Biocompatibility of Plasma-Treated Polymeric Implants
Abstract
1. Introduction
2. Biomaterials
2.1. Artificial Vascular Grafts
2.2. Biocompatibility of Synthetic Materials
2.3. Factors That Influence the Biocompatibility of Biomaterials
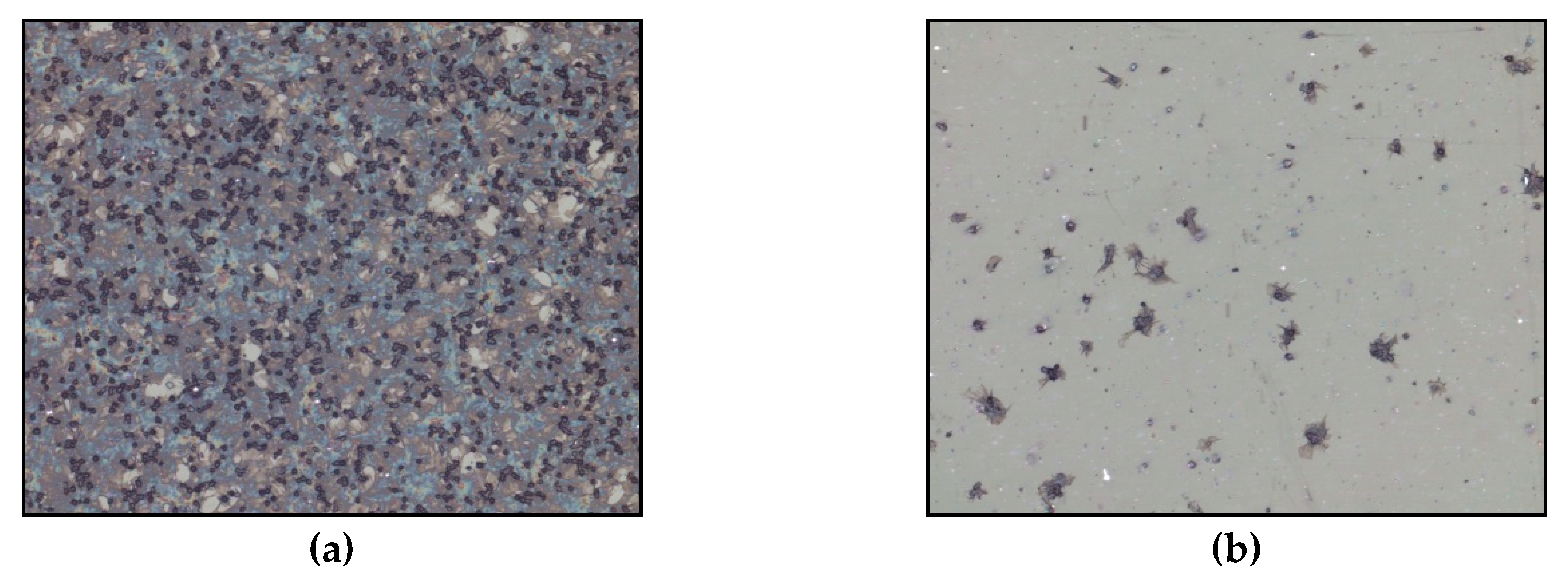
2.3.1. Impact of Plasma Treatment on the Hemo- and Biocompatibility of Synthetic Materials
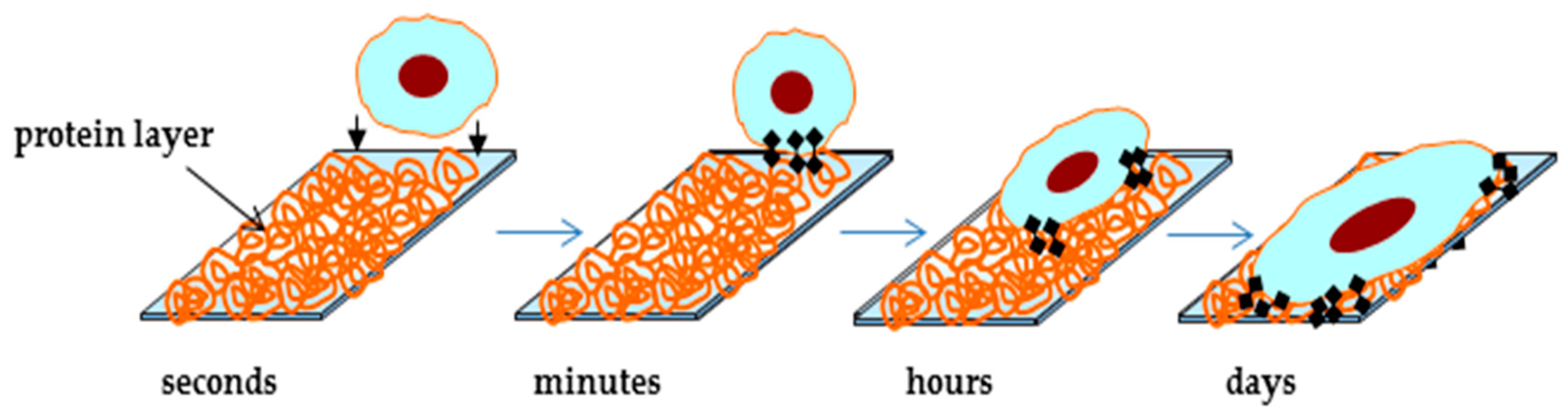
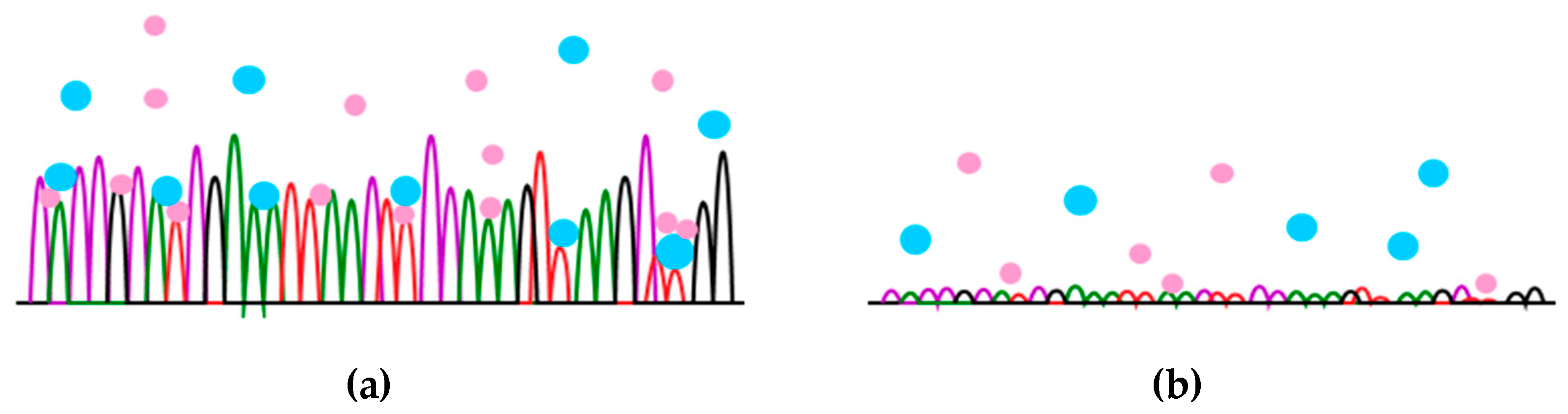
2.3.2. The Effect of the Surface Chemical Composition on the Hemocompatibility of Biomaterials and on Cell Response
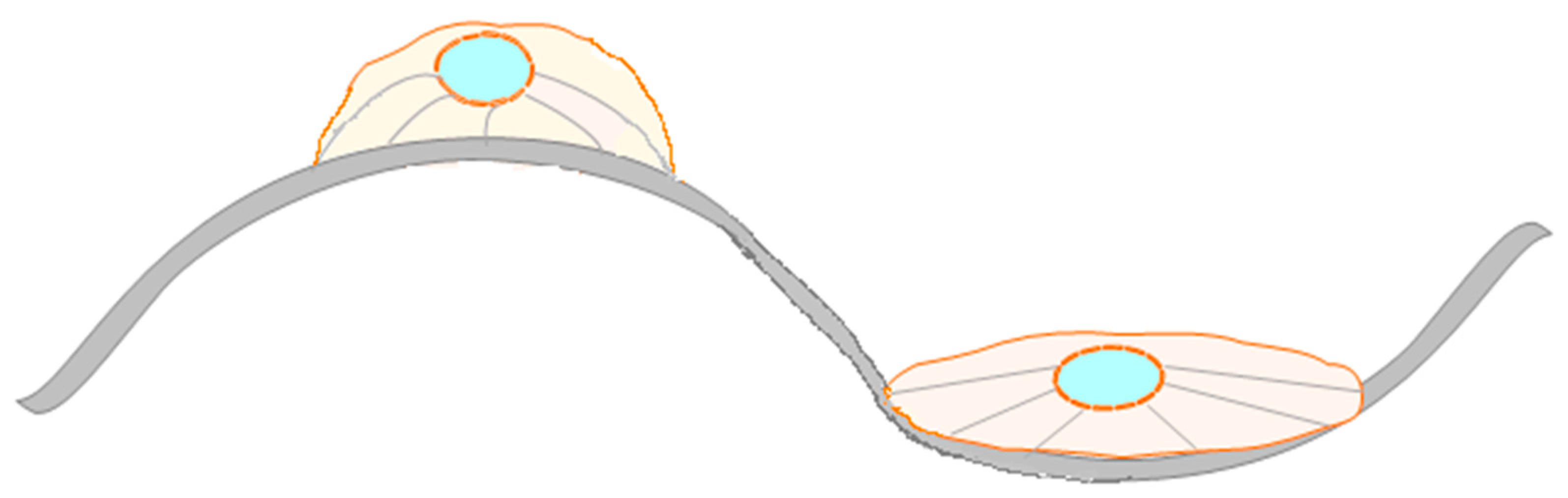
2.3.3. The Influence of the Topography on the Hemocompatibility of Biomaterials and on Cell Response
2.3.4. The Effect of Wettability on the Hemocompatibility of Biomaterials and on Cell Response
3. Biomaterial-Blood Interactions
4. Methods for Improving the Biocompatibility of Synthetic Materials
4.1. Plasma Treatment of Polymers
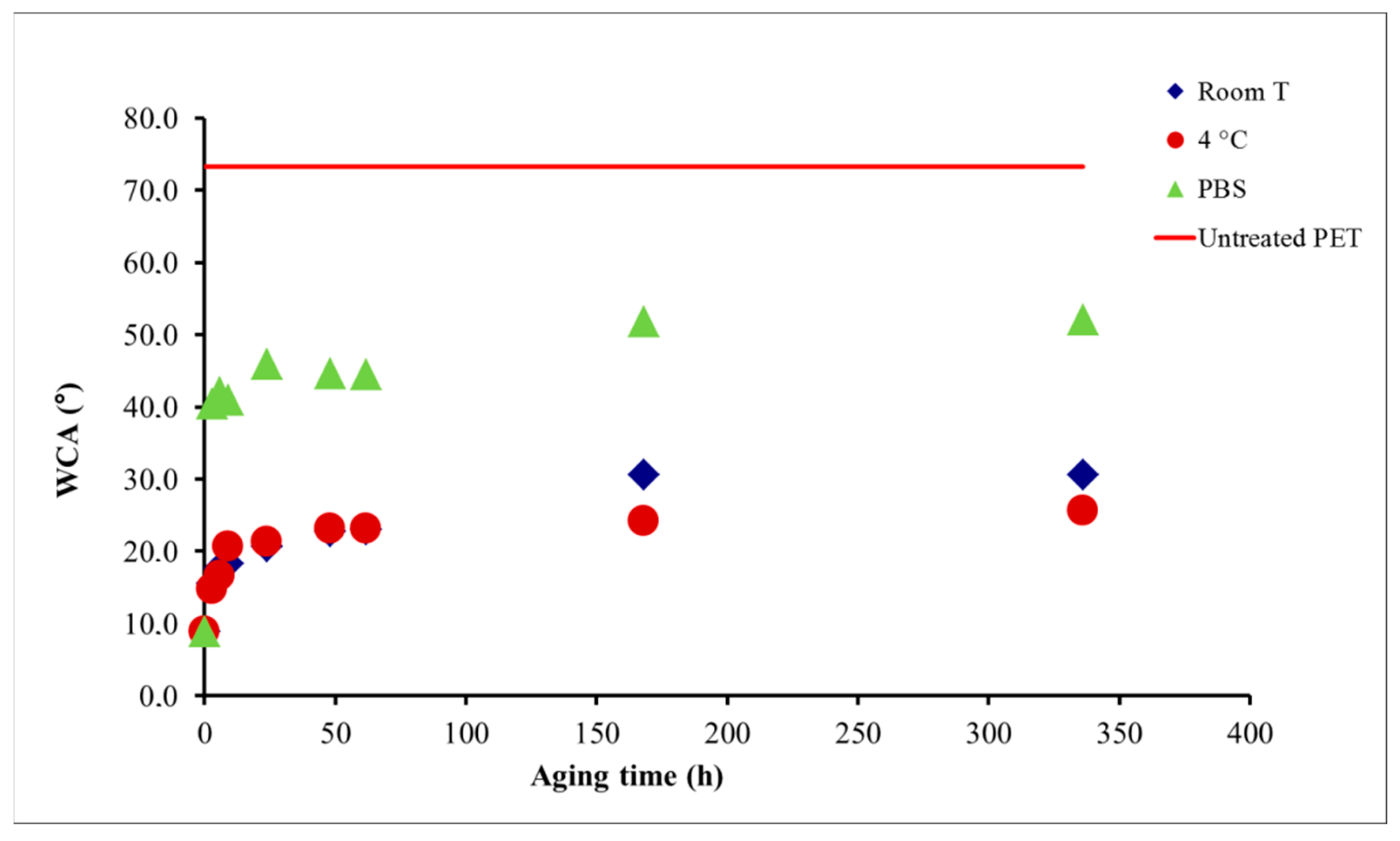
4.1.1. Aging of Plasma-Treated Materials
- Reorientation and relocation of polar groups from the surface of the polymer into the bulk of the material due to thermodynamic relaxation,
- Diffusion of low molecular weight oligomers from the interior to the surface and products that are formed during plasma treatment on the surface of polymers,
- Reactions of free radicals and other active species and groups formed during treatment, with each other and with the environment in which the polymer is located.
5. Conclusions and Future Perspectives
Funding
Acknowledgments
Conflicts of Interest
References
- Allender, S.; Foster, C.; Hutchinson, L.; Arambepola, C. Quantification of urbanization in relation to chronic diseases in developing countries: A systematic review. J. Urban Health 2008, 85, 938–951. [Google Scholar] [CrossRef] [PubMed]
- Helmus, M.N.; Hubbell, J.A. Materials selection. Cardiovasc. Pathol. 1993, 2, 53–71. [Google Scholar] [CrossRef]
- Tsuruta, T.; Hayashi, T.; Kataoka, K.; Ishihara, K.; Kimura, Y. Biomedical Applications of Polymeric Materials; CRC Press: Boca Raton, FL, USA, 1993; Volume 340. [Google Scholar]
- Jaganathan, S.K.; Supriyanto, E.; Murugesan, S.; Balaji, A.; Asokan, M.K. Biomaterials in cardiovascular research: applications and clinical implications. BioMed Res. Int. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Roald, H.; Barstad, R.; Bakken, I.; Roald, B.; Lyberg, T.; Sakariassen, K. Initial interactions of platelets and plasma proteins in flowing non-anticoagulated human blood with the artificial surfaces Dacron and PTFE. Blood Coagul. Fibrinol. Int. J. Haemost. Thromb. 1994, 5, 355–363. [Google Scholar]
- Ormiston, J.A.; Serruys, P.W.; Regar, E.; Dudek, D.; Thuesen, L.; Webster, M.W.; Onuma, Y.; Garcia-Garcia, H.M.; McGreevy, R.; Veldhof, S. A bioabsorbable everolimus-eluting coronary stent system for patients with single de-novo coronary artery lesions (ABSORB): A prospective open-label trial. Lancet 2008, 371, 899–907. [Google Scholar] [CrossRef]
- Serruys, P.W.; Ormiston, J.A.; Onuma, Y.; Regar, E.; Gonzalo, N.; Garcia-Garcia, H.M.; Nieman, K.; Bruining, N.; Dorange, C.; Miquel-Hébert, K. A bioabsorbable everolimus-eluting coronary stent system (ABSORB): 2-year outcomes and results from multiple imaging methods. Lancet 2009, 373, 897–910. [Google Scholar] [CrossRef]
- Damman, P.; Iñiguez, A.; Klomp, M.; Beijk, M.; Woudstra, P.; Silber, S.; Ribeiro, E.E.; Suryapranata, H.; Sim, K.H.; Tijssen, J.G. Coronary Stenting With the GenousTM Bio-Engineered R StentTM in Elderly Patients. Circ. J. 2011, 75, 2590–2597. [Google Scholar] [CrossRef]
- Herring, M.; Gardner, A.; Glover, J. A single-staged technique for seeding vascular grafts with autogenous endothelium. Surgery 1978, 84, 498–504. [Google Scholar]
- Bruck, S.D. Interactions of synthetic and natural surfaces with blood in the physiological environment. J. Biomed. Mater. Res. 1977, 11, 1–21. [Google Scholar] [CrossRef]
- Chan, C.-M.; Ko, T.-M.; Hiraoka, H. Polymer surface modification by plasmas and photons. Surf. Sci. rep. 1996, 24, 1–54. [Google Scholar] [CrossRef]
- Grunkemeier, J.; Tsai, W.; Horbett, T. Hemocompatibility of treated polystyrene substrates: Contact activation, platelet adhesion, and procoagulant activity of adherent platelets. J. Biomed. Mater. Res. Off. J. Soc. Biomater. Jpn. Soc. Biomater. Aust. Soc. Biomater. 1998, 41, 657–670. [Google Scholar] [CrossRef]
- Ratner, B.D.; Hoffman, A.S.; Schoen, F.J.; Lemons, J.E. Biomaterials Science: An Introduction to Materials in Medicine; Elsevier: Amsterdam, The Netherlands, 2004. [Google Scholar]
- Junkar, I.; Cvelbar, U.; Vesel, A.; Hauptman, N.; Mozetič, M. The Role of Crystallinity on Polymer Interaction with Oxygen Plasma. Plasma Process. Polym. 2009, 6, 667–675. [Google Scholar] [CrossRef]
- Cui, H.; Sinko, P. The role of crystallinity on differential attachment/proliferation of osteoblasts and fibroblasts on poly (caprolactone-co-glycolide) polymeric surfaces. Front. Mater. Sci. 2012, 6, 47–59. [Google Scholar] [CrossRef]
- Vesel, A. Hydrophobization of polymer polystirene in fluorine plasma. Mater. Tehnol. 2011, 45, 217–220. [Google Scholar]
- Vesel, A.; Junkar, I.; Cvelbar, U.; Kovac, J.; Mozetic, M. Surface modification of polyester by oxygen- and nitrogen-plasma treatment. Surf. Interface Anal. 2008, 40, 1444–1453. [Google Scholar] [CrossRef]
- Di Mundo, R.; Palumbo, F.; d’Agostino, R. Influence of chemistry on wetting dynamics of nanotextured hydrophobic surfaces. Langmuir 2010, 26, 5196–5201. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Ji, J.; Zhang, W.; Wang, W.; Zhang, Y.; Wu, Z.; Zhang, Y.; Chu, P.K. Rat calvaria osteoblast behavior and antibacterial properties of O2 and N2 plasma-implanted biodegradable poly(butylene succinate). Acta Biomater. 2010, 6, 154–159. [Google Scholar] [CrossRef]
- John, M.J.; Thomas, S. Biofibres and biocomposites. Carbohydr. Polym. 2008, 71, 343–364. [Google Scholar] [CrossRef]
- Dolenc, A.; Homar, M.; Gašperlin, M.; Kristl, J. Z nanooblaganjem do izboljšanja biokompatibilnosti vsadkov. Medicinski Razgledi 2006, 4, 411–420. [Google Scholar]
- Lemons, J.; Ratner, B.; Hoffman, A.; Schoen, F. Biomaterials Science: An Introduction to Materials in Medicine; Academic Press: San Diego, CA, USA, 1996. [Google Scholar]
- Kerdjoudj, H.; Berthelemy, N.; Rinckenbach, S.; Kearney-Schwartz, A.; Montagne, K.; Schaaf, P.; Lacolley, P.; Stoltz, J.-F.; Voegel, J.-C.; Menu, P. Small vessel replacement by human umbilical arteries with polyelectrolyte film-treated arteries: In vivo behavior. J. Am. Coll. Cardiol. 2008, 52, 1589–1597. [Google Scholar] [CrossRef]
- Moris, D.; Sigala, F.; Georgopoulos, S.; Bramis, I. The choice of the appropriate graft in the treatment of vascular diseases. Hellenic J. Surg. 2010, 82, 274–283. [Google Scholar] [CrossRef]
- Nunn, D.B.; Freeman, M.H.; Hudgins, P.C. Postoperative alterations in size of Dacron aortic grafts: An ultrasonic evaluation. Ann. Surg. 1979, 189, 741. [Google Scholar] [CrossRef] [PubMed]
- Drury, J.K.; Ashton, T.R.; Cunningham, J.D.; Maini, R.; Pollock, J.G. Experimental and clinical experience with a gelatin impregnated Dacron prosthesis. Ann. Vasc. Surg. 1987, 1, 542–547. [Google Scholar] [CrossRef]
- Guidoin, R.; Snyder, R.; Martin, L.; Botzko, K.; Marois, M.; Awad, J.; King, M.; Domurado, D.; Bedros, M.; Gosselin, C. Albumin Coating of a Knitted Polyester Arterial Prosthesis: An Alternative to Preclotting. Ann. Thorac. Surg. 1984, 37, 457–465. [Google Scholar] [CrossRef]
- Kito, H.; Matsuda, T. Biocompatible coatings for luminal and outer surfaces of small-caliber artificial grafts. J. Biomed. Mater. Res. 1996, 30, 321–330. [Google Scholar] [CrossRef]
- Callow, A.D. Problems in the Construction of A Small Diameter Graft. Int. Angiol. 1988, 7, 246–253. [Google Scholar]
- Charpentier, P.A.; Maguire, A.; Wan, W.K. Surface modification of polyester to produce a bacterial cellulose-based vascular prosthetic device. Appl. Surf. Sci. 2006, 252, 6360–6367. [Google Scholar] [CrossRef]
- Kolar, M.; Mozetič, M.; Stana-Kleinschek, K.; Fröhlich, M.; Turk, B.; Vesel, A. Covalent binding of heparin to functionalized PET materials for improved haemocompatibility. Materials 2015, 8, 1526–1544. [Google Scholar] [CrossRef]
- Doliška, A.; Ribitsch, V.; Stana Kleinschek, K.; Strnad, S. Viscoelastic properties of fibrinogen adsorbed onto poly(ethylene terephthalate) surfaces by QCM-D. Carbohydr. Polym. 2013, 93, 246–255. [Google Scholar] [CrossRef]
- Gorbet, M.B.; Sefton, M.V. Biomaterial-associated thrombosis: Roles of coagulation factors, complement, platelets and leukocytes. Biomaterials 2004, 25, 5681–5703. [Google Scholar] [CrossRef]
- Otto, M.; Franzen, A.; Hansen, T.; Kirkpatrick, C.J. Modification of human platelet adhesion on biomaterial surfaces by protein preadsorption under static and flow conditions. J. Mater. Sci. Mater. Med. 2004, 15, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Ratner, B.D. The catastrophe revisited: Blood compatibility in the 21st century. Biomaterials 2007, 28, 5144–5147. [Google Scholar] [CrossRef] [PubMed]
- Rihova, B. Biocompatibility of biomaterials: Hemocompatibility, immunocompatibility and biocompatibility of solid polymeric materials and soluble targetable polymeric carriers. Adv. Drug Deliv. Rev. 1996, 2, 157–176. [Google Scholar] [CrossRef]
- Ratner, B.D. The blood compatibility catastrophe. J. Biomed. Mater. Res. 1993, 27, 283–287. [Google Scholar] [CrossRef] [PubMed]
- Ninan, N.; Muthiah, M.; Park, I.-K.; Elain, A.; Wong, T.W.; Thomas, S.; Grohens, Y. Faujasites Incorporated Tissue Engineering Scaffolds for Wound Healing: In Vitro and In Vivo Analysis. ACS Appl. Mater. Interfaces 2013, 5, 11194–11206. [Google Scholar] [CrossRef] [PubMed]
- Ghosal, K.; Latha, M.S.; Thomas, S. Poly(ester amides) (PEAs)—Scaffold for tissue engineering applications. Eur. Polym. J. 2014, 60, 58–68. [Google Scholar] [CrossRef]
- Garcia, J.L.; Asadinezhad, A.; Pachernik, J.; Lehocky, M.; Junkar, I.; Humpolicek, P.; Saha, P.; Valasek, P. Cell Proliferation of HaCaT Keratinocytes on Collagen Films Modified by Argon Plasma Treatment. Molecules 2010, 15, 2845–2856. [Google Scholar] [CrossRef] [PubMed]
- Asadinezhad, A.; Novák, I.; Lehocký, M.; Bílek, F.; Vesel, A.; Junkar, I.; Sáha, P.; Popelka, A. Polysaccharides Coatings on Medical-Grade PVC: A Probe into Surface Characteristics and the Extent of Bacterial Adhesion. Molecules 2010, 15, 1007–1027. [Google Scholar] [CrossRef] [PubMed]
- Modic, M.; Junkar, I.; Vesel, A.; Mozetic, M. Aging of plasma treated surfaces and their effects on platelet adhesion and activation. Surface Coat. Technol. 2012, 213, 98–104. [Google Scholar] [CrossRef]
- Junkar, I.; Cvelbar, U.; Lehocky, M. Plasma treatment of biomedical materials. Mater. Tehnol. 2011, 45, 221–226. [Google Scholar]
- Uroš, C.; Ita, J.; Martina, M. Hemocompatible Poly(ethylene terephthalate) Polymer Modified via Reactive Plasma Treatment. Jpn. J. Appl. Phys. 2011, 50, 08JF02. [Google Scholar]
- Vesel, A.; Mozetic, M.; Jaganjac, M.; Milkovic, L.; Cipak, A.; Zarkovic, N. Biocompatibility of oxygen-plasma-treated polystyrene substrates. Eur. Phys. J. Appl. Phys. 2011, 56, 24024. [Google Scholar] [CrossRef]
- Jaganjac, M.; Milkovic, L.; Cipak, A.; Mozetic, M.; Recek, N.; Zarkovic, N.; Vesel, A. Cell adhesion on hydrophobic polymer surfaces. Mater. Tehnol. 2012, 46, 53–56. [Google Scholar]
- Recek, N.; Mozetič, M.; Jaganjac, M.; Milkovič, L.; Žarkovic, N.; Vesel, A. Improved proliferation of human osteosarcoma cells on oxygen plasma treated polystyrene. Vacuum 2013, 98, 116–121. [Google Scholar] [CrossRef]
- Recek, N.; Mozetic, M.; Jaganjac, M.; Milkovic, L.; Zarkovic, N.; Vesel, A. Adsorption of Proteins and Cell Adhesion to Plasma Treated Polymer Substrates. Int. J. Polym. Mater. Polym. Biomater. 2014, 63, 685–691. [Google Scholar] [CrossRef]
- Jaganjac, M.; Vesel, A.; Milkovic, L.; Recek, N.; Kolar, M.; Zarkovic, N.; Latiff, A.; Kleinschek, K.-S.; Mozetic, M. Oxygen-rich coating promotes binding of proteins and endothelialization of polyethylene terephthalate polymers. J. Biomed. Mater. Res. Part A 2014, 102, 2305–2314. [Google Scholar] [CrossRef] [PubMed]
- Recek, N.; Vesel, A.; Mozetic, M.; Jaganjac, M.; Milkovic, L.; Žarković, N. Influence of polymer surface on cell proliferation and cell oxidation momeostatis. In Proceedings of the ICPM5, 5th International Conference on Plasma Medicine, Nara, Japan, 18–23 May 2014. [Google Scholar]
- Seifert, B.; Mihanetzis, G.; Groth, T.; Albrecht, W.; Richau, K.; Missirlis, Y.; Paul, D.; von Sengbusch, G. Polyetherimide: A new membrane-forming polymer for biomedical applications. Artif. Organs 2002, 26, 189–199. [Google Scholar] [CrossRef]
- Sperling, C.; Schweiss, R.B.; Streller, U.; Werner, C. In vitro hemocompatibility of self-assembled monolayers displaying various functional groups. Biomaterials 2005, 26, 6547–6557. [Google Scholar] [CrossRef]
- Wilson, C.J.; Clegg, R.E.; Leavesley, D.I.; Pearcy, M.J. Mediation of Biomaterial–Cell Interactions by Adsorbed Proteins: A Review. Tissue Eng. 2005, 11, 1–18. [Google Scholar] [CrossRef]
- Anselme, K. Osteoblast adhesion on biomaterials. Biomaterials 2000, 21, 667–681. [Google Scholar] [CrossRef]
- Ingber, D.E. Cellular mechanotransduction: Putting all the pieces together again. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2006, 20, 811–827. [Google Scholar] [CrossRef] [PubMed]
- Trepat, X.; Lenormand, G.; Fredberg, J.J. Universality in cell mechanics. Soft Matter 2008, 4, 1750–1759. [Google Scholar] [CrossRef]
- Rodrigues, S.N.; Goncalves, I.C.; Martins, M.C.; Barbosa, M.A.; Ratner, B.D. Fibrinogen adsorption, platelet adhesion and activation on mixed hydroxyl-/methyl-terminated self-assembled monolayers. Biomaterials 2006, 27, 5357–5367. [Google Scholar] [CrossRef] [PubMed]
- Tengvall, P.; Askendal, A.; Lundstrom, I.; Elwing, H. Studies of surface activated coagulation: Antisera binding onto methyl gradients on silicon incubated in human plasma in vitro. Biomaterials 1992, 13, 367–374. [Google Scholar] [CrossRef]
- Wang, J.; Chen, J.Y.; Yang, P.; Leng, Y.X.; Wan, G.J.; Sun, H.; Zhao, A.S.; Huang, N.; Chu, P.K. In vitro platelet adhesion and activation of polyethylene terephthalate modified by acetylene plasma immersion ion implantation and deposition. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. Atoms. 2006, 242, 12–14. [Google Scholar] [CrossRef]
- Wilson, D.J.; Williams, R.L.; Pond, R.C. Plasma modification of PTFE surfaces. Part II: Plasma-treated surfaces following storage in air or PBS. Surf. Interface Anal. 2001, 31, 397–408. [Google Scholar] [CrossRef]
- Anselme, K.; Davidson, P.; Popa, A.M.; Giazzon, M.; Liley, M.; Ploux, L. The interaction of cells and bacteria with surfaces structured at the nanometre scale. Acta Biomater. 2010, 6, 3824–3846. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Han, D.; Jiang, L. On improving blood compatibility: From bioinspired to synthetic design and fabrication of biointerfacial topography at micro/nano scales. Colloids Surf. B Biointerfaces 2011, 85, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Chu, P.K.; Ding, C. Surface nano-functionalization of biomaterials. Mater. Sci. Eng. R Rep. 2010, 70, 275–302. [Google Scholar] [CrossRef]
- Fan, H.; Chen, P.; Qi, R.; Zhai, J.; Wang, J.; Chen, L.; Chen, L.; Sun, Q.; Song, Y.; Han, D.; et al. Greatly Improved Blood Compatibility by Microscopic Multiscale Design of Surface Architectures. Small 2009, 5, 2144–2148. [Google Scholar] [CrossRef]
- Ferraz, N.; Carlsson, J.; Hong, J.; Ott, M.K. Influence of nanoporesize on platelet adhesion and activation. J. Mater. Sci. Mater. Med. 2008, 19, 3115–3121. [Google Scholar] [CrossRef] [PubMed]
- Sivaraman, B.; Latour, R.A. The relationship between platelet adhesion on surfaces and the structure versus the amount of adsorbed fibrinogen. Biomaterials 2010, 31, 832–839. [Google Scholar] [CrossRef] [PubMed]
- Mozetič, M.; Primc, G.; Vesel, A.; Modic, M.; Junkar, I.; Recek, N.; Klanjšek-Gunde, M.; Guhy, L.; Sunkara, M.K.; Assensio, M.C.; et al. Application of extremely non-equilibrium plasmas in the processing of nano and biomedical materials. Plasma Sources Sci. Technol. 2015, 24, 015026. [Google Scholar] [CrossRef]
- Curtis, A.; Wilkinson, C. Topographical control of cells. Biomaterials 1997, 18, 1573–1583. [Google Scholar] [CrossRef]
- Anselme, K.; Bigerelle, M. Topography effects of pure titanium substrates on human osteoblast long-term adhesion. Acta Biomater. 2005, 1, 211–222. [Google Scholar] [CrossRef] [PubMed]
- Bigerelle, M.; Anselme, K.; Noel, B.; Ruderman, I.; Hardouin, P.; Iost, A. Improvement in the morphology of Ti-based surfaces: A new process to increase in vitro human osteoblast response. Biomaterials 2002, 23, 1563–1577. [Google Scholar] [CrossRef]
- Dalby, M.J.; Gadegaard, N.; Tare, R.; Andar, A.; Riehle, M.O.; Herzyk, P.; Wilkinson, C.D.W.; Oreffo, R.O.C. The control of human mesenchymal cell differentiation using nanoscale symmetry and disorder. Nat. Mater. 2007, 6, 997. [Google Scholar] [CrossRef]
- Bershadsky, A.; Kozlov, M.; Geiger, B. Adhesion-mediated mechanosensitivity: A time to experiment, and a time to theorize. Curr. Opin. Cell Biol. 2006, 18, 472–481. [Google Scholar] [CrossRef]
- Chen, Q.; Espey, M.G.; Sun, A.Y.; Lee, J.H.; Krishna, M.C.; Shacter, E.; Choyke, P.L.; Pooput, C.; Kirk, K.L.; Buettner, G.R.; et al. Ascorbate in pharmacologic concentrations selectively generates ascorbate radical and hydrogen peroxide in extracellular fluid in vivo. Proc. Natl. Acad. Sci. USA 2007, 104, 8749–8754. [Google Scholar] [CrossRef]
- Feinberg, A.W.; Wilkerson, W.R.; Seegert, C.A.; Gibson, A.L.; Hoipkemeier-Wilson, L.; Brennan, A.B. Systematic variation of microtopography, surface chemistry and elastic modulus and the state dependent effect on endothelial cell alignment. J. Biomed. Mater. Res. Part A 2008, 86A, 522–534. [Google Scholar] [CrossRef]
- Stevenson, P.M.; Donald, A.M. Identification of Three Regimes of Behavior for Cell Attachment on Topographically Patterned Substrates. Langmuir 2009, 25, 367–376. [Google Scholar] [CrossRef] [PubMed]
- Berry, C.C.; Campbell, G.; Spadiccino, A.; Robertson, M.; Curtis, A.S.G. The influence of microscale topography on fibroblast attachment and motility. Biomaterials 2004, 25, 5781–5788. [Google Scholar] [CrossRef] [PubMed]
- Théry, M.; Pépin, A.; Dressaire, E.; Chen, Y.; Bornens, M. Cell distribution of stress fibres in response to the geometry of the adhesive environment. Cell Motil. 2006, 63, 341–355. [Google Scholar] [CrossRef] [PubMed]
- Hallab, N.J.; Bundy, K.J.; O’Connor, K.; Moses, R.L.; Jacobs, J.J. Evaluation of Metallic and Polymeric Biomaterial Surface Energy and Surface Roughness Characteristics for Directed Cell Adhesion. Tissue Eng. 2001, 7, 55–71. [Google Scholar] [CrossRef] [PubMed]
- Anselme, K.; Bigerelle, M.; Noel, B.; Dufresne, E.; Judas, D.; Iost, A.; Hardouin, P. Qualitative and quantitative study of human osteoblast adhesion on materials with various surface roughnesses. J. Biomed. Mater. Res. 2000, 49, 155–166. [Google Scholar] [CrossRef]
- Anselme, K.; Bigerelle, M. Modelling approach in cell/material interactions studies. Biomaterials 2006, 27, 1187–1199. [Google Scholar] [CrossRef]
- Anselme, K.; Ploux, L.; Ponche, A. Cell/Material Interfaces: Influence of Surface Chemistry and Surface Topography on Cell Adhesion. J. Adhes. Sci. Technol. 2010, 24, 831–852. [Google Scholar] [CrossRef]
- Bigerelle, M.; Anselme, K. Statistical correlation between cell adhesion and proliferation on biocompatible metallic materials. J. Biomed. Mater. Res. Part A 2005, 72A, 36–46. [Google Scholar] [CrossRef]
- Ponsonnet, L.; Comte, V.; Othmane, A.; Lagneau, C.; Charbonnier, M.; Lissac, M.; Jaffrezic, N. Effect of surface topography and chemistry on adhesion, orientation and growth of fibroblasts on nickel–titanium substrates. Mater. Sci. Eng. C 2002, 21, 157–165. [Google Scholar] [CrossRef]
- Ponsonnet, L.; Reybier, K.; Jaffrezic, N.; Comte, V.; Lagneau, C.; Lissac, M.; Martelet, C. Relationship between surface properties (roughness, wettability) of titanium and titanium alloys and cell behaviour. Mater. Sci. Eng. C 2003, 23, 551–560. [Google Scholar] [CrossRef]
- Agnihotri, A.; Soman, P.; Siedlecki, C.A. AFM measurements of interactions between the platelet integrin receptor GPIIbIIIa and fibrinogen. Colloids Surf. B Biointerfaces 2009, 71, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Savage, B.; Bottini, E.; Ruggeri, Z.M. Interaction of Integrin αIIbβ3 with Multiple Fibrinogen Domains during Platelet Adhesion. J. Biol. Chem. 1995, 270, 28812–28817. [Google Scholar] [CrossRef] [PubMed]
- Shaw, D.E.; Maragakis, P.; Lindorff-Larsen, K.; Piana, S.; Dror, R.O.; Eastwood, M.P.; Bank, J.A.; Jumper, J.M.; Salmon, J.K.; Shan, Y.; et al. Atomic-level characterization of the structural dynamics of proteins. Science 2010, 330, 341–346. [Google Scholar] [CrossRef]
- Xu, L.C.; Siedlecki, C.A. Effects of surface wettability and contact time on protein adhesion to biomaterial surfaces. Biomaterials 2007, 28, 3273–3283. [Google Scholar] [CrossRef]
- Xu, L.C.; Vadillo-Rodriguez, V.; Logan, B.E. Residence time, loading force, pH, and ionic strength affect adhesion forces between colloids and biopolymer-coated surfaces. Langmuir 2005, 21, 7491–7500. [Google Scholar] [CrossRef]
- Spijker, H.T.; Bos, R.; Busscher, H.J.; van Kooten, T.; van Oeveren, W. Platelet adhesion and activation on a shielded plasma gradient prepared on polyethylene. Biomaterials 2002, 23, 757–766. [Google Scholar] [CrossRef]
- Vogler, E.A.; Graper, J.C.; Harper, G.R.; Sugg, H.W.; Lander, L.M.; Brittain, W.J. Contact activation of the plasma coagulation cascade. I. Procoagulant surface chemistry and energy. J. Biomed. Mater. Res. 1995, 29, 1005–1016. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Lee, H.B. Platelet adhesion onto wettability gradient surfaces in the absence and presence of plasma proteins. J. Biomed. Mater. Res. 1998, 41, 304–311. [Google Scholar] [CrossRef]
- Yanagisawa, I.; Sakuma, H.; Shimura, M.; Wakamatsu, Y.; Yanagisawa, S.; Sairenji, E. Effects of “wettability” of biomaterials on culture cells. J. Oral Implantol. 1989, 15, 168–177. [Google Scholar]
- Moller, K.; Meyer, U.; Szulczewski, D.; Heide, H.; Priessnitz, B.; Jones, D. The influence of zeta-potential and interfacial-tension on osteoblast-like cells. Cells Mater. 1994, 4, 263–274. [Google Scholar]
- Lee, J.H.; Lee, S.J.; Khang, G.; Lee, H.B. The Effect of Fluid Shear Stress on Endothelial Cell Adhesiveness to Polymer Surfaces with Wettability Gradient. J. Colloid Interface Sci. 2000, 230, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Khang, G.; Lee, Y.M.; Lee, H.B. The effect of surface wettability on induction and growth of neurites from the PC-12 cell on a polymer surface. J. Colloid Interface Sci. 2003, 259, 228–235. [Google Scholar] [CrossRef]
- Lim, J.Y.; Liu, X.; Vogler, E.A.; Donahue, H.J. Systematic variation in osteoblast adhesion and phenotype with substratum surface characteristics. J. Biomed. Mater. Res. Part A 2004, 68A, 504–512. [Google Scholar] [CrossRef] [PubMed]
- George, J.; Sreekala, M.S.; Thomas, S. A review on interface modification and characterization of natural fiber reinforced plastic composites. Polym. Eng. Sci. 2001, 41, 1471–1485. [Google Scholar] [CrossRef]
- Jurk, K.; Kehrel, B.E. Platelets: Physiology and biochemistry. Semin. Thromb. Hemost. 2005, 31, 381–392. [Google Scholar] [CrossRef]
- Anderson, J.M.; Kottke-Marchant, K. Platelet Interactions With Biomaterials and Artificial Devices; CRC Press Inc: Boca Raton, FL, USA, 1987. [Google Scholar]
- Klinkmann, H.; Falkenhagen, D.; Courtney, J.M. Clinical Relevance of Biocompatibility—The Material Cannot Be Divorced from the Device. In Uremia Therapy; Gurland, H., Ed.; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 1987; pp. 125–140. [Google Scholar] [CrossRef]
- Schaller, J.; Gerber, S.; Kaempfer, U.; Lejon, S.; Trachsel, C. Human Blood Plasma Proteins: Structure and Function; John Wiley & Sons: New York, NY, USA, 2008. [Google Scholar]
- Courtney, J.; Lamba, N.; Sundaram, S.; Forbes, C. Biomaterials for blood-contacting applications. Biomaterials 1994, 15, 737–744. [Google Scholar] [CrossRef]
- Salzman, E.W. Interaction of the blood with natural and artificial surfaces. In Blood Material Interaction; Dekker Inc.: Ney York, NY, USA, 1986; p. 39. [Google Scholar]
- Meyer, J.G. Blutgerinnung und Fibrinolyse; Deutsche Ärzte Verlag: Köln, Germany, 1986. [Google Scholar]
- Brash, J.L. Mechanism of adsorption of proteins to solid surfaces and its relationship to blood compatibility. In Biocompatible Polymers, Metals and Composites; Technomic: Lancaster, UK, 1983; p. 35. [Google Scholar]
- Baszkin, A. The effect of polymer surface composition and structure on adsorption of plasma proteins. In Blood Compatible Materials and Their Testing; Dawids, S., Bantjes, A., Eds.; Martins Nijhoff Publishers: Dortrecht, The Netherlands, 1986; p. 39. [Google Scholar]
- Kottke-Marchant, K.; Anderson, J.M.; Umemura, Y.; Marchant, R.E. Effect of albumin coating on the in vitro blood compatibility of Dacron arterial prostheses. Biomaterials 1989, 10, 147–155. [Google Scholar] [CrossRef]
- Ikada, Y. Blood-Compatible Polymers; Springer: Berlin/Heidelberg, Germany, 1984; pp. 103–140. [Google Scholar]
- Andrade, J.D. Interfacial phenomena and biomaterials. Med. Instrum. 1973, 7, 110–119. [Google Scholar]
- Baier, R.E. The role of surface energy in thrombogenesis. Bull. N. Y. Acad. Med. 1972, 48, 257–272. [Google Scholar]
- Ratner, B.D.; Hoffman, A.S.; Hanson, S.R.; Harker, L.A.; Whiffen, J.D. Blood-compatibility-water-content relationships for radiation-grafted hydrogels. J. Polym. Sci. Polym. Symp. 1979, 66, 363–375. [Google Scholar] [CrossRef]
- Bantjes, A. Clotting Phenomena at the Blood-Polymer Interface and Development of Blood Compatible Polymeric Surfaces. Br. Polym. J. 1978, 10, 267–274. [Google Scholar] [CrossRef]
- Norde, W.; Lyklema, J. Proportion titration and electrokinetic studies of adsorbed protein layers. In Surface and Interfacial Aspects of Biomedical Polymers; Andrade, J.D., Ed.; Plenum Press: New York, NY, USA, 1985; p. 241. [Google Scholar]
- Bruck, S.D. Physicochemical aspects of the blood compatibility of polymeric surfaces. J. Polym. Sci. Polym. Symp. 1979, 66, 283–312. [Google Scholar] [CrossRef]
- Sawyer, P.N.; Srinivasan, S. Studies on the biophysics of intravascular thrombosis. Am. J. Surg. 1967, 114, 42–60. [Google Scholar] [CrossRef]
- Szycher, M. (Ed.) Biocomatible Polymers, Metals and Composites; Technomic: Lanchester, UK, 1983. [Google Scholar]
- Yu, J.; Sundaram, S.; Weng, D.; Courtney, J.M.; Moran, C.R.; Graham, N.B. Blood interactions with novel polyurethaneurea hydrogels. Biomaterials 1991, 12, 119–120. [Google Scholar] [CrossRef]
- Beissinger, R.L.; Leonard, E.F. Plasma protein adsorption and desorption rates on quartz: Approach to multi-component systems. Trans.Am. Soc. Artif. Intern. Organs 1981, 27, 225–230. [Google Scholar] [PubMed]
- Soderquist, M.E.; Walton, A.G. Structural changes in proteins adsorbed on polymer surfaces. J. Colloid Interface Sci. 1980, 75, 386–397. [Google Scholar] [CrossRef]
- Lundström, I.; Elwing, H. Simple kinetic models for protein exchange reactions on solid surfaces. J. Colloid Interface Sci. 1990, 136, 68–84. [Google Scholar] [CrossRef]
- Giangrande, P.L.F. Six Characters in Search of An Author: The History of the Nomenclature of Coagulation Factors. Br. J. Haematol. 2003, 121, 703–712. [Google Scholar] [CrossRef]
- Brummel, K.E.; Butenas, S.; Mann, K.G. An Integrated Study of Fibrinogen during Blood Coagulation. J. Biol. Chem. 1999, 274, 22862–22870. [Google Scholar] [CrossRef]
- Macfarlane, R.G. A. The blood clotting mechanism The development of a theory of blood coagulation. Proc. R. Soc. Lond. Ser. B Biol. Sci. 1969, 173, 261–268. [Google Scholar] [CrossRef]
- Van Wachem, P.; Beugeling, T.; Feijen, J.; Bantjes, A.; Detmers, J.; Van Aken, W. Interaction of cultured human endothelial cells with polymeric surfaces of different wettabilities. Biomaterials 1985, 6, 403–408. [Google Scholar] [CrossRef]
- Joseph, K.; Thomas, S.; Pavithran, C. Effect of chemical treatment on the tensile properties of short sisal fibre-reinforced polyethylene composites. Polymer 1996, 37, 5139–5149. [Google Scholar] [CrossRef]
- France, R.M.; Short, R.D. Plasma treatment of polymers Effects of energy transfer from an argon plasma on the surface chemistry of poly(styrene), low density poly(ethylene), poly(propylene) and poly(ethylene terephthalate). J. Chem. Soc. Faraday Trans. 1997, 93, 3173–3178. [Google Scholar] [CrossRef]
- Morent, R.; De Geyter, N.; Desmet, T.; Dubruel, P.; Leys, C. Plasma surface modification of biodegradable polymers: a review. Plasma Process. Polym. 2011, 8, 171–190. [Google Scholar] [CrossRef]
- Vesel, A.; Mozetic, M. New developments in surface functionalization of polymers using controlled plasma treatments. J. Phys. D Appl. Phys. 2017, 50, 293001. [Google Scholar] [CrossRef]
- Plasma—Materials Interactions. Plasma Deposition, Treatment, and Etching of Polymers; d’Agostino, R., Ed.; Academic Press: San Diego, CA, USA, 1990; p. ii. [Google Scholar] [CrossRef]
- Yasuda, H.K.; Yeh, Y.S.; Fusselman, S. A growth mechanism for the vacuum deposition of polymeric materials. Pure nd Appl. Chem. 1990, 62, 1689. [Google Scholar] [CrossRef]
- Kirkpatrick, M.J.; Locke, B.R. Hydrogen, oxygen, and hydrogen peroxide formation in aqueous phase pulsed corona electrical discharge. Ind. Eng. Chem. Res. 2005, 44, 4243–4248. [Google Scholar] [CrossRef]
- Locke, B.R.; Shih, K.-Y. Review of the methods to form hydrogen peroxide in electrical discharge plasma with liquid water. Plasma Sources Sci. Technol. 2011, 20, 034006. [Google Scholar] [CrossRef]
- Rajesh, D.; Mark, J.K. A model for plasma modification of polypropylene using atmospheric pressure discharges. J. Phys. D Appl. Phys. 2003, 36, 666. [Google Scholar]
- Fridman, A. Plasma Chemistry; Cambridge University Press: Cambridge, UK, 2008; pp. 848–857. [Google Scholar]
- Inagaki, N.; Narushim, K.; Tuchida, N.; Miyazaki, K. Surface characterization of plasma-modified poly(ethylene terephthalate) film surfaces. J. Polym. Sci. Part B Polym. Phys. 2004, 42, 3727–3740. [Google Scholar] [CrossRef]
- Williams, R.L.; Wilson, D.J.; Rhodes, N.P. Stability of plasma-treated silicone rubber and its influence on the interfacial aspects of blood compatibility. Biomaterials 2004, 25, 4659–4673. [Google Scholar] [CrossRef] [PubMed]
- Wavhal, D.S.; Fisher, E.R. Modification of polysulfone ultrafiltration membranes by CO2 plasma treatment. Desalination 2005, 172, 189–205. [Google Scholar] [CrossRef]
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Recek, N. Biocompatibility of Plasma-Treated Polymeric Implants. Materials 2019, 12, 240. https://doi.org/10.3390/ma12020240
Recek N. Biocompatibility of Plasma-Treated Polymeric Implants. Materials. 2019; 12(2):240. https://doi.org/10.3390/ma12020240
Chicago/Turabian StyleRecek, Nina. 2019. "Biocompatibility of Plasma-Treated Polymeric Implants" Materials 12, no. 2: 240. https://doi.org/10.3390/ma12020240
APA StyleRecek, N. (2019). Biocompatibility of Plasma-Treated Polymeric Implants. Materials, 12(2), 240. https://doi.org/10.3390/ma12020240




