Developing 9,10-anthracene Derivatives: Optical, Electrochemical, Thermal, and Electrical Characterization
Abstract
1. Introduction
2. Results and Discussion
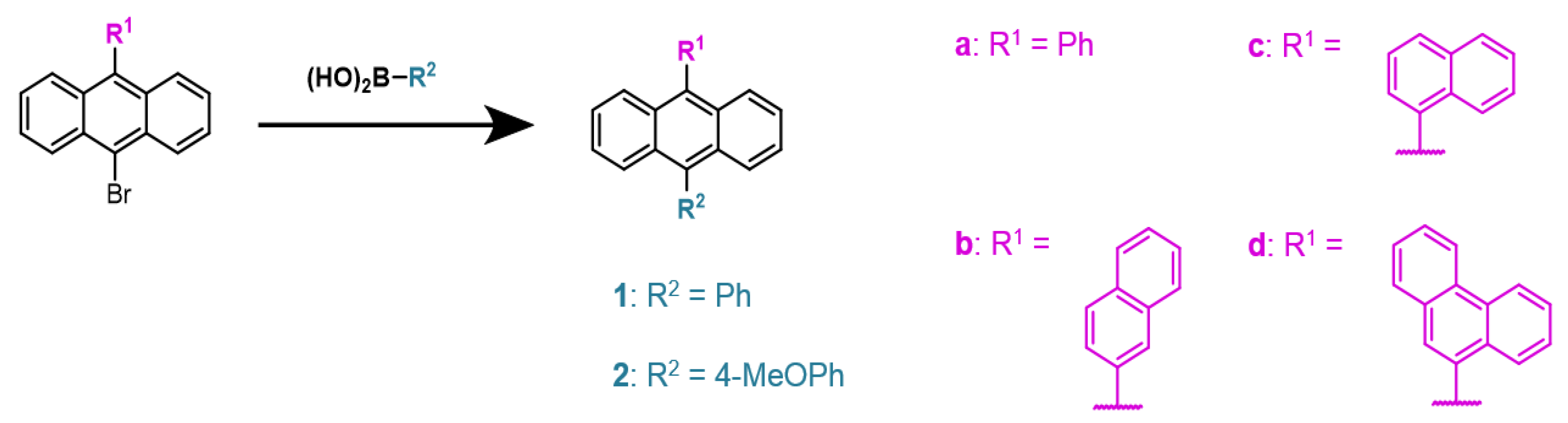
2.1. Synthesis of 9,10-Disubstiuted Anthracenes
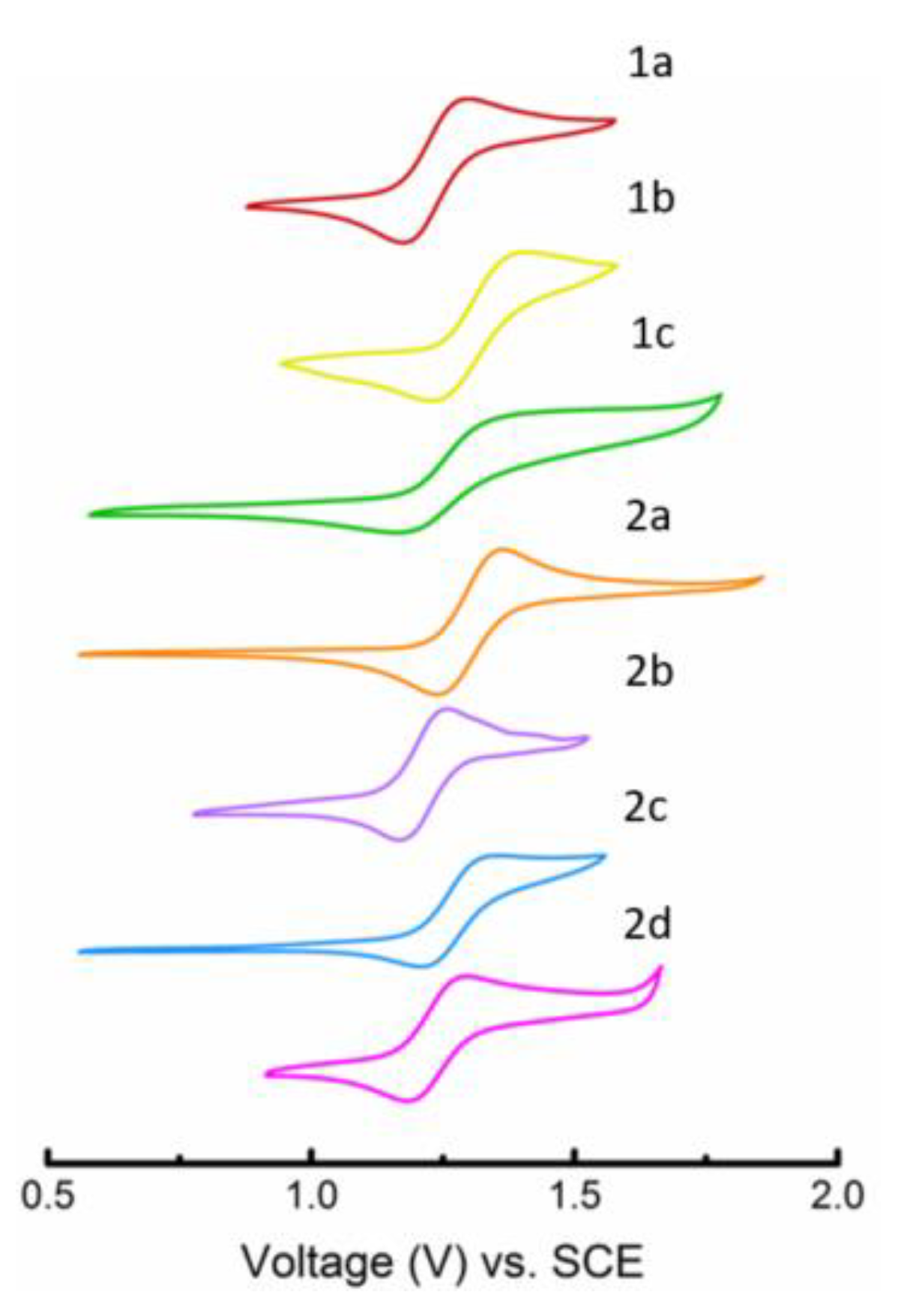
2.2. Optical and Electrochemical Properties
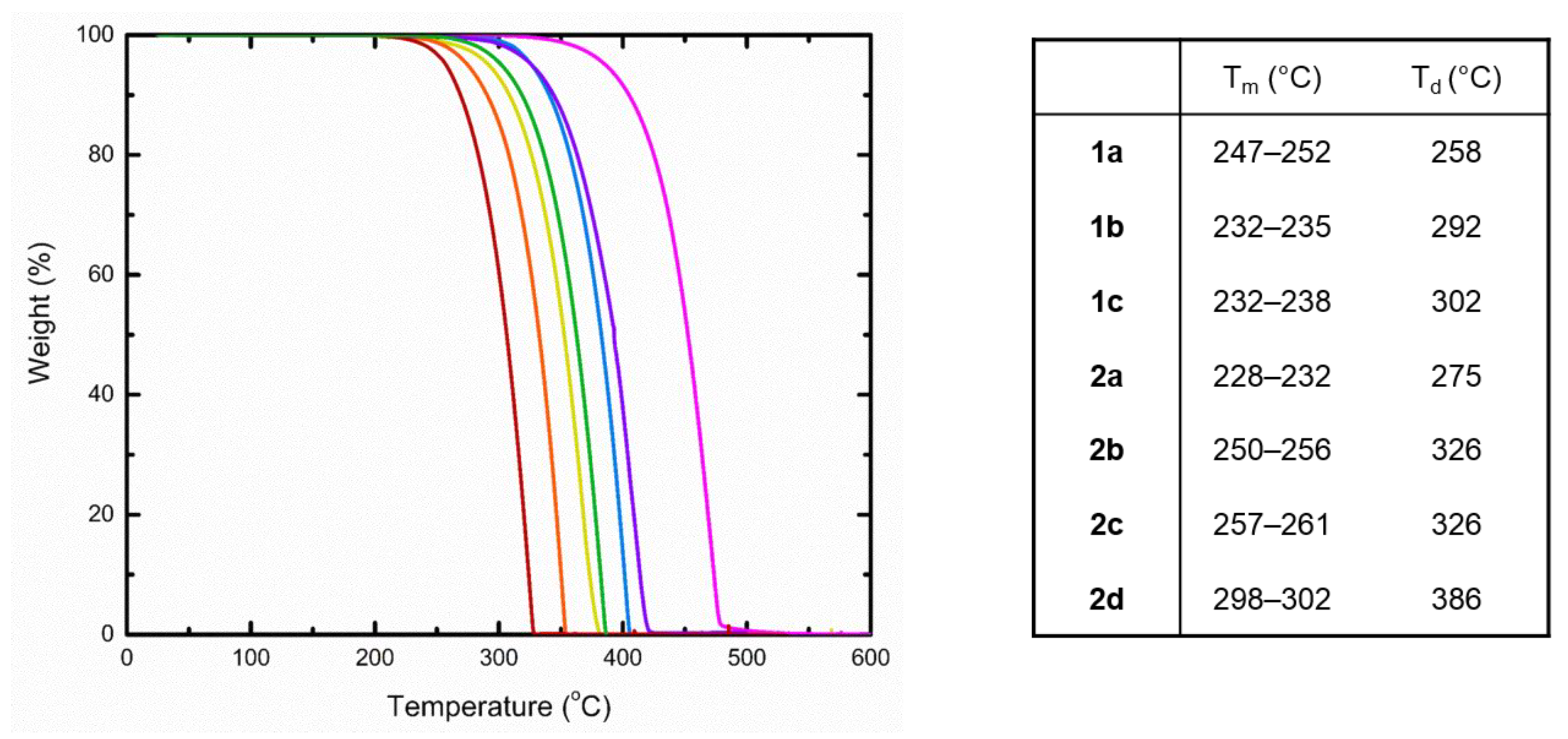
2.3. Thermogravimetric Analysis
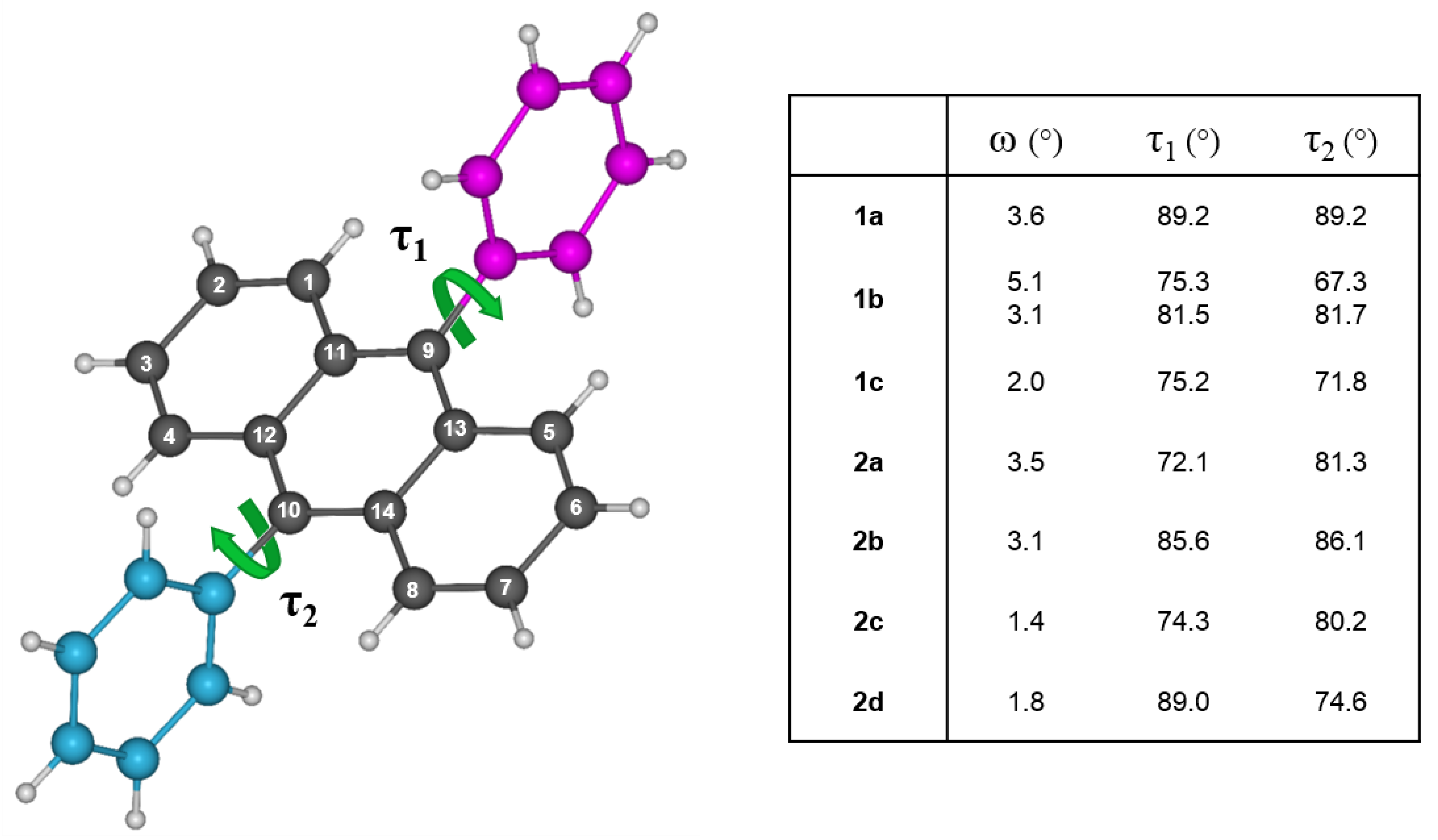
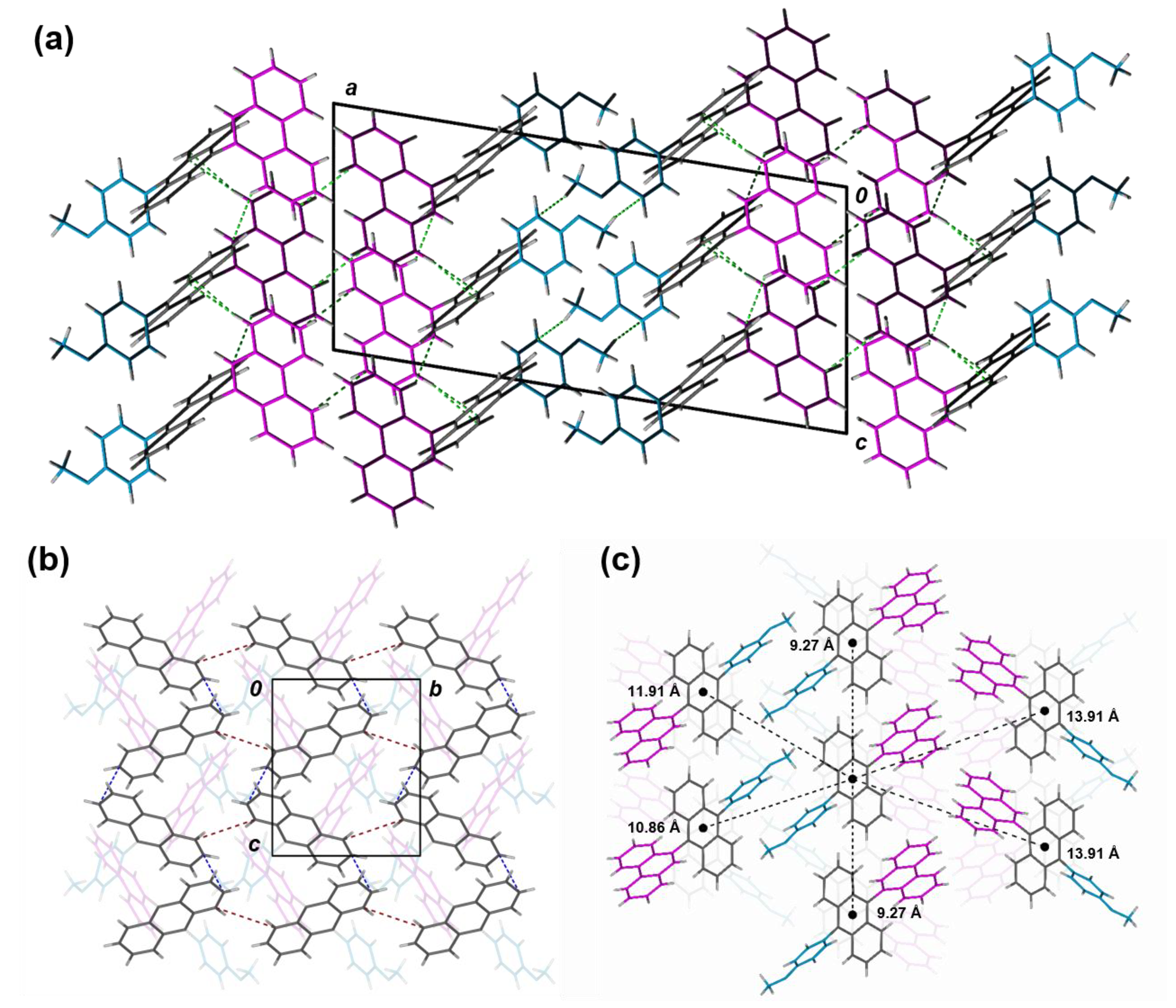
2.4. Single Crystal X-Ray Diffraction
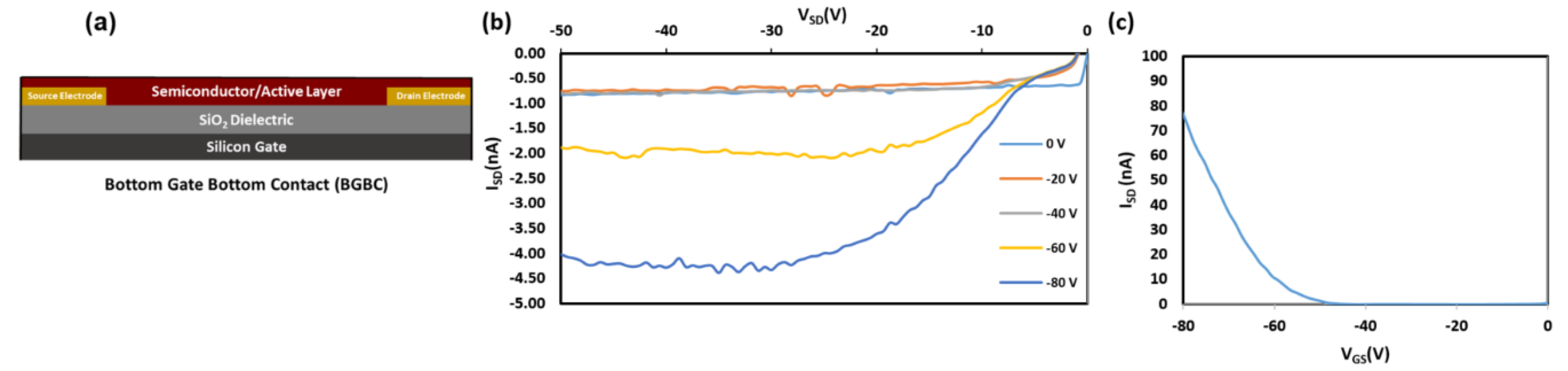
2.5. OTFT Performance
3. Conclusions
4. Materials and Methods
4.1. General Methods and Procedures
4.1.1. Preparation of 9,10-diphenylanthracene (1a)
4.1.2. Preparation of 9-phenyl-10-(1-naphthalenyl)-anthracene (1b)
4.1.3. Preparation of 9-phenyl-10-(2-naphthalenyl)-anthracene (1c)
4.1.4. Preparation of 9-(4-methoxyphenyl)-10-phenylanthracene (2a)
4.1.5. Preparation of 9-(4-(methoxyphenyl))-10-(1-naphthalenyl)anthracene (2b)
4.1.6. Preparation of 9-(4-methoxyphenyl)-10-(naphthalen-2-yl)anthracene (2c)
4.1.7. Preparation of 9-(4-methoxyphenyl)-10-(phenanthrene-10-yl)anthracene (2d)
4.2. Electrochemistry
4.3. Thermogravimetric Analysis
4.4. Crystallographic Characterization
4.5. Electrical Characterization
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Chang, Y.L.; Song, Y.; Wang, Z.; Helander, M.G.; Qiu, J.; Chai, L.; Liu, Z.; Scholes, G.D.; Lu, Z. Highly efficient warm white organic light-emitting diodes by triplet exciton conversion. Adv. Funct. Mater. 2013, 23, 705–712. [Google Scholar] [CrossRef]
- Wang, Z.B.; Helander, M.G.; Qiu, J.; Puzzo, D.P.; Greiner, M.T.; Hudson, Z.M.; Wang, S.; Liu, Z.W.; Lu, Z.H. Unlocking the full potential of organic light-emitting diodes on flexible plastic. Nat. Photonics 2011, 5, 753. [Google Scholar] [CrossRef]
- Piliego, C.; Jarzab, D.; Gigli, G.; Chen, Z.; Facchetti, A.; Loi, M.A. High Electron mobility and ambient stability in solution-processed perylene-based organic field-effect transistors. Adv. Mater. 2009, 21, 1573–1576. [Google Scholar] [CrossRef]
- Bettinger, C.J.; Becerril, H.A.; Kim, D.H.; Lee, B.L.; Lee, S.; Bao, Z. Microfluidic Arrays for rapid characterization of organic thin-film transistor performance. Adv. Mater. 2011, 23, 1257–1261. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.Z.; Shi, M.M.; Aernouts, T.; Wang, M.; Borghs, G.; Heremans, P. A Novel organic N-Type material: Fluorinated perylene diimide. Sol. Energy Mater. Sol. Cells 2005, 87, 521–527. [Google Scholar] [CrossRef]
- Tian, H.; Han, Y.; Bao, C.; Yan, D.; Geng, Y.; Wang, F. An asymmetric oligomer based on thienoacene for solution processed crystal organic thin-film transistors. Chem. Commun. 2012, 48, 3557–3559. [Google Scholar] [CrossRef] [PubMed]
- Ma, R.-Q.; Hewitt, R.; Rajan, K.; Silvernail, J.; Urbanik, K.; Hack, M.; Brown, J.J. Flexible active-matrix OLED displays: Challenges and progress. J. Soc. Inf. Disp. 2007, 16, 169. [Google Scholar] [CrossRef]
- Glawe, A.; Eggerath, D.; Schäfer, F. Printing versus Coating - What Will Be the Future Production Technology for Printed Electronics? In AIP Conference Proceedings; American Institute of Physics: College Park, MD, USA, 2015; Volume 1646, pp. 87–90. [Google Scholar]
- Boileau, N.T.; Melville, O.A.; Mirka, B.; Cranston, R.; Lessard, B.H. P and N type copper phthalocyanines as effective semiconductors in organic thin-film transistor based DNA biosensors at elevated temperatures. Rsc Adv. 2019, 9, 2133–2142. [Google Scholar] [CrossRef]
- Chen, M.; Yan, L.; Zhao, Y.; Murtaza, I.; Meng, H.; Huang, W. Anthracene-based semiconductors for organic field-effect transistors. J. Mater. Chem. C 2018, 6, 7416–7444. [Google Scholar] [CrossRef]
- Xie, H.; Cheng, C.Y.; Li, L.; Deng, X.Y.; Yang, K.K.; Wang, Y.Z. Integrating shape-memory technology and photo-imaging on a polymer platform for a high-security information storage medium. J. Mater. Chem. C 2018, 6, 10422–10427. [Google Scholar] [CrossRef]
- Kommandeur, J. Photoconductivity in organic single crystals. J. Phys. Chem. Solids 1961, 22, 339–349. [Google Scholar] [CrossRef]
- LeBlanc, O.H. Band Structure and Transport of Holes and Electrons in Anthracene. J. Chem. Phys. 1961, 35, 1275–1280. [Google Scholar] [CrossRef]
- Quinn, J.T.E.; Zhu, J.; Li, X.; Wang, J.; Li, Y. Recent progress in the development of n-type organic semiconductors for organic field effect transistors. J. Mater. Chem. C 2017, 5, 8654–8681. [Google Scholar] [CrossRef]
- Dong, H.; Fu, X.; Liu, J.; Wang, Z.; Hu, W. 25th anniversary article: Key points for high-mobility organic field-effect transistors. Adv. Mater. 2013, 25, 6158–6183. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhao, G.; Zhen, Y.; Tu, Z.; He, P.; Yi, Y.; Dong, H.; Hu, W. The position effect of an ethynyl spacer on the carrier mobility of anthracene derivatives. J. Mater. Chem. C 2015, 3, 5368–5371. [Google Scholar] [CrossRef]
- Chaari, M.; Kelemen, Z.; Planas, J.G.; Teixidor, F.; Choquesillo-Lazarte, D.; Ben Salah, A.; Viñas, C.; Núñez, R. Photoluminescence in: M -Carborane-Anthracene Triads: A combined experimental and computational study. J. Mater. Chem. C 2018, 6, 11336–11347. [Google Scholar] [CrossRef]
- Liu, J.; Dong, H.; Wang, Z.; Ji, D.; Cheng, C.; Geng, H.; Zhang, H.; Zhen, Y.; Jiang, L.; Fu, H.; et al. Thin film Field-Effect transistors of 2,6-Diphenyl Anthracene (DPA). Chem. Commun. 2015, 51, 11777–11779. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, H.; Dong, H.; Meng, L.; Jiang, L.; Jiang, L.; Wang, Y.; Yu, J.; Sun, Y.; Hu, W.; et al. High mobility emissive organic semiconductor. Nat. Commun. 2015, 6, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Zhao, Y.; Yu, H.; Hu, Z.; He, Y.; Li, A.; Goto, O.; Yan, C.; Chen, T.; Chen, R.; et al. Influence of heteroatoms on the charge mobility of anthracene derivatives. J. Mater. Chem. C 2016, 4, 3517–3522. [Google Scholar] [CrossRef]
- Li, A.; Yan, L.; Liu, M.; Murtaza, I.; He, C.; Zhang, D.; He, Y.; Meng, H. Highly responsive phototransistors based on 2,6-bis(4-methoxyphenyl)anthracene single crystal. J. Mater. Chem. C 2017, 5, 5304–5309. [Google Scholar] [CrossRef]
- Payne, M.M.; Parkin, S.R.; Anthony, J.E.; Kuo, C.C.; Jackson, T.N. Organic field-effect transistors from solution-deposited functionalized acenes with mobilities as high as 1 Cm2/V·s. J. Am. Chem. Soc. 2005, 127, 4986–4987. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liu, J.; Zhang, Z.; Xu, C.; Li, Q.; Zhou, K.; Dong, H.; Zhang, X.; Hu, W. Enhancing field-effect mobility and maintaining solid-state emission by incorporating 2,6-diphenyl substitution to 9,10-bis(phenylethynyl)anthracene. J. Mater. Chem. C 2017, 5, 2519–2523. [Google Scholar] [CrossRef]
- Zafar, M.N.; Mohsin, M.A.; Danish, M.; Nazar, M.F.; Murtaza, S. Palladium catalyzed heck − mizoroki and suzuki − miyaura coupling reactions (Review). Russ. J. Coord. Chem. 2014, 40, 781–800. [Google Scholar] [CrossRef]
- Smith, G.B.; Dezeny, G.C.; Hughes, D.L.; King, A.O.; Verhoeven, T.R. Mechanistic studies of the suzuki cross-coupling reaction. J. Org. Chem. 1994, 59, 8151–8156. [Google Scholar] [CrossRef]
- Wan, Z.; Qi, W.; Xing, F.; Li, Y. Anthracene-Based Derivatives: Synthesis, Photophysical Properties and Electrochemical Properties. Chem. Res. Chin. Univ. 2017, 33, 603–610. [Google Scholar]
- Kim, R.; Lee, S.; Kim, K.H.; Lee, Y.J.; Kwon, S.K.; Kim, J.J.; Kim, Y.H. Extremely deep blue and highly efficient non-doped organic light emitting diodes using an asymmetric anthracene derivative with a xylene unit. Chem. Commun. 2013, 49, 4664–4666. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.Q.; Xu, C.; Wang, W.Z.; Duan, L.M.; Li, Z.; Zhao, B.T.; Ji, B.M. High-color-purity and high-efficiency non-doped deep-blue electroluminescent devices based on novel anthracene derivatives. New J. Chem. 2012, 36, 662–667. [Google Scholar] [CrossRef]
- Serevičius, T.; Komskis, R.; Adomėnas, P.; Adomėnienė, O.; Jankauskas, V.; Gruodis, A.; Juršėnas, S. Non-symmetric 9, 10-diphenylanthracene-based deep-blue emitters with enhanced charge transport properties. Phys. Chem. Chem. Phys. 2014, 16, 7089–7101. [Google Scholar] [CrossRef]
- Roberto, P.; Pietro, T.; Carlo, S.; Paolo, P.; Roberta, D.A. Synthesis and Photophysical Properties of 9,10-Disubstituted Anthracenes. Sci. Res. 2015, 6, 943–952. [Google Scholar]
- Gidron, O.; Dadvand, A.; Wei-Hsin Sun, E.; Chung, I.; Shimon, L.J.W.; Bendikov, M.; Perepichka, D.F. Oligofuran-containing molecules for organic electronics. J. Mater. Chem. C 2013, 1, 4358–4367. [Google Scholar] [CrossRef]
- Dadvand, A.; Sun, W.H.; Moiseev, A.G.; Bélanger-Gariépy, F.; Rosei, F.; Meng, H.; Perepichka, D.F. 1,5-, 2,6- and 9,10-distyrylanthracenes as luminescent organic semiconductors. J. Mater. Chem. C 2013, 1, 2817–2825. [Google Scholar] [CrossRef]
- Usta, H.; Kim, C.; Wang, Z.; Lu, S.; Huang, H.; Facchetti, A.; Marks, T.J. Anthracenedicarboximide-based semiconductors for air-stable, n-channel organic thin-film transistors: Materials design, synthesis, and structural characterization. J. Mater. Chem. 2012, 22, 4459–4472. [Google Scholar] [CrossRef]
- Chen, M.; Zhao, Y.; Yan, L.; Yang, S.; Zhu, Y.; Murtaza, I.; He, G.; Meng, H.; Huang, W. A unique blend of 2-fluorenyl-2-anthracene and 2-anthryl-2-anthracence showing white emission and high charge mobility. Angew. Chem. - Int. Ed. 2017, 56, 722–727. [Google Scholar] [CrossRef]
- Dadvand, A.; Moiseev, A.G.; Sawabe, K.; Sun, W.H.; Djukic, B.; Chung, I.; Takenobu, T.; Rosei, F.; Perepichka, D.F. Maximizing field-effect mobility and solid-state luminescence in organic semiconductors. Angew. Chem. - Int. Ed. 2012, 51, 3837–3841. [Google Scholar] [CrossRef]
- Ding, S.Y.; Gao, J.; Wang, Q.; Zhang, Y.; Song, W.G.; Su, C.Y.; Wang, W. Construction of covalent organic framework for catalysis: Pd/cof-lzu1 in suzuki-miyaura coupling reaction. J. Am. Chem. Soc. 2011, 133, 19816–19822. [Google Scholar] [CrossRef]
- Aranzaes, J.R.; Daniel, M.-C.; Astruc, D. Metallocenes as references for the determination of redox potentials by cyclic voltammetry—permethylated iron and cobalt sandwich complexes, inhibition by polyamine dendrimers, and the role of hydroxy-containing ferrocenes. Can. J. Chem. 2006, 84, 288–299. [Google Scholar] [CrossRef]
- Li, Y.; Cao, Y.; Gao, J.; Wang, D.; Yu, G.; Heeger, A.J. Electrochemical properties of luminescent polymers and polymer light-emitting electrochemical cells. Synth. Met. 2002, 99, 243–248. [Google Scholar] [CrossRef]
- Dang, M.T.; Grant, T.M.; Yan, H.; Seferos, D.S.; Lessard, B.H.; Bender, T.P. Bis(Tri-n-Alkylsilyl Oxide) silicon phthalocyanines: A start to establishing a structure property relationship as both ternary additives and non-fullerene electron acceptors in bulk heterojunction organic photovoltaic devices. J. Mater. Chem. A 2017, 5, 12168–12182. [Google Scholar] [CrossRef]
- Lohrman, J.; Zhang, C.; Zhang, W.; Ren, S. Semiconducting carbon nanotube and covalent organic polyhedron-c60nanohybrids for light harvesting. Chem. Commun. 2012, 48, 8377–8379. [Google Scholar] [CrossRef]
- Silvestri, F.; Marrocchi, A.; Seri, M.; Kim, C.; Marks, T.J.; Facchetti, A.; Taticchi, A. Solution-processable low-molecular weight extended arylacetylenes: Versatile p-type semiconductors for field-effect transistors and bulk heterojunction solar cells. J. Am. Chem. Soc. 2010, 132, 6108–6123. [Google Scholar] [CrossRef]
- Schmidt, R.; Göttling, S.; Leusser, D.; Stalke, D.; Krause, A.M.; Würthner, F. Highly Soluble Acenes as semiconductors for thin film transistors. J. Mater. Chem. 2006, 16, 3708–3714. [Google Scholar] [CrossRef]
- APEX Softward Suite v 2010; Bruker AXS Inc: Madison Wisconsin, WI, USA, 2010.
- Blessing, R.H. An empirical correction for absorption anisotropy. Acta Crystallogr. Sect. A: Found. Crystallogr. 1995, 51, 33–38. [Google Scholar] [CrossRef]
- Hübschle, C.B.; Sheldrick, G.M.; Dittrich, B. ShelXle: A Qt graphical user interface for SHELXL. J. Appl. Crystallogr. 2011, 44, 1281–1284. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. Acta Crystallogr. Sect. A: Found. Crystallogr. 2008, 64, 112–122. [Google Scholar] [CrossRef]
- Brixi, S.; Melville, O.A.; Boileau, N.T.; Lessard, B.H. The influence of air and temperature on the performance of pbdb-t and p3ht in organic thin film transistors. J. Mater. Chem. C 2018, 6, 11972–11979. [Google Scholar] [CrossRef]
| Stokes Shift (nm) | ||||||
|---|---|---|---|---|---|---|
| 1a | 1.241 | −5.68 | 342, 358, 376, 397 | 2.98 | 420, 435 | 23 |
| 1b | 1.323 | −5.73 | 341, 357, 376, 396 | 2.98 | 414, 430 | 18 |
| 1c | 1.377 | −5.73 | 341, 357, 376, 396 | 2.99 | 420, 435 | 24 |
| 2a | 1.303 | −5.69 | 339, 358, 376, 396 | 2.96 | 421, 431 | 25 |
| 2b | 1.215 | −5.59 | 343, 359, 377, 398 | 2.96 | 405, 430 | 7 |
| 2c | 1.279 | −5.68 | 343, 358, 377, 398 | 2.97 | 428 | 30 |
| 2d | 1.237 | −5.61 | 342, 358, 377, 397 | 2.97 | 427 | 30 |
| π–π Distance (Å)b | Ion/off | μavg (cm2 V−1 s−1) | μmax (cm2 V−1 s−1) | VT,Avg (V) | VT,max (V) | |
|---|---|---|---|---|---|---|
| 1a | 3.67, 3.79, 3.92 | |||||
| 1b | 3.46, 3.58, 3.60, 3.71, 3.74 | 102 | 5.31 × 10−6 | 7.26 × 10−6 | −37 | −21 |
| 1c | 3.72, 3.88 | |||||
| 2a | 3.63, 3.90 | 102 | 8.08 × 10−7 | 4.44 × 10−6 | −41 | −14 |
| 2bc | 4.82 | |||||
| 2c | 3.71, 3.91 | 101 | 1.48 × 10−7 | 1.91 × 10−6 | −29 | −13 |
| 2d | 3.54, 3.97 | 102 | 6.68 × 10−6 | 7.07 × 10−6 | −43 | −34 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vorona, M.Y.; Yutronkie, N.J.; Melville, O.A.; Daszczynski, A.J.; Agyei, K.T.; Ovens, J.S.; Brusso, J.L.; Lessard, B.H. Developing 9,10-anthracene Derivatives: Optical, Electrochemical, Thermal, and Electrical Characterization. Materials 2019, 12, 2726. https://doi.org/10.3390/ma12172726
Vorona MY, Yutronkie NJ, Melville OA, Daszczynski AJ, Agyei KT, Ovens JS, Brusso JL, Lessard BH. Developing 9,10-anthracene Derivatives: Optical, Electrochemical, Thermal, and Electrical Characterization. Materials. 2019; 12(17):2726. https://doi.org/10.3390/ma12172726
Chicago/Turabian StyleVorona, Mikhail Y., Nathan J. Yutronkie, Owen A. Melville, Andrew J. Daszczynski, Kwame T. Agyei, Jeffrey S. Ovens, Jaclyn L. Brusso, and Benoît H. Lessard. 2019. "Developing 9,10-anthracene Derivatives: Optical, Electrochemical, Thermal, and Electrical Characterization" Materials 12, no. 17: 2726. https://doi.org/10.3390/ma12172726
APA StyleVorona, M. Y., Yutronkie, N. J., Melville, O. A., Daszczynski, A. J., Agyei, K. T., Ovens, J. S., Brusso, J. L., & Lessard, B. H. (2019). Developing 9,10-anthracene Derivatives: Optical, Electrochemical, Thermal, and Electrical Characterization. Materials, 12(17), 2726. https://doi.org/10.3390/ma12172726







