Effects of Corrosion on Mechanical Properties of Welded Carbon Steel Pipe in District Heating Water
Abstract
1. Introduction
2. Materials and Methods
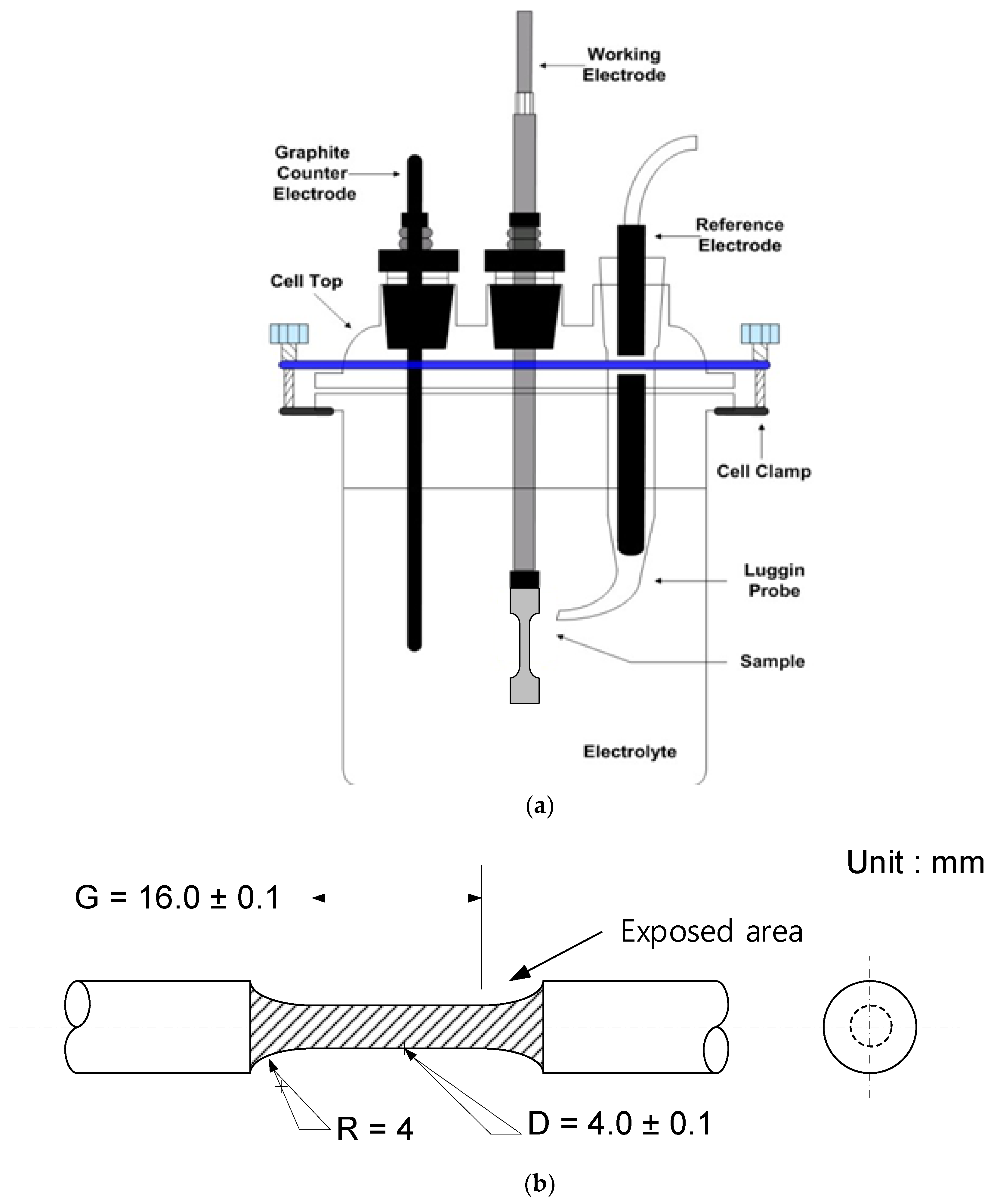
2.1. Specimen and Solution Preparation
2.2. Potentiodynamic Polarization Test
2.3. Galvanostatic Method
2.4. Tensile Test
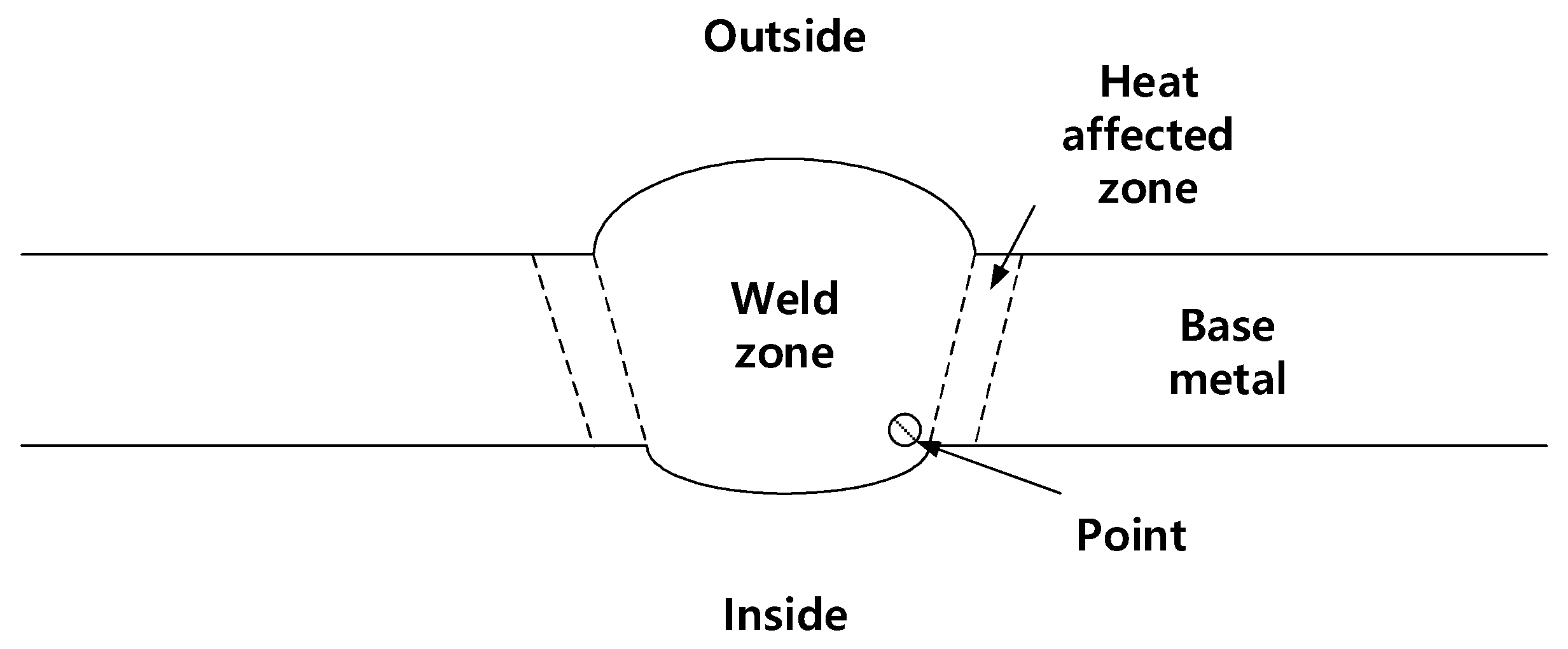
2.5. Microstructure Observation
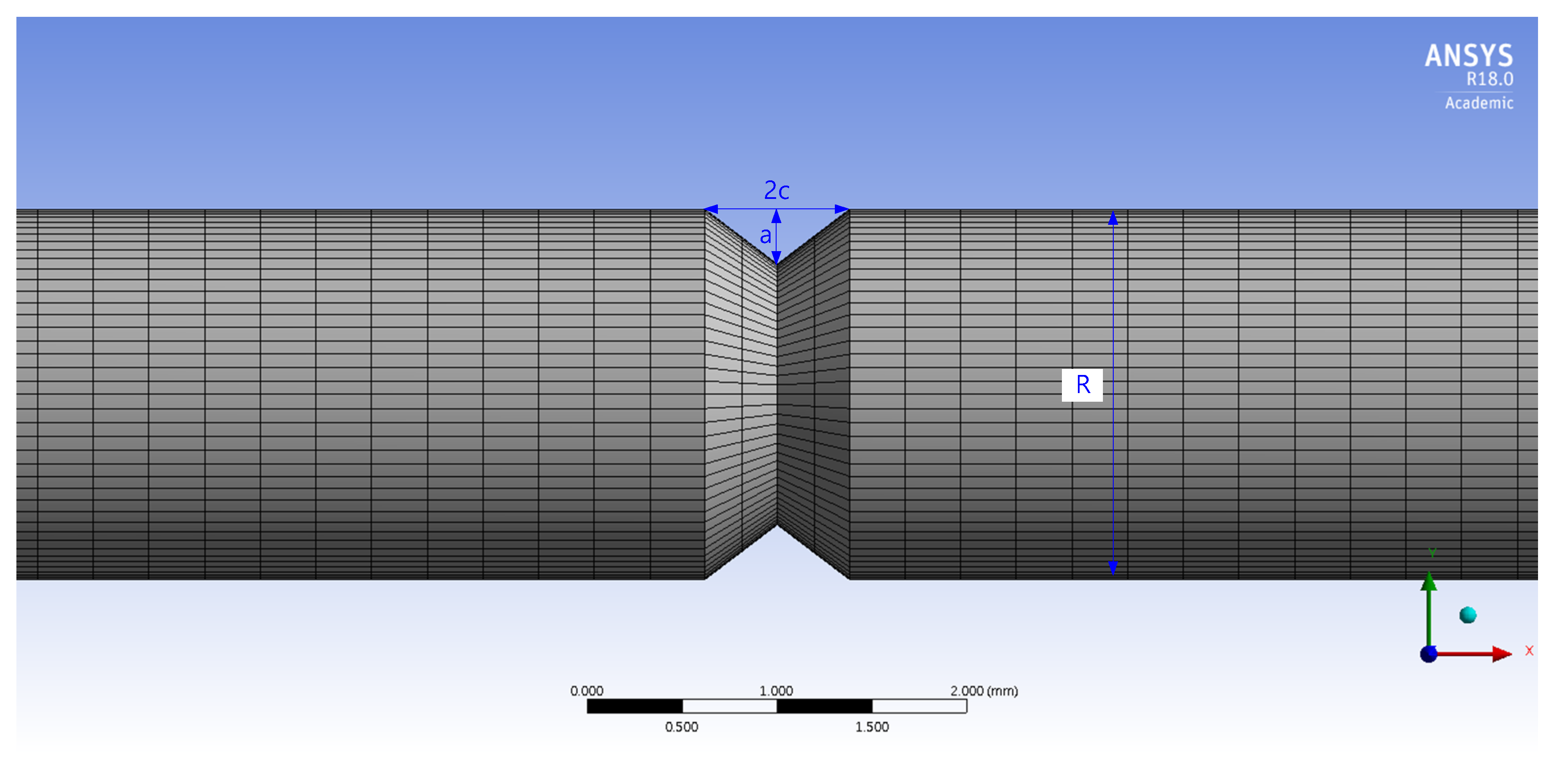
2.6. FEM Simulation
2.7. Instrumented Indentation Test
3. Results
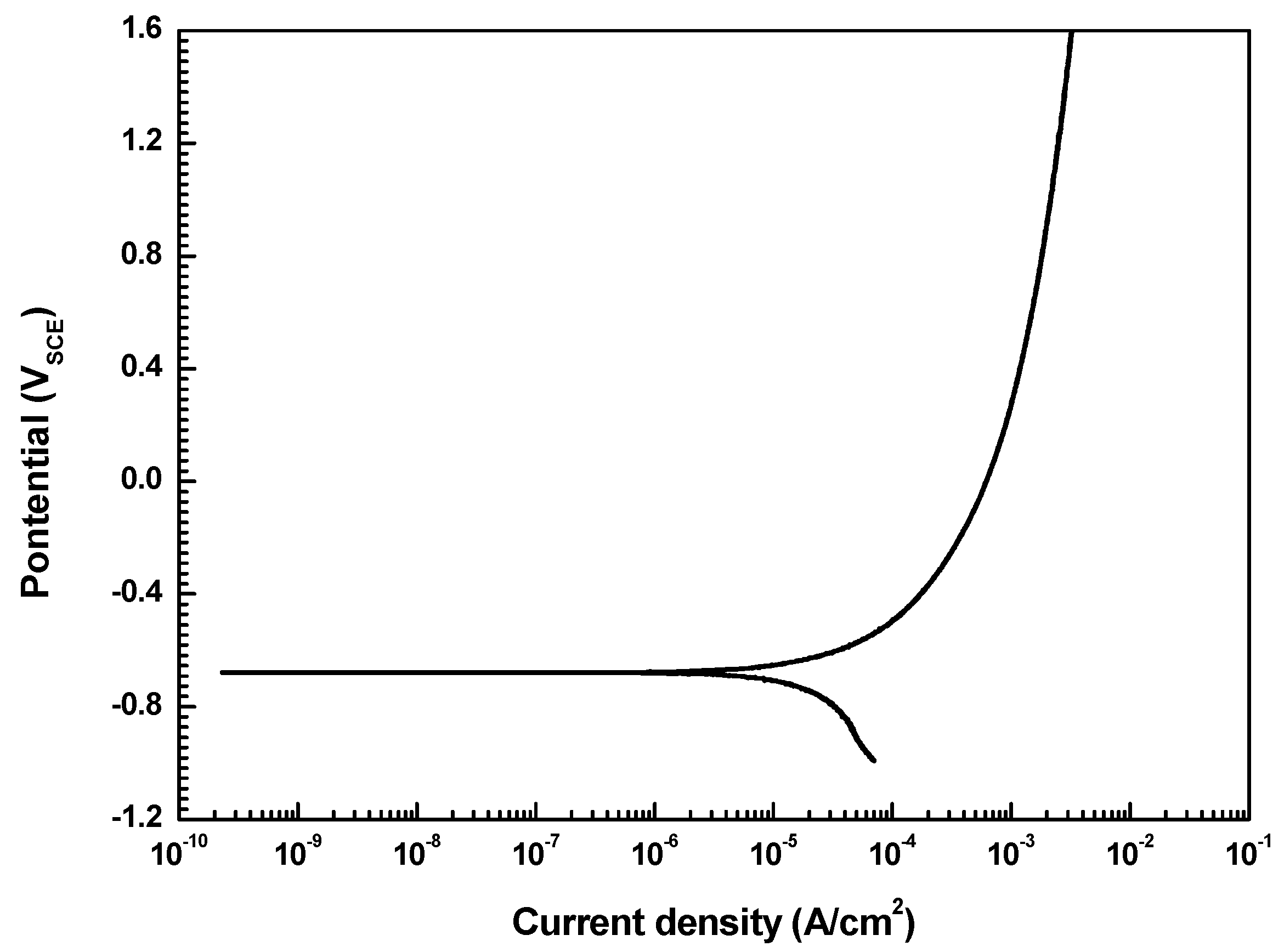
3.1. Potentiodynamic Polarization Test
3.2. Corrosion Acceleration by Galvanostatic Test Method
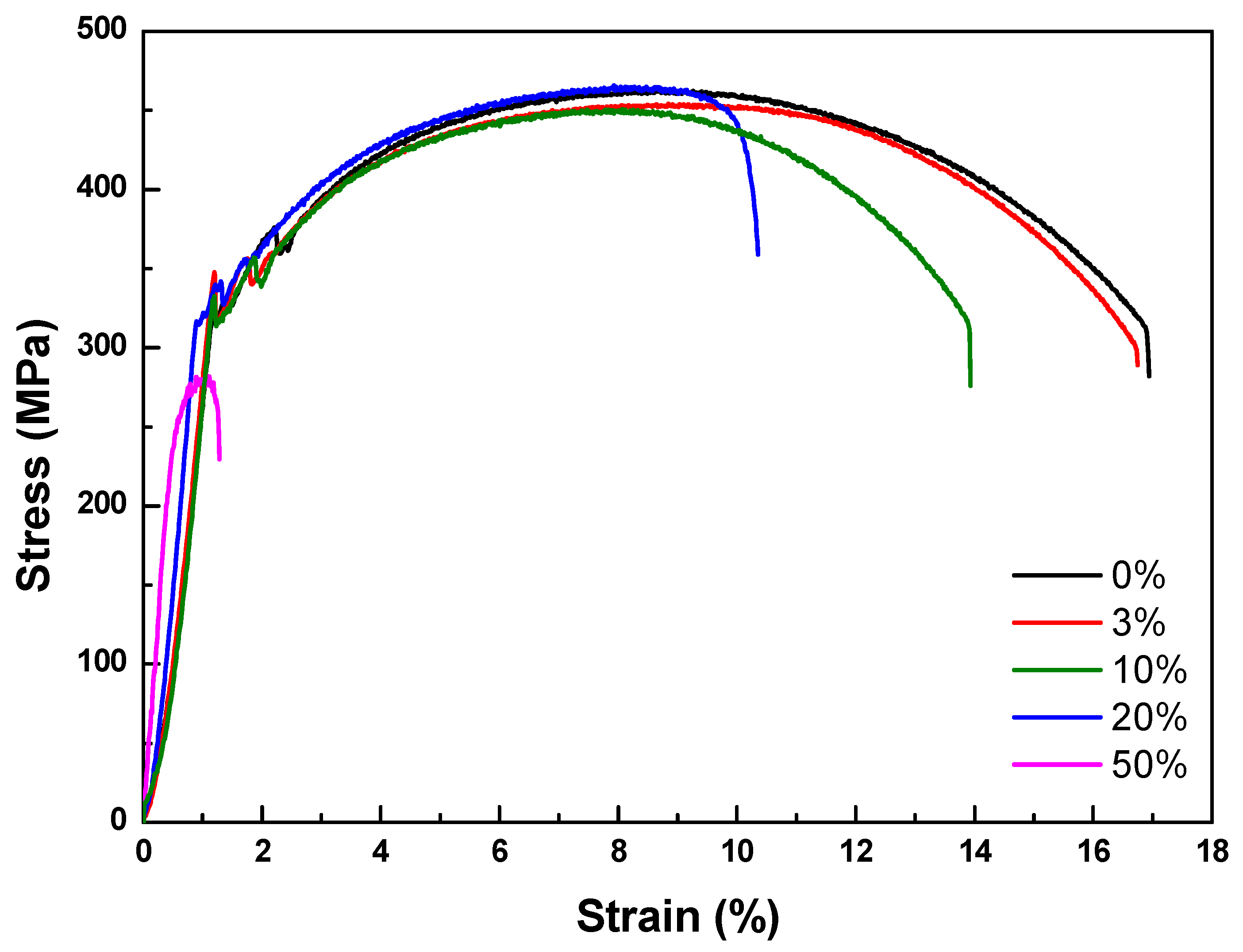
3.3. Tensile Test
3.4. Microstructural Analysis
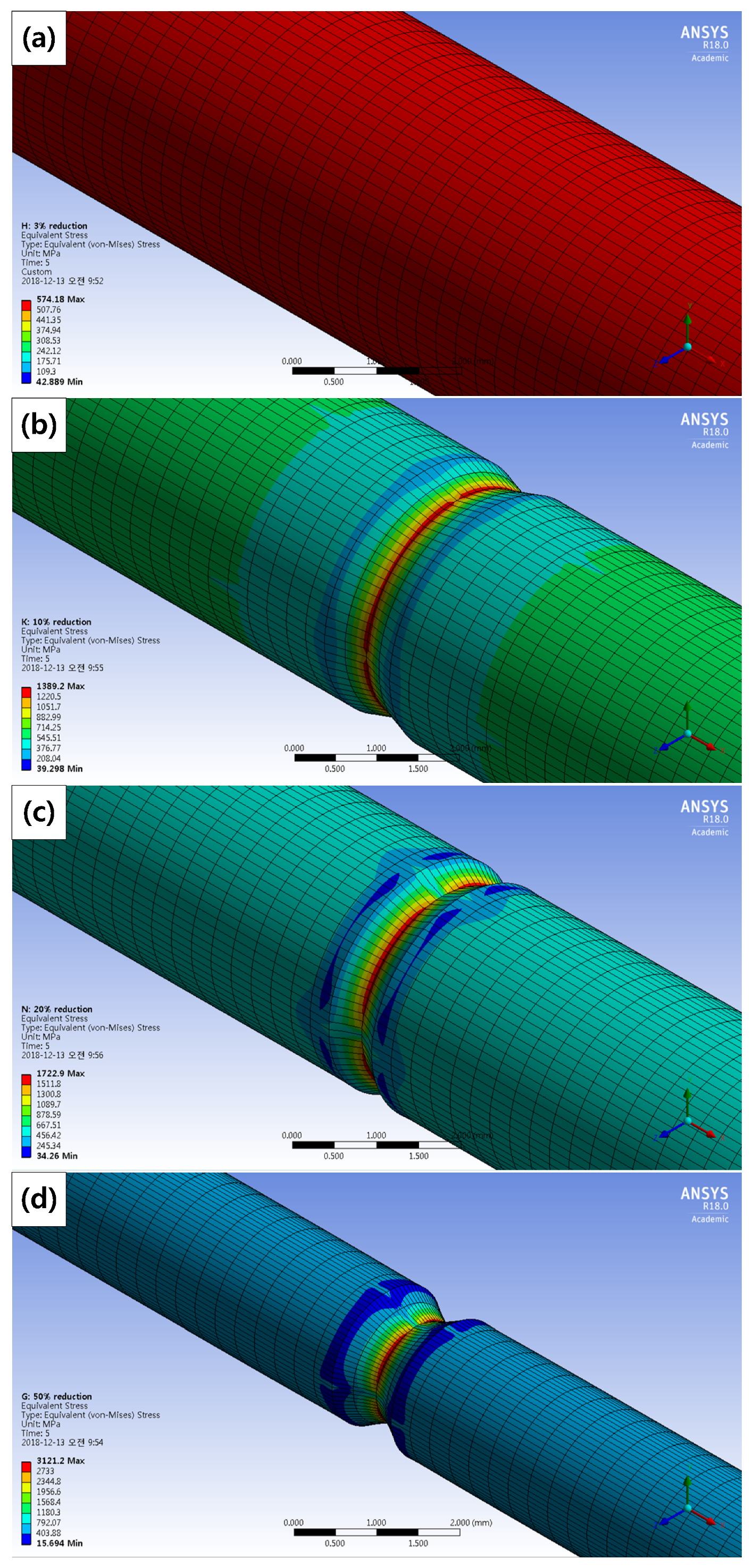
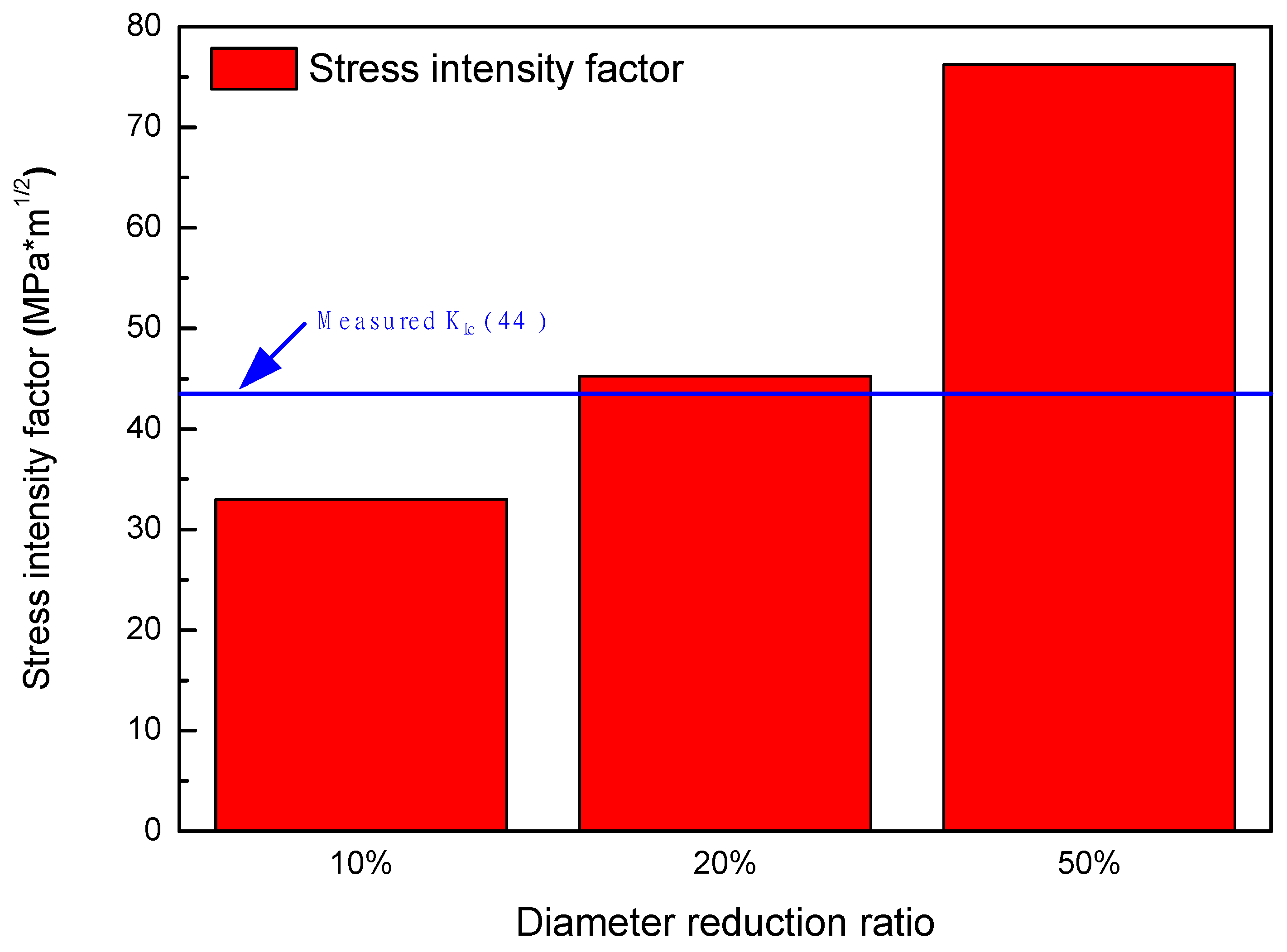
3.5. Stress Analysis and Stress Intensity Factor
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Protić, M.; Shamshirband, S.; Petković, D.; Abbasi, A.; Kiah, M.L.; Unar, J.A.; Živković, L.; Raos, M. Forecasting of consumers heat load in district heating systems using the support vector machine with a discrete wavelet transform algorithm. Energy 2015, 87, 343–351. [Google Scholar] [CrossRef]
- Kwon, O.K.; Cha, D.A.; Park, C.S. Performance evaluation of a two-stage compression heat pump system for district heating using waste energy. Energy 2013, 57, 375–381. [Google Scholar] [CrossRef]
- Roberge, P.R. Corrosion Inspection and Monitoring; John Wiley & Sons: Hoboken, NJ, USA, 2007; Volume 2, pp. 154–196. [Google Scholar]
- Garverick, L. Corrosion in Petrochemical Industry, 2nd ed.; ASM International: Novelty, OH, USA, 1994; pp. 3–48. [Google Scholar]
- Gutzeit, J.; Merrick, R.D.; Scharfstein, L.K. ASM Metals Handbook; ASM International: Novelty, OH, USA, 2006; Volume 13, pp. 1262–1287. [Google Scholar]
- Nasirpouri, F.; Mostafaei, A.; Fathyunes, L.; Jafari, R. Assessment of localized corrosion in carbon steel tube-grade AISI 1045 used in output oil-gas separator vessel of desalination unit in oil refinery industry. Eng. Fail. Anal. 2014, 40, 75–88. [Google Scholar] [CrossRef]
- Daneshvar-Fatah, F.; Mostafaei, A.; Hosseinzadeh-Taghani, R.; Nasirpouri, F. Caustic corrosion in a boiler waterside tube: Root cause and mechanism. Eng. Fail. Anal. 2013, 28, 69–77. [Google Scholar] [CrossRef]
- Mostafaei, A.; Peighambari, S.M.; Nasirpouri, F. Failure analysis of monel packing in atmospheric distillation tower under the service in the presence of corrosive gases. Eng. Fail. Anal. 2013, 28, 241–251. [Google Scholar] [CrossRef]
- Shirinzadeh-Dastgiri, M.; Mohammadi, J.; Behnamian, Y.; Eghlimi, A.; Mostafaei, A. Metallurgical investigations and corrosion behavior of failed weld joint in AISI 1518 low carbon steel pipeline. Eng. Fail. Anal. 2015, 53, 78–96. [Google Scholar] [CrossRef]
- Srivastava, B.K.; Tewari, S.P.; Prakash, J. A reivew on effect of preheating and/or post weld heat treatment (PWHT) on mechanical behaviour of ferrous metals. Int. J. Eng. Sci. Tech. 2010, 2, 625–631. [Google Scholar]
- Xue, Q.; Benson, D.; Meyers, M.A.; Nesterenko, V.F.; Olevsky, E.A. Constitutive response of welded HSLA 100 steel. Mat. Sci. Eng. 2003, A354, 166–179. [Google Scholar] [CrossRef]
- Raamirez, J.E.; Mishael, S.; Shockley, R. Properties and sulfide stress cracking resistance of coarse-grained heat-affected zones in V-micro alloyed X60 Steel Pipe. Weld. J. 2005, 84, 113–123. [Google Scholar]
- Samuel, E.I.; Choudhary, B.K.; Rao, B.S. Influence of post-weld heat treatment on tensile properties of modified 9Cr-1Mo ferritic steel base metal. Mater. Sci. Tech. 2007, 23, 992–999. [Google Scholar] [CrossRef]
- Tavakoli Shoushtari, M.R.; Moayed, M.H.; Davoodi, A. Post-weld heat treatment influence on galvanic corrosion of GTAW of 17-4PH stainless steel in 3.5%NaCl. Corr. Eng. Sci. Tech. 2011, 46, 415–424. [Google Scholar] [CrossRef]
- Wang, W.; Zhou, A.; Fu, G.; Li, C.Q.; Robert, D.; Mahmoodian, M. Evaluation of stress intensity factor for cast iron pipes with sharp corrosion pits. Eng. Fail. Anal. 2017, 81, 254–269. [Google Scholar] [CrossRef]
- Apostolopoulos, C.A.; Papadakis, V.G. Consequences of steel corrosion on the ductility properties of reinforcement bar. Constr. Build. Mater. 2008, 22, 2316–2324. [Google Scholar] [CrossRef]
- Almusallam, A.A. Effect of degree of corrosion on the properties of reinforcing steel bars. Constr. Build. Mater. 2001, 15, 361–368. [Google Scholar] [CrossRef]
- Du, Y.G.; Clark, L.A.; Chan, A.H.C. Effect of corrosion on ductility of reinforcing bars. Mag. Concrete. Res. 2005, 57, 407–419. [Google Scholar] [CrossRef]
- Du, Y.G.; Clark, L.A.; Chan, A.H.C. Residual capacity of corroded reinforcing bars. Mag. Concrete. Res. 2005, 57, 135–147. [Google Scholar] [CrossRef]
- ASTM, G5.-5, Test Method for Making Potentiostatic and Potentiodynamic Anodic Polarization Measurements; ASTM International: West Conshohocken, PA, USA, 2004.
- Ahn, W.; Reddy, D.V. Galvanostatic testing for the durability of marine concrete under fatigue loading. Cement. Concrete. Res. 2001, 31, 343–349. [Google Scholar] [CrossRef]
- ASTM, E8–99-Standard test methods for tension testing of metallic materials; ASTM International: West Conshohocken, PA, USA, 2001.
- Jones, D.A. Principles and Prevention of Corrosion, 2nd ed.; Prentice-Hall: Hoboken, NJ, USA, 1996; pp. 75–77. [Google Scholar]
- Macdonald, J.R.; Kenan, W.R. Impedance Spectroscopy: Emphasizing Solid Materials and Systems; John Wiley & Sons: New York, NY, USA, 1987; p. 13. [Google Scholar]
- Linnert, G.E. Welding Metallurgy: Carbon and Alloy Steels, 4th ed.; American Welding Society: Miami, FL, USA, 1994; pp. 74–88. [Google Scholar]
- Ralston, K.D.; Birbilis, N. Effect of grain size on corrosion: A review. Corrosion 2010, 66, 075005. [Google Scholar] [CrossRef]
- Lazzarin, P.; Tovo, R. A unified approach to the evaluation of linear elastic stress fields in the neighborhood of cracks and notches. Int. J. Fract. 1996, 78, 3–19. [Google Scholar] [CrossRef]
- Hertzberg, R.W. Deformation and Fracture Mechanics of Engineering Materials, 5th ed.; Wiley: New York, NY, USA, 1996; pp. 571–600. [Google Scholar]
- Williams, M.L. Stress singularities resulting from various boundary conditions. J. Appl. Mech. 1952, 19, 526–528. [Google Scholar]









| Elements | C | Mn | P | S | Fe |
|---|---|---|---|---|---|
| Composition (wt.%) | 0.25 | 1.00 | 0.040 | 0.040 | Bal. |
| Welding Process | Gas Tungsten Arc Welding (GTAW) |
|---|---|
| Joint design | Single-V joint with a 65 ± 5 degree included angle and a 1.6 ± 0.8 mm root face |
| Number of passes | 1–2 or higher |
| Electrode | ER70S-G (2.4 or 3.2 mm diameter, Thoriated, TGC-50) |
| Voltage | 11–15 V or 22–28 V |
| Current | 80–130 A or 100–170 A |
| Polarity | Direct current straight polarity (DCSP) |
| Travel speed | 5–10 cm/min or 6–16 cm/min |
| Welding atmosphere | Argon, 7–17 L/min |
| Elements | NaCl | Mg(OH)2 | CaCO3 | NH4OH |
|---|---|---|---|---|
| Composition | 15.01 | 0.48 | 2.65 | 10.28 |
| Specimens | Ecorr (mVSCE) | icorr (μA/cm2) | βa (V/Decade) | βc (V/decade) | Corrosion Rate (mm/Year) |
|---|---|---|---|---|---|
| As-received | −678.5 | 16.4 | 0.021 | 0.038 | 0.2 |
| Specimens (% Diameter Reduction) | a (mm) | 2c (mm) | R (mm) |
|---|---|---|---|
| 0% | – | – | 3.95 |
| 3% | – | – | 3.87 |
| 10% | 0.18 | 0.96 | 3.53 |
| 20% | 0.22 | 0.58 | 3.18 |
| 50% | 0.19 | 0.76 | 1.96 |
| Charge Amounts | icorr | Equivalent Aging Time (Year) | iapp | Test Time (h) |
|---|---|---|---|---|
| 0 | 16 uA/cm2 | 0 | 1 mA/cm2 | 0 |
| 811.7 | 0.4 | 57.8 | ||
| 2435.2 | 1 | 173.5 | ||
| 4058.7 | 2 | 289.1 | ||
| 8117.4 | 4 | 578.2 |
| Charge Amounts | Diameter Reduction Ratio (%) | Diameter (mm) | Diameter Reduction (mm) |
|---|---|---|---|
| – | 0 | 3.95 | – |
| 811.7 | 3 | 3.87 | 0.08 |
| 2435.2 | 10 | 3.53 | 0.42 |
| 4058.7 | 20 | 3.18 | 0.77 |
| 8117.4 | 50 | 1.96 | 1.99 |
| Specimens | Mechanical Properties | ||
|---|---|---|---|
| Yield Stress (MPa) | Tensile Stress (MPa) | Elongation (%) | |
| 0% reduction | 374.54 | 463.40 | 16.94 |
| 3% reduction | 347.74 | 454.13 | 16.75 |
| 10% reduction | 332.62 | 450.62 | 13.93 |
| 20% reduction | 336.27 | 465.62 | 10.36 |
| 50% reduction | 208.09 | 282.12 | 1.28 |
| Specimens | Maximum Stress (MPa) | a (mm) | Stress Intensity Factor |
|---|---|---|---|
| 0% reduction | – | – | – |
| 3% reduction | 578.06 | – | – |
| 10% reduction | 1389.2 | 0.18 | 33.03 |
| 20% reduction | 1722.9 | 0.22 | 45.28 |
| 50% reduction | 3121.2 | 0.19 | 76.23 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ko, S.-J.; An, J.-H.; Kim, Y.-S.; Kim, W.-C.; Kim, J.-G. Effects of Corrosion on Mechanical Properties of Welded Carbon Steel Pipe in District Heating Water. Materials 2019, 12, 3682. https://doi.org/10.3390/ma12223682
Ko S-J, An J-H, Kim Y-S, Kim W-C, Kim J-G. Effects of Corrosion on Mechanical Properties of Welded Carbon Steel Pipe in District Heating Water. Materials. 2019; 12(22):3682. https://doi.org/10.3390/ma12223682
Chicago/Turabian StyleKo, Sang-Jin, Jeong-Hun An, Yong-Sang Kim, Woo-Cheol Kim, and Jung-Gu Kim. 2019. "Effects of Corrosion on Mechanical Properties of Welded Carbon Steel Pipe in District Heating Water" Materials 12, no. 22: 3682. https://doi.org/10.3390/ma12223682
APA StyleKo, S.-J., An, J.-H., Kim, Y.-S., Kim, W.-C., & Kim, J.-G. (2019). Effects of Corrosion on Mechanical Properties of Welded Carbon Steel Pipe in District Heating Water. Materials, 12(22), 3682. https://doi.org/10.3390/ma12223682





