Effects of Aluminum Sulfate and Quicklime/Fluorgypsum Ratio on the Properties of Calcium Sulfoaluminate (CSA) Cement-Based Double Liquid Grouting Materials
Abstract
1. Introduction
→ 3CaO∙Al2O3∙3CaSO4∙32H2O + 2(Al2O3∙3H2O)
2. Materials and Methods
2.1. Materials
2.2. Test Methods
3. Results
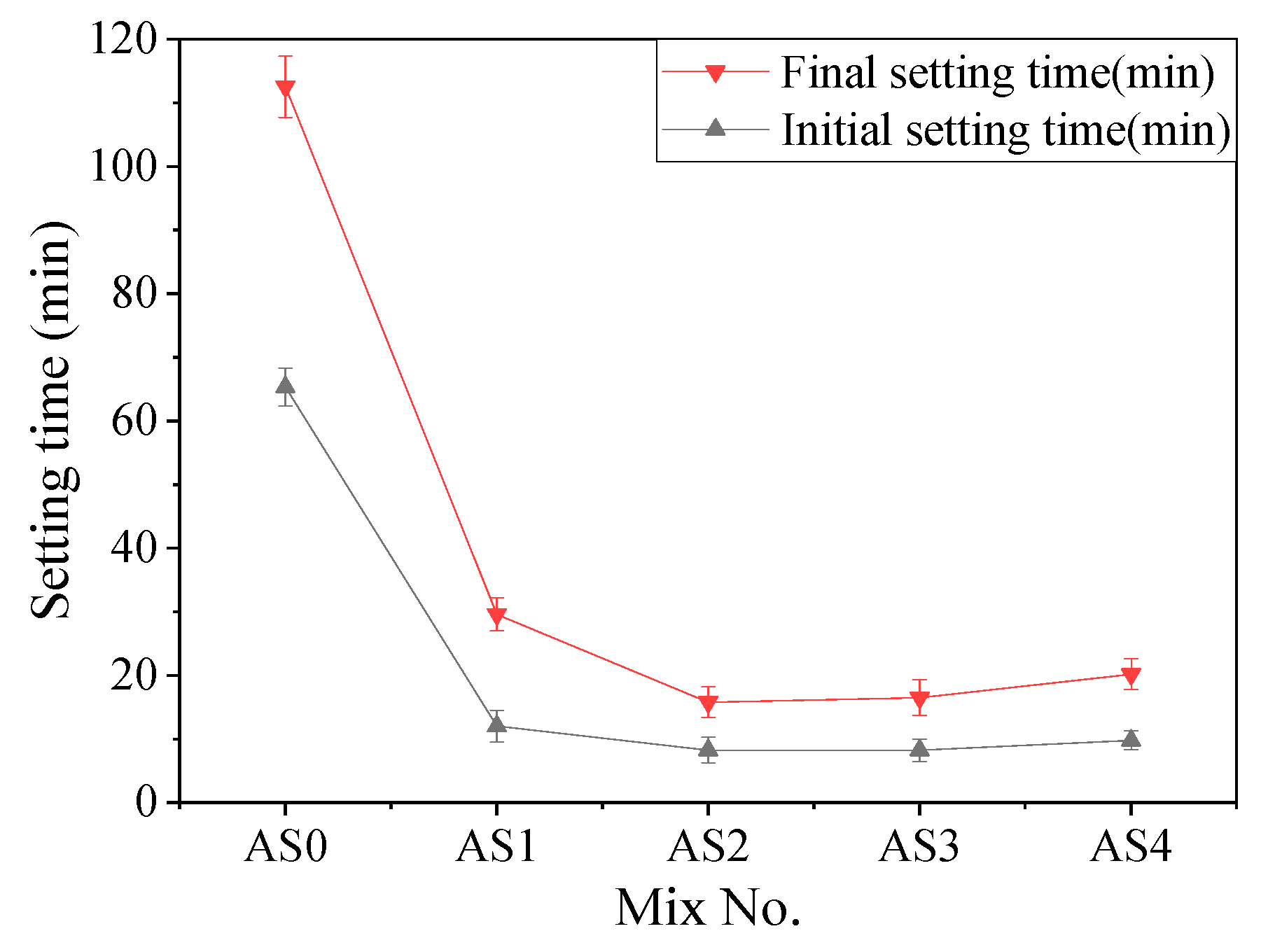
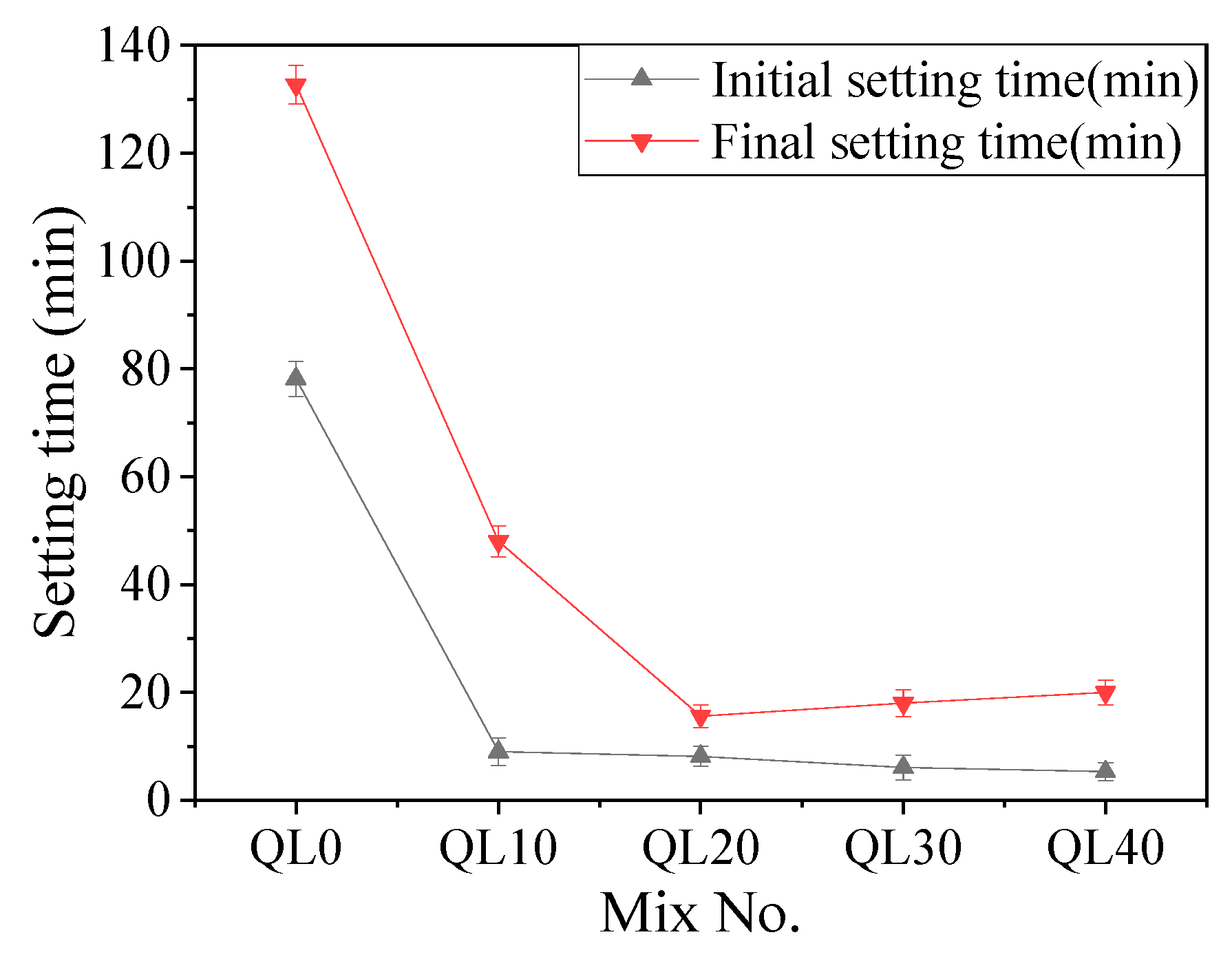
3.1. Setting Time
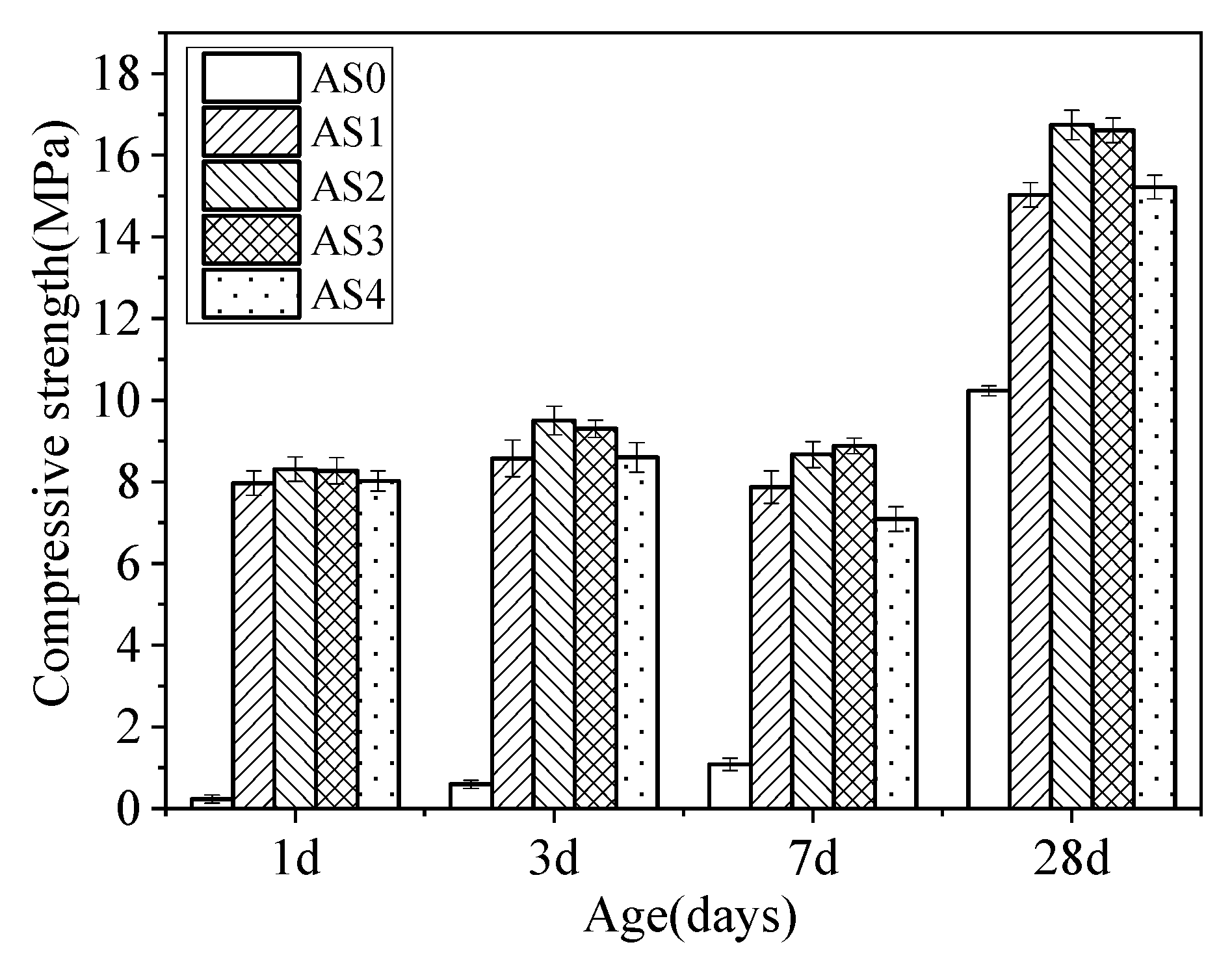
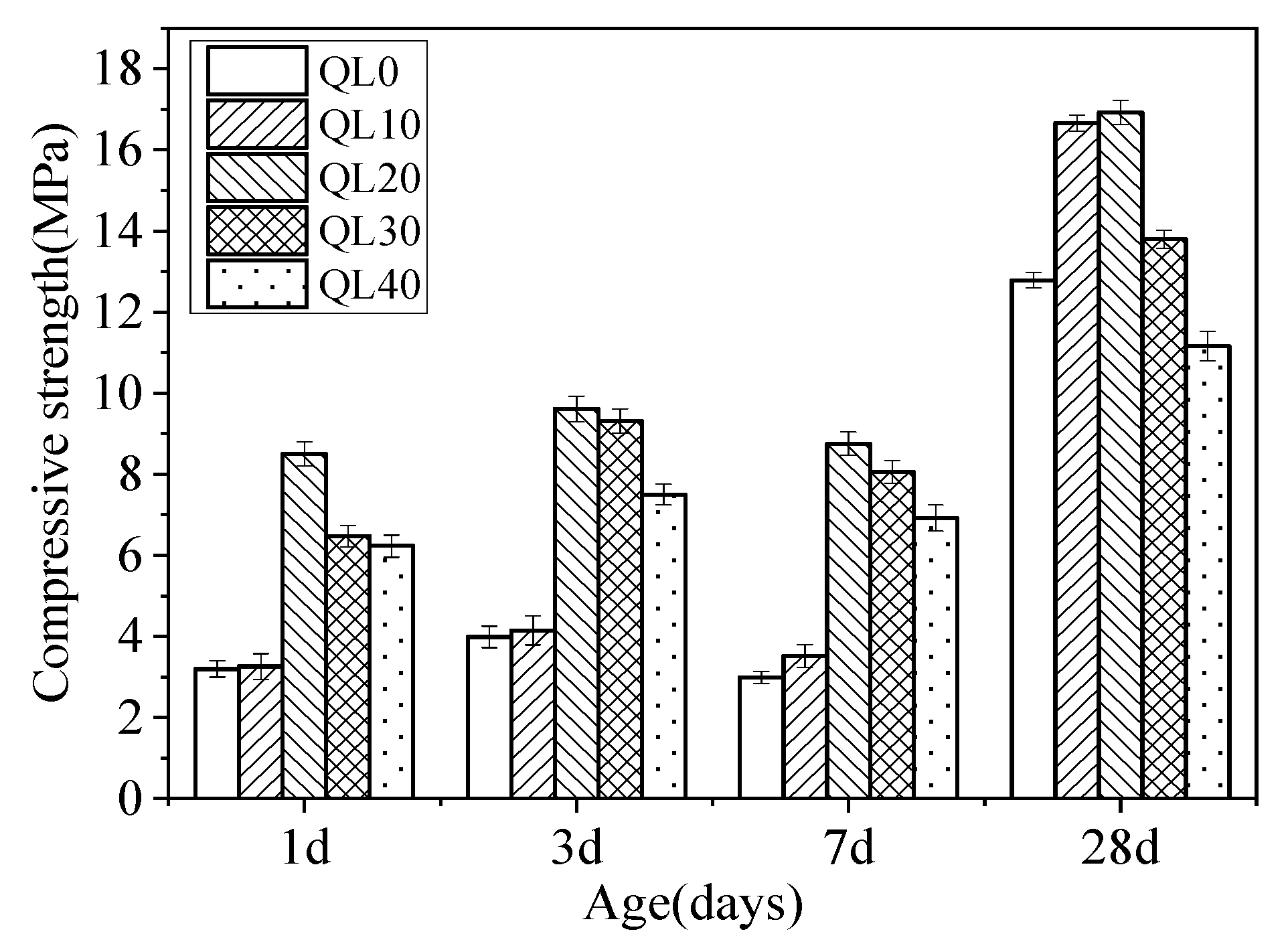
3.2. Compressive Strength
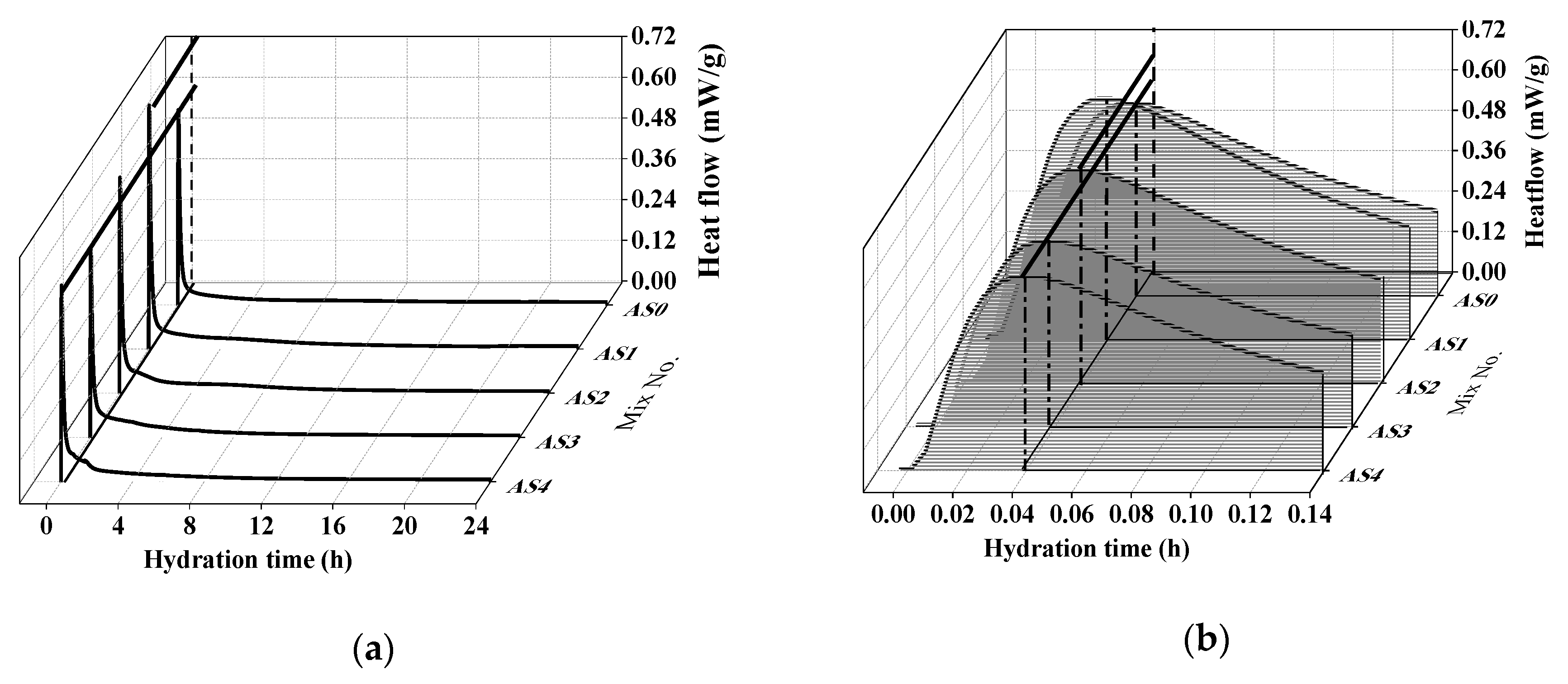
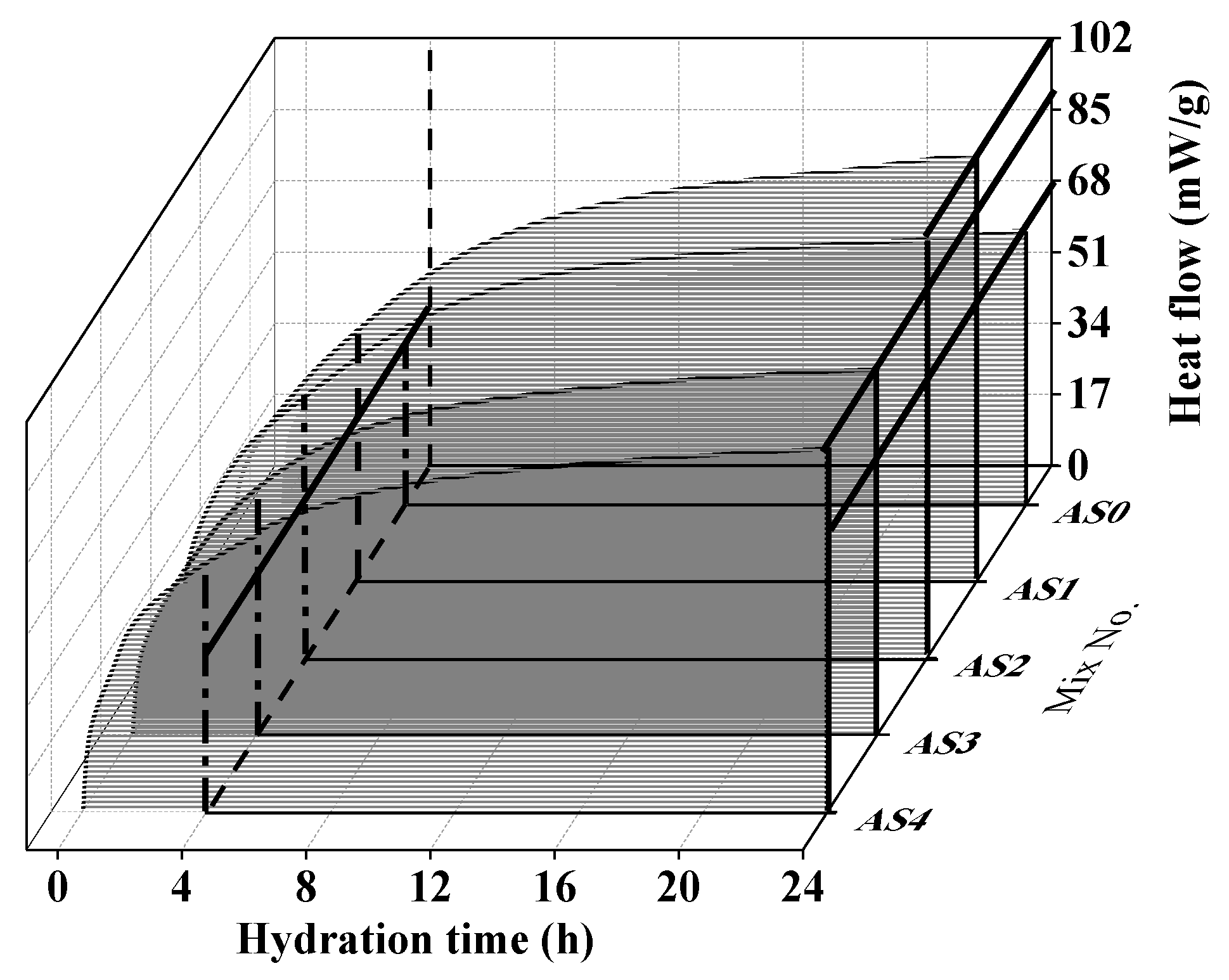
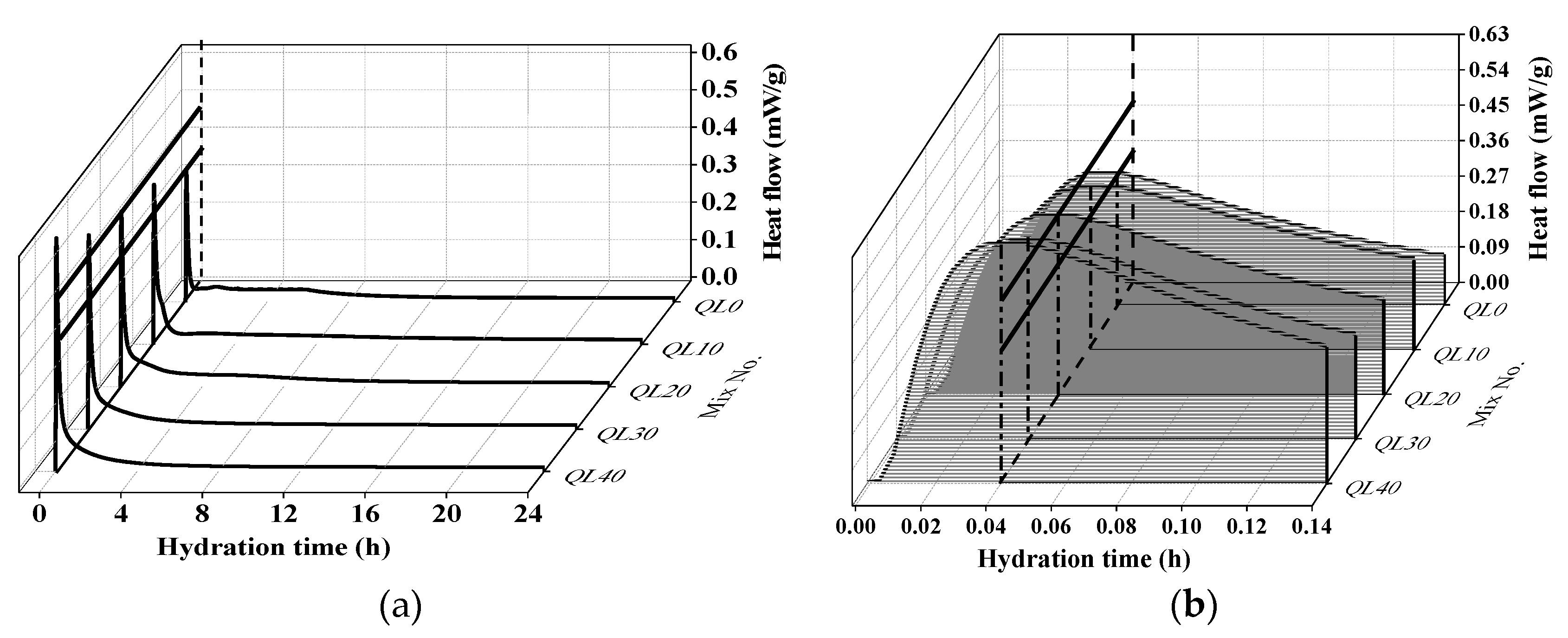
3.3. Hydration Heat
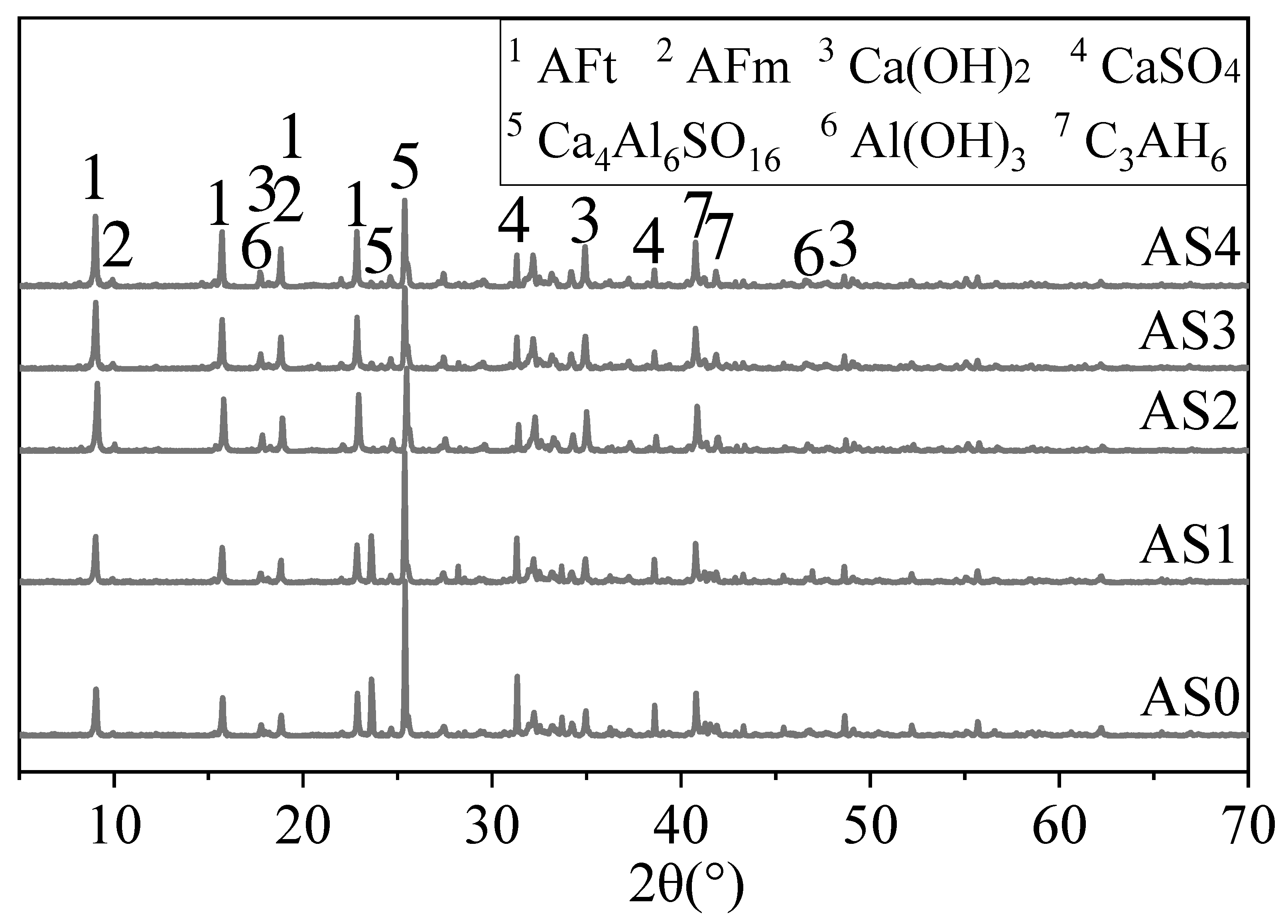
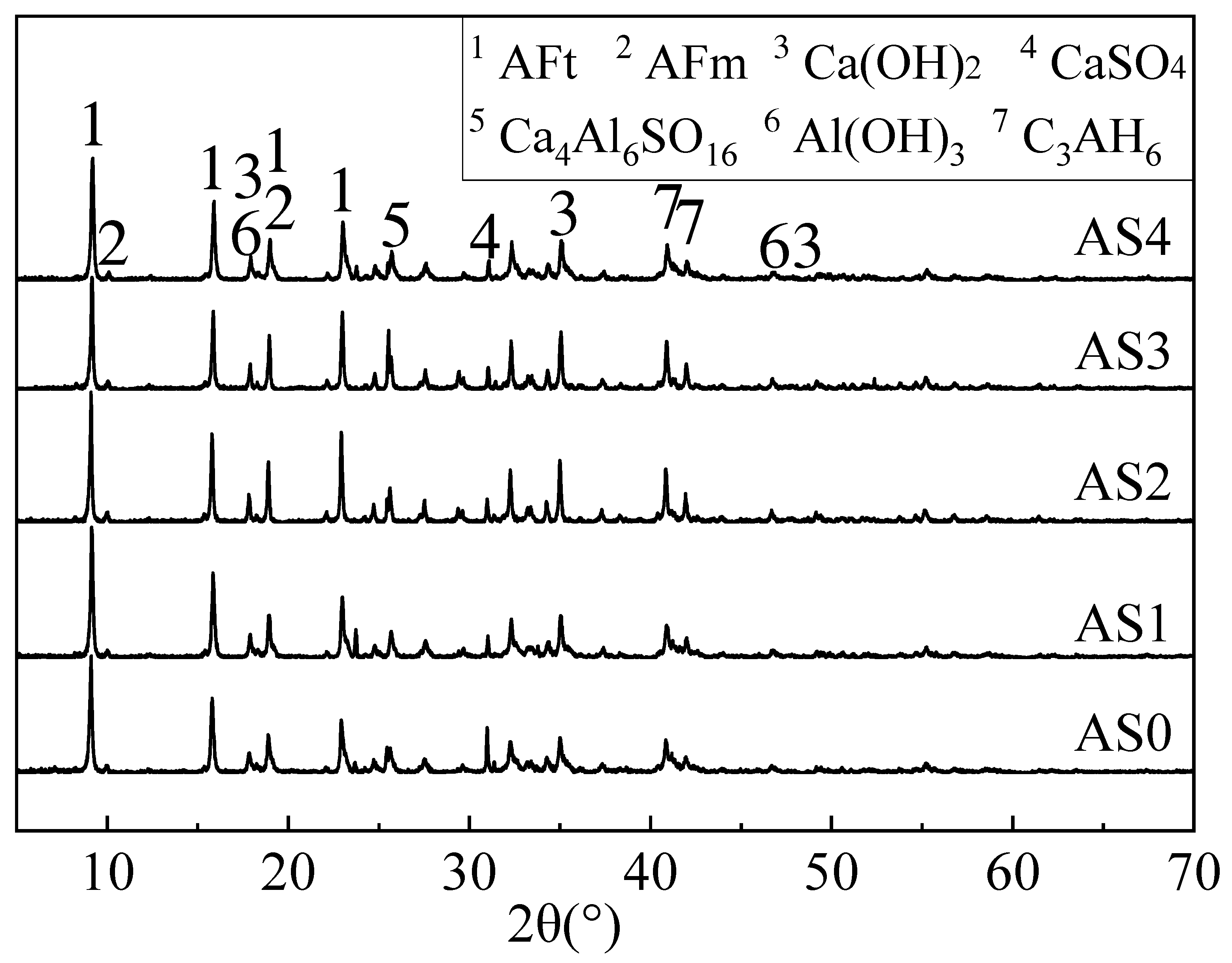
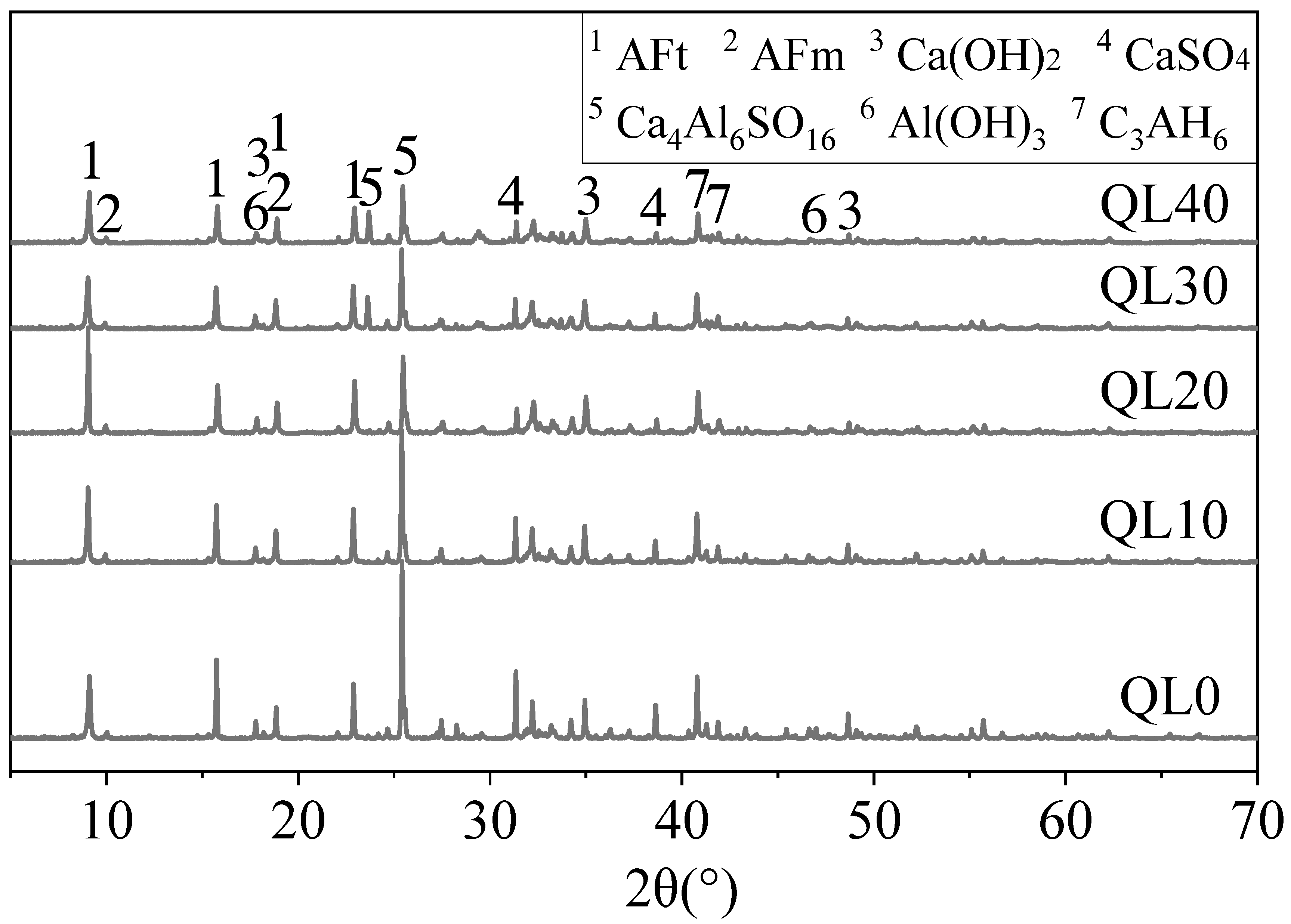
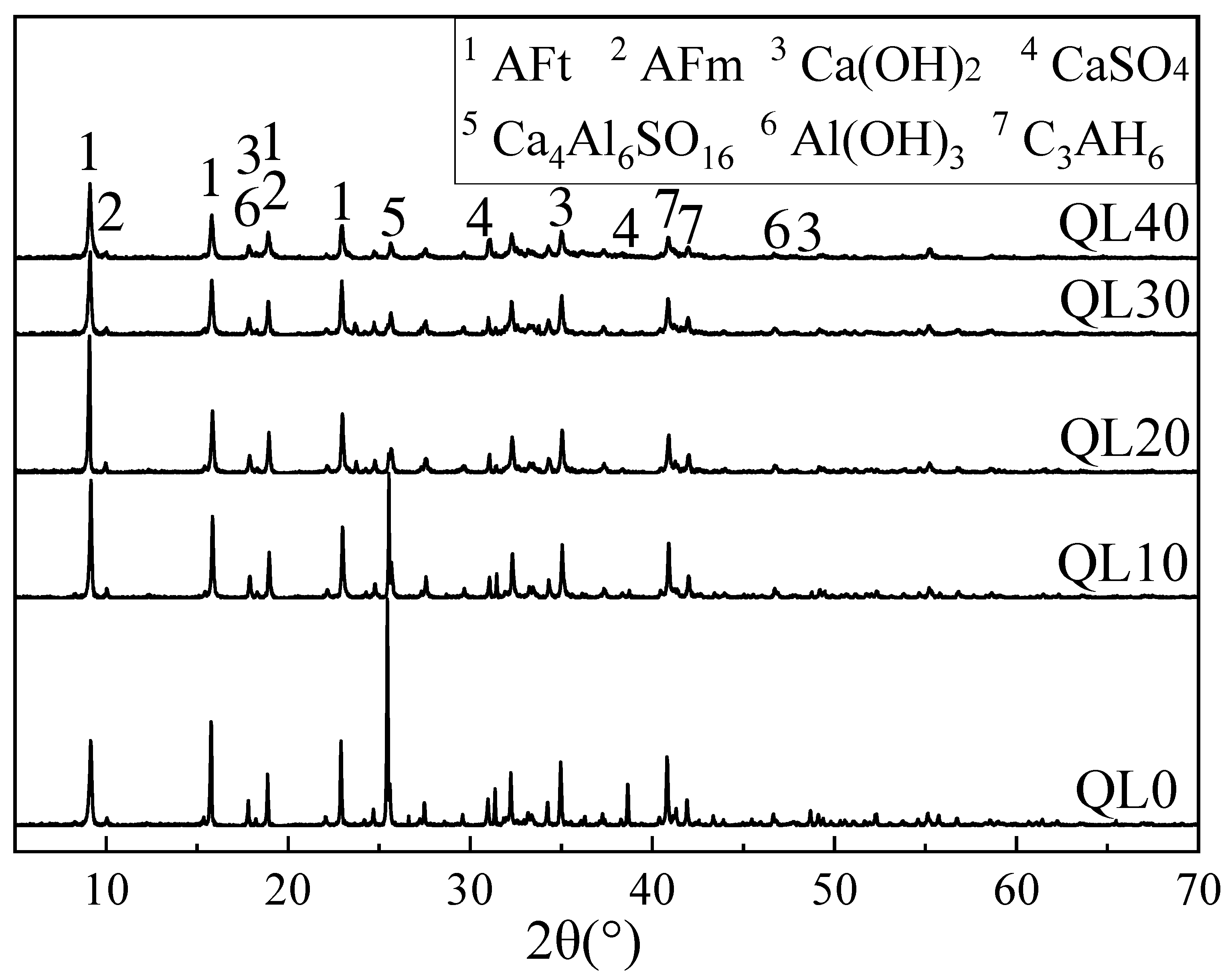
3.4. XRD Results
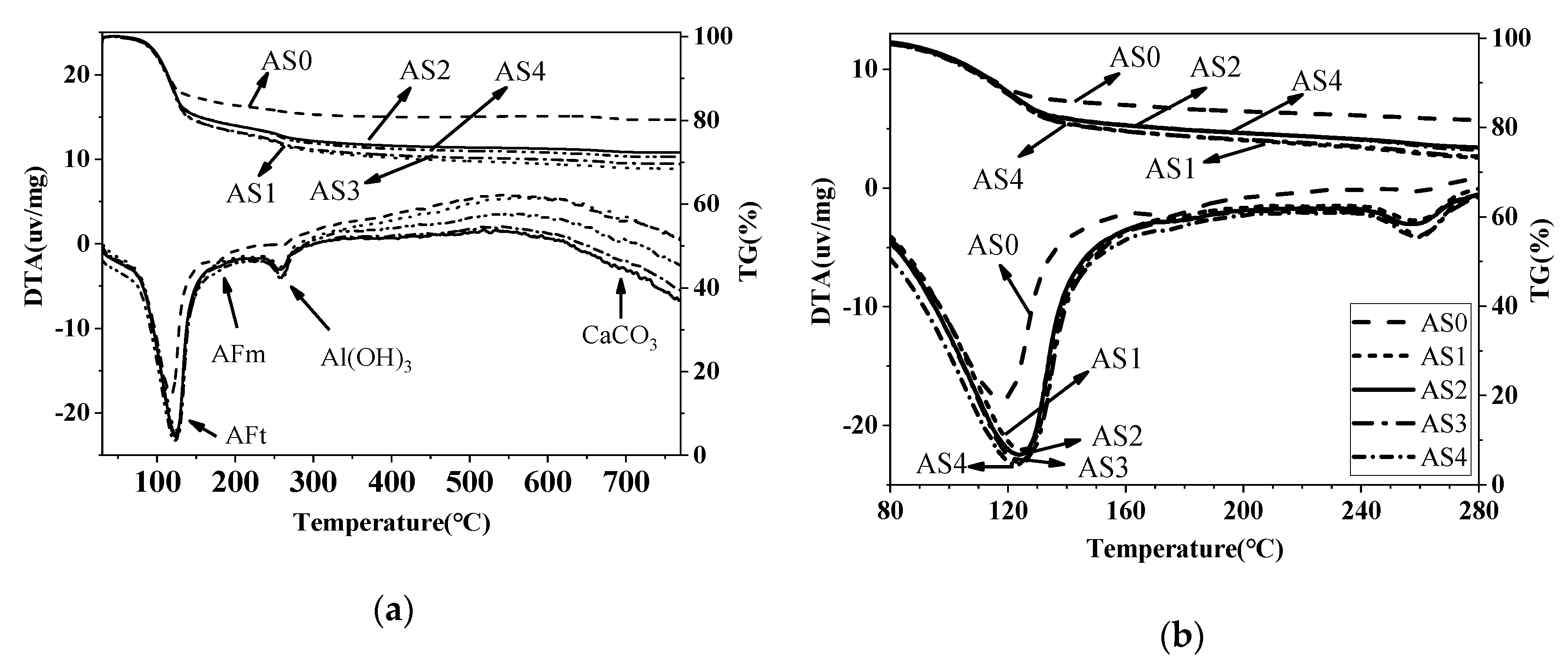
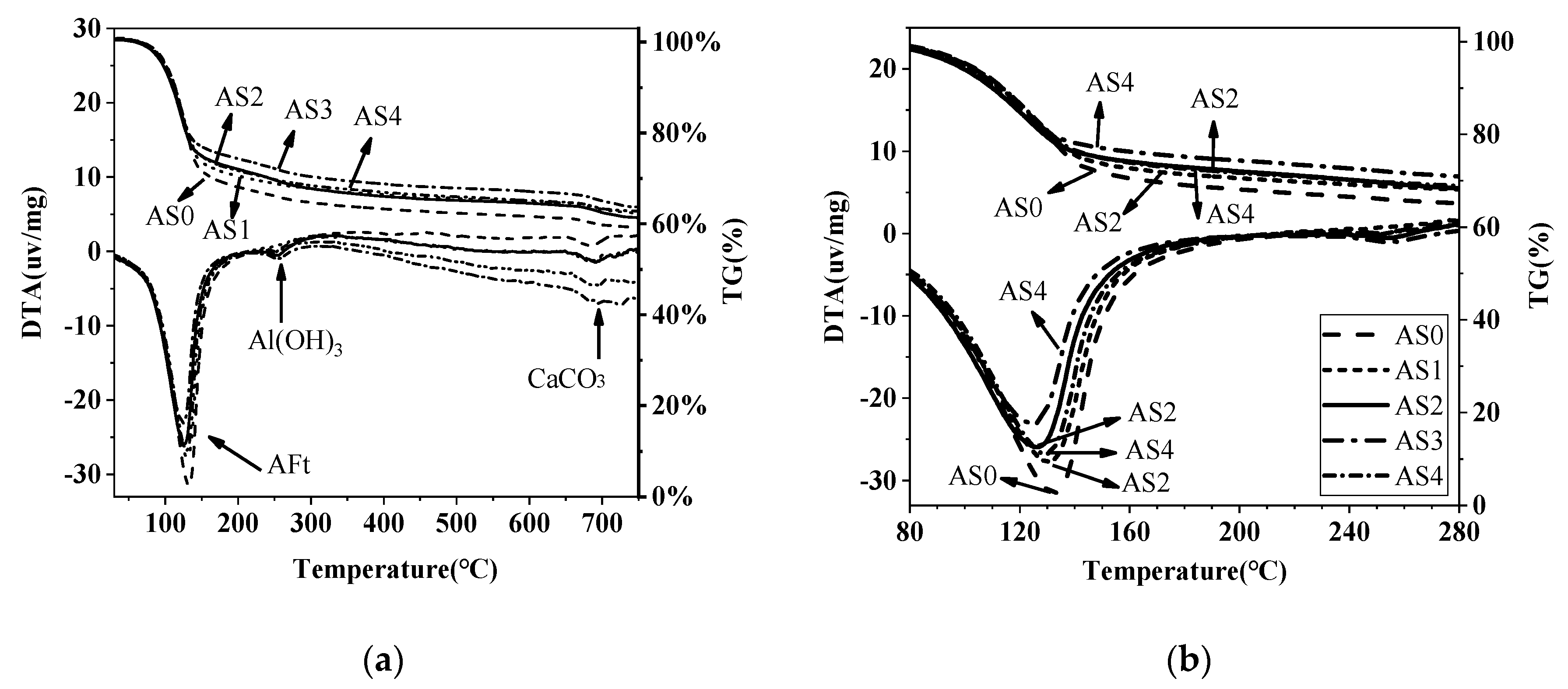
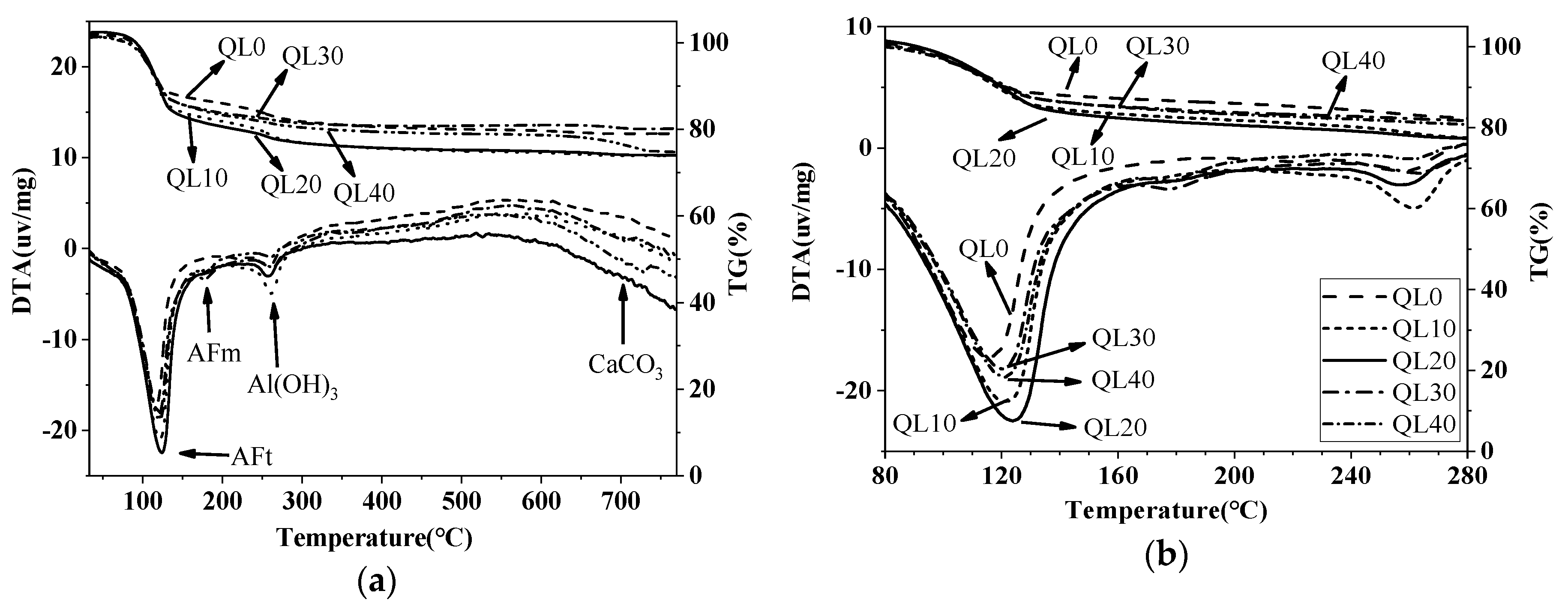
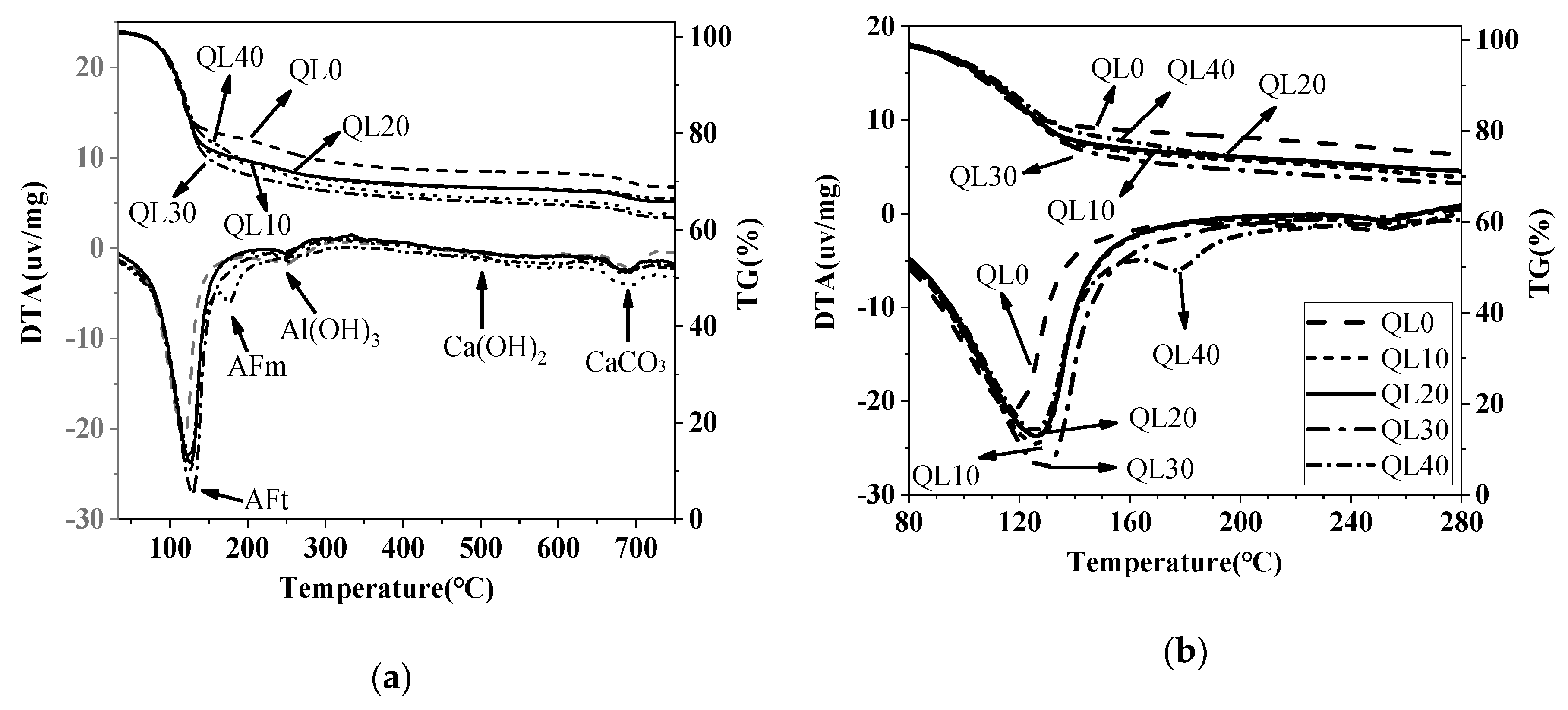
3.5. DTA-TG Results
3.6. SEM Results
4. Discussion
4.1. Optimum Ratio of Quicklime/Fluorgypsum
→ 3(3CaO∙Al2O3∙3CaSO4∙32 H2O)
4.2. Optimum Content of Aluminum Sulfate
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Seo, H.J.; Choi, H.; Lee, I.M. Numerical and experimental investigation of pillar reinforcement with pressurized grouting and pre-stress. Tunn. Undergr. Space Technol. 2016, 54, 135–144. [Google Scholar] [CrossRef]
- Juenger, M.C.G.; Winnefeld, F.; Provis, J.L.; Ideker, J.H. Advances in alternative cementitious binders. Cem. Concr. Res. 2011, 41, 1232–1243. [Google Scholar] [CrossRef]
- Cabrera, J.G.; Al-Hasan, A.S. Performance properties of concrete repair materials. Constr. Build. Mater. 1997, 11, 283–290. [Google Scholar] [CrossRef]
- Zhang, J.; Guan, X.; Li, H.; Liu, X. Performance and hydration study of ultra-fine sulfoaluminate cement-based double liquid grouting material. Constr. Build. Mater. 2017, 132, 262–270. [Google Scholar] [CrossRef]
- Nonveiller, E. Grouting Theory and Practice, 1st ed.; Elsevier: New York, NY, USA, 1989. [Google Scholar]
- Kasselouri, V.; Tsakiridis, P.; Malami, C.; Georgali, B.; Alexandridou, C. A study on the hydration products of a non-expansive sulfoaluminate cement. Cem. Concr. Res. 1995, 25, 1726–1736. [Google Scholar] [CrossRef]
- Péra, J.; Ambroise, J. New applications of calcium sulfoaluminate cement. Cem. Concr. Res. 2004, 34, 671–676. [Google Scholar] [CrossRef]
- García-Maté, M.; Londono-Zuluaga, D.; Torre, A.G.D.L.; Cabeza, A.; Aranda, M.A.G.; Santacruz, I. Tailored setting times with high compressive strengths in bassanite calcium sulfoaluminate eco-cements. Cem. Concr. Compos. 2016, 72, 39–47. [Google Scholar] [CrossRef]
- Coumes, C.C.D.; Dhoury, M.; Champenois, J.B.; Mercier, C.; Damidot, D. Physico-chemical mechanisms involved in the acceleration of the hydration of calcium sulfoaluminate cement by lithium ions. Cem. Concr. Res. 2017, 96, 42–51. [Google Scholar] [CrossRef]
- Coumes, C.C.D.; Dhoury, M.; Champenois, J.B.; Mercier, C.; Damidot, D. Combined effects of lithium and borate ions on the hydration of calcium sulfoaluminate cement. Cem. Concr. Res. 2017, 97, 50–60. [Google Scholar] [CrossRef]
- Zhang, G.; Li, G.; Li, Y. Effects of superplasticizers and retarders on the fluidity and strength of sulphoaluminate cement. Constr. Build. Mater. 2016, 126, 44–54. [Google Scholar] [CrossRef]
- Tan, H.; Guo, Y.; Zou, F.; Jian, S.; Ma, B.; Zhi, Z. Effect of borax on rheology of calcium sulphoaluminate cement paste in the presence of polycarboxylate superplasticizer. Constr. Build. Mater. 2017, 139, 277–285. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, Y.; Li, T.; Xiong, Z.; Sun, Y. Effects of lithium carbonate on performances of sulphoaluminate cement-based dual liquid high water material and its mechanisms. Constr. Build. Mater. 2018, 161, 374–380. [Google Scholar] [CrossRef]
- Guan, X.; Li, H.; Luo, S.; Liu, X.; Zhang, J. Influence of LiAl-layered double hydroxides with 3D micro-nano structures on the properties of calcium sulphoaluminate cement clinker. Cem. Concr. Compos. 2016, 70, 15–23. [Google Scholar] [CrossRef]
- Rashad, A.M. Effects of ZnO2, ZrO2, Cu2O3, CuO, CaCO3, SF, FA, cement and geothermal silica waste nanoparticles on properties of cementitious materials—A short guide for Civil Engineer. Constr. Build. Mater. 2013, 48, 1120–1133. [Google Scholar] [CrossRef]
- Li, W.; Huang, Z.; Zu, T.; Shi, C.; Duan, W.H.; Shah, S.P. Influence of nanolimestone on the hydration, mechanical strength, and autogenous shrinkage of ultrahigh-performance concrete. J. Mater. Civ. Eng. 2016, 28, 04015068. [Google Scholar] [CrossRef]
- Meng, T.; Yu, Y.; Qian, X.; Zhan, S.; Qian, K. Effect of nano-TiO2 on the mechanical properties of cement mortar. Constr. Build. Mater. 2012, 29, 241–245. [Google Scholar] [CrossRef]
- Li, W.; Huang, Z.; Cao, F.; Sun, Z.; Shah, S.P. Effects of nano-silica and nano-limestone on flowability and mechanical properties of ultra-high-performance concrete matrix. Constr. Build. Mater. 2015, 95, 366–374. [Google Scholar] [CrossRef]
- Morsy, M.S.; Alsayed, S.H.; Aqel, M. Hybrid effect of carbon nanotube and nano-clay on physico-mechanical properties of cement mortar. Constr. Build. Mater. 2011, 25, 145–149. [Google Scholar] [CrossRef]
- Clark, S.M.; Colas, B.; Kunz, M.; Speziale, S.; Monteiro, P.J.M. Effect of pressure on the crystal structure of ettringite. Cem. Concr. Res. 2008, 38, 19–26. [Google Scholar] [CrossRef]
- Adams, L.D. Ettringite, the positive side. Proc. Int. Cem. Microsc. Assoc. 1997, 19, 1–13. [Google Scholar]
- Li, H.; Guan, X.; Zhang, X.; Ge, P.; Hu, X.; Zou, D. Influence of superfine ettringite on the properties of sulphoaluminate cement-based grouting materials. Constr. Build. Mater. 2018, 166, 723–731. [Google Scholar] [CrossRef]
- Fraire-Luna, P.E.; Escalante-Garcia, J.I.; Gorokhovsky, A. Composite systems fluorgypsum–blastfurnance slag–metakaolin, strength and microstructures. Cem. Concr. Res. 2006, 36, 1048–1055. [Google Scholar] [CrossRef]
- Yan, P.; Yang, W.; Qin, X.; You, Y. Microstructure and properties of the binder of fly ash-fluorogypsum-Portland cement. Cem. Concr. Res. 1999, 29, 349–354. [Google Scholar] [CrossRef]
- Xu, L.L.; Wang, P.M.; Wu, G.M.; Zhang, G.F. Effect of calcium sulfate on the formation of ettringite in calcium aluminate and sulfoaluminate blended systems. Key Eng. Mater. 2014, 599, 23–28. [Google Scholar] [CrossRef]
- Palou, M.T.; Majling, J. Effects of sulphate, calcium and aluminum ions upon the hydration of sulphoaluminate belite cement. J. Therm. Anal. Calorim. 1996, 46, 549–556. [Google Scholar] [CrossRef]
- Wang, J.; Liu, E.; Li, L. Characterization on the recycling of waste seashells with Portland cement towards sustainable cementitious materials. J. Clean. Prod. 2019, 220, 235–252. [Google Scholar] [CrossRef]
- Xie, J.; Wang, J.; Zhang, B.; Fang, C.; Li, L. Physicochemical properties of alkali activated GGBS and fly ash geopolymeric recycled concrete. Constr. Build. Mater. 2019, 204, 384–398. [Google Scholar] [CrossRef]
- Xie, J.; Wang, J.; Wang, C.; Fang, C. Effects of combined usage of GGBS and fly ash on workability and mechanical properties of alkali activated geopolymer concrete with recycled aggregate. Compos. B Eng. 2019, 164, 179–190. [Google Scholar] [CrossRef]
- Xie, J.; Huang, L.; Guo, Y.; Li, Z.; Fang, C.; Li, L.; Wang, J. Experimental study on the compressive and flexural behaviour of recycled aggregate concrete modified with silica fume and fibers. Constr. Build. Mater. 2018, 178, 612–623. [Google Scholar] [CrossRef]
- Wang, H.; Sun, X.; Wang, J.; Monteiro, P.J.M. Permeability of concrete with recycled concrete aggregate and pozzolanic materials under stress. Materials 2016, 9, 252. [Google Scholar] [CrossRef]
- Wang, H.; Wang, J.; Sun, X.; Jin, W. Improving performance of recycled aggregate concrete with superfine pozzolanic powders. J. Cent. South Univ. 2013, 20, 3715–3722. [Google Scholar] [CrossRef]
- Wang, L.; Yu, K.Q.; Li, J.; Tsang, D.C.W.; Poon, C.S.; Yoo, J.C.; Baek, K.; Ding, S.M.; Hou, D.Y.; Dai, J.G. Low-carbon and low-alkalinity stabilization/solidification of high-Pb contaminated soil. Chem. Eng. J. 2018, 351, 418–427. [Google Scholar] [CrossRef]
- Wang, L.; Chen, L.; Tsang, D.C.W.; Li, J.; Poon, C.S.; Baek, K.; Hou, D.Y.; Ding, S.M. Recycling dredged sediment into fill materials, partition blocks, and paving blocks: Technical and economic assessment. J. Clean. Prod. 2018, 199, 69–76. [Google Scholar] [CrossRef]
- Wang, J.; Mu, M.; Liu, Y. Recycled cement. Constr. Build. Mater. 2018, 190, 1124–1132. [Google Scholar] [CrossRef]
- He, Z.; Zhu, X.; Wang, J.; Mu, M.; Wang, Y. Comparison of CO2 emissions from OPC and recycled cement production. Constr. Build. Mater. 2019, 211, 965–973. [Google Scholar] [CrossRef]
- Chen, L.; Wang, L.; Cho, D.W.; Tsang, D.C.W.; Tong, L.Z.; Zhou, Y.Y.; Yang, J.; Hu, Q.; Poon, C.S. Sustainable stabilization/solidification of municipal solid waste incinerator fly ash by incorporation of green materials. J. Clean. Prod. 2019, 222, 335–343. [Google Scholar] [CrossRef]
- Wang, Y.; He, H.; He, F. Effect of slaked lime and aluminum sulfate on the properties of dry-mixed masonry mortar. Constr. Build. Mater. 2018, 180, 117–123. [Google Scholar] [CrossRef]
- GB/T 176-2017: Methods for Chemical Analysis of Cement; Standardization Administration of China: Beijing, China, 2017.
- ASTM. C191-13: Standard Test Methods for Time of Setting of Hydraulic Cement by Vicat Needle; American Society for Testing and Materials, Annual Book of ASTM Standards: New York, NY, USA, 2014. [Google Scholar]
- Martin, L.H.; Winnefeld, F.; Müller, C.J.; Lothenbach, B. Contribution of limestone to the hydration of calcium sulfoaluminate cement. Cem. Concr. Compos. 2015, 62, 204–211. [Google Scholar] [CrossRef]
- Winnefeld, F.; Barlag, S. Influence of calcium sulfate and calcium hydroxide on the hydration of calcium sulfoaluminate clinker. ZKG Int. 2009, 62, 42–53. [Google Scholar]
- Zhang, L.; Glasser, F.P. Hydration of calcium sulfoaluminate cement at less than 24 h. Adv. Cem. Res. 2002, 14, 141–155. [Google Scholar] [CrossRef]
- Taylor, H.F.W. Cement Chemistry, 2nd ed.; Academic Press: London, UK, 1997. [Google Scholar]
- Telesca, A.; Marroccoli, M.; Pace, M.L.; Tomasulo, M.; Valenti, G.L.; Monteiro, P.J.M. A hydration study of various calcium sulfoaluminate cements. Cem. Concr. Compos. 2014, 53, 224–232. [Google Scholar] [CrossRef]
- Paul, S.C.; van Rooyen, A.S.; van Zijl, G.P.A.G.; Petrik, L.F. Properties of cement-based composites using nanoparticles: A comprehensive review. Constr. Build. Mater. 2018, 189, 1019–1034. [Google Scholar] [CrossRef]




| SO3 | SiO2 | CaO | Al2O3 | Fe2O3 | MgO | LOI |
|---|---|---|---|---|---|---|
| 9.44 | 8.59 | 45.21 | 31.68 | 3.86 | 0.82 | 0.4 |
| Ca4Al6SO16 | 2CaO∙SiO2 | C4AF | f-SO3 | f-CaO |
|---|---|---|---|---|
| 64.12 | 25.53 | 6.41 | 1.92 | 2.02 |
| SO3 | SiO2 | Al2O3 | Fe2O3 | CaO | MgO | CaF2 | LOI |
|---|---|---|---|---|---|---|---|
| 53.79 | 4.71 | 2.6 | 0.24 | 34.56 | 1.15 | 0.96 | 1.99 |
| Mix No. | Slurry A 1 | Slurry B 2 | ||||
|---|---|---|---|---|---|---|
| CSA 3 (wt.%) | Water (wt.%) | Fluorgypsum (wt.%) | Quicklime (wt.%) | Aluminum Sulfate (wt.%) | Water (wt.%) | |
| AS0 | 100 | 100 | 80 | 20 | 0 | 100 |
| AS1 | 100 | 100 | 80 | 20 | 1 | 100 |
| AS2 | 100 | 100 | 80 | 20 | 2 | 100 |
| AS3 | 100 | 100 | 80 | 20 | 3 | 100 |
| AS4 | 100 | 100 | 80 | 20 | 4 | 100 |
| QL0 | 100 | 100 | 100 | 0 | 2 | 100 |
| QL10 | 100 | 100 | 90 | 10 | 2 | 100 |
| QL20 | 100 | 100 | 80 | 20 | 2 | 100 |
| QL30 | 100 | 100 | 70 | 30 | 2 | 100 |
| QL40 | 100 | 100 | 60 | 40 | 2 | 100 |
| Slurry A | Initial Setting Time (min) | Final Setting Time (min) | ||
|---|---|---|---|---|
| Aluminum Sulfate (wt.%) | CSA (wt.%) | Water (wt.%) | ||
| 0 | 100 | 100 | 491 | 619 |
| 1 | 100 | 100 | 489 | 614 |
| 2 | 100 | 100 | 485 | 607 |
| 3 | 100 | 100 | 486 | 611 |
| 4 | 100 | 100 | 479 | 599 |
| Mix No. | 1 Day Weight Loss (%) | 28 Days Weight Loss (%) | ||||
|---|---|---|---|---|---|---|
| AFt | AFm | Al(OH)3 | AFt | AFm | Al(OH)3 | |
| AS0 | 14.45 | 1.82 | 1.76 | 28.88 | (3.60) | 2.61 |
| AS1 | 18.75 | (2.37) * | 3.58 | 26.86 | (3.11) | 2.15 |
| AS2 | 18.99 | (2.34) | 3.00 | 23.70 | (2.54) | 2.75 |
| AS3 | 19.93 | (2.85) | 3.66 | 23.67 | (2.66) | 3.16 |
| AS4 | 20.22 | (2.46) | 3.56 | 25.65 | (3.17) | 2.47 |
| Mix No. | 1d Weight Loss (%) | 28d Weight Loss (%) | ||||
|---|---|---|---|---|---|---|
| AFt | AFm | Al(OH)3 | AFt | AFm | Al(OH)3 | |
| QL0 | 14.24 | (1.59) * | 3.49 | 20.10 | (1.90) | 3.54 |
| QL10 | 17.35 | (2.11) | 4.19 | 24.38 | (2.56) | 3.52 |
| QL20 | 18.99 | (2.34) | 3.00 | 23.70 | (2.54) | 2.75 |
| QL30 | 16.45 | 2.02 | 3.16 | 26.11 | (3.33) | (2.49) |
| QL40 | 15.61 | 2.34 | 2.33 | 22.19 | 4.32 | (2.77) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Yu, J.; Wang, J.; Guan, X. Effects of Aluminum Sulfate and Quicklime/Fluorgypsum Ratio on the Properties of Calcium Sulfoaluminate (CSA) Cement-Based Double Liquid Grouting Materials. Materials 2019, 12, 1222. https://doi.org/10.3390/ma12081222
Wang Y, Yu J, Wang J, Guan X. Effects of Aluminum Sulfate and Quicklime/Fluorgypsum Ratio on the Properties of Calcium Sulfoaluminate (CSA) Cement-Based Double Liquid Grouting Materials. Materials. 2019; 12(8):1222. https://doi.org/10.3390/ma12081222
Chicago/Turabian StyleWang, Yuli, Jie Yu, Junjie Wang, and Xuemao Guan. 2019. "Effects of Aluminum Sulfate and Quicklime/Fluorgypsum Ratio on the Properties of Calcium Sulfoaluminate (CSA) Cement-Based Double Liquid Grouting Materials" Materials 12, no. 8: 1222. https://doi.org/10.3390/ma12081222
APA StyleWang, Y., Yu, J., Wang, J., & Guan, X. (2019). Effects of Aluminum Sulfate and Quicklime/Fluorgypsum Ratio on the Properties of Calcium Sulfoaluminate (CSA) Cement-Based Double Liquid Grouting Materials. Materials, 12(8), 1222. https://doi.org/10.3390/ma12081222






