Rapid Fabrication of High-Performance CaO-Based Integral Ceramic Mould by Stereolithography and Non-Aqueous Gelcasting
Abstract
1. Introduction
2. Experimental Procedure
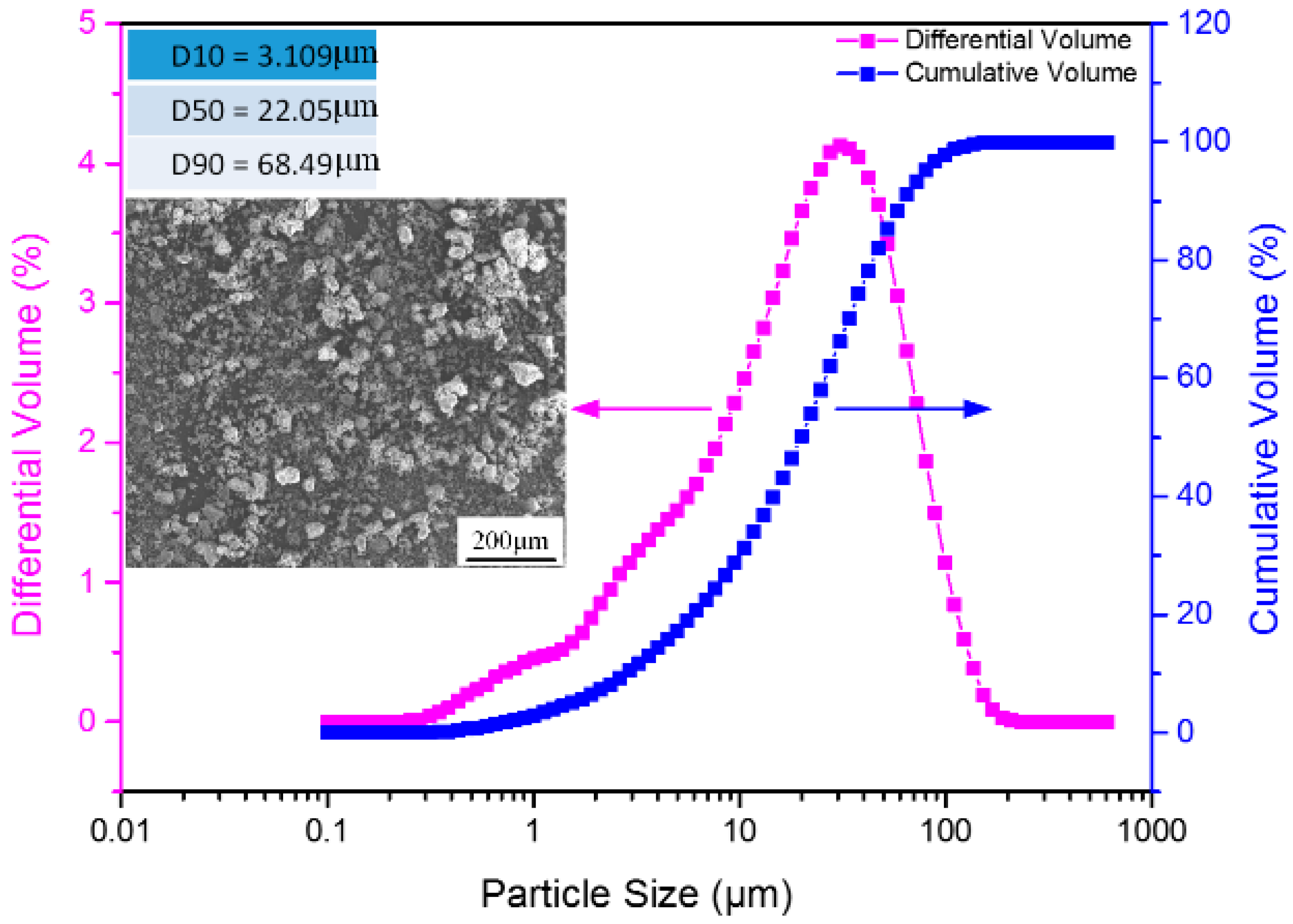
2.1. Raw Materials and Reagents
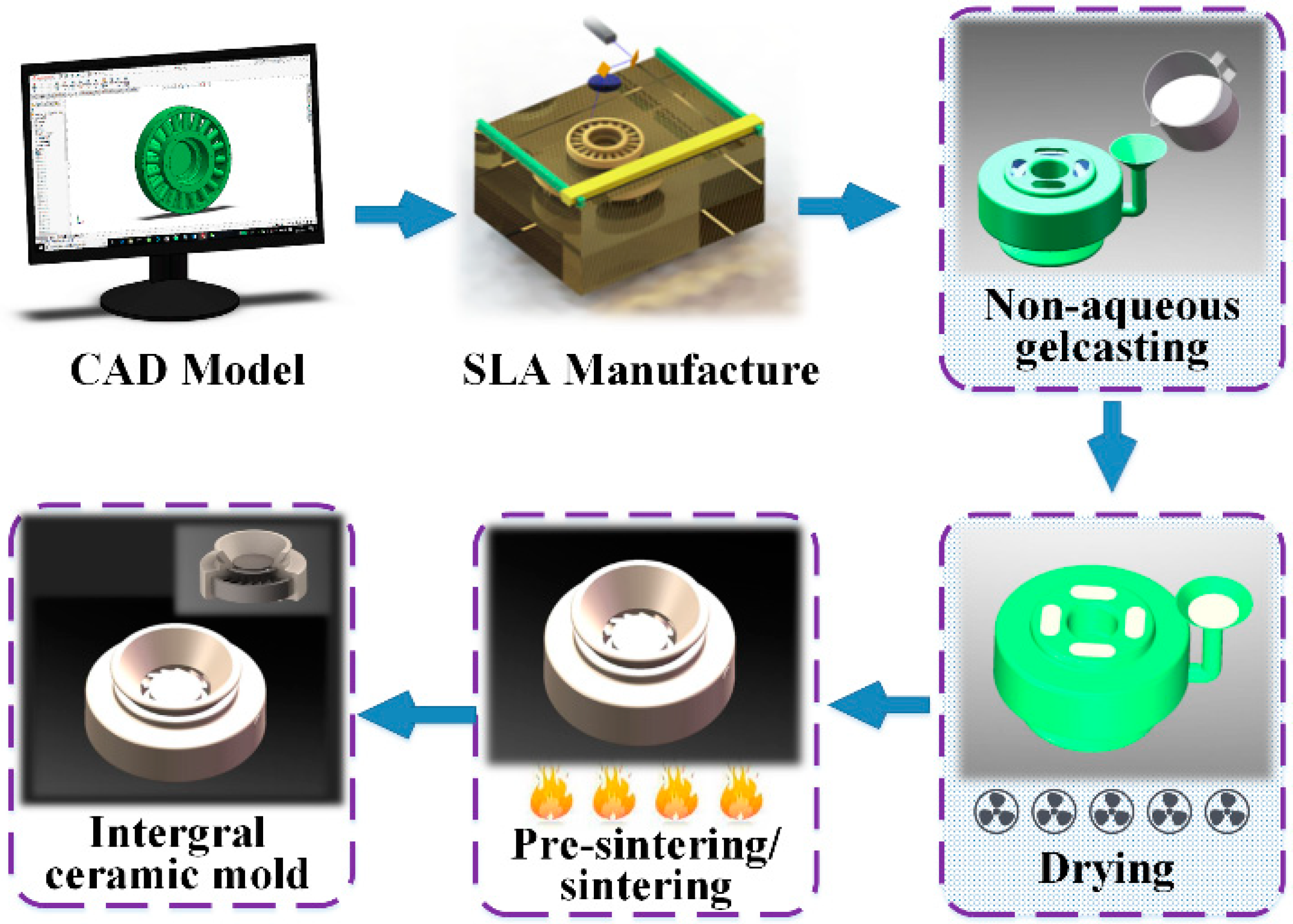
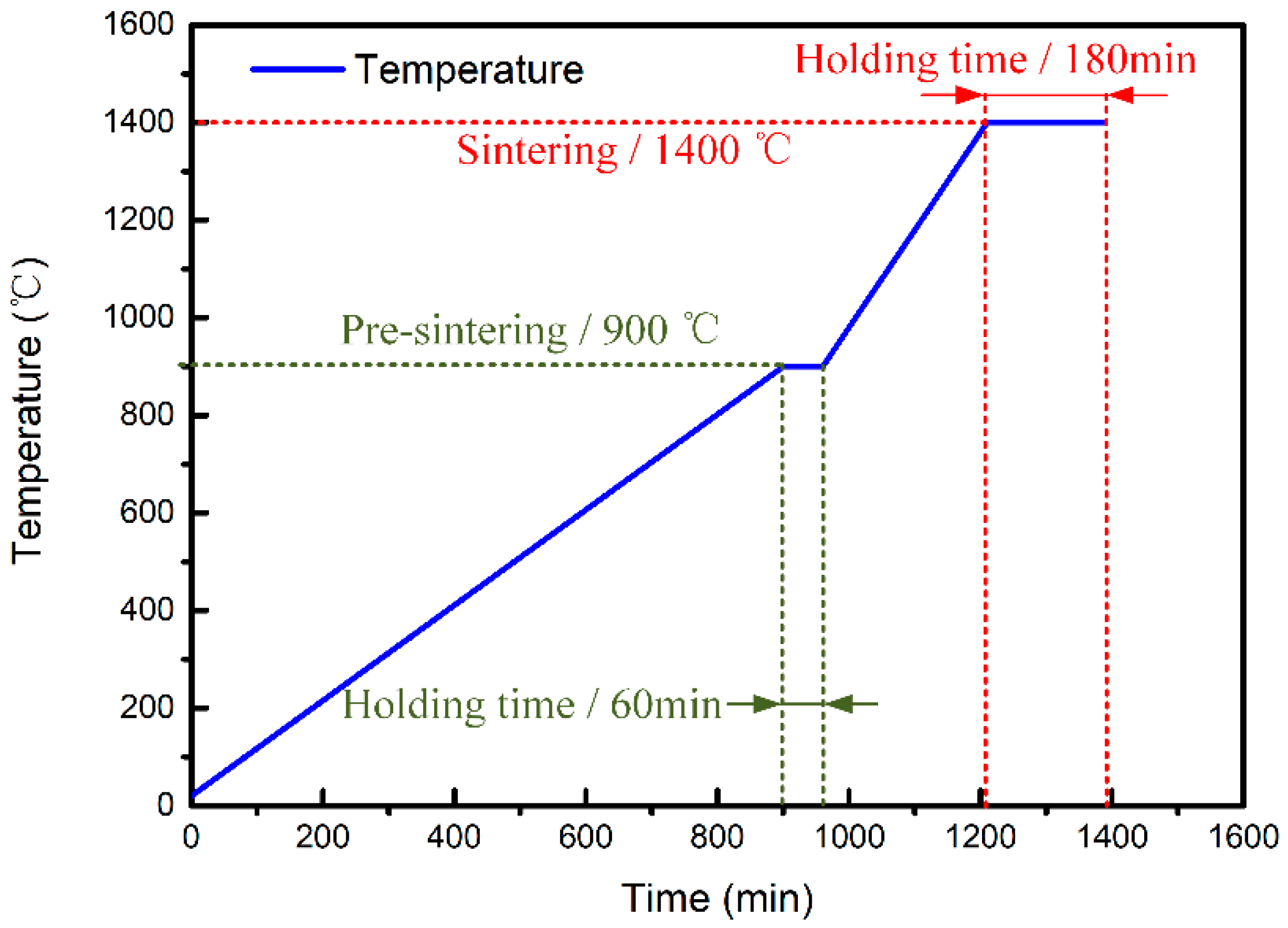
2.2. Fabrication of CaO-Based Integral Ceramic Mould
2.3. Characterization and Testing
3. Results and Discussions
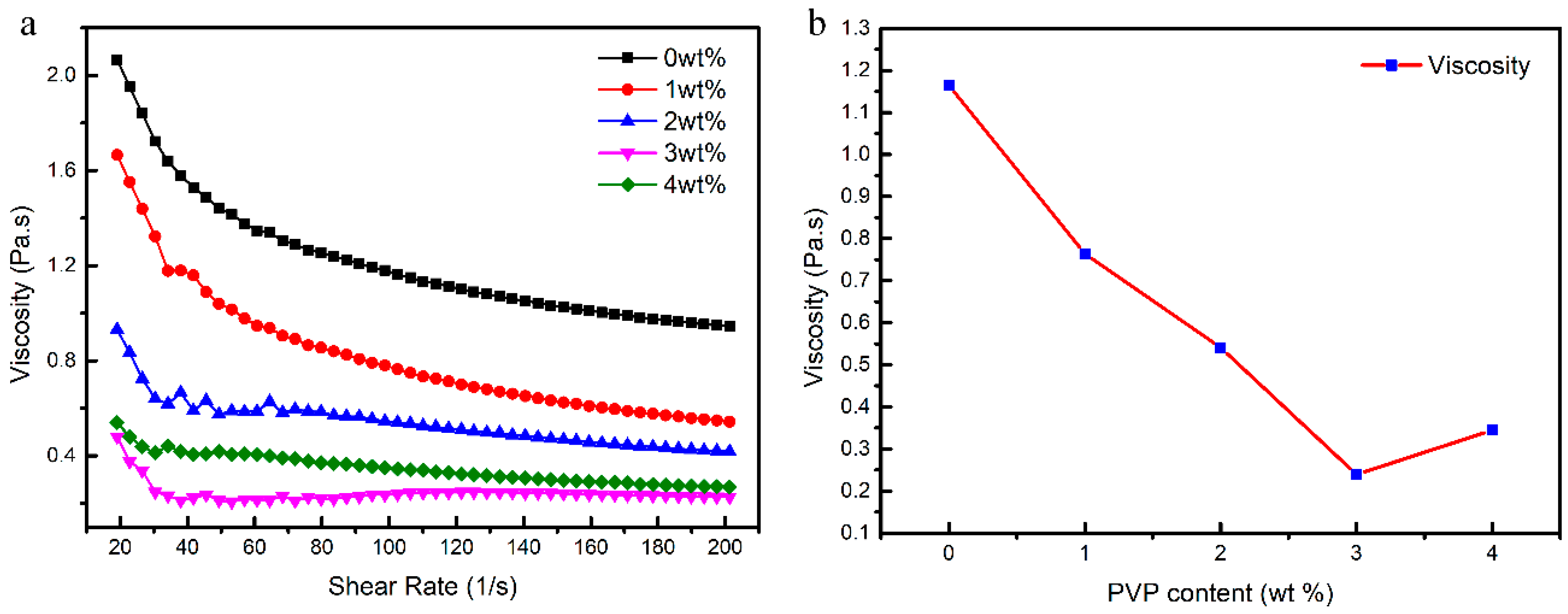
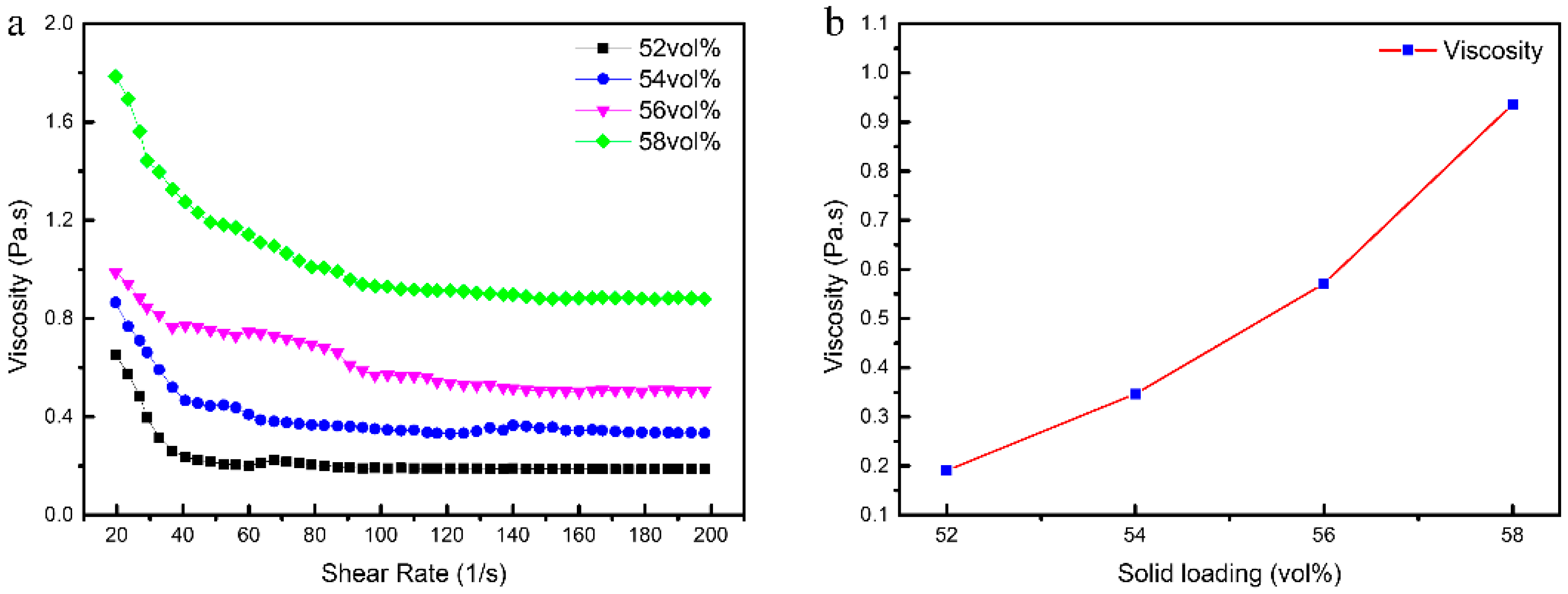
3.1. Rheology of CaO Slurries
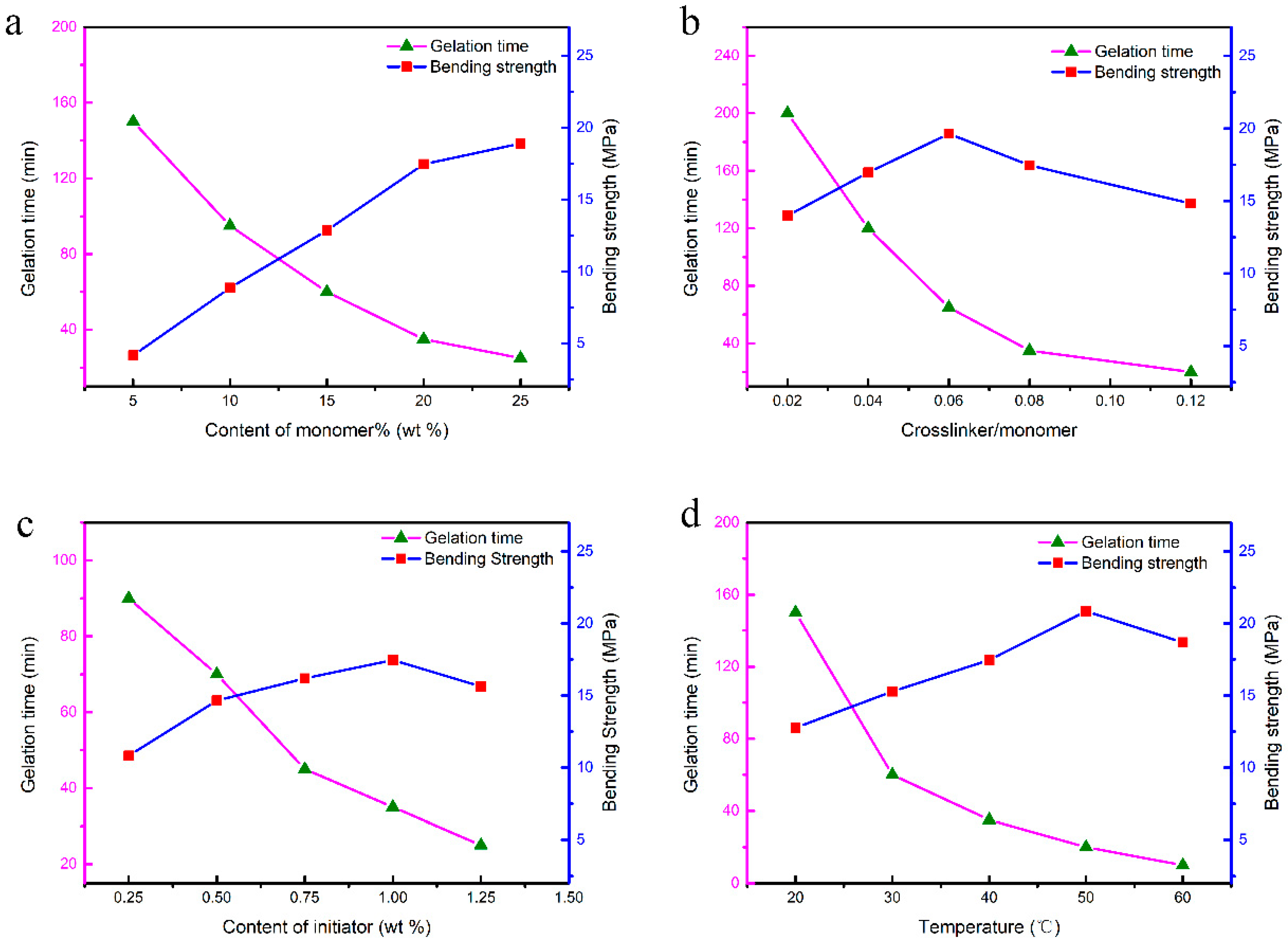
3.2. Gelation Process of CaO-Based Slurries
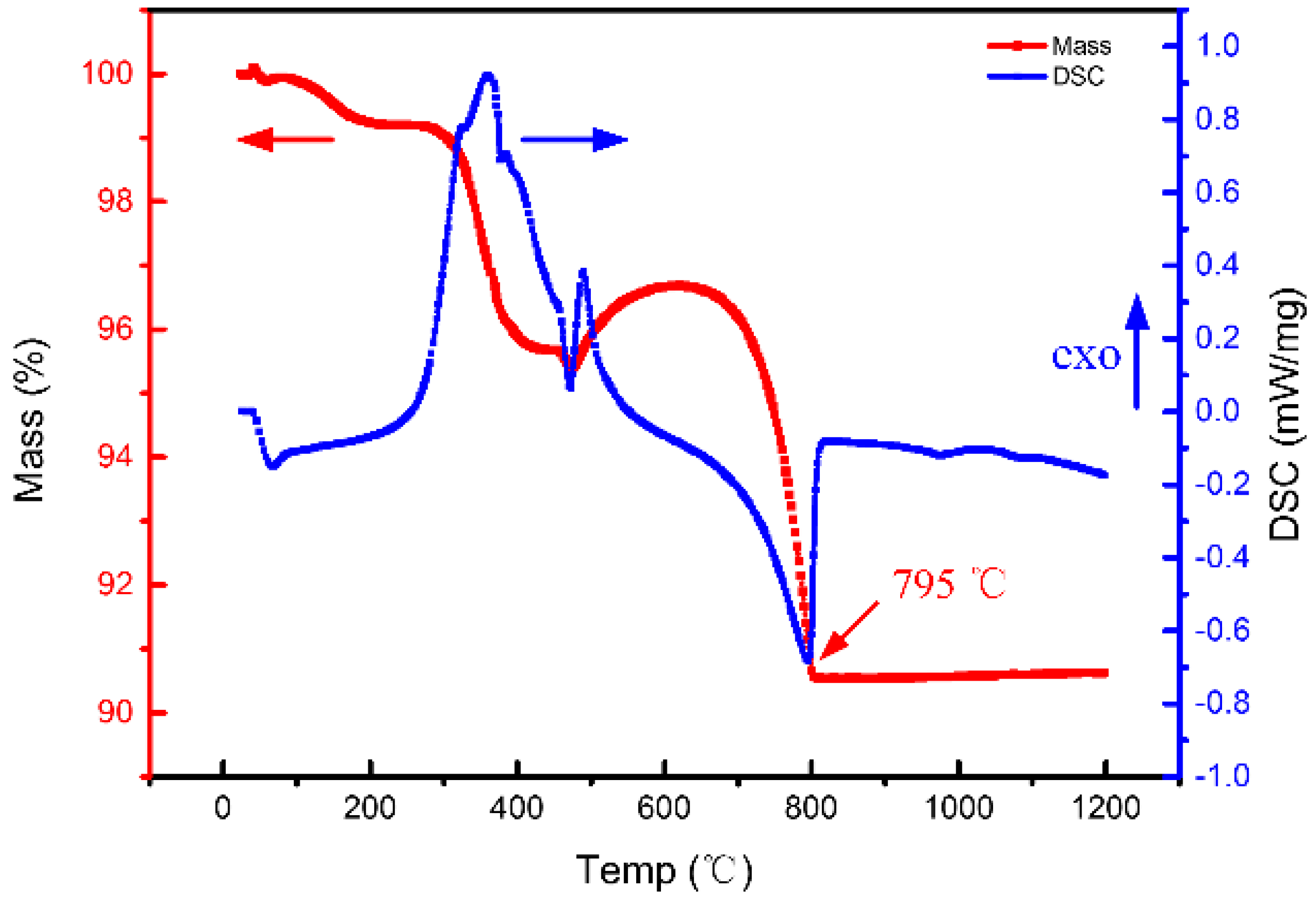
3.3. Cracks during Pre-Sintering
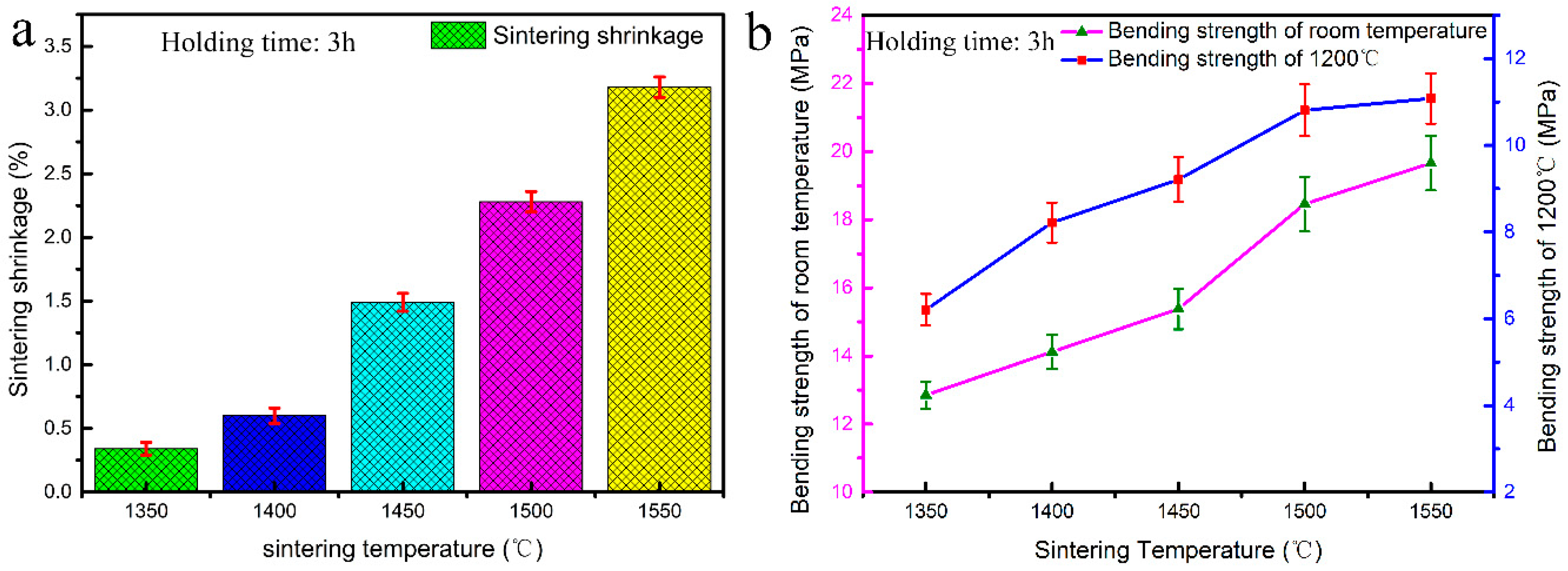
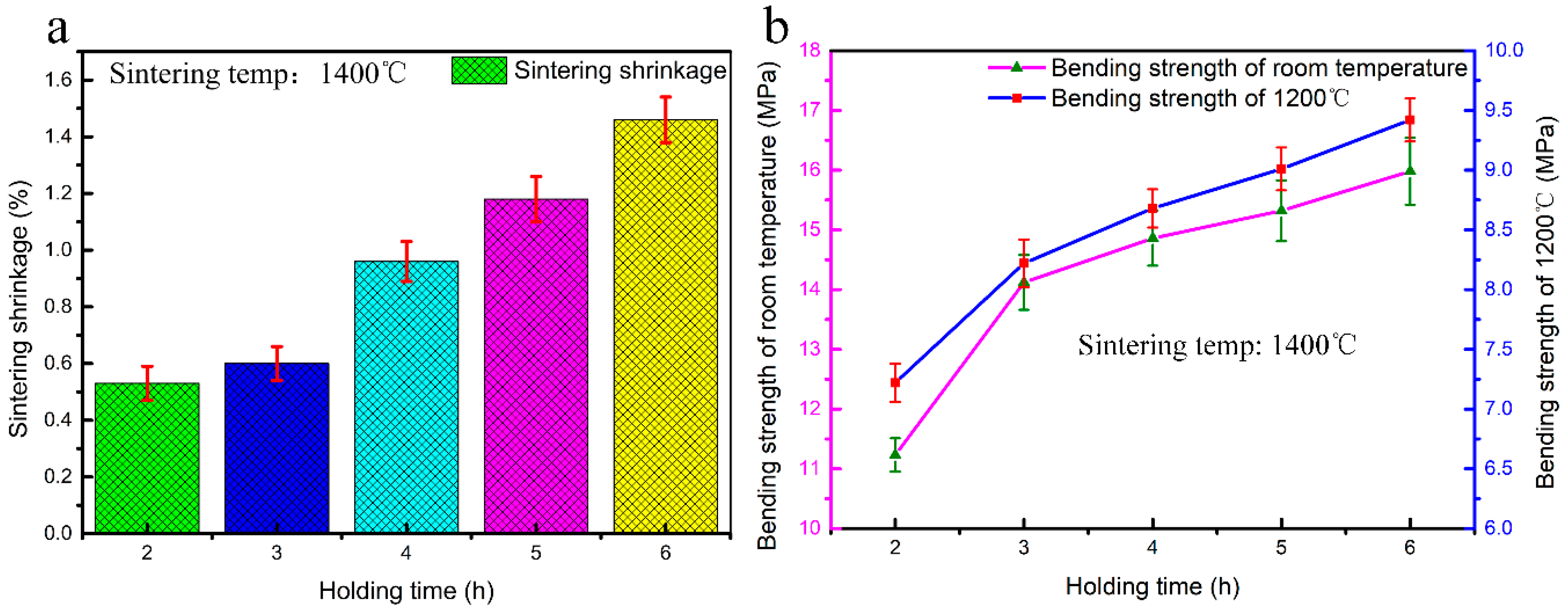
3.4. Sintering Shrinkage and Strength of CaO-Based Mould
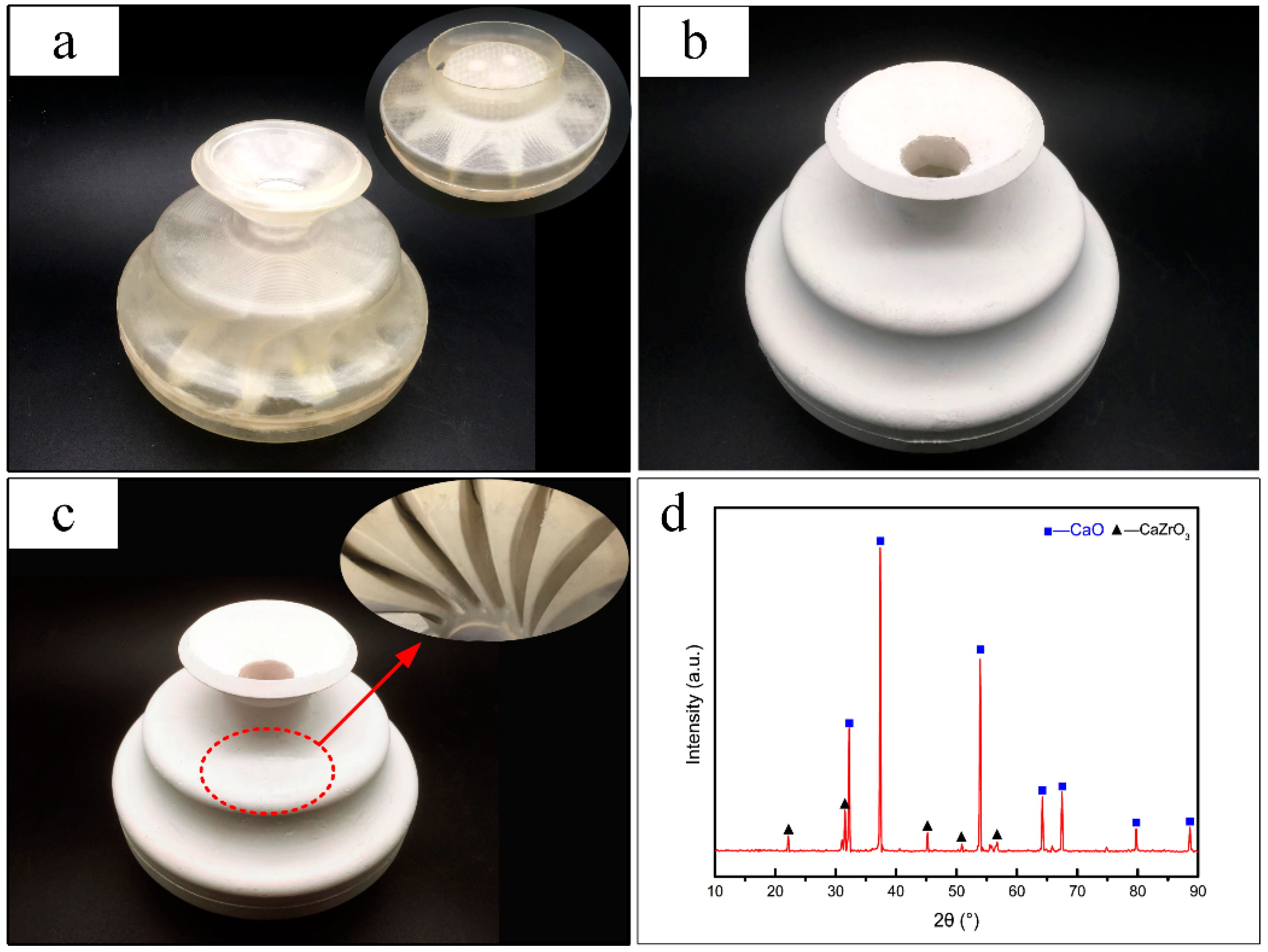
4. Case Study
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pattnaik, S.; Karunakar, D.B.; Jha, P.K. Developments in investment casting process-A review. J. Mater. Process. Technol. 2012, 212, 2332–2348. [Google Scholar] [CrossRef]
- Xu, Z.; Zhong, J.; Su, X.; Xu, Q.; Liu, B. Experimental study on mechanical properties of silica-based ceramic core for directional solidification of single crystal superalloy. Ceram. Int. 2018, 44, 394–401. [Google Scholar] [CrossRef]
- Qin, Y.; Pan, W. Effect of silica sol on the properties of alumina-based ceramic core composites. Mater. Sci. Eng. A 2009, 508, 71–75. [Google Scholar] [CrossRef]
- Wang, F.; Li, F.; He, B.; Wang, D.; Sun, B. Gel-casting of fused silica based core packing for investment casting using silica sol as a binder. J. Eur. Ceram. Soc. 2013, 33, 2745–2749. [Google Scholar] [CrossRef]
- Barbosa, J.; Puga, H.; Ribeiro, C.S.; Teodoro, O.M.N.D.; Monteiro, A.C. Characterisation of metal/mould interface on investment casting of gamma-TiAl. Inter. J. Cast Metals Res. 2006, 19, 331–338. [Google Scholar] [CrossRef]
- Zhang, H.; Lu, Z.; Ji, Z.; Li, D. Basis for the alkaline removal process design of the alumina-based ceramic core. J. Ceram. Soc. Jpn. 2017, 125, 616–622. [Google Scholar] [CrossRef]
- Wang, Y.; Guo, X.; Qiao, Y. Interactions between Nb-Si based ultrahigh temperature alloy and yttria matrix mould shells. Mater. Des. 2017, 116, 461–471. [Google Scholar]
- Hung, C.C.; Lai, P.L.; Tsai, C.C.; Huang, T.K.; Liao, Y.Y. Pure titanium casting into titanium-modified calcia-based and magnesia-based investment molds. Mater. Sci. Eng. A 2007, 454, 178–182. [Google Scholar] [CrossRef]
- Klotz, U.E.; Heiss, T. Evaluation of crucible and investment materials for lost wax investment casting of Ti and NiTi alloys. Int. J. Cast Metals Res. 2014, 27, 341–348. [Google Scholar] [CrossRef]
- Liu, F.; Fan, Z.; Liu, X.; He, J.; Li, F. Aqueous gel casting of water-soluble calcia-based ceramic core for investment casting using epoxy resin as a binder. Int. J. Adv. Manuf. Technol. 2016, 86, 1235–1242. [Google Scholar] [CrossRef]
- Hulse, C.O. Process of Casting Nickel Base Alloys Using Water-Soluble Calcia Cores. U.S. Patent No. 3,643,728, 22 February 1972. [Google Scholar]
- Wu, G.; Zhou, P.; Zhao, J.; Nan, H. Calcium Oxide-Based Ceramic Core and Preparation Method Thereof. U.S. Patent No. 9,308,579, 12 April 2016. [Google Scholar]
- Niinomi, M.; Akahori, T.; Takeuchi, T.; Katsura, S. Dental Precision Casting of Ti-29Nb-13Ta-4.6Zr Using Calcia Mold. Mater. Trans. 2009, 50, 2057–2063. [Google Scholar]
- Ren, X.; Shao, H.; Lin, T.; Zheng, H. 3D gel-printing-An additive manufacturing method for producing complex shape parts. Mater. Des. 2016, 101, 80–87. [Google Scholar]
- Zhao, H.; Ye, C.; Fan, Z.; Wang, C. 3D printing of CaO-based ceramic core using nanozirconia suspension as a binder. J. Eur. Ceram. Soc. 2017, 37, 5119–5125. [Google Scholar] [CrossRef]
- Wu, H.; Li, D.; Guo, N. Fabrication of integral ceramic mold for investment casting of hollow turbine blade based on stereolithography. Rapid Prototyp. J. 2009, 15, 232–237. [Google Scholar] [CrossRef]
- Wu, H.; Li, D.; Tang, Y.; Sun, B.; Xu, D. Rapid fabrication of alumina-based ceramic cores for gas turbine blades by stereolithography and gelcasting. J. Mater. Process. Technol. 2009, 209, 5886–5891. [Google Scholar] [CrossRef]
- Yang, Q.; Lu, Z.; Zhou, J.; Miao, K.; Li, D. A novel method for improving surface finish of stereolithography apparatus. Int. J. Adv. Manuf. Technol. 2017, 93, 1537–1544. [Google Scholar] [CrossRef]
- Young, A.C.; Omatete, O.O.; Janney, M.A.; Menchhofer, P.A. Gelcasting of Alumina. J. Am. Ceram. Soc. 1991, 74, 612–618. [Google Scholar] [CrossRef]
- Omatete, O.O.; Janney, M.A.; Nunn, S.D. Gelcasting: From laboratory development toward industrial production. J. Eur. Ceram. Soc. 1997, 17, 407–413. [Google Scholar] [CrossRef]
- Jiao, C.R.; Chen, D.M.; Tong, J.F. Preparation of the Barium Strontium Titanate/Magnesium Oxide Plates by Non-Aqueous Gel-Casting. Adv. Mater. Res-Switz. 2010, 105, 794–797. [Google Scholar] [CrossRef]
- Wei, G.U.O.; Hongbin, L.U.; Chunxia, F.E.N.G. Influence of La2O3 on preparation and performance of porous cordierite from rice husk. J. Rare Earths 2010, 28, 614–617. [Google Scholar]
- Jiang, C.; Gan, X.P.; Zhang, D.; Xie, R.; Zhou, K.C. Gelcasting of aluminum nitride ceramics using hydantion epoxy resin as gelling agent. Ceram. Int. 2013, 39, 9429–9433. [Google Scholar] [CrossRef]
- Liu, S.; Liu, J.C.; Hou, F.; Du, H.Y. Microstructure and properties of inter-locked mullite framework prepared by the TBA-based gel-casting process. Ceram. Int. 2016, 42, 15459–15463. [Google Scholar] [CrossRef]
- Hou, Z.G.; Du, H.Y.; Liu, J.C.; Hao, R.H.; Dong, X.; Liu, M.X. Fabrication and properties of mullite fiber matrix porous ceramics by a TBA-based gel-casting process. J. Eur. Ceram. Soc. 2013, 33, 717–725. [Google Scholar] [CrossRef]
- Yang, J.; Yu, J.; Huang, Y. Recent developments in gelcasting of ceramics. J. Eur. Ceram. Soc. 2011, 31, 2569–2591. [Google Scholar] [CrossRef]
- Montanaro, L.; Coppola, B.; Palmero, P.; Tulliani, J.-M. A review on aqueous gelcasting: A versatile and low-toxic technique to shape ceramics. Ceram. Int. 2018, 45, 9653–9673. [Google Scholar] [CrossRef]
- Zhang, C.; Yang, J.; Qiu, T.; Guo, J. Preparation of ZTA ceramic by aqueous gelcasting with a low-toxic monomer DMAA. Ceram. Int. 2012, 38, 3063–3068. [Google Scholar] [CrossRef]
- Shen, L.Y.; Xu, X.S.; Lu, W.; Shi, B. Aluminum nitride shaping by non-aqueous gelcasting of low-viscosity and high solid-loading slurry. Ceram. Int. 2016, 42, 5569–5574. [Google Scholar] [CrossRef]
- Wan, W.; Yang, J.; Zeng, J.Z.; Yao, L.C.; Qiu, T. Effect of solid loading on gelcasting of silica ceramics using DMAA. Ceram. Int. 2014, 40, 1735–1740. [Google Scholar] [CrossRef]
- Wu, H.H.; Li, D.C.; Tang, Y.P.; Sun, B.; Xu, D.Y. Gelcasting of alumina based ceramic cores containing yttria for single crystal and directional solidification blades. Adv. Appl. Ceram. 2009, 108, 406–411. [Google Scholar] [CrossRef]


| Chemical | CaO | MgO | SiO2 | Al2O3 | Fe2O3 |
|---|---|---|---|---|---|
| wt % | 98.10 | 0.81 | 0.72 | 0.25 | 0.12 |
| Properties | CaO-Based Integral Ceramic Moulds | Al2O3-Based AC-1 Ceramic Cores [31] | CaO-Based Ceramic Cores [10] |
|---|---|---|---|
| Bending strength at room temperature/MPa | 14.12 ± 0.28 | 9–12 | 25 ± 1.2 |
| Bending strength at high temperature (1200 °C)/MPa | 8.22 ± 0.26 | 0.8–1.6 | Not mentioned |
| Sintering shrinkage/% | 0.6 ± 0.06 | 1.5 | 11.31 |
| Apparent porosity/% | 32.8 ± 1.2 | 34 | 30.5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, Q.; Zhu, W.; Lu, Z.; Li, D.; Wang, Z.; Wang, F. Rapid Fabrication of High-Performance CaO-Based Integral Ceramic Mould by Stereolithography and Non-Aqueous Gelcasting. Materials 2019, 12, 934. https://doi.org/10.3390/ma12060934
Yang Q, Zhu W, Lu Z, Li D, Wang Z, Wang F. Rapid Fabrication of High-Performance CaO-Based Integral Ceramic Mould by Stereolithography and Non-Aqueous Gelcasting. Materials. 2019; 12(6):934. https://doi.org/10.3390/ma12060934
Chicago/Turabian StyleYang, Qiang, Weijun Zhu, Zhongliang Lu, Dichen Li, Zhongrui Wang, and Fu Wang. 2019. "Rapid Fabrication of High-Performance CaO-Based Integral Ceramic Mould by Stereolithography and Non-Aqueous Gelcasting" Materials 12, no. 6: 934. https://doi.org/10.3390/ma12060934
APA StyleYang, Q., Zhu, W., Lu, Z., Li, D., Wang, Z., & Wang, F. (2019). Rapid Fabrication of High-Performance CaO-Based Integral Ceramic Mould by Stereolithography and Non-Aqueous Gelcasting. Materials, 12(6), 934. https://doi.org/10.3390/ma12060934




