Bimetallic Nanoparticles as a Model System for an Industrial NiMo Catalyst
Abstract
1. Introduction
2. Materials and Methods
2.1. Spark-Discharge-Generated NiMo Nanoparticles
2.2. Production of Industrial Catalysts
3. Catalyst Characterization
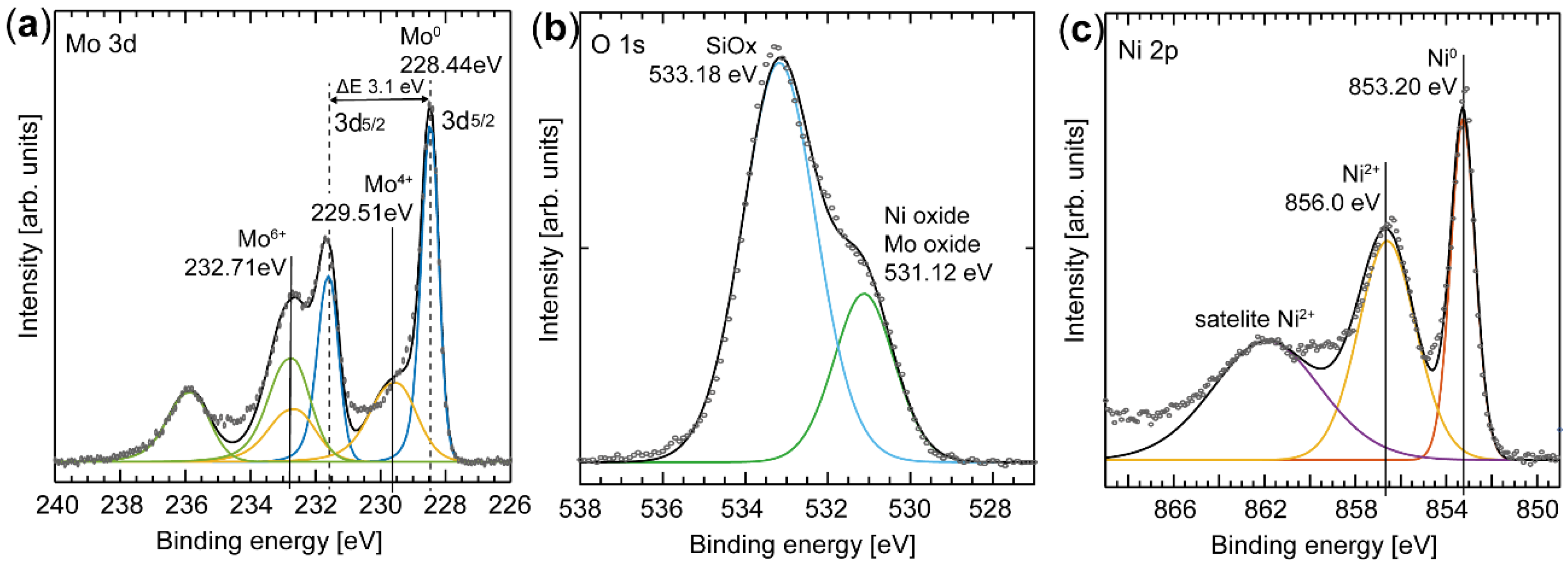
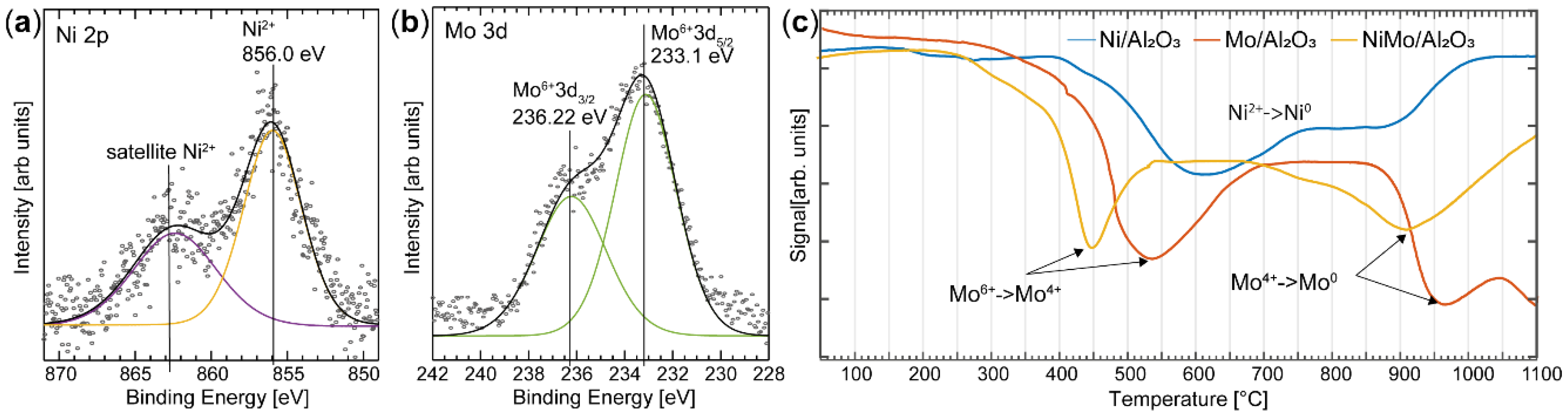
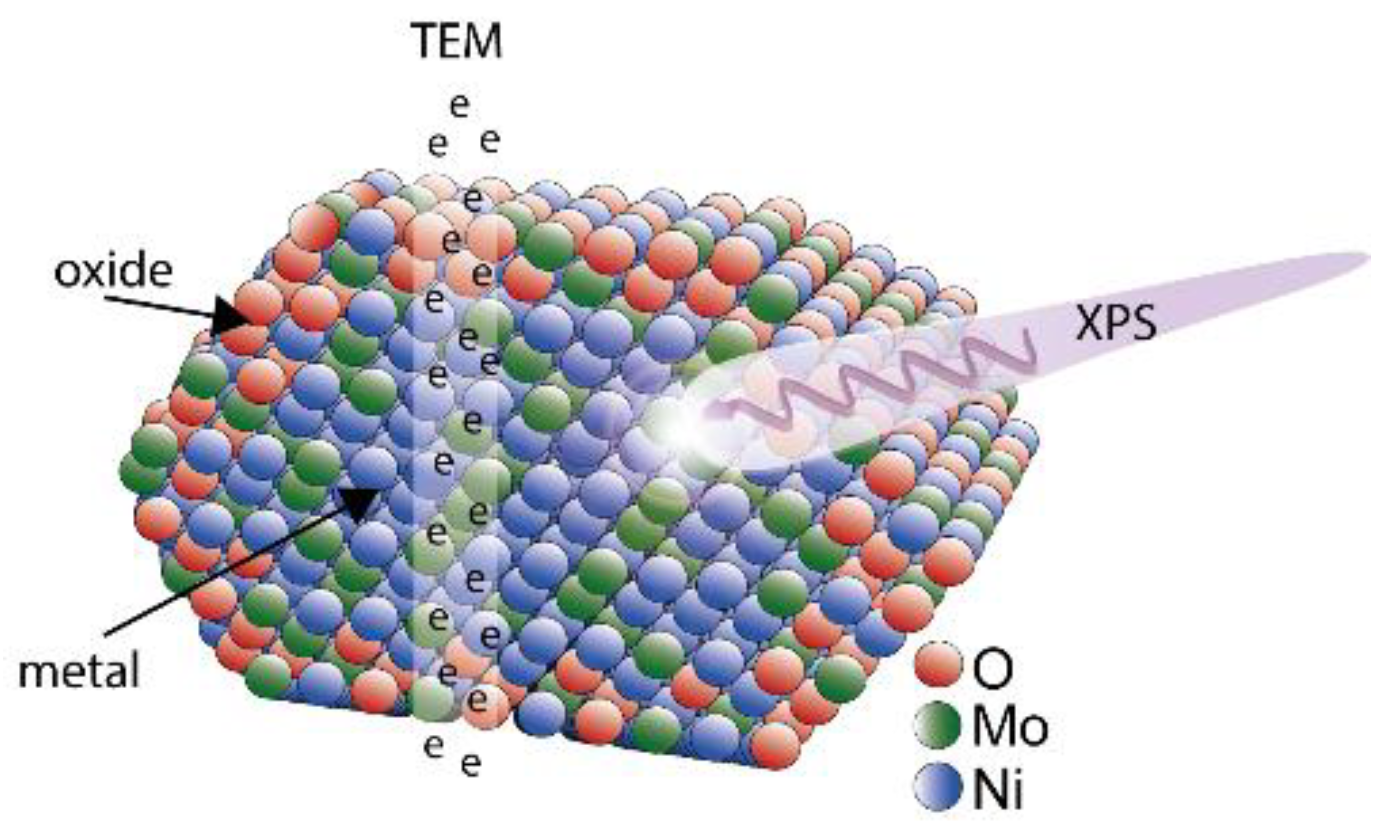
3.1. X-Ray Photoelectron Spectroscopy
3.2. Transmission Electron Spectroscopy
3.3. Scanning Electron Spectroscopy
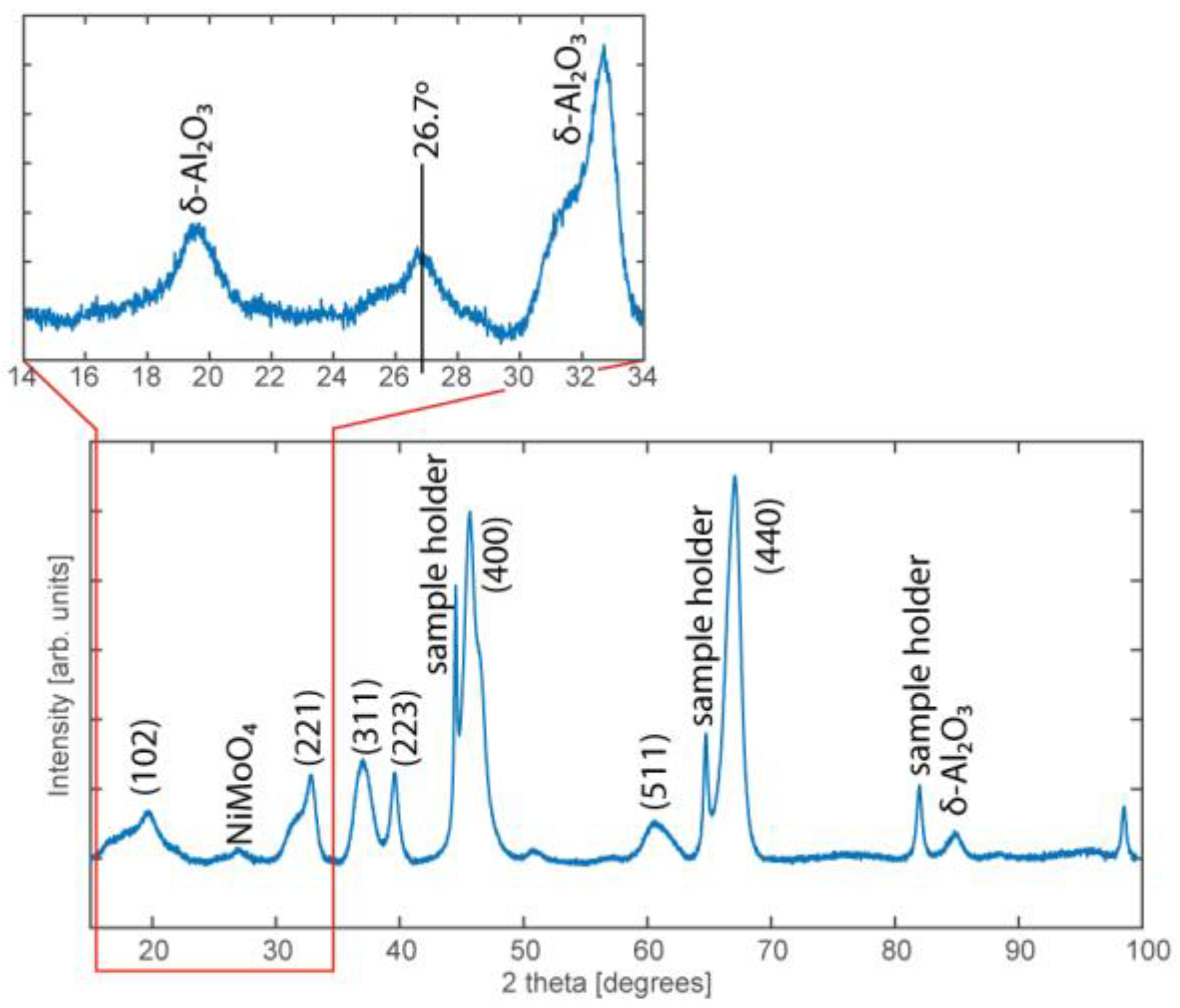
3.4. Powder X-Ray Diffraction
3.5. H2-Temperature Programmed Reduction and Nitrogen Physisorption
4. Results
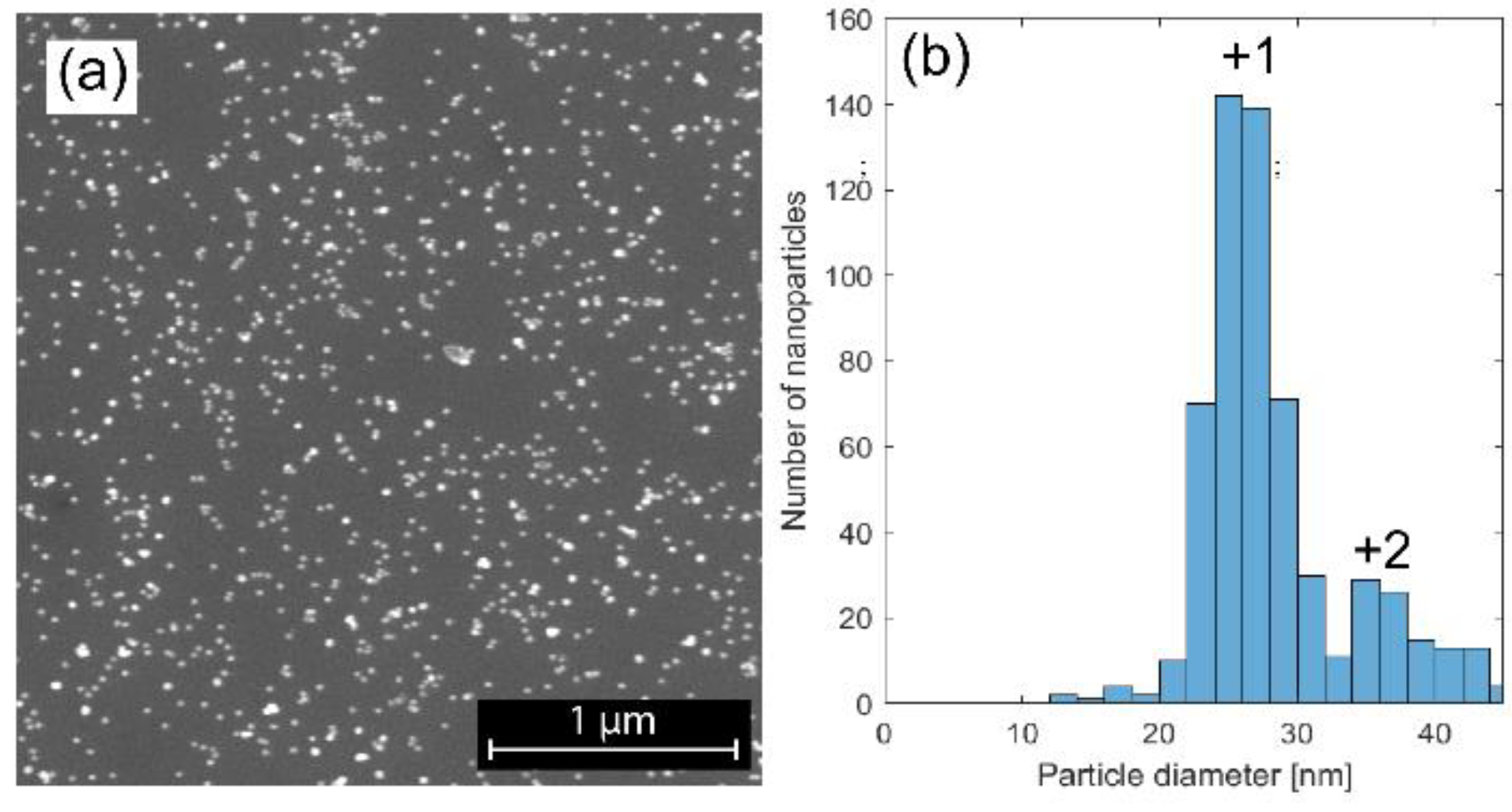
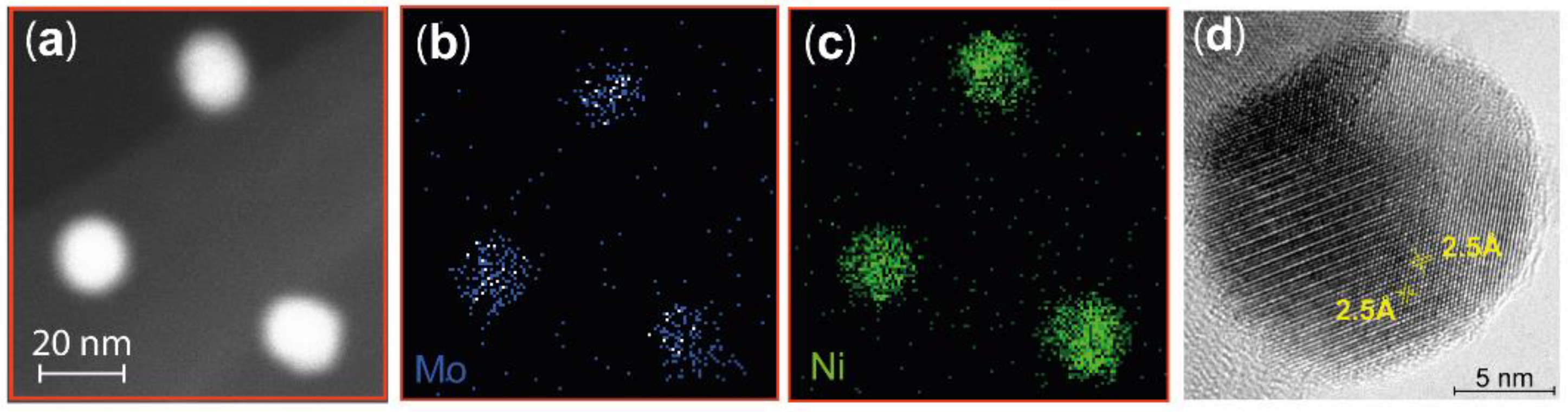
4.1. Characterization of NiMo Nanoparticles
4.2. Characterization of the Industrial NiMo/δ-Al2O3 Catalyst
5. Discussion
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gnansounou, E.; Pandey, A. Life-Cycle Assessment of Biorefineries; Elsevier: Amsterdam, The Netherlands, 2017; p. 312. [Google Scholar]
- Abhilash, P.; Thomas, D. Biopolymer Composites in Electronics-Biopolymers for Biocomposites and Chemical Sensor Applications; Elsevier: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Mortensen, P.M.; Grunwaldt, J.D.; Jensen, P.A.; Knudsen, K.G.; Jensen, A.D. A review of catalytic upgrading of bio-oil to engine fuels. Appl. Catal. Gen. 2011, 407, 1–19. [Google Scholar] [CrossRef]
- Abdelaziz, O.Y.; Meier, S.; Prothmann, J.; Turner, C.; Riisager, A.; Hulteberg, C.P. Oxidative Depolymerisation of Lignosulphonate Lignin into Low-Molecular-Weight Products with Cu–Mn/δ-Al2O3. Top. Catal. 2019, 62, 639–648. [Google Scholar] [CrossRef]
- Dupont, C.; Lemeur, R.; Daudin, A.; Raybaud, P. Hydrodeoxygenation pathways catalyzed by MoS2 and NiMoS active phases: A DFT study. J. Catal. 2011, 279, 276–286. [Google Scholar] [CrossRef]
- Hocevar, B.; Grilc, M.; Hus, M.; Likozar, B. Mechanism, ab initio calculations and microkinetics of hydrogenation, hydrodeoxygenation, double bond migration and cis-trans isomerisation during hydrotreatment of C-6 secondary alcohol species and ketones. Appl. Catal. B Environ. 2017, 218, 147–162. [Google Scholar] [CrossRef]
- Topsoe, H.; Clausen, B.S. Importance of Co-Mo-S Type Structures in Hydrodesulfurization. Catal. Rev. 1984, 26, 395–420. [Google Scholar] [CrossRef]
- Lauritsen, J.V.; Helveg, S.; Laegsgaard, E.; Stensgaard, I.; Clausen, B.S.; Topsoe, H.; Besenbacher, E. Atomic-scale structure of Co-Mo-S nanoclusters in hydrotreating catalysts. J. Catal. 2001, 197, 1–5. [Google Scholar] [CrossRef]
- Gronborg, S.S.; Salazar, N.; Bruix, A.; Rodriguez-Fernandez, J.; Thomsen, S.D.; Hammer, B.; Lauritsen, J.V. Visualizing hydrogen-induced reshaping and edge activation in MoS2 and Co-promoted MoS2 catalyst clusters. Nat. Commun. 2018, 9, 2211. [Google Scholar] [CrossRef] [PubMed]
- Dahl-Petersen, C.; Saric, M.; Brorson, M.; Moses, P.G.; Rossmeisl, J.; Lauritsen, J.V.; Helveg, S. Topotactic Growth of Edge-Terminated MoS2 from MoO2 Nanocrystals. ACS Nano 2018, 12, 5351–5358. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Yin, C.L.; Li, X.H.; Chai, Y.M.; Li, Y.P.; Liu, C.G. Effect of NiMo phases on the hydrodesulfurization activities of dibenzothiophene. Catal. Today 2017, 282, 222–229. [Google Scholar] [CrossRef]
- Besenbacher, F.; Brorson, M.; Clausen, B.S.; Helveg, S.; Hinnemann, B.; Kibsgaard, J.; Lauritsen, J.; Moses, P.G.; Norskov, J.K.; Topsoe, H. Recent STM, DFT and HAADF-STEM studies of sulfide-based hydrotreating catalysts: Insight into mechanistic, structural and particle size effects. Catal. Today 2008, 130, 86–96. [Google Scholar] [CrossRef]
- Topsoe, H.; Clausen, B.S.; Candia, R.; Wivel, C.; Morup, S. Insitu Mossbauer Emission-Spectroscopy Studies of Unsupported and Supported Sulfided Co-Mo Hydrodesulfurization Catalysts—Evidence for and Nature of a Co-Mo-S Phase. J. Catal. 1981, 68, 433–452. [Google Scholar] [CrossRef]
- Bruix, A.; Lauritsen, J.V.; Hammer, B. Effects of particle size and edge structure on the electronic structure, spectroscopic features, and chemical properties of Au(111)-supported MoS2 nanoparticles. Faraday Discuss. 2016, 188, 323–343. [Google Scholar] [CrossRef] [PubMed]
- Balmes, O.; Resta, A.; Wermeille, D.; Felici, R.; Messing, M.E.; Deppert, K.; Liu, Z.; Grass, M.E.; Bluhm, H.; van Rijn, R.; et al. Reversible formation of a PdCx phase in Pd nanoparticles upon CO and O-2 exposure. Phys. Chem. Chem. Phys. 2012, 14, 4796–4801. [Google Scholar] [CrossRef] [PubMed]
- Blomberg, S.; Gustafson, J.; Martin, N.M.; Messing, M.E.; Deppert, K.; Liu, Z.; Chang, R.; Fernandes, V.R.; Borg, A.; Gronbeck, H.; et al. Generation and oxidation of aerosol deposited PdAg nanoparticles. Surf. Sci. 2013, 616, 186–191. [Google Scholar] [CrossRef]
- Westerstrom, R.; Messing, M.E.; Blomberg, S.; Hellman, A.; Gronbeck, H.; Gustafson, J.; Martin, N.M.; Balmes, O.; van Rijn, R.; Andersen, J.N.; et al. Oxidation and reduction of Pd(100) and aerosol-deposited Pd nanoparticles. Phys. Rev. B 2011, 83, 115440. [Google Scholar] [CrossRef]
- Messing, M.E.; Dick, K.A.; Wallenberg, L.R.; Deppert, K. Generation of size-selected gold nanoparticles by spark discharge—For growth of epitaxial nanowires. Gold Bull. 2009, 42, 20–26. [Google Scholar] [CrossRef]
- Hallberg, R.T.; Ludvigsson, L.; Preger, C.; Meuller, B.O.; Dick, K.A.; Messing, M.E. Hydrogen-assisted spark discharge generated metal nanoparticles to prevent oxide formation. Aerosol Sci. Technol. 2018, 52, 347–358. [Google Scholar] [CrossRef]
- Preger, C.; Bulbucan, C.; Meuller, B.O.; Ludvigsson, L.; Kostanyan, A.; Muntwiler, M.; Deppert, K.; Westerstrom, R.; Messing, M.E. Controlled Oxidation and Self-Passivation of Bimetallic Magnetic FeCr and FeMn Aerosol Nanoparticles. J. Phys. Chem. C 2019, 123, 16083–16090. [Google Scholar] [CrossRef]
- Urpelainen, S.; Sathe, C.; Grizolli, W.; Agaker, M.; Head, A.R.; Andersson, M.; Huang, S.W.; Jensen, B.N.; Wallen, E.; Tarawneh, H.; et al. The SPECIES beamline at the MAX IV Laboratory: A facility for soft X-ray RIXS and APXPS. J. Synchrotron Radiat. 2017, 24, 344–353. [Google Scholar] [CrossRef] [PubMed]
- Schnadt, J.; Knudsen, J.; Andersen, J.N.; Siegbahn, H.; Pietzsch, A.; Hennies, F.; Johansson, N.; Martensson, N.; Ohrwall, G.; Bahr, S.; et al. The new ambient-pressure X-ray photoelectron spectroscopy instrument at MAX-lab. J. Synchrotron Radiat. 2012, 19, 701–704. [Google Scholar] [CrossRef] [PubMed]
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of Gases in Multimolecular Layers. J. Am. Chem. Soc. 1938, 60, 309–319. [Google Scholar] [CrossRef]
- Barrett, E.P.; Joyner, L.G.; Halenda, P.P. The Determination of Pore Volume and Area Distributions in Porous Substances. I. Computations from Nitrogen Isotherms. J. Am. Chem. Soc. 1951, 73, 373–380. [Google Scholar] [CrossRef]
- Baltrusaitis, J.; Mendoza-Sanchez, B.; Fernandez, V.; Veenstra, R.; Dukstiene, N.; Roberts, A.; Fairley, N. Generalized molybdenum oxide surface chemical state XPS determination via informed amorphous sample model. Appl. Surf. Sci. 2015, 326, 151–161. [Google Scholar] [CrossRef]
- Song, Z.; Cai, T.H.; Chang, Z.P.; Liu, G.; Rodriguez, J.A.; Hrbek, J. Molecular level study of the formation and the spread of MoO3 on Au (111) by scanning tunneling microscopy and X-ray photoelectron spectroscopy. J. Am. Chem. Soc. 2003, 125, 8059–8066. [Google Scholar] [CrossRef] [PubMed]
- Swartz, W.E.; Hercules, D.M. X-ray Photoelectron Spectroscopy of Molybdenum Compounds—Use of Esca in Quantitative Analysis. Anal. Chem. 1971, 43, 1774–1779. [Google Scholar] [CrossRef]
- Guimond, S.; Gobke, D.; Sturm, J.M.; Romanyshyn, Y.; Kuhlenbeck, H.; Cavalleri, M.; Freund, H.J. Well-Ordered Molybdenum Oxide Layers on Au(111): Preparation and Properties. J. Phys. Chem. C 2013, 117, 8746–8757. [Google Scholar] [CrossRef]
- Salazar, N.; Beinik, I.; Lauritsen, J.V. Single-layer MoS2 formation by sulfidation of molybdenum oxides in different oxidation states on Au(111). Phys. Chem. Chem. Phys. 2017, 19, 14020–14029. [Google Scholar] [CrossRef] [PubMed]
- Scott, C.E.; Perez-Zurita, M.J.; Carbognani, L.A.; Molero, H.; Vitale, G.; Guzman, H.J.; Pereira-Almao, P. Preparation of NiMoS nanoparticles for hydrotreating. Catal. Today 2015, 250, 21–27. [Google Scholar] [CrossRef]
- Zhang, M.H.; Fan, J.Y.; Chi, K.; Duan, A.J.; Zhao, Z.; Meng, X.L.; Zhang, H.L. Synthesis, characterization, and catalytic performance of NiMo catalysts supported on different crystal alumina materials in the hydrodesulfurization of diesel. Fuel. Process. Technol. 2017, 156, 446–453. [Google Scholar] [CrossRef]
- Zou, J.; Schrader, G.L. Multicomponent thin film molybdate catalysts for the selective oxidation of 1,3-butadiene. J. Catal. 1996, 161, 667–686. [Google Scholar] [CrossRef]
- Grosvenor, A.P.; Biesinger, M.C.; Smart, R.S.; McIntyre, N.S. New interpretations of XPS spectra of nickel metal and oxides. Surf. Sci. 2006, 600, 1771–1779. [Google Scholar] [CrossRef]
- Wang, Y.G.; Xiong, G.; Liu, X.; Yu, X.C.; Liu, L.P.; Wang, J.Y.; Feng, Z.C.; Li, C. Structure and Reducibility of NiO-MoO3/gamma-Al2O3 Catalysts: Effects of Loading and Molar Ratio. J. Phys. Chem. C 2008, 112, 17265–17271. [Google Scholar] [CrossRef]
- Hong, S.T.; Park, D.R.; Yoo, S.J.; Kim, J.D.; Park, H.S. Characterization of the active phase of NiMo/Al2O3 hydrodesulfurization catalysts. Res. Chem. Intermed. 2006, 32, 857–870. [Google Scholar] [CrossRef]
- Finster, J.; Schulze, D.; Meisel, A. Characterization of Amorphous Siox Layers with Esca. Surf. Sci. 1985, 162, 671–679. [Google Scholar] [CrossRef]
- Biesinger, M.C.; Payne, B.P.; Lau, L.W.M.; Gerson, A.; Smart, R.S.C. X-ray photoelectron spectroscopic chemical state quantification of mixed nickel metal, oxide and hydroxide systems. Surf. Interface Anal. 2009, 41, 324–332. [Google Scholar] [CrossRef]
- Shi, B.Y.; Xu, A.J.; Wang, J. The impact of preparation methods on the structure and catalytic performance of NiMoO4 for oxidative dehydrogenation of propane. Integr. Ferroelectr. 2016, 171, 16–22. [Google Scholar] [CrossRef]
- Merte, L.R.; Gustafson, J.; Shipilin, M.; Zhang, C.; Lundgren, E. Redox behavior of iron at the surface of an O(100) single crystal studied by ambient-pressure photoelectron spectroscopy. Catal. Struct. React. 2017, 3, 95–103. [Google Scholar] [CrossRef]
- Plyuto, Y.V.; Babich, I.V.; Plyuto, I.V.; VanLangeveld, A.D.; Moulijn, J.A. XPS studies of MoO3/Al2O3 and MoO3/SiO2 systems. Appl. Surf. Sci. 1997, 119, 11–18. [Google Scholar] [CrossRef]
- Massoth, F.E. Characterization of Molybdena Catalysts. In Advances in Catalysis; Academic Press: Cambridge, MA, USA, 1974; Volume 27, pp. 265–310. [Google Scholar]
- Ojagh, H.; Creaser, D.; Tamm, S.; Hu, C.Q.; Olsson, L. Effect of Thermal Treatment on Hydrogen Uptake and Characteristics of Ni-, Co-, and Mo-Containing Catalysts. Ind. Eng. Chem. Res. 2015, 54, 11511–11524. [Google Scholar] [CrossRef]
- Liu, F.; Xu, S.P.; Cao, L.; Chi, Y.; Zhang, T.; Xue, D.F. A comparison of NiMo/Al2O3 catalysts prepared by impregnation and coprecipitation methods for hydrodesulfurization of dibenzothiophene. J. Phys. Chem. C 2007, 111, 7396–7402. [Google Scholar] [CrossRef]
- Qu, L.L.; Zhang, W.P.; Kooyman, P.J.; Prins, R. MAS NMR, TPR, and TEM studies of the interaction of NiMo with alumina and silica-alumina supports. J. Catal. 2003, 215, 7–13. [Google Scholar] [CrossRef]
- Jin, S.H.; Xiao, Z.H.; Li, C.; Chen, X.; Wang, L.; Xing, J.C.; Li, W.Z.; Liang, C.H. Catalytic hydrodeoxygenation of anisole as lignin model compound over supported nickel catalysts. Catal. Today 2014, 234, 125–132. [Google Scholar] [CrossRef]
- Jiang, S.J.; Zhou, Y.S.; Ding, S.J.; Wei, Q.; Zhou, W.W.; Shan, Y.C. Effect of direct synthesis Al-SBA-15 supports on the morphology and catalytic activity of the NiMoS phase in HDS of DBT. RSC Adv. 2016, 6, 106680–106689. [Google Scholar] [CrossRef]
- Brito, J.; Laine, J. Characterization of Supported MoO3 by Temperature-Programmed Reduction. Polyhedron 1986, 5, 179–182. [Google Scholar] [CrossRef]
- Kala, S.; Theissmann, R.; Kruis, F.E. Generation of AuGe nanocomposites by co-sparking technique and their photoluminescence properties. J. Nanopart. Res. 2013, 15, 1963. [Google Scholar] [CrossRef]

| Technique | Ni (Atomic %) | Mo (Atomic %) | Ni:Mo |
|---|---|---|---|
| STEM-XEDS | 66 | 34 | 1.9 |
| XPS | 72 | 28 | 2.6 |
| Technique | Ni (Atomic %) | Mo (Atomic %) | Ni:Mo |
|---|---|---|---|
| STEM-XEDS | 29 | 71 | 0.4 |
| XPS | 23 | 77 | 0.3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blomberg, S.; Johansson, N.; Kokkonen, E.; Rissler, J.; Kollberg, L.; Preger, C.; Franzén, S.M.; Messing, M.E.; Hulteberg, C. Bimetallic Nanoparticles as a Model System for an Industrial NiMo Catalyst. Materials 2019, 12, 3727. https://doi.org/10.3390/ma12223727
Blomberg S, Johansson N, Kokkonen E, Rissler J, Kollberg L, Preger C, Franzén SM, Messing ME, Hulteberg C. Bimetallic Nanoparticles as a Model System for an Industrial NiMo Catalyst. Materials. 2019; 12(22):3727. https://doi.org/10.3390/ma12223727
Chicago/Turabian StyleBlomberg, Sara, Niclas Johansson, Esko Kokkonen, Jenny Rissler, Linnéa Kollberg, Calle Preger, Sara M Franzén, Maria E Messing, and Christian Hulteberg. 2019. "Bimetallic Nanoparticles as a Model System for an Industrial NiMo Catalyst" Materials 12, no. 22: 3727. https://doi.org/10.3390/ma12223727
APA StyleBlomberg, S., Johansson, N., Kokkonen, E., Rissler, J., Kollberg, L., Preger, C., Franzén, S. M., Messing, M. E., & Hulteberg, C. (2019). Bimetallic Nanoparticles as a Model System for an Industrial NiMo Catalyst. Materials, 12(22), 3727. https://doi.org/10.3390/ma12223727






