10-MDP Based Dental Adhesives: Adhesive Interface Characterization and Adhesive Stability—A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Inclusion and Exclusion Criteria
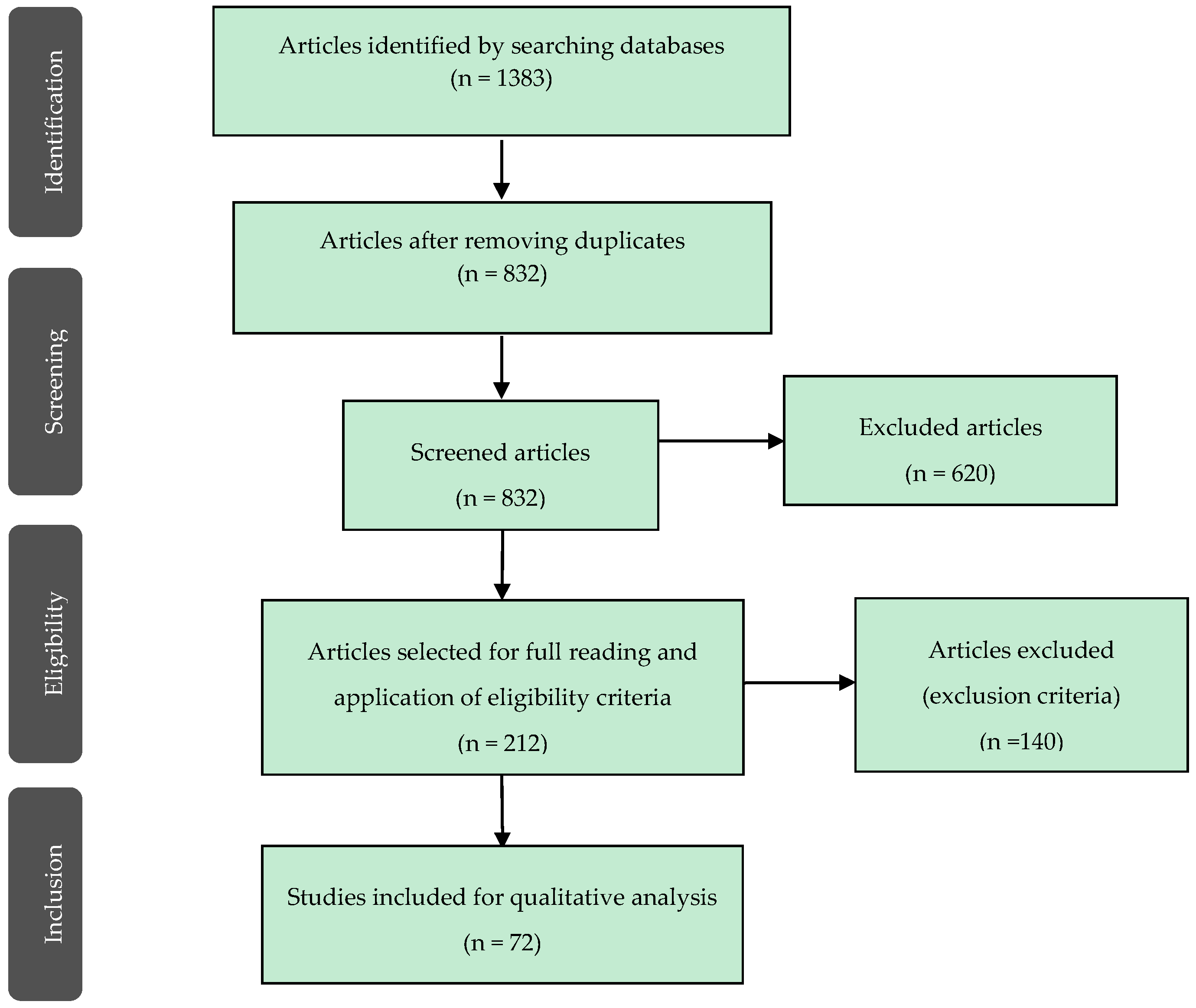
3. Results
- Formation of nano-layered structures (MDP-Ca salts)—Formation/absence of nano-layered structures and morphology (Table 4);
- Acid-base resistant zone (ABRZ)—Formation or absence of ABRZ, thickness, and differences between dentin ABRZ and enamel ABRZ (Table 5);
- Adhesive stability—Measurement of adhesion forces (Table 6).
3.1. 10-MDP Monomer: Molecular Structure, Hydrophilicity and Nano-Layered Structures
3.2. Capacity to Create an Acid-Base Resistant Zone (ABRZ)
3.3. Adhesive Stability
4. Discussion
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhang, Z.; Tian, F.; Niu, L.; Ochala, K.; Chen, C.; Fu, B.; Wang, X.; Pashley, D.; Tay, F. Defying ageing: An expectation for dentine bonding with universal adhesives? J. Dent. 2016, 45, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Nikaido, T.; Ichikawa, C.; Li, N.; Takagaki, T.; Sadr, A.; Yoshida, Y.; Suzuki, K.; Tagami, J. Effect of functional monomers in all-in-one adhesive systems on formation of enamel/dentin acid-base resistant zone. Dent. Mater. J. 2011, 30, 576–582. [Google Scholar] [CrossRef] [PubMed]
- Imai, T.; Itoh, K.; Tani, C.; Manabe, A.; Yamashita, T.; Hisamitsu, H.; Wakumoto, S. Effectiveness of simplified dentin bonding systems. Dent. Mater. J. 1998, 17, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Yoshihara, K.; Hayakawa, S.; Nagaoka, N.; Okihara, T.; Yoshida, Y.; Van Meerbeek, B. Etching Efficacy of Self-Etching Functional Monomers. J. Dent. Res. 2018, 97, 1010–1016. [Google Scholar] [CrossRef] [PubMed]
- Yaguchi, T. Layering mechanism of MDP-Ca salt produced in demineralization of enamel and dentin apatite. Dent. Mater. 2017, 33, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Tian, F.; Zhou, L.; Zhang, Z.; Niu, L.; Zhang, L.; Chen, C.; Zhou, J.; Yang, H.; Wang, X.; Fu, B.; et al. Paucity of Nanolayering in Resin-Dentin Interfaces of MDP-based Adhesives. J. Dent. Res. 2016, 95, 380–387. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, Y.; Nagakane, K.; Fukuda, R.; Nakayama, Y.; Okazaki, M.; Shintani, H.; Inoue, S.; Tagawa, Y.; Suzuki, K.; De Munck, J.; et al. Comparative study on adhesive performance of functional monomers. J. Dent. Res. 2004, 83, 454–458. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Wang, X.; Zhang, L.; Liang, B.; Tang, T.; Fu, B.; Hannig, M. The contribution of chemical bonding to the short- and long-term enamel bond strengths. Dent. Mater. 2013, 29, e103–e112. [Google Scholar] [CrossRef] [PubMed]
- Van Landuyt, K.L.; Yoshida, Y.; Hirata, I.; Snauwaert, J.; De Munck, J.; Okazaki, M.; Suzuki, K.; Lambrechts, P.; Van Meerbeek, B. Influence of the chemical structure of functional monomers on their adhesive performance. J. Dent. Res. 2008, 87, 757–761. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Nikaido, T.; Takagaki, T.; Sadr, A.; Makishi, P.; Chen, J.; Tagami, J. The role of functional monomers in bonding to enamel: acid-base resistant zone and bonding performance. J. Dent. 2010, 38, 722–730. [Google Scholar] [CrossRef] [PubMed]
- Yoshihara, K.; Nagaoka, N.; Okihara, T.; Kuroboshi, M.; Hayakawa, S.; Maruo, Y.; Nishigawa, G.; De Munck, J.; Yoshida, Y.; Van Meerbeek, B. Functional monomer impurity affects adhesive performance. Dent. Mater. 2015, 31, 1493–1501. [Google Scholar] [CrossRef] [PubMed]
- Iwai, H.; Nishiyama, N. Effect of calcium salt of functional monomer on bonding performance. J. Dent. Res. 2012, 91, 1043–1048. [Google Scholar] [CrossRef] [PubMed]
- Feitosa, V.P.; Ogliari, F.A.; Van Meerbeek, B.; Watson, T.F.; Yoshihara, K.; Ogliari, A.O.; Sinhoreti, M.A.; Correr, A.B.; Cama, G.; Sauro, S. Can the hydrophilicity of functional monomers affect chemical interaction? J. Dent. Res. 2014, 93, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Fukegawa, D.; Hayakawa, S.; Yoshida, Y.; Suzuki, K.; Osaka, A.; Van Meerbeek, B. Chemical interaction of phosphoric acid ester with hydroxyapatite. J. Dent. Res. 2006, 85, 941–944. [Google Scholar] [CrossRef] [PubMed]
- Feitosa, V.P.; Sauro, S.; Ogliari, F.A.; Ogliari, A.O.; Yoshihara, K.; Zanchi, C.H.; Correr-Sobrinho, L.; Sinhoreti, M.A.; Correr, A.B.; Watson, T.F.; et al. Impact of hydrophilicity and length of spacer chains on the bonding of functional monomers. Dent. Mater. 2014, 30, e317–e323. [Google Scholar] [CrossRef] [PubMed]
- Salz, U.; Mucke, A.; Zimmermann, J.; Tay, F.R.; Pashley, D.H. pKa value and buffering capacity of acidic monomers commonly used in self-etching primers. J. Adhes. Dent. 2006, 8, 143–150. [Google Scholar] [PubMed]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef] [PubMed]
- Yoshihara, K.; Yoshida, Y.; Hayakawa, S.; Nagaoka, N.; Irie, M.; Ogawa, T.; Van Landuyt, K.L.; Osaka, A.; Suzuki, K.; Minagi, S.; et al. Nanolayering of phosphoric acid ester monomer on enamel and dentin. Acta Biomater. 2011, 7, 3187–3195. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, Y.; Yoshihara, K.; Nagaoka, N.; Hayakawa, S.; Torii, Y.; Ogawa, T.; Osaka, A.; Meerbeek, B.V. Self-assembled Nano-layering at the Adhesive interface. J. Dent. Res. 2012, 91, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Yoshihara, K.; Yoshida, Y.; Nagaoka, N.; Hayakawa, S.; Okihara, T.; De Munck, J.; Maruo, Y.; Nishigawa, G.; Minagi, S.; Osaka, A.; et al. Adhesive interfacial interaction affected by different carbon-chain monomers. Dent. Mater. 2013, 29, 888–897. [Google Scholar] [CrossRef] [PubMed]
- Hiraishi, N.; Tochio, N.; Kigawa, T.; Otsuki, M.; Tagami, J. Role of 2-hydroxyethyl methacrylate in the interaction of dental monomers with collagen studied by saturation transfer difference NMR. J. Dent. 2014, 42, 484–489. [Google Scholar] [CrossRef] [PubMed]
- Yokota, Y.; Nishiyama, N. Determination of molecular species of calcium salts of MDP produced through decalcification of enamel and dentin by MDP-based one-step adhesive. Dent. Mater. J. 2015, 34, 270–279. [Google Scholar] [CrossRef] [PubMed]
- Matsui, N.; Takagaki, T.; Sadr, A.; Ikeda, M.; Ichinose, S.; Nikaido, T.; Tagami, J. The role of MDP in a bonding resin of a two-step self-etching adhesive system. Dent. Mater. J. 2015, 34, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Nurrohman, H.; Nikaido, T.; Takagaki, T.; Sadr, A.; Ichinose, S.; Tagami, J. Apatite crystal protection against acid-attack beneath resin-dentin interface with four adhesives: TEM and crystallography evidence. Dent. Mater. 2012, 28, e89–e98. [Google Scholar] [CrossRef] [PubMed]
- Guan, R.; Takagaki, T.; Matsui, N.; Sato, T.; Burrow, M.F.; Palamara, J.; Nikaido, T.; Tagami, J. Dentin bonding performance using Weibull statistics and evaluation of acid-base resistant zone formation of recently introduced adhesives. Dent. Mater. J. 2016, 35, 684–693. [Google Scholar] [CrossRef] [PubMed]
- Nikaido, T.; Nurrohman, H.; Takagaki, T.; Sadr, A.; Ichinose, S.; Tagami, J. Nanoleakage in Hybrid Layer and Acid-Base Resistant Zone at the Adhesive/Dentin Interface. Microsc. Microanal. 2015, 21, 1271–1277. [Google Scholar] [CrossRef] [PubMed]
- Hayakawa, T.; Kikutake, K.; Nemoto, K. Influence of self-etching primer treatment on the adhesion of resin composite to polished dentin and enamel. Dent. Mater. 1998, 14, 99–105. [Google Scholar] [CrossRef]
- Inoue, S.; Koshiro, K.; Yoshida, Y.; De Munck, J.; Nagakane, K.; Suzuki, K.; Sano, H.; Van Meerbeek, B. Hydrolytic stability of self-etch adhesives bonded to dentin. J. Dent. Res. 2005, 84, 1160–1164. [Google Scholar] [CrossRef] [PubMed]
- Fujita, K.; Ma, S.; Aida, M.; Maeda, T.; Ikemi, T.; Hirata, M.; Nishiyama, N. Effect of reacted acidic monomer with calcium on bonding performance. J. Dent. Res. 2011, 90, 607–612. [Google Scholar] [CrossRef] [PubMed]
- Harnirattisai, C.; Roengrungreang, P.; Rangsisiripaiboon, U.; Senawongse, P. Shear and micro-shear bond strengths of four self-etching adhesives measured immediately and 24 hours after application. Dent. Mater. J. 2012, 31, 779–787. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, H. Effect of calcium salt of 10-methacryloyloxydecyl dihydrogen phosphate produced on the bond durability of one-step self-etch adhesive. Dent. Mater. J. 2014, 33, 394–401. [Google Scholar] [CrossRef] [PubMed]
- Anchieta, R.B.; Machado, L.S.; Martini, A.P.; Santos, P.H.; Giannini, M.; Janal, M.; Tovar, N.; Sundfeld, R.H.; Rocha, E.P.; Coelho, P.G. Effect of long-term storage on nanomechanical and morphological properties of dentin-adhesive interfaces. Dent. Mater. 2015, 31, 141–153. [Google Scholar] [CrossRef] [PubMed]
- Munoz, M.A.; Luque-Martinez, I.; Malaquias, P.; Hass, V.; Reis, A.; Campanha, N.H.; Loguercio, A.D. In Vitro Longevity of Bonding Properties of Universal Adhesives to Dentin. Oper. Dent. 2015, 40, 282–292. [Google Scholar] [CrossRef] [PubMed]
- Thanatvarakorn, O.; Prasansuttiporn, T.; Takahashi, M.; Thittaweerat, S.; Foxton, R.M.; Ichinose, S.; Tagami, J.; Nakajima, M. Effect of Scrubbing Technique with Mild Self-etching Adhesives on Dentin Bond Strengths and Nanoleakage Expression. J. Adhes. Dent. 2016, 18, 197–204. [Google Scholar] [PubMed]
- Wang, R.; Shi, Y.; Li, T.; Pan, Y.; Cui, Y.; Xia, W. Adhesive interfacial characteristics and the related bonding performance of four self-etching adhesives with different functional monomers applied to dentin. J. Dent. 2017, 62, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Niu, L.N.; Xie, H.; Zhang, Z.Y.; Zhou, L.Q.; Jiao, K.; Chen, J.H.; Pashley, D.H.; Tay, F.R. Bonding of universal adhesives to dentine—Old wine in new bottles? J. Dent. 2015, 43, 525–536. [Google Scholar] [CrossRef] [PubMed]
- Farias, D.C.S.; de Andrada, M.A.C.; Boushell, L.W.; Walter, R. Assessment of the initial and aged dentin bond strength of universal adhesives. Int. J. Adhes. Adhes. 2016, 70, 53–61. [Google Scholar] [CrossRef]
- Tsuchiya, K.; Takamizawa, T.; Barkmeier, W.W.; Tsubota, K.; Tsujimoto, A.; Berry, T.P.; Erickson, R.L.; Latta, M.A.; Miyazaki, M. Effect of a functional monomer (MDP) on the enamel bond durability of single-step self-etch adhesives. Eur. J. Oral Sci. 2016, 124, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Tsujimoto, A.; Barkmeier, W.W.; Takamizawa, T.; Watanabe, H.; Johnson, W.W.; Latta, M.A.; Miyazaki, M. Comparison between universal adhesives and two-step self-etch adhesives in terms of dentin bond fatigue durability in self-etch mode. Eur. J. Oral Sci. 2017, 125, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Hass, V.; Abuna, G.; Feitosa, V.P.; Martini, E.C.; Sinhoreti, M.A.; Carvalho, R.F.; Bandéca, M.; Sauro, S.; Loguercio, A.D. Self-Etching Enamel Bonding Using Acidic Functional Monomers with Different-length Carbon Chains and Hydrophilicity. J. Adhes. Dent. 2017, 19, 497–505. [Google Scholar] [PubMed]
- Hoshika, S.; Kameyama, A.; Suyama, Y.; De Munck, J.; Sano, H.; Van Meerbeek, B. GPDM- and 10-MDP-based Self-etch Adhesives Bonded to Bur-cut and Uncut Enamel—“Immediate” and “Aged” µTBS. J. Adhes. Dent. 2018, 20, 113–120. [Google Scholar] [PubMed]
- Mine, A.; De Munck, J.; Cardoso, M.V.; Van Landuyt, K.L.; Poitevin, A.; Van Ende, A.; Matsumoto, M.; Yoshida, Y.; Kuboki, T.; Yatani, H.; et al. Dentin-smear remains at self-etch adhesive interface. Dent. Mater. 2014, 30, 1147–1153. [Google Scholar] [CrossRef] [PubMed]
- Van Meerbeek, B.; Yoshihara, K.; Yoshida, Y.; Mine, A.; De Munck, J.; Van Landuyt, K.L. State of the art of self-etch adhesives. Dent. Mater. 2011, 27, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Leal, F.B.; Madruga, F.C.; Prochnow, E.P.; Lima, G.S.; Ogliari, F.A.; Piva, E.; Moraes, R. Effect of acidic monomer concentration on the dentin bond stability of self-etch adhesives. Int. J. Adhes. Adhes. 2011, 31, 571–574. [Google Scholar] [CrossRef]
- Wang, X.M.; Wang, C.Y.; Zhang, L.; Zhang, Z.L.; Fu, B.P.; Hannig, M. Influence of priming time and primer’s concentrations on bovine enamel bond strengths. J. Adhes. Sci. Technol. 2013, 27, 2558–2570. [Google Scholar] [CrossRef]
- Hiraishi, N.; Tochio, N.; Kigawa, T.; Otsuki, M.; Tagami, J. Monomer-collagen interactions studied by saturation transfer difference NMR. J. Dent. Res. 2013, 92, 284–288. [Google Scholar] [CrossRef] [PubMed]
- Yokota, Y.; Fujita, K.N.; Uchida, R.; Aida, E.; Aoki, N.T.; Aida, M.; Nishiyama, N. Quantitative Evaluation of MDP-Ca Salt and DCPD after Application of an MDP-based One-step Self-etching Adhesive on Enamel and Dentin. J. Adhes. Dent. 2016, 18, 205–213. [Google Scholar] [PubMed]
- Tian, F.C.; Wang, X.Y.; Huang, Q.; Niu, L.N.; Mitchell, J.; Zhang, Z.Y.; Prananik, C.; Zhang, L.; Chen, J.H.; Breschi, L.; et al. Effect of nanolayering of calcium salts of phosphoric acid ester monomers on the durability of resin-dentin bonds. Acta Biomater. 2016, 38, 190–200. [Google Scholar] [CrossRef] [PubMed]
- Yoshihara, K.; Yoshida, Y.; Hayakawa, S.; Nagaoka, N.; Torii, Y.; Osaka, A.; Suzuki, K.; Minagi, S.; Van Meerbeek, B.; Van Landuyt, K.L. Self-etch monomer-calcium salt deposition on dentin. J. Dent. Res. 2011, 90, 602–606. [Google Scholar] [CrossRef] [PubMed]
- Hanabusa, M.; Yoshihara, K.; Yoshida, Y.; Okihara, T.; Yamamoto, T.; Momoi, Y.; Van Meerbeek, B. Interference of functional monomers with polymerization efficiency of adhesives. Eur. J. Oral Sci. 2016, 124, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Yazdi, F.M.; Moosavi, H.; Atai, M.; Zeynali, M. Dentin bond strength and degree of conversion evaluation of experimental self-etch adhesive systems. J. Clin. Exp. Dent. 2015, 7, e243–e249. [Google Scholar] [CrossRef] [PubMed]
- Feitosa, V.P.; Sauro, S.; Ogliari, F.A.; Stansbury, J.W.; Carpenter, G.H.; Watson, T.F.; Sinhoreti, M.A.; Correr, A.B. The role of spacer carbon chain in acidic functional monomers on the physicochemical properties of self-etch dental adhesives. J. Dent. 2014, 42, 565–574. [Google Scholar] [CrossRef] [PubMed]
- Fujita-Nakajima, K.; Aoki-Tabei, N.; Arita, A.; Nishiyama, N. NMR study on the demineralization mechanism of the enamel and dentin surfaces in MDP-based all-in-one adhesive. Dent. Mater. J. 2018, 37, 693–701. [Google Scholar] [CrossRef] [PubMed]
- Kakuda, S.; Fu, J.; Nakaoki, Y.; Ikeda, T.; Tanaka, T.; Sano, H. Improved long-term bonding performance of an experimental all-in-one adhesive. Dent. Mater. J. 2013, 32, 600–607. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, H.; Tsubota, K.; Iwasa, M.; Ando, S.; Miyazaki, M.; Platt, J.A. Influence of Adhesive Application Time on Enamel Bond Strength of Single-step Self-etch Adhesive Systems. Oper. Dent. 2010, 35, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Mine, A.; De Munck, J.; Cardoso, M.V.; Van Landuyt, K.L.; Poitevin, A.; Kuboki, T.; Yoshida, Y.; Suzuki, K.; Van Meerbeek, B. Enamel-Smear Compromises Bonding by Mild Self-Etch Adhesives. J. Dent. Res. 2010, 89, 1505–1509. [Google Scholar] [CrossRef] [PubMed]
- Reis, A.; Leite, T.M.; Matte, K.; Michels, R.; Amaral, R.C.; Geraldeli, S.; Loguercio, A.D. Improving clinical retention of one-step self-etching adhesive systems, with an additional hydrophobic adhesive layer. J. Am. Dent. Assoc. 2009, 140, 877–885. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Takagaki, T.; Matsui, N.; Hamba, H.; Sadr, A.; Nikaido, T.; Tagami, J. Morphological Evaluation of the Adhesive/Enamel Interfaces of Two-step Self-etching Adhesives and Multimode One-bottle Self-etching Adhesives. J. Adhes. Dent. 2016, 18, 223–229. [Google Scholar] [PubMed]
- Beltrami, R.; Chiesa, M.; Scribante, A.; Allegretti, J.; Poggio, C. Comparison of shear bond strength of universal adhesives on etched and nonetched enamel. J. Appl. Biomater. Funct. Mater. 2016, 14, E78–E83. [Google Scholar] [CrossRef] [PubMed]
- Fujita Nakajima, K.; Nikaido, T.; Arita, A.; Hirayama, S.; Nishiyama, N. Demineralization capacity of commercial 10-methacryloyloxydecyl dihydrogen phosphate-based all-in-one adhesive. Dent. Mater. 2018, 34, 1555–1565. [Google Scholar] [CrossRef] [PubMed]
- Teshima, M. Effect of the concentration of water in an MDP-based all-in-one adhesive on the efficacy of smear layer removal and on dentin bonding performance. Dent. Mater. J. 2018, 37, 685–692. [Google Scholar] [CrossRef] [PubMed]
- Miyazaki, M.; Tsujimoto, A.; Tsubota, K.; Takamizawa, T.; Kurokawa, H.; Platt, J.A. Important compositional characteristics in the clinical use of adhesive systems. J. Oral Sci. 2014, 56, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Iwai, H.; Fujita, K.; Iwai, H.; Ikemi, T.; Goto, H.; Aida, M.; Nishiyama, N. Development of MDP-based one-step self-etch adhesive -Effect of additional 4-META on bonding performance. Dent. Mater. J. 2013, 32, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Fujita Nakajima, K.; Nikaido, T.; Francis Burrow, M.; Iwasaki, T.; Tanimoto, Y.; Hirayama, S.; Nishiyama, N. Effect of the demineralisation efficacy of MDP utilized on the bonding performance of MDP-based all-in-one adhesives. J. Dent. 2018, 77, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Kirihara, M.; Inoue, G.; Nikaido, T.; Ikeda, M.; Sadr, A.; Tagami, J. Effect of fluoride concentration in adhesives on morphology of acid-base resistant zones. Dent. Mater. J. 2013, 32, 578–584. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, Y.; Yoshihara, K.; Hayakawa, S.; Nagaoka, N.; Okihara, T.; Matsumoto, T.; Minagi, S.; Osaka, A.; Van Landuyt, K.; Van Meerbeek, B. HEMA inhibits interfacial nano-layering of the functional monomer MDP. J. Dent. Res. 2012, 91, 1060–1065. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Wurihan; Shibata, Y.; Tanaka, R.; Zhang, Z.; Zheng, K.; Li, Q.; Ikeda, S.; Gao, P.; Miyazaki, T. Quantitative/qualitative analysis of adhesive-dentin interface in the presence of 10-methacryloyloxydecyl dihydrogen phosphate. J. Mech. Behav. Biomed. Mater. 2019, 92, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Feitosa, V.P.; Pomacondor-Hernandez, C.; Ogliari, F.A.; Leal, F.; Correr, A.B.; Sauro, S. Chemical interaction of 10-MDP (methacryloyloxi-decyl-dihydrogen-phosphate) in zinc-doped self-etch adhesives. J. Dent. 2014, 42, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Garcia, M.G.; Poskus, L.T.; Hass, V.; Amaral, C.M.; Noronha-Filho, J.D.; Silva, E.M.D. Effect of Calcium Hydroxide on Bonding Performance of an Experimental Self-etch Adhesive. J. Adhes. Dent. 2018, 20, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.C.; Park, H.; Lee, S.I.; Kim, S.Y. Effect of the Acidic Dental Resin Monomer 10-methacryloyloxydecyl Dihydrogen Phosphate on Odontoblastic Differentiation of Human Dental Pulp Cells. Basic Clin. Pharmacol. Toxicol. 2015, 117, 340–349. [Google Scholar] [CrossRef] [PubMed]
- Putzeys, E.; Duca, R.C.; Coppens, L.; Vanoirbeek, J.; Godderis, L.; Van Meerbeek, B.; Van Landuyt, K.L. In-vitro transdentinal diffusion of monomers from adhesives. J. Dent. 2018, 75, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Teshima, I. Degradation of 10-Methacryloyloxydecyl Dihydrogen Phosphate. J. Dent. Res. 2010, 89, 1281–1286. [Google Scholar] [CrossRef] [PubMed]
- Aida, M.; Odaki, M.; Fujita, K.; Kitagawa, T.; Teshima, I.; Suzuki, K.; Nishiyama, N. Degradation-stage effect of self-etching primer on dentin bond durability. J. Dent. Res. 2009, 88, 443–448. [Google Scholar] [CrossRef] [PubMed]
| P (Problem) | Permanent teeth with need for restoration. |
| I (Intervention) | Direct restoration with composite, using adhesives with 10-MDP. |
| C (Comparison) | Adhesives with different functional monomers other than 10-MDP. Different adhesives with 10-MDP monomer. |
| O (Outcome) | Capacity to create an acid-base resistant zone (ABRZ). Formation of nano-layered structures. Adhesive stability. |
| Database | Search Strategy |
|---|---|
| PubMed | (“methacryloyloxydecyl dihydrogen phosphate” OR “10-MDP” OR “Functional monomer*”) AND (“dental cements [Mesh]” OR “adhesive*” OR “bond*”). |
| Cochrane Library | (“methacryloyloxydecyl dihydrogen phosphate” OR “10-MDP” OR “functional monomer*”) AND (“adhesive*” OR “bond*”). |
| Web of Science | TS = (“methacryloyloxydecyl dihydrogen phosphate” OR “10-MDP” OR “FUNCTIONAL MONOMER*”) AND TS = (“adhesive*” OR “bond*”). |
| Embase | (‘methacryloyloxydecyl dihydrogen phosphate’:ti,ab,kw OR ‘10-mdp’:ti,ab,kw OR’functional monomer*’:ti,ab,kw) AND (‘adhesive*’:ti,ab,kw OR ‘bond*’:ti,ab,kw). |
| Inclusion Criteria | Studies on permanent teeth |
| Direct restorations | |
| Dental adhesives | |
| Exclusion Criteria | Studies on deciduous teeth |
| Indirect restorations | |
| Dental cements | |
| Adhesion to metal alloys, ceramics, posts | |
| Plaque inhibitors/Antibacterial activity | |
| Deproteinized dentin |
| Author, Year | Groups | Results |
|---|---|---|
| Yoshihara et al., 2011 [18] | T1*: mixed solution containing 15% 10-MDP C*: Clearfil SE Bond primer (Kuraray) | Nano-layering was stronger on dentin than on enamel; Rubbing the primer for 20 s enhanced nano-layering; Nano-layering was reduced with lower [MDP] |
| Yoshida et al., 2012 [19] | T1*: Clearfil SE Bond (Kuraray) T2*: Scotchbond Universal (3M ESPE) | Hybrid layer: T1* thicker than T2*; T1*—at the top of the hybrid layer, regular longitudinally layered structures, often curved; T2*—nano-layering near the tubule orifices where the adhesive infiltrated residual smear. |
| Yoshihara et al., 2013 [20] | T1: 2-MEP T2: 6-MHP T3*: 10-MDP T4: Adper Easy Bond (3M ESPE) T5*: All-Bond Universal (Bisco) T6*: Clearfil S3 Bond (Kuraray) T7*: Scotchbond Universal (3M ESPE) | T1—Results not obtainable (failure at preparation); T2—hybrid layer thinner than T3 and HAp-rich; T3*—thicker hybrid layer, intense nano-layering through the whole adhesive layer; Nano-layering formation for all 10-MDP based adhesives; T4 (6-MHP) only formed some nano-layering; In contrast to T4, the 3 commercially available 10-MDP containing adhesives revealed the 3 characteristic nano-layering peaks (XRD) already after 20 s interaction. |
| Hiraishi et al., 2014 [21] | T1*: 10-MDP T2: 4-META | 10-MDP long chain makes it quite hydrophobic; Atelocollagen and MDP tend to aggregate in water; Reduction in the STD intensity when HEMA was added to T1*, resulting in a weak interaction with atelocollagen. |
| Yokota et al., 2015 [22] | T1*: Experimental adhesive | Several types of MDP-Ca salts and amorphous DCPD were developed during decalcification; Enamel and dentin produced MCS-MM and MD; Dentin produced DCS-MD. |
| Tian et al., 2016 [6] | T1*: 5% 10-MDP primer T2*: 10% 10-MDP primer T3*: 15% 10-MDP primer T4*: Adhese Universal (Ivoclar-Vivadent) T5*: All-Bond Universal (Bisco) T6*: Clearfil S3 Bond Plus (Kuraray) T7*: Clearfil SE Bond 2 (Kuraray) T8*: Clearfil Universal Bond (Kuraray) T9*: G-Premio Bond (GC Corp.) T10*: Scotchbond Universal (3M ESPE) | Nano-layering became sparser with reduction in [MDP] (T3* > T1*, T2*); Nano-layering was identified in limited sites when using T6* and T7*; T4*, T5*, T8*, T9*, T10*: no nano-layered structures were identified; T7*: limited and less well-organized patterns of nano-layering when compared to pure 10-MDP. |
| Yaguchi, 2017 [5] | T1*: 25.6 mg T2*: 49.9 mg T3*: 80.5 mg T4*: 116.1 mg (quantity of 10-MDP in 1g of the experimental adhesive) | ↑ [10-MDP] on enamel led to ↑ production of MCS-MD and ↓ production of MCS-MM, and then it leveled; Dentin produced ↑ [MCS-MD] and [DCS-MD] than enamel did (p < 0.05); ↑ [10-MDP] on dentin led to ↑ production ratios of both MCS-MM and MCS-MD; Dentin showed a greater production of MDP-Ca salts than enamel did (p < 0.05); Dentin produced greater amounts of mono- and di-calcium salts of the MDP dimer that were able to form nano-layered structures; dentin and enamel predominantly produced a mono-calcium salt. |
| Author, Year | Groups | Results |
|---|---|---|
| Na li et al., 2010 [10] | C*: 10-MDP in primer and bond (Clearfil SE Bond, Kuraray) T1*: 10-MDP (primer) and Phenyl-P (bond) T2*: Phenyl-P (primer) and 10-MDP (bond) T3: Phenyl-P in primer and bond | Enamel ABRZ thickness (µm): C*: 0.5; T1*: <0.2; T2*: 1; T3: <0.1. T1* ABRZ morphology similar to C* but sparser distribution of crystals; T2* crystals with ↑ length, but similar width (ABRZ), with clear intercrystallite spaces. T3 had low capacity to create an ABRZ and presence of funnel-shaped erosions; Adhesive interface produced by 10-MDP containing adhesive systems remained after acid-base challenge. |
| Nikaido et al., 2011 [2] | T1*: 10-MDP T2: 3D-SR T3: 4-META (similar compositions, different functional monomers, all universal adhesives) | Dentin ABRZ thickness: T1* > T2 > T3; Enamel ABRZ is very thin, compared to dentin ABRZ; Enamel ABRZ thickness < 0.5 µm in all groups but for T1* it appeared to be thicker. Dentin ABRZ formed under the hybrid layer, while enamel ABRZ was created along the interface between adhesive and enamel; ABRZ was confirmed at both enamel and dentin; it was influenced by the functional monomer contained in the adhesive system; Funnel-shaped erosion found at bonding interface between enamel and outer lesion in T3. |
| Nurrohman et al., 2012 [24] | C: Scothbond multi-purpose (3M ESPE) T2*: Clearfil photo bond (Kuraray) T3*: Clearfil SE Bond (Kuraray) T4: Adper Easy Bond (3M ESPE) | C: 4 µm HL and some regions with absence of a crystalline phase; deep funnel-shaped lesion into intact dentin; similar lesion in T2*; T2*: 5 µm HL and regions with low density and partially dissolved apatite crystals; T3*: 1 µm HL and denser overall crystallite arrangement in the base of the HL; approximately 0.5 µm thick ABRZ with densely arranged crystals and no funnel-shaped lesions in all specimens of this group; T4: partially demineralize HL, approximately 0.5 µm; funnel-shaped lesions along the apatite-rich zone. |
| Matsui et al., 2015 [23] | C*: Clearfil SE Bond (Kuraray) T1*: Experimental adhesive (10-MDP in primer) | Dentin ABRZ formed beneath the HL in both groups; Funnel-shaped erosion observed at the junction of dentin and bonding layer in T1*; Excluding 10-MDP from the bonding resin resulted in ↓ resistance against acid attack at ABRZ. |
| Nikaido et al., 2015 [26] | T1*: Clearfil SE Bond (Kuraray) T2*: Clearfil Bond SE One (Kuraray) T3: G-Bond Plus (GC) | T3 ABRZ was the thinnest (p < 0.05), and had the highest NL (p < 0.05); Funnel-shaped lesion not observed for T1*. |
| Guan et al., 2016 [25] | T1*: Clearfil SE Bond 2 (Kuraray) T2: Optibond XTR (KERR) T3*: Scotchbond Universal (3M ESPE), applied as SE, ERM (Moist) and ERD (Dry) | ABRZ at the front of demineralization for SE groups; Slope at bottom of outer lesion in T2; T3*SE: funnel-shaped lesion at bottom of outer lesion; T3*ERM and T3*ERD: 5 µm HL without appearance of ABRZ. |
| Author, Year | Groups | Results | Comments |
|---|---|---|---|
| Hayakawa et al., 1998 [27] | T1: 5% Phenyl-P + 60% H2O T2: 10% Phenyl-P + 55% H2O T3: 20% Phenyl-P + 45% H2O T4: 30% Phenyl-P + 35% H2O T5*: 5% 10-MDP + 60% H2O T6*: 10% 10-MDP + 55% H2O T7*: 20% 10-MDP + 45% H2O T8*: 30% 10-MDP + 35% H2O | Dentin T3, T4—30 s treatment: ↑TBS than T1 and T2 (p < 0.05); Dentin T8*—15 s treatment: ↑adhesion than T1, T2, T3 (p < 0.05); Dentin T8*—60 s treatment: ↑adhesion than T5*; Different patterns after treatment with T1/T4 and T5*/T8*. | Adhesives partially dissolved the smear layer which restricted the resin penetration. Monomers could infiltrate into the dentin to create the hybrid layer, resulting in a tight adhesion to dentin; Insufficient infiltration of monomers into the dentin, preserving more of the smear layer, resulted in lower BS. |
| Inoue et al., 2005 [28] | T1*: Clearfil SE Bond (Kuraray) T2: Unifil Bond (GC) T3: Clearfil Liner Bond II (Kuraray) | T1*: µTBS to dentin after 100,000 thermocycles = 0 thermocycles; T2: ↓µTBS (41%) after 100,000 thermocycles; T3: ↓µTBS (48%) after 30,000 and 100,000 thermocycles; HAp crystals remained at the hybrid layer (T1* > T2 > T3). | Long-term durability of the dentin-adhesive interface of two-step self-etching adhesives differed, depending on the particular adhesive; T1* showed no signs of degradation in bond strength and interfacial ultrastructure. |
| Na Li et al., 2010 [10] | C*: Clearfil SE Bond (Kuraray) T1*: 10-MDP (primer) and Phenyl-P (bond) T2*: Phenyl-P (primer) and 10-MDP (bond) T3: Phenyl-P in primer and bond | C*: ↑BS than the other groups (p < 0.005); T1*, T2*, T3: no differences in BS (p > 0.05); Significant distribution of failure modes among groups (p < 0.05); C*, T1*, T2*: adhesive and cohesive failure, while major failure of T3 was adhesive failure; Micro-shear bond-strength values showed ↑bond strength in C (p < 0.005). Among test groups, no significant difference was found. | |
| Fujita et al., 2011 [29] | T1*: Clearfil Tri-S Bond (Kuraray) T2*: Clearfil SE Bond (Kuraray) | T2*: ↑ [reacted 10-MDP] (16.1%) compared to T1 (9.2%); T1*: blank outline of the enamel prisms; dentinal tubes were widened, with deposits on the intertubular dentin, without exposure of collagen fibrils; T2*: typical etching pattern on enamel; dentinal tubes were more widened and blocked by precipitates, with collagen fibrils exposed; conditioning of enamel and dentin allowed enhancement in the initial BS (p < 0.05); a reduction was observed in conditioned dentin after 20,000 thermocycles. | Superior BS of T2* correlated to the demineralized amount of tooth apatite by 10-MDP; Unreacted 10-MDP polymer within the adhesive layer did not ↓ the bond strength, despite application of 20,000 thermocycles. |
| Harnirattisai et al., 2012 [30] | T1*: Clearfil SE Bond (Kuraray) T2*: Clearfil Tri-S Bond (Kuraray) T3: G-Bond (GC) T4: i-Bond (Kulzer) (T2, T3, T4: all-in-one adhesives) | Bond strength at 10 min was lower than that at 24 h for all adhesives; T1*: ↑bond strength (10 min and 24 h); SBt: ↑adhesive failure (66.04–97.44%) for all-in-one adhesives, compared to T1* (10 min and 24 h); µSBt: ↑cohesive failures in resin; µSBt: T1* > T4 > T2* and T1 = T3. | Dentin cohesive failure was found to be lower in the µSBt of T1 at 24 h; µSBt results in divergency of behavior between systems, not seen with SBt; |
| Iwai et al., 2012 [12] | T1*: 0 mg T2*: 25.6 mg T3*: 49.9 mg T4*: 80.5 mg T5*: 116.1 mg (quantity of 10-MDP in 1g of experimental adhesive) | ↑ [10-MDP] resulted in ↑amounts of MDP-Ca salts, which resulted in ↑BS for enamel and dentin; Further ↑ in the amount of MDP-Ca salt resulted in ↓BS. | |
| Zhang et al., 2013 [8] | C: Durafill Bond (Heraeus Kulzer) T1*: MDP/HEMA/Bis-GMA (1:1:1) (Kuraray) T2*: MDP/HEMA/Bis-GMA (2:1:1) (Kuraray) T3*: MDP/Bis-GMA (1:1) (Kuraray) | µTBS: C lower than test groups (24 h and 1 year water-storage) (p < 0.05); No differences between test groups (p > 0.05); C: clearly visible enamel HAp crystallites partly and adhesive mixed with fractured HAp crystallites partly (solely micromechanical interlocking at the interface); Test groups: Faintly visible enamel HAp crystallites partly. | Etched enamel surfaces treated with the MDP-containing primers revealed that the etched enamel surfaces were covered by a layer of variable network-like/fibril-like HAp crystallites; C: cannot chemically react with HAp |
| Feitosa et al., 2014 [13] | T1: MEP T2*: MDP T3: MDDP T4: CAP-P T5: MTEP | T2*, T3: lowest free-calcium concentrations (p < 0.001); T1 had the highest; Monomer-Ca salt on dentin present in all groups; T2*, T3: ↑µTBS than T1, T4, T5 (p < 0.05). | Formation of monomer-Ca salts and initial BS were influenced by the length and hydrophilicity of the spacer chain of functional monomers. |
| Feitosa et al., 2014 [15] | T1: MEP T2*: MDP T3: MDDP T4: CAP-P T5: MTEP | T1: lowest monomer-calcium formation (p < 0.05); T2*, T3: ↑µTBS than those of T1, T4, T5; After 1-year aging: drop in µTBS was observed for T5 (enamel and dentin), T1 (enamel) and T4 (enamel) (p < 0.005); T5: highest micro-permeability; T1, T4, T5: ↑NL after aging. | Length and hydrophilicity of the spacer chain influenced the monomer-calcium salt formation, the dentin/enamel bonding performance, the interfacial micro-permeability and NL. |
| Takahashi, 2014 [31] | T1*: 0 g; T2*: 3.0 g; T3*: 6.0 g; T4*: 10.0 g; T5*: 15.0 g. (quantity of 10-MDP in 1 g of the experimental adhesive) | T1*: thermocycling led to a ↓ in the BS, with no MDP-Ca salt produced (p < 0.05); ↑ of MDP-Ca salts to above:
| |
| Anchieta et al., 2015 [32] | C: Scotchbond Multi-Purpose (3M ESPE) T1*: Clearfil SE Bond (Kuraray) T2: One Up Bond F (Tokuyama) T3: Adper Easy One (3M ESPE) T4: Filtek LS adhesive (3M ESPE) | C: thickest hybrid layer (p < 0.05); longest resin tags ( = T2) (p < 0.05); T1*: thinnest hybrid layer; highest elastic modulus of the hybrid layer (p < 0.05); T2: thinnest adhesive layer (p < 0.05); highest degree of silver impregnation at 24 h (p < 0.05); T3, T4: highest infiltration (p < 0.05). T4: thickest adhesive layer; Storage for 12 months ↑ silver impregnation for all groups (p < 0.05), except for T1* (p > 0.05); ↓Elastic modulus along time in all groups (p < 0.05); ↑ NL over time except for T1*. | Partially demineralized dentin below the hybrid layer occurred for all adhesives; After 12 months storage, degradation occurred at the DAI in all groups and the intensity of degradation differed depending on the type of adhesive used; 10-MDP containing adhesive system (T1*) DAI formed showed the best stability among all adhesive systems. |
| Matsui et al., 2015 [23] | T1*: Clearfil SE Bond (Kuraray) T2*: Experimental adhesive with 10-MDP primer | T1* µTBS > T2* µTBS without thermocycling (p < 0.001); after thermocycling: T2* > T1* (p < 0.001); T2* µTBS remained stable after thermocycling (p < 0.001); UTS: T2* > T1* in all evaluation periods; ↓ UTS after storage in water. | |
| Muñoz et al., 2015 [33] | C1: Adper Single Bond 2 (3M ESPE) C2*: Clearfil SE Bond (Kuraray) T1: Peak Universal Adhesive System (Ultradent Products Inc.) T2*: Scotchbond Universal Adhesive (3M ESPE) T3*: All Bond Universal (Bisco) C1, T1, T2 and T3 as ER C2, T1, T2 and T3 as SE | Most of the specimens showed adhesive or adhesive/mixed failures; T1 (SE) and T1 (ER) showed the ↑ immediate µTBS, similar to C1, C2* (p > 0.05) with a ↓ after 6-months of water storage (p > 0.05); T2* (SE), T2* (ER), T3* (SE), T3* (ER): lower immediate µTBS, compared to C1, C2* (p < 0.05); ER: only T3* had lower µTBS after 6-months (p < 0.05); T1: highest NL at immediate time (p < 0.05), ↑ after 6 months (p < 0.05); | Universal adhesives demonstrated heterogenous behavior, since some adhesives diminished the bonding performance over the course of time. |
| Yoshihara et al., 2015 [11] | Three 10-MDP molecules by different companies: T1*: 83% purity T2*: 90% purity T3*: ↑% than T1 and T2 | T1*: µTBS did not ↓ after 100,000 thermocycles, contrarily to T1* and T2*; T3*: ↑Immediate µTBS than T1*, T2*; No pre-testing failure recorded for T3*, but several failures happened with the “aged specimens” of T1* and T2*. | Differences in the ultrastructure of the hybrid layer were observed between the different monomers used. |
| Chen et al., 2015 [36] | T1: Prime and Bond Elect (Dentsply) T2*: Scotchbond Universal (3M ESPE) T3*: All Bond Universal (Bisco) T4*: Clearfil Universal Bond (Kuraray) T5: Futurabond U (VOCO) | Comparisons between test groups were all significant (p < 0.01), except between T1/T2*, T1/T3*, T2*/T3*, T4*/T5; T2* (p = 0.004), T4* (0.006) and T5 (p < 0.001) had different results between with and without thermocycling; T1 and T3* were resistant to thermocycling (p > 0.01). | |
| Farias et al., 2016 [37] | T1*: Scotchbond Universal (3M ESPE) T2*: All Bond Universal (Bisco) T3: Optibond FL (Kerr) T4: Adper Single Bond Plus (3M ESPE) T5*: Clearfil SE Bond (Kuraray) T6: Adper Prompt L-Pop (3M ESPE) | Similar µTBS means, before and after thermocycling for T1*, T2*, T3, T4 (p < 005); Before thermocycling: similar µTBS means between groups: T1*/T2*ER/T3/T4/T5* (p < 0.05), T1*SE/T2*SE/T3/T6(p < 0.05) T1*SE/T2*SE/T3/T5* (p < 0.05). After thermocycling: similar µTBS means between groups T1*/T2*ER/T3/T4/T5* and T1*SE/T2*SE/T3/T4. | |
| Tsuchiya et al., 2016 [38] | T1*: Clearfil SE Bond (Kuraray) T2: Experimental adhesive (equal to T1*, without MDP) | T1*: ↑SB (p < 0.05) with pre-etching, for same storage period; ↑SB at 6-months and 1-year storage; ↑SFS (p < 0.05) with pre-etching for same storage period; ↑SB for both test groups at 6 months storage. | |
| Zhang et al., 2016 [1] | T1*: All-Bond Universal (Bisco) T2*: Clearfil Universal Bond (Kuraray) T3: Futurabond U (VOCO) T4: Prime&Bond Elect (Dentsply) T5*: Scotchbond Universal (3M ESPE) | µTBS was affected by the bonding strategy and aging tests (p < 0.005); 12 months: ↑µTBS for T1* as ER (p < 0.001), while for the rest of the groups was ↑ when in SE mode (p < 0.001); T1* to T5*: ↓µTBS when in ER mode (p < 0.001); T1*, T2*, T3: ↓µTBS when in SE mode (p < 0.001); T4, T5*: no changes in µTBS between 24 h and 12 months. | Universal adhesive systems with 10-MDP monomer did not show better performance than those without; Bonds created in SE mode were more durable than those created in ER mode; With exception of bonds created by T4 and T5, universal adhesives at test were incapable of defying ageing. |
| Thanatvarakorn et al., 2016 [34] | T1*: Clearfil SE Bond (Kuraray) T2*: Scotchbond Universal (3M ESPE) T1*s, T2*s: applied with scrubbing technique T1*ns, T2*ns: passively applied | T1*s did not affect µTBS (p > 0.05); T2*s exhibited ↑µTBS than T2*ns (p < 0.05); T2*ns: adhesive failure at adhesive interface was predominant and larger than in other groups; T2*s had the highest etching ability, while T2*ns had the lowest; T1*s and T2*s were free of NL. | Scrubbing technique not only improved immediate µTBS but also ↑ the stability of a one-step self-etching adhesive bond to dentin. |
| Guan et al., 2016 [25] | T1*: Clearfil SE Bond 2 (Kuraray) T2: Optibond XTR (KERR) T3*: Scotchbond Universal (3M ESPE), applied as SE, ERM (Moist) and ERD (Dry) | ↓ 24 h BS of ER than SE groups (p < 0.05); 5000 thermal cycles: ↓µTBS of T3*ERM (p = 0.001); 10,000 thermal cycles: T1* µTBS remained stable, T2 ↑ and all T3* ↓ (p < 0.05); Immediate BS of T3*ERD was lower than other groups (p < 0.05); 10,000 thermal cycles: ↓BS on SE and ER (p < 0.05); | |
| Tsujimoto et al., 2017 [39] | T1*: Clearfil Universal Bond (Kuraray) T2*: G-Premio Bond (GC) T3*: Scotchbond Universal (3M ESPE) T4*: Clearfil SE Bond (Kuraray) T5*: Clearfil SE Bond 2 (Kuraray) T6: Optibond XTR (Kerr) | Initial BS: T6 > T4* > T5* > T3* > T1* > T2*; SFS: T6 > T4* > T5* > T3* > T1* > T2*; Initial BS of universal adhesives is influenced by the type of adhesive, but lower than that of two-step self-etching adhesive systems. | |
| Wang et al., 2017 [35] | T1*: Clearfil SE Bond (Kuraray) T2*: Scotchbond Universal (3M ESPE) T3: Optibond XTR (Kerr) T4: Adper Easy Bond (3M ESPE) | TF-XRD: T1* and T2* revealed production of 10-MDP-Ca salts; T2*: slightly shifted and ↓intensity; no detected peaks in T3 and T4; SEM: T1*, T2*: after ethanol rinsing most of the adhesive was retained; T4: smear debris remained; T3: all of the hybrid layer was removed; T1* and T2* µTBS stable before and after thermocycling (p > 0.05); Dentin µTBS: T2* and T3 ↑ than T1* and T4 at 24 h; T4 ↓ after thermocycling and T3 after aging; NL: T1 and T2—slight ↑ impregnation after thermocycling; T3 and T4: ↑ infiltration after thermocycling and in many cases the entire length of the hybrid layer was infiltrated. | Differences in T1* and T2* for TF-XRD analysis are related to the ratio of 10-MDP contained in each formulation; T2* and T3 gained ↑ bonding strength even after aging than the traditional T1* and T4, although T3 showed ↑ NL after thermocycling. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carrilho, E.; Cardoso, M.; Marques Ferreira, M.; Marto, C.M.; Paula, A.; Coelho, A.S. 10-MDP Based Dental Adhesives: Adhesive Interface Characterization and Adhesive Stability—A Systematic Review. Materials 2019, 12, 790. https://doi.org/10.3390/ma12050790
Carrilho E, Cardoso M, Marques Ferreira M, Marto CM, Paula A, Coelho AS. 10-MDP Based Dental Adhesives: Adhesive Interface Characterization and Adhesive Stability—A Systematic Review. Materials. 2019; 12(5):790. https://doi.org/10.3390/ma12050790
Chicago/Turabian StyleCarrilho, Eunice, Miguel Cardoso, Manuel Marques Ferreira, Carlos Miguel Marto, Anabela Paula, and Ana Sofia Coelho. 2019. "10-MDP Based Dental Adhesives: Adhesive Interface Characterization and Adhesive Stability—A Systematic Review" Materials 12, no. 5: 790. https://doi.org/10.3390/ma12050790
APA StyleCarrilho, E., Cardoso, M., Marques Ferreira, M., Marto, C. M., Paula, A., & Coelho, A. S. (2019). 10-MDP Based Dental Adhesives: Adhesive Interface Characterization and Adhesive Stability—A Systematic Review. Materials, 12(5), 790. https://doi.org/10.3390/ma12050790








