Morphological and Spectroscopic Study of an Apatite Layer Induced by Fast-Set Versus Regular-Set EndoSequence Root Repair Materials
Abstract
1. Introduction
2. Materials and Methods
2.1. Specimen Preparation
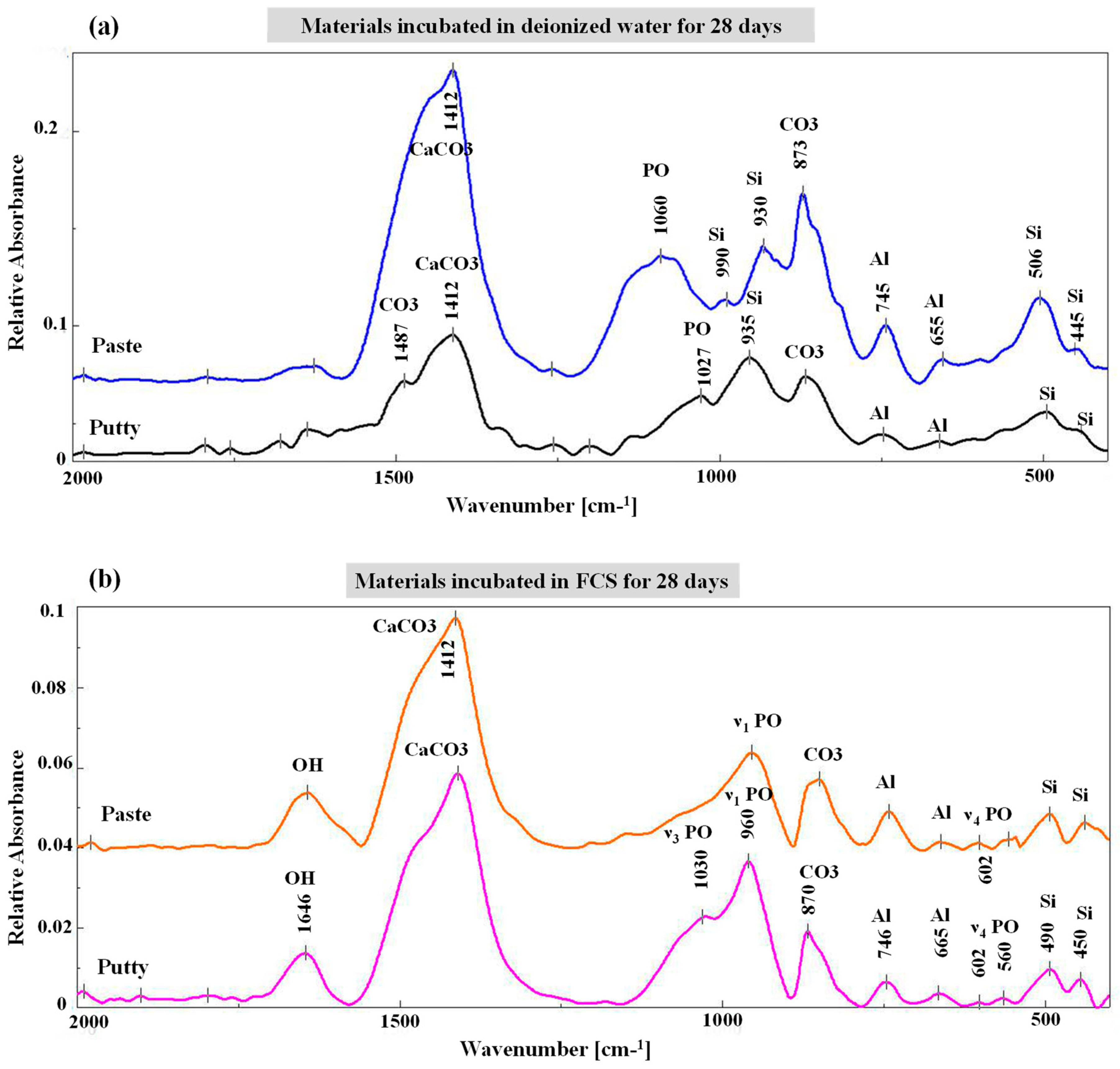
2.2. Raman Spectroscopy
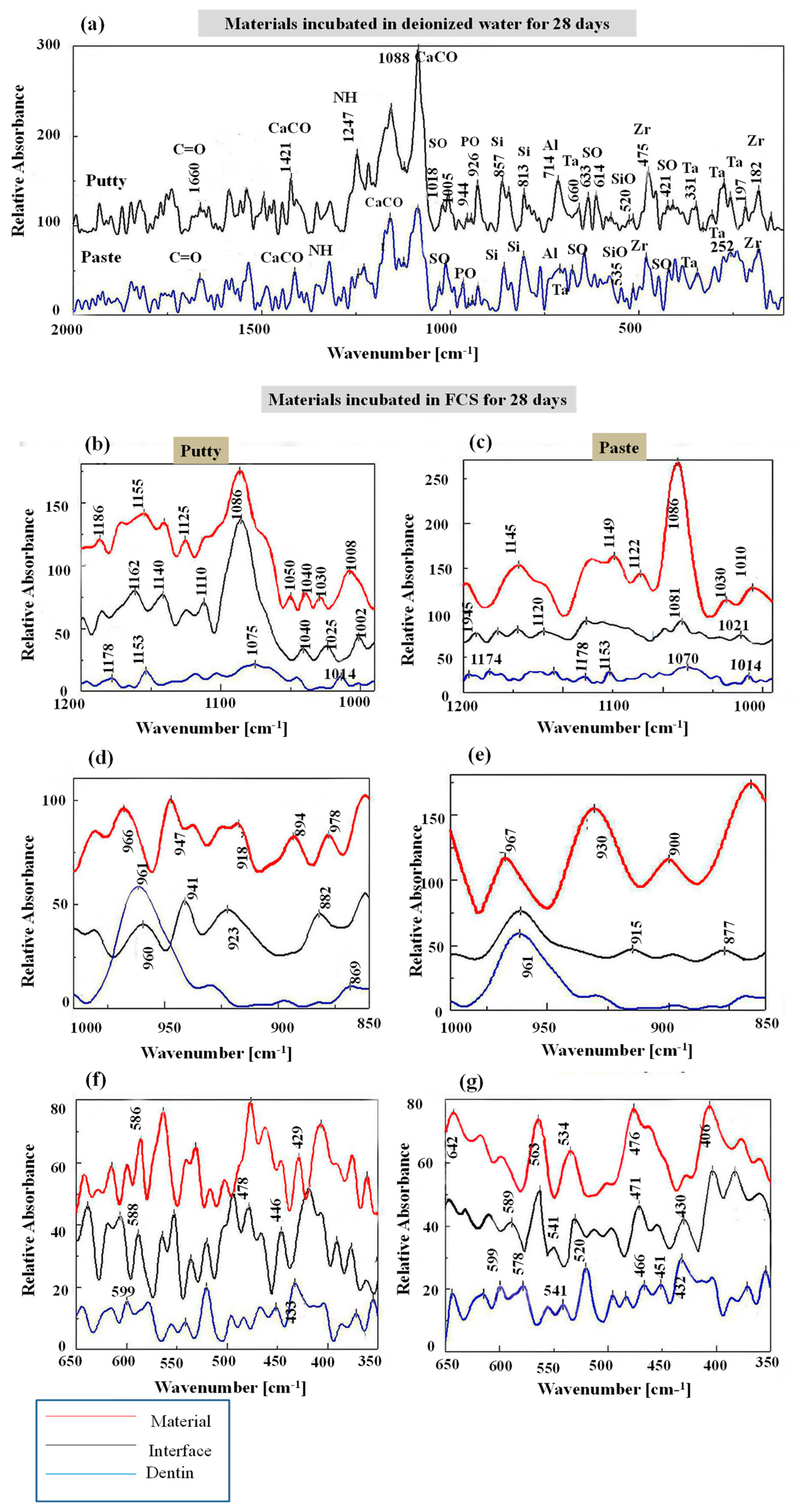
2.3. Fourier Transform Infrared Spectroscopy
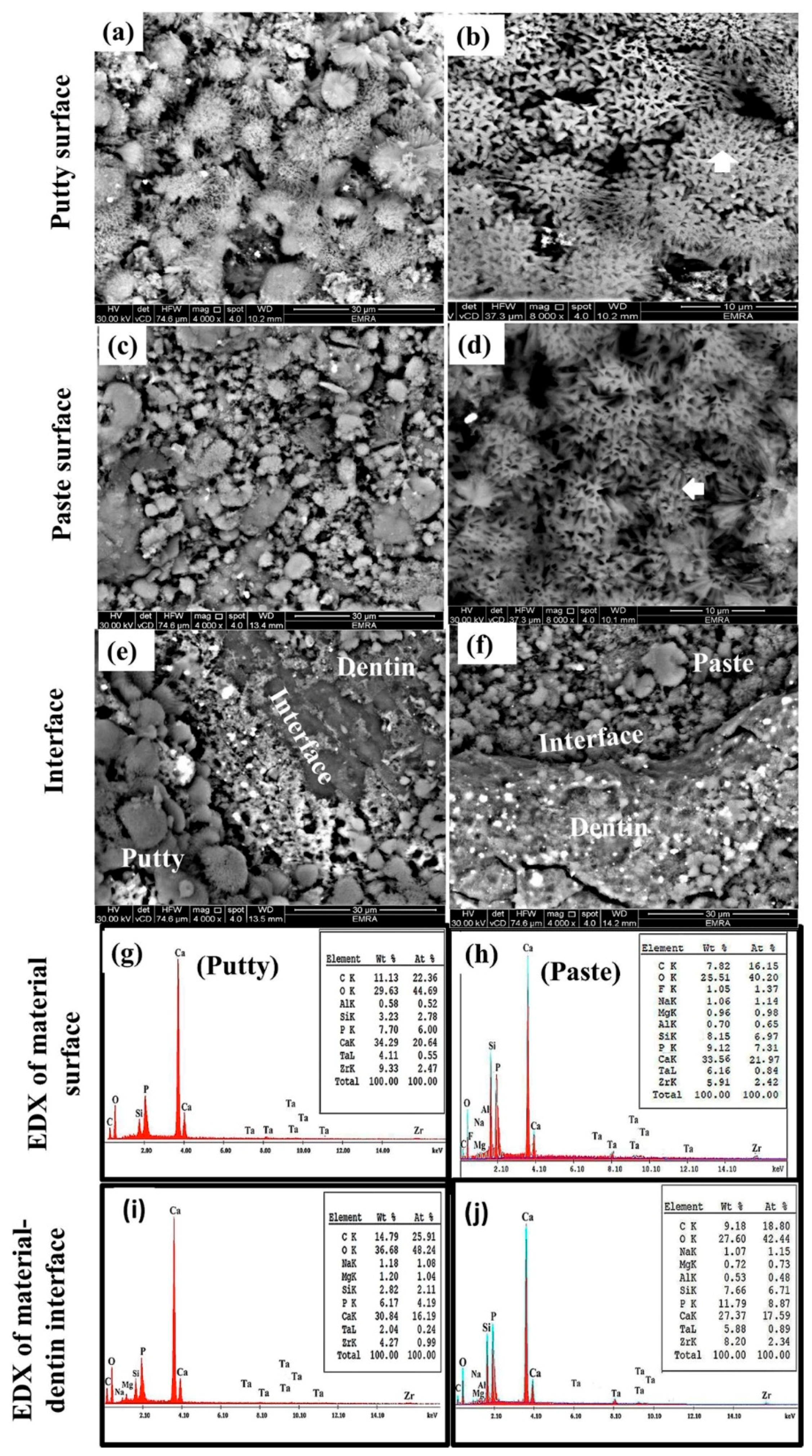
2.4. Scanning Electron Microscope/Energy Dispersed X-ray Analysis (SEM/EDX)
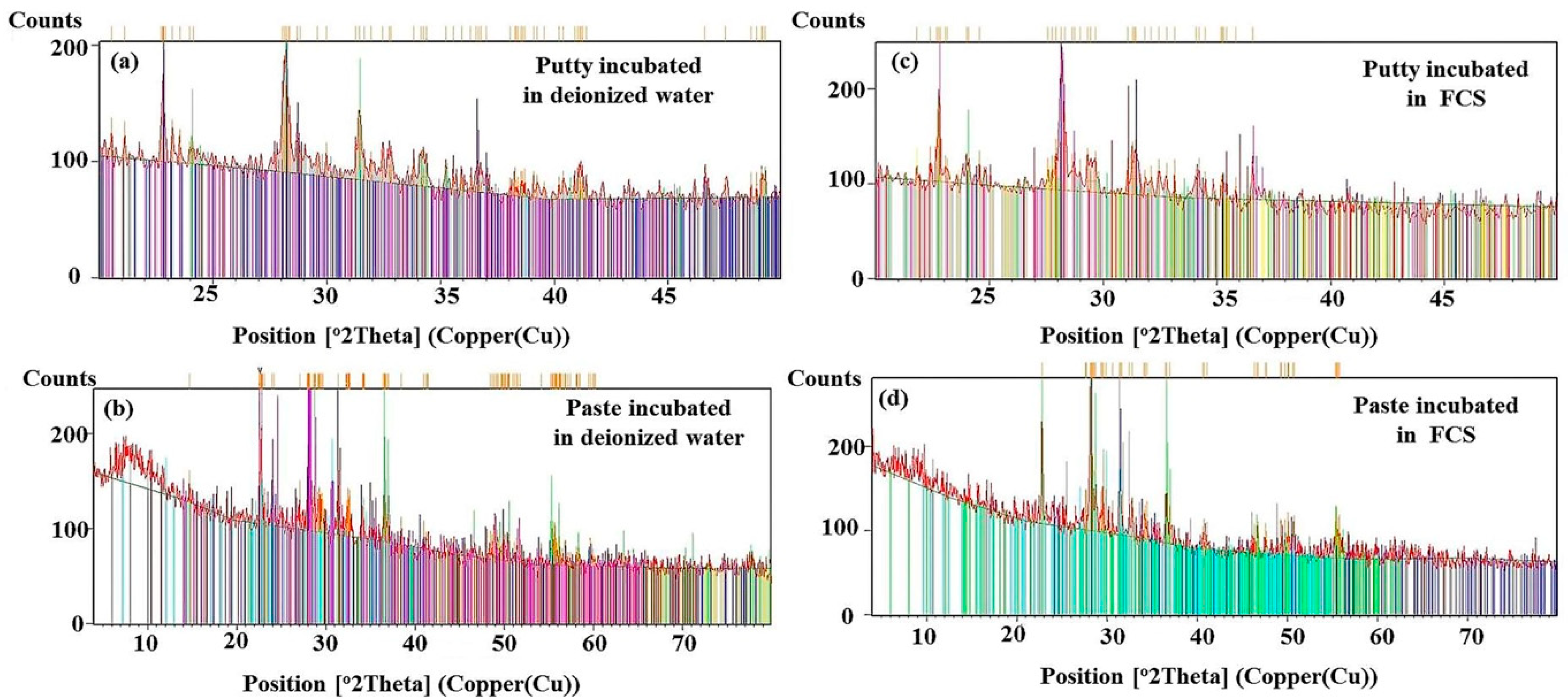
2.5. X-ray Diffraction (XRD)
2.6. Statistical Analyses
3. Results
3.1. Raman Spectroscopy
3.2. Fourier Transform Infrared Spectroscopy
3.3. Scanning Electron Microscopy/Energy Dispersed X-ray Analysis
3.4. X-ray Diffraction Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Parirokh, M.; Torabinejad, M. Mineral trioxide aggregate: A comprehensive literature review—Part III: Clinical applications, drawbacks, and mechanism of action. J. Endod. 2010, 36, 400–413. [Google Scholar] [CrossRef] [PubMed]
- Roberts, H.W.; Toth, J.M.; Berzins, D.W.; Charlton, D.G. Mineral trioxide aggregate material use in endodontic treatment: A review of the literature. Dent. Mater. 2008, 24, 149–164. [Google Scholar] [CrossRef] [PubMed]
- Main, C.; Mirzayan, N.; Shabahang, S.; Torabinejad, M. Repair of root perforations using mineral trioxide aggregate: A long-term study. J. Endod. 2004, 30, 80–83. [Google Scholar] [CrossRef] [PubMed]
- Abu Zeid, S.; Saleh, A.M.; Khafagi, M.; Neel, E.A. Setting Reaction of New Bioceramic Root Canal Sealers. Spectrosc. Lett. 2018, 51, 426–430. [Google Scholar] [CrossRef]
- Han, L.; Okiji, T.; Okawa, S. Morphological and chemical analysis of different precipitates on mineral trioxide aggregate immersed in different fluids. Dent. Mater. J. 2010, 29, 512–517. [Google Scholar] [CrossRef] [PubMed]
- Gandolfi, M.G.; Taddei, P.; Tinti, A.; Prati, C. Apatite-forming ability (bioactivity) of ProRoot MTA. Int. Endod. J. 2010, 43, 917–929. [Google Scholar] [CrossRef]
- Alamoudi, R.A.; Abu Zeid, S.T. Effect of Irrigants on the Push-Out Bond Strength of Two Bioceramic Root Repair Materials. Materials 2019, 12, 1921. [Google Scholar] [CrossRef]
- Moinzadeh, A.T.; Portoles, C.A.; Wismayer, P.S.; Camilleri, J. Bioactivity potential of EndoSequence BC RRM putty. J. Endod. 2016, 42, 615–621. [Google Scholar] [CrossRef]
- Shokouhinejad, N.; Nekoofar, M.H.; Razmi, H.; Sajadi, S.; Davies, T.E.; Saghiri, M.A.; Gorjestani, H.; Dummer, P.M.H. Bioactivity of EndoSequence root repair material and bioaggregate. Int. Endod. J. 2012, 45, 1127–1134. [Google Scholar] [CrossRef]
- Zamparini, F.; Siboni, F.; Prati, C.; Taddei, P.; Gandolfi, M.G. Properties of calcium silicate-monobasic calcium phosphate materials for endodontics containing tantalum pentoxide and zirconium oxide. Clinl. Oral. Investig. 2018, 23, 445–457. [Google Scholar] [CrossRef]
- Tingey, M.C.; Bush, P.; Levine, M.S. Analysis of mineral trioxide aggregate surface when set in the presence of fetal bovine serum. J. Endod. 2008, 34, 45–49. [Google Scholar] [CrossRef]
- Gandolfi, M.G.; Ciapetti, G.; Taddei, P.; Perut, F.; Tinti, A.; Cardoso, M.V.; Van Meerbeek, B.; Prati, C. Apatite formation on bioactive calcium-silicate cements for dentistry affects surface topography and human marrow stromal cells proliferation. Dent. Mater. 2010, 26, 974–992. [Google Scholar] [CrossRef] [PubMed]
- Abu Zeid, S.T.; Alamoudi, N.M.; Khafagi, M.G.; Abou Neel, E.A. Chemistry and Bioactivity of NeoMTA Plus™ versus MTA Angelus® Root Repair Materials. J. Spectrosc. 2017, 2017, 8736428. [Google Scholar] [CrossRef]
- Farlay, D.; Panczer, G.; Rey, C.; Delmas, P.D.; Boivin, G. Mineral maturity and crystallinity index are distinct characteristics of bone mineral. J. Bone Miner. Metab. 2010, 28, 433–445. [Google Scholar] [CrossRef] [PubMed]
- Sauer, G.; Zunic, W.; Durig, J.; Wuthier, R. Fourier transform Raman spectroscopy of synthetic and biological calcium phosphates. Calcif. Tissue Int. 1994, 54, 414–420. [Google Scholar] [CrossRef] [PubMed]
- Siboni, F.; Taddei, P.; Zamparini, F.; Prati, C.; Gandolfi, M.G. Properties of BioRoot RCS, a tricalcium silicate endodontic sealer modified with povidone and polycarboxylate. Int. Endod. J. 2017, 50, e120–e136. [Google Scholar] [CrossRef] [PubMed]
- Tesch, W.; Eidelman, N.; Roschger, P.; Goldenberg, F.; Klaushofer, K.; Fratzl, P. Graded microstructure and mechanical properties of human crown dentin. Calcif. Tissue Int. 2001, 69, 147–157. [Google Scholar] [CrossRef]
- Notingher, I.; Jones, J.; Verrier, S.; Bisson, I.; Embanga, P.; Edwards, P.; Polak, J.; Hench, L. Application of FTIR and Raman spectroscopy to characterisation of bioactive materials and living cells. J. Spectrosc. 2003, 17, 275–288. [Google Scholar] [CrossRef]
- Frost, R.L. Raman spectroscopic study of the magnesium carbonate mineral hydromagnesite (Mg5[(CO3)4(OH)2]·4H2O). J. Raman Spectrosc. 2011, 42, 1690–1694. [Google Scholar] [CrossRef]
- Gandolfi, M.G.; Taddei, P.; Siboni, F.; Modena, E.; Ginebra, M.P.; Prati, C. Fluoride-containing nanoporous calcium-silicate MTA cements for endodontics and oral surgery: Early fluorapatite formation in a phosphate-containing solution. Int. Endod. J. 2011, 44, 938–949. [Google Scholar] [CrossRef]
- Taddei, P.; Modena, E.; Tinti, A.; Siboni, F.; Prati, C.; Gandolfi, M.G. Vibrational investigation of calcium-silicate cements for endodontics in simulated body fluids. J. Mol. Struct. 2011, 993, 367–375. [Google Scholar] [CrossRef]
- Taddei, P.; Tinti, A.; Gandolfi, M.G.; Rossi, P.L.; Prati, C. Ageing of calcium silicate cements for endodontic use in simulated body fluids: A micro-Raman study. J. Raman Spectrosc. 2009, 40, 1858–1866. [Google Scholar] [CrossRef]
- Gandolfi, M.G.; Van Landuyt, K.; Taddei, P.; Modena, E.; Van Meerbeek, B.; Prati, C. Environmental scanning electron microscopy connected with energy dispersive X-ray analysis and Raman techniques to study ProRoot mineral trioxide aggregate and calcium silicate cements in wet conditions and in real time. J. Endod. 2010, 36, 851–857. [Google Scholar] [CrossRef] [PubMed]
- Turunen, M.J.; Saarakkala, S.; Rieppo, L.; Helminen, H.J.; Jurvelin, J.S.; Isaksson, H. Comparison between infrared and Raman spectroscopic analysis of maturing rabbit cortical bone. Appl. Spectrosc. 2011, 65, 595–603. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.S.; Khalid, H.; Sarfraz, Z.; Khan, M.; Iqbal, J.; Muhammad, N.; Fareed, M.A.; Rehman, I.U. Vibrational spectroscopy of selective dental restorative materials. Appl. Spectrosc. Rev. 2017, 52, 507–540. [Google Scholar] [CrossRef]
- Chen, Y.; Fierro, J.L.; Tanaka, T.; Wachs, I.E. Supported tantalum oxide catalysts: Synthesis, physical characterization, and methanol oxidation chemical probe reaction. J. Phys. Chem. B 2003, 107, 5243–5250. [Google Scholar] [CrossRef]
- Siboni, F.; Taddei, P.; Prati, C.; Gandolfi, M. Properties of Neo MTA Plus and MTA Plus cements for endodontics. Int. Endod. J. 2017, 50, e83–e94. [Google Scholar] [CrossRef]
- Timchenko, P.E.; Timchenko, E.V.; Pisareva, E.V.; Vlasov, M.Y.; Red’kin, N.A.; Frolov, O.O. Spectral Analysis of Allogeneic Hydroxyapatite Powders. J. Phys. Conf. Ser. 2017. [Google Scholar] [CrossRef]
- Ciobotă, V.; Salama, W.; Jentzsch, P.V.; Tarcea, N.; Rösch, P.; El Kammar, A.; Morsy, R.S.; Popp, J. Raman investigations of upper cretaceous phosphorite and black shale from Safaga District, Red Sea, Egypt. Spectrochim. Acta Part A: Mol. Biomol. Spectrosc. 2014, 118, 42–47. [Google Scholar] [CrossRef]
- Ribeiro, T.J.; Lima, O.J.; Faria, E.H.; Rocha, L.A.; Calefi, P.S.; Ciuffi, K.J.; Nassar, E.J. Sol-gel as methodology to obtain bioactive materials. Ann. Acad. Bras. Ciê 2014, 86, 27–36. [Google Scholar] [CrossRef]
- Singh, N.B.; Das, S.S.; Singh, N.P.; Dwivedi, V.N. Studies on SCLA composite Portland cement. Indian J. Eng. Mater. Sci. 2009, 16, 415–422. [Google Scholar]
- Gandolfi, M.G.; Taddei, P.; Modena, E.; Siboni, F.; Prati, C. Biointeractivity-related versus chemi/physisorption-related apatite precursor-forming ability of current root end filling materials. J. Biomed. Mater. Res. Part B: Appl. Biomater. 2013, 101, 1107–1123. [Google Scholar] [CrossRef] [PubMed]
- Berzina-Cimdina, L.; Borodajenko, N. Research of calcium phosphates using Fourier transform infrared spectroscopy. In Infrared Spectroscopy-Materials Science, Engineering and Technology; InTech: Rijeka, Croatia, 2012; pp. 251–263. [Google Scholar]
- Best, S.; Porter, A.; Thian, E.; Huang, J. Bioceramics: Past, present and for the future. J. Eur. Ceram. Soc. 2008, 28, 1319–1327. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abu Zeid, S.T.; Alamoudi, R.A.; Abou Neel, E.A.; Mokeem Saleh, A.A. Morphological and Spectroscopic Study of an Apatite Layer Induced by Fast-Set Versus Regular-Set EndoSequence Root Repair Materials. Materials 2019, 12, 3678. https://doi.org/10.3390/ma12223678
Abu Zeid ST, Alamoudi RA, Abou Neel EA, Mokeem Saleh AA. Morphological and Spectroscopic Study of an Apatite Layer Induced by Fast-Set Versus Regular-Set EndoSequence Root Repair Materials. Materials. 2019; 12(22):3678. https://doi.org/10.3390/ma12223678
Chicago/Turabian StyleAbu Zeid, Sawsan T., Ruaa A. Alamoudi, Ensanya A. Abou Neel, and Abeer A. Mokeem Saleh. 2019. "Morphological and Spectroscopic Study of an Apatite Layer Induced by Fast-Set Versus Regular-Set EndoSequence Root Repair Materials" Materials 12, no. 22: 3678. https://doi.org/10.3390/ma12223678
APA StyleAbu Zeid, S. T., Alamoudi, R. A., Abou Neel, E. A., & Mokeem Saleh, A. A. (2019). Morphological and Spectroscopic Study of an Apatite Layer Induced by Fast-Set Versus Regular-Set EndoSequence Root Repair Materials. Materials, 12(22), 3678. https://doi.org/10.3390/ma12223678



