Enamel Matrix Derivative and Autogenous Bone Graft for Periodontal Regeneration of Intrabony Defects in Humans: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Focused Question
2.2. Search Strategy
2.3. Inclusion Criteria
- English language.
- Randomized controlled trials (RCTs) comparing a combination of EMD and autogenous bone graft with the EMD alone for the treatment of periodontal intrabony defects.
- Studies including patients with advanced chronic or aggressive periodontitis with the presence of at least one intrabony defect with a probing depth of at least 6 mm and an intrabony component of at least 3 mm as detected on the radiographs.
- Studies with at least 6-month follow-up after surgery for the radiographic and clinical evaluation.
2.4. Exclusion Criteria
- Studies not reporting clinical/radiographical data.
- Studies that considered the use of EMD in combination with other biomaterials.
- Studies comparing the use of EMD in combination with autogenous bone graft with open-flap debridement, guided tissue regeneration, or autogenous bone graft alone.
- Preclinical studies, case series, case reports, retrospective studies, letters to the editor, technical reports, narrative reviews, conference abstracts.
2.5. Data Extraction and Analysis
2.6. Outcome Measures
- Change in clinical attachment level (CAL) or relative attachment level (RAL).
- Change in probing depth (PD);
- Change in gingival recession (REC).
2.7. Methodological Quality Assessment
2.8. Data Analysis
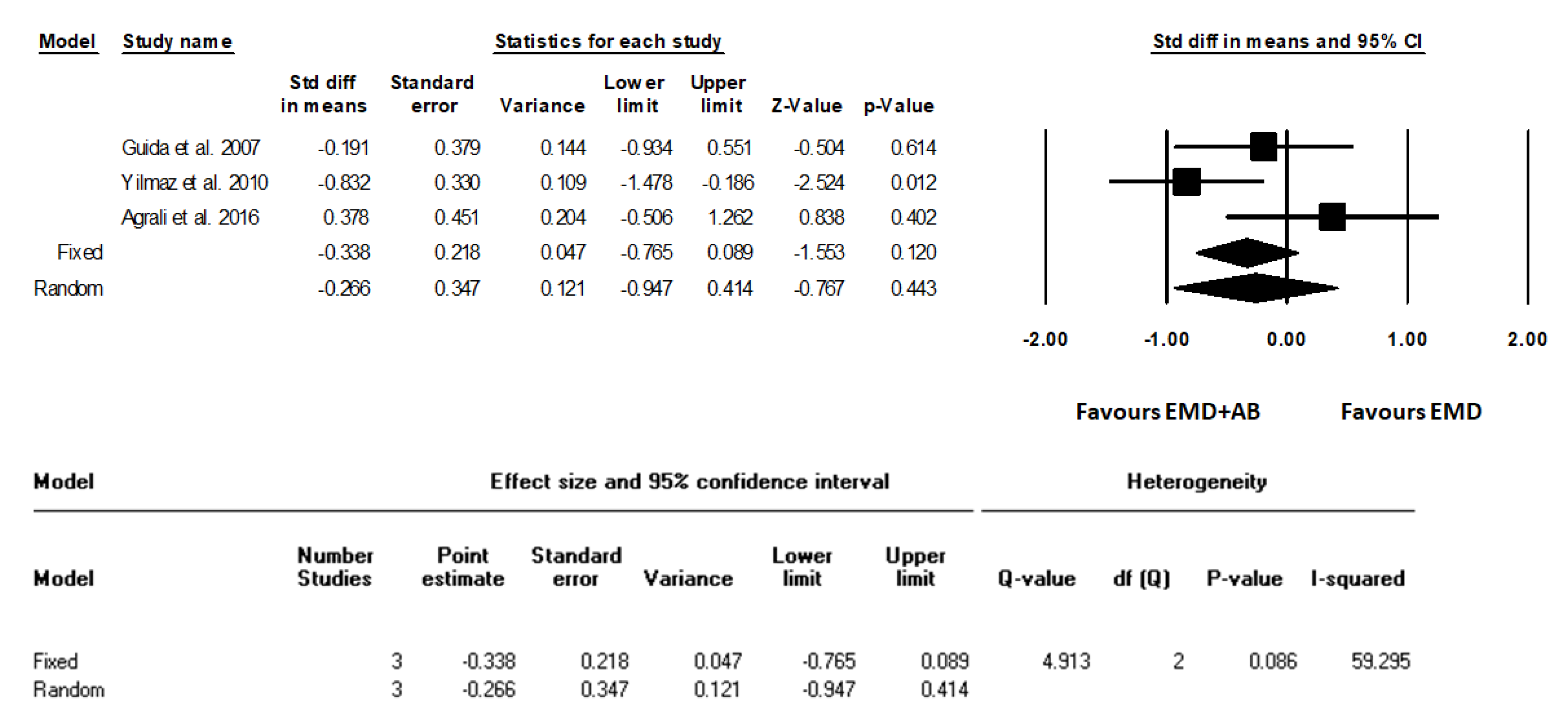
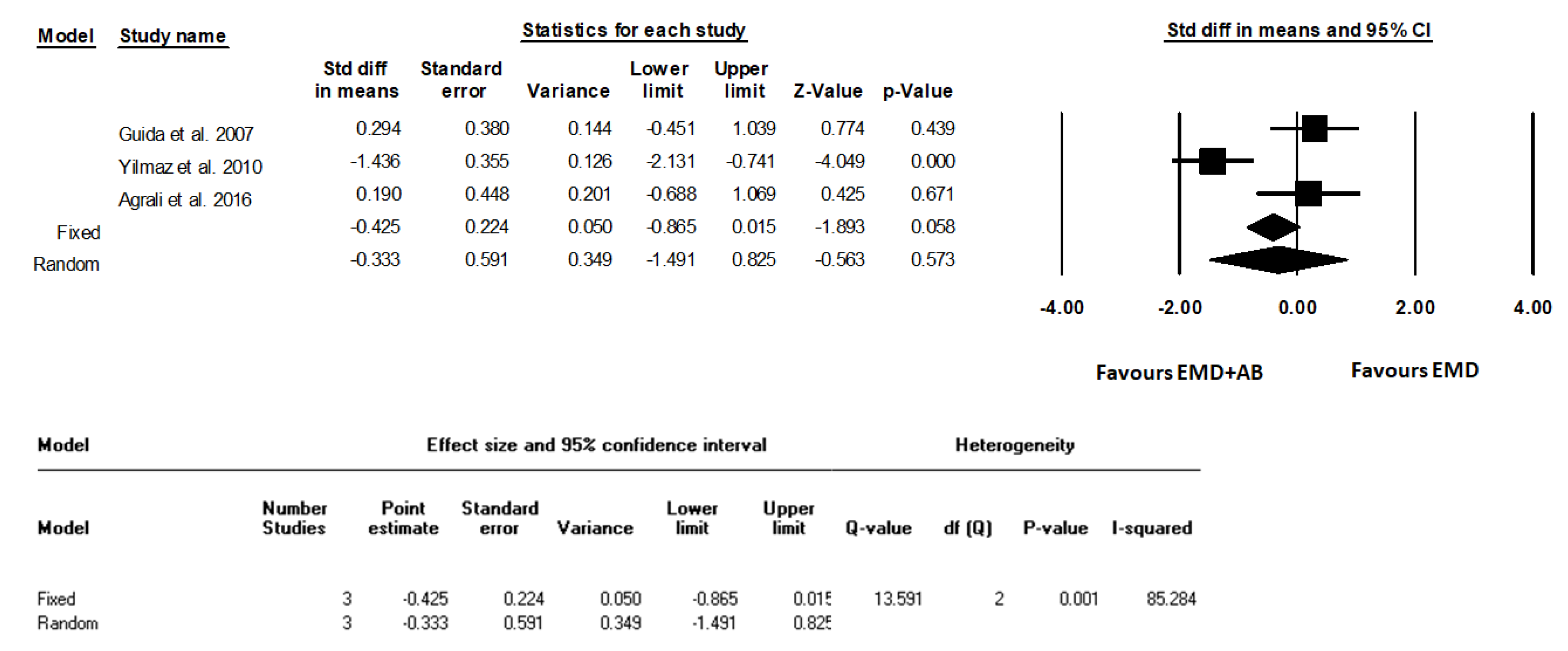
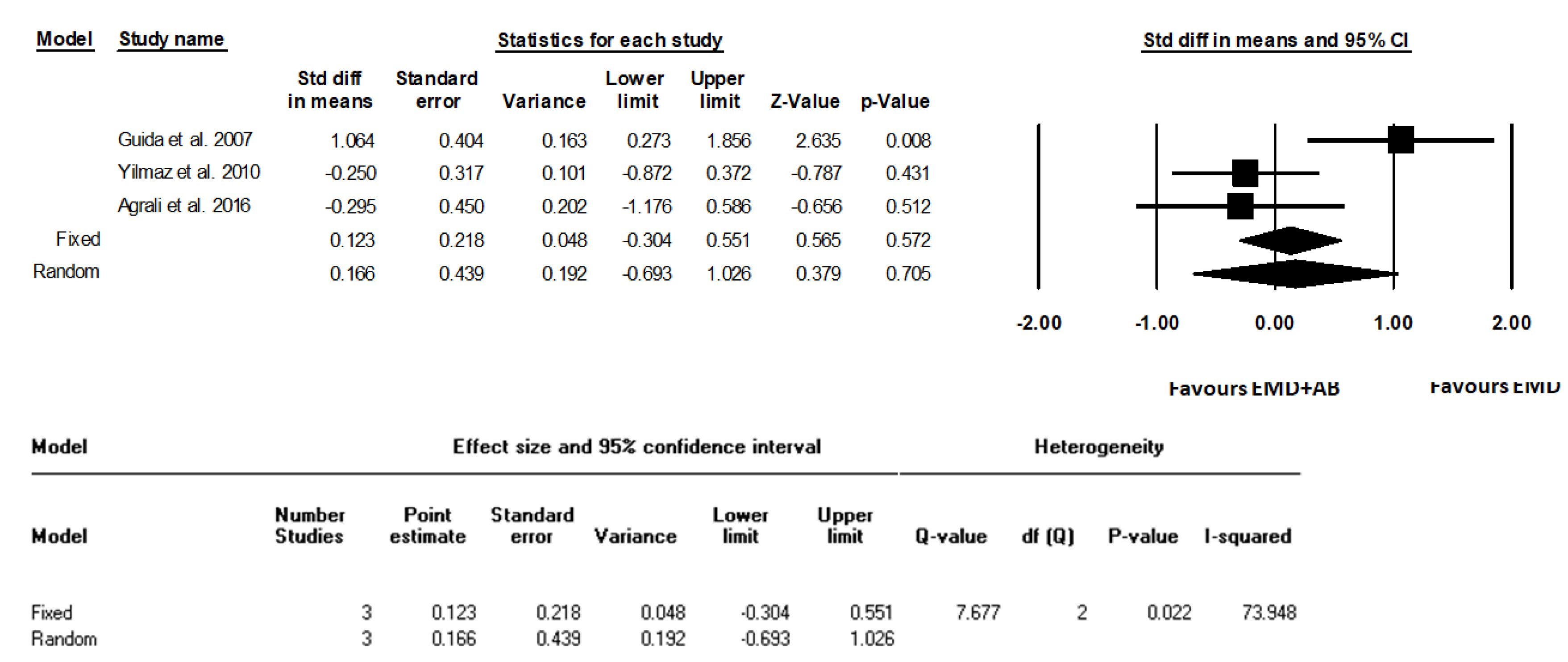
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Bartold, P.M.; Van Dyke, T.E. A host-mediated disruption of microbial homeostasis. Unlearning learned concepts. Periodontology 2000, 62, 203–217. [Google Scholar] [CrossRef] [PubMed]
- Cortellini, P.; Tonetti, M.S. Regenerative Periodontal Therapy. In Clinical Periodontology and Implant Dentistry (sixth edition); Blackwell-Munksgaard: Hoboken, NJ, USA, 2015; pp. 901–918. [Google Scholar]
- Scantlebury, T.V. 1982–1992: A Decade of Technology Development for Guided Tissue Regeneration. J. Periodontol. 1993, 64, 1129–1137. [Google Scholar] [CrossRef] [PubMed]
- Needleman, I.; Worthington, H.V.; Giedrys-Leeper, E.; Tucker, R. WITHDRAWN: Guided tissue regeneration for periodontal infra-bony defects. Cochrane Data. Syst. Rev. 2019, 5, CD001724. [Google Scholar] [CrossRef]
- Guida, L.; Annunziata, M.; Carinci, F.; Di Feo, A.; Passaro, I.; Oliva, A. In vitro biologic response of human bone marrow stromal cells to enamel matrix derivative. J. Periodontol. 2007, 78, 2190–2196. [Google Scholar] [CrossRef] [PubMed]
- Carinci, F.; Piattelli, A.; Guida, L.; Perrotti, V.; Laino, G.; Oliva, A.; Annunziata, M.; Palmieri, A.; Pezzetti, F. Effects of Emdogain on osteoblast gene expression. Oral Dis. 2006, 12, 329–342. [Google Scholar] [CrossRef] [PubMed]
- Hammarström, L. Enamel matrix, cementum development and regeneration. J. Clin. Periodontol. 1997, 24, 658–668. [Google Scholar] [CrossRef] [PubMed]
- Lekovic, V.; Camargo, P.M.; Weinlaender, M.; Nedic, M.; Aleksic, Z.; Kenney, E.B. A comparison between enamel matrix proteins used alone or in combination with bovine porous bone mineral in the treatment of intrabony periodontal defects in humans. J. Periodontol. 2000, 71, 1110–1116. [Google Scholar] [CrossRef]
- Velasquez-Plata, D.; Scheyer, E.T.; Mellonig, J.T. Clinical comparison of an esame matrix derivative used alone or in combination with a bovine-derived xenograft for the treatment of periodontal osseous defects in humans. J. Periodontol. 2002, 73, 433–440. [Google Scholar] [CrossRef]
- Sculean, A.; Windisch, P.; Keglevich, T.; Chiantella, G.C.; Gera, I.; Donos, N. Clinical and histologic evaluation of human intrabony defects treated with an enamel matrix protein derivative combined with a bovine-derived xenograft. Int. J. Periodontics Restorative Dent. 2003, 23, 47–55. [Google Scholar]
- Sculean, A.; Windisch, P.; Szendröi-Kiss, D.; Horváth, A.; Rosta, P.; Becker, J.; Gera, I.; Schwarz, F. Clinical and histologic evaluation of an enamel matrix derivative combined with a biphasic calcium phosphate for the treatment of human intrabony periodontal defects. J. Periodontol. 2008, 79, 1991–1999. [Google Scholar] [CrossRef]
- Zucchelli, G.; Amore, C.; Montebugnoli, L.; De Sanctis, M. Enamel matrix proteins and bovine porous bone mineral in the treatment of intrabony defects: A comparative controlled clinical trial. J. Periodontol. 2003, 74, 1725–1735. [Google Scholar] [CrossRef] [PubMed]
- Gurinsky, B.S.; Mills, M.P.; Mellonig, J.T. Clinical evaluation of demineralised freeze-dried bone allograft and enamel matrix derivative versus enamel matrix derivative alone for the treatment of periodontal osseous defects in humans. J. Periodontol. 2004, 75, 1309–1318. [Google Scholar] [CrossRef] [PubMed]
- Kuru, B.; Yilmaz, S.; Argin, K.; Noyan, U. Enamel matrix derivative alone or in combination with a bioactive glass in wide intrabony defects. Clin. Oral Investig. 2006, 10, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Trombelli, L.; Annunziata, M.; Belardo, S.; Farina, R.; Scabbia, A.; Guida, L. Autogenous bone graft in conjunction with enamel matrix derivative in the treatment of deep periodontal intra-osseous defects: A report of 13 consecutively treated patients. J. Clin. Periodontol. 2006, 33, 69–75. [Google Scholar] [CrossRef]
- Urist, M.R. Bone formation by autoinduction. Science 1965, 150, 893–899. [Google Scholar] [CrossRef] [PubMed]
- Miron, R.J.; Hedbom, E.; Saulacic, N.; Zhang, Y.; Sculean, A.; Bosshardt, D.D.; Buser, D. Osteogenic potential of autogenous bone grafts harvested with four different surgical techniques. J. Dent. Res. 2011, 90, 1428–1433. [Google Scholar] [CrossRef] [PubMed]
- Guida, L.; Annunziata, M.; Belardo, S.; Farina, R.; Scabbia, A.; Trombelli, L. Effect of autogenous cortical bone particulate in conjunction with enamel matrix derivative in the treatment of periodontal intraosseous defects. J. Periodontol. 2007, 78, 231–238. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Prisma Group: Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Int. J. Surg. 2010, 8, 336–341. [Google Scholar] [CrossRef]
- Schardt, C.; Adams, M.B.; Owens, T.; Keitz, S.; Fontelo, P. Utilization of the PICO framework to improve searching PubMed for clinical questions. BMC Med. Inform. Decis. Mak. 2007, 15, 7–16. [Google Scholar] [CrossRef]
- Van der Weijden, F.; Dell’acqua, F.; Slot, D.E. Alveolar bone dimensional changes of post-extraction sockets in humans: A systematic review. J. Clin. Periodontol. 2009, 36, 1048–1058. [Google Scholar] [CrossRef]
- Yilmaz, S.; Cakar, G.; Yildirim, B.; Sculean, A. Healing of two and three wall intrabony periodontal defects following treatment with an enamel matrix derivative combined with autogenous bone. J. Clin. Periodontol. 2010, 37, 544–550. [Google Scholar] [CrossRef]
- Agrali, Ö.B.; Kuru, B.E.; Yarat, A.; Kuru, L. Evaluation of gingival crevicular fluid transforming growth factor-β1 level after treatment of intrabony periodontal defects with enamel matrix derivatives and autogenous bone graft: A randomized controlled clinical trial. Niger. J. Clin Pract. 2016, 19, 535–543. [Google Scholar] [CrossRef]
- Matarasso, M.; Iorio-Siciliano, V.; Blasi, A.; Ramaglia, L.; Salvi, G.E.; Sculean, A. Enamel matrix derivative and bone grafts for periodontal regeneration of intrabony defects. A systematic review and meta-analysis. Clin. Oral Investig. 2015, 19, 1581–1593. [Google Scholar] [CrossRef]
- Miron, R.J.; Gruber, R.; Hedbom, E.; Saulacic, N.; Zhang, Y.; Sculean, A.; Buser, D. Impact of bone harvesting techniques on cell viability and the release of growth factors of autografts. Clin. Implant Dent. Relat. Res. 2013, 15, 481–489. [Google Scholar] [CrossRef]
- Ferrarotti, F.; Romano, F.; Quirico, A.; Di Bella, M.; Pallotti, S.; Aimetti, M. Effectiveness of Enamel Matrix Derivative in Conjunction with Particulate Autologous Bone in the Treatment of Noncontained Intrabony Defects: A 2-Year Prospective Case Series. Int. J. Periodontics Restorative Dent. 2018, 38, 673–680. [Google Scholar] [CrossRef]
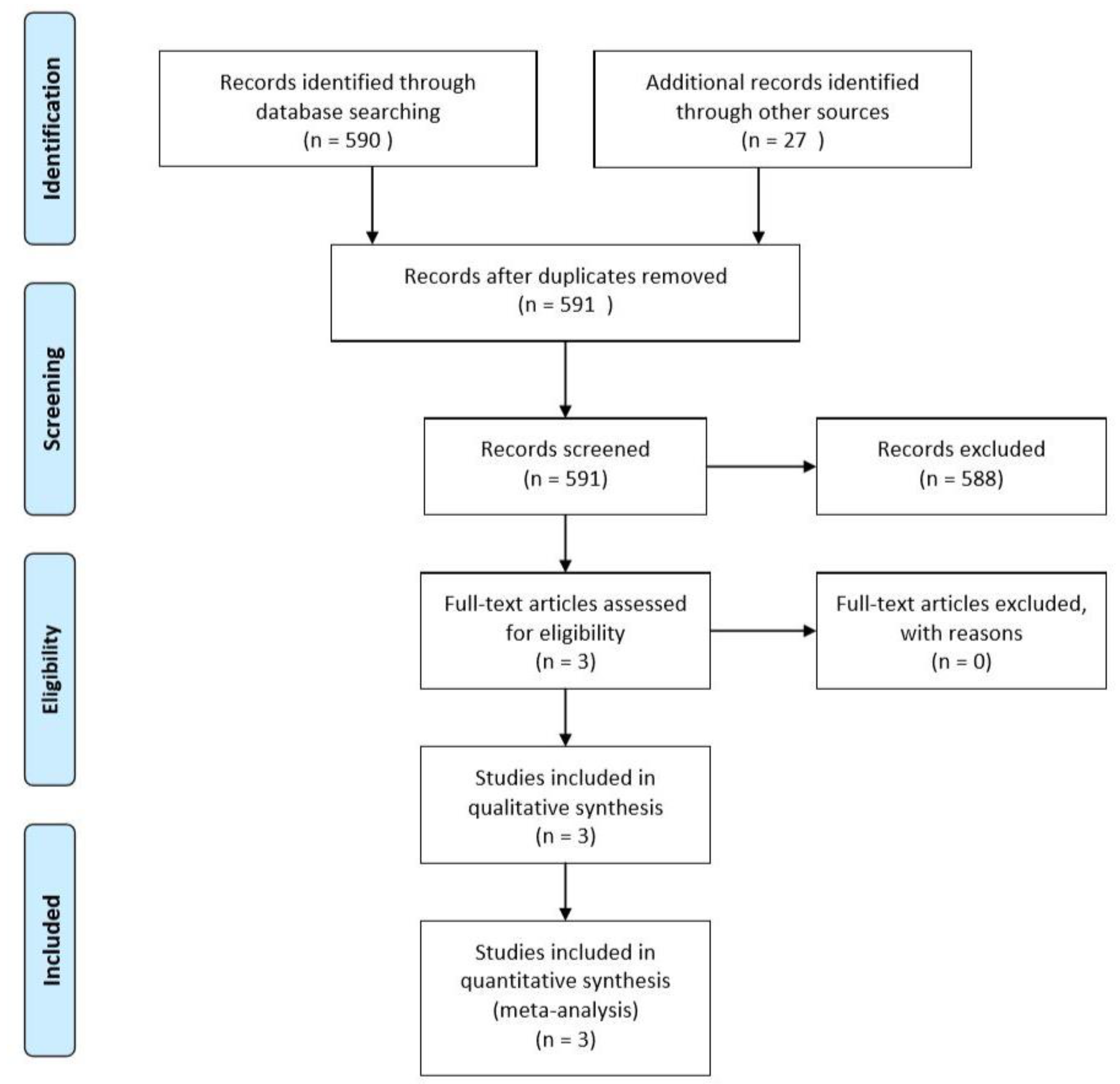
| Study | Design | Patients/Defects Gender (M/F) | Mean Age (Range or ±SD) | Study Groups (Patients/Defects) | Defect Localization | Number of Defect Walls | Type of Bone Harvested | |
|---|---|---|---|---|---|---|---|---|
| Guida et al., 2007 [18] | RCT (Pa) | 27/28 13/14 | 46.3 ± 8.7 (30–65 years) | Test EMD + AB (14) | Ctr EMD (14) | Max: 13 def. (7 EMD + AB, 6 EMD) Mdb: 15 def. (7 EMD + AB, 8 EMD) IN/CA: 12 def. (5 EMD + AB, 7 EMD) PM/MO: 16 def. (9 EMD + AB, 7 EMD) | A predominant 1- to 2-wall component | Cortical autogenous bone particles were harvested from the buccal cortical plate by means of a bone scraper. The bone graft was collected from the surgical site adjacent to the intraosseous defect. |
| Yilmaz et al., 2010 [22] | RCT (Pa) | 40/40 24/16 | (30–50 years) | Test EMD + AB (20) | Ctr EMD (20) | Max: 18 def. (8 EMD + AB, 10 EMD) Mdb: 22 def. (12 EMD + AB, 10 EMD) IN/CA: 12 def. (6 EMD + AB, 6 EMD) PM: 14 def. (8 EMD + AB, 6 EMD) MO: 14 def. (6 EMD + AB, 8 EMD) | 2 walls: 15 defects (7 EMD + AB, 8 EMD) 2-3 wall: 25 defects (13 EMD + AB, 12 EMD) | Cortico-cancellous autogenous bone was harvested from the retromolar area using a trephine bur with a diameter of 3 mm. |
| Agrali et al., 2016 [23] | RCT (Pa) | 12/30 6/6 | 44.17 ± 7.80 | Test EMD + AB (10)EMD (10) | Ctr OFD (10) | Max: NR Mdb: NR IN/CA: 10 def. (5 EMD + AB, 2 EMD alone, 3 OFD) PM: 10 def. (2 EMD + AB, 6 EMD alone, 2 OFD) MO: 10 def. (3 EMD + AB, 2 EMD alone, 5 OFD) | 1-walled: 6 def. (1 EMD + AB, 4 EMD alone, 1 OFD) 1-2-walled: 20 def. (6 EMD + AB, 5 EMD alone, 9 OFD) 1-2-3-walled: 4 def. (3 EMD + AB, 1 EMD alone) | Autogenous bone was obtained from adjacent bone surfaces by using hand instruments Ochsenbein Periodontal Chisel CO2, Rhodes Back Action Periodontal Chisel C36/37, Hu-Friedy Inst. Co., Chicago, IL, USA). |
| Study | Aim | Inclusion Criteria | Surgical Protocol |
|---|---|---|---|
| Guida et al., 2007 [18] | Assess the additional clinical benefit of autogenous cortical bone particles when added to EMD, compared to EMD alone, in the treatment of deep periodontal intraosseous defects. | 1) No systemic diseases that contraindicated periodontal surgery; 2) No medications affecting periodontal status; 3) No pregnancy or lactation; 4) At least one intraosseous defect in need of surgical treatment after initial periodontal treatment and revaluation; 5) PD ≥ 6 mm; 3) Radiographic intraosseous defect ≥4 mm. 6) No full-mouth plaque score and full-mouth bleeding score >20% at the time of surgical procedure. Furthermore, third molars, teeth with Class III mobility, furcation involvement, inadequate endodontic treatment, or restoration were excluded. | After flap reflection, all soft tissue was removed from the defect, and the root surface was scaled and planed with hand and ultrasonic instruments. In all cases, the exposed root surfaces were conditioned with 24% EDTA gel for 2 min. The defect was then thoroughly rinsed with saline to remove gel remnants. For the EMD + AB group, particulate cortical bone was harvested from the buccal cortical plate by means of a bone scraper. A first layer of EMD was injected to condition the bone defect and the more apical portion of the root surface. AB was positioned to fill only the intrabony component of the defect. Finally, a second layer of EMD was injected to cover the grafted autogenous bone particles and to condition the portion of the root surface coronal to the bone crest. Therefore, a “sandwich” technique was adopted to treat the defect (i.e., apical layer of EMD, AB, and coronal layer of EMD). For the EMD group, the EMD gel alone was injected into the defect. Finally, flaps were positioned at the pre-surgery level or slightly coronal. |
| Yilmaz et al., 2010 [22] | Evaluate the healing of deep intrabony defects treated with either a combination EMD + AB or EMD alone | (1) No systemic diseases such as diabetes mellitus or cardiovascular diseases that could influence the outcome of the therapy; (2) No smokers; (3) A good level of oral hygiene (plaque index (PI) < 1); (4) Compliance with the maintenance programme; (5) Presence of one intrabony defect with a probing depth of at least 6 mm and an intrabony component of at least 3 mm, as detected on the radiographs. | Intracrevicular incisions were placed, and full-thickness flaps were raised vestibularly and orally. If necessary, vertical releasing incisions were performed. Following removal of granulation tissue from the defects, the roots were thoroughly scaled and planed using hand and ultrasonic instruments. In both groups, the root surfaces adjacent to the defects were conditioned for 2 min with an EDTA gel in order to remove the smear layer. The defects and the adjacent mucoperiosteal flaps were then thoroughly rinsed with sterile saline in order to remove all EDTA residues. Following root conditioning, EMD was applied to the root surfaces and into the defects with a sterile syringe. Cortico-cancellous AB was harvested from the retromolar area using a trephine bur. The remaining EMD was then mixed with AB and the defects were completely filled with the mixture of EMD + AB. Finally, the flaps were advanced coronally and closed with vertical or horizontal mattress sutures. The sites treated with EMD received exactly the same treatment, including root conditioning with EDTA, but without the application of AB. |
| Agrali et al., 2016 [23] | Evaluate the effects of EMD either alone or combined with AB applied to intrabony defects in chronic periodontitis patients on clinical/radiographic parameters and GCF TGF-β1 level and to compare with OFD. | (1) No systemic diseases that contraindicated periodontal surgery and could affect the consequences of the therapy; (2) No smoking; (3) No medications; (4) No pregnancy or lactation; (5) Good oral hygiene level (plaque index (PI) < 1) and full-mouth bleeding on probing score <20% after initial periodontal treatment (IPT); (6) Compliance with the maintenance program; (7) Minimum one intrabony defect existence with a probing depth (PD) ≥ 6 mm, radiographic depth ≥ 3 mm, as detected on radiographs. | After local anesthesia, sulcular incisions were made and full-thickness flaps were raised buccally and lingually, granulation tissues removed, and the root surfaces gently scaled and planed. In the EMD and combination groups, the exposed root surfaces were conditioned with 24% EDTA gel for 2 min. The surgical area was then rinsed with saline. EMD gel was injected onto the intrabony defects and root surfaces. Then, in the combination group, the adequate amount of AB obtained from adjacent bone surfaces by using hand instruments was mixed with the gel and placed into the bone defects. Finally, a second layer of EMD gel was injected to cover the AB. Then, the flaps were sutured. |
| Study | Time | Outcomes | Method of Evaluation | Conclusions |
|---|---|---|---|---|
| Guida et al., 2007 [18] | 12 m | Clinical parameters -LPS (%) -LBS (%) -CAL (mm) -PD (mm) -REC (mm) Intrasurgical parameters -PBL (mm) -IBD (mm) Radiographical parameters -DEPTH (mm) -radiographic defect fill (percentage) -ANGLE (degrees) | -Periodontal probe (UNC 15, Hu-Friedy Mfg. Inc., Chicago, IL, USA). -A manual pressure-sensitive probe at approximately 0.3 N force with 1 mm increments). -LPS and LBS recorded dichotomously at surgical site as the presence or absence of supragingival plaque and bleeding on probing, respectively. -DEPTH, measured as the linear distance (in mm) -Radiographic defect fill (percentage) calculated as follows: (baseline DEPTH-12-month DEPTH)/baseline DEPTH 100 -ANGLE (degrees) the radiographic defect angle (ANGLE) at baseline, determined in degrees as the angle formed between the lines that represent the root surface of the involved tooth and the bone defect surface | Data support the clinical effectiveness of a regenerative procedure based on EMD application, either alone or in combination with a cortical AB, in the treatment of deep intraosseous defects without statistically significant differences. The combined EMD + AB procedure led to a reduced post-surgery recession and increased the proportion of defects with substantial clinical attachment level gain (≥6 mm). |
| Yilmaz et al., 2010 [22] | 12 m | Clinical parameters -PI -GI -BOP (%) -PD (mm) -RAL (mm) -REC (mm) -PBL (mm) Intrasurgical parameters -INTRA (mm) | -Periodontal probe (UNC 15, Hu-Friedy Mfg. Inc, Chicago, IL, USA). | At 1 year after surgery, both therapies resulted in statistically significant clinical improvements compared with baseline, and although the combination of EMD + AB resulted in statistically significant higher soft and hard tissue improvements compared with treatment with EMD, the clinical relevance of this finding is unclear. |
| Agrali et al., 2016 [23] | 6 m | Clinical parameters -PI -GI -BOP (%) -PD (mm) -RAL (mm) -REC (mm) Intrasurgical measurements -IDD (mm) Radiographic parameters -Bone fill (%) Other parameters -Determination of gingival crevicular fluid (GCF) transforming growth factor-β1 levels | -Periodontal probe (UNC 15, Hu-Friedy, Chicago, IL, USA) using an adapted acrylic stent with reference holes. -Long cone paralleling technique using an appropriate screening device (RWT Roentgenographic-System, Kentzler-Kaschner Dental GmbH, Germany). -GCF samples were collected with paper strips (PerioPaper® Oraflow Inc., New York, USA) just before surgery and 7, 14, 30, 90, 180 days after surgery and evaluated by enzyme-linked immunosorbent assay using a commercially available kit for TGF-β1 (Quantikine Human TGF-β1, R&D Systems, Inc., Minneapolis, MN, USA) | All treatment procedures led to significant improvements at 6 months (p < 0.01). Gain in attachment level (p < 0.01) and radiographic defect fill (p < 0.05) of the combination and EMD groups were found to be significantly higher than those of the control group, while the use of EMD either with AB or alone was observed to produce significantly less recession than the OFD (p < 0.05). |
| Validity | Quality Criteria | Guida et al., 2007 [18] | Yilmaz et al., 2010 [22] | Agrali et al., 2016 [23] |
|---|---|---|---|---|
| External | Declared the use of specific protocol guidelines | no | no | no |
| Representative population group | yes | yes | yes | |
| Eligibility criteria defined | yes | yes | yes | |
| Internal | Consecutive enrollment | yes | yes | yes |
| Random allocation | yes | yes | yes | |
| Allocation concealment | NR | yes | NR | |
| Blinding of the patient | NA | NA | NA | |
| Blinding of the examiner | yes | yes | no | |
| Blinding of the statistician | NR | NR | NR | |
| Reported loss to follow-up | yes | yes | yes | |
| No. (%) of dropouts | 0 | 0 | 0 | |
| Treatment identical, except for intervention | yes | yes | yes | |
| Statistical | Sample size calculation and power | yes | yes | yes |
| Point estimates presented for primary outcome | yes | yes | yes | |
| Measures of variability for the primary outcome | yes | yes | yes | |
| Intention to treat analysis | NR | NR | NR | |
| Coherent data presentation | yes | yes | yes | |
| Clinical aspects | Study design | RCT parallel | RCT parallel | RCT parallel |
| Validated measurement | yes | yes | yes | |
| Calibration of examiner | yes | yes | yes | |
| Estimated potential risk of bias | Low | Low | Low |
| Authors | PD (mm) | CAL/RAL (mm) | REC (mm) | PI | GI | BOP (%) | LPS (%) | LBS (%) |
|---|---|---|---|---|---|---|---|---|
| Guida et al., 2007 [18] | EMD (9.6 ± 1.7) EMD + AB (9.1 ± 1.6) | EMD (10.6 ± 1.3) EMD + AB (10.3 ± 1.5) | EMD (1.1 ± 1.0) EMD + AB (1.1 ± 0.9) | NA | NA | NA | EMD (21.4) EMD + AB (21.4) | EMD (71.4) EMD + AB (50.0) |
| Yilmaz et al., 2010 [22] | EMD (8.2 ± 0.7) EMD + AB (8.4 ± 1.2) | EMD (11.3 ± 0.9) EMD + AB (11.7 ± 1.0) | EMD (3.1 ± 1.1) EMD + AB (3.3 ± 1.5) | EMD (0.4 ± 0.1) EMD + AB (0.5 ± 0.1) | EMD (1.3 ± 0.3) EMD + AB (1.2 ± 0.2) | EMD (49.00) EMD + AB (50.00) | NA | NA |
| Agrali et al., 2016 [23] | EMD (8.30 ± 1.70) EMD + AB (7.93 ± 1.66) OFD (7.60 ± 1.51) | EMD (13.70 ± 2.58) EMD + AB (13.06 ± 1.77) OFD (12.10 ± 2.13) | EMD (5.40 ± 1.96) EMD + AB (5.12 ± 1.91) OFD (4.70 ± 1.70) | EMD (0.65 ± 0.24) EMD + AB (0.55 ± 0.16) OFD (0.75 ± 0.26) | EMD (0.90 ± 0.21) EMD + AB (0.78 ± 0.24) OFD (0.90 ± 0.21) | EMD (55.00 ± 10.54) EMD + AB (60.33 ± 17.29) OFD (62.50 ± 17.68) | NA | NA |
| Authors | Radiographic Parameters | Intrasurgical Parameters | ||
|---|---|---|---|---|
| DEPTH (mm) | ANGLE (degrees) | PBL (mm) | IBD/IDD/INTRA (mm) | |
| Guida et al., 2007 [18] | EMD (6.5 ± 2.9) EMD + AB (6.5 ± 1.8) | EMD (31.5 ± 2.4) EMD + AB (30.9 ± 12.6) | EMD (11.7 ± 1.7) EMD + AB (10.9 ± 2.0) | IBD EMD (6.2 ± 2.0) EMD + AB (7.0 ± 1.2) |
| Yilmaz et al., 2010 [22] | NA | NA | EMD (12.1 ± 0.9) EMD + AB (12.3 ± 1.0) | INTRA EMD (5.2 ± 0.7) EMD + AB (5.4 ± 1.0) |
| Agrali et al., 2016 [23] | NA | NA | NA | IDD EMD (6.40 ± 1.95) EMD + AB (5.20 ± 1.39) OFD (5.60 ± 1.64) |
| Authors | BOP (%) | PD (mm) | CAL/RAL (mm) | REC (mm) | DEPTH (mm) | BONE FILL (%) |
|---|---|---|---|---|---|---|
| Guida et al., 2007 [18] | NA | EMD (5.6 ± 1.7) EMD + AB (5.1 ± 1.7) NS | EMD (4.6 ± 1.3) EMD + AB (4.9 ± 1.8) NS CALgain < 2 mm EMD (0%) EMD + AB (0%) CALgain 2–3 mm EMD (21%) EMD + AB (29%) CALgain 4–5 mm EMD (57%) EMD + AB (21%) CALgain ≥ 6 mm EMD (21%) EMD + AB (50%) | EMD (1.1 ± 0.7) EMD + AB (0.3 ± 0.8) * | EMD (4.3 ± 2.4) EMD + AB (4.3 ± 1.3) NS DEPTH gain < 2 mm EMD (7%) EMD + AB (0%) DEPTH gain 2–3mm EMD (29%) EMD + AB (43%) DEPTH gain 4–5 mm EMD (50%) EMD + AB (43%) DEPTH gain ≥ 6 mm EMD (14%) EMD + AB (14%) | EMD (64.8 ± 24.1) EMD + AB (68 ± 17.3) NS |
| Yilmaz et al., 2010 [22] | EMD (16.00); EMD + AB (15.00) | EMD (4.6 ± 0.4) EMD + AB (5.6 ± 0.9) * | EMD (3.4 ± 0.8) EMD + AB (4.2 ± 1.1) * RALgain < 2 mm EMD (0%) EMD + AB (0%) RALgain 2–3 mm EMD (35%) EMD + AB (10%) RALgain 4–5 mm EMD (55%) EMD + AB (85%) RALgain 6 mm EMD (0%) EMD + AB (5%) | EMD (1.2 ± 0.8) EMD + AB (1.4 ± 0.9) NS | NA | NA |
| Agrali et al., 2016 [23] | EMD (42.50 ± 12.08) EMD + AB (47.71 ± 18.49) OFD (25.00 ± 0.00) | EMD (5.00 ± 1.41) EMD + AB (4.71 ± 1.63) OFD (4.40 ± 1.17) | EMD (4.50 ± 3.24) EMD + AB (3.55 ± 1.46) OFD (1.60 ± 0.70) | EMD (−0.50 ± 2.72) EMD + AB (−1.16 ± 1.62) OFD (−2.70 ± 0.95) | NA | EMD (65.98% ± 14.76%) EMD + AB (64.56% ± 24.23%) OFD (35.31% ± 20.56%) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Annunziata, M.; Piccirillo, A.; Perillo, F.; Cecoro, G.; Nastri, L.; Guida, L. Enamel Matrix Derivative and Autogenous Bone Graft for Periodontal Regeneration of Intrabony Defects in Humans: A Systematic Review and Meta-Analysis. Materials 2019, 12, 2634. https://doi.org/10.3390/ma12162634
Annunziata M, Piccirillo A, Perillo F, Cecoro G, Nastri L, Guida L. Enamel Matrix Derivative and Autogenous Bone Graft for Periodontal Regeneration of Intrabony Defects in Humans: A Systematic Review and Meta-Analysis. Materials. 2019; 12(16):2634. https://doi.org/10.3390/ma12162634
Chicago/Turabian StyleAnnunziata, Marco, Angelantonio Piccirillo, Francesco Perillo, Gennaro Cecoro, Livia Nastri, and Luigi Guida. 2019. "Enamel Matrix Derivative and Autogenous Bone Graft for Periodontal Regeneration of Intrabony Defects in Humans: A Systematic Review and Meta-Analysis" Materials 12, no. 16: 2634. https://doi.org/10.3390/ma12162634
APA StyleAnnunziata, M., Piccirillo, A., Perillo, F., Cecoro, G., Nastri, L., & Guida, L. (2019). Enamel Matrix Derivative and Autogenous Bone Graft for Periodontal Regeneration of Intrabony Defects in Humans: A Systematic Review and Meta-Analysis. Materials, 12(16), 2634. https://doi.org/10.3390/ma12162634






