The Influence of Lignin Diversity on the Structural and Thermal Properties of Polymeric Microspheres Derived from Lignin, Styrene, and/or Divinylbenzene
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Materials
2.2. Lignin Fractionation, Modification, and Characterization
2.3. Synthesis of Microspheres
2.4. Characterization Methods of Microspheres
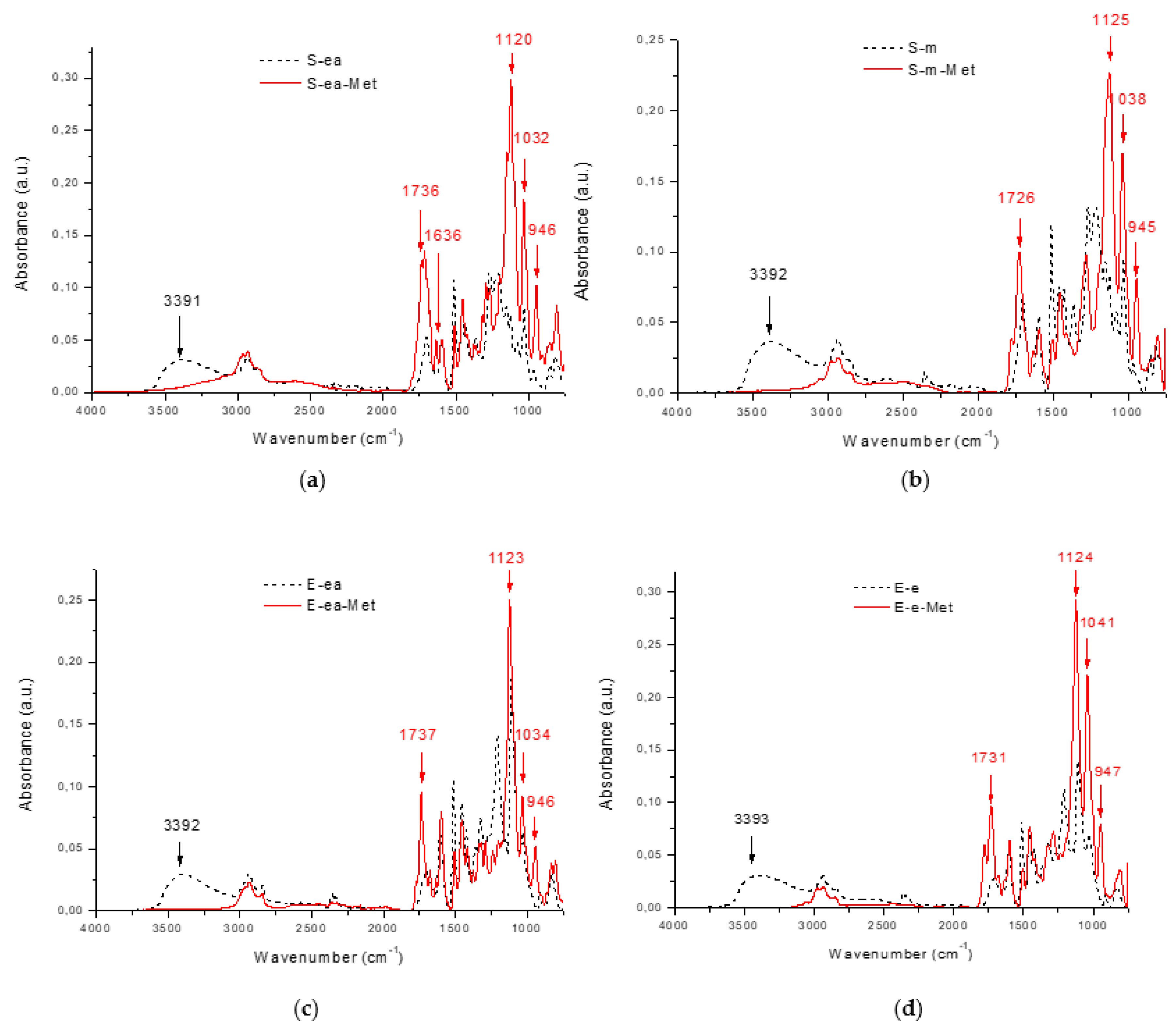
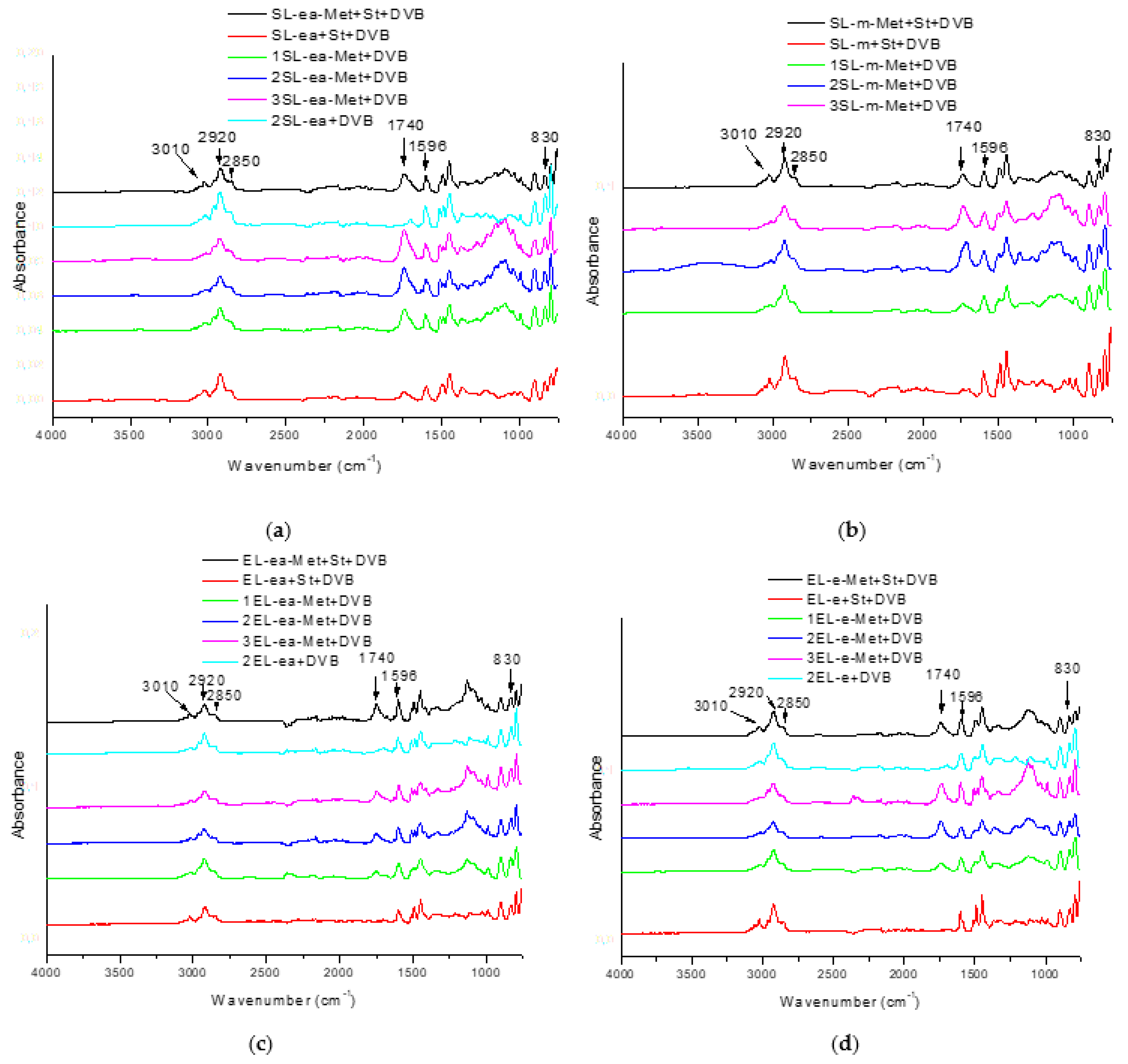
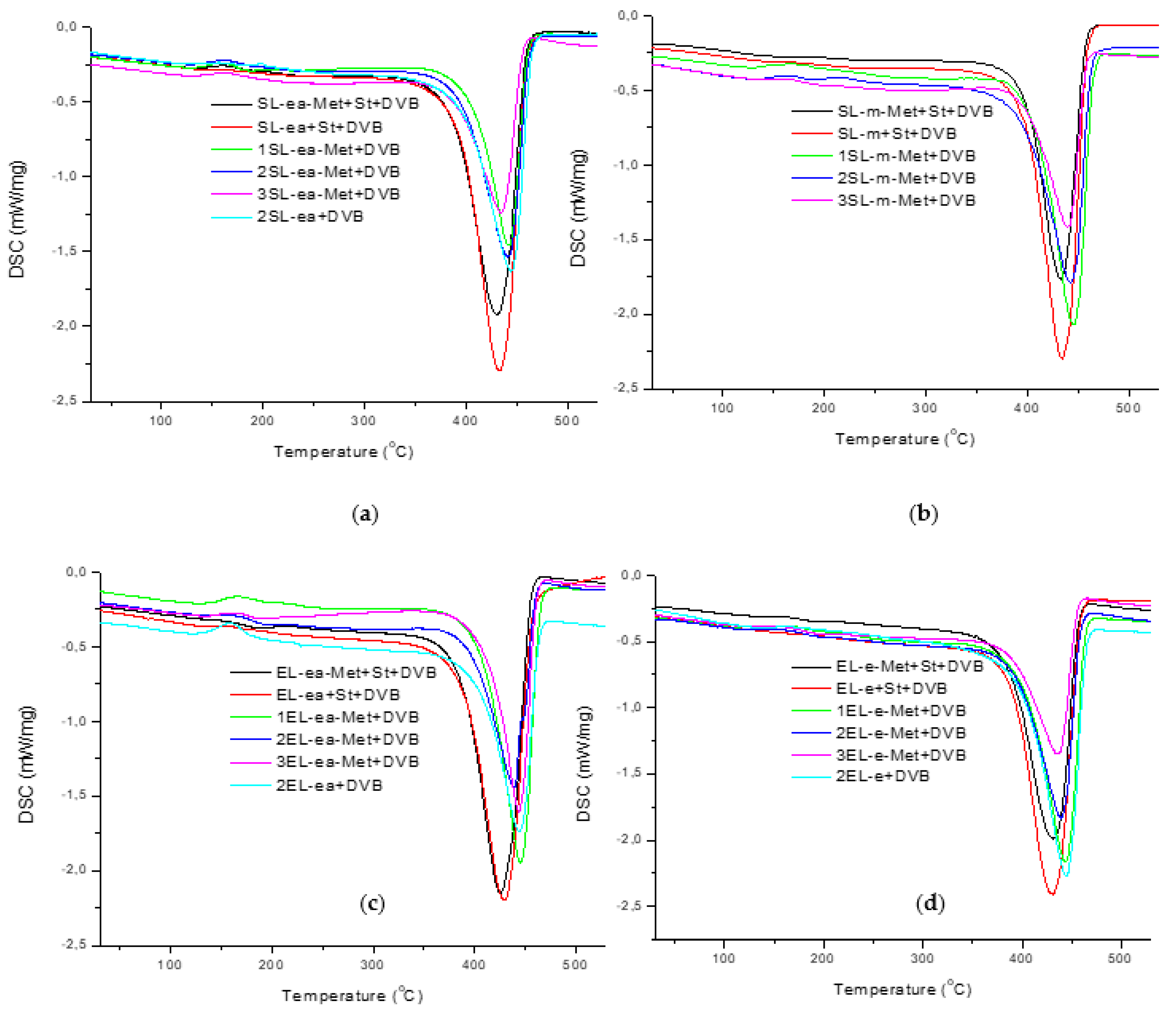
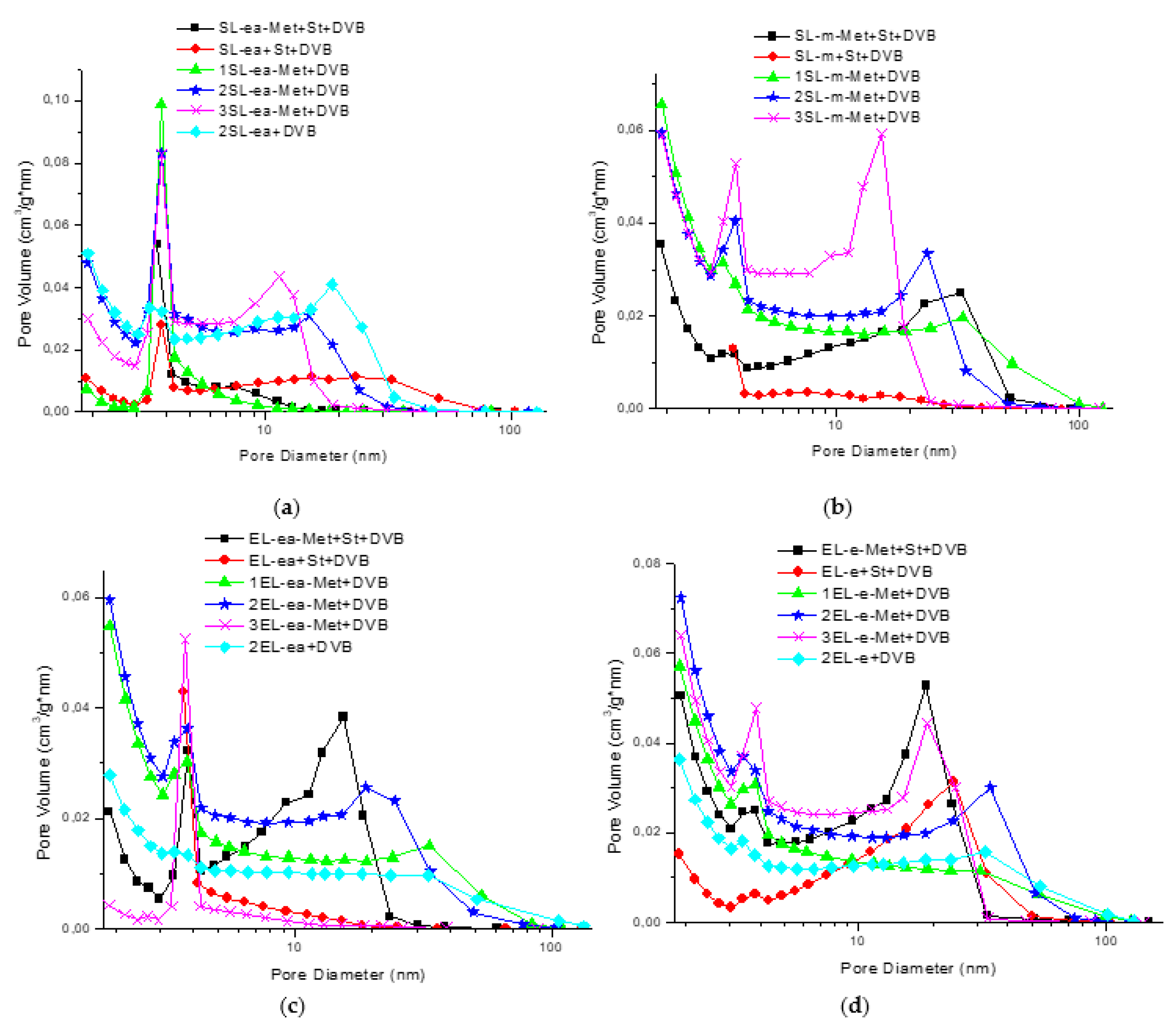
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Manjarrez Nevárez, L.A.; Ballinas Casarrubias, L.; Celzard, A.; Fierro, V.; Torres Muñoz, V.; Camacho Davila, A.; Torres Lubian, J.R.; González Sánchez, G. Biopolymer-based nanocomposites: Effect of lignin acetylation in cellulose triacetate films. Sci. Technol. Adv. Mater. 2011, 12, 045006. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Dean, K.; Li, L. Polymer blends and composites from renewable resources. Prog. Polym. Sci. 2006, 31, 576–602. [Google Scholar] [CrossRef]
- Saito, T.; Brown, R.H.; Hunt, M.A.; Pickel, D.L.; Pickel, J.M.; Messman, J.M.; Baker, F.S.; Keller, M.; Naskar, A.K. Turning renewable resources into value-added polymer: Development of lignin-based thermoplastic. Green Chem. 2012, 14, 3295–3303. [Google Scholar] [CrossRef]
- Meier, M.A.R.; Meier, M. Renewable Resources for Polymer Chemistry: A Sustainable Alternative? Macromol. Rapid Commun. 2011, 32, 1297–1298. [Google Scholar] [CrossRef] [PubMed]
- Imre, B.; Pukánszky, B. Compatibilization in bio-based and biodegradable polymer blends. Eur. Polym. J. 2013, 49, 1215–1233. [Google Scholar] [CrossRef]
- Tănase, E.E.; Râpă, M.; Popa, O. Biopolymers based on renewable resources—A review. In Proceedings of the International Conference Agriculture for Life, Life for Agriculture, Bucharest, Romania, 5–7 June 2014; Scientific Bulletin, Series F, Biotechnologies: Bucharest, Romania; Volume XVIII. [Google Scholar]
- Kaplan, D.L. Introduction to Biopolymers from Renewable Resources. In Biopolymers from Renewable Resources. Macromolecular Systems—Materials Approach; Kaplan, D.L., Ed.; Springer: Berlin/Heidelberg, Germany, 1998; pp. 1–29. [Google Scholar]
- Zhang, Z. Lignin Modification and Degradation for Advanced Composites and Chemicals. Ph.D. Thesis, Georgia Institute of Technology, Atlanta, GA, USA, December 2017. [Google Scholar]
- Saini, J.K.; Saini, R.; Tewari, L. Lignocellulosic agriculture wastes as biomass feedstocks for second-generation bioethanol production: Concepts and recent developments. 3 Biotech 2015, 5, 337–353. [Google Scholar] [CrossRef]
- Väisänen, T.; Haapala, A.; Lappalainen, R.; Tomppo, L. Utilization of agricultural and forest industry waste and residues in natural fiber-polymer composites: A review. Waste Manag. 2016, 54, 62–73. [Google Scholar] [CrossRef] [PubMed]
- Treinyte, J.; Bridziuviene, D.; Fataraite-Urboniene, E.; Rainosalo, E.; Rajan, R.; Cesoniene, L.; Grazuleviciene, V. Forestry wastes filled polymer composites for agricultural use. J. Clean Prod. 2018, 205, 388–406. [Google Scholar] [CrossRef]
- Whetten, R.; Sederoff, R. Lignin Biosynthesis. Plant Cell 1995, 7, 1001–1013. [Google Scholar] [CrossRef]
- Sen, S.; Patil, S.; Argyropoulos, D.S. Thermal Properties of Lignin in Copolymers, Blends, and Composites: A Review. Green Chem. 2015, 17, 4862–4887. [Google Scholar] [CrossRef]
- Hatakeyama, H.; Hatakeyama, T. Lignin Structure, Properties, and Applications. In Biopolymers Advances in Polymer Science; Abe, A., Dusek, K., Kobayashi, S., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; Volume 232, pp. 1–63. [Google Scholar]
- Crestini, C.; Melone, F.; Sette, M.; Saladino, R. Milled wood lignin: A linear oligomer. Biomacromolecules 2011, 12, 3928–3935. [Google Scholar] [CrossRef] [PubMed]
- Sette, M.; Wechselberger, R.; Crestini, C. Elucidation of Lignin Structure by Quantitative 2D NMR. Chem. Eur. J. 2011, 17, 9529–9535. [Google Scholar] [CrossRef] [PubMed]
- Goliszek, M.; Sobiesiak, M.; Fila, K.; Podkościelna, B. Evaluation of sorption capabilities of biopolymeric microspheres by the solid-phase extraction. Adsorption 2019, 25, 289–300. [Google Scholar] [CrossRef]
- Laurichesse, S.; Avérous, L. Chemical modification of lignins: Towards biobased polymers. Prog. Polym. Sci. 2014, 39, 1266–1290. [Google Scholar] [CrossRef]
- Thielemans, W.; Wool, R.P. Lignin Esters for Use in Unsaturated Thermosets: Lignin Modification and Solubility Modeling. Biomacromolecules 2005, 6, 1895–1905. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Weng, J.K.; Chapple, C. Improvement of biomass through lignin modification. Plant J. 2008, 54, 569–581. [Google Scholar] [CrossRef] [PubMed]
- Bernier, E.; Lavigne, C.; Robidoux, P. Life cycle assessment of kraft lignin for polymer applications. Int. J. Life Cycle Assess. 2013, 18, 520–528. [Google Scholar] [CrossRef]
- Atifi, S.; Miao, C.; Hamad, W.Y. Surface modification of lignin for applications in polypropylene blends. J. Appl. Polym. Sci. 2017, 134, 45103. [Google Scholar] [CrossRef]
- Alekhina, M.; Ershova, O.; Ebert, A.; Heikkinen, S.; Sixta, H. Softwood kraft lignin for value-added applications: Fractionation and structural characterization. Ind. Crops Prod. 2015, 66, 220–228. [Google Scholar] [CrossRef]
- Ghaffar, S.H.; Fan, M. Lignin in straw and its applications as an adhesive. Int. J. Adhes. Adhes. 2014, 48, 92–101. [Google Scholar] [CrossRef]
- Thakur, V.K.; Thakur, M.K.; Raghavan, P.; Kessler, M.R. Progress in Green Polymer Composites from Lignin for Multifunctional Applications: A Review. ACS Sustain. Chem. Eng. 2014, 2, 1072–1092. [Google Scholar] [CrossRef]
- Gopalakrishnan, K.; Ceylan, H.; Kim, S. Renewable biomass-derived lignin in transportation infrastructure strengthening applications. Int. J. Sustain. Eng. 2013, 6, 316–325. [Google Scholar] [CrossRef]
- Berlin, A.; Balakshin, M. Industrial Lignins: Analysis, Properties, and Applications. In Bioenergy Research: Advances and Applications; Vijai, G., Maria Tuohy, G., Kubicek, C.P., Saddler, J., Xu, F., Eds.; Elsevier: New York, NY, USA, 2014; pp. 315–336. [Google Scholar]
- Duval, A.; Lawoko, M. A review on lignin-based polymeric, micro- and nano-structured materials. React. Funct. Polym. 2014, 85, 78–96. [Google Scholar] [CrossRef]
- Gordobil, O.; Moriana, R.; Zhang, L.; Labidi, J.; Sevastyanova, O. Assesment of technical lignins for uses in biofuels and biomaterials: Structure-related properties, proximate analysis and chemical modification. Ind. Crops Prod. 2016, 83, 155–165. [Google Scholar] [CrossRef]
- Naseem, A.; Tabasum, S.; Zia, K.M.; Zuber, M.; Ali, M.; Noreen, A. Lignin-derivatives based polymers, blends and composites: A review. Int. J. Biol. Macromol. 2016, 93, 296–313. [Google Scholar] [CrossRef]
- Podkościelna, B.; Sobiesiak, M.; Gawdzik, B.; Zhao, Y.; Sevastyanova, O. Preparation of lignin-containing porous microspheres through the copolymerization of lignin acrylate derivatives with St and DVB. Holzforschung 2015, 69, 769–776. [Google Scholar] [CrossRef]
- Kun, D.; Pukánszky, B. Polymer/lignin blends: Interactions, properties, applications. Eur. Polym. J. 2017, 93, 618–641. [Google Scholar] [CrossRef]
- Yin, Q.; Yang, W.; Sun, C.; Di, M. Preparation and properties of lignin-epoxy resin composite. BioResources 2012, 7, 5737–5748. [Google Scholar] [CrossRef]
- Jesionowski, T.; Klapiszewski, Ł.; Milczarek, G. Kraft lignin and silica as precursors of advanced composite materials and electroactive blends. J. Mater. Sci. 2014, 49, 1376–1385. [Google Scholar] [CrossRef]
- He, Z.W.; Lü, Q.F.; Zhang, J.Y. Facile preparation of hierarchical polyaniline-lignin composite with a reactive silver-ion adsorbability. ACS Appl. Mater. Interfaces 2012, 4, 369–374. [Google Scholar] [CrossRef]
- Ballner, D.; Herzele, S.; Keckes, J.; Edler, M.; Griesser, T.; Saake, B.; Liebner, F.; Potthast, A.; Paulik, C.; Gindl-Altmutter, W. Lignocellulose Nanofiber-Reinforced Polystyrene Produced from Composite Microspheres Obtained in Suspension Polymerization Shows Superior Mechanical Performance. ACS Appl. Mater. Interfaces 2016, 8, 13520–13525. [Google Scholar] [CrossRef]
- Wang, K.; Xu, F.; Sun, R. Molecular characteristics of Kraft-AQ pulping lignin fractionated by sequential organic solvent extraction. Int. J. Mol. Sci. 2010, 11, 2988–3001. [Google Scholar] [CrossRef]
- Li, M.F.; Sun, S.N.; Xu, F.; Sun, R.C. Sequential solvent fractionation of heterogeneous bamboo organosolv lignin for value-added application. Sep. Purif. Technol. 2012, 101, 18–25. [Google Scholar] [CrossRef]
- Park, S.Y.; Kim, J.Y.; Youn, H.J.; Choi, J.W. Fractionation of lignin macromolecules by sequential organic solvents systems and their characterization for further valuable applications. Int. J. Biol. Macromol. 2018, 106, 793–802. [Google Scholar] [CrossRef]
- Jääskeläinen, A.S.; Liitiä, T.; Mikkelson, A.; Tamminen, T. Aqueous organic solvent fractionation as means to improve lignin homogeneity and purity. Ind. Crops Prod. 2017, 103, 51–58. [Google Scholar] [CrossRef]
- Tagami, A.; Gioia, C.; Lauberts, M.; Budnyak, T.; Moriana, R.; Lindström, M.E.; Sevastyanova, O. Solvent fractionation of softwood and hardwood kraft lignins for more efficient uses: Compositional, structural, thermal, antioxidant and adsorption properties. Ind. Crops Prod. 2019, 129, 123–134. [Google Scholar] [CrossRef]
- Duval, A.; Vilaplana, F.; Crestini, C.; Lawoko, M. Solvent screening for the fractionation of industrial kraft lignin. Holzforschung 2016, 70, 11–20. [Google Scholar] [CrossRef]
- Toledano, A.; Serrano, l.; Garcia, A.; Mondragon, I.; Labidi, J. Comparative study of lignin fractionation by ultrafiltration and selective precipitation. Chem. Eng. J. 2010, 157, 93–99. [Google Scholar] [CrossRef]
- Jönsson, A.S.; Wallberg, O. Cost estimates of kraft lignin recovery by ultrafiltration. Desalination 2009, 237, 254–267. [Google Scholar] [CrossRef]
- Lauberts, M.; Sevastyanova, O.; Ponomarenko, J.; Dizhbite, T.; Dobele, G.; Volperts, A.; Lauberte, L.; Telysheva, G. Fractionation of technical lignin with ionic liquids as a method for improving purity and antioxidant activity. Ind. Crops Prod. 2017, 95, 512–520. [Google Scholar] [CrossRef]
- Sevastyanova, O.; Helander, M.; Chowdhury, S.; Lange, H.; Wedin, H.; Zhang, L.; Ek, M.; Kadla, J.F.; Crestini, C.; Lindström, M.E. Tailoring the Molecular and Thermo-Mechanical Properties of Kraft Lignin by Ultrafiltration. J. Appl. Polym. Sci. 2014, 131, 9505–9515. [Google Scholar] [CrossRef]
- Zhao, Y.; Tagami, A.; Dobele, G.; Lindström, M.E.; Sevastyanova, O. The Impact of Lignin Structural Diversity on Performance of Cellulose Nanofiber (CNF)-Starch Composite Films. Polymers 2019, 11, 538. [Google Scholar] [CrossRef]
- Aminzadeh, S.; Lauberts, M.; Dobele, G.; Ponomarenko, J.; Mattsson, T.; Lindström, M.E.; Sevastyanova, O. Membrane filtration of kraft lignin: Structural properties and anti-oxidant activity of the low-molecular-weight fraction. Ind. Crop. Prod. 2018, 112, 200–209. [Google Scholar] [CrossRef]
- Passoni, V.; Scarica, C.; Levi, M.; Turri, S.; Griffini, G. Fractionation of Industrial Softwood Kraft Lignin: Solvent Selection as a Tool for Tailored Material Properties. ACS Sustain. Chem. Eng. 2016, 4, 2232–2242. [Google Scholar] [CrossRef]
- Goliszek, M.; Podkościelna, B.; Fila, K.; Riazanova, A.; Aminzadeh, S.; Sevastyanova, O.; Gun’ko, V. Synthesis and structure characterization of polymeric nanoporous microspheres with lignin. Cellulose 2018, 25, 5843–5862. [Google Scholar] [CrossRef]
- Tomani, P. The LignoBoost process. Cell. Chem. Technol. 2010, 44, 53–58. [Google Scholar]
- Tagami, A. Towards Molecular Weight-Dependent uses of Kraft Lignin. Ph.D. Thesis, KTH Royal Institute of Technology, Stockholm, Sweden, 2018. Available online: http://kth.diva-portal.org/smash/get/diva2:1240150/FULLTEXT01.pdf (accessed on 14 September 2018).
- Podkościelna, B.; Goliszek, M.; Sevastyanova, O. New approach in the application of lignin for the synthesis of hybrid materials. Pure Appl. Chem. 2017, 89, 161–171. [Google Scholar] [CrossRef]
- Faix, O. Classification of lignins from different botanical origins by FT-IR spectroscopy. Holzforschung 1991, 45, 21–28. [Google Scholar] [CrossRef]
- Jaswal, S.; Gaur, B. Green methacrylated lignin model compounds as reactive monomers with low VOC emission for thermosetting resins. Green Process. Synth. 2015, 4, 191–202. [Google Scholar] [CrossRef]
- Silverstein, R.M.; Webster, F.X.; Kiemle, D.J. Spectrometric Identification of Organic Compounds, 7th ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2005. [Google Scholar]
- Hermán, V.; Takacs, H.; Duclairoir, F.; Renault, O.; Tortai, J.H.; Viala, B. Core double–shell cobalt/graphene/polystyrene magnetic nanocomposites synthesized by in situ sonochemical polymerization. RSC Adv. 2015, 5, 51371–51381. [Google Scholar] [CrossRef]

| Polymer | Monomers (g) | ||
|---|---|---|---|
| L 1 | L-Met 2 | St 3 | |
| SL-ea-Met+St+DVB | 0 | 2 | 4 |
| SL-ea +St+DVB | 2 | 0 | 4 |
| 1SL-ea-Met+DVB | 0 | 1 | 0 |
| 2SL-ea-Met+DVB | 0 | 2 | 0 |
| 3SL-ea-Met+DVB | 0 | 3 | 0 |
| 2SL-ea+DVB | 2 | 0 | 0 |
| SL-m-Met+St+DVB | 0 | 2 | 4 |
| SL-m +St+DVB | 2 | 0 | 4 |
| 1SL-m-Met+DVB | 0 | 1 | 0 |
| 2SL-m-Met+DVB | 0 | 2 | 0 |
| 3SL-m-Met+DVB | 0 | 3 | 0 |
| EL-ea-Met+St+DVB | 0 | 2 | 4 |
| EL-ea +St+DVB | 2 | 0 | 4 |
| 1EL-ea-Met+DVB | 0 | 1 | 0 |
| 2EL-ea-Met+DVB | 0 | 2 | 0 |
| 3EL-ea-Met+DVB | 0 | 3 | 0 |
| 2EL-ea+DVB | 2 | 0 | 0 |
| EL-e-Met+St+DVB | 0 | 2 | 4 |
| EL-e +St+DVB | 2 | 0 | 4 |
| 1EL-e-Met+DVB | 0 | 1 | 4 |
| 2EL-e-Met+DVB | 0 | 2 | 0 |
| 3EL-e-Met+DVB | 0 | 3 | 0 |
| 2EL-e+DVB | 2 | 0 | 0 |
| Lignin Fraction | Mn 1 (g/mol) | Mw 2 (g/mol) | Đ 3 | Aliphatic-OH (mmol/g) | Carboxyl-OH (mmol/g) | Phenolic-OH (mmol/g) | ||
|---|---|---|---|---|---|---|---|---|
| Condensed G 4 | Non-Condensed (G+S 5) | Total | ||||||
| Spruce-ethyl acetate (SL-ea) | 720 | 1160 | 1.6 | 0.7 | 0.7 | 1.8 | 3.2 | 5.0 |
| Spruce-methanol (SL-m) | 1400 | 2900 | 2.1 | 1.8 | 0.4 | 2.0 | 2.5 | 4.5 |
| Eucalyptus-ethyl acetate (EL-ea) | 630 | 940 | 1.5 | 0.6 | 0.3 | 0.8 | 4.1 | 4.9 |
| Eucalyptus-ethanol (EL-e) | 870 | 1420 | 1.6 | 1.4 | 0.4 | 0.9 | 3.5 | 4.4 |
| Polymer | Td 1 (°C) | ΔHd 2 (J/g) |
|---|---|---|
| SL-ea-Met + St + DVB | 430.2 | 539.8 |
| SL-ea + St + DVB | 432.7 | 595.1 |
| 1SL-ea-Met + DVB | 441.6 | 321.0 |
| 2SL-ea-Met + DVB | 440.6 | 378.0 |
| 3SL-ea-Met + DVB | 434.3 | 296.8 |
| 2SL-ea + DVB | 444.1 | 428.3 |
| SL-m-Met + St + DVB | 432.2 | 568.9 |
| SL-m + St + DVB | 434.3 | 529.9 |
| 1SL-m-Met + DVB | 444.4 | 409.3 |
| 2SL-m-Met + DVB | 442.0 | 420.5 |
| 3SL-m-Met + DVB | 439.5 | 251 |
| EL-ea-Met + St + DVB | 425.3 | 558.7 |
| EL-ea + St + DVB | 428.6 | 556.3 |
| 1EL-ea-Met + DVB | 445.1 | 407.7 |
| 2EL-ea-Met + DVB | 438.3 | 318.9 |
| 3EL-ea-Met + DVB | 443.7 | 315.2 |
| 2EL-ea + DVB | 444.2 | 326.7 |
| EL-e-Met + St + DVB | 431.5 | 505.1 |
| EL-e + St + DVB | 430.5 | 579.6 |
| 1EL-e-Met + DVB | 443.6 | 446.9 |
| 2EL-e-Met + DVB | 439.4 | 377.2 |
| 3EL-e-Met + DVB | 435.2 | 281.6 |
| 2EL-e + DVB | 444.7 | 479.0 |
| Polymer | SBET 1 (m2/g) | VTOT 2 (cm3/g) | DA 3 (nm) |
|---|---|---|---|
| SL-ea-Met + St + DVB | 51 | 0.104 | 8.2 |
| SL-ea + St + DVB | 139 | 0.597 | 17.1 |
| 1SL-ea-Met + DVB | 103 | 0.126 | 4.9 |
| 2SL-ea-Met + DVB | 396 | 0.700 | 7.1 |
| 3SL-ea-Met + DVB | 314 | 0.542 | 6.9 |
| 2SL-ea + DVB | 416 | 0.996 | 9.6 |
| SL-m-Met + St + DVB | 291 | 0.983 | 13.5 |
| SL-m + St + DVB | 23 | 0.077 | 13.2 |
| 1SL-m-Met + DVB | 474 | 1.405 | 11.9 |
| 2SL-m-Met + DVB | 442 | 0.969 | 8.8 |
| 3SL-m-Met + DVB | 462 | 0.803 | 6.9 |
| EL-ea-Met + St + DVB | 229 | 0.517 | 9.0 |
| EL-ea + St + DVB | 30 | 0.080 | 10.6 |
| 1EL-ea-Met + DVB | 384 | 0.937 | 9.7 |
| 2EL-ea-Met + DVB | 434 | 0.953 | 8.8 |
| 3EL-ea-Met + DVB | 62 | 0.083 | 5.4 |
| 2EL-ea + DVB | 212 | 0.802 | 15.1 |
| EL-e-Met + St + DVB | 410 | 0.938 | 9.1 |
| EL-e + St + DVB | 195 | 0.785 | 16.1 |
| 1EL-e-Met + DVB | 394 | 0.996 | 10.1 |
| 2EL-e-Met + DVB | 506 | 1.342 | 10.6 |
| 3EL-e-Met + DVB | 483 | 0.981 | 8.1 |
| 2EL-e + DVB | 299 | 1.135 | 15.2 |
| Polymer | Swellability Coefficient, B (%) | ||||||
|---|---|---|---|---|---|---|---|
| Acetone | THF 1 | Chloroform | ACN 2 | Methanol | Toluene | Aqua dest. | |
| SL-ea-Met + St + DVB | 122 | 122 | 122 | 122 | 100 | 100 | 0 |
| SL-ea + St + DVB | 100 | 30 | 91 | 67 | 58 | 100 | 0 |
| 1SL-ea-Met + DVB | 113 | 78 | 75 | 63 | 63 | 63 | 0 |
| 2SL-ea-Met + DVB | 67 | 46 | 85 | 82 | 64 | 75 | 0 |
| 3SL-ea-Met + DVB | 58 | 83 | 45 | 55 | 80 | 45 | 0 |
| 2SL-ea + DVB | 73 | 47 | 67 | 67 | 60 | 83 | 0 |
| SL-m-Met + St + DVB | 0 | 0 | 0 | 10 | 0 | 0 | 0 |
| SL-m + St + DVB | 100 | 60 | 120 | 100 | 80 | 209 | 0 |
| 1SL-m-Met + DVB | 8 | 8 | 8 | 15 | 0 | 0 | 0 |
| 2SL-m-Met + DVB | 0 | 0 | 0 | 0 | 0 | 11 | 0 |
| 3SL-m-Met + DVB | 22 | 10 | 11 | 11 | 11 | 10 | 0 |
| EL-ea-Met + St + DVB | 23 | 25 | 55 | 36 | 27 | 55 | 0 |
| EL-ea + St + DVB | 109 | 120 | 136 | 91 | 118 | 127 | 0 |
| 1EL-ea-Met + DVB | 7 | 13 | 7 | 7 | 7 | 22 | 0 |
| 2EL-ea-Met + DVB | 20 | 27 | 6 | 8 | 15 | 7 | 0 |
| 3EL-ea-Met + DVB | 40 | 40 | 80 | 70 | 40 | 70 | 0 |
| 2EL-ea + DVB | 0 | 6 | 0 | 0 | 0 | 6 | 0 |
| EL-e-Met + St + DVB | 22 | 10 | 11 | 0 | 0 | 0 | 0 |
| EL-e + St + DVB | 0 | 9 | 0 | 0 | 0 | 0 | 0 |
| 1EL-e-Met + DVB | 6 | 0 | 0 | 10 | 0 | 0 | 0 |
| 2EL-e-Met + DVB | 22 | 0 | 0 | 11 | 0 | 0 | 0 |
| 3EL-e-Met + DVB | 25 | 0 | 13 | 13 | 13 | 14 | 0 |
| 2EL-e + DVB | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goliszek, M.; Podkościelna, B.; Sevastyanova, O.; Gawdzik, B.; Chabros, A. The Influence of Lignin Diversity on the Structural and Thermal Properties of Polymeric Microspheres Derived from Lignin, Styrene, and/or Divinylbenzene. Materials 2019, 12, 2847. https://doi.org/10.3390/ma12182847
Goliszek M, Podkościelna B, Sevastyanova O, Gawdzik B, Chabros A. The Influence of Lignin Diversity on the Structural and Thermal Properties of Polymeric Microspheres Derived from Lignin, Styrene, and/or Divinylbenzene. Materials. 2019; 12(18):2847. https://doi.org/10.3390/ma12182847
Chicago/Turabian StyleGoliszek, Marta, Beata Podkościelna, Olena Sevastyanova, Barbara Gawdzik, and Artur Chabros. 2019. "The Influence of Lignin Diversity on the Structural and Thermal Properties of Polymeric Microspheres Derived from Lignin, Styrene, and/or Divinylbenzene" Materials 12, no. 18: 2847. https://doi.org/10.3390/ma12182847
APA StyleGoliszek, M., Podkościelna, B., Sevastyanova, O., Gawdzik, B., & Chabros, A. (2019). The Influence of Lignin Diversity on the Structural and Thermal Properties of Polymeric Microspheres Derived from Lignin, Styrene, and/or Divinylbenzene. Materials, 12(18), 2847. https://doi.org/10.3390/ma12182847








