Glass-Ceramic Foams from ‘Weak Alkali Activation’ and Gel-Casting of Waste Glass/Fly Ash Mixtures
Abstract
1. Introduction
2. Materials and Methods
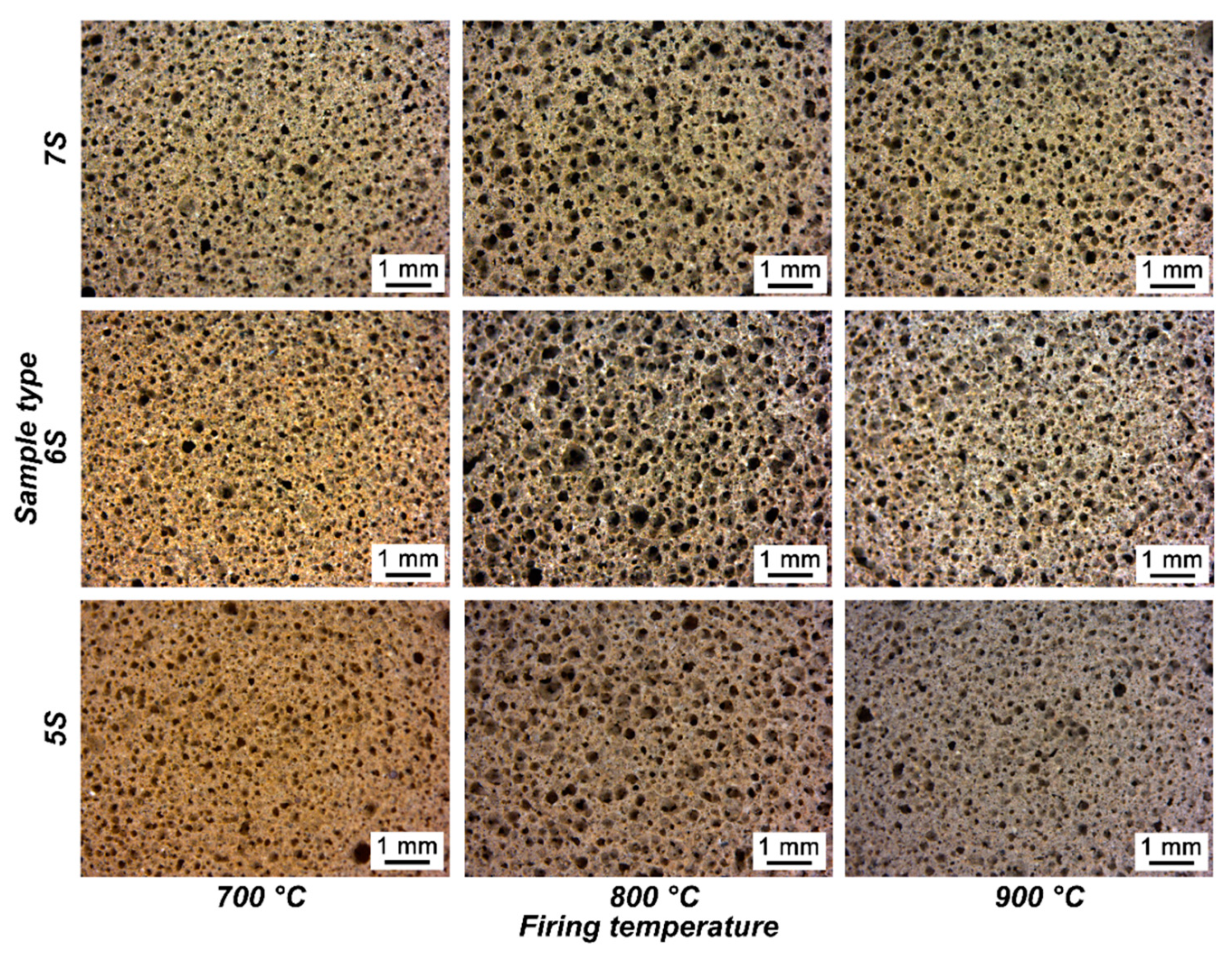
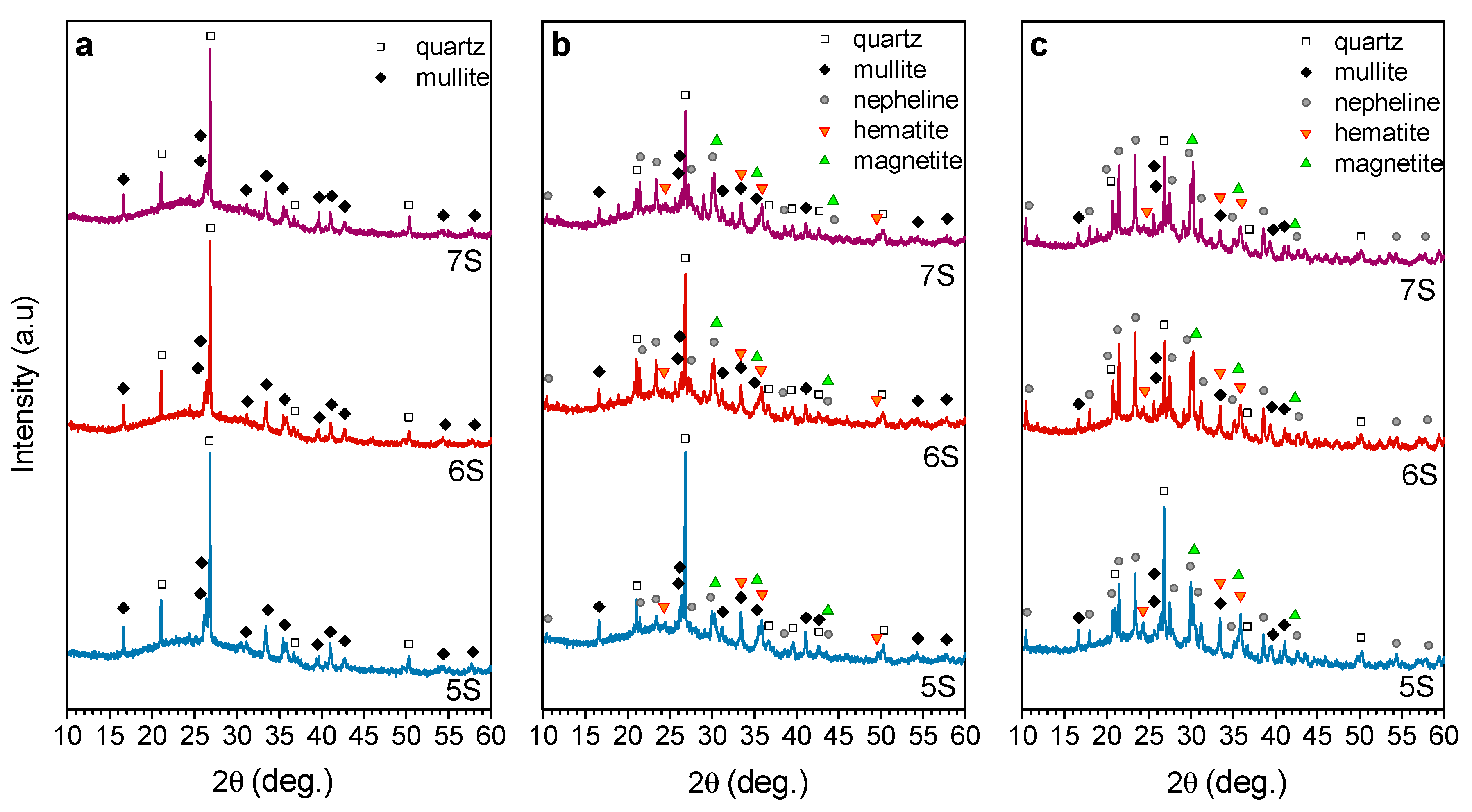
3. Results and Discussion
4. Conclusions
- The technique ensures an excellent approach to produce glass–ceramic foams allowing the incorporation of high proportions of fly ash.
- This approach provides a recycling route to glass fraction currently landfilled, providing a solution to the landfill derived problems as well as a significant economic advantage.
- The possibility to use low alkali activator concentrations to produce a geopolymer-like gel, which acts as a binding phase, is demonstrated.
- The decomposition of the gel and the SLG/FA interactions upon firing promote the formation of the glass–ceramic foams.
- The developed glass–ceramic foams have high porosity, low thermal conductivity, and reasonable mechanical properties to be applied as thermal insulation materials.
- The chemical stability of the glass ceramic foams was assessed by leaching tests; the release of heavy metals remained below the threshold specification for inert materials.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Blissett, R.S.; Rowson, N.A. A review of the multi-component utilisation of coal fly ash. Fuel 2012, 97, 1–23. [Google Scholar] [CrossRef]
- Carlson, C.L.; Adriano, D.C. Environmental impacts of coal combustion residues. J. Environ. Qual. 1993, 22, 227–247. [Google Scholar] [CrossRef]
- Fernandes, H.R.; Tulyaganov, D.U.; Ferreira, J.M.F. Preparation and characterization of foams from sheet glass and fly ash using carbonates as foaming agents. Ceram. Int. 2009, 35, 229–235. [Google Scholar] [CrossRef]
- Asokan, P.; Saxena, M.; Asolekar, S.R. Coal combustion residues–Environmental implications and recycling potentials. Resour. Conserv. Recycl. 2005, 43, 239–262. [Google Scholar] [CrossRef]
- Kulasuriya, C.; Vimonsatit, V.; Dias, W.; De Silva, P. Design and development of Alkali Pozzolan Cement (APC). Constr. Build. Mater. 2014, 68, 426–433. [Google Scholar] [CrossRef]
- Bui, P.T.; Ogawa, Y.; Nakarai, K.; Kawai, K. Effect of internal alkali activation on pozzolanic reaction of low-calcium fly ash cement paste. Mater. Struct. Mater. Constr. 2016, 49, 3039–3053. [Google Scholar] [CrossRef]
- Leroy, C.; Ferro, M.C.; Monteiro, R.C.C.; Fernandes, M.H.V. Production of glass-ceramics from coal ashes. J. Eur. Ceram. Soc. 2001, 21, 195–202. [Google Scholar] [CrossRef]
- Barbieri, L.; Lancellotti, I.; Manfredini, T.; Ignasi, Q.; Rincon, J.; Romero, M. Design, obtainment and properties of glasses and glass–ceramics from coal fly ash. Fuel 1999, 78, 271–276. [Google Scholar] [CrossRef]
- Wu, J.P.; Boccaccini, A.R.; Lee, P.D.; Kershaw, M.J.; Rawlings, R.D. Glass ceramic foams from coal ash and waste glass: Production and characterisation. Adv. Appl. Ceram. 2006, 105, 32–39. [Google Scholar] [CrossRef]
- Bai, J.; Yang, X.; Xu, S.; Jing, W.; Yang, J. Preparation of foam glass from waste glass and fly ash. Mater. Lett. 2014, 136, 52–54. [Google Scholar] [CrossRef]
- Mi, H.; Yang, J.; Su, Z.; Wang, T.; Li, Z.; Huo, W.; Qu, Y. Preparation of ultra-light ceramic foams from waste glass and fly ash. Adv. Appl. Ceram. 2017, 116, 400–408. [Google Scholar] [CrossRef]
- Wei, Y.-L.; Cheng, S.-H.; Ko, G.-W. Effect of waste glass addition on lightweight aggregates prepared from F-class coal fly ash. Constr. Build. Mater. 2016, 112, 773–782. [Google Scholar] [CrossRef]
- Feng, J.; Zhang, R.; Gong, L.; Li, Y.; Cao, W.; Cheng, X. Development of porous fly ash-based geopolymer with low thermal conductivity. Mater. Des. 2015, 65, 529–533. [Google Scholar] [CrossRef]
- Xu, H.; Van Deventer, J.S.J. The geopolymerisation of alumino-silicate minerals. Int. J. Miner. Process. 2000, 59, 247–266. [Google Scholar] [CrossRef]
- Palomo, A.; Grutzeck, M.W.; Blanco, M.T. Alkali-activated fly ashes: A cement for the future. Cem. Concr. Res. 1999, 29, 1323–1329. [Google Scholar] [CrossRef]
- Toniolo, N.; Boccaccini, A.R. Fly ash-based geopolymers containing added silicate waste. A review. Ceram. Int. 2017, 43, 14545–14551. [Google Scholar] [CrossRef]
- Swanepoel, J.C.; Strydom, C.A. Utilisation of fly ash in a geopolymeric material. Appl. Geochem. 2002, 17, 1143–1148. [Google Scholar] [CrossRef]
- Fernández-Jiménez, A.; Palomo, A.; Criado, M. Microstructure development of alkali-activated fly ash cement: A descriptive model. Cem. Concr. Res. 2005, 35, 1204–1209. [Google Scholar] [CrossRef]
- McLellan, B.C.; Williams, R.P.; Lay, J.; van Riessen, A.; Corder, G.D. Costs and carbon emissions for geopolymer pastes in comparison to ordinary portland cement. J. Clean. Prod. 2011, 19, 1080–1090. [Google Scholar] [CrossRef]
- Komljenović, M.; Baščarević, Z.; Bradić, V. Mechanical and microstructural properties of alkali-activated fly ash geopolymers. J. Hazard. Mater. 2010, 181, 35–42. [Google Scholar] [CrossRef]
- Somna, K.; Jaturapitakkul, C.; Kajitvichyanukul, P.; Chindaprasirt, P. NaOH-activated ground fly ash geopolymer cured at ambient temperature. Fuel 2011, 90, 2118–2124. [Google Scholar] [CrossRef]
- Puertas, F.; Torres-Carrasco, M. Use of glass waste as an activator in the preparation of alkali-activated slag. Mechanical strength and paste characterisation. Cem. Concr. Res. 2014, 57, 95–104. [Google Scholar] [CrossRef]
- Fernández-Jiménez, A.; Cristelo, N.; Miranda, T.; Palomo, Á. Sustainable alkali activated materials: Precursor and activator derived from industrial wastes. J. Clean. Prod. 2017, 162, 1200–1209. [Google Scholar] [CrossRef]
- Zhang, S.; Keulen, A.; Arbi, K.; Ye, G. Waste glass as partial mineral precursor in alkali-activated slag/fly ash system. Cem. Concr. Res. 2017, 102, 29–40. [Google Scholar] [CrossRef]
- Toniolo, N.; Rincón, A.; Roether, J.A.; Ercole, P.; Bernardo, E.; Boccaccini, A.R. Extensive reuse of soda-lime waste glass in fly ash-based geopolymers. Constr. Build. Mater. 2018, 188, 1077–1084. [Google Scholar] [CrossRef]
- Toniolo, N.; Rincón, A.; Avadhut, Y.S.; Hartmann, M.; Bernardo, E.; Boccaccini, A.R. Novel geopolymers incorporating red mud and waste glass cullet. Mater. Lett. 2018, 219, 152–154. [Google Scholar] [CrossRef]
- Standard Specification for Coal Fly Ash and Raw or Calcined Natural Pozzolan for Use in Concrete; ASTM Committee C-09 on Concrete and Concrete Aggregates: West Conshohocken, PA, USA, 2013.
- Ranjbar, N.; Mehrali, M.; Alengaram, U.J.; Metselaar, H.S.C.; Jumaat, M.Z. Compressive strength and microstructural analysis of fly ash/palm oil fuel ash based geopolymer mortar under elevated temperatures. Constr. Build. Mater. 2014, 65, 114–121. [Google Scholar] [CrossRef]
- Studart, A.R.; Gonzenbach, U.T.; Tervoort, E.; Gauckler, L.J. Processing Routes to Macroporous Ceramics: A Review. J. Am. Ceram. Soc. 2006, 89, 1771–1789. [Google Scholar] [CrossRef]
- Álvarez-Ayuso, E.; Querol, X.; Plana, F.; Alastuey, A.; Moreno, N.; Izquierdo, M.; Font, O.; Moreno, T.; Diez, S.; Vázquez, E.; et al. Environmental, physical and structural characterisation of geopolymer matrixes synthesised from coal (co-)combustion fly ashes. J. Hazard. Mater. 2008, 154, 175–183. [Google Scholar] [CrossRef]
- Torres-Carrasco, M.; Puertas, F. Waste glass in the geopolymer preparation. Mechanical and microstructural characterisation. J. Clean. Prod. 2015, 90, 397–408. [Google Scholar] [CrossRef]
- Rattanasak, U.; Chindaprasirt, P. Influence of NaOH solution on the synthesis of fly ash geopolymer. Miner. Eng. 2009, 22, 1073–1078. [Google Scholar] [CrossRef]
- Desbats-Le Chequer, C.; Frizon, F. Impact of sulfate and nitrate incorporation on potassium- and sodium-based geopolymers: Geopolymerization and materials properties. J. Mater. Sci. 2011, 46, 5657–5664. [Google Scholar] [CrossRef]
- Hu, N.; Bernsmeier, D.; Grathoff, G.H.; Warr, L.N. The influence of alkali activator type, curing temperature and gibbsite on the geopolymerization of an interstratified illite-smectite rich clay from Friedland. Appl. Clay Sci. 2017, 135, 386–393. [Google Scholar] [CrossRef]
- Fernández-Jiménez, A.; Palomo, A. Composition and microstructure of alkali activated fly ash binder: Effect of the activator. Cem. Concr. Res. 2005, 35, 1984–1992. [Google Scholar] [CrossRef]
- Katz, A. Microscopic Study of Alkali-Activated Fly Ash. Cem. Concr. Res. 1998, 28, 197–208. [Google Scholar] [CrossRef]
- Li, J.; Zhuang, X.; Monfort, E.; Querol, X.; Llaudis, A.S.; Font, O.; Moreno, N.; Ten, F.J.G.; Izquierdo, M. Utilization of coal fly ash from a Chinese power plant for manufacturing highly insulating foam glass: Implications of physical, mechanical properties and environmental features. Constr. Build. Mater. 2018, 175, 64–76. [Google Scholar] [CrossRef]
- ul Haq, E.; Kunjalukkal Padmanabhan, S.; Licciulli, A. Synthesis and characteristics of fly ash and bottom ash based geopolymers—A comparative study. Ceram. Int. 2014, 40, 2965–2971. [Google Scholar] [CrossRef]
- Bakharev, T. Thermal behaviour of geopolymers prepared using class F fly ash and elevated temperature curing. Cem. Concr. Res. 2006, 36, 1134–1147. [Google Scholar] [CrossRef]
- Dombrowski, K.; Buchwald, A.; Weil, M. The influence of calcium content on the structure and thermal performance of fly ash based geopolymers. J. Mater. Sci. 2007, 42, 3033–3043. [Google Scholar] [CrossRef]
- Kuenzel, C.; Grover, L.M.; Vandeperre, L.; Boccaccini, A.R.; Cheeseman, C.R. Production of nepheline/quartz ceramics from geopolymer mortars. J. Eur. Ceram. Soc. 2013, 33, 251–258. [Google Scholar] [CrossRef]
- Gibson, L.J.; Ashby, M.F. Cellular Solids: Structure and Properties, 2nd ed.; Cambridge University Press: Cambridge, UK, 2014; pp. 1–510. [Google Scholar]
- Bernardo, E.; Scarinci, G.; Maddalena, A.; Hreglich, S. Development and mechanical properties of metal–particulate glass matrix composites from recycled glasses. Compos. Part A Appl. Sci. Manuf. 2004, 35, 17–22. [Google Scholar] [CrossRef]
- Petersen, R.R.; König, J.; Yue, Y. The mechanism of foaming and thermal conductivity of glasses foamed with MnO2. J. Non-Cryst. Solids 2015, 425, 74–82. [Google Scholar] [CrossRef]
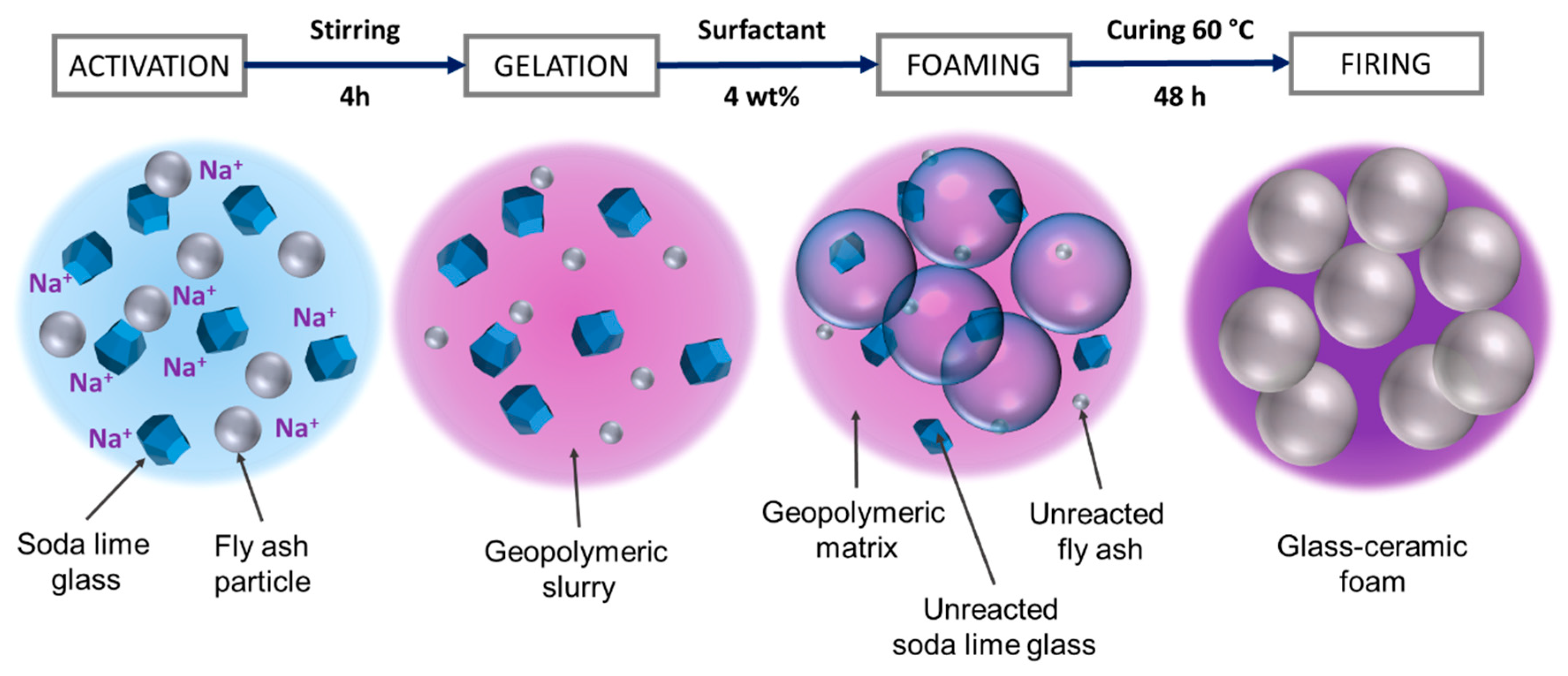

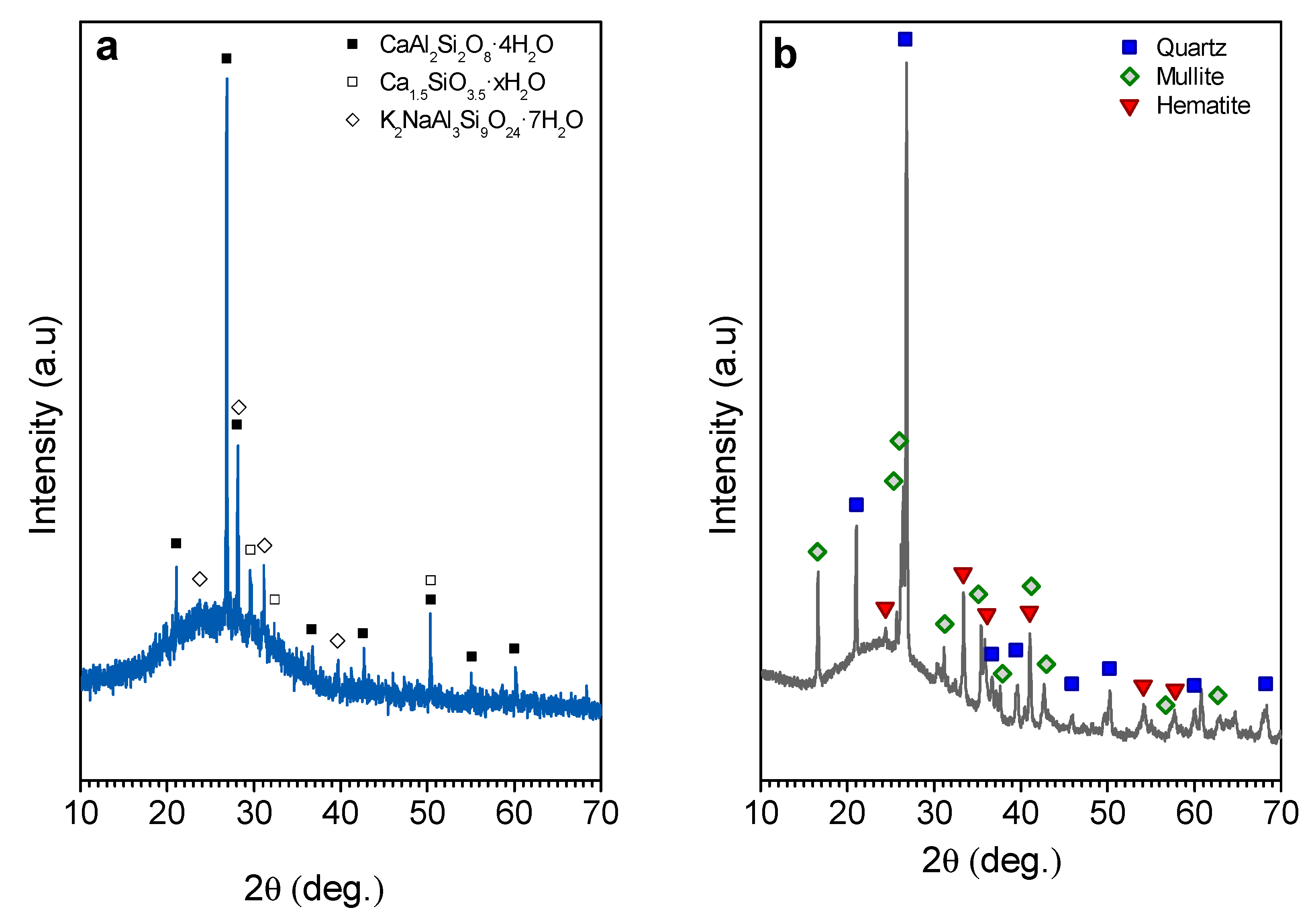
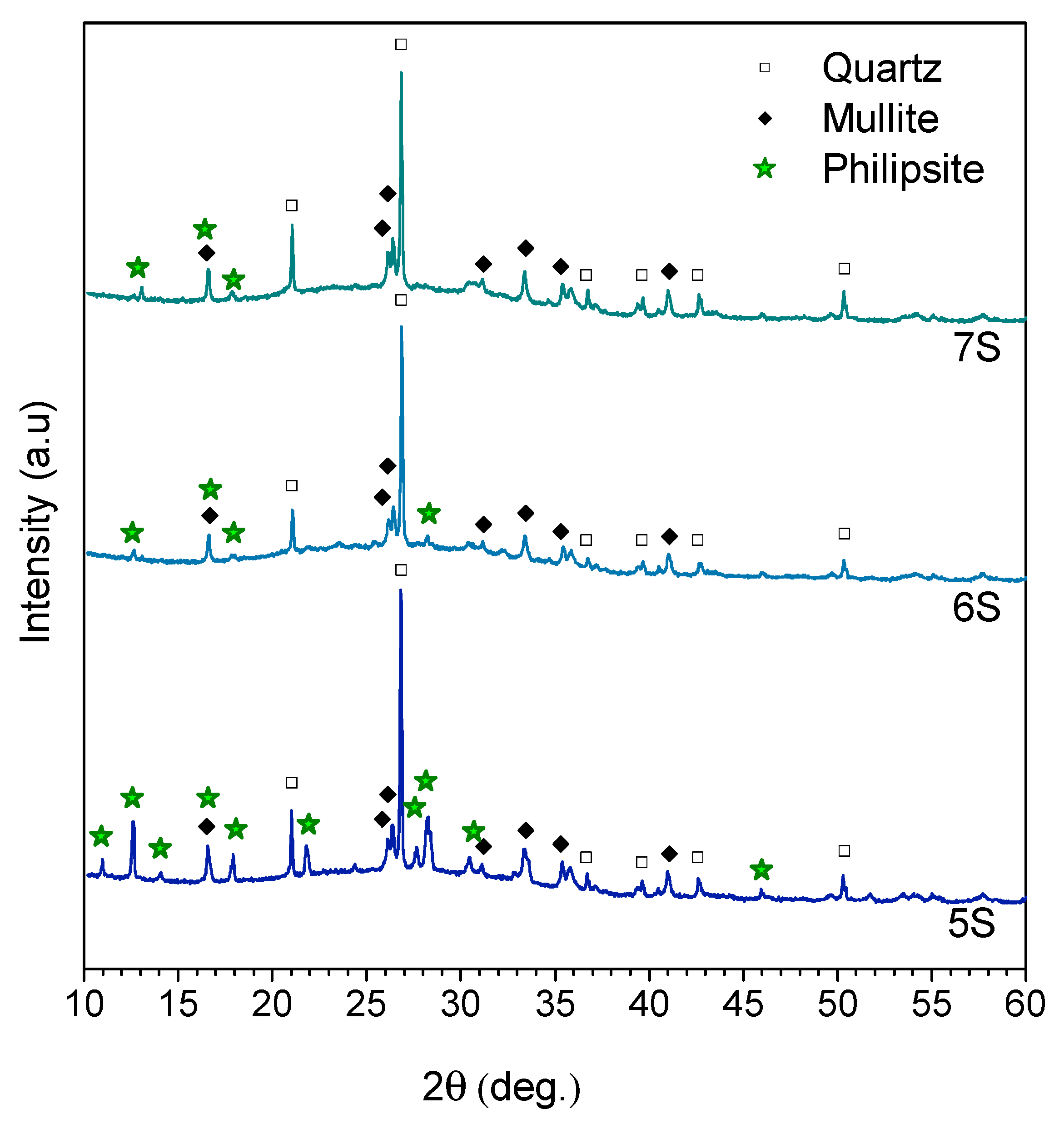

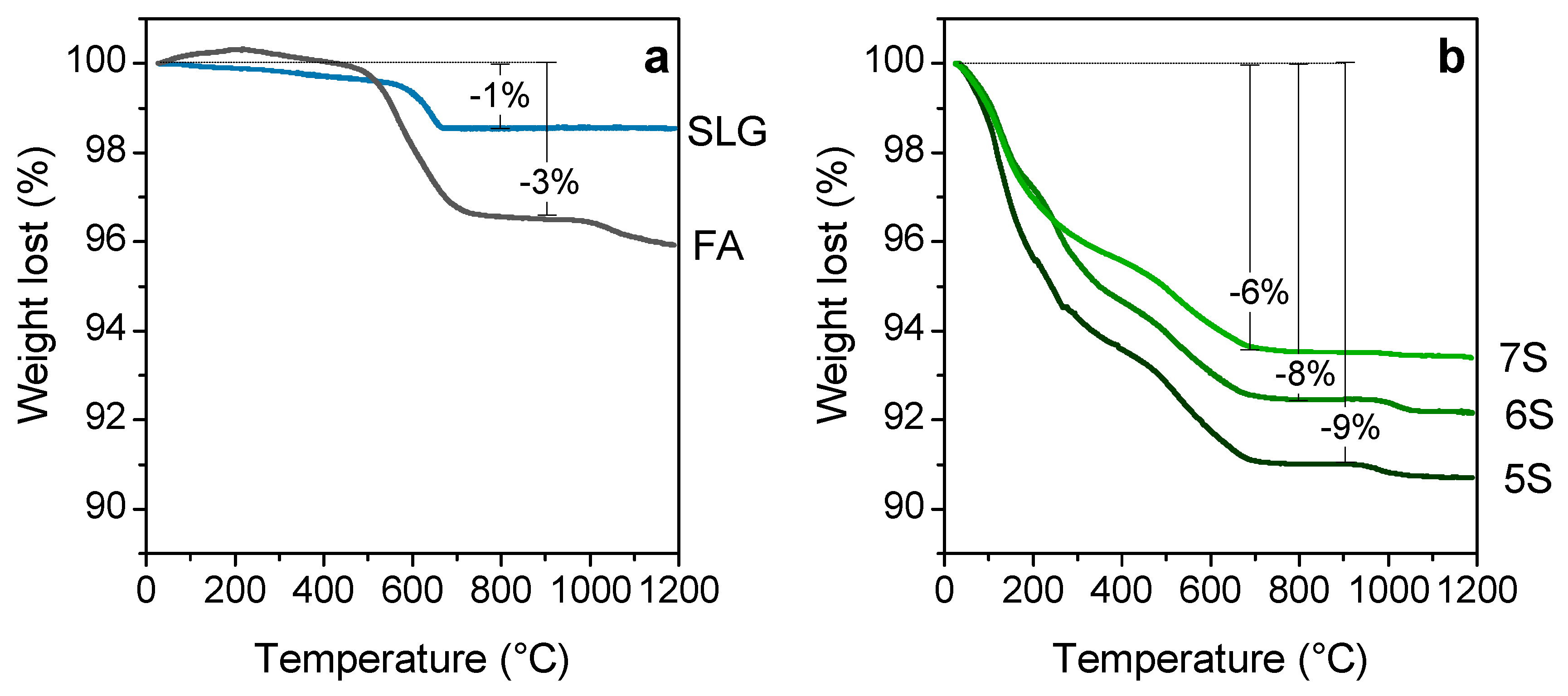



| Oxide (wt%) | SiO2 | Al2O3 | Na2O | K2O | CaO | MgO | Fe2O3 | TiO2 |
|---|---|---|---|---|---|---|---|---|
| FA | 54.36 | 24.84 | 0.83 | 3.03 | 2.56 | 2.06 | 8.28 | 1.07 |
| SLG | 70.5 | 3.2 | 12 | 1 | 10 | 2.3 | 0.42 | 0.07 |
| Sample | Firing Tª (°C) | Density, ρ (g/cm3) | Porosity (%) | σcomp (MPa) | ||||
|---|---|---|---|---|---|---|---|---|
| Geometric | Apparent | True | Total | Open | Closed | |||
| 5 | 700 | 0.71 ± 0.01 | 2.28 ± 0.03 | 2.46 ± 0.01 | 71 ± 1 | 68 ± 2 | 2 ± 1 | 1.8 ± 0.2 |
| 800 | 0.82 ± 0.01 | 2.32 ± 0.01 | 2.49 ± 0.01 | 67 ± 1 | 64 ± 1 | 2 ± 1 | 5.1 ± 0.3 | |
| 900 | 0.87 ± 0.01 | 2.36 ± 0.01 | 2.51 ± 0.01 | 65 ± 1 | 62 ± 1 | 2 ± 1 | 7.4 ± 0.3 | |
| 6 | 700 | 0.54 ± 0.08 | 2.25 ± 0.02 | 2.42 ± 0.01 | 78 ± 2 | 75 ± 3 | 2 ± 1 | 1.9 ± 0.2 |
| 800 | 0.66 ± 0.01 | 2.26 ± 0.02 | 2.43 ± 0.01 | 72 ± 1 | 70 ± 2 | 2 ± 1 | 4.5 ± 0.2 | |
| 900 | 0.71 ± 0.01 | 2.26 ± 0.01 | 2.45 ± 0.01 | 70 ± 3 | 68 ± 3 | 2 ± 1 | 5.4 ± 0.4 | |
| 7 | 700 | 0.71 ± 0.01 | 2.31 ± 0.06 | 2.46 ± 0.01 | 71 ± 1 | 69 ± 2 | 2 ± 1 | 3.8 ± 0.2 |
| 800 | 0.83 ± 0.01 | 2.13 ± 0.05 | 2.49 ± 0.01 | 66 ± 2 | 61 ± 2 | 6 ± 1 | 5.3 ± 0.4 | |
| 900 | 1.02 ± 0.3 | 2.08 ± 0.02 | 2.44 ± 0.01 | 58 ± 3 | 50 ± 4 | 7 ± 2 | 8.7 ± 0.6 | |
| Element (ppm) | 5S3M | 6S3M | 7S3M | Initial Materials | Limits [UE] (ppm) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| 800 | 900 | 800 | 900 | 800 | 900 | FA | SLG | Inert material | Non-hazardous material | |
| As | 0.0316 | 0.0635 | 0.068 | 0.0503 | 0.0491 | 0.0795 | <0.0049 | <0.0049 | 0.5 | 2 |
| Ba | >al | >al | >al | >al | 0.0672 | 0.1108 | <0.000 | >al | 20 | 100 |
| Cd | <0.0002 | <0.0002 | <0.0002 | <0.0002 | <0.0002 | <0.0002 | <0.0002 | 0.001 | 0.04 | 1 |
| Cr | 0.3406 | 0.0805 | 0.0255 | 0.0598 | 0.0689 | 0.3001 | 0.4672 | 0.0043 | 0.5 | 10 |
| Cu | 0.0029 | 0.0183 | 0.0024 | 0.0065 | 0.0053 | 0.0207 | 0.0282 | 0.0036 | 2 | 50 |
| Hg | 0.0032 | <0.0004 | 0.0006 | <0.0004 | 0.0020 | 0.0017 | 0.8983 | <0.0004 | 0.01 | 0.2 |
| Mo | 0.5324 | 0.0472 | 0.2107 | 0.0087 | 0.1973 | 0.2435 | <0.0004 | 0.007 | 0.5 | 10 |
| Ni | <0.0014 | <0.0014 | <0.0014 | <0.0014 | <0.0014 | <0.0014 | <0.0014 | <0.0014 | 0.4 | 10 |
| Pb | <0.0047 | <0.0047 | <0.0047 | <0.0047 | <0.0047 | <0.0047 | <0.0047 | 0.018 | 0.5 | 10 |
| Se | 0.0133 | <0.0122 | 0.0255 | <0.0122 | 0.0226 | <0.0122 | <0.0122 | 0.018 | 0.1 | 0.5 |
| Zn | <0.0203 | <0.0203 | <0.0203 | <0.0203 | <0.0203 | <0.0203 | <0.0203 | 0.088 | 4 | 50 |
| pH | 9.4 | 8.6 | 8.2 | 7.7 | 8.0 | 7.6 | ||||
| Abbreviation: >al, above detection limit | ||||||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rincón Romero, A.; Toniolo, N.; Boccaccini, A.R.; Bernardo, E. Glass-Ceramic Foams from ‘Weak Alkali Activation’ and Gel-Casting of Waste Glass/Fly Ash Mixtures. Materials 2019, 12, 588. https://doi.org/10.3390/ma12040588
Rincón Romero A, Toniolo N, Boccaccini AR, Bernardo E. Glass-Ceramic Foams from ‘Weak Alkali Activation’ and Gel-Casting of Waste Glass/Fly Ash Mixtures. Materials. 2019; 12(4):588. https://doi.org/10.3390/ma12040588
Chicago/Turabian StyleRincón Romero, Acacio, Nicoletta Toniolo, Aldo R. Boccaccini, and Enrico Bernardo. 2019. "Glass-Ceramic Foams from ‘Weak Alkali Activation’ and Gel-Casting of Waste Glass/Fly Ash Mixtures" Materials 12, no. 4: 588. https://doi.org/10.3390/ma12040588
APA StyleRincón Romero, A., Toniolo, N., Boccaccini, A. R., & Bernardo, E. (2019). Glass-Ceramic Foams from ‘Weak Alkali Activation’ and Gel-Casting of Waste Glass/Fly Ash Mixtures. Materials, 12(4), 588. https://doi.org/10.3390/ma12040588







