Respirometric Study of Optical Brighteners in Textile Wastewater
Abstract
1. Introduction
2. Materials and Methods
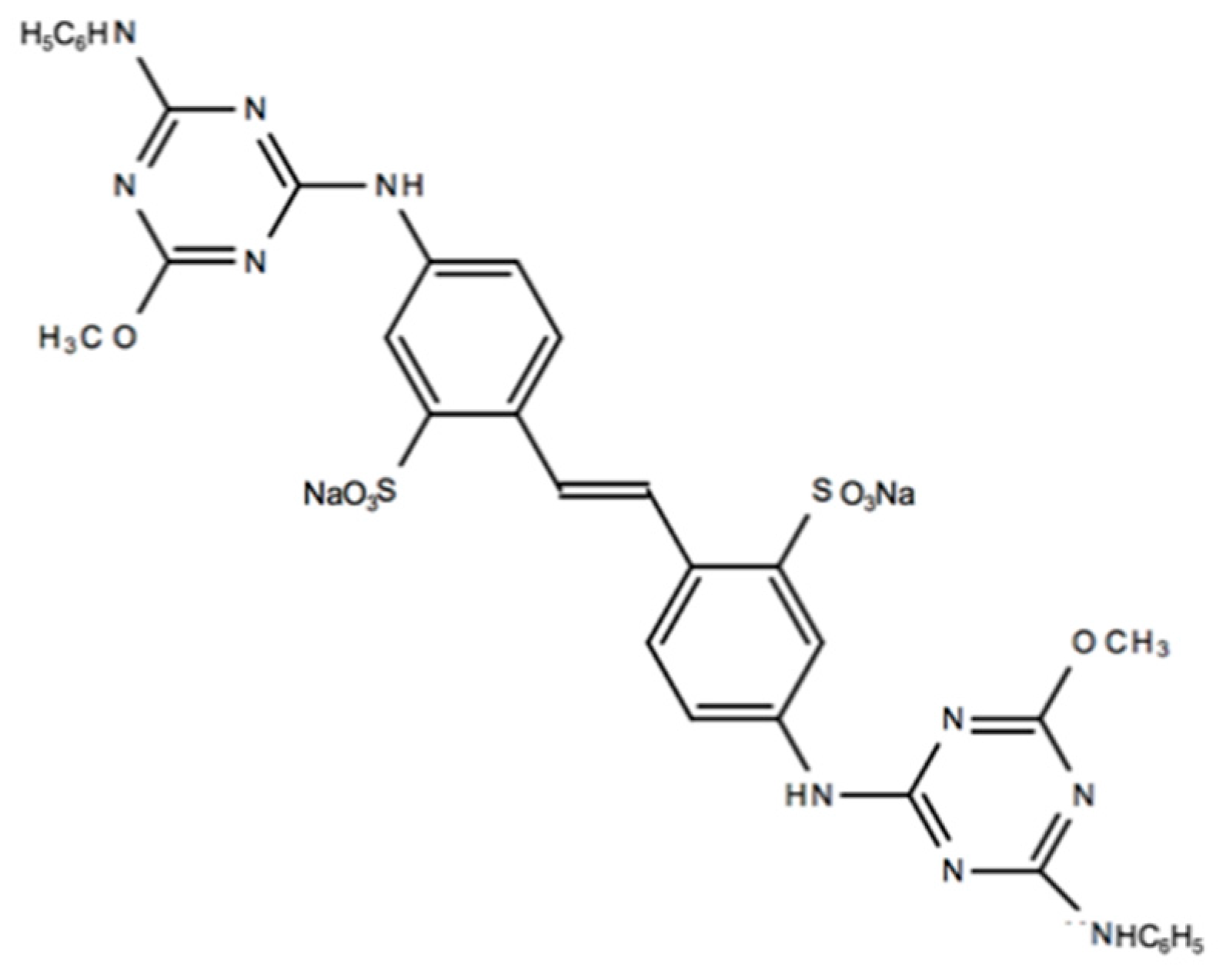
- Fluorescent Brightener 134, commercial name Leucophor PC (Clariant Iberica, Sant Joan Despí, Spain) and identified in this document as “L-PC”. It is a diaminostilbene-disulphonic acid derivate product with CAS Number 3426-43-5. It can be found in the Colour Index as Fluorescent Brightener N/A 134. Its chemical structure is presented in Figure 1.
- A commercial optical brightener provided by Golden Technology Inc. (Golden Technology, São Paulo, Brazil), named Goldblanc BHA and referred in this document as “BHA”. It is a derivative of 4,4′-diaminostilbene-2,2′-disulfonic acid (DSD acid). This OB is currently used in the textile industry. It was not possible to obtain more information beyond the data provided by the manufacturer. For this product, the full chemical composition and structure has not been published because it is confidential information of the provider.
2.1. Organic Load of OBs
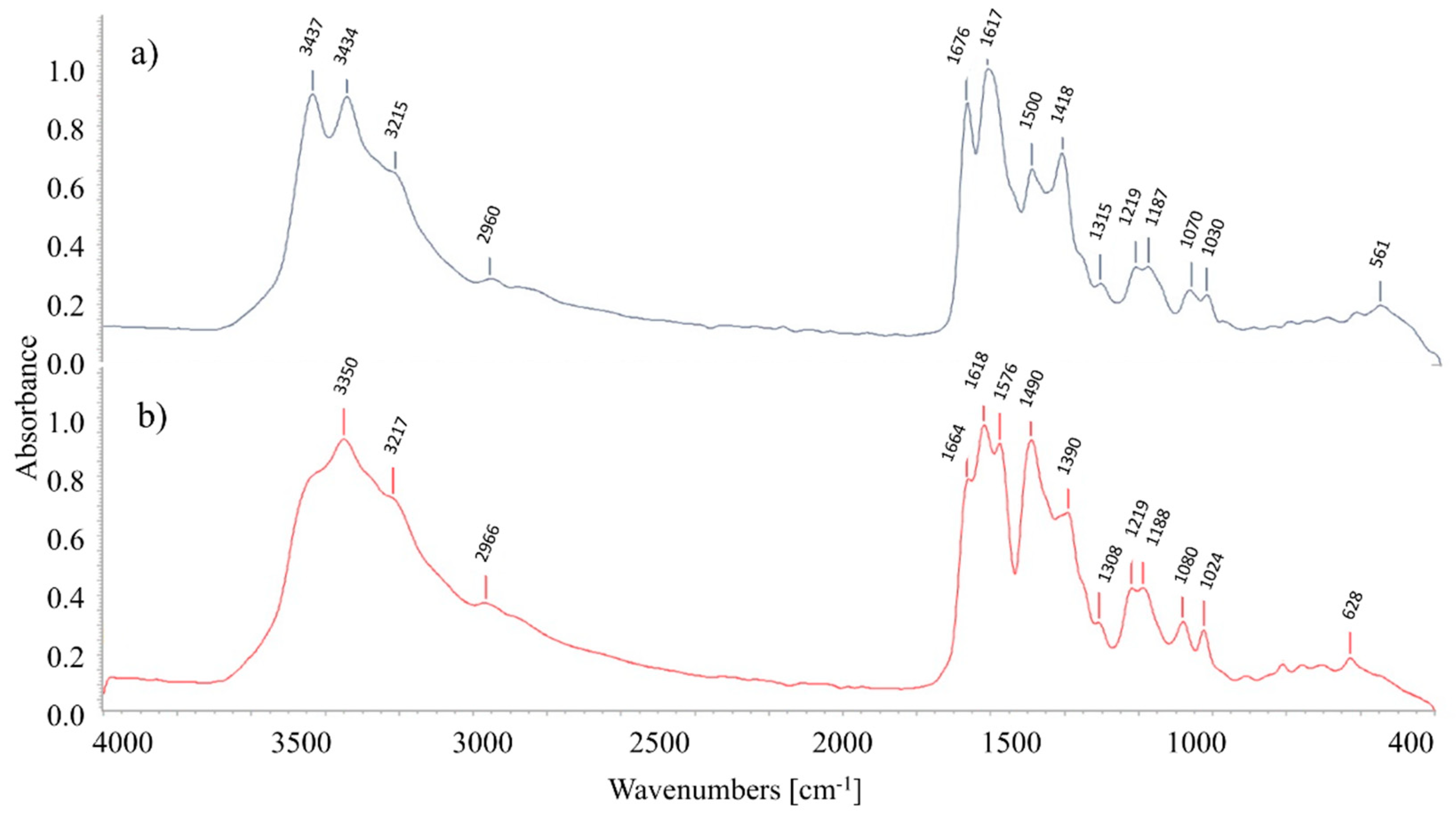
2.2. FTIR Analysis
2.3. Toxicity
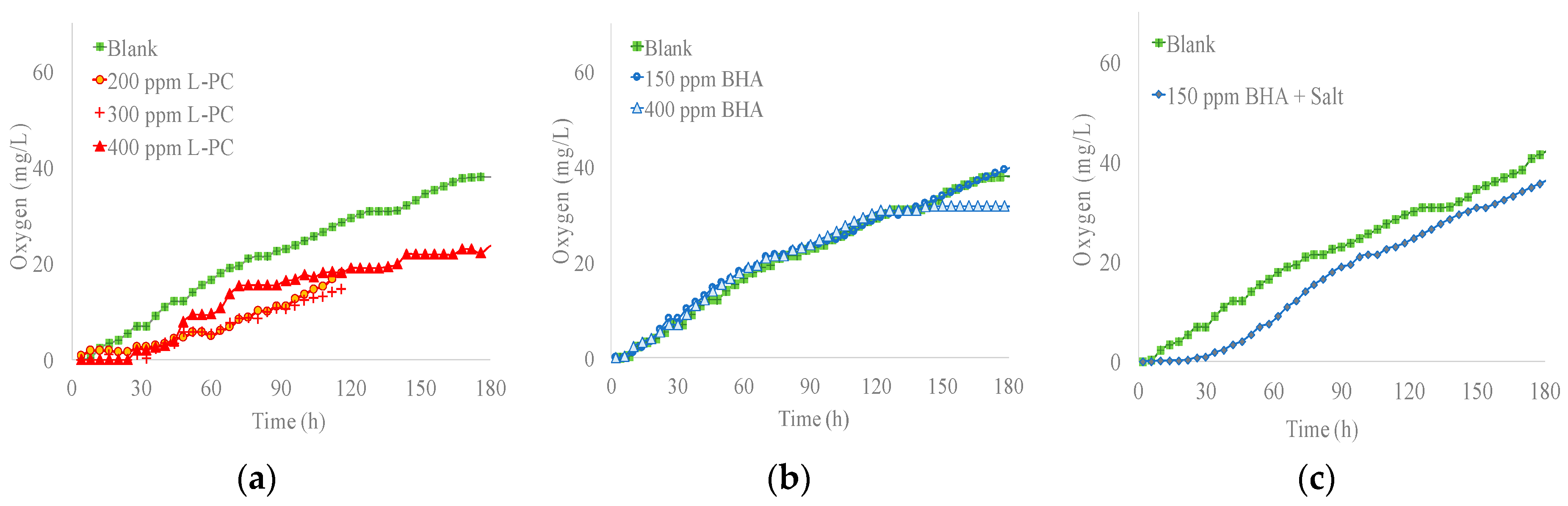
2.4. Respirometry
3. Results and Discussion
3.1. Organic Load Results
3.2. FTIR Analysis Results
3.3. Toxicity Results
3.4. Respirometry Results
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kabir, S.M.F.; Iqbal, M.I.; Sikdar, P.P.; Rahman, M.M.; Akhter, S. Optimization of parameters of cotton fabric whiteness. Eur. Sci. J. 2014, 10, 200–210. [Google Scholar]
- Tiki, A.; Amin, A.; Kanwal, A. Chemistry of optical brighteners and uses in textile industries. Pak. Text. J. 2010, 59, 42–43. [Google Scholar]
- Karmakar, S.R. Chapter 15 Application of biotechnology in the pre-treatment processes of textiles. In Chemical Technology in the Pre-Treatment Processes of Textiles; Elsevier Science: Amsterdam, The Netherlands, 1999; Volume 12, pp. 418–440. ISBN 044450060X. [Google Scholar]
- Ciba Specialty Chemicals. Optical Brighteners—Fluorescent Whitening Agents for Plastics, Paints, Imaging and Fibers; 016263.040; Ciba: Basel, Switzerland, 1999. [Google Scholar]
- Bueno, L.; Amador, C.; Bakalis, S. Modeling the deposition of fluorescent whitening agents on cotton fabrics. AIChE J. 2017, 64, 1305–1316. [Google Scholar] [CrossRef]
- Assaad, A.; Pontvianne, S.; Pons, M.N. Photodegradation-based detection of fluorescent whitening agents in a mountain river. Chemosphere 2014, 100, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Jackson, A. Optical brightening agents: Past, present and future. Pulp Pap. Int. 2009, 51, 32–35. [Google Scholar]
- Han, J.; Tang, X.; Wang, Y.; Li, J.; Ni, L.; Wang, L. A Simple two-step cloud point extraction process for removing fluorescent whitening agents VBL in industrial wastewater and recycling of surfactant. Water Environ. Res. 2017, 89, 281–287. [Google Scholar] [CrossRef] [PubMed]
- Poiger, T.; Jennifer, A.; Field, M.; Field, T.; Siegrist, H.; Giger, W. Behavior of fluorescent whitening agents during sewage treatment. Water Res. 1998, 32, 1939–1947. [Google Scholar] [CrossRef]
- Scheringer, M.; Halder, D.; Hungerbühler, K. Comparing the environmental performance of fluorescent whitening agents with peroxide bleaching of Mechanical Pulp. J. Ind. Ecol. 1999, 3, 77–95. [Google Scholar] [CrossRef]
- Santa Cruz Biotechnology Fluorescent Brightener 134—Ficha de Datos de Seguridad. Available online: http://datasheets.scbt.com/sds/eghs/es/sc-495741.pdf (accessed on 16 October 2018).
- Water Analysis—Guidelines for the Determination of Total Organic Carbon (TOC) and Dissolved Organic Carbon (DOC); EN 1484:1997; European Committee for Standardization, CEN: Brussels, Belgium, 1997.
- ISO 6060:1989-Water Quality—Determination of the Chemical Oxygen Demand; International Organization for Standardization: Geneva, Switzerland, 1989.
- APHA-AWWA-WEF Standard Methods for the Examination of Water and Wastewater, 22nd ed.; American Public Health Association: Washington, DC, USA, 2012.
- Vilaseca Vallvè, M.M. Observación microscópica de fangos activados en los tratamientos de depuración biológica. Bol. Intexter Inst. Investig. Text. Coop. Ind. 2001, 119, 67–74. [Google Scholar]
- ISO 11348-3:2007-Water Quality—Determination of the Inhibitory Effect of Water Samples on the Light Emission of Vibrio Fischeri (luminescent Bacteria Test)—Part Ш: Method Using Freeze-Dried Bacteria. ISO/TC 147/SC5—Water Quality Methods; International Organization for Standardization: Geneva, Switzerland, 2007.
- Vilaseca, M.M.; Gutiérrez, M.C.; Crespi, M. Biodegradability of textile effluents treated with electrochemical oxidation techniques. Bol. Intexter Inst. Investig. Text. Coop. Ind. 2003, 123, 45–50. [Google Scholar]
- Idel-aouad, R.; Valiente, M.; Gutiérrez-Bouzán, C.; Vilaseca, M.; Yaacoubi, A.; Tanouti, B.; López-Mesas, M. Relevance of Toxicity Assessment in Wastewater Treatments: Case Study—Four Fenton Processes Applied to the Mineralization of C.I. Acid Red 14. J. Anal. Methods Chem. 2015, 2015, 945489. [Google Scholar] [CrossRef] [PubMed]
- Abbas, M.; Adil, M.; Ehtisham-ul-haque, S.; Munir, B.; Yameen, M. Vibrio fischeri bioluminescence inhibition assay for ecotoxicity assessment: A review Science of the Total Environment Vibrio fi scheri bioluminescence inhibition assay for ecotoxicity assessment: A review. Sci. Total Environ. 2018, 626, 1296–1305. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Yediler, A.; Lienert, D.; Wang, Z.; Kettrup, A. Toxicity evaluation of reactive dyestuffs, auxiliaries and selected effluents in textile finishing industry to luminescent bacteria Vibrio fischeri. Chemosphere 2002, 46, 339–344. [Google Scholar] [CrossRef]
- Gottlieb, A.; Shaw, C.; Smith, A.; Wheatley, A.; Forsythe, S. The toxicity of textile reactive azo dyes after hydrolysis and decolourisation. J. Biotechnol. 2003, 101, 49–56. [Google Scholar] [CrossRef]
- Vilaseca, M.; Gutiérrez, M.-C.; López-Grimau, V.; López-Mesas, M.; Crespi, M. Biological treatment of a textile effluent after electrochemical oxidation of reactive dyes. Water Environ. Res. 2010, 82, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Bioscienceinc.com MICROCAT Microbial Products—Biological Wastewater Treatment. Available online: https://www.bioscienceinc.com/microbials/ (accessed on 29 May 2018).
- Vilaseca, M. Respirometría electrolítica en el diseño y explotación de depuradoras. Bol. Intexter Inst. Investig. Text. Coop. Ind. 2007, 132, 51–56. [Google Scholar]
- Algaba, I.M.; Pepió, M.; Riva, A. Modelization of the influence of the treatment with two optical brighteners on the ultraviolet protection factor of cellulosic fabrics. Ind. Eng. Chem. Res. 2007, 46, 2677–2682. [Google Scholar] [CrossRef]
- Zaher, K.; Hammam, G. Correlation between biochemical oxygen demand and chemical oxygen demand for various wastewater treatment plants in Egypt to obtain the biodegradability indices. Int. J. Sci. Basic Appl. Res. 2014, 13, 42–48. [Google Scholar]
- Metcalf & Eddy Inc. Wastewater Engineering: Treatment and Reuse, 4th ed.; McGraw-Hill Education: New York, NY, USA, 2003; ISBN 0-07-041878-0. [Google Scholar]
- Pretsch, E.; Buhlmann, P.; Badertscher, M. Structure Determination of Organic Compounds, 4th ed.; Springer: Berlin, Germany, 2009; ISBN 978-3-540-93809-5. [Google Scholar]
- Guo, M.; Zhang, G.; Du, L.; Zheng, H.; Liu, G. Water-soluble, antiyellowing polymeric fluorescence brighteners. J. Appl. Polym. Sci. 2017, 134, 45536. [Google Scholar] [CrossRef]
- Hanyz, I.; Ion, R.M.; Nuta, A.; Wróbel, D. Spectral characterization of selected stilbentriazine dyes-Structural trans-cis isomerisation. J. Mol. Struct. 2008, 887, 165–171. [Google Scholar] [CrossRef]
- Guo, M.; Zhang, G.; Du, L.; Zheng, H.; Liu, G.; Li, Y. Multifunctional water-soluble polymeric fluorescent whitening agents based on 4,4′-bis (1,3,5-Triazinyl)-diamino stilbene-2,2′-disulfonic acid structure. BioResources 2017, 12, 8901–8919. [Google Scholar] [CrossRef]
- Saeed, A.; Shabir, G.; Batool, I. Novel stilbene-triazine symmetrical optical brighteners: Synthesis and applications. J. Fluoresc. 2014, 24, 1119–1127. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Zhou, J.; Wang, D.; Tian, C.; Wang, P.; Uddin, M.S. A novel moderately halophilic bacterium for decolorizing azo dye under high salt condition. Biodegradation 2008, 19, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Rao, R.N.; Venkateswarlu, N. Removal of amino and nitro-substituted stilbenesulfonic acids from aqueous environment: Biosorption and biodegradation by isolated Aspergillus awamori (Nakazawa). Process Biochem. 2006, 41, 1097–1105. [Google Scholar] [CrossRef]
| OB | COD (mg O2/mg) | BOD5 (mg O2/mg) | TOC (mg C/mg) | BOD5/COD Ratio |
|---|---|---|---|---|
| L-PC | 1.28 | 0.20 | 0.39 | 0.16 |
| BHA | 0.77 | 0.29 | 0.33 | 0.37 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salas, H.; Gutiérrez-Bouzán, C.; López-Grimau, V.; Vilaseca, M. Respirometric Study of Optical Brighteners in Textile Wastewater. Materials 2019, 12, 785. https://doi.org/10.3390/ma12050785
Salas H, Gutiérrez-Bouzán C, López-Grimau V, Vilaseca M. Respirometric Study of Optical Brighteners in Textile Wastewater. Materials. 2019; 12(5):785. https://doi.org/10.3390/ma12050785
Chicago/Turabian StyleSalas, Héctor, Carmen Gutiérrez-Bouzán, Víctor López-Grimau, and Mercedes Vilaseca. 2019. "Respirometric Study of Optical Brighteners in Textile Wastewater" Materials 12, no. 5: 785. https://doi.org/10.3390/ma12050785
APA StyleSalas, H., Gutiérrez-Bouzán, C., López-Grimau, V., & Vilaseca, M. (2019). Respirometric Study of Optical Brighteners in Textile Wastewater. Materials, 12(5), 785. https://doi.org/10.3390/ma12050785





