Gd/Sm-Pr Co-Doped Ceria: A First Report of the Precipitation Method Effect on Flash Sintering
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
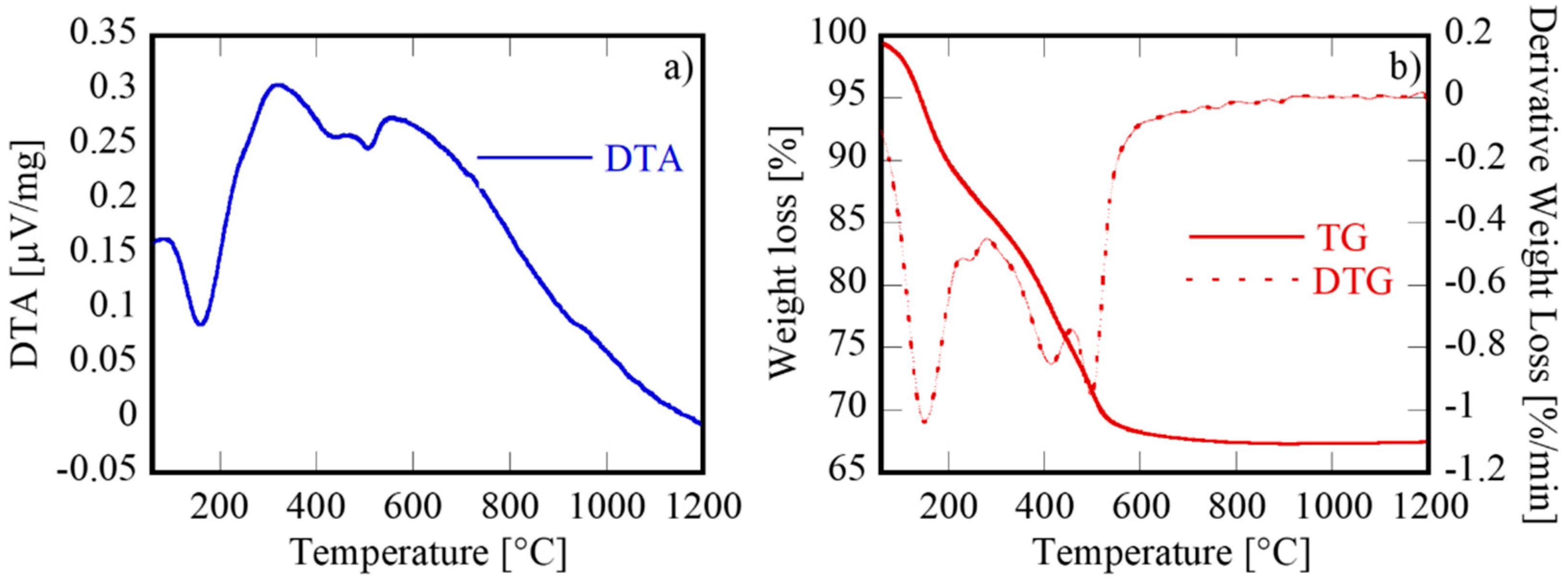
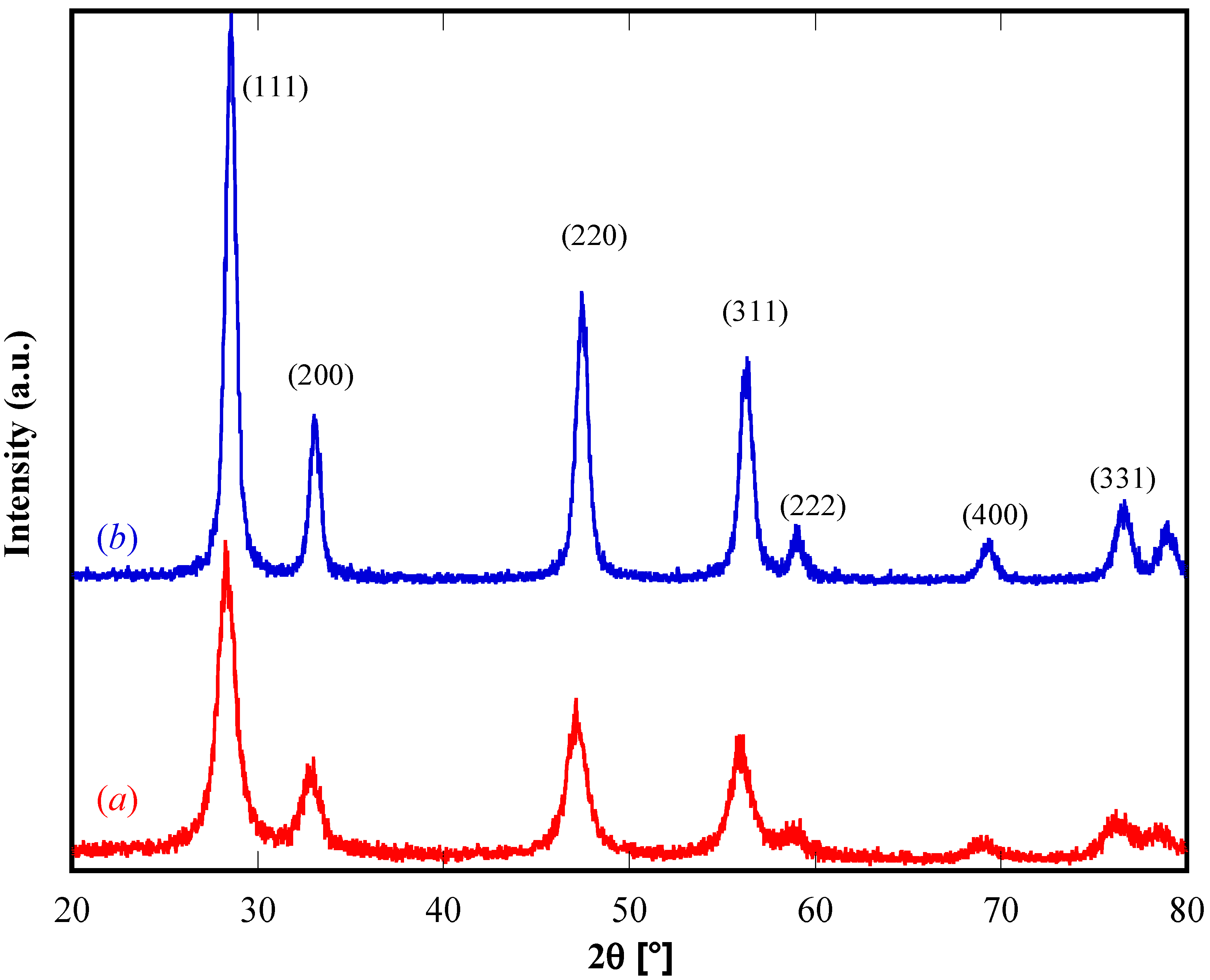
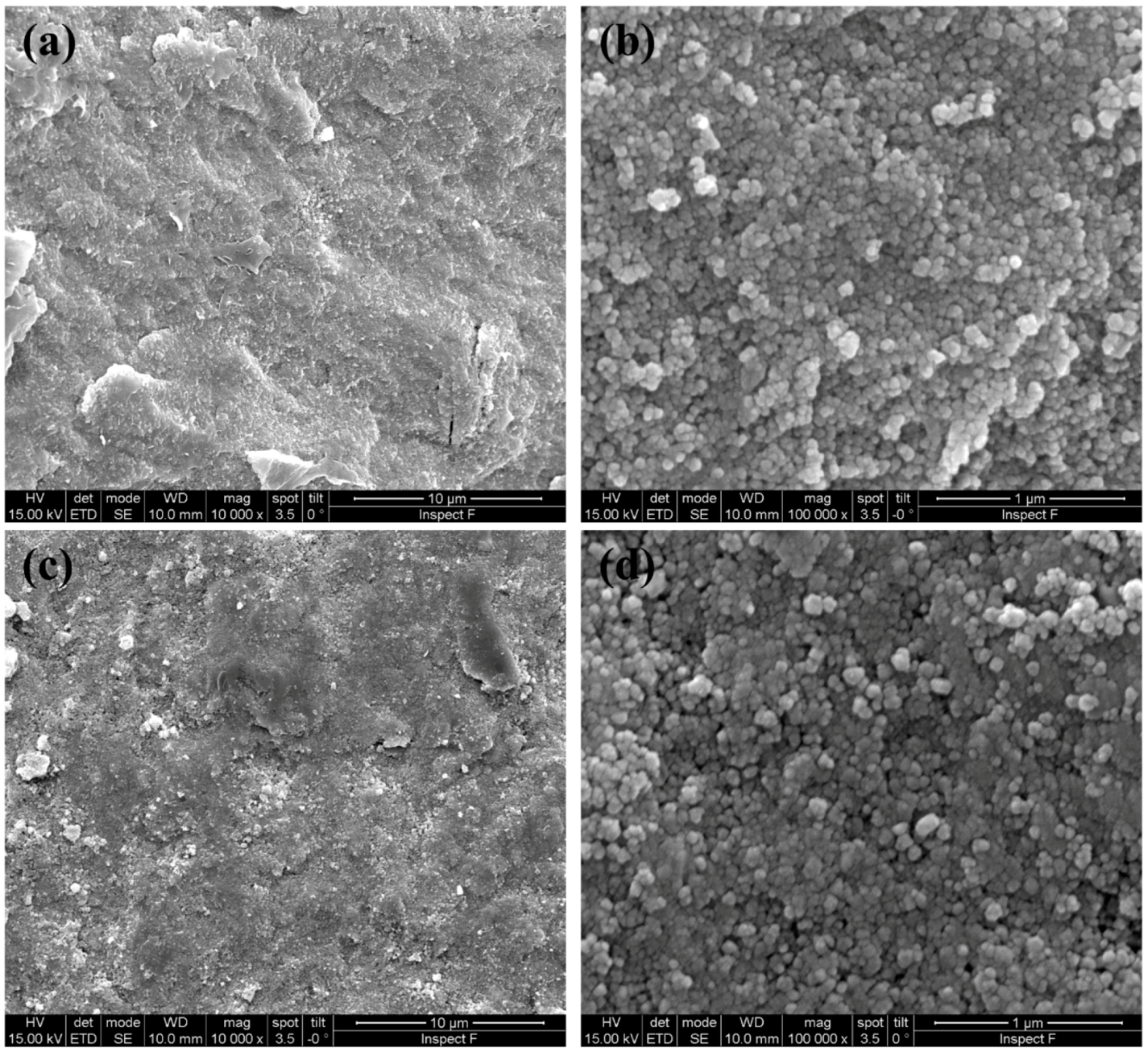
3.1. Characterization of the Samples
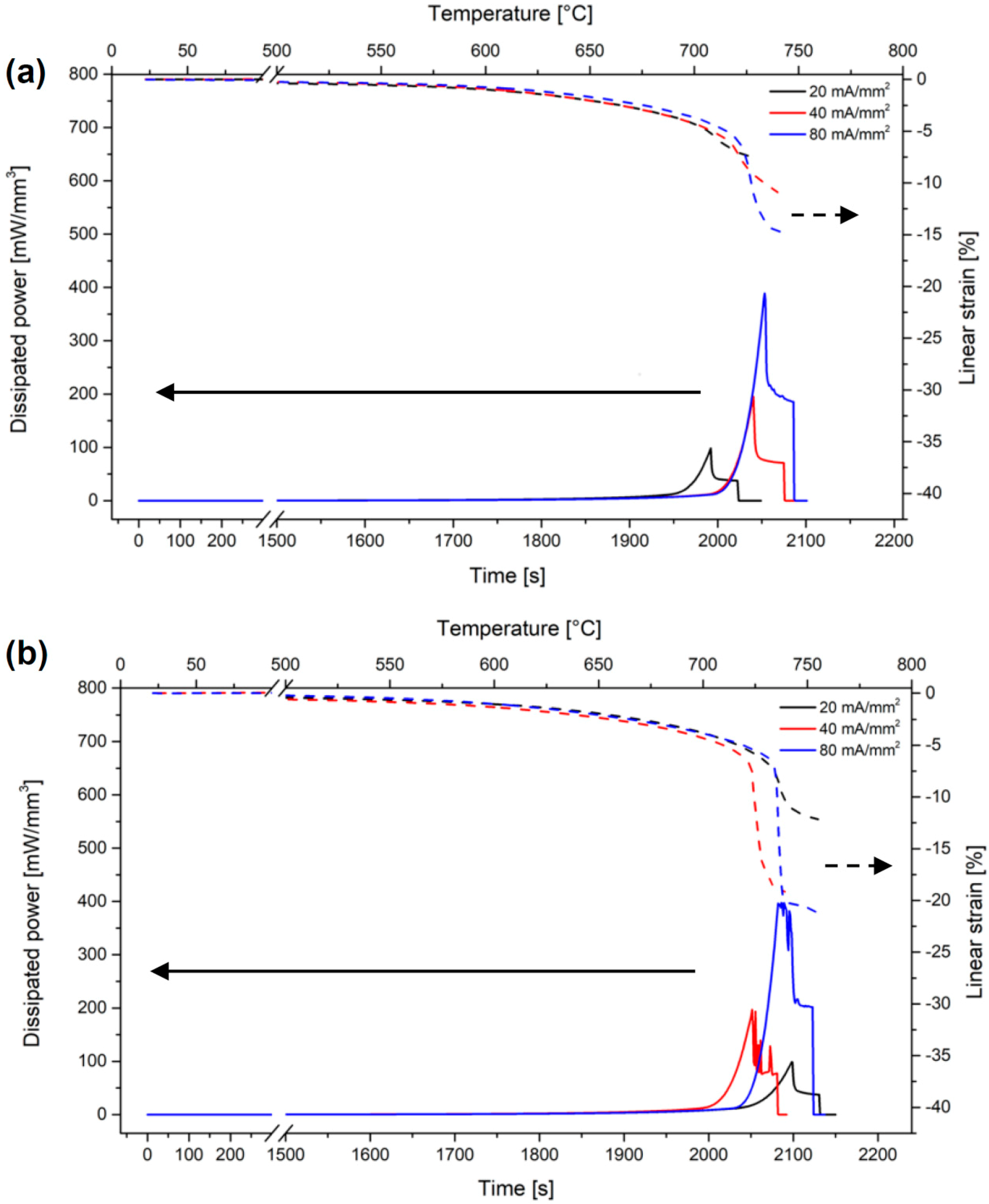
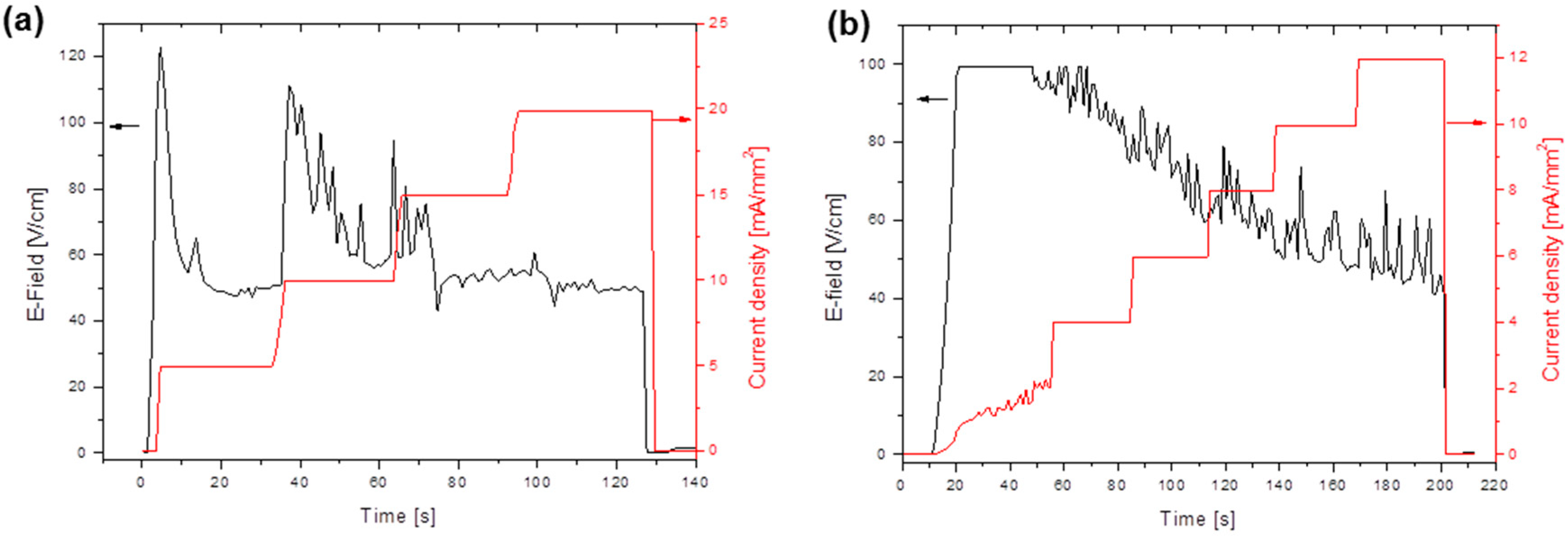
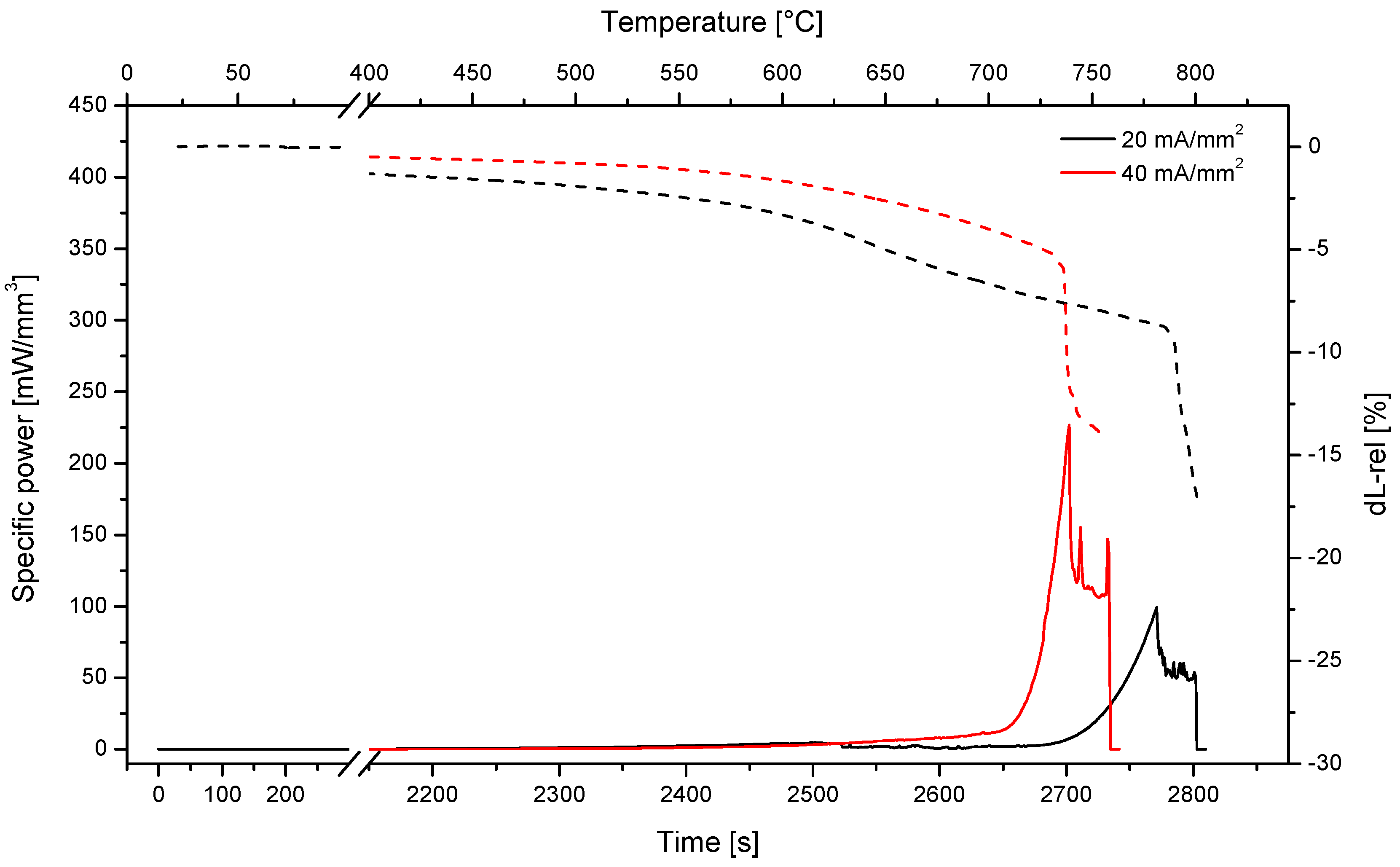
3.2. Flash Sintering
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hao, X.; Liu, Y.; Wang, Z.; Qiao, J.; Sun, K. A novel sintering method to obtain fully dense gadolinia doped ceria by applying a direct current. J. Power Source 2012, 210, 86–91. [Google Scholar] [CrossRef]
- Jaiswala, N.; Tanwarb, K.; Sumana, R.; Uppadhyac, D.K.S.; Parkash, O. A Brief Review on Ceria Based Solid Electrolytes for Solid Oxide Fuel Cells. J. Alloys Compd. 2019, 781, 984–1005. [Google Scholar] [CrossRef]
- Santos, T.H.; Grilo, J.P.F.; Loureiro, F.J.A.; Fagg, D.P.; Fonseca, F.C.; Macedo, D.A. Structure, densification and electrical properties of Gd3+ and Cu2+ co-doped ceria solid electrolytes for SOFC applications: Effects of Gd2O3 content. Ceram. Int. 2018, 44, 2745–2751. [Google Scholar] [CrossRef]
- Anwar, M.; Muhammad Ali, S.A.; Muchtar, A.; Somalu, M.R. Influence of strontium co-doping on the structural, optical, and electrical properties of erbium-doped ceria electrolyte for intermediate temperature solid oxide fuel cells. Ceram. Int. 2019, 45, 5627–5636. [Google Scholar] [CrossRef]
- Liu, Y.; Mushtaq, M.N.; Zhang, W.; Teng, A.; Liu, X. Single-phase electronic-ionic conducting Sm3+/Pr3+/Nd3+ triple-doped ceria for new generation fuel cell technology. Int. J. Hydrog. Energy 2018, 43, 12817–12824. [Google Scholar] [CrossRef]
- Bowman, W.J.; Zhu, J.; Sharma, R.; Crozier, P.A. Electrical conductivity and grain boundary composition of Gd-doped and Gd/Pr co-doped ceria. Solid State Ion. 2015, 272, 9–17. [Google Scholar] [CrossRef]
- Spiridigliozzi, L.; Dell’Agli, G.; Accardo, G.; Yoon, S.P.; Frattini, D. Electro-morphological, structural, thermal and ionic conduction properties of Gd/Pr co-doped ceria electrolytes exhibiting mixed Pr3+/Pr4+ cations. Ceram. Int. 2019, 45, 4570–4580. [Google Scholar] [CrossRef]
- Spiridigliozzi, L. Doped Ceria Electrolytes: Synthesis Methods. In Doped-Ceria Electrolytes; SpringerNature: Cham, Switzerland, 2018; pp. 25–55. ISBN 978-3-319-99394-2. [Google Scholar]
- Spiridigliozzi, L.; Dell’Agli, G.; Marocco, A.; Accardo, G.; Pansini, M.; Kwon, Y.; Yoon, S.P.; Frattini, D. Engineered co-precipitation chemistry with ammonium carbonate for scalable synthesis and sintering of improved Sm0.2Ce0.8O1.90 and Gd0.16Pr0.04Ce0.8O1.90 electrolytes for IT-SOFCs. J. Ind. Eng. Chem. 2018, 59, 17–27. [Google Scholar] [CrossRef]
- Accardo, G.; Spiridigliozzi, L.; Cioffi, R.; Ferone, C.; Di Bartolomeo, E.; Yoon, S.; Dell’Agli, G. Gadolinium-doped ceria nanopowders synthesized by Urea-Based Homogeneous co-Precipitation (UBHP). Mater. Chem. Phys. 2017, 187, 149–155. [Google Scholar] [CrossRef]
- Joh, D.W.; Rath, M.K.; Park, W.J.; Park, H.J.; Cho, H.K.; Lee, S.; Yoon, J.K.; Lee, J.H.; Lee, T.K. Sintering behavior and electrochemical performances of nano-sized gadolinium-doped ceria via ammonium carbonate assisted co-precipitation for solid oxide fuel cells. J. Alloys Compd. 2016, 682, 188–195. [Google Scholar] [CrossRef]
- Robert, C.L.; Long, J.W.; Lucas, E.M.; Pettigrew, K.A.; Stroud, R.M.; Doescher, M.S.; Rolison, D.R. Sol-Gel-Derived Ceria Nanoarchitectures: Synthesis, Characterization and Electrical Properties. Chem. Mater. 2006, 18, 50–58. [Google Scholar] [CrossRef]
- Dell’Agli, G.; Spiridigliozzi, L.; Marocco, A.; Accardo, G.; Frattini, D.; Kwon, Y.; Yoon, S.P. Morphological and crystalline evolution of Sm-(20 mol%)–doped ceria nanopowders prepared by a combined co-precipitation/hydrothermal synthesis for solid oxide fuel cell applications. Ceram. Int. 2017, 43, 12799–12808. [Google Scholar] [CrossRef]
- Dell’Agli, G.; Spiridigliozzi, L.; Pansini, M.; Accardo, G.; Yoon, S.P.; Frattini, D. Effect of the carbonate environment on morphology and sintering behaviour of variously co-doped (Ca, Sr, Er, Pr) Samarium-doped Ceria in co-precipitation/hydrothermal synthesis. Ceram. Int. 2018, 44, 17935–17944. [Google Scholar] [CrossRef]
- Zhang, X.; Deces-Petit, C.; Yick, S.; Robertson, M.; Kesler, O.; Maric, R.; Ghosh, D. A study on sintering aids for Sm0.2Ce0.8O1.9 electrolyte. J. Power Sources 2006, 162, 480–485. [Google Scholar] [CrossRef]
- Biesuz, M.; Spiridigliozzi, L.; Frasnelli, M.; Dell’Agli, G.; Sglavo, V.M. Rapid densification of Samarium-doped Ceria ceramic with nanometric grain size at 900–1100 °C. Mater. Lett. 2017, 190, 17–19. [Google Scholar] [CrossRef]
- Charoonsuk, T.; Sukkha, U.; Kolodiazhnyi, T.; Vittayakorn, N. Enhancing the densification of ceria ceramic at low temperature via the cold sintering assisted two-step sintering process. Ceram. Int. 2018, 44, S54–S57. [Google Scholar] [CrossRef]
- Cologna, M.; Rashkova, B.; Raj, R. Flash sintering of nanograin zirconia in <5 s at 850 °C. J. Am. Ceram. Soc. 2010, 93, 3556–3559. [Google Scholar] [CrossRef]
- Biesuz, M.; Sglavo, V.M. Flash sintering of ceramics. J. Eur. Ceram. Soc. 2019, 39, 115–143. [Google Scholar] [CrossRef]
- Becker, M.Z.; Shomrat, N.; Tsur, Y. Recent Advances in Mechanism Research and Methods for Electric-Field-Assisted Sintering of Ceramics. Adv. Mater. 2018, 30, 1706369. [Google Scholar] [CrossRef]
- Todd, R.I.; Zapata-Solvas, E.; Bonilla, R.S.; Sneddon, T.; Wilshaw, P.R. Electrical characteristics of flash sintering: Thermal runaway of Joule heating. J. Eur. Ceram. Soc. 2015, 35, 1865–1877. [Google Scholar] [CrossRef]
- da Silva, J.G.P.; Al-Qureshi, H.A.; Keil, F.; Janssen, R. A dynamic bifurcation criterion for thermal runaway during the flash sintering of ceramics. J. Eur. Ceram. Soc. 2016, 36, 1261–1267. [Google Scholar] [CrossRef]
- Dong, Y. On the Hotspot Problem in Flash Sintering; Department of Materials Science and Engineering, University of Pennsylvania: Philadelphia, PA, USA, 2017. [Google Scholar]
- Charalambous, H.; Jha, S.K.; Christian, K.; Lay, R.; Tsakalakos, T. Flash Sintering using Controlled Current Ramp. J. Eur. Ceram. Soc. 2018, 38, 3689–3693. [Google Scholar] [CrossRef]
- Kumar, P.; Yadav, D.; Lebrun, J.; Raj, R. Flash Sintering with Current-Rate: A Different Approach Punith. J. Am. Ceram. Soc. 2019, 102, 823–835. [Google Scholar] [CrossRef]
- Campos, J.V.; Lavagnini, I.R.; de Sousa, R.V.; Ferreira, J.A.; de Pallone, E.M. Development of an instrumented and automated flash sintering setup for enhanced process monitoring and parameter control. J. Eur. Ceram. Soc. 2019, 39, 531–538. [Google Scholar] [CrossRef]
- Biesuz, M.; Dell’Agli, G.; Spiridigliozzi, L.; Ferone, C.; Sglavo, V.M. Conventional and Field-Assisted Sintering of Nanosized Gd-doped Ceria Synthesized by Co-precipitation. Ceram. Int. 2016, 42, 11766–11771. [Google Scholar] [CrossRef]
- Spiridigliozzi, L.; Biesuz, M.; Dell’Agli, G.; Di Bartolomeo, E.; Zurlo, F.; Sglavo, V.M. Microstructural and electrical investigation of flash-sintered Gd/Sm-doped ceria. J. Mater. Sci. 2017, 52, 7479–7488. [Google Scholar] [CrossRef]
- Li, J.; Guan, W.; Luo, M.; Song, J.; Song, X.; An, S. Sintering behavior of samarium doped ceria under DC electrical field. Ceram. Int. 2018, 44, 2470–2477. [Google Scholar] [CrossRef]
- Jiang, T.; Wang, Z.; Zhang, J.; Hao, X.; Rooney, D.; Liu, Y.; Sun, W.; Qiao, J.; Sun, K. Understanding the Flash Sintering of Rare-Earth-Doped Ceria for Solid Oxide Fuel Cell. J. Am. Ceram. Soc. 2015, 98, 1717–1723. [Google Scholar] [CrossRef]
- Kim, P.; Anderko, A.; Navrotsky, A.; Riman, R.E. Trends in Structure and Thermodynamic Properties of Normal Rare Earth Carbonates and Rare Earth Hydroxycarbonates. Minerals 2018, 8, 106. [Google Scholar] [CrossRef]
- Accardo, G.; Dell’Agli, G.; Mascolo, M.C.; Spiridigliozzi, L.; Yoon, S.P. Controlled Coprecipitation of Amorphous Cerium-Based Carbonates with Suitable Morphology as Precursors of Ceramic Electrolytes for IT-SOFCs. Materials 2019, 12, 702. [Google Scholar] [CrossRef]
- Li, J.-G.; Ikegami, T.; Wang, Y.; Mori, T. Reactive Ceria Nanopowders via carbonate precipitation. J. Am. Ceram. Soc. 2002, 85, 2376–2378. [Google Scholar] [CrossRef]
- Spiridigliozzi, L.; Dell’Agli, G.; Biesuz, M.; Sglavo, V.M.; Pansini, M. Effect of the Precipitating Agent on the Synthesis and Sintering Behaviour of 20 mol% Sm-doped Ceria. Adv. Mater. Sci. Eng. 2016, 6096123. [Google Scholar] [CrossRef]
- Lutterotti, L.; Bortolotti, M.; Ischia, G.; Lonardelli, I.; Wenk, H.R. Rietveld texture analysis from diffraction images. Z. Kristallogr. 2007, 26, 125–130. [Google Scholar] [CrossRef]
- Shannon, R.D. Revised Effective Ionic Radii and Systematic Studies of Interatomic Distances in Halides and Chalcogenides. Acta Cryst. 1976, A32, 751–767. [Google Scholar] [CrossRef]
- Nicholas, J.D.; De Jonghe, L.C. Prediction and Evaluation of Sintering Aids for Cerium Gadolinium Oxide. Solid State Ion. 2007, 178, 1187–1194. [Google Scholar] [CrossRef]



| Composition | Precipitating Agent | Flash Cycle | Electric Data | Relative Density [%] |
|---|---|---|---|---|
| 6PrSDC20 | (NH4)2CO3 | Traditional | 50V/cm, 20 mA/mm2 | 81 |
| 6PrSDC20 | (NH4)2CO3 | Traditional | 50V/cm, 40 mA/mm2 | 61 |
| 6PrSDC20 | (NH4)2CO3 | Traditional | 50V/cm, 80 mA/mm2 | 74 |
| 6PrGDC20 | (NH4)2CO3 | Traditional | 50V/cm, 20 mA/mm2 | 88 |
| 6PrGDC20 | (NH4)2CO3 | Traditional | 50V/cm, 40 mA/mm2 | 77 |
| 6PrGDC20 | (NH4)2CO3 | Traditional | 50V/cm, 80 mA/mm2 | 60 |
| GDC20 | (NH4)2CO3 | Traditional | 50V/cm, 15 mA/mm2 | 66 |
| GDC20 | (NH4)2CO3 | Traditional | 50V/cm, 10 mA/mm2 | 68 |
| SDC20 | (NH4)2CO3 | Traditional | 50V/cm, 15 mA/mm2 | 84 |
| 6PrSDC20 | (NH4)2CO3 | Ramp | 5 mA/mm2 steps up to 20 mA/mm2 | 91 |
| 6PrSDC20 | (NH4)2CO3 | Ramp | 2 mA/mm2 steps up to 12 mA/mm2 | 88 |
| 6PrGDC20 | (NH4)2CO3 | Ramp | 2 mA/mm2 steps up to 12 mA/mm2 | 93 |
| 6PrSDC20 | NH3 | Traditional | 50V/cm, 20 mA/mm2 | 94 |
| 6PrSDC20 | NH3 | Traditional | 50V/cm, 40 mA/mm2 | 93 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spiridigliozzi, L.; Pinter, L.; Biesuz, M.; Dell’Agli, G.; Accardo, G.; Sglavo, V.M. Gd/Sm-Pr Co-Doped Ceria: A First Report of the Precipitation Method Effect on Flash Sintering. Materials 2019, 12, 1218. https://doi.org/10.3390/ma12081218
Spiridigliozzi L, Pinter L, Biesuz M, Dell’Agli G, Accardo G, Sglavo VM. Gd/Sm-Pr Co-Doped Ceria: A First Report of the Precipitation Method Effect on Flash Sintering. Materials. 2019; 12(8):1218. https://doi.org/10.3390/ma12081218
Chicago/Turabian StyleSpiridigliozzi, Luca, Lorenzo Pinter, Mattia Biesuz, Gianfranco Dell’Agli, Grazia Accardo, and Vincenzo M. Sglavo. 2019. "Gd/Sm-Pr Co-Doped Ceria: A First Report of the Precipitation Method Effect on Flash Sintering" Materials 12, no. 8: 1218. https://doi.org/10.3390/ma12081218
APA StyleSpiridigliozzi, L., Pinter, L., Biesuz, M., Dell’Agli, G., Accardo, G., & Sglavo, V. M. (2019). Gd/Sm-Pr Co-Doped Ceria: A First Report of the Precipitation Method Effect on Flash Sintering. Materials, 12(8), 1218. https://doi.org/10.3390/ma12081218










