitrón, C.; Sáez, C. Electrochemical Technologies for the Regeneration of Urban Wastewaters. Electrochim. Acta 2010. [Google Scholar] [CrossRef]
- Martínez-Huitle, C.A.; Brillas, E. Decontamination of Wastewaters Containing Synthetic Organic Dyes by Electrochemical Methods: A General Review. Appl. Catal. B Environ. 2009. [Google Scholar] [CrossRef]
- Garcia-Segura, S.; Ocon, J.D.; Chong, M.N. Electrochemical Oxidation Remediation of Real Wastewater Effluents—A Review. Process Saf. Environ. Prot. 2018. [Google Scholar] [CrossRef]
- Moreira, F.C.; Boaventura, R.A.R.; Brillas, E.; Vilar, V.J.P. Electrochemical Advanced Oxidation Processes: A Review on Their Application to Synthetic and Real Wastewaters. Appl. Catal. B Environ. 2017. [Google Scholar] [CrossRef]
- Zhang, C.; Jiang, Y.; Li, Y.; Hu, Z.; Zhou, L.; Zhou, M. Three-Dimensional Electrochemical Process for Wastewater Treatment: A General Review. Chem. Eng. J. 2013. [Google Scholar] [CrossRef]
- Patel, P.S.; Bandre, N.; Saraf, A.; Ruparelia, J.P. Electro-Catalytic Materials (Electrode Materials) in Electrochemical Wastewater Treatment. Procedia Eng. 2013. [Google Scholar] [CrossRef]
- Nasr, B.; Abdellatif, G.; Cañizares, P.; Sáez, C.; Lobato, J.; Rodrigo, M.A. Electrochemical Oxidation of Hydroquinone, Resorcinol and Catechol on Boron-Doped Diamond Anodes. Environ. Sci. Technol. 2005. [Google Scholar] [CrossRef]
- Cañizares, P.; Gadri, A.; Lobato, J.; Nasr, B.; Paz, R.; Rodrigo, M.A.; Saez, C. Electrochemical Oxidation of Azoic Dyes with Conductive-Diamond Anodes. Ind. Eng. Chem. Res. 2006, 45. [Google Scholar] [CrossRef]
- Cañizares, P.; Louhichi, B.; Gadri, A.; Nasr, B.; Paz, R.; Rodrigo, M.A.; Saez, C. Electrochemical Treatment of the Pollutants Generated in an Ink-Manufacturing Process. J. Hazard. Mater. 2007, 146. [Google Scholar] [CrossRef]
- Rabaaoui, N.; Saad, M.E.K.; Moussaoui, Y.; Allagui, M.S.; Bedoui, A.; Elaloui, E. Anodic Oxidation of O-Nitrophenol on BDD Electrode: Variable Effects and Mechanisms of Degradation. J. Hazard. Mater. 2013. [Google Scholar] [CrossRef] [PubMed]
- Bensalah, N.; Bedoui, A. Enhancing the Performance of Electro-Peroxone by Incorporation of UV Irradiation and BDD Anodes. Environ. Technol. 2017, 38. [Google Scholar] [CrossRef]
- Panizza, M.; Brillas, E.; Comninellis, C. Application of Boron-Doped Diamond Electrodes for Wastewater Treatment. J. Environ. Eng. Manag. 2008, 13, 139–153. [Google Scholar]
- Panizza, M. Importance of Electrode Material in the Electrochemical Treatment of Wastewater Containing Organic Pollutants. Electrochem. Environ. 2010, 25–54. [Google Scholar] [CrossRef]
- Daghrir, R.; Drogui, P.; Tshibangu, J. Efficient Treatment of Domestic Wastewater by Electrochemical Oxidation Process Using Bored Doped Diamond Anode. Sep. Purif. Technol. 2014, 131, 79–83. [Google Scholar] [CrossRef]
- Labiadh, L.; Fernandes, A.; Ciríaco, L.; Pacheco, M.J.; Gadri, A.; Ammar, S.; Lopes, A. Electrochemical Treatment of Concentrate from Reverse Osmosis of Sanitary Landfill Leachate. J. Environ. Manag. 2016. [Google Scholar] [CrossRef]
- Lan, Y.; Coetsier, C.; Causserand, C.; Groenen Serrano, K. On the Role of Salts for the Treatment of Wastewaters Containing Pharmaceuticals by Electrochemical Oxidation Using a Boron Doped Diamond Anode. Electrochim. Acta 2017. [Google Scholar] [CrossRef]
- Dbira, S.; Bensalah, N.; Bedoui, A.; Cañizares, P.; Rodrigo, M.A. Treatment of Synthetic Urine by Electrochemical Oxidation Using Conductive-Diamond Anodes. Environ. Sci. Pollut. Res. 2015, 22. [Google Scholar] [CrossRef] [PubMed]
- Dbira, S.; Bensalah, N.; Cañizares, P.; Rodrigo, M.A.; Bedoui, A. The Electrolytic Treatment of Synthetic Urine Using DSA Electrodes. J. Electroanal. Chem. 2015, 744. [Google Scholar] [CrossRef]
- Dbira, S.; Bensalah, N.; Bedoui, A. Mechanism and Kinetics of Electrochemical Degradation of Uric Acid Using Conductive-Diamond Anodes. Environ. Technol. 2016, 37. [Google Scholar] [CrossRef]
- Bensalah, N.; Dbira, S.; Bedoui, A. The Contribution of Mediated Oxidation Mechanisms in the Electrolytic Degradation of Cyanuric Acid Using Diamond Anodes. J. Environ. Sci. 2015, 45. [Google Scholar] [CrossRef]
- Martínez-Huitle, C.A.; Brillas, E. Electrochemical Alternatives for Drinking Water Disinfection. Angew. Chem. Int. Ed. 2008, 47, 1998–2005. [Google Scholar] [CrossRef]
- Li, H.; Zhu, X.; Ni, J. Comparison of Electrochemical Method with Ozonation, Chlorination and Monochloramination in Drinking Water Disinfection. Electrochim. Acta 2011. [Google Scholar] [CrossRef]
- Cashman, S.; Gaglione, A.; Mosley, J.; Weiss, L.; Hawkins, T.R.; Ashbolt, N.J.; Cashdollar, J.; Xue, X.; Ma, C.; Arden, S. Environmental and Cost Life Cycle Assessment of Disinfection Options for Municipal Drinking Water Treatment; US Environmental Protection Agency: Cincinnati, OH, USA, 2014.
- Jeong, J.; Kim, C.; Yoon, J. The Effect of Electrode Material on the Generation of Oxidants and Microbial Inactivation in the Electrochemical Disinfection Processes. Water Res. 2009. [Google Scholar] [CrossRef]
- Jeong, J.; Kim, J.Y.; Yoon, J. The Role of Reactive Oxygen Species in the Electrochemical Inactivation of Microorganisms. Environ. Sci. Technol. 2006. [Google Scholar] [CrossRef]
- Bensalah, N.; Abdel-Wahab, A. Electrochemical Inactivation of P. Aeruginosa, A. Hydrophila, L. Pneumophila Using Boron Doped Diamond Anodes. J. Adv. Oxid. Technol. 2013, 16. [Google Scholar] [CrossRef]
- Llanos, J.; Cotillas, S.; Cañizares, P.; Rodrigo, M.A. Effect of Bipolar Electrode Material on the Reclamation of Urban Wastewater by an Integrated Electrodisinfection/Electrocoagulation Process. Water Res. 2014. [Google Scholar] [CrossRef]
- Huang, X.; Qu, Y.; Cid, C.A.; Finke, C.; Hoffmann, M.R.; Lim, K.; Jiang, S.C. Electrochemical Disinfection of Toilet Wastewater Using Wastewater Electrolysis Cell. Water Res. 2016. [Google Scholar] [CrossRef]
- Särkkä, H.; Bhatnagar, A.; Sillanpää, M. Recent Developments of Electro-Oxidation in Water Treatment—A Review. J. Electroanal. Chem. 2015. [Google Scholar] [CrossRef]
- Bensalah, N.; Alfaro, M.A.Q.; Martínez-Huitle, C.A. Electrochemical Treatment of Synthetic Wastewaters Containing Alphazurine A Dye. Chem. Eng. J. 2009, 149. [Google Scholar] [CrossRef]
- Bergmann, M.E.H.; Rollin, J.; Iourtchouk, T. The Occurrence of Perchlorate during Drinking Water Electrolysis Using BDD Anodes. Electrochim. Acta 2009. [Google Scholar] [CrossRef]
- Ninomiya, K.; Arakawa, M.; Ogino, C.; Shimizu, N. Inactivation of Escherichia Coli by Sonoelectrocatalytic Disinfection Using TiO2 as Electrode. Ultrason. Sonochem. 2013. [Google Scholar] [CrossRef]
- Jeong, J.; Kim, J.Y.; Cho, M.; Choi, W.; Yoon, J. Inactivation of Escherichia Coli in the Electrochemical Disinfection Process Using a Pt Anode. Chemosphere 2007. [Google Scholar] [CrossRef]
- Ghernaout, D.; Naceur, M.W.; Aouabed, A. On the Dependence of Chlorine By-Products Generated Species Formation of the Electrode Material and Applied Charge during Electrochemical Water Treatment. Desalination 2011. [Google Scholar] [CrossRef]
- Schmalz, V.; Dittmar, T.; Haaken, D.; Worch, E. Electrochemical Disinfection of Biologically Treated Wastewater from Small Treatment Systems by Using Boron-Doped Diamond (BDD) Electrodes—Contribution for Direct Reuse of Domestic Wastewater. Water Res. 2009. [Google Scholar] [CrossRef]
- Lacasa, E.; Tsolaki, E.; Sbokou, Z.; Rodrigo, M.A.; Mantzavinos, D.; Diamadopoulos, E. Electrochemical Disinfection of Simulated Ballast Water on Conductive Diamond Electrodes. Chem. Eng. J. 2013. [Google Scholar] [CrossRef]
- Haaken, D.; Dittmar, T.; Schmalz, V.; Worch, E. Influence of Operating Conditions and Wastewater-Specific Parameters on the Electrochemical Bulk Disinfection of Biologically Treated Sewage at Boron-Doped Diamond (BDD) Electrodes. Desalin. Water Treat. 2012. [Google Scholar] [CrossRef]
- Cotillas, S.; Llanos, J.; Cañizares, P.; Mateo, S.; Rodrigo, M.A. Optimization of an Integrated Electrodisinfection/Electrocoagulation Process with Al Bipolar Electrodes for Urban Wastewater Reclamation. Water Res. 2013. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Kubota, Y.; Murakami, T.; Ochiai, T.; Ishiguro, H.; Nakata, K.; Fujishima, A. Electrochemical Inactivation Kinetics of Boron-Doped Diamond Electrode on Waterborne Pathogens. J. Water Health 2011. [Google Scholar] [CrossRef]
- Cano, A.; Cañizares, P.; Barrera-Díaz, C.; Sáez, C.; Rodrigo, M.A. Use of Conductive-Diamond Electrochemical-Oxidation for the Disinfection of Several Actual Treated Wastewaters. Chem. Eng. J. 2012. [Google Scholar] [CrossRef]
- Guinea, E.; Arias, C.; Cabot, P.L.; Garrido, J.A.; Rodríguez, R.M.; Centellas, F.; Brillas, E. Mineralization of Salicylic Acid in Acidic Aqueous Medium by Electrochemical Advanced Oxidation Processes Using Platinum and Boron-Doped Diamond as Anode and Cathodically Generated Hydrogen Peroxide. Water Res. 2008. [Google Scholar] [CrossRef] [PubMed]
- Pieczyńska, A.; Ofiarska, A.; Borzyszkowska, A.F.; Białk-Bielińska, A.; Stepnowski, P.; Stolte, S.; Siedlecka, E.M. A Comparative Study of Electrochemical Degradation of Imidazolium and Pyridinium Ionic Liquids: A Reaction Pathway and Ecotoxicity Evaluation. Sep. Purif. Technol. 2015. [Google Scholar] [CrossRef]
- Loos, G.; Scheers, T.; Van Eyck, K.; Van Schepdael, A.; Adams, E.; Van der Bruggen, B.; Cabooter, D.; Dewil, R. Electrochemical Oxidation of Key Pharmaceuticals Using a Boron Doped Diamond Electrode. Sep. Purif. Technol. 2018. [Google Scholar] [CrossRef]
- Marselli, B.; Garcia-Gomez, J.; Michaud, P.; Rodrigo, M.A.; Comninellis, C. Electrogeneration of Hydroxyl Radicals on Boron-Doped Diamond Electrodes. J. Electrochem. Soc. 2003. [Google Scholar] [CrossRef]
- Costa, C.R.; Olivi, P. Effect of Chloride Concentration on the Electrochemical Treatment of a Synthetic Tannery Wastewater. Electrochim. Acta 2009. [Google Scholar] [CrossRef]
- Radjenovic, J.; Sedlak, D.L. Challenges and Opportunities for Electrochemical Processes as Next-Generation Technologies for the Treatment of Contaminated Water. Environ. Sci. Technol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Scialdone, O.; Randazzo, S.; Galia, A.; Silvestri, G. Electrochemical Oxidation of Organics in Water: Role of Operative Parameters in the Absence and in the Presence of NaCl. Water Res. 2009. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Särkkä, H.; Sillanpää, M. A Comparative Experimental Study on Methyl Orange Degradation by Electrochemical Oxidation on BDD and MMO Electrodes. Sep. Purif. Technol. 2011. [Google Scholar] [CrossRef]
- Li, M.; Feng, C.; Zhang, Z.; Sugiura, N. Efficient Electrochemical Reduction of Nitrate to Nitrogen Using Ti/IrO2-Pt Anode and Different Cathodes. Electrochim. Acta 2009. [Google Scholar] [CrossRef]
- Lacasa, E.; Llanos, J.; Cañizares, P.; Rodrigo, M.A. Electrochemical Denitrificacion with Chlorides Using DSA and BDD Anodes. Chem. Eng. J. 2012. [Google Scholar] [CrossRef]
- Polcaro, A.M.; Vacca, A.; Mascia, M.; Palmas, S.; Rodiguez Ruiz, J. Electrochemical Treatment of Waters with BDD Anodes: Kinetics of the Reactions Involving Chlorides. J. Appl. Electrochem. 2009. [Google Scholar] [CrossRef]
- Wächter, N.; Pereira, G.F.; Rocha-Filho, R.C.; Bocchi, N.; Biaggio, S.R. Comparative Electrochemical Degradation of the Acid Yellow 49 Dye Using Boron-Doped Diamond, β-PbO2 and DSA® Anodes in a Flow Reactor. Int. J. Electrochem. Sci. 2015, 10, 1361–1371. [Google Scholar]
- Gilcreas, F.W. Standard Methods for the Examination of Water and Waste Water. Am. J. Public Health Nations Health 2008. [Google Scholar] [CrossRef]
- APHA. Standard Methods for Examination of Water and Wastewater (Standard Methods for the Examination of Water and Wastewater); American Public Health Association (APHA): Washington, DC, USA, 2012; ISBN 9780875532356. [Google Scholar]
- Valero, D.; García-García, V.; Expósito, E.; Aldaz, A.; Montiel, V. Electrochemical Treatment of Wastewater from Almond Industry Using DSA-Type Anodes: Direct Connection to a PV Generator. Sep. Purif. Technol. 2014. [Google Scholar] [CrossRef]
- Oturan, M.A.; Brillas, E. Electrochemical Advanced Oxidation Processes (EAOPs) for Environmental Applications. Port. Electrochim. Acta 2009. [Google Scholar] [CrossRef]
- Brillas, E.; Martınez-Huitle, C.A. Environmental Decontamination of Wastewaters Containing Synthetic Organic Dyes by Electrochemical Methods: A updated review. Appl. Catal. B Environ. 2015. [Google Scholar] [CrossRef]
- Bedoui, A.; Ahmadi, M.F.; Bensalah, N.; Gadri, A. Comparative Study of Eriochrome Black T Treatment by BDD-Anodic Oxidation and Fenton Process. Chem. Eng. J. 2009, 146. [Google Scholar] [CrossRef]
- Faouzi, M.; Cañizares, P.; Gadri, A.; Lobato, J.; Nasr, B.; Paz, R.; Rodrigo, M.A.; Saez, C. Advanced Oxidation Processes for the Treatment of Wastes Polluted with Azoic Dyes. Electrochim. Acta 2006, 52. [Google Scholar] [CrossRef]
- Faouzi Elahmadi, M.; Bensalah, N.; Gadri, A. Treatment of Aqueous Wastes Contaminated with Congo Red Dye by Electrochemical Oxidation and Ozonation Processes. J. Hazard. Mater. 2009, 168. [Google Scholar] [CrossRef]
- Bensalah, N.; Ahmadi, M.F.; Gadri, A. Electrochemical Treatment of Wastewaters Containing 4-Nitrocathecol Using Boron-Doped Diamond Anodes. J. Environ. Eng. Sci. 2013. [Google Scholar] [CrossRef]
- Orescanin, V.; Kollar, R.; Nad, K.; Halkijevic, I.; Kuspilic, M.; Findri Gustek, S. Removal of Arsenic, Phosphates and Ammonia from Well Water Using Electrochemical/Chemical Methods and Advanced Oxidation: A Pilot Plant Approach. J. Environ. Sci. Heal. Part A 2014. [Google Scholar] [CrossRef] [PubMed]
- Lacasa, E.; Cañizares, P.; Sáez, C.; Fernández, F.J.; Rodrigo, M.A. Electrochemical Phosphates Removal Using Iron and Aluminium Electrodes. Chem. Eng. J. 2011. [Google Scholar] [CrossRef]
- Cañizares, P.; Sáez, C.; Sánchez-Carretero, A.; Rodrigo, M.A. Influence of the Characteristics of P-Si BDD Anodes on the Efficiency of Peroxodiphosphate Electrosynthesis Process. Electrochem. Commun. 2008. [Google Scholar] [CrossRef]
- Garcia-Segura, S.; Lanzarini-Lopes, M.; Hristovski, K.; Westerhoff, P. Electrocatalytic Reduction of Nitrate: Fundamentals to Full-Scale Water Treatment Applications. Appl. Catal. B Environ. 2018. [Google Scholar] [CrossRef]
- Kuang, P.; Feng, C.; Li, M.; Chen, N.; Hu, Q.; Wang, G.; Li, R. Improvement on Electrochemical Reduction of Nitrate in Synthetic Groundwater by Reducing Anode Surface Area. J. Electrochem. Soc. 2017. [Google Scholar] [CrossRef]
- Raut, A.S.; Glass, J.T.; Deshusses, M.A.; Cunningham, G.B.; Parker, C.B.; Stoner, B.R.; Klem, E.J.D. Electrochemical Disinfection of Human Urine for Water-Free and Additive-Free Toilets Using Boron-Doped Diamond Electrodes. ECS Trans. 2013. [Google Scholar] [CrossRef]
- Raut, A.S.; Cunningham, G.B.; Parker, C.B.; Klem, E.J.D.; Stoner, B.R.; Deshusses, M.A.; Glass, J.T. Disinfection of E. coli Contaminated Urine Using Boron-Doped Diamond Electrodes. J. Electrochem. Soc. 2014. [Google Scholar] [CrossRef]
- Rajab, M.; Heim, C.; Letzel, T.; Drewes, J.E.; Helmreich, B. Electrochemical Disinfection Using Boron-Doped Diamond Electrode—The Synergetic Effects of in Situ Ozone and Free Chlorine Generation. Chemosphere 2015. [Google Scholar] [CrossRef]
- Cotillas, S.; Lacasa, E.; Sáez, C.; Cañizares, P.; Rodrigo, M.A. Disinfection of Urine by Conductive-Diamond Electrochemical Oxidation. Appl. Catal. B Environ. 2018. [Google Scholar] [CrossRef]
- Cano, A.; Cañizares, P.; Barrera, C.; Sáez, C.; Rodrigo, M.A. Use of Low Current Densities in Electrolyses with Conductive-Diamond Electrochemical—Oxidation to Disinfect Treated Wastewaters for Reuse. Electrochem. Commun. 2011. [Google Scholar] [CrossRef]
- Mostafa, E.; Reinsberg, P.; Garcia-Segura, S.; Baltruschat, H. Chlorine Species Evolution during Electrochlorination on Boron-Doped Diamond Anodes: In-Situ Electrogeneration of Cl2, Cl2O and ClO2. Electrochim. Acta 2018. [Google Scholar] [CrossRef]
- Sánchez-Carretero, A.; Sáez, C.; Cañizares, P.; Rodrigo, M.A. Electrochemical Production of Perchlorates Using Conductive Diamond Electrolyses. Chem. Eng. J. 2011. [Google Scholar] [CrossRef]
- Mousset, E.; Oturan, N.; van Hullebusch, E.D.; Guibaud, G.; Esposito, G.; Oturan, M.A. Treatment of Synthetic Soil Washing Solutions Containing Phenanthrene and Cyclodextrin by Electro-Oxidation. Influence of Anode Materials on Toxicity Removal and Biodegradability Enhancement. Appl. Catal. B Environ. 2014. [Google Scholar] [CrossRef]
- Bergmann, M.E.H.; Rollin, J.; Koparal, A.S. Chlorate and Perchlorate—New Criterions for Environmentally-Friendly Processes in Advanced Oxidation. Water Pract. Technol. 2010. [Google Scholar] [CrossRef]
- Bergmann, M.E.H.; Koparal, A.S.; Iourtchouk, T. Electrochemical Advanced Oxidation Processes, Formation of Halogenate and Perhalogenate Species: A Critical Review. Crit. Rev. Environ. Sci. Technol. 2014. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dbira, S.; Bensalah, N.; Ahmad, M.I.; Bedoui, A. Electrochemical Oxidation/Disinfection of Urine Wastewaters with Different Anode Materials. Materials 2019, 12, 1254. https://doi.org/10.3390/ma12081254
Dbira S, Bensalah N, Ahmad MI, Bedoui A. Electrochemical Oxidation/Disinfection of Urine Wastewaters with Different Anode Materials. Materials. 2019; 12(8):1254. https://doi.org/10.3390/ma12081254
Chicago/Turabian StyleDbira, Sondos, Nasr Bensalah, Mohammad I. Ahmad, and Ahmed Bedoui. 2019. "Electrochemical Oxidation/Disinfection of Urine Wastewaters with Different Anode Materials" Materials 12, no. 8: 1254. https://doi.org/10.3390/ma12081254
APA StyleDbira, S., Bensalah, N., Ahmad, M. I., & Bedoui, A. (2019). Electrochemical Oxidation/Disinfection of Urine Wastewaters with Different Anode Materials. Materials, 12(8), 1254. https://doi.org/10.3390/ma12081254





