Bio- and Fossil-Based Polymeric Blends and Nanocomposites for Packaging: Structure–Property Relationship
Abstract
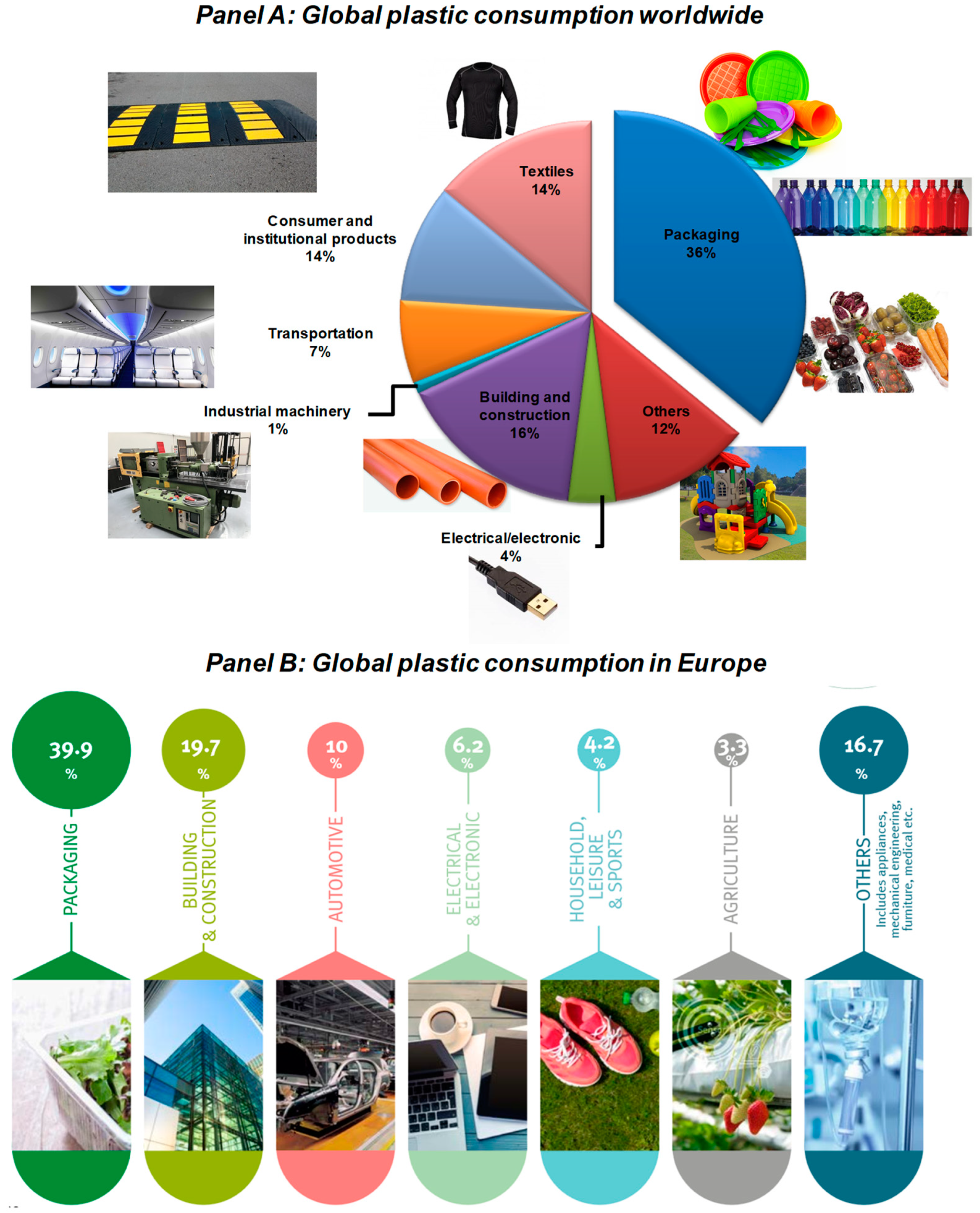
1. Introduction
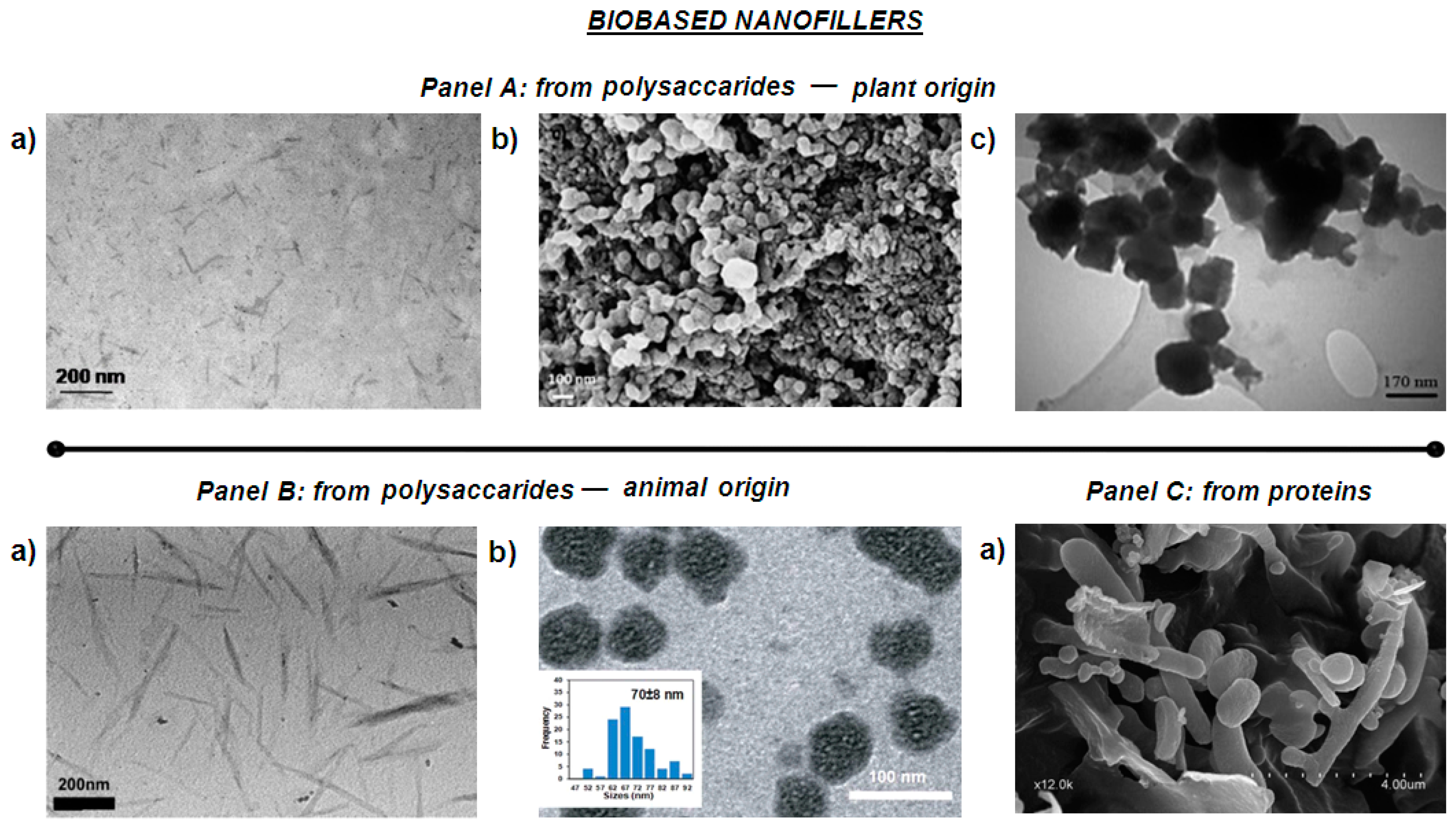
2. Bio-Based Nanofillers in the Packaging Sector
2.1. Nanofillers from Polysaccharides—Plant Origin
2.1.1. Cellulose Nanofibers/Nanocrystals
2.1.2. Lignin Nanoparticles
2.1.3. Starch Nanoparticles
2.2. Nanofillers from Polysaccharides—Animal Origin
2.2.1. Chitin Nanoparticles/Nanofibers
2.2.2. Chitosan Nanoparticles
2.3. Nanofillers from Proteins
2.3.1. Nanokeratin
2.3.2. Nanogelatin
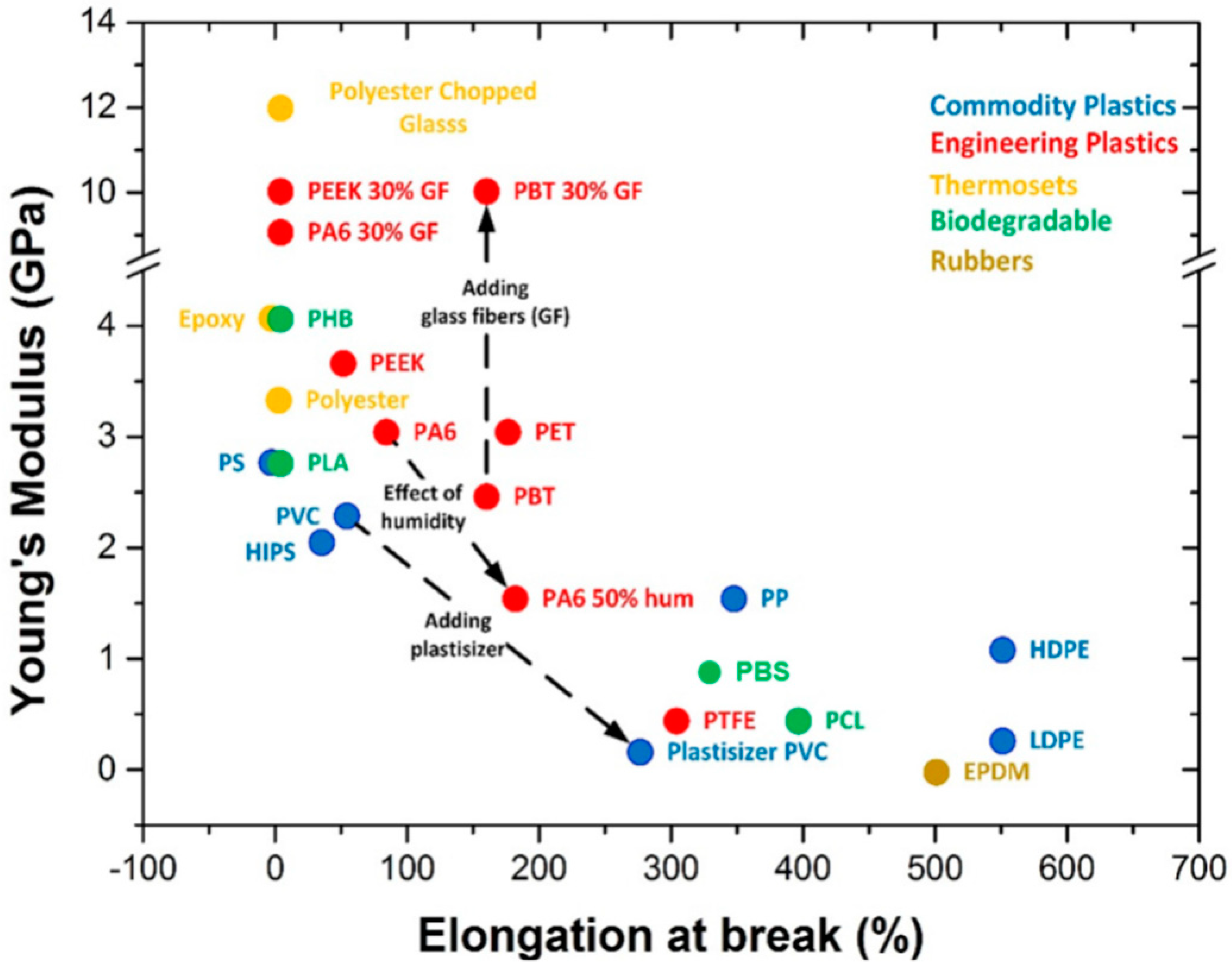
3. Conventional Matrices for Packaging
4. Bio-Based Matrices for Packaging Applications
4.1. Starch
4.2. Cellulose
4.3. Chitin/Chitosan
4.4. Biopolyesters from Microorganisms—Polyhydroxyalcanoates (PHAs)
4.5. Biopolyesters from Biotechnology and Conventional Synthesis from Synthetic Monomers
4.5.1. PLA
4.5.2. PBS
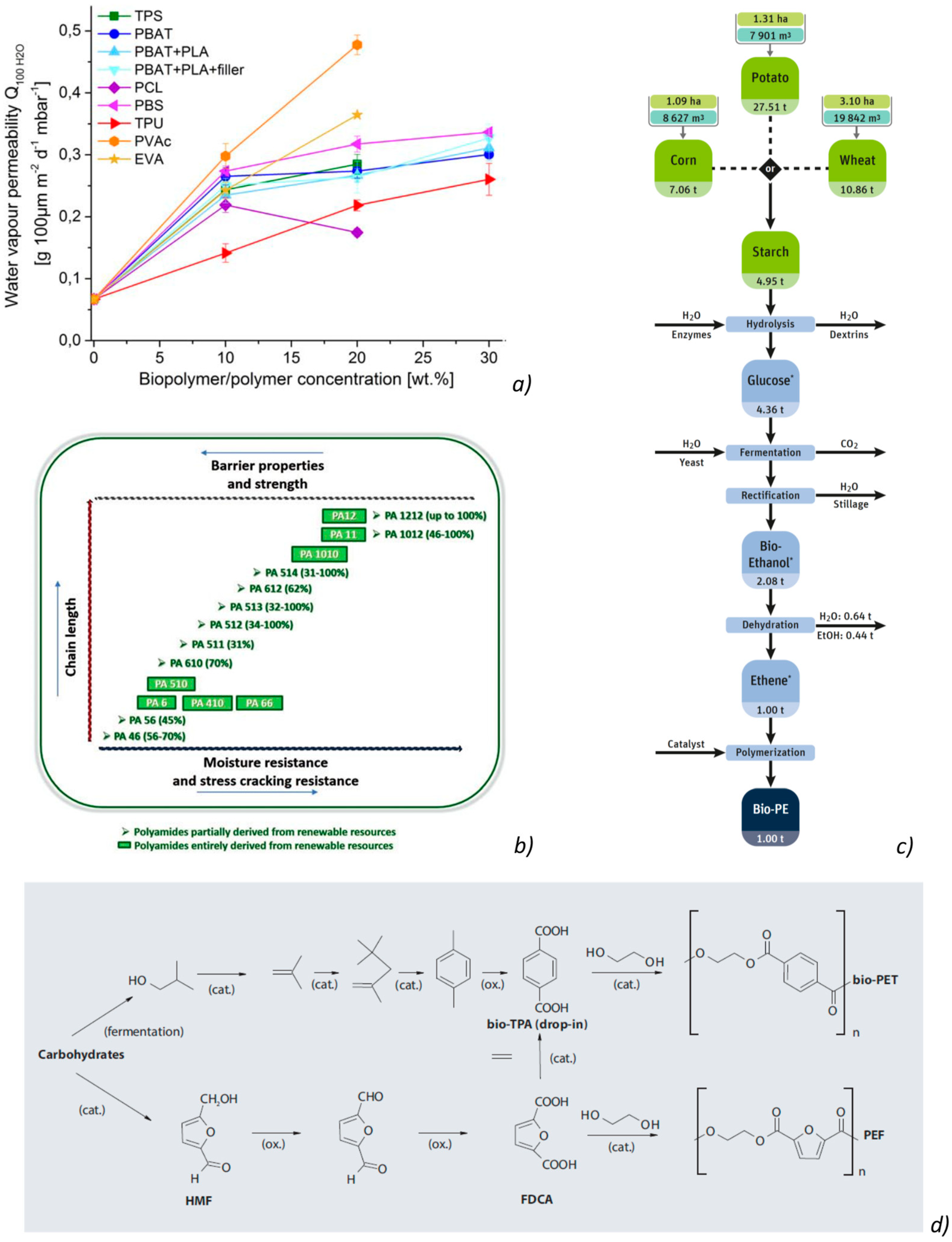
4.5.3. PBAT
4.6. Bio-Polyamides
4.7. Bio-Polyolefins
4.8. Bio-Poly (Ethylene Terephtalic Acid) (Bio-PET or PEF)
5. Hybrid Blends Based on Bio-Based and Fossil Fuel-Derived Polymers
5.1. Starch
5.2. Chitosan
5.3. Cellulose
5.4. PHB
5.5. PLA
5.6. PBS
5.7. PBAT
5.8. Bio-PE-Based Blends
6. Nanocomposites of Hybrid Blends and Bio-Based Nanofillers
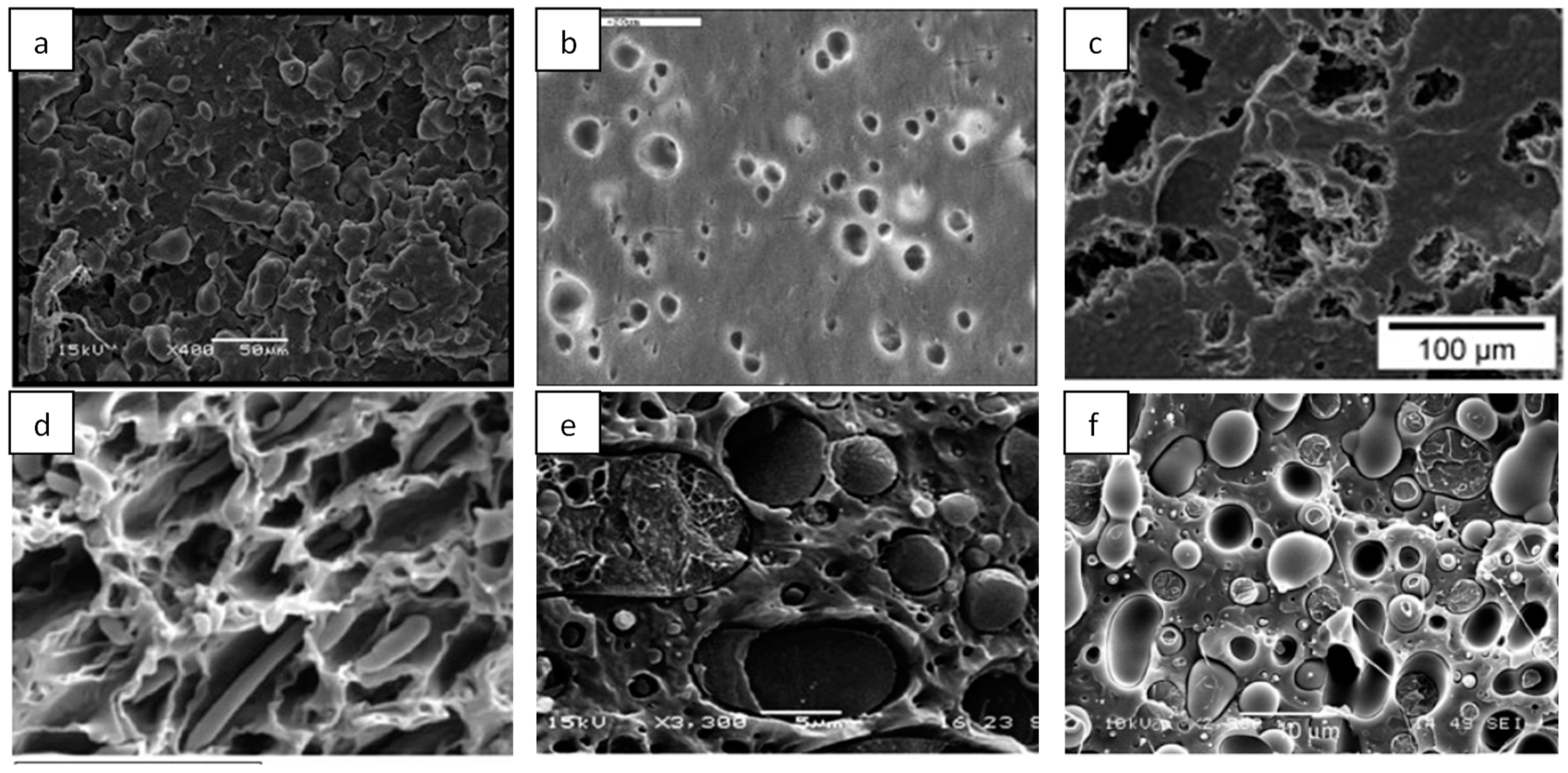
6.1. Starch Based Hybrid Nanocomposites
6.2. Cellulose-Based Hybrid Nanocomposites
6.3. Chitosan-Based Hybrid Nanocomposites
6.4. Polyester-Based Hybrid Nanocomposites
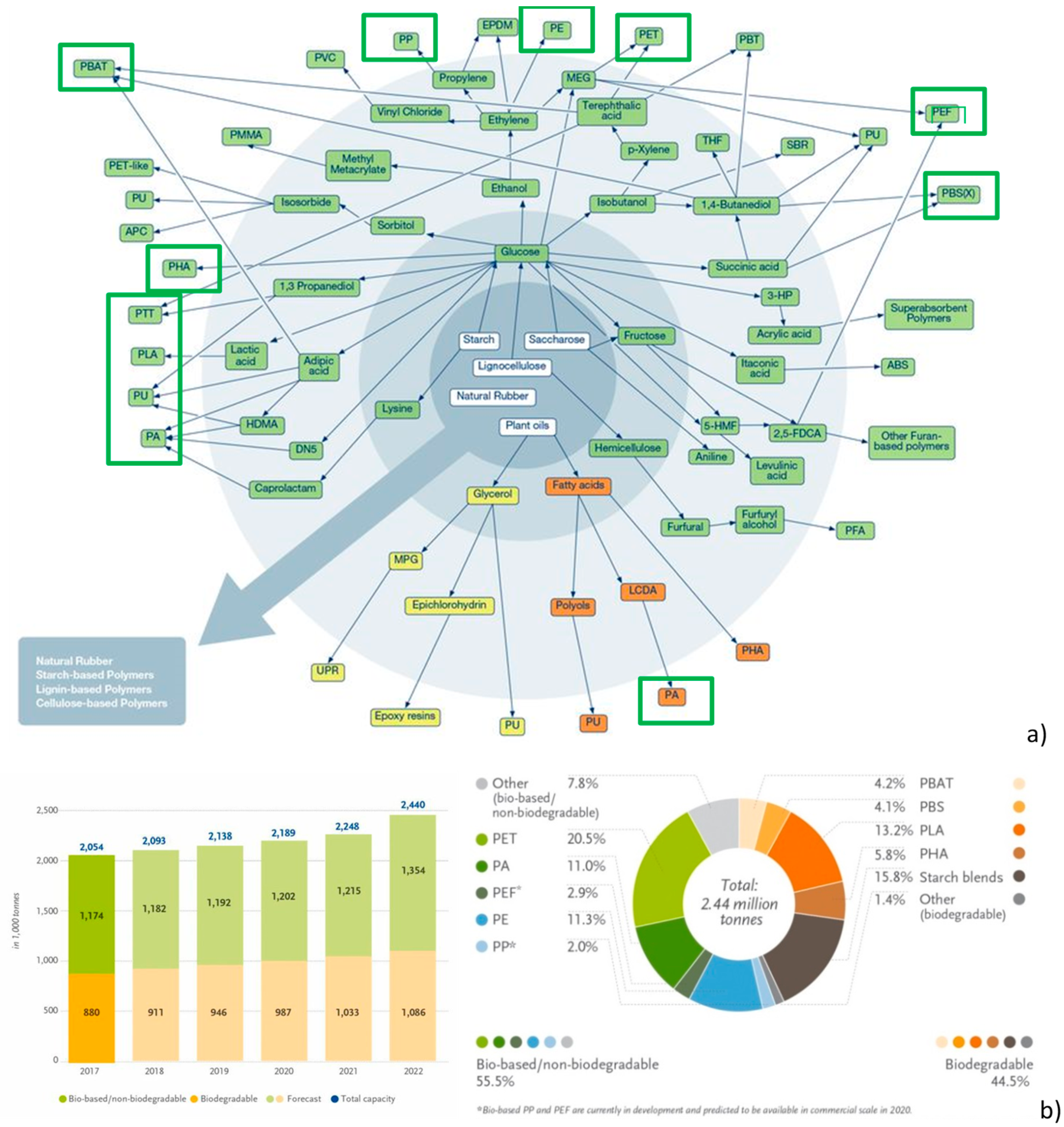
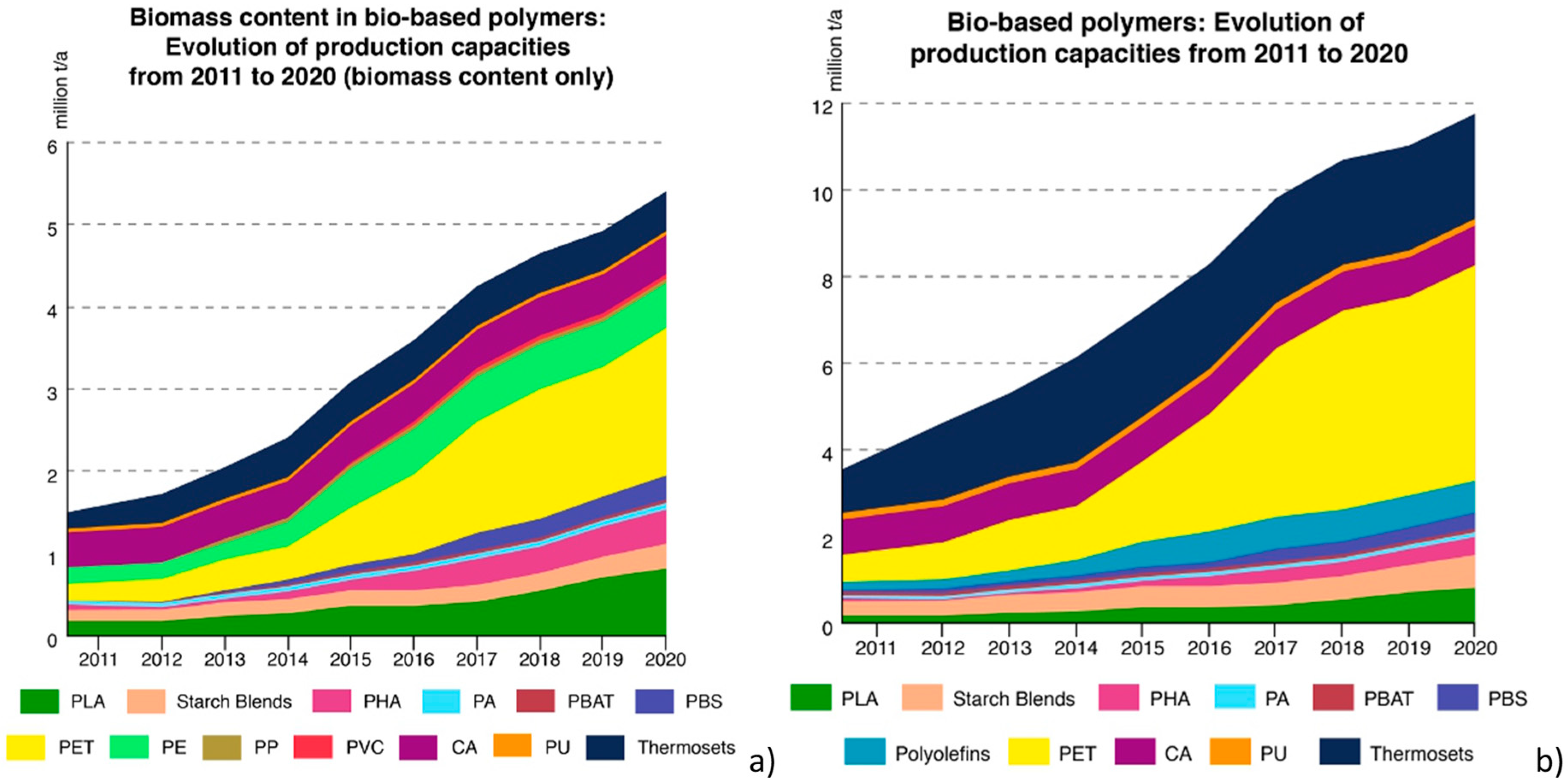
7. Production, Market, and Future Perspective of Hybrid Bio-Based Polymers
8. Conclusions
Author Contributions
Conflicts of Interest
References
- Geyer, R.; Jambeck, J.R.; Law, K.L. Production, use, and fate of all plastics ever made. Sci. Adv. 2017, 3, e1700782. [Google Scholar] [CrossRef] [PubMed]
- Ellen MacArthur Foundation and McKinsey and Company. The New Plastics Economy-Rethinking the Future of Plastics; Ellen MacArthur Foundation: Cowes, UK, 2016. [Google Scholar]
- Pongrácz, E. The environmental impacts of packaging. Environ. Conscious Mater. Chem. Process. 2007, 2, 237. [Google Scholar]
- Europe, F.D. Environmental Sustainability Vision towards 2030; Achievements, Challenges and Opportunities: Brussels, Belgium, 2012. [Google Scholar]
- Hestin, M.; Faninger, T.; Milios, L. Increased EU Plastics Recycling Targets: Environmental, Economic and Social Impact Assessment; Deloitte: New York, NY, USA, 2015. [Google Scholar]
- Robertson, G. State-of-the-art biobased food packaging materials. In Environmentally Compatible Food Packaging; Woodhead Publishing: Sawston, CA, USA, 2008. [Google Scholar]
- Babu, R.; O’Connor, K.; Seeram, R. Current progress on bio-based polymers and their future trends. Prog. Biomater. 2013, 2, 8. [Google Scholar] [CrossRef] [PubMed]
- Narancic, T.; Verstichel, S.; Reddy Chaganti, S.; Morales-Gamez, L.; Kenny, S.T.; De Wilde, B.; Babu Padamati, R.; O’Connor, K.E. Biodegradable plastic blends create new possibilities for end-of-life management of plastics but they are not a panacea for plastic pollution. Environ. Sci. Technol. 2018, 52, 10441–10452. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Yu, J.; Yu, J. Influence of maleic anhydride on the compatibility of thermal plasticized starch and linear low-density polyethylene. J. Appl. Polym. Sci. 2004, 93, 686–695. [Google Scholar] [CrossRef]
- Bikiaris, D.; Prinos, J.; Koutsopoulos, K.; Vouroutzis, N.; Pavlidou, E.; Frangis, N.; Panayiotou, C. LDPE/plasticized starch blends containing PE-g-MA copolymer as compatibilizer. Polym. Degrad. Stab. 1998, 59, 287–291. [Google Scholar] [CrossRef]
- Biresaw, G.; Carriere, C.J. Compatibility and mechanical properties of blends of polystyrene with biodegradable polyesters. Compos. Part A Appl. Sci. Manuf. 2004, 35, 313–320. [Google Scholar] [CrossRef]
- Swift, G. Degradability of commodity plastics and specialty polymers. An overview. Degrad. Commod. Plast. Spe. Polym. 1990, 433, 2–12. [Google Scholar]
- Otey, F.H.; Westhoff, R.P.; Russell, C.R. Biodegradable films from starch and ethylene-acrylic acid copolymer. Ind. Eng. Chem. Prod. Res. Dev. 1977, 16, 305–308. [Google Scholar] [CrossRef]
- Swanson, C.L.; Shogren, R.L.; Fanta, G.F.; Imam, S.H. Starch-plastic materials—Preparation, physical properties, and biodegradability (a review of recent USDA research). J. Environ. Polym. Degrad. 1993, 1, 155–166. [Google Scholar] [CrossRef]
- Schneiderman, D.K.; Hillmyer, M.A. 50th anniversary perspective: There is a great future in sustainable polymers. Macromolecules 2017, 50, 3733–3749. [Google Scholar] [CrossRef]
- Dartee, M. It’s time to get to know your way around bioplastics. Plast. Technol. 2010, 56, 18–22. [Google Scholar]
- Sam, S.T.; Nuradibah, M.A.; Ismail, H.; Noriman, N.Z.; Ragunathan, S. Recent advances in polyolefins/natural polymer blends used for packaging application. Polym. Plast. Technol. Eng. 2014, 53, 631–644. [Google Scholar] [CrossRef]
- Rallini, M.; Kenny, J.M. Nanofillers in Polymers. In Modification of Polymer Properties; Elsevier: New York, NY, USA, 2017; pp. 47–86. [Google Scholar]
- Luzi, F.; Fortunati, E.; Di Michele, A.; Pannucci, E.; Botticella, E.; Santi, L.; Kenny, J.M.; Torre, L.; Bernini, R. Nanostructured starch combined with hydroxytyrosol in poly(vinyl alcohol) based ternary films as active packaging system. Carbohydr. Polym. 2018, 193, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Seoane, I.T.; Luzi, F.; Puglia, D.; Cyras, V.P.; Manfredi, L.B. Enhancement of paperboard performance as packaging material by layering with plasticized polyhydroxybutyrate/nanocellulose coatings. J. Appl. Polym. Sci. 2018, 135, 46872. [Google Scholar] [CrossRef]
- Le Corre, D.; Bras, J.; Dufresne, A. Starch nanoparticles: A review. Biomacromolecules 2010, 11, 1139–1153. [Google Scholar] [CrossRef] [PubMed]
- Peponi, L.; Puglia, D.; Torre, L.; Valentini, L.; Kenny, J.M. Processing of nanostructured polymers and advanced polymeric based nanocomposites. Mater. Sci. Eng. 2014, 85, 1–46. [Google Scholar] [CrossRef]
- Khosravi-Darani, K.; Bucci, D.Z. Application of Poly (hydroxyalkanoate) In Food Packaging: Improvements by Nanotechnology. Chem. Biochem. Eng. Q. 2015, 29, 275–285. [Google Scholar] [CrossRef]
- Luzi, F.; Fortunati, E.; Jiménez, A.; Puglia, D.; Pezzolla, D.; Gigliotti, G.; Kenny, J.M.; Chiralt, A.; Torre, L. Production and characterization of PLA_PBS biodegradable blends reinforced with cellulose nanocrystals extracted from hemp fibres. Ind. Crops Prod. 2016, 93, 276–289. [Google Scholar] [CrossRef]
- Yang, W.; Fortunati, E.; Luzi, F.; Kenny, J.M.; Torre, L.; Puglia, D. Lignocellulosic Based Bionanocomposites for Different Industrial Applications. Curr. Org. Chem. 2018, 22, 1205–1221. [Google Scholar] [CrossRef]
- Mohanty, A.K.; Misra, M.; Drzal, L.T. Sustainable bio-composites from renewable resources: Opportunities and challenges in the green materials world. J. Polym. Environ. 2002, 10, 19–26. [Google Scholar] [CrossRef]
- Luzi, F.; Fortunati, E.; Puglia, D.; Lavorgna, M.; Santulli, C.; Kenny, J.M.; Torre, L. Optimized extraction of cellulose nanocrystals from pristine and carded hemp fibres. Ind. Crops Prod. 2014, 56, 175–186. [Google Scholar] [CrossRef]
- Alila, S.; Besbes, I.; Vilar, M.R.; Mutjé, P.; Boufi, S. Non-woody plants as raw materials for production of microfibrillated cellulose (MFC): A comparative study. Ind. Crops Prod. 2013, 41, 250–259. [Google Scholar] [CrossRef]
- Lavoine, N.; Desloges, I.; Dufresne, A.; Bras, J. Microfibrillated cellulose–Its barrier properties and applications in cellulosic materials: A review. Carbohydr. Polym. 2012, 90, 735–764. [Google Scholar] [CrossRef] [PubMed]
- Brinchi, L.; Cotana, F.; Fortunati, E.; Kenny, J.M. Production of nanocrystalline cellulose from lignocellulosic biomass: Technology and applications. Carbohydr. Polym. 2013, 94, 154–169. [Google Scholar] [CrossRef] [PubMed]
- Habibi, Y.; Lucia, L.A.; Rojas, O.J. Cellulose nanocrystals: Chemistry, self-assembly, and applications. Chem. Rev. 2010, 110, 3479–3500. [Google Scholar] [CrossRef] [PubMed]
- Dufresne, A. Nanocellulose: A new ageless bionanomaterial. Mater. Today 2013, 16, 220–227. [Google Scholar] [CrossRef]
- Rescignano, N.; Fortunati, E.; Montesano, S.; Emiliani, C.; Kenny, J.M.; Martino, S.; Armentano, I. PVA bio-nanocomposites: A new take-off using cellulose nanocrystals and PLGA nanoparticles. Carbohydr. Polym. 2014, 99, 47–58. [Google Scholar] [CrossRef]
- He, X.; Luzi, F.; Yang, W.; Xiao, Z.; Torre, L.; Xie, Y.; Puglia, D. Citric acid as green modifier for tuned hydrophilicity of surface modified cellulose and lignin nanoparticles. ACS Sustain. Chem. Eng. 2018, 6, 9966–9978. [Google Scholar] [CrossRef]
- Nasseri, R.; Mohammadi, N. Starch-based nanocomposites: A comparative performance study of cellulose whiskers and starch nanoparticles. Carbohydr. Polym. 2014, 106, 432–439. [Google Scholar] [CrossRef]
- Zhu, L.; Liang, K.; Ji, Y. Prominent reinforcing effect of chitin nanocrystals on electrospun polydioxanone nanocomposite fiber mats. J. Mech. Behave. Biomed. Mater. 2015, 44, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Kongkaoroptham, P.; Piroonpan, T.; Hemvichian, K.; Suwanmala, P.; Rattanasakulthong, W.; Pasanphan, W. Poly (ethylene glycol) methyl ether methacrylate-graft-chitosan nanoparticles as a biobased nanofiller for a poly (lactic acid) blend: Radiation-induced grafting and performance studies. J. Appl. Polym. Sci. 2015, 132. [Google Scholar] [CrossRef]
- Fabra, M.J.; Pardo, P.; Martínez-Sanz, M.; Lopez-Rubio, A.; Lagarón, J.M. Combining polyhydroxyalkanoates with nanokeratin to develop novel biopackaging structures. J. Appl. Polym. Sci. 2016, 133. [Google Scholar] [CrossRef]
- Fortunati, E.; Luzi, F.; Puglia, D.; Dominici, F.; Santulli, C.; Kenny, J.M.; Torre, L. Investigation of thermo-mechanical, chemical and degradative properties of PLA-limonene films reinforced with cellulose nanocrystals extracted from Phormium tenax leaves. Eur. Polym. J. 2014, 56, 77–91. [Google Scholar] [CrossRef]
- Fortunati, E.; Luzi, F.; Puglia, D.; Petrucci, R.; Kenny, J.M.; Torre, L. Processing of PLA nanocomposites with cellulose nanocrystals extracted from Posidonia oceanica waste: Innovative reuse of coastal plant. Ind. Crops Prod. 2015, 67, 439–447. [Google Scholar] [CrossRef]
- Fortunati, E.; Armentano, I.; Zhou, Q.; Iannoni, A.; Saino, E.; Visai, L.; Berglund, L.A.; Kenny, J.M. Multifunctional bionanocomposite films of poly(lactic acid), cellulose nanocrystals and silver nanoparticles. Carbohydr. Polym. 2012, 87, 1596–1605. [Google Scholar] [CrossRef]
- Fortunati, E.; Puglia, D.; Luzi, F.; Santulli, C.; Kenny, J.M.; Torre, L. Binary PVA bio-nanocomposites containing cellulose nanocrystals extracted from different natural sources: Part I. Carbohydr. Polym. 2013, 97, 825–836. [Google Scholar] [CrossRef]
- Fortunati, E.; Luzi, F.; Puglia, D.; Terenzi, A.; Vercellino, M.; Visai, L.; Santulli, C.; Torre, L.; Kenny, J.M. Ternary PVA nanocomposites containing cellulose nanocrystals from different sources and silver particles: Part II. Carbohydr. Polym. 2013, 97, 837–848. [Google Scholar] [CrossRef] [PubMed]
- Fortunati, E.; Benincasa, P.; Balestra, G.M.; Luzi, F.; Mazzaglia, A.; Del Buono, D.; Puglia, D.; Torre, L. Revalorization of barley straw and husk as precursors for cellulose nanocrystals extraction and their effect on PVA_CH nanocomposites. Ind. Crops Prod. 2016, 92, 201–217. [Google Scholar] [CrossRef]
- Cano, A.; Fortunati, E.; Cháfer, M.; González-Martínez, C.; Chiralt, A.; Kenny, J.M. Effect of cellulose nanocrystals on the properties of pea starch–poly (vinyl alcohol) blend films. J. Mater. Sci. 2015, 50, 6979–6992. [Google Scholar] [CrossRef]
- Habibi, Y. Key advances in the chemical modification of nanocelluloses. Chem. Soc. Rev. 2014, 43, 1519–1542. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Kenny, J.M.; Puglia, D. Structure and properties of biodegradable wheat gluten bionanocomposites containing lignin nanoparticles. Ind. Crops Prod. 2015, 74, 348–356. [Google Scholar] [CrossRef]
- Yang, W.; Owczarek, J.S.; Fortunati, E.; Kozanecki, M.; Mazzaglia, A.; Balestra, G.M.; Kenny, J.M.; Torre, L.; Puglia, D. Antioxidant and antibacterial lignin nanoparticles in polyvinyl alcohol/chitosan films for active packaging. Ind. Crops Prod. 2016, 94, 800–811. [Google Scholar] [CrossRef]
- Yang, W.; Fortunati, E.; Bertoglio, F.; Owczarek, J.S.; Bruni, G.; Kozanecki, M.; Kenny, J.M.; Torre, L.; Visai, L.; Puglia, D. Polyvinyl alcohol/chitosan hydrogels with enhanced antioxidant and antibacterial properties induced by lignin nanoparticles. Carbohydr. Polym. 2018, 181, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Thakur, V.K.; Thakur, M.K.; Raghavan, P.; Kessler, M.R. Progress in green polymer composites from lignin for multifunctional applications: A review. ACS Sustain. Chem. Eng. 2014, 2, 1072–1092. [Google Scholar] [CrossRef]
- Beisl, S.; Miltner, A.; Friedl, A. Lignin from micro-to nanosize: Production methods. Int. J. Mol. Sci. 2017, 18, 1244. [Google Scholar] [CrossRef] [PubMed]
- Tian, D.; Hu, J.; Bao, J.; Chandra, R.P.; Saddler, J.N.; Lu, C. Lignin valorization: Lignin nanoparticles as high-value bio-additive for multifunctional nanocomposites. Biotechnol. Biofuels 2017, 10, 192. [Google Scholar]
- Bertoft, E. Understanding starch structure: Recent progress. Agronomy 2017, 7, 56. [Google Scholar] [CrossRef]
- Chaudhary, D.; Adhikari, B. Effect of temperature and plasticizer molecular size on moisture diffusion of plasticized-starch biopolymer. Starch Stärke 2010, 62, 364–372. [Google Scholar] [CrossRef]
- Mäkelä, M.J.; Korpela, T.; Laakso, S. Studies of starch size and distribution in 33 barley varieties with a celloscope. Starch Stärke 1982, 34, 329–334. [Google Scholar] [CrossRef]
- De la Concha, B.B.S.; Agama-Acevedo, E.; Nuñez-Santiago, M.C.; Bello-Perez, L.A.; Garcia, H.S.; Alvarez-Ramirez, J. Acid hydrolysis of waxy starches with different granule size for nanocrystal production. J. Cereal Sci. 2018, 79, 193–200. [Google Scholar] [CrossRef]
- Salaberria, A.M.; Diaz, R.H.; Labidi, J.; Fernandes, S.C.M. Role of chitin nanocrystals and nanofibers on physical, mechanical and functional properties in thermoplastic starch films. Food Hydrocoll. 2015, 46, 93–102. [Google Scholar] [CrossRef]
- Salaberria, A.M.; Fernandes, S.C.M.; Diaz, R.H.; Labidi, J. Processing of α-chitin nanofibers by dynamic high pressure homogenization: Characterization and antifungal activity against A. niger. Carbohydr. Polym. 2015, 116, 286–291. [Google Scholar] [CrossRef]
- Wang, C.; Xiong, Y.; Fan, B.; Yao, Q.; Wang, H.; Jin, C.; Sun, Q. Cellulose as an adhesion agent for the synthesis of lignin aerogel with strong mechanical performance, Sound-absorption and thermal Insulation. Sci. Rep. 2016, 6, 32383. [Google Scholar] [CrossRef]
- Goodrich, J.D.; Winter, W.T. α-Chitin nanocrystals prepared from shrimp shells and their specific surface area measurement. Biomacromolecules 2007, 8, 252–257. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Fukuzumi, H.; Saito, T.; Isogai, A. Comparative characterization of aqueous dispersions and cast films of different chitin nanowhiskers/nanofibers. Int. J. Biol. Macromol. 2012, 50, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Gopalan Nair, K.; Dufresne, A. Crab shell chitin whisker reinforced natural rubber nanocomposites. 1. Processing and swelling behavior. Biomacromolecules 2003, 4, 657–665. [Google Scholar] [CrossRef]
- Ifuku, S.; Saimoto, H. Chitin nanofibers: Preparations, modifications, and applications. Nanoscale 2012, 4, 3308–3318. [Google Scholar] [CrossRef] [PubMed]
- Butchosa, N.; Brown, C.; Larsson, P.T.; Berglund, L.A.; Bulone, V.; Zhou, Q. Nanocomposites of bacterial cellulose nanofibers and chitin nanocrystals: Fabrication, characterization and bactericidal activity. Green Chem. 2013, 15, 3404–3413. [Google Scholar] [CrossRef]
- Zhang, H.; Li, R.; Liu, W. Effects of chitin and its derivative chitosan on postharvest decay of fruits: A review. Int. J. Mol. Sci. 2011, 12, 917–934. [Google Scholar] [CrossRef] [PubMed]
- Averous, L. Biodegradable multiphase systems based on plasticized starch: A review. J. Macromol. Sci. Polym. Rev. 2004, C44, 231–274. [Google Scholar] [CrossRef]
- Ezekiel Mushi, N.; Butchosa, N.; Zhou, Q.; Berglund, L.A. Nanopaper membranes from chitin–protein composite nanofibers—Structure and mechanical properties. J. Appl. Polym. Sci. 2014, 131, 40121. [Google Scholar] [CrossRef]
- Gopalan Nair, K.; Dufresne, A.; Gandini, A.; Belgacem, M.N. Crab shell chitin whiskers reinforced natural rubber nanocomposites. 3. Effect of chemical modification of chitin whiskers. Biomacromolecules 2003, 4, 1835–1842. [Google Scholar] [CrossRef] [PubMed]
- Morin, A.; Dufresne, A. Nanocomposites of chitin whiskers from Riftia tubes and poly (caprolactone). Macromolecules 2002, 35, 2190–2199. [Google Scholar] [CrossRef]
- Lu, Y.; Weng, L.; Zhang, L. Morphology and properties of soy protein isolate thermoplastics reinforced with chitin whiskers. Biomacromolecules 2004, 5, 1046–1051. [Google Scholar] [CrossRef] [PubMed]
- Sriupayo, J.; Supaphol, P.; Blackwell, J.; Rujiravanit, R. Preparation and characterization of α-chitin whisker-reinforced chitosan nanocomposite films with or without heat treatment. Carbohydr. Polym. 2005, 62, 130–136. [Google Scholar] [CrossRef]
- De Moura, M.R.; Avena-Bustillos, R.J.; McHugh, T.H.; Krochta, J.M.; Mattoso, L.H.C. Properties of novel hydroxypropyl methylcellulose films containing chitosan nanoparticles. J. Food Sci. 2008, 73, N31–N37. [Google Scholar] [CrossRef]
- Li, M.-C.; Wu, Q.; Song, K.; Cheng, H.N.; Suzuki, S.; Lei, T. Chitin nanofibers as reinforcing and antimicrobial agents in carboxymethyl cellulose films: Influence of partial deacetylation. ACS Sustain. Chem. Eng. 2016, 4, 4385–4395. [Google Scholar] [CrossRef]
- Mincea, M.; Negrulescu, A.; Ostafe, V. Preparation, modification, and applications of chitin nanowhiskers: A review. Rev. Adv. Mater. Sci. 2012, 30, 225–242. [Google Scholar]
- Wu, J.; Zhang, K.; Girouard, N.; Meredith, J.C. Facile route to produce chitin nanofibers as precursors for flexible and transparent gas barrier materials. Biomacromolecules 2014, 15, 4614–4620. [Google Scholar] [CrossRef]
- Huang, K.-S.; Sheu, Y.-R.; Chao, I.-C. Preparation and properties of nanochitosan. Polym. Plast. Technol. Eng. 2009, 48, 1239–1243. [Google Scholar] [CrossRef]
- Ding, D.-R.; Shen, Y. Antibacterial finishing with chitosan derivatives and their nano particles. Dye. Finish. 2005, 14, 002. [Google Scholar]
- Berthold, A.; Cremer, K.; Kreuter, J. Preparation and characterization of chitosan microspheres as drug carrier for prednisolone sodium phosphate as model for anti-inflammatory drugs. J. Control. Release 1996, 39, 17–25. [Google Scholar] [CrossRef]
- Tian, X.X.; Groves, M.J. Formulation and biological activity of antineoplastic proteoglycans derived from Mycobacterium vaccae in chitosan nanoparticles. J. Pharm. Pharmacol. 1999, 51, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Ohya, Y.; Shiratani, M.; Kobayashi, H.; Ouchi, T. Release behavior of 5-fluorouracil from chitosan-gel nanospheres immobilizing 5-fluorouracil coated with polysaccharides and their cell specific cytotoxicity. J. Macromol. Sci.—Pure Appl. Chem. 1994, 31, 629–642. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M.; Díez-Vicente, A.L. Antimicrobial and sustainable food packaging based on poly (butylene adipate-co-terephthalate) and electrospun chitosan nanofibers. RSC Adv. 2015, 5, 93095–93107. [Google Scholar] [CrossRef]
- Lee, H.; Noh, K.; Lee, S.C.; Kwon, I.-K.; Han, D.-W.; Lee, I.-S.; Hwang, Y.-S. Human hair keratin and its-based biomaterials for biomedical applications. Tissue Eng. Reg. Med. 2014, 11, 255–265. [Google Scholar] [CrossRef]
- Wang, J.; Hao, S.; Luo, T.; Yang, Q.; Wang, B. Development of feather keratin nanoparticles and investigation of their hemostatic efficacy. Mater. Sci. Eng. C 2016, 68, 768–773. [Google Scholar] [CrossRef]
- Li, L.; Wang, N.; Jin, X.; Deng, R.; Nie, S.; Sun, L.; Wu, Q.; Wei, Y.; Gong, C. Biodegradable and injectable in situ cross-linking chitosan-hyaluronic acid based hydrogels for postoperative adhesion prevention. Biomaterials 2014, 35, 3903–3917. [Google Scholar] [CrossRef]
- Pace, L.A.; Plate, J.F.; Smith, T.L.; Van Dyke, M.E. The effect of human hair keratin hydrogel on early cellular response to sciatic nerve injury in a rat model. Biomaterials 2013, 34, 5907–5914. [Google Scholar] [CrossRef]
- Poranki, D.; Whitener, W.; Howse, S.; Mesen, T.; Howse, E.; Burnell, J.; Greengauz-Roberts, O.; Molnar, J.; Van Dyke, M. Evaluation of skin regeneration after burns in vivo and rescue of cells after thermal stress in vitro following treatment with a keratin biomaterial. J. Biomater. Appl. 2014, 29, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Wang, Z.; Foo, S.E.M.; Tan, N.S.; Yuan, Y.; Lin, W.; Zhang, Z.; Ng, K.W. Culturing fibroblasts in 3D human hair keratin hydrogels. ACS Appl. Mater. Interfaces 2015, 7, 5187–5198. [Google Scholar] [CrossRef] [PubMed]
- Burnett, L.R.; Rahmany, M.B.; Richter, J.R.; Aboushwareb, T.A.; Eberli, D.; Ward, C.L.; Orlando, G.; Hantgan, R.R.; Van Dyke, M.E. Hemostatic properties and the role of cell receptor recognition in human hair keratin protein hydrogels. Biomaterials 2013, 34, 2632–2640. [Google Scholar] [CrossRef] [PubMed]
- Elzoghby, A.O. Gelatin-based nanoparticles as drug and gene delivery systems: Reviewing three decades of research. J. Control. Release 2013, 172, 1075–1091. [Google Scholar] [CrossRef] [PubMed]
- Flory, P.J.; Weaver, E.S. Helix [unk] coil transitions in dilute aqueous collagen solutions1. J. Am. Chem. Soc. 1960, 82, 4518–4525. [Google Scholar] [CrossRef]
- Kumari, A.; Yadav, S.K.; Yadav, S.C. Biodegradable polymeric nanoparticles based drug delivery systems. Colloids Surf. B Biointerfaces 2010, 75, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, B.; Aswal, V.K.; Kohlbrecher, J.; Bohidar, H.B. Synthesis of gelatin nanoparticles via simple coacervation. J. Surf. Sci. Technol. 2005, 21, 149. [Google Scholar]
- Zhao, Y.-Z.; Li, X.; Lu, C.-T.; Xu, Y.-Y.; Lv, H.-F.; Dai, D.-D.; Zhang, L.; Sun, C.-Z.; Yang, W.; Li, X.-K. Experiment on the feasibility of using modified gelatin nanoparticles as insulin pulmonary administration system for diabetes therapy. Acta Diabetol. 2012, 49, 315–325. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.K.; Gupta, M.; Yarwood, S.J.; Curtis, A.S.G. Effect of cellular uptake of gelatin nanoparticles on adhesion, morphology and cytoskeleton organisation of human fibroblasts. J. Control. Release 2004, 95, 197–207. [Google Scholar] [CrossRef]
- Fessi, H.; Puisieux, F.; Devissaguet, J.P.; Ammoury, N.; Benita, S. Nanocapsule formation by interfacial polymer deposition following solvent displacement. Int. J. Pharm. 1989, 55, R1–R4. [Google Scholar] [CrossRef]
- Galindo-Rodriguez, S.; Allemann, E.; Fessi, H.; Doelker, E. Physicochemical parameters associated with nanoparticle formation in the salting-out, emulsification-diffusion, and nanoprecipitation methods. Pharm. Res. 2004, 21, 1428–1439. [Google Scholar] [CrossRef] [PubMed]
- Ganachaud, F.; Katz, J.L. Nanoparticles and nanocapsules created using the Ouzo effect: Spontaneous emulsification as an alternative to ultrasonic and high-shear devices. ChemPhysChem 2005, 6, 209–216. [Google Scholar] [CrossRef]
- Sahoo, N.; Sahoo, R.K.; Biswas, N.; Guha, A.; Kuotsu, K. Recent advancement of gelatin nanoparticles in drug and vaccine delivery. Int. J. Biol. Macromol. 2015, 81, 317–331. [Google Scholar] [CrossRef] [PubMed]
- Arfat, Y.A.; Ahmed, J.; Hiremath, N.; Auras, R.; Joseph, A. Thermo-mechanical, rheological, structural and antimicrobial properties of bionanocomposite films based on fish skin gelatin and silver-copper nanoparticles. Food Hydrocoll. 2017, 62, 191–202. [Google Scholar] [CrossRef]
- Kumar, S.; Shukla, A.; Baul, P.P.; Mitra, A.; Halder, D. Biodegradable hybrid nanocomposites of chitosan/gelatin and silver nanoparticles for active food packaging applications. Food Package. Shelf Life 2018, 16, 178–184. [Google Scholar] [CrossRef]
- Plastics—The Facts 2017. Available online: www.plasticseurope.org/application/files/5715/1717/4180/Plastics_the_facts_2017_FINAL_for_website_one_page.pdf (accessed on 10 December 2018).
- Barlow, C.Y.; Morgan, D.C. Polymer film packaging for food: An environmental assessment. Resour. Conserv. Recycl. 2013, 78, 74–80. [Google Scholar] [CrossRef]
- Marsh, K.; Bugusu, B. Food packaging—Roles, materials, and environmental issues. J. Food Sci. 2007, 72, R39–R55. [Google Scholar] [CrossRef]
- Ramos, M.; Valdés, A.; Mellinas, A.; Garrigós, M. New trends in beverage packaging systems: A review. Beverages 2015, 1, 248–272. [Google Scholar] [CrossRef]
- Hilliou, L.; Covas, J.A. Production and Processing of Polymer-Based Nanocomposites. In Nanomaterials for Food Packaging; Elsevier: New York, NY, USA, 2018; pp. 111–146. [Google Scholar]
- Brandsch, J.; Piringer, O. Characteristics of plastic materials. In Plastic Packaging Materials for Food: Barrier Function, Mass Transport, Quality Assurance, and Legislation; John Wiley & Sons: Hoboken, NJ, USA, 2000; pp. 9–45. [Google Scholar]
- Selke, S.E.M.; Culter, J.D. Plastics Packaging: Properties, Processing, Applications, and Regulations; Carl Hanser Verlag GmbH Co KG: Munich, Germany, 2016. [Google Scholar]
- Scarfato, P.; Di Maio, L.; Incarnato, L.; Acierno, D.; Mariano, A. Influence of co-monomer structure on properties of co-polyamide packaging films. Packag. Technol. Sci. 2002, 15, 9–16. [Google Scholar] [CrossRef]
- Cabedo, L.; Lagarón, J.M.; Cava, D.; Saura, J.J.; Giménez, E. The effect of ethylene content on the interaction between ethylene-vinyl alcohol copolymers and water—II: Influence of water sorption on the mechanical properties of EVOH copolymers. Polym. Test. 2006, 25, 860–867. [Google Scholar] [CrossRef]
- López-Rubio, A.; Lagaron, J.M.; Giménez, E.; Cava, D.; Hernandez-Muñoz, P.; Yamamoto, T.; Gavara, R. Morphological Alterations Induced by Temperature and Humidity in Ethylene−Vinyl Alcohol Copolymers. Macromolecules 2003, 36, 9467–9476. [Google Scholar] [CrossRef]
- Lagaron, J.M.; Powell, A.K.; Bonner, G. Permeation of water, methanol, fuel and alcohol-containing fuels in high-barrier ethylene–vinyl alcohol copolymer. Polym. Test. 2001, 20, 569–577. [Google Scholar] [CrossRef]
- Luzi, F.; Puglia, D.; Dominici, F.; Fortunati, E.; Giovanale, G.; Balestra, G.M.; Torre, L. Effect of gallic acid and umbelliferone on thermal, mechanical, antioxidant and antimicrobial properties of poly (vinyl alcohol-co-ethylene) films. Polym. Degrad. Stab. 2018, 152, 162–176. [Google Scholar] [CrossRef]
- Blanchard, A.; Gouanvé, F.; Espuche, E. Effect of humidity on mechanical, thermal and barrier properties of EVOH films. J. Membr. Sci. 2017, 540, 1–9. [Google Scholar] [CrossRef]
- Lagarón, J.M.; Giménez, E.; Gavara, R.; Saura, J.J. Study of the influence of water sorption in pure components and binary blends of high barrier ethylene–vinyl alcohol copolymer and amorphous polyamide and nylon-containing ionomer. Polymer 2001, 42, 9531–9540. [Google Scholar] [CrossRef]
- Abdullah, Z.W.; Dong, Y.; Davies, I.J.; Barbhuiya, S. PVA, PVA blends, and their nanocomposites for biodegradable packaging application. Polym. Plast. Technol. Eng. 2017, 56, 1307–1344. [Google Scholar] [CrossRef]
- Zhou, J.; Ma, Y.; Ren, L.; Tong, J.; Liu, Z.; Xie, L. Preparation and characterization of surface crosslinked TPS/PVA blend films. Carbohydr. Polym. 2009, 76, 632–638. [Google Scholar] [CrossRef]
- Rahman, W.; Sin, L.T.; Rahmat, A.R.; Samad, A.A. Thermal behaviour and interactions of cassava starch filled with glycerol plasticized polyvinyl alcohol blends. Carbohydr. Polym. 2010, 81, 805–810. [Google Scholar] [CrossRef]
- López-Rubio, A.; Lagarón, J.M.; Hernandez-Munoz, P.; Almenar, E.; Catalá, R.; Gavara, R.; Pascall, M.A. Effect of high pressure treatments on the properties of EVOH-based food packaging materials. Innov. Food Sci. Emerg. Technol. 2005, 6, 51–58. [Google Scholar] [CrossRef]
- Liu, X.; Wu, Q.; Berglund, L.A.; Fan, J.; Qi, Z. Polyamide 6-clay nanocomposites/polypropylene-grafted-maleic anhydride alloys. Polymer 2001, 42, 8235–8239. [Google Scholar] [CrossRef]
- Birley, A.W. Plastics used in food packaging and the rôle of additives. Food Chem. 1982, 8, 81–84. [Google Scholar] [CrossRef]
- Kirwan, M.J.; Plant, S.; Strawbridge, J.W. Plastics in Food Packaging. In Food and Beverage Packaging Technology; Coles, R., Kirwan, M., Eds.; Blackwell Publishing Ltd.: Hoboken, NJ, USA, 2011; pp. 157–211. [Google Scholar]
- Finnigan, B. Barrier polymers. In The Wiley Encyclopedia of Packaging Technology; Yam, K.L., Ed.; John Wiley and Sons, Inc.: New York, NY, USA, 2009; pp. 103–109. [Google Scholar]
- Armentano, I.; Fortunati, E.; Burgos, N.; Dominici, F.; Luzi, F.; Fiori, S.; Jiménez, A.; Yoon, K.; Ahn, J.; Kang, S. Processing and characterization of plasticized PLA/PHB blends for biodegradable multiphase systems. Express Polym. Lett. 2015, 9, 583–596. [Google Scholar] [CrossRef]
- Fortunati, E.; Yang, W.; Luzi, F.; Kenny, J.; Torre, L.; Puglia, D. Lignocellulosic nanostructures as reinforcement in extruded and solvent casted polymeric nanocomposites: An overview. Eur. Polym. J. 2016, 80, 295–316. [Google Scholar] [CrossRef]
- Peelman, N.; Ragaert, P.; De Meulenaer, B.; Adons, D.; Peeters, R.; Cardon, L.; Van Impe, F.; Devlieghere, F. Application of bioplastics for food packaging. Trends Food Sci. Technol. 2013, 32, 128–141. [Google Scholar] [CrossRef]
- Tang, X.Z.; Kumar, P.; Alavi, S.; Sandeep, K.P. Recent advances in biopolymers and biopolymer-based nanocomposites for food packaging materials. Crit. Rev. Food sci. Nutr. 2012, 52, 426–442. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, V.; Samyn, P. Bio-based coatings for paper applications. Coatings 2015, 5, 887–930. [Google Scholar] [CrossRef]
- Rubie van Crevel, Bio-Based Food Packaging in Sustainable Development. 2016, Forest Products Team, Forestry Policy and Resources Division. Available online: http://www.fao.org/forestry/45849-023667e93ce5f79f4df3c74688c2067cc.pdf (accessed on 10 December 2018).
- Abbrescia, M.; Colaleo, A.; Iaselli, G.; Loddo, F.; Maggi, M.; Marangelli, B.; Natali, S.; Nuzzo, S.; Pugliese, G.; Ranieri, A. New developments on front-end electronics for the CMS Resistive Plate Chambers. Nucl. Instrum. Methods Phys. Res. Sect. A 2000, 456, 143–149. [Google Scholar] [CrossRef]
- Nakajima, H.; Dijkstra, P.; Loos, K. The recent developments in biobased polymers toward general and engineering applications: Polymers that are upgraded from biodegradable polymers, analogous to petroleum-derived polymers, and newly developed. Polymers 2017, 9, 523. [Google Scholar] [CrossRef]
- Assessment Conducted by Nova-Institut Concludes That First-Generation Fermentable Sugar Is Appropriate for a Sustainable Raw Material Strategy of the European Chemical Industry. Available online: https://www.european-bioplastics.org/tag/nova-institut/ (accessed on 10 December 2018).
- Thakur, V.K.; Thakur, M.K. Handbook of Sustainable Polymers: Processing and Applications; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar]
- Satam, C.C.; Irvin, C.W.; Lang, A.W.; Jallorina, J.C.R.; Shofner, M.L.; Reynolds, J.R.; Meredith, J.C. Spray-Coated Multilayer Cellulose Nanocrystal—Chitin Nanofiber Films for Barrier Applications. ACS Sustain. Chem. Eng. 2018, 6, 10637–10644. [Google Scholar] [CrossRef]
- Narayan, R. Biobased & biodegradable polymer materials: Rationale, drivers and technology examples. ACS Polym. Prepr. 2005, 46, 319–320. [Google Scholar]
- Vilar, M. Starch-Based Materials in Food Packaging: Processing, Characterization and Applications; Academic Press: Cambridge, MA, USA, 2017. [Google Scholar]
- Castro-Aguirre, E.; Iñiguez-Franco, F.; Samsudin, H.; Fang, X.; Auras, R. Poly (lactic acid)—Mass production, processing, industrial applications, and end of life. Adv. Drug Deliv. Rev. 2016, 107, 333–366. [Google Scholar] [CrossRef] [PubMed]
- Gadhave, R.V.; Das, A.; Mahanwar, P.A.; Gadekar, P.T. Starch Based Bio-Plastics: The Future of Sustainable Packaging. Open J. Polym. Chem. 2018, 8, 21. [Google Scholar] [CrossRef]
- Glenn, G.M.; Orts, W.; Imam, S.; Chiou, B.-S.; Wood, D.F. Starch plastic packaging and agriculture applications. In Starch Polymers; Elsevier: New York, NY, USA, 2014; pp. 421–452. [Google Scholar]
- Ferreira, A.; Alves, V.; Coelhoso, I. Polysaccharide-based membranes in food packaging applications. Membranes 2016, 6, 22. [Google Scholar] [CrossRef] [PubMed]
- Swain, S.K.; Mohanty, F. Polysaccharides-Based Bionanocomposites for Food Packaging Applications. In Bionanocomposites for Packaging Applications; Springer: Berlin, Germany, 2018; pp. 191–208. [Google Scholar]
- Credou, J.; Berthelot, T. Cellulose: From biocompatible to bioactive material. J. Mater. Chem. B 2014, 2, 4767–4788. [Google Scholar] [CrossRef]
- Tajeddin, B. Cellulose-Based Polymers for Packaging Applications. In Lignocellulosic Polymer Composites: Processing, Characterization, and Properties; John Wiley & Sons: Hoboken, NJ, USA, 2014; pp. 477–498. [Google Scholar]
- Shaghaleh, H.; Xu, X.; Wang, S. Current progress in production of biopolymeric materials based on cellulose, cellulose nanofibers, and cellulose derivatives. RSC Adv. 2018, 8, 825–842. [Google Scholar] [CrossRef]
- Hu, B. Biopolymer-based lightweight materials for packaging applications. Lightw. Mater. Biopolym. Biofibers 2014, 1175, 239–255. [Google Scholar]
- Arvanitoyannis, I.S. The use of chitin and chitosan for food packaging applications. In Environmentally Compatible Food Packaging; Woodhead Publishing: Sawston, CA, USA, 2008; Volume 137. [Google Scholar]
- Srinivasa, P.C.; Tharanathan, R.N. Chitin/Chitosan—Safe, Ecofriendly Packaging Materials with Multiple Potential Uses. Food Rev. Int. 2007, 23, 53–72. [Google Scholar] [CrossRef]
- Van den Broek, L.A.M.; Knoop, R.J.I.; Kappen, F.H.J.; Boeriu, C.G. Chitosan films and blends for packaging material. Carbohydr. Polym. 2015, 116, 237–242. [Google Scholar] [CrossRef]
- Rinaudo, M. Chitin and chitosan: Properties and applications. Prog. Polym. Sci. 2006, 31, 603–632. [Google Scholar] [CrossRef]
- Xu, Y.X.; Kim, K.M.; Hanna, M.A.; Nag, D. Chitosan–starch composite film: Preparation and characterization. Ind. Crops Prod. 2005, 21, 185–192. [Google Scholar] [CrossRef]
- Kurek, M.; Galus, S.; Debeaufort, F. Surface, mechanical and barrier properties of bio-based composite films based on chitosan and whey protein. Food Package. Shelf Life 2014, 1, 56–67. [Google Scholar] [CrossRef]
- Wang, H.; Qian, J.; Ding, F. Emerging Chitosan-Based Films for Food Packaging Applications. J. Agric. Food Chem. 2018, 66, 395–413. [Google Scholar] [CrossRef] [PubMed]
- Vijayendra, S.V.N.; Shamala, T.R. Film forming microbial biopolymers for commercial applications—A review. Crit. Rev. Biotechnol. 2014, 34, 338–357. [Google Scholar] [CrossRef] [PubMed]
- Arrieta, P.M.; Samper, D.M.; Aldas, M.; López, J. On the Use of PLA-PHB Blends for Sustainable Food Packaging Applications. Materials 2017, 10. [Google Scholar] [CrossRef]
- Plackett, D.; Siró, I. 18—Polyhydroxyalkanoates (PHAs) for food packaging. In Multifunctional and Nanoreinforced Polymers for Food Packaging; Lagarón, J.-M., Ed.; Woodhead Publishing: Sawston, CA, USA, 2011; pp. 498–526. [Google Scholar]
- Rhim, J.-W.; Hong, S.-I.; Ha, C.-S. Tensile, water vapor barrier and antimicrobial properties of PLA/nanoclay composite films. LWT Food Sci. Technol. 2009, 42, 612–617. [Google Scholar] [CrossRef]
- Rasal, R.M.; Janorkar, A.V.; Hirt, D.E. Poly (lactic acid) modifications. Prog. Polym. Sci. 2010, 35, 338–356. [Google Scholar] [CrossRef]
- Auras, R.; Harte, B.; Selke, S. An Overview of Polylactides as Packaging Materials. Macromol. Biosci. 2004, 4, 835–864. [Google Scholar] [CrossRef] [PubMed]
- Murariu, M.; Dubois, P. PLA composites: From production to properties. Adv. Drug Deliv. Rev. 2016, 107, 17–46. [Google Scholar] [CrossRef] [PubMed]
- Vroman, I.; Tighzert, L. Biodegradable Polymers. Materials 2009, 2. [Google Scholar] [CrossRef]
- Kijchavengkul, T.; Auras, R.; Rubino, M.; Selke, S.; Ngouajio, M.; Fernandez, R.T. Biodegradation and hydrolysis rate of aliphatic aromatic polyester. Polym. Degrad. Stab. 2010, 95, 2641–2647. [Google Scholar] [CrossRef]
- Rychter, P.; Kawalec, M.; Sobota, M.; Kurcok, P.; Kowalczuk, M. Study of Aliphatic-Aromatic Copolyester Degradation in Sandy Soil and Its Ecotoxicological Impact. Biomacromolecules 2010, 11, 839–847. [Google Scholar] [CrossRef] [PubMed]
- Biron, M. Industrial Applications of Renewable Plastics: Environmental, Technological, and Economic Advances; William Andrew: Norwich, NY, USA, 2016. [Google Scholar]
- Kyulavska, M.; Toncheva-Moncheva, N.; Rydz, J. Biobased Polyamide Ecomaterials and Their Susceptibility to Biodegradation. In Handbook of Ecomaterials; Martínez, L.M.T., Kharissova, O.V., Kharisov, B.I., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 1–34. [Google Scholar]
- Shi, B.; Wideman, G.; Wang, J.; Shlepr, M. Bio-based polyolefin composites and functional films for reducing total carbon footprint. J. Compos. Mater. 2014, 49, 2349–2355. [Google Scholar] [CrossRef]
- Kamigaito, M.; Satoh, K. Bio-based Hydrocarbon Polymers. In Encyclopedia of Polymeric Nanomaterials; Kobayashi, S., Müllen, K., Eds.; Springer Berlin Heidelberg: Berlin/Heidelberg, Germany, 2015; pp. 109–118. [Google Scholar]
- Sousa, A.F.; Vilela, C.; Fonseca, A.C.; Matos, M.; Freire, C.S.R.; Gruter, G.-J.M.; Coelho, J.F.J.; Silvestre, A.J.D. Biobased polyesters and other polymers from 2,5-furandicarboxylic acid: A tribute to furan excellency. Polym. Chem. 2015, 6, 5961–5983. [Google Scholar] [CrossRef]
- Gross, R.A.; Cheng, H.N.; Smith, P.B. Green Polymer Chemistry: Biobased Materials and Biocatalysis; American Chemical Society: Washington, DC, USA, 2015. [Google Scholar]
- Jost, V.; Miesbauer, O. Effect of different biopolymers and polymers on the mechanical and permeation properties of extruded PHBV cast films. J. Appl. Polym. Sci. 2018, 135, 46153. [Google Scholar] [CrossRef]
- Institute for Bioplastics and Biocomposites. Biopolymers—Facts and Statistics, Hannover 2016; IFBB—Institute for Bioplastics and Biocomposites: London, UK, 2016. [Google Scholar]
- Storz, H.; Vorlop, K.-D. Bio-based plastics: Status, challenges and trends. Landbauforschung 2013, 63, 321–332. [Google Scholar]
- Imam, S.; Glenn, G.; Chiou, B.S.; Shey, J.; Narayan, R.; Orts, W. Types, production and assessment of biobased food packaging materials. In Environmentally Compatible Food Packaging; Woodhead Publishing: Sawston, CA, USA, 2008; Volume 28. [Google Scholar]
- Aontee, A.; Sutapun, W. Effect of Blend Ratio on Phase Morphology and Mechanical Properties of High Density Polyethylene and Poly (Butylene Succinate) Blend; Trans Tech Publications: London, UK, 2013; pp. 555–559. [Google Scholar]
- Lackner, M. Bioplastics—Biobased Plastics as Renewable and/or Biodegradable Alternatives to Petroplastics, Kirk-Othmer Encyclopedia of Chemical Technology; John Wiley & Sons: Hoboken, NJ, USA, 2015. [Google Scholar]
- Alaerts, L.; Augustinus, M.; Van Acker, K. Impact of Bio-Based Plastics on Current Recycling of Plastics. Sustainability 2018, 10, 1487. [Google Scholar] [CrossRef]
- Lambert, S.; Wagner, M. Environmental performance of bio-based and biodegradable plastics: The road ahead. Chem. Soc. Rev. 2017, 46, 6855–6871. [Google Scholar] [CrossRef]
- Gironi, F.; Piemonte, V. Bioplastics and petroleum-based plastics: Strengths and weaknesses. Energy Sources Part A 2011, 33, 1949–1959. [Google Scholar] [CrossRef]
- Peres, A.M.; Pires, R.R.; Oréfice, R.L. Evaluation of the effect of reprocessing on the structure and properties of low density polyethylene/thermoplastic starch blends. Carbohydr. Polym. 2016, 136, 210–215. [Google Scholar] [CrossRef]
- Okan, M.; Aydin, H.M.; Barsbay, M. Current approaches to waste polymer utilization and minimization: A review. J. Chem. Technol. Biotechnol. 2019, 94, 8–21. [Google Scholar] [CrossRef]
- Álvarez-Chávez, C.R.; Edwards, S.; Moure-Eraso, R.; Geiser, K. Sustainability of bio-based plastics: General comparative analysis and recommendations for improvement. J. Clean. Prod. 2012, 23, 47–56. [Google Scholar] [CrossRef]
- Imre, B.; Pukánszky, B. Compatibilization in bio-based and biodegradable polymer blends. Eur. Polym. J. 2013, 49, 1215–1233. [Google Scholar] [CrossRef]
- Khan, B.; Bilal Khan Niazi, M.; Samin, G.; Jahan, Z. Thermoplastic Starch: A Possible Biodegradable Food Packaging Material—A Review. J. Food Process Eng. 2016, 40, e12447. [Google Scholar] [CrossRef]
- Tabasum, S.; Younas, M.; Zaeem, M.A.; Majeed, I.; Majeed, M.; Noreen, A.; Iqbal, M.N.; Zia, K.M. A review on blending of corn starch with natural and synthetic polymers, and inorganic nanoparticles with mathematical modeling. Int. J. Biol. Macromol. 2019, 122, 969–996. [Google Scholar] [CrossRef] [PubMed]
- Rosa, D.S.; Guedes, C.G.F.; Carvalho, C.L. Processing and thermal, mechanical and morphological characterization of post-consumer polyolefins/thermoplastic starch blends. J. Mater. Sci. 2007, 42, 551–557. [Google Scholar] [CrossRef]
- Martins, A.B.; Santana, R.M.C. Effect of carboxylic acids as compatibilizer agent on mechanical properties of thermoplastic starch and polypropylene blends. Carbohydr. Polym. 2016, 135, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Debiagi, F.; Mello, L.R.P.F.; Mali, S. Chapter 6—Thermoplastic Starch-Based Blends: Processing, Structural, and Final Properties. In Starch-Based Materials in Food Packaging; Villar, M.A., Barbosa, S.E., García, M.A., Castillo, L.A., López, O.V., Eds.; Academic Press: Cambridge, MA, USA, 2017; pp. 153–186. [Google Scholar]
- Cerclé, C.; Sarazin, P.; Favis, B.D. High performance polyethylene/thermoplastic starch blends through controlled emulsification phenomena. Carbohydr. Polym. 2013, 92, 138–148. [Google Scholar] [CrossRef]
- Nguyen, D.M.; Vu, T.T.; Grillet, A.-C.; Ha Thuc, H.; Ha Thuc, C.N. Effect of organoclay on morphology and properties of linear low density polyethylene and Vietnamese cassava starch biobased blend. Carbohydr. Polym. 2016, 136, 163–170. [Google Scholar] [CrossRef]
- Kuciel, S.; Kuźniar, P.; Nykiel, M. Biodegradable polymers in the general waste stream—The issue of recycling with polyethylene packaging materials. Polimery 2018, 63, 1–8. [Google Scholar] [CrossRef]
- Soroudi, A.; Jakubowicz, I. Recycling of bioplastics, their blends and biocomposites: A review. Eur. Polym. J. 2013, 49, 2839–2858. [Google Scholar] [CrossRef]
- Teyssandier, F.; Cassagnau, P.; Gérard, J.F.; Mignard, N. Reactive compatibilization of PA12/plasticized starch blends: Towards improved mechanical properties. Eur. Polym. J. 2011, 47, 2361–2371. [Google Scholar] [CrossRef]
- Burlein, G.A.D.; Rocha, M.C.G. Mechanical and morphological properties of LDPE/PHB blends filled with castor oil pressed cake. Mater. Res. 2014, 17, 97–105. [Google Scholar] [CrossRef]
- Abdelwahab, M.A.; Martinelli, E.; Alderighi, M.; Grillo Fernandes, E.; Imam, S.; Morelli, A.; Chiellini, E. Poly[(R)-3-hydroxybutyrate)]/poly(styrene) blends compatibilized with the relevant block copolymer. J. Polym. Sci. Part A Polym. Chem. 2012, 50, 5151–5160. [Google Scholar] [CrossRef]
- Imre, B.; Renner, K.; Pukánszky, B. Interactions, structure and properties in poly (lactic acid)/thermoplastic polymer blends. Express Polym. Lett. 2014, 8, 2–14. [Google Scholar] [CrossRef]
- Sabetzadeh, M.; Bagheri, R.; Masoomi, M. Study on ternary low density polyethylene/linear low density polyethylene/thermoplastic starch blend films. Carbohydr. Polym. 2015, 119, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Shujun, W.; Jiugao, Y.; Jinglin, Y. Preparation and characterization of compatible thermoplastic starch/polyethylene blends. Polym. Degrad. Stab. 2005, 87, 395–401. [Google Scholar] [CrossRef]
- Arvanitoyannis, I.; Biliaderis, C.G.; Ogawa, H.; Kawasaki, N. Biodegradable films made from low-density polyethylene (LDPE), rice starch and potato starch for food packaging applications: Part 1. Carbohydr. Polym. 1998, 36, 89–104. [Google Scholar] [CrossRef]
- Pedroso, A.G.; Rosa, D.S. Mechanical, thermal and morphological characterization of recycled LDPE/corn starch blends. Carbohydr. Polym. 2005, 59, 1–9. [Google Scholar] [CrossRef]
- Euaphantasate, N.; Prachayawasin, P.; Uasopon, S.; Methacanon, P. Moisture sorption characteristic and their relative properties of thermoplastic starch/linear low density polyethylene films for food packaging. J. Met. Mater. Min. 2008, 18, 103–109. [Google Scholar]
- Landreau, E.; Tighzert, L.; Bliard, C.; Berzin, F.; Lacoste, C. Morphologies and properties of plasticized starch/polyamide compatibilized blends. Eur. Polym. J. 2009, 45, 2609–2618. [Google Scholar] [CrossRef]
- TureČKovÁ, J.; Prokopova, I.; Niklova, P.; ŠImek, J.A.N.; ŠMejkalovÁ, P.; KeclÍK, F. Biodegradable copolyester/starch blends-preparation, mechanical properties, wettability, biodegradation course (in English). Polimery 2008, 53, 639–643. [Google Scholar] [CrossRef]
- Gutiérrez, T.J.; Alvarez, V.A. Properties of native and oxidized corn starch/polystyrene blends under conditions of reactive extrusion using zinc octanoate as a catalyst. React. Funct. Polym. 2017, 112, 33–44. [Google Scholar] [CrossRef]
- Men, Y.; Du, X.; Shen, J.; Wang, L.; Liu, Z. Preparation of corn starch-g-polystyrene copolymer in ionic liquid: 1-Ethyl-3-methylimidazolium acetate. Carbohydr. Polym. 2015, 121, 348–354. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.I.; Ahmed, S.; Javed, I.; Ali, N.; Atiq, N.; Hameed, A.; Robson, G. Biodegradation of starch blended polyvinyl chloride films by isolated Phanerochaete chrysosporium PV1. Int. J. Environ. Sci. Technol. 2014, 11, 339–348. [Google Scholar] [CrossRef]
- Jiang, W.; Qiao, X.; Sun, K. Mechanical and thermal properties of thermoplastic acetylated starch/poly (ethylene-co-vinyl alcohol) blends. Carbohydr. Polym. 2006, 65, 139–143. [Google Scholar] [CrossRef]
- George, E.R.; Sullivan, T.M.; Park, E.H. Thermoplastic starch blends with a poly (ethylene-co-vinyl alcohol): Processability and physical properties. Polym. Eng. Sci. 1994, 34, 17–23. [Google Scholar] [CrossRef]
- Orts, W.J.; Nobes, G.A.R.; Glenn, G.M.; Gray, G.M.; Imam, S.; Chiou, B.S. Blends of starch with ethylene vinyl alcohol copolymers: Effect of water, glycerol, and amino acids as plasticizers. Polym. Adv. Technol. 2007, 18, 629–635. [Google Scholar] [CrossRef]
- Simmons, S.; Thomas, E.L. Structural characteristics of biodegradable thermoplastic starch/poly (ethylene–vinyl alcohol) blends. J. Appl. Polym. Sci. 1995, 58, 2259–2285. [Google Scholar] [CrossRef]
- Wu, Z.; Wu, J.; Peng, T.; Li, Y.; Lin, D.; Xing, B.; Li, C.; Yang, Y.; Yang, L.; Zhang, L. Preparation and application of starch/polyvinyl alcohol/citric acid ternary blend antimicrobial functional food packaging films. Polymers 2017, 9, 102. [Google Scholar] [CrossRef]
- Guohua, Z.; Ya, L.; Cuilan, F.; Min, Z.; Caiqiong, Z.; Zongdao, C. Water resistance, mechanical properties and biodegradability of methylated-cornstarch/poly (vinyl alcohol) blend film. Polym. Degrad. Stab. 2006, 91, 703–711. [Google Scholar] [CrossRef]
- Jayasekara, R.; Harding, I.; Bowater, I.; Christie, G.B.Y.; Lonergan, G.T. Preparation, surface modification and characterisation of solution cast starch PVA blended films. Polym. Test. 2004, 23, 17–27. [Google Scholar] [CrossRef]
- El-Hefian, E.A.; Nasef, M.M.; Yahaya, A.H. Chitosan-Based Polymer Blends: Current Status and Applications. J. Chem. Soc. Pak. 2014, 36, 11–27. [Google Scholar]
- Kausar, A. Scientific potential of chitosan blending with different polymeric materials: A review. J. Plast. Film Sheet. 2017, 33, 384–412. [Google Scholar] [CrossRef]
- Li, H.-Z.; Chen, S.-C.; Wang, Y.-Z. Preparation and characterization of nanocomposites of polyvinyl alcohol/cellulose nanowhiskers/chitosan. Compos. Sci. Technol. 2015, 115, 60–65. [Google Scholar] [CrossRef]
- Lima, P.S.; Brito, R.S.F.; Santos, B.F.F.; Tavares, A.A.; Agrawal, P.; Andrade, D.L.; Wellen, R.M.R.; Canedo, E.L.; Silva, S.M.L. Rheological properties of HDPE/chitosan composites modified with PE-g-MA. J. Mater. Res. 2017, 32, 775–787. [Google Scholar] [CrossRef]
- Quiroz-Castillo, J.M.; Rodríguez-Félix, D.E.; Grijalva-Monteverde, H.; del Castillo-Castro, T.; Plascencia-Jatomea, M.; Rodríguez-Félix, F.; Herrera-Franco, P.J. Preparation of extruded polyethylene/chitosan blends compatibilized with polyethylene-graft-maleic anhydride. Carbohydr. Polym. 2014, 101, 1094–1100. [Google Scholar] [CrossRef] [PubMed]
- Kuo, P.-C.; Sahu, D.; Yu, H.H. Properties and biodegradability of chitosan/nylon 11 blending films. Polym. Degrad. Stab. 2006, 91, 3097–3102. [Google Scholar] [CrossRef]
- Smitha, B.; Dhanuja, G.; Sridhar, S. Dehydration of 1, 4-dioxane by pervaporation using modified blend membranes of chitosan and nylon 66. Carbohydr. Polym. 2006, 66, 463–472. [Google Scholar] [CrossRef]
- Masoomi, M.; Tavangar, M.; Razavi, S.M.R. Preparation and investigation of mechanical and antibacterial properties of poly (ethylene terephthalate)/chitosan blend. RSC Adv. 2015, 5, 79200–79206. [Google Scholar] [CrossRef]
- Mascarenhas, N.P.; Gonsalves, R.A.; Goveas, J.J.; Shetty, T.C.S.; Crasta, V. Preparation and characterization of chitosan-polystyrene polymer blends. AIP Conf. 2016, 1731, 140039. [Google Scholar]
- Carrasco-Guigón, F.J.; Rodríguez-Félix, D.E.; Castillo-Ortega, M.M.; Santacruz-Ortega, H.C.; Burruel-Ibarra, S.E.; Encinas-Encinas, J.C.; Plascencia-Jatomea, M.; Herrera-Franco, P.J.; Madera-Santana, T.J. Preparation and Characterization of Extruded Composites Based on Polypropylene and Chitosan Compatibilized with Polypropylene-Graft-Maleic Anhydride. Materials 2017, 10, 105. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Saiz, P.; Ocio, M.J.; Lagaron, J.M. Antibacterial chitosan-based blends with ethylene–vinyl alcohol copolymer. Carbohydr. Polym. 2010, 80, 874–884. [Google Scholar] [CrossRef]
- Ouattara, B.; Simard, R.E.; Piette, G.; Begin, A.; Holley, R.A. Diffusion of acetic and propionic acids from chitosan-based antimicrobial packaging films. J. Food Sci. 2000, 65, 768–773. [Google Scholar] [CrossRef]
- Dutta, P.K.; Tripathi, S.; Mehrotra, G.K.; Dutta, J. Perspectives for chitosan based antimicrobial films in food applications. Food Chem. 2009, 114, 1173–1182. [Google Scholar] [CrossRef]
- Campos, C.A.; Gerschenson, L.N.; Flores, S.K. Development of edible films and coatings with antimicrobial activity. Food Bioprocess Technol. 2011, 4, 849–875. [Google Scholar] [CrossRef]
- Douglass, E.F.; Avci, H.; Boy, R.; Rojas, O.J.; Kotek, R. A review of cellulose and cellulose blends for preparation of bio-derived and conventional membranes, nanostructured thin films, and composites. Polym. Rev. 2018, 58, 102–163. [Google Scholar] [CrossRef]
- Suvorova, A.I.; Tyukova, I.S.; Trufanova, E.I. Biodegradable starch-based polymeric materials. Russ. Chem. Rev. 2000, 69, 451. [Google Scholar] [CrossRef]
- Yoo, S.; Krochta, J.M. Starch–methylcellulose–whey protein film properties. Int. J. Food Sci. Technol. 2012, 47, 255–261. [Google Scholar] [CrossRef]
- Paunonen, S. Strength and barrier enhancements of cellophane and cellulose derivative films: A review. BioResources 2013, 8, 3098–3121. [Google Scholar] [CrossRef]
- Khan, R.A.; Salmieri, S.; Dussault, D.; Sharmin, N.; Lacroix, M. Mechanical, barrier, and interfacial properties of biodegradable composite films made of methylcellulose and poly (caprolactone). J. Appl. Polym. Sci. 2012, 123, 1690–1697. [Google Scholar] [CrossRef]
- Nabar, Y.U.; Gupta, A.; Narayan, R. Isothermal crystallization kinetics of poly (ethylene terephthalate)–cellulose acetate blends. Polym. Bull. 2005, 53, 117–125. [Google Scholar] [CrossRef]
- El-Gendi, A.; Abdallah, H.; Amin, A.; Amin, S.K. Investigation of polyvinylchloride and cellulose acetate blend membranes for desalination. J. Mol. Struct. 2017, 1146, 14–22. [Google Scholar] [CrossRef]
- Miyashita, Y.; Suzuki, T.; Nishio, Y. Miscibility of cellulose acetate with vinyl polymers. Cellulose 2002, 9, 215–223. [Google Scholar] [CrossRef]
- Abdel-Naby, A.S.; Al-Ghamdi, A.A. Poly (vinyl chloride) blend with biodegradable cellulose acetate in presence of N-(phenyl amino) maleimides. Int. J. Biol. Macromol. 2014, 70, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Khaparde, D. Preparation and prediction of physical properties of cellulose acetate and polyamide polymer blend. Carbohydr. Polym. 2017, 173, 338–343. [Google Scholar] [CrossRef] [PubMed]
- Hopmann, C.; Hendriks, S.; Spicker, C.; Zepnik, S.; van Lück, F. Surface roughness and foam morphology of cellulose acetate sheets foamed with 1, 3, 3, 3-tetrafluoropropene. Polym. Eng. Sci. 2017, 57, 441–449. [Google Scholar] [CrossRef]
- Meenakshi, P.; Noorjahan, S.E.; Rajini, R.; Venkateswarlu, U.; Rose, C.; Sastry, T.P. Mechanical and microstructure studies on the modification of CA film by blending with PS. Bull. Mater. Sci. 2002, 25, 25–29. [Google Scholar] [CrossRef]
- Meireles, C.d.S.; Filho, G.R.; de Assunção, R.M.N.; Zeni, M.; Mello, K. Blend compatibility of waste materials—Cellulose acetate (from sugarcane bagasse) with polystyrene (from plastic cups): Diffusion of water, FTIR, DSC, TGA, and SEM study. J. Appl. Polym. Sci. 2007, 104, 909–914. [Google Scholar] [CrossRef]
- Dias, D.S.; Crespi, M.S.; Kobelnik, M.; Ribeiro, C.A. Calorimetric and SEM studies of PHB–PET polymeric blends. J. Therm. Anal. Calorim. 2009, 97, 581–584. [Google Scholar] [CrossRef]
- Ol’khov, A.A.; Iordanskii, A.L.; Zaikov, G.E.; Shibryaeva, L.S.; Litwinov, I.A.; Vlasov, S.V. Morphological Features of Poly (3-Hydroxybutyrate)/Low Density Polyethylene Blends. Int. J. Polym. Mater. Polym. Biomater. 2000, 47, 457–468. [Google Scholar] [CrossRef]
- Ol’Khov, A.A.; Iordanskii, A.L.; Zaikov, G.E.; Shibryaeva, L.S.; Litvinov†, I.A.; Vlasov, S.V. Morphologically Special Features of Poly(3-Hydroxybutyrate)/Low-Density Polyethylene Blends. Polym. Plast. Technol. Eng. 2000, 39, 783–792. [Google Scholar] [CrossRef]
- Fabbri, P.; Bassoli, E.; Bon, S.B.; Valentini, L. Preparation and characterization of poly (butylene terephthalate)/graphene composites by in-situ polymerization of cyclic butylene terephthalate. Polymer 2012, 53, 897–902. [Google Scholar] [CrossRef]
- Pachekoski, W.M.; Marcondes Agnelli, J.A.; Belem Thermal, L.P. Mechanical and morphological properties of poly (hydroxybutyrate) and polypropylene blends after processing. Mat. Res. 2009, 12, 159–164. [Google Scholar] [CrossRef]
- Olkhov, A.A.; Pankova, Y.N.; Goldshtrakh, M.A.; Kosenko, R.Y.; Markin, V.S.; Ischenko, A.A.; Iordanskiy, A.L. Structure and properties of films based on blends of polyamide–polyhydroxybutyrate. Inorg. Mater. 2016, 7, 471–477. [Google Scholar] [CrossRef]
- El-Hadi, A.; Schnabel, R.; Straube, E.; Müller, G.; Riemschneider, M. Effect of Melt Processing on Crystallization Behavior and Rheology of Poly(3-hydroxybutyrate) (PHB) and its Blends. Macromol. Mater. Eng. 2002, 287, 363–372. [Google Scholar] [CrossRef]
- You, X.; Snowdon, M.R.; Misra, M.; Mohanty, A.K. Biobased Poly(ethylene terephthalate)/Poly(lactic acid) Blends Tailored with Epoxide Compatibilizers. ACS Omega 2018, 3, 11759–11769. [Google Scholar] [CrossRef]
- McLauchlin, A.R.; Ghita, O.R. Studies on the thermal and mechanical behavior of PLA-PET blends. J. Appl. Polym. Sci. 2016, 133. [Google Scholar] [CrossRef]
- Sangermano, M.; Marchi, S.; Valentini, L.; Bon, S.B.; Fabbri, P. Transparent and Conductive Graphene Oxide/Poly(ethylene glycol) diacrylate Coatings Obtained by Photopolymerization. Macromol. Mater. Eng. 2010, 296, 401–407. [Google Scholar] [CrossRef]
- Hachemi, R.; Belhaneche-Bensemra, N.; Massardier, V. Elaboration and characterization of bioblends based on PVC/PLA. J. Appl. Polym. Sci. 2013, 131. [Google Scholar] [CrossRef]
- Kaseem, M.; Ko, Y.G. Melt Flow Behavior and Processability of Polylactic Acid/Polystyrene (PLA/PS) Polymer Blends. J. Polym. Environ. 2017, 25, 994–998. [Google Scholar] [CrossRef]
- Vital, A.; Vayer, M.; Tillocher, T.; Dussart, R.; Boufnichel, M.; Sinturel, C. Morphology control in thin films of PS:PLA homopolymer blends by dip-coating deposition. Appl. Surf. Sci. 2017, 393, 127–133. [Google Scholar] [CrossRef]
- Feng, F.; Ye, L. Structure and Property of Polylactide/Polyamide Blends. J. Macromol. Sci. Part B 2010, 49, 1117–1127. [Google Scholar] [CrossRef]
- Patel, R.; Ruehle, D.A.; Dorgan, J.R.; Halley, P.; Martin, D. Biorenewable blends of polyamide-11 and polylactide. Polym. Eng. Sci. 2013, 54, 1523–1532. [Google Scholar] [CrossRef]
- Stoclet, G.; Seguela, R.; Lefebvre, J.M. Morphology, thermal behavior and mechanical properties of binary blends of compatible biosourced polymers: Polylactide/polyamide11. Polymer 2011, 52, 1417–1425. [Google Scholar] [CrossRef]
- Dong, W.; Cao, X.; Li, Y. High-performance biosourced poly(lactic acid)/polyamide 11 blends with controlled salami structure. Polym. Int. 2013, 63, 1094–1100. [Google Scholar] [CrossRef]
- Walha, F.; Lamnawar, K.; Maazouz, A.; Jaziri, M. Biosourced blends based on poly (lactic acid) and polyamide 11: Structure-properties relationships and enhancement of film blowing processability. Adv. Polym. Technol. 2017, 37, 2061–2074. [Google Scholar] [CrossRef]
- Pai, F.-C.; Lai, S.-M.; Chu, H.-H. Characterization and Properties of Reactive Poly(lactic acid)/Polyamide 610 Biomass Blends. J. Appl. Polym. Sci. 2013, 130, 2563–2571. [Google Scholar] [CrossRef]
- Li, H.-Z.; Chen, S.-C.; Wang, Y.-Z. Thermoplastic PVA/PLA Blends with Improved Processability and Hydrophobicity. Ind. Eng. Chem. Res. 2014, 53, 17355–17361. [Google Scholar] [CrossRef]
- Shuai, X.; He, Y.; Asakawa, N.; Inoue, Y. Miscibility and phase structure of binary blends of poly(L-lactide) and poly(vinyl alcohol). J. Appl. Polym. Sci. 2001, 81, 762–772. [Google Scholar] [CrossRef]
- Threepopnatkul, P.; Wongnarat, C.; Intolo, W.; Suato, S.; Kulsetthanchalee, C. Effect of TiO2 and ZnO on Thin Film Properties of PET/PBS Blend for Food Packaging Applications. Energy Procedia 2014, 56, 102–111. [Google Scholar] [CrossRef]
- Chuayjuljit, S.; Kongthan, J.; Chaiwutthinan, P.; Boonmahitthisud, A. Poly(vinyl chloride)/Poly(butylene succinate)/wood flour composites: Physical properties and biodegradability. Polym. Compos. 2016, 39, 1543–1552. [Google Scholar] [CrossRef]
- Thongsong, W.; Kulsetthanchalee, C.; Threepopnatkul, P. Effect of polybutylene adipate-co-terephthalate on properties of polyethylene terephthalate thin films. Mater. Today 2017, 4, 6597–6604. [Google Scholar] [CrossRef]
- Brito, G.F.; Agrawal, P.; Mélo, T.J.A. Mechanical and Morphological Properties of PLA/BioPE Blend Compatibilized with E-GMA and EMA-GMA Copolymers. Macromol. Symp. 2016, 367, 176–182. [Google Scholar] [CrossRef]
- Muller, J.; González-Martínez, C.; Chiralt, A. Combination of Poly(lactic) Acid and Starch for Biodegradable Food Packaging. Materials 2017, 10. [Google Scholar] [CrossRef]
- Rhim, J.-W.; Kim, Y.-T. Chapter 17—Biopolymer-Based Composite Packaging Materials with Nanoparticles. In Innovations in Food Packaging, 2nd ed.; Han, J.H., Ed.; Academic Press: San Diego, CA, USA, 2014; pp. 413–442. [Google Scholar]
- Ahmed, S. (Ed.) Bio-Based Materials for Food Packaging; Springer: Berlin, Germany, 2018. [Google Scholar]
- Swain, S.K. Gas Barrier Properties of Biopolymer-based Nanocomposites: Application in Food Packaging. Adv. Mater. Agric. Food Environ. Saf. 2014, 369–384. [Google Scholar] [CrossRef]
- Lagaron, J.M.; Núñez, E. Nanocomposites of moisture-sensitive polymers and biopolymers with enhanced performance for flexible packaging applications. J. Plast. Film Sheet. 2011, 28, 79–89. [Google Scholar] [CrossRef]
- Wolf, C.; Angellier-Coussy, H.; Gontard, N.; Doghieri, F.; Guillard, V. How the shape of fillers affects the barrier properties of polymer/non-porous particles nanocomposites: A review. J. Membr. Sci. 2018, 556, 393–418. [Google Scholar] [CrossRef]
- Majid, I.; Thakur, M.; Nanda, V. Biodegradable Packaging Materials. Ref. Mod. Mater. Sci. Mater. Eng. 2018. [Google Scholar] [CrossRef]
- Sadeghi, G.M.M.; Mahsa, S. Compostable Polymers and Nanocomposites—A Big Chance for Planet Earth, Recycling Materials Based on Environmentally Friendly Techniques; Achilias, D.S., Ed.; IntechOpen: Rijeka, Croatia, 2015. [Google Scholar]
- Gutiérrez, T.J. Biodegradability and Compostability of Food Nanopackaging Materials. In Composites Materials for Food Packaging; Cirillo, G., Kozlowski, M.A., Spizzirri, U.G., Eds.; Springer: Berlin, Germany, 2018; pp. 269–296. [Google Scholar]
- Zinoviadou, K.G.; Gougouli, M.; Biliaderis, C.G. Chapter 9—Innovative Biobased Materials for Packaging Sustainability. In Innovation Strategies in the Food Industry; Galanakis, C.M., Ed.; Academic Press: Cambridge, MA, USA, 2016; pp. 167–189. [Google Scholar]
- Naderizadeh, S.; Shakeri, A.; Mahdavi, H.; Nikfarjam, N.; Taheri Qazvini, N. Hybrid Nanocomposite Films of Starch, Poly(vinyl alcohol) (PVA), Starch Nanocrystals (SNCs), and Montmorillonite (Na-MMT): Structure–Properties Relationship. Starch Stärke 2018, 1800027. [Google Scholar] [CrossRef]
- Ashori, A. Effects of graphene on the behavior of chitosan and starch nanocomposite films. Polym. Eng. Sci. 2013, 54, 2258–2263. [Google Scholar] [CrossRef]
- Vertuccio, L.; Gorrasi, G.; Sorrentino, A.; Vittoria, V. Nano clay reinforced PCL/starch blends obtained by high energy ball milling. Carbohydr. Polym. 2009, 75, 172–179. [Google Scholar] [CrossRef]
- Ikeo, Y.; Aoki, K.; Kishi, H.; Matsuda, S.; Murakami, A. Nano clay reinforced biodegradable plastics of PCL starch blends. Polym. Adv. Technol. 2006, 17, 940–944. [Google Scholar] [CrossRef]
- Liao, H.-T.; Wu, C.-S. Synthesis and characterization of polyethylene-octene elastomer/clay/biodegradable starch nanocomposites. J. Appl. Polym. Sci. 2005, 97, 397–404. [Google Scholar] [CrossRef]
- DeLeo, C.; Pinotti, C.A.; do Carmo Gonçalves, M.; Velankar, S. Preparation and Characterization of Clay Nanocomposites of Plasticized Starch and Polypropylene Polymer Blends. J. Polym. Environ. 2011, 19, 689. [Google Scholar] [CrossRef]
- Tang, S.; Zou, P.; Xiong, H.; Tang, H. Effect of nano-SiO2 on the performance of starch/polyvinyl alcohol blend films. Carbohydr. Polym. 2008, 72, 521–526. [Google Scholar] [CrossRef]
- Spiridon, I.; Popescu, M.C.; Bodârlău, R.; Vasile, C. Enzymatic degradation of some nanocomposites of poly(vinyl alcohol) with starch. Polym. Degrad. Stab. 2008, 93, 1884–1890. [Google Scholar] [CrossRef]
- Tang, X.; Alavi, S. Structure and Physical Properties of Starch/Poly Vinyl Alcohol/Laponite RD Nanocomposite Films. J. Agric. Food Chem. 2012, 60, 1954–1962. [Google Scholar] [CrossRef]
- Alix, S.; Mahieu, A.; Terrie, C.; Soulestin, J.; Gerault, E.; Feuilloley, M.G.J.; Gattin, R.; Edon, V.; Ait-Younes, T.; Leblanc, N. Active pseudo-multilayered films from polycaprolactone and starch based matrix for food-packaging applications. Eur. Polym. J. 2013, 49, 1234–1242. [Google Scholar] [CrossRef]
- Tang, X.; Alavi, S. Recent advances in starch, polyvinyl alcohol based polymer blends, nanocomposites and their biodegradability. Carbohydr. Polym. 2011, 85, 7–16. [Google Scholar] [CrossRef]
- El Achaby, M.; El Miri, N.; Aboulkas, A.; Zahouily, M.; Bilal, E.; Barakat, A.; Solhy, A. Processing and properties of eco-friendly bio-nanocomposite films filled with cellulose nanocrystals from sugarcane bagasse. Int. J. Biol. Macromol. 2017, 96, 340–352. [Google Scholar] [CrossRef]
- Taghizadeh, M.T.; Sabouri, N.; Ghanbarzadeh, B. Polyvinyl alcohol:starch:carboxymethyl cellulose containing sodium montmorillonite clay blends; mechanical properties and biodegradation behavior. SpringerPlus 2013, 2, 376. [Google Scholar] [CrossRef] [PubMed]
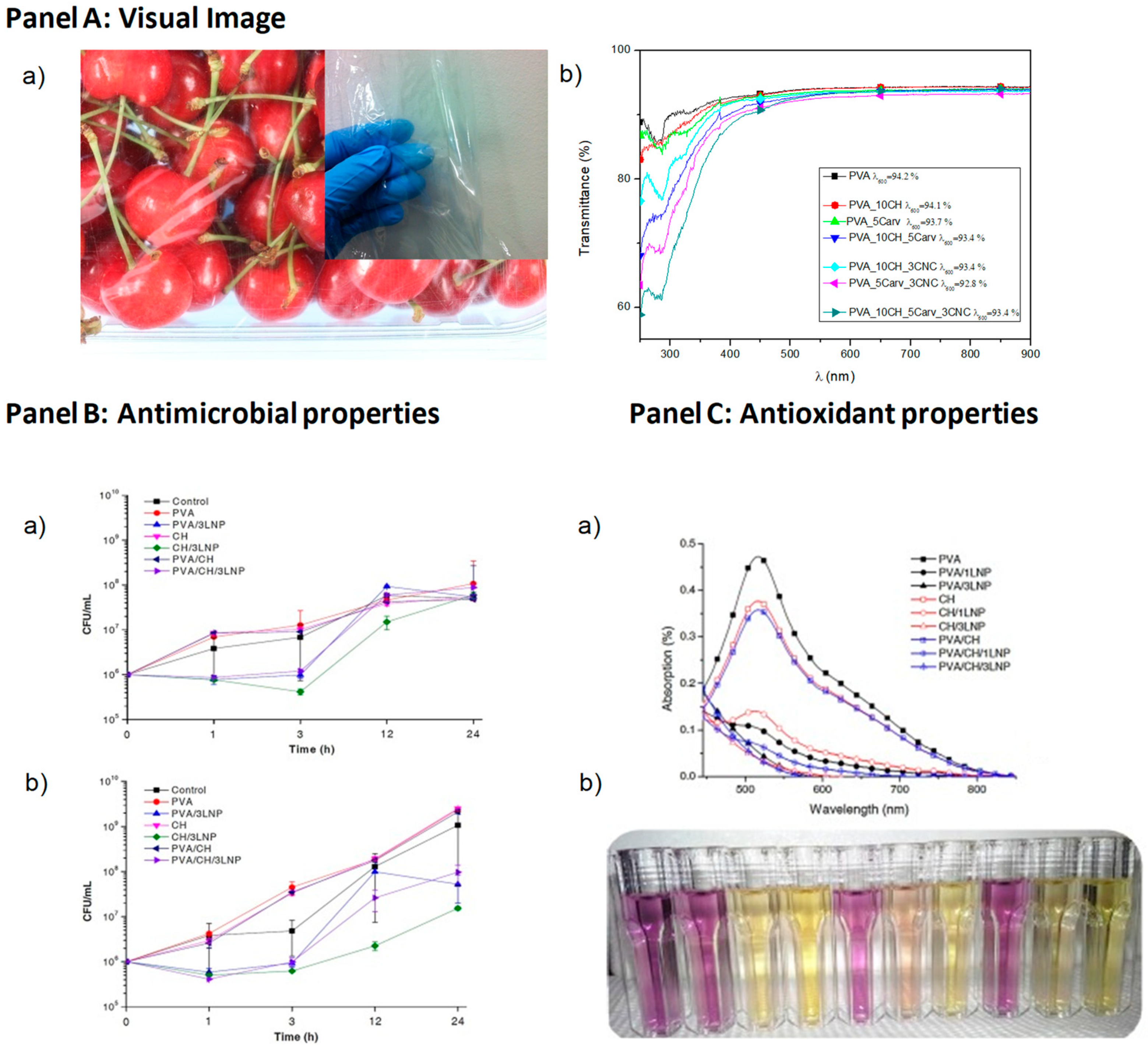
- Luzi, F.; Fortunati, E.; Giovanale, G.; Mazzaglia, A.; Torre, L.; Balestra, G.M. Cellulose nanocrystals from Actinidia deliciosa pruning residues combined with carvacrol in PVA_CH films with antioxidant/antimicrobial properties for packaging applications. Int. J. Biol. Macromol. 2017, 104, 43–55. [Google Scholar] [CrossRef] [PubMed]
- Sadeghi, K.; Shahedi, M. Physical, mechanical, and antimicrobial properties of ethylene vinyl alcohol copolymer/chitosan/nano-ZnO (ECNZn) nanocomposite films incorporating glycerol plasticizer. J. Food Meas. Charact. 2016, 10, 137–147. [Google Scholar] [CrossRef]
- Nuñez, K.; Rosales, C.; Perera, R.; Villarreal, N.; Pastor, J.M. Poly(lactic acid)/low-density polyethylene blends and its nanocomposites based on sepiolite. Polym. Eng. Sci. 2011, 52, 988–1004. [Google Scholar] [CrossRef]
- As’habi, L.; Jafari, S.H.; Khonakdar, H.A.; Boldt, R.; Wagenknecht, U.; Heinrich, G. Tuning the processability, morphology and biodegradability of clay incorporated PLA/LLDPE blends via selective localization of nanoclay induced by melt mixing sequence. Express Polym. Lett. 2013, 7, 21–39. [Google Scholar] [CrossRef]
- Weber, C.J.; Haugaard, V.; Festersen, R.; Bertelsen, G. Production and applications of biobased packaging materials for the food industry. Food Addit. Contam. 2002, 19, 172–177. [Google Scholar] [CrossRef] [PubMed]
- Wg1 Brochure: Moving From Oil-Based To Biobased Packaging—Key Material Aspects For Consideration Cellulose. Chem. Technol. 2015, 49, 709–713.
- Enabling Biobased Nylons, Polyurethanes, Plasticizers, and Other Sustainable Materials. Available online: http://www.rennovia.com/markets/ (accessed on 10 December 2018).
- Bio-Based Polymers—Production Capacity Will Triple from 3.5 Million Tonnes in 2011 to nearly 12 Million Tonnes in 2020. Available online: http://www.bio-based.eu/market_study/media/13-03-06PRMSBiopolymerslongnova.pdf (accessed on 10 December 2018).
- Tullo, A.H. The cost of plastic packaging. Chem. Eng. News 2016, 94, 32–37. [Google Scholar]

| Polymer | Packaging Types | Thermal | Mechanical | Permeability | |||||
|---|---|---|---|---|---|---|---|---|---|
| Tg (°C) | Tm (°C) | T (MPa) | εB (%) | OP | H2O: WVTR (g/m2/day) | O2 (g/m2/day) | CO2 (ml μm m−2 day−1 atm−1) | ||
| PET | Bottles, microwaveable and ovenable trays, boil in-the-bag products | 70–87 | 243–268 | 48–72 | 20–300 | ++ | 15–20 | 100–150 | 300–600 |
| HDPE | Jars and other rigid containers, pallets, films, or layers for dry food | −125 to −90 | 135 | 22–31 | 100 ≥ 1000 | +++ | 7–10 | 1600–2000 | 12,000–14,000 |
| PVC | Wrapping films, bottles, trays, containers | 60–100 | n.d. | 40–51 | 40–75 | ++ | 0.5–1.0 | 2–4 | 400–10,000 |
| LDPE | Films (wrapping, carrier bags, pouches), bottles | −125 to −100 | 112–135 | 8–31 | 200–900 | ++ | 10–20 | 6500–8500 | 20,000–40,000 |
| PP | Cups and containers for frozen and microwaveable food, lids, thin-walled containers (yoghurt) | −10 | 167–177 | 31–41 | 100–600 | + | 10–12 | 3500–4500 | 10,000–14,000 |
| PS | Disposable cups, plates and trays, boxes (egg cartons), rigid containers (yoghurt) | 100 | n.d | 35–51 | 1–4 | ++ | 4500–6000 | 14,000–30,000 | |
| PA | flexible packaging of perishable food, such as cheese and meat | 50–60 | 220 | 40–52 | 5–10 | ++ | 300–400 | 50–75 | n.d |
| PVA | Films for moisture barrier, confectionery products | 70–75 | 215–220 | 25–30 | 220–250 | ++ | n.d. | n.d. | n.d |
| EVOH | Thin films for dry/fatty food, multilayer | 60–65 | 180–150 | 45–110 | 180–250 | +++ | 1000 | 0.5 | n.d |
| Bioplastic | Main Food Applications |
|---|---|
| Starch-based polymers | Substitute for polystyrene (PS). Used in food packaging, disposable tableware and cutlery, coffee machine capsules, bottles. |
| Cellulose-based polymers | Low water vapor barrier, poor mechanical properties, bad processability, brittleness (pure cellulosic polymer), Regulated under 2007/42/EC. Coated, compostable cellulose films. Used in the packaging of bread, fruits, meat, dried products, etc. |
| Polylactide (PLA) | Possible alternative of low- and high-density polyethylene (LDPE and HDPE), polystyrene (PS), and poly terephthalate (PET). Transparent, rigid containers, bags, jars, films. |
| Polyhydroxyalkanoates (PHA) | Family of many, chemically different polymers Brittleness, stiffness, thermal instability. |
| Bio-based polypropylene (PP) and polyethylene (PE) | Mainly based on sugar cane. Identical physicochemical properties. |
| Partially bio-based (PET) | Alternative to conventional PET. Up to 30% bio-based raw materials. Used in bottles. |
| Bio-based polyethylene furanoate (PEF) | Better barrier function than PET. Up to 100% bio-based raw materials. May be used in the future in bottles, fibers, films. |
| Aliphatic (co)polyesters | Includes polybutylene succinate (PBS), polyethylene succinate (PES), and polyethylene adipate (PEA). Used in disposable cutlery. |
| Aliphatic-aromatic (co)polyesters | Includes polybutylene adipate terephthalate (PBAT), polybutylene, and succinate terephthalate (PBST). Used as fast food disposable packaging, PBAT for plastic films. |
| Polycaprolactone (PCL) | Biodegradable polyester. Low melting temperature, easily biodegradable. Used in medical applications. |
| Polyvinyl alcohol (PVOH) | Used for coatings, adhesives, and as additive in paper and board production. |
| Polysaccharide | Properties | Main Food Applications |
|---|---|---|
Starch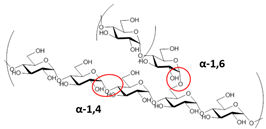 | Biodegradable Transparent Odorless and tasteless Retrogradation high elongation and tensile strength | Flexible packaging:
|
Cellulose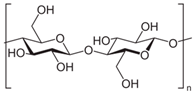 | Biodegradable Good mechanical properties Transparent Highly sensitive to water Resistance to fats and oils Need to perform modification, use of plasticizer, or polymer blend | Cellophane membranes. |
Chitin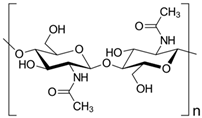 | Biodegradable Antibacterial and fungistatic properties Biocompatible and non-toxic Highly transparent | Coffee capsules Food bags Packaging films |
Chitosan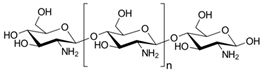 | Biodegradable Biocompatible and non-toxic Antifungal and antibacterial properties Good mechanical properties Barrier to gases High water vapor permeability Brittle—need to use plasticizer | Edible membranes and coatings (strawberries, cherries, mango, guava, among others) Packaging membranes for vegetables and fruit |
| Packed Product | Barrier Requirements | Classic Packaging Solution | Bio-Based Packaging Solution | Technology Readiness Level |
|---|---|---|---|---|
| Meat/fish | High barrier against oxygen and gas (aroma); | Trays (PS, PP, PVC with EVOH + LDPE or PVC as coating) + foil (PVC) or lid, bags, for short term storage; waxed paper (wrapping), paperboard external packaging; transparent films (PP, PE) | Multilayer packaging materials, functional bio-based coating (modified starches) + antimicrobial and anti-fogging systems | On the market (as pilot packaging on selected markets); still more expensive than conventional solutions |
| Fresh cheese | High barrier properties; grease, water, O2, CO2 and N2, aroma and light. MAP (80% N2, 20% CO2) | Transparent films/foils; bags (e.g., LDPE/ EVA /PVdC /EVA), trays, wrapping films (PE, laminated), plastic cups (HDPE, PP, PS) + high barrier lid (PA/LDPE) | Eco-paper for short term storage (wrapping); PHA/modified PLA films | On the market, still more expensive than conventional plastics |
| Dairy products/liq uids | High barrier properties; water vapor (scavenging moisture), O2, light high/moderate for grease and aroma | Waxed paper, LDPE, PVC, or aluminum-coated/laminated paper or paperboard, plastic films (BOPP), metal cans | Paper/paperboard coated with bio-based materials | Close to market |
| Salad (flexible packaging) | High oxygen barrier, water resistant | Transparent laminated PP films | PLA films (perforated) Coated paper with bio-based films + transparent window | On the market, still more expensive than conventional plastics |
| Fruits/vegetables | Medium barrier properties (water vapor) | Perforated PP, OPP, LDPE; PVC films/bags, trays, pouches, overwraps; PS/PP trays | Molding pulp—trays PLA films (perforated) Edible coatings (polysaccharides: xanthan gum, starch, cellulose, HPC, MC, CMC, proteins: chitosan, corn zein, wheat gluten) + low barrier packaging films | On the market (molded pulp trays); on the market (PLA as pilot packaging in selected markets, e.g., for tomatoes); still more expensive than conventional solutions |
| Take-away food | Grease, thermal insulation | Polystyrene foam trays | Paperboard with grease barrier coating on the inside | On the market |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luzi, F.; Torre, L.; Kenny, J.M.; Puglia, D. Bio- and Fossil-Based Polymeric Blends and Nanocomposites for Packaging: Structure–Property Relationship. Materials 2019, 12, 471. https://doi.org/10.3390/ma12030471
Luzi F, Torre L, Kenny JM, Puglia D. Bio- and Fossil-Based Polymeric Blends and Nanocomposites for Packaging: Structure–Property Relationship. Materials. 2019; 12(3):471. https://doi.org/10.3390/ma12030471
Chicago/Turabian StyleLuzi, Francesca, Luigi Torre, José Maria Kenny, and Debora Puglia. 2019. "Bio- and Fossil-Based Polymeric Blends and Nanocomposites for Packaging: Structure–Property Relationship" Materials 12, no. 3: 471. https://doi.org/10.3390/ma12030471
APA StyleLuzi, F., Torre, L., Kenny, J. M., & Puglia, D. (2019). Bio- and Fossil-Based Polymeric Blends and Nanocomposites for Packaging: Structure–Property Relationship. Materials, 12(3), 471. https://doi.org/10.3390/ma12030471








