Surface, Microstructural, and Mechanical Characterization of Prefabricated Pediatric Zirconia Crowns
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
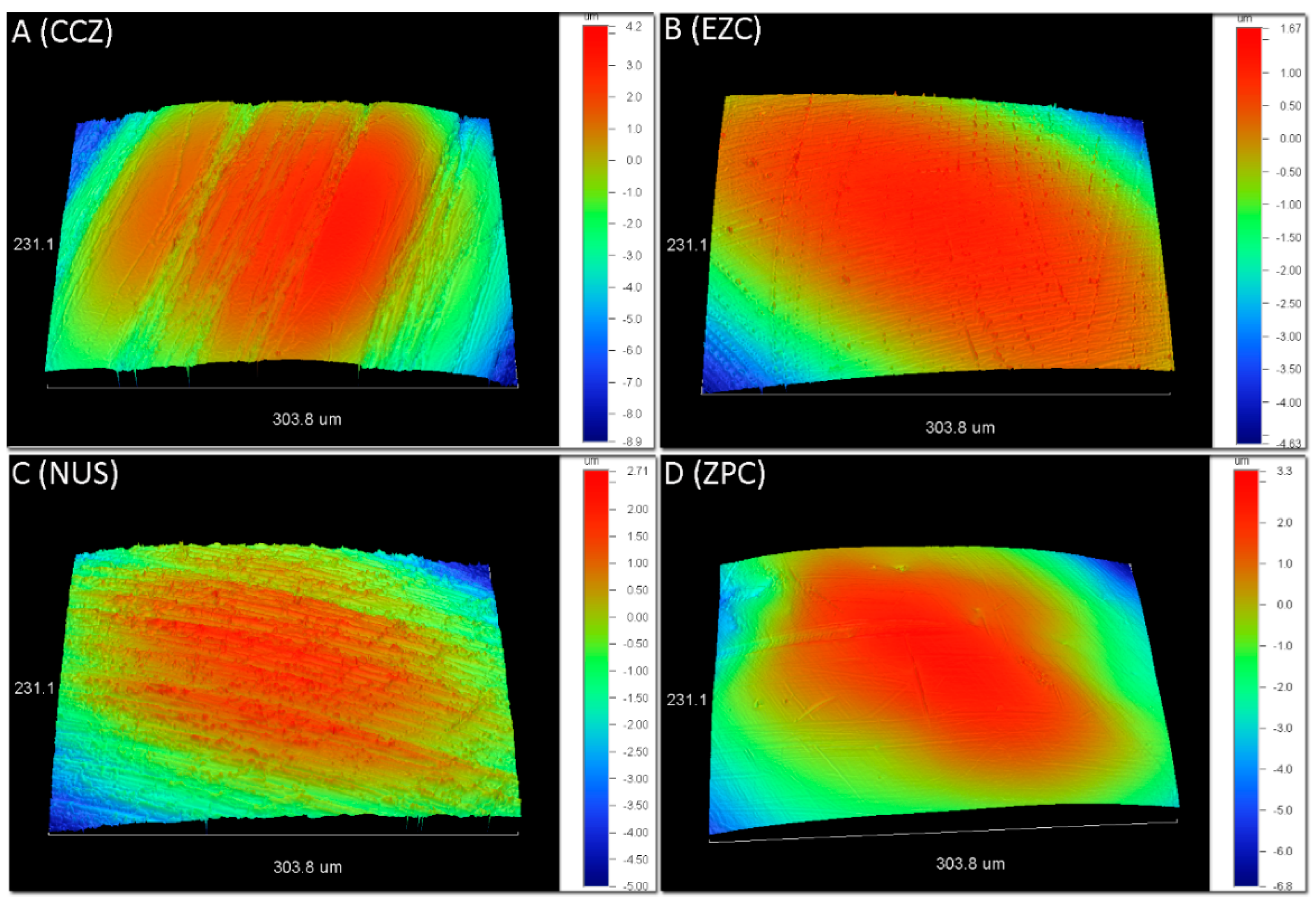
2.2. Optical Profilometry
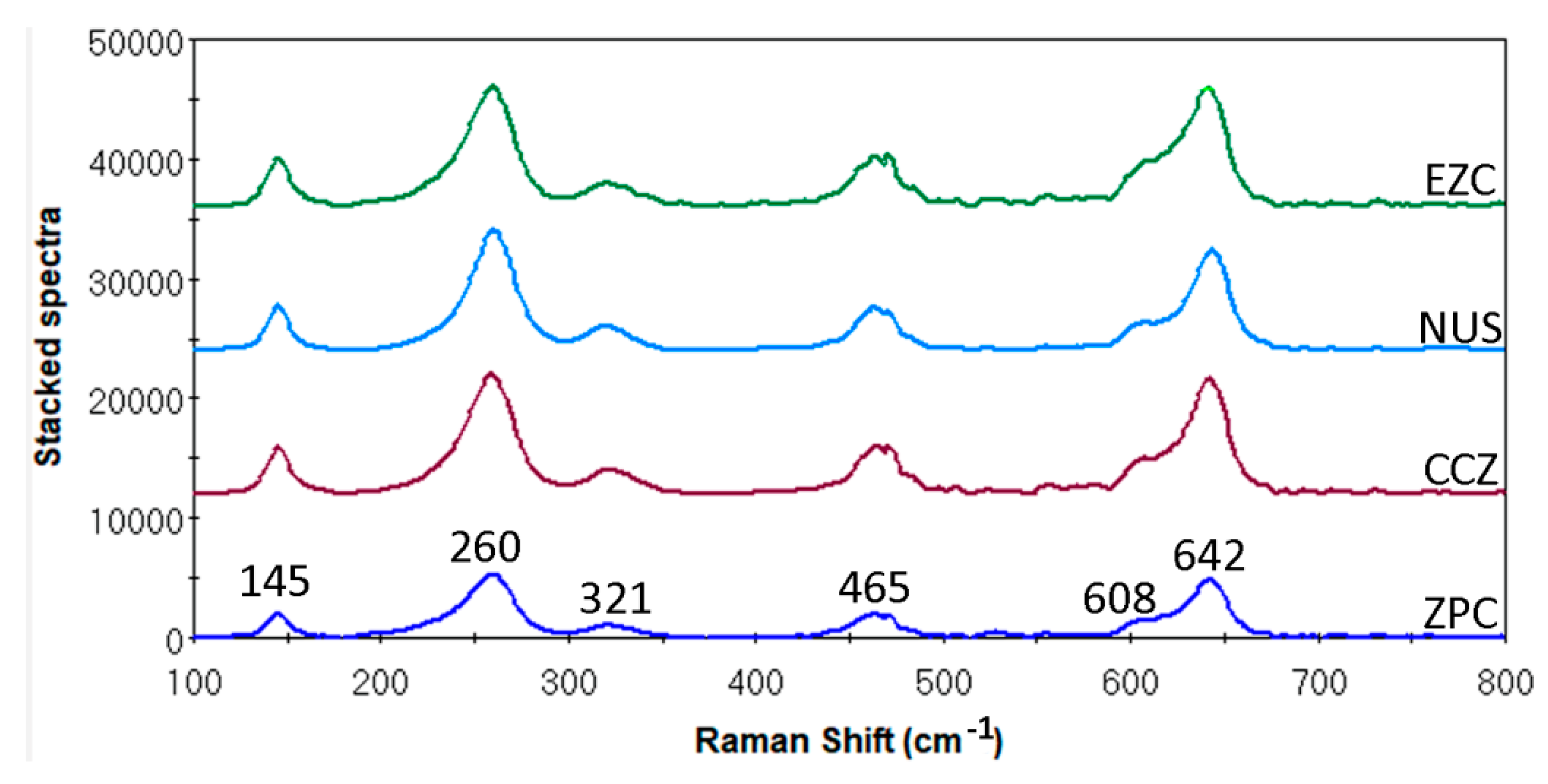
2.3. Raman Microscopy
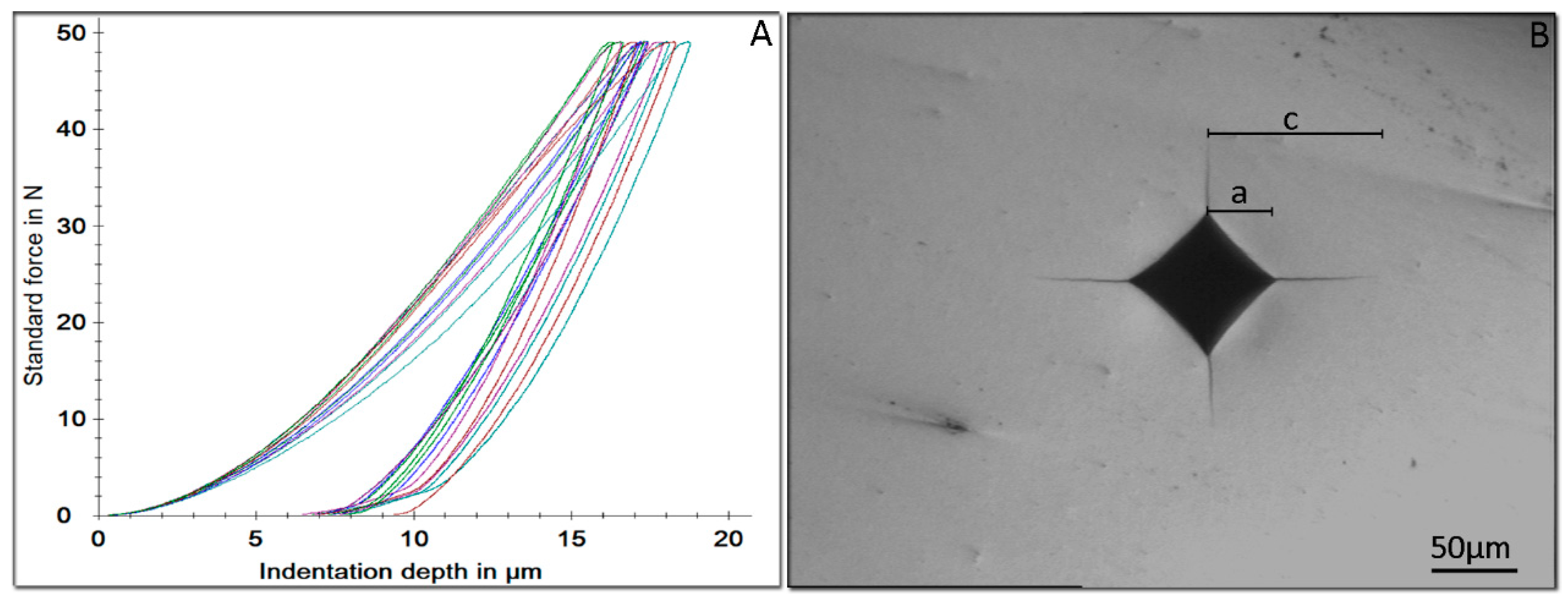
2.4. Instrumented Indentation Testing (IIT) and Fracture Toughness
2.5. Statistical Analysis
3. Results
3.1. Optical Profilometry
3.2. Raman Microscopy
3.3. Instrumented Indentation Testing (IIT) and Fracture Toughness
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- De Grauwe, A.; Aps, J.K.; Martens, L.C. Early Childhood Caries (ECC): what’s in a name? Eur. J. Paediatr. Dent. 2004, 5, 62–70. [Google Scholar] [PubMed]
- Meyer, F.; Enax, J. Early Childhood Caries: Epidemiology, Aetiology, and Prevention. Int. J. Dent. 2018, 2018, 1–7. [Google Scholar] [CrossRef] [PubMed]
- López-Gómez, S.A.; Villalobos-Rodelo, J.J.; Ávila-Burgos, L.; Casanova-Rosado, J.F.; Vallejos-Sánchez, A.A.; Lucas-Rincón, S.E.; Patiño-Marin, N.; Medina-Solís, C.E. Relationship between premature loss of primary teeth with oral hygiene, consumption of soft drinks, dental care and previous caries experience. Sci. Rep. 2016, 6, 21147. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Jorge, J.; Alencar, B.M.; Pordeus, I.A.; Soares, M.E.; Marques, L.S.; Ramos-Jorge, M.L.; Paiva, S.M. Impact of dental caries on quality of life among preschool children: Emphasis on the type of tooth and stages of progression. Eur. J. Oral Sci. 2015, 123, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Attari, N.; Roberts, J.F. Restoration of Primary Teeth with crowns: A systematic review of the literature. Eur. Arch. Paediatr. Dent. 2006, 1, 58–62. [Google Scholar] [CrossRef]
- Belduz Kara, N.; Yilmaz, Y. Assessment of oral hygiene and periodontal health around posterior primary molars after their restoration with various crown types. Int. J. Paediatr. Dent. 2014, 24, 303–313. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, J.A.; Feigal, R.J.; Till, M.J.; Hodges, J.S. Parental attitudes on restorative materials as factors influencing current use in pediatric dentistry. Pediatr. Dent. 2009, 31, 63–70. [Google Scholar] [PubMed]
- Manicone, P.F.; Iommetti, P.R.; Raffaelli, L. An overview of zirconia ceramics: Basic properties and clinical applications. J. Dent. 2007, 35, 819–826. [Google Scholar] [CrossRef] [PubMed]
- Piconi, C.; Maccauro, G. Zirconia as a ceramic biomaterial. Biomaterials 1999, 20, 1–25. [Google Scholar] [CrossRef]
- Aiem, E.; Smail-Faugeron, V.; Muller-Bolla, M. Aesthetic preformed paediatric crowns: Systematic review. Int. J. Paediatr. Dent. 2017, 27, 273–282. [Google Scholar] [CrossRef] [PubMed]
- Ram, D.; Fuks, A.B.; Eidelman, E. Long-term clinical performance of esthetic primary molar crowns. Pediatr. Dent. 2003, 25, 582–584. [Google Scholar] [PubMed]
- Salami, A.; Walia, T.; Bashiri, R. Comparison of Parental Satisfaction with Three Tooth-Colored Full-Coronal Restorations in Primary Maxillary Incisors. J. Clin. Pediatr. Dent. 2015, 39, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Walia, T.; Salami, A.A.; Bashiri, R.; Hamoodi, O.M.; Rashid, F. A randomised controlled trial of three aesthetic full-coronal restorations in primary maxillary teeth. Eur. J. Paediatr. Dent. 2014, 15, 113–118. [Google Scholar] [PubMed]
- Ashima, G.; Sarabjot, K.B.; Gauba, K.; Mittal, H.C. Zirconia crowns for rehabilitation of decayed primary incisors: An esthetic alternative. J. Clin. Pediatr. Dent. 2014, 39, 18–22. [Google Scholar] [CrossRef]
- Holsinger, D.M.; Wells, M.H.; Scarbecz, M.; Donaldson, M. Clinical Evaluation and Parental Satisfaction with Pediatric Zirconia Anterior Crowns. Pediatr. Dent. 2016, 38, 192–197. [Google Scholar] [PubMed]
- Taran, P.K.; Kaya, M.S. A comparison of periodontal health in primary molars restored with prefabricated stainless steel and zirconia crowns. Pediatr. Dent. 2018, 40, 334–339. [Google Scholar]
- Lee, H.; Chae, Y.K.; Lee, H.S.; Choi, S.C.; Nam, O.H. Three-Dimensional Digitalized Surface and Volumetric Analysis of Posterior Prefabricated Zirconia Crowns for Children. J. Clin. Pediatr. Dent. 2019, 43, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Clark, L.; Wells, M.H.; Harris, E.F.; Lou, J. Comparison of Amount of Primary Tooth Reduction Required for Anterior and Posterior Zirconia and Stainless Steel Crowns. Pediatr. Dent. 2016, 38, 42–46. [Google Scholar]
- Karimipour-Saryazdi, M.; Sadid-Zadeh, R.; Givan, D.; Burgess, J.O.; Ramp, L.C.; Liu, P.-R. Influence of surface treatment of yttrium-stabilized tetragonal zirconium oxides and cement type on crown retention after artificial aging. J. Prosthet. Dent. 2014, 111, 395–403. [Google Scholar] [CrossRef]
- Amaral, R.; Özcan, M.; Valandro, L.F.; Balducci, I.; Bottino, M.A. Effect of conditioning methods on the microtensile bond strength of phosphate monomer-based cement on zirconia ceramic in dry and aged conditions. J. Biomed. Mater. Res. Part B Appl. Biomater. 2008, 85, 1–9. [Google Scholar] [CrossRef]
- Kelly, J.R.; Denry, I. Stabilized zirconia as a structural ceramic: An overview. Dent. Mater. 2008, 24, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Sundh, A.; Sjögren, G. Fracture resistance of all-ceramic zirconia bridges with differing phase stabilizers and quality of sintering. Dent. Mater. 2006, 22, 778–784. [Google Scholar] [CrossRef] [PubMed]
- Stepp, P.; Morrow, B.R.; Wells, M.; Tipton, D.A.; Garcia-Godoy, F. Microleakage of cements in prefabricated zirconia crowns. Pediatr. Dent. 2018, 40, 136–139. [Google Scholar]
- Theriot, A.L.; Frey, G.N.; Ontiveros, J.C.; Badger, G. Gloss and surface roughness of anterior pediatric zirconia crowns. J. Dent. Child. (Chic.) 2017, 84, 115–119. [Google Scholar]
- Al Shobber, M.Z.; Alkhadra, T.A. Fracture resistance of different primary anterior esthetic crowns. Saudi Dent. J. 2017, 29, 179–184. [Google Scholar] [CrossRef] [PubMed]
- ISO14577-1. Metallic Materials—Instrumented Indentation Test for Hardness and Materials Parameters; International Organization for Standardization: Geneva, Switzerland, 2002. [Google Scholar]
- Guden, M.; Şakar-Deliormanli, A.; Şakar-Deliormanli, A. Microhardness and fracture toughness of dental materials by indentation method. J. Biomed. Mater. Res. Part B Appl. Biomater. 2006, 76, 257–264. [Google Scholar]
- Walia, T.; Brigi, C.; KhirAllah, A. Comparative evaluation of surface roughness of posterior primary zirconia crowns. Eur. Arch. Paediatr. Dent. 2019, 20, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Oh, W.S.; Delong, R.; Anusavice, K.J. Factors affecting enamel and ceramic wear: A literature review. J. Prosthet. Dent. 2002, 87, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Chevalier, J.; Gremillard, L.; Deville, S. Low-Temperature Degradation of Zirconia and Implications for Biomedical Implants. Annu. Rev. Mater. Res. 2007, 37, 1–32. [Google Scholar] [CrossRef]
- Wulfman, C.; Sadoun, M.; De La Chapelle, M.L. Interest of Raman spectroscopy for the study of dental material: The zirconia material example. IRBM 2010, 31, 257–262. [Google Scholar] [CrossRef]
- Zhuang, Y.X.; Zhu, Z.Y.; Jiao, T.; Sun, J. Effect of aging time and thickness on low-temperature degradation of dental zirconia. J. Prosthodont. 2019, 28, E404–E410. [Google Scholar] [CrossRef] [PubMed]
- Coric, D.; Renjo, M.M.; Ćurković, L. Vickers indentation fracture toughness of Y-TZP dental ceramics. Int. J. Refract. Met. Hard Mater. 2017, 64, 14–19. [Google Scholar] [CrossRef]
- Suresh, S.; Giannakopoulos, A. A new method for estimating residual stresses by instrumented sharp indentation. Acta Mater. 1998, 46, 5755–5767. [Google Scholar] [CrossRef]
- Shahdad, S.A.; McCabe, J.F.; Bull, S.; Rusby, S.; Wassell, R.W. Hardness measured with traditional Vickers and Martens hardness methods. Dent. Mater. 2007, 23, 1079–1085. [Google Scholar] [CrossRef] [PubMed]
- Turp, V.; Tuncelli, B.; Sen, D.; Goller, G. Evaluation of hardness and fracture toughness, coupled with microstructural analysis, of zirconia ceramics stored in environments with different pH values. Dent. Mater. J. 2012, 31, 891–902. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.S.; Lee, J.W.; Choi, Y.J.; Ahn, J.S.; Shin, S.W.; Huh, J.B. A study on the in-vitro wear of the natural tooth structure by opposing zirconia or dental porcelain. J. Adv. Prosthodont. 2010, 2, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Bramanti, E.; Cervino, G.; Lauritano, F.; Fiorillo, L.; D’Amico, C.; Sambataro, S.; Denaro, D.; Famà, F.; Ierardo, G.; Polimeni, A.; et al. FEM and Von Mises Analysis on Prosthetic Crowns Structural Elements: Evaluation of Different Applied Materials. Sci. World J. 2017, 2017, 1–7. [Google Scholar] [CrossRef]
| Brand Name | Manufacturer | Code |
|---|---|---|
| Cheng Crowns Zirconia | Cheng Crowns Exton, PA, USA | CCZ |
| EZCrowns | Sprig Suite L Loomis, CA, USA | EZC |
| NuSmile ZR | NuSmile, Houston, TX, USA | NUS |
| Zirconia Pediatric Crowns | Kinder Krowns, St Louis Park, MN, USA | ZPC |
| Group | Sa (μm) | Sq (μm) | Sz (μm) | Sc (μm3/μm2) | Sv (nm3/nm2) |
|---|---|---|---|---|---|
| CCZ | 2.7 (0.7) a | 3.4 (1.0) a | 18.7 (6.5) a | 3.1 (0.6) a | 563 (184) a |
| EZC | 1.1 (0.2) b | 1.4 (0.3) b | 8.2 (1.4) b | 1.3 (0.1) b | 265 (62) b |
| NUS | 1.7 (0.5) ab | 2.2 (0.7) ab | 11.3 (2.6) ab | 2.2 (0.7) ab | 265 (50) ab |
| ZPC | 1.6 (1.0) ab | 1.9 (1.2) ab | 9.6 (6.2) ab | 1.7 (0.8) b | 188 (53) b |
| Material | HM (N/mm2) | EIT (GPa) | nIT (%) | HV5 | KIC (MPa m1/2) |
|---|---|---|---|---|---|
| CCZ | 6653 (635) a | 123.1 (15.7) a | 52.0 (4.5) a | 1365 (20) a | 6.1 (0.3) a |
| EZC | 6390 (584) a | 118.1 (12.4) a | 54.4 (1.6) a | 1347 (12) a | 4.7 (0.3) b |
| NUS | 6142 (430) a | 114.3 (13.9) a | 54.9 (5.3) a | 1305 (18) b | 8.0 (1.8) a |
| ZPC | 6318 (402) a | 114.2 (13.7) a | 53.3 (2.9) a | 1325 (14) b | 6.0 (0.5) a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Diener, V.; Polychronis, G.; Erb, J.; Zinelis, S.; Eliades, T. Surface, Microstructural, and Mechanical Characterization of Prefabricated Pediatric Zirconia Crowns. Materials 2019, 12, 3280. https://doi.org/10.3390/ma12203280
Diener V, Polychronis G, Erb J, Zinelis S, Eliades T. Surface, Microstructural, and Mechanical Characterization of Prefabricated Pediatric Zirconia Crowns. Materials. 2019; 12(20):3280. https://doi.org/10.3390/ma12203280
Chicago/Turabian StyleDiener, Valeria, Georgrios Polychronis, Juliane Erb, Spiros Zinelis, and Theodore Eliades. 2019. "Surface, Microstructural, and Mechanical Characterization of Prefabricated Pediatric Zirconia Crowns" Materials 12, no. 20: 3280. https://doi.org/10.3390/ma12203280
APA StyleDiener, V., Polychronis, G., Erb, J., Zinelis, S., & Eliades, T. (2019). Surface, Microstructural, and Mechanical Characterization of Prefabricated Pediatric Zirconia Crowns. Materials, 12(20), 3280. https://doi.org/10.3390/ma12203280







