Influence of Alloyed Ga on the Microstructure and Corrosion Properties of As-Cast Mg–5Sn Alloys
Abstract
1. Introduction
2. Materials and Methods
2.1. Specimen Preparation
2.2. Microstructural Characterization
2.3. Corrosion Tests
3. Results and Discussion
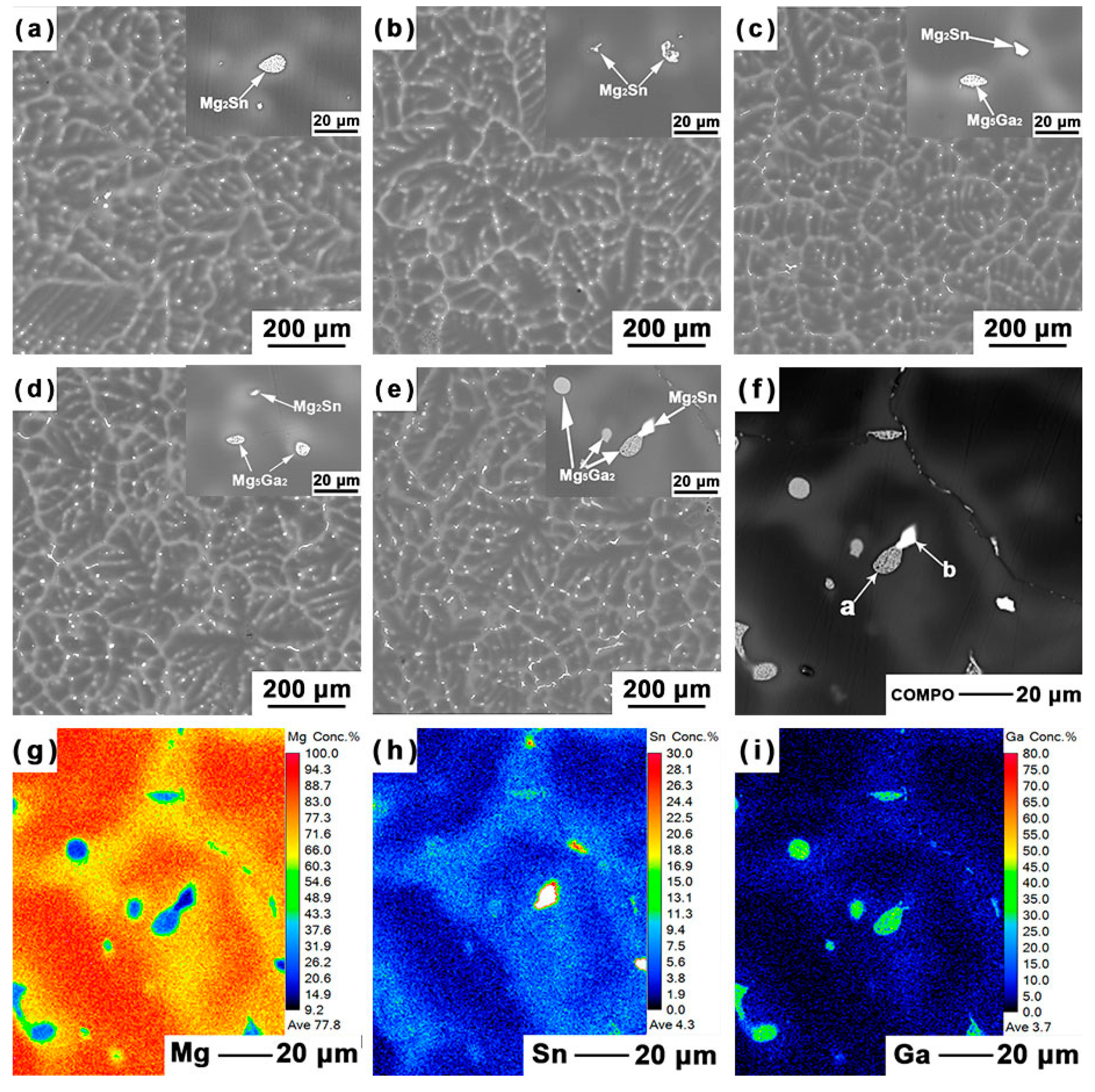
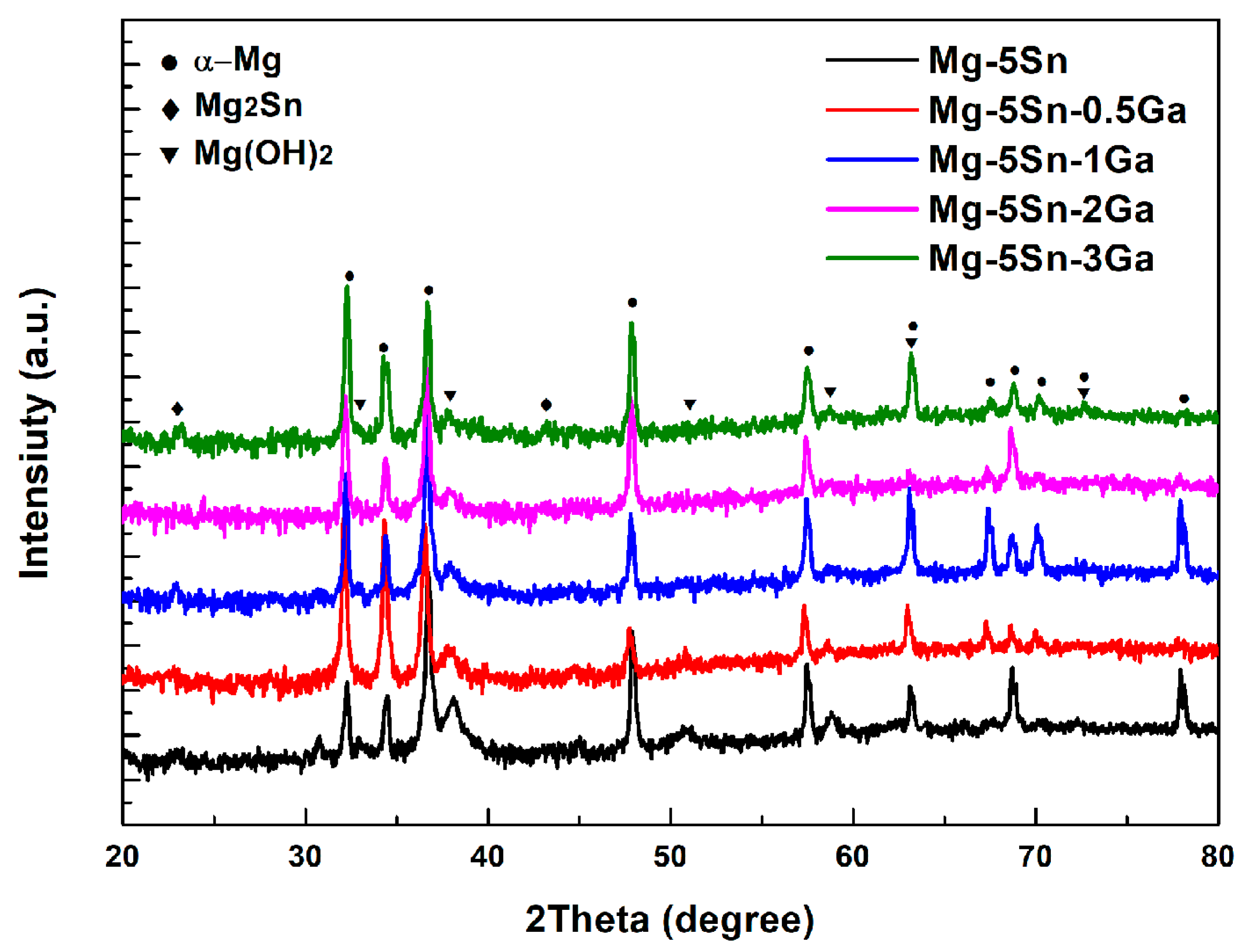
3.1. Microstructure Analysis
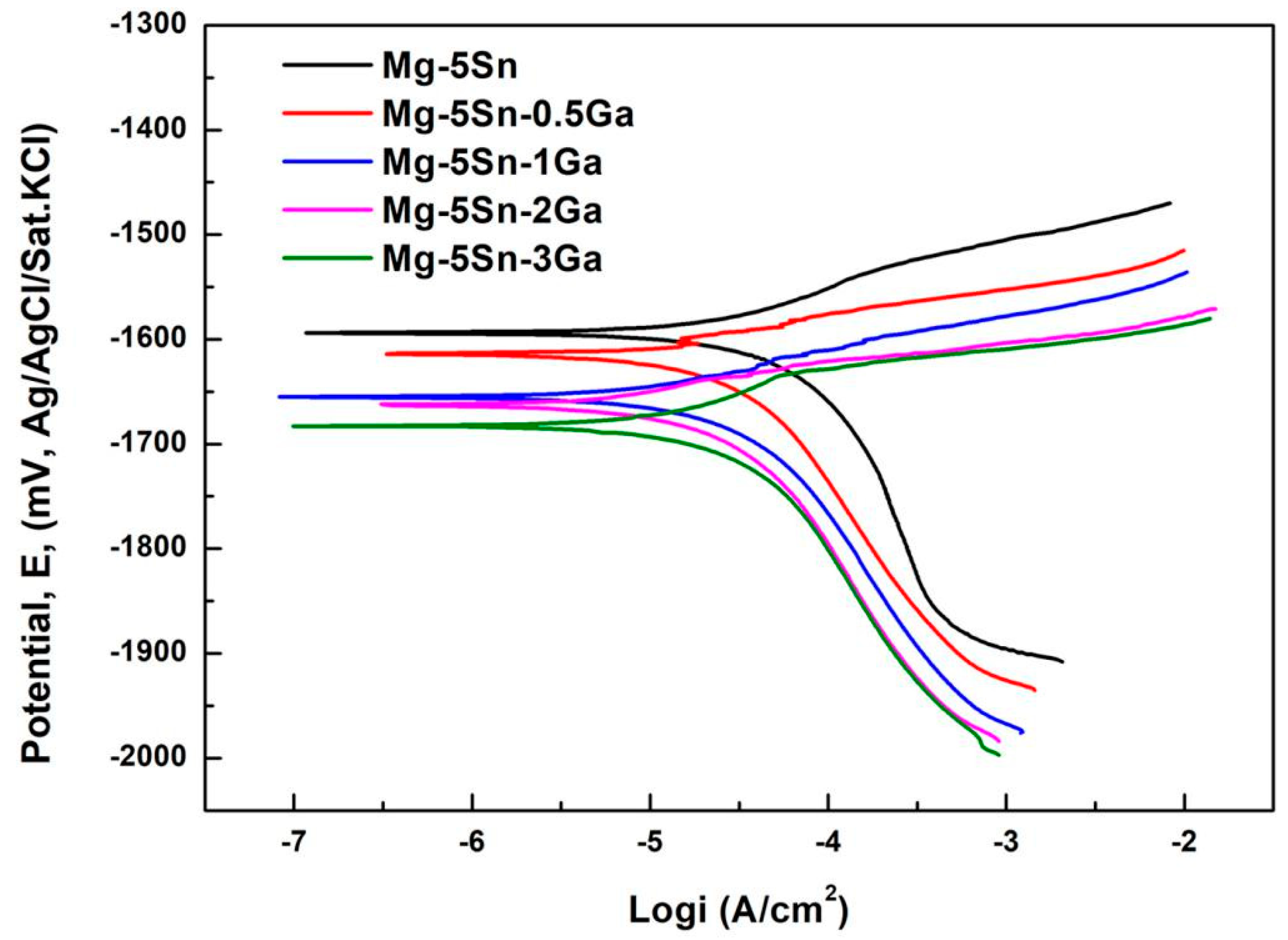
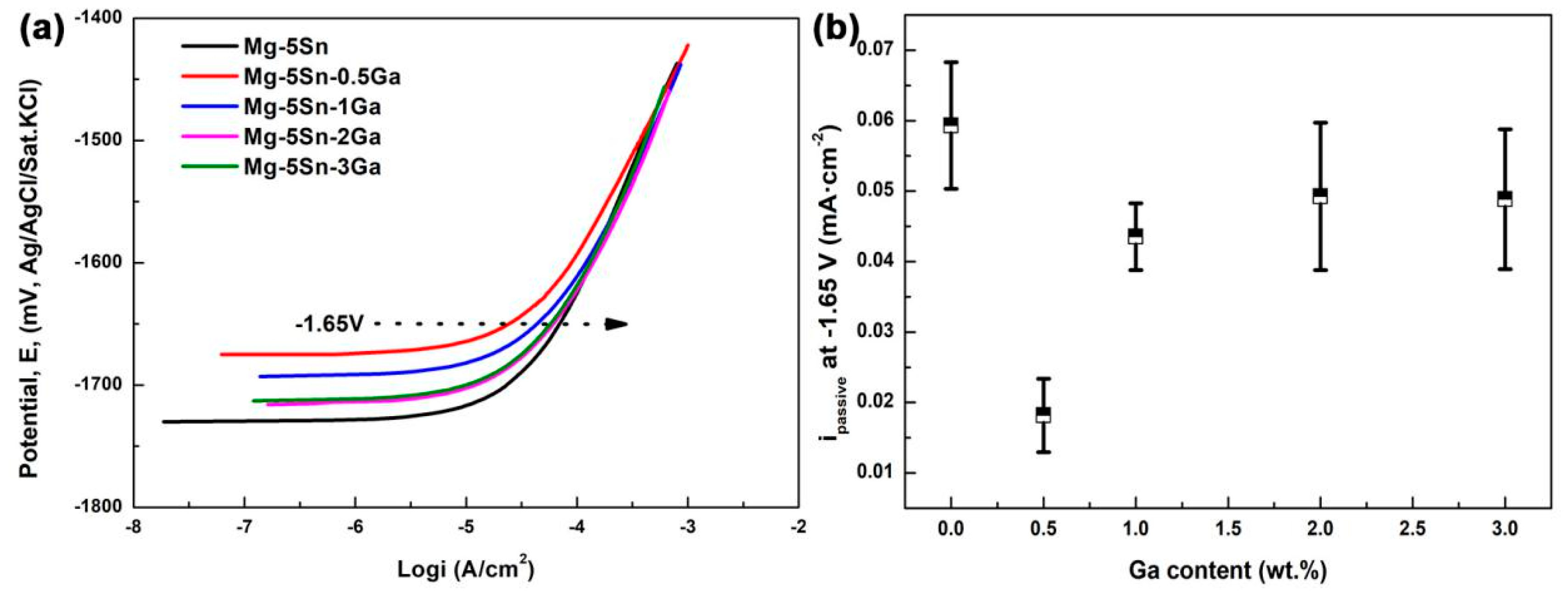
3.2. Polarization Tests
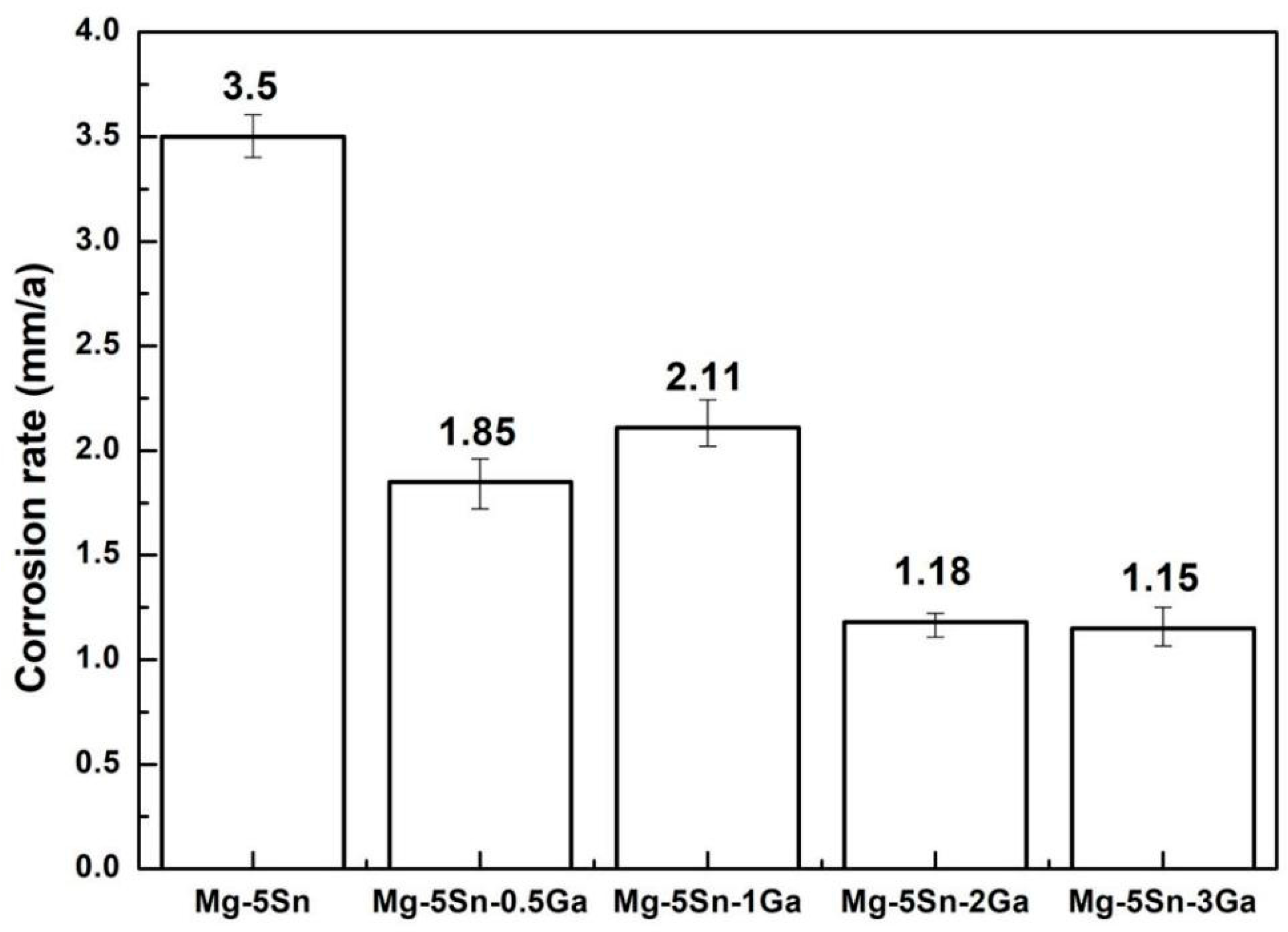
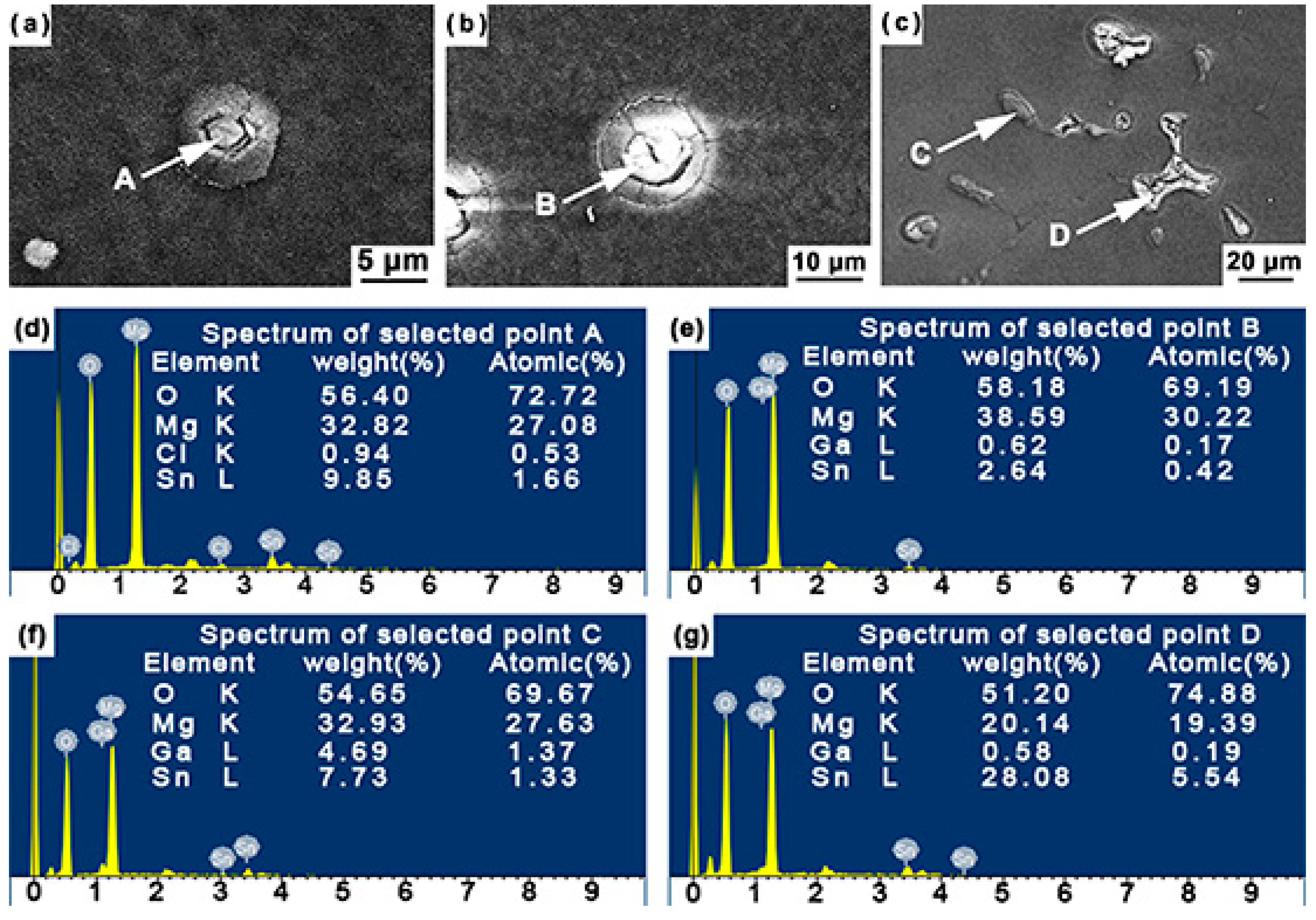
3.3. The Immersion Tests
4. Conclusions
- The microstructures of the Mg–5Sn–xGa alloys similarly present a typical dendritic morphology, regardless of the content of Ga. Ga refines the grain structure adequately. An average grain size of 155.9 ± 8.6 μm was obtained for the Mg–5Sn–3Ga alloy, recording a 48.3% reduction in grain size as compared to the Mg–5Sn alloy;
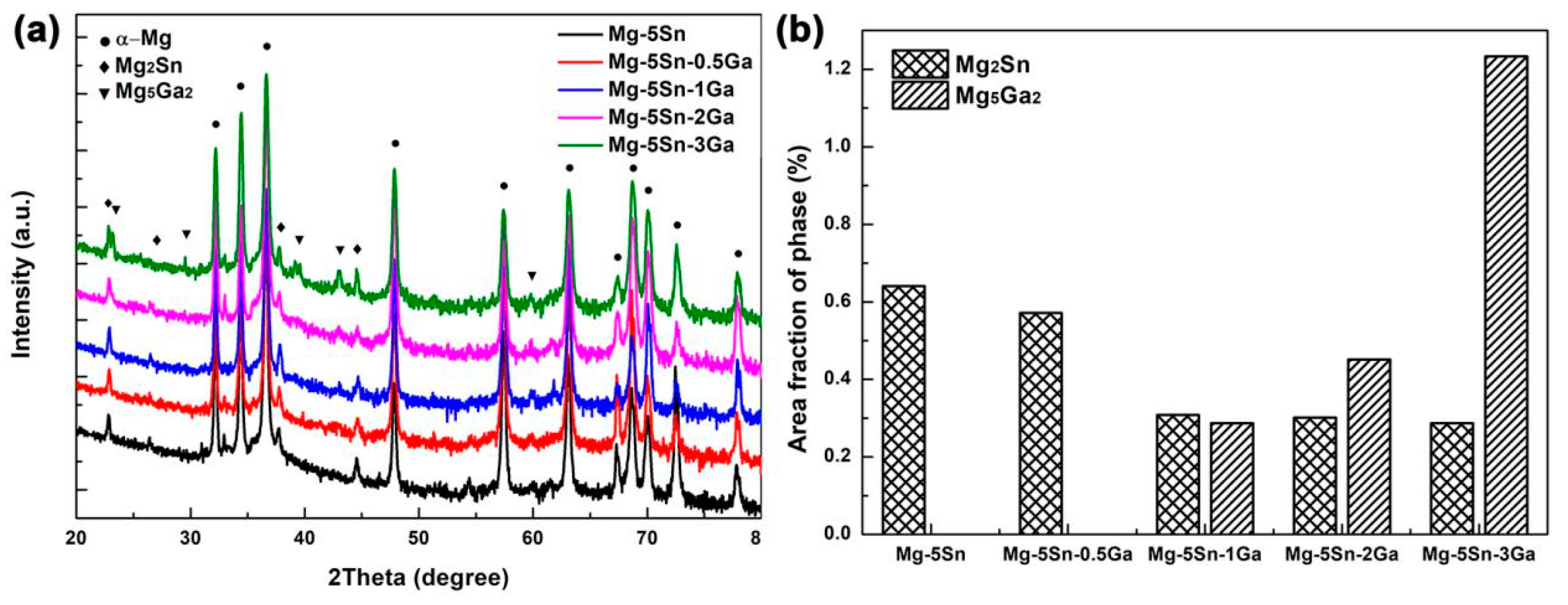
- When 0.5 wt % Ga is added to Mg–5Sn alloy, no new phase is formed. When Ga content exceeds 0.5 wt %, a new eutectic phase, identified as Mg5Ga2, is found in the interdendritic region. Increasing the Ga content decreases the area fraction of Mg2Sn phase, while gradually increasing that of Mg5Ga2 phase;
- Immersion test in 3.5 wt % NaCl solution shows that the corrosion of the studied Mg–Sn–Ga alloys is initiated in a pitting mode, which rapidly propagates with intense H2 evolution on the surface. With the addition of 3 wt % Ga, the overall corrosion rate is decreased significantly. Despite the fact that corrosion is also initiated in a pitting mode, the number of pits is reduced, and the propagation rate is decelerated. The potentiodynamic polarization tests are basically in accordance with the immersion tests.
Author Contributions
Funding
Conflicts of Interest
References
- Ghali, E.; Dietzel, W.; Kainer, K. Testing of General and Localized Corrosion of Magnesium Alloys: A Critical Review. J. Mater. Eng. Perform. 2004, 13, 517–529. [Google Scholar] [CrossRef]
- Kang, D.H.; Park, S.S.; Kim, N.J. Development of Creep Resistant Die Cast Mg–Sn–Al–Si Alloy. Mater. Sci. Eng. A 2005, 413–414, 555–560. [Google Scholar] [CrossRef]
- Luo, A.A. Recent Magnesium Alloy Development for Elevated Temperature Applications. Int. Mater. Rev. 2013, 49, 13–30. [Google Scholar] [CrossRef]
- Song, G.; Atrens, A. Understanding Magnesium Corrosion—A Framework for Improved Alloy Performance. Adv. Eng. Mater. 2003, 5, 837–858. [Google Scholar] [CrossRef]
- Mendis, C.L.; Bettles, C.J.; Gibson, M.A.; Hutchinson, C.R. An Enhanced Age Hardening Response in Mg–Sn Based Alloys Containing Zn. Mater. Sci. Eng. A 2006, 435–436, 163–171. [Google Scholar] [CrossRef]
- Cheng, W.; Zhang, Y.; Ma, S.; Arthanari, S.; Cui, Z.; Wang, H.; Wang, L. Tensile Properties and Corrosion Behavior of Extruded Low-Alloyed Mg–1Sn–1Al–1Zn Alloy: The Influence of Microstructural Characteristics. Materials 2018, 11, 1157. [Google Scholar] [CrossRef]
- Sasaki, T.T.; Oh-ishi, K.; Ohkubo, T.; Hono, K. Enhanced Age Hardening Response by the Addition of Zn in Mg–Sn Alloys. Scr. Mater. 2006, 55, 251–254. [Google Scholar] [CrossRef]
- Liu, H.; Chen, Y.; Tang, Y.; Wei, S.; Niu, G. The Microstructure, Tensile Properties, and Creep Behavior of as-Cast Mg–(1–10)%Sn Alloys. J. Alloys Compd. 2007, 440, 122–126. [Google Scholar] [CrossRef]
- Huang, X.; Han, G.; Huang, W. T6 Treatment and its Effects on Corrosion Properties of an Mg–4Sn–4Zn–2Al Alloy. Materials 2018, 11, 628. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Hua, Z.; Li, D.; Wei, D.; Liu, Y.; Wang, J.; Luo, D.; Wang, H. Effect of Sn Content on the Microstructure, Mechanical Properties and Corrosion Behavior of Biodegradable Mg–X (1, 3 and 5 wt.%) Sn–1Zn–0.5Ca Alloys. Materials 2018, 11, 2378. [Google Scholar] [CrossRef] [PubMed]
- Park, K.C.; Kim, B.H.; Kimura, H.; Park, Y.H.; Park, I.M. Microstructure and Corrosion Properties of Mg–xSn–5Al–1Zn (x = 0, 1, 5 and 9 mass%) Alloys. Mater. Trans. 2010, 51, 472–476. [Google Scholar] [CrossRef]
- Song, G. Effect of Tin Modification on Corrosion of Am70 Magnesium Alloy. Corros. Sci. 2009, 51, 2063–2070. [Google Scholar] [CrossRef]
- Ha, H.; Kang, J.; Yang, J.; Yim, C.D.; You, B.S. Role of Sn in Corrosion and Passive Behavior of Extruded Mg-5 wt % Sn Alloy. Corros. Sci. 2016, 102, 355–362. [Google Scholar] [CrossRef]
- Ha, H.; Kang, J.; Kim, S.G.; Kim, B.; Park, S.S.; Yim, C.D.; You, B.S. Influences of Metallurgical Factors on the Corrosion Behaviour of Extruded Binary Mg-Sn Alloys. Corros. Sci. 2014, 82, 369–379. [Google Scholar] [CrossRef]
- Kubásek, J.; Vojtěch, D.; Lipov, J.; Ruml, T. Structure, Mechanical Properties, Corrosion Behavior and Cytotoxicity of Biodegradable Mg–X (X = Sn, Ga, in) Alloys. Mater. Sci. Eng. C 2013, 33, 2421–2432. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Shan, D.; Song, Y.; Chen, R.; Han, E. Influences of the Quantity of Mg2Sn Phase on the Corrosion Behavior of Mg–7Sn Magnesium Alloy. Electrochim. Acta 2011, 56, 2582–2590. [Google Scholar] [CrossRef]
- Feng, Y.; Wang, R.; Peng, C.; Wang, N. Influence of Mg21Ga5Hg3 Compound on Electrochemical Properties of Mg-5%Hg-5%Ga Alloy. Trans. Nonferr. Met. Soc. China 2009, 19, 154–159. [Google Scholar] [CrossRef]
- Feng, Y.; Wang, R.-c.; Peng, C.-q.; Tang, H.-p.; Liu, H.-y. Influence of Mg5Ga2 Compound on Microstructures and Electrochemical Properties of Mg-5%Hg-22%Ga Alloy. Prog. Nat. Sci. 2011, 21, 73–79. [Google Scholar] [CrossRef]
- Mohedano, M.; Blawert, C.; Yasakau, K.A.; Arrabal, R.; Matykina, E.; Mingo, B.; Scharnagl, N.; Ferreira, M.G.S.; Zheludkevich, M.L. Characterization and Corrosion Behavior of Binary Mg-Ga Alloys. Mater. Charact. 2017, 128, 85–99. [Google Scholar] [CrossRef]
- Shi, Z.; Liu, M.; Atrens, A. Measurement of the Corrosion Rate of Magnesium Alloys Using Tafel Extrapolation. Corros. Sci. 2010, 52, 579–588. [Google Scholar] [CrossRef]
- Cai, S.; Lei, T.; Li, N.; Feng, F. Effects of Zn On Microstructure, Mechanical Properties and Corrosion Behavior of Mg–Zn Alloys. Mater. Sci. Eng. C 2012, 32, 2570–2577. [Google Scholar] [CrossRef]
- Gu, X.; Zheng, Y.; Zhong, S.; Xi, T.; Wang, J.; Wang, W. Corrosion of, and Cellular Responses to Mg–Zn–Ca Bulk Metallic Glasses. Biomaterials 2010, 31, 1093–1103. [Google Scholar] [CrossRef] [PubMed]
- Ha, H.; Kang, J.; Yang, J.; Yim, C.D.; You, B.S. Limitations in the Use of the Potentiodynamic Polarisation Curves to Investigate the Effect of Zn On the Corrosion Behaviour of as-Extruded Mg–Zn Binary Alloy. Corros. Sci. 2013, 75, 426–433. [Google Scholar] [CrossRef]
- Yang, F.; Kang, H.; Guo, E.; Li, R.; Chen, Z.; Zeng, Y.; Wang, T. The Role of Nickel in Mechanical Performance and Corrosion Behaviour of Nickel-Aluminium Bronze in 3.5 Wt.% Nacl Solution. Corros. Sci. 2018, 139, 333–345. [Google Scholar] [CrossRef]
- Jayalakshmi, S.; Sankaranarayanan, S.; Koh, S.P.X.; Gupta, M. Effect of Ag and Cu Trace Additions on the Microstructural Evolution and Mechanical Properties of Mg–5Sn Alloy. J. Alloys Compd. 2013, 565, 56–65. [Google Scholar] [CrossRef]
- Lee, Y.C.; Dahle, A.K.; StJohn, D.H. The Role of Solute in Grain Refinement of Magnesium. Metall. Mater. Trans. A 2000, 31, 2895–2906. [Google Scholar] [CrossRef]
- Easton, M.; StJohn, D. Grain Refinement of Aluminum Alloys: Part II. Confirmation of, and a Mechanism for, the Solute Paradigm. Metall. Mater. Trans. A 1999, 30, 1625–1633. [Google Scholar] [CrossRef]
- Easton, M.; StJohn, D. Grain Refinement of Aluminum Alloys: Part I. The Nucleant and Solute Paradigms—A Review of the Literature. Metall. Mater. Trans. A 1999, 30, 1613–1623. [Google Scholar] [CrossRef]
- Fu, J.W.; Yang, Y.S. Formation of the Solidified Microstructure in Mg–Sn Binary Alloy. J. Cryst. Growth 2011, 322, 84–90. [Google Scholar] [CrossRef]
- Liu, H.; Zhao, G.; Liu, C.; Zuo, L. Effects of Different Tempers On Precipitation Hardening of 6000 Series Aluminium Alloys. Trans. Nonferr. Met. Soc. China 2007, 17, 122–127. [Google Scholar] [CrossRef]
- Liu, C.; Zhu, X.; Zhou, H. Magnesium Alloy Phase Atlas; Central South University Press: Changsha, China, 2006; p. 23. [Google Scholar]
- Zhao, C.; Pan, F.; Zhao, S.; Pan, H.; Song, K.; Tang, A. Preparation and Characterization of as-Extruded Mg–Sn Alloys for Orthopedic Applications. Mater. Des. 2015, 70, 60–67. [Google Scholar] [CrossRef]
- Hamu, G.B.; Eliezer, D.; Wagner, L. The Relation Between Severe Plastic Deformation Microstructure and Corrosion Behavior of Az31 Magnesium Alloy. J. Alloys Compd. 2009, 468, 222–229. [Google Scholar] [CrossRef]
- Liao, J.; Hotta, M.; Yamamoto, N. Corrosion Behavior of Fine-Grained Az31B Magnesium Alloy. Corros. Sci. 2012, 61, 208–214. [Google Scholar] [CrossRef]
- Liu, H.; Qi, G.; Ma, Y.; Hao, H.; Jia, F.; Ji, S.; Zhang, H.; Zhang, X. Microstructure and Mechanical Property of Mg–2.0Ga Alloys. Mater. Sci. Eng. A 2009, 526, 7–10. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Zhen, Z.; Xi, T.; Zheng, Y.; Li, L.; Li, L. In Vitro Study on Mg–Sn–Mn Alloy as Biodegradable Metals. J. Mater. Sci. Technol. 2014, 30, 675–685. [Google Scholar] [CrossRef]
- Song, G.; Unocic, K.A. The Anodic Surface Film and Hydrogen Evolution on Mg. Corros. Sci. 2015, 98, 758–765. [Google Scholar] [CrossRef]
- Ha, H.; Kang, J.; Yim, C.D.; Yang, J.; You, B.S. Role of Hydrogen Evolution Rate in Determining the Corrosion Rate of Extruded Mg–5Sn–(1–4 Wt.%) Zn Alloys. Corros. Sci. 2014, 89, 275–285. [Google Scholar] [CrossRef]
- Ha, H.; Kim, H.J.; Baek, S.; Kim, B.; Sohn, S.; Shin, H.; Jeong, H.Y.; Park, S.H.; Yim, C.D.; You, B.S.; et al. Improved Corrosion Resistance of Extruded Mg–8Sn–1Zn–1Al Alloy by Microalloying with Mn. Scr. Mater. 2015, 109, 38–43. [Google Scholar] [CrossRef]
- Kim, H.J.; Kim, B.; Baek, S.; Sohn, S.; Shin, H.; Jeong, H.Y.; Yim, C.D.; You, B.S.; Ha, H.; Park, S.S. Influence of Alloyed Al On the Microstructure and Corrosion Properties of Extruded Mg–8Sn–1Zn Alloys. Corros. Sci. 2015, 95, 133–142. [Google Scholar] [CrossRef]
- Zhao, C.; Pan, F.; Zhao, S.; Pan, H.; Song, K.; Tang, A. Microstructure, Corrosion Behavior and Cytotoxicity of Biodegradable Mg–Sn Implant Alloys Prepared by Sub-Rapid Solidification. Mater. Sci. Eng. C 2015, 54, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Rad, H.R.B.; Idris, M.H.; Kadir, M.R.A.; Farahany, S. Microstructure Analysis and Corrosion Behavior of Biodegradable Mg–Ca Implant Alloys. Mater. Des. 2012, 33, 88–97. [Google Scholar] [CrossRef]
- Yim, C.D.; Yang, J.; Woo, S.K.; Ha, H.; You, B.S. The Effects of Microstructural Factors on the Corrosion Behaviour of Mg–5Sn–X Zn (X = 1, 3 wt%) Extrusions. Corros. Sci. 2015, 90, 597–605. [Google Scholar] [CrossRef]
- Bratsch, S.G. Standard Electrode Potentials and Temperature Coefficients in Water at 298.15 K. J. Phys. Chem. Ref. Data 1989, 18, 1–21. [Google Scholar] [CrossRef]
- Chen, J.; Wang, J.; Han, E.; Ke, W. In Situ Observation of Pit Initiation of Passivated Az91 Magnesium Alloy. Corros. Sci. 2009, 51, 477–484. [Google Scholar] [CrossRef]



| Nominal Composition | Element Content (wt %) | ||
|---|---|---|---|
| Sn | Ga | Mg | |
| Mg–5Sn | 5.08 | - | Bal. |
| Mg–5Sn–0.5Ga | 4.98 | 0.56 | Bal. |
| Mg–5Sn–1Ga | 4.81 | 1.12 | Bal. |
| Mg–5Sn–2Ga | 5.08 | 2.02 | Bal. |
| Mg–5Sn–3Ga | 4.67 | 2.81 | Bal. |
| Area | Sn | Ga | Mg |
|---|---|---|---|
| a | 9.778 | 1.929 | 88.293 |
| b | 25.115 | 0.741 | 74.144 |
| c | 1.589 | 0.167 | 98.244 |
| Point | Sn | Ga | Mg |
|---|---|---|---|
| a | 2.2 | 19.6 | 78.2 |
| b | 29.3 | 4.4 | 66.3 |
| Alloy | Mg–5Sn | Mg–5Sn–0.5Ga | Mg–5Sn–1Ga | Mg–5Sn–2Ga | Mg–5Sn–3Ga |
|---|---|---|---|---|---|
| Ecorr (V) | −1.596 | −1.614 | −1.658 | −1.665 | −1.684 |
| icorr (mA/cm2) | 9.73 × 10−2 | 3.28 × 10−2 | 3.82 × 10−2 | 3.16 × 10−2 | 2.79 × 10−2 |
| Ri (mm/y) | 2.223 | 0.770 | 0.873 | 0.722 | 0.638 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ren, J.; Guo, E.; Wang, X.; Kang, H.; Chen, Z.; Wang, T. Influence of Alloyed Ga on the Microstructure and Corrosion Properties of As-Cast Mg–5Sn Alloys. Materials 2019, 12, 3686. https://doi.org/10.3390/ma12223686
Ren J, Guo E, Wang X, Kang H, Chen Z, Wang T. Influence of Alloyed Ga on the Microstructure and Corrosion Properties of As-Cast Mg–5Sn Alloys. Materials. 2019; 12(22):3686. https://doi.org/10.3390/ma12223686
Chicago/Turabian StyleRen, Jing, Enyu Guo, Xuejian Wang, Huijun Kang, Zongning Chen, and Tongmin Wang. 2019. "Influence of Alloyed Ga on the Microstructure and Corrosion Properties of As-Cast Mg–5Sn Alloys" Materials 12, no. 22: 3686. https://doi.org/10.3390/ma12223686
APA StyleRen, J., Guo, E., Wang, X., Kang, H., Chen, Z., & Wang, T. (2019). Influence of Alloyed Ga on the Microstructure and Corrosion Properties of As-Cast Mg–5Sn Alloys. Materials, 12(22), 3686. https://doi.org/10.3390/ma12223686






