Band to Band Tunneling at the Zinc Oxide (ZnO) and Lead Selenide (PbSe) Quantum Dot Contact; Interfacial Charge Transfer at a ZnO/PbSe/ZnO Probe Device
Abstract
:1. Introduction
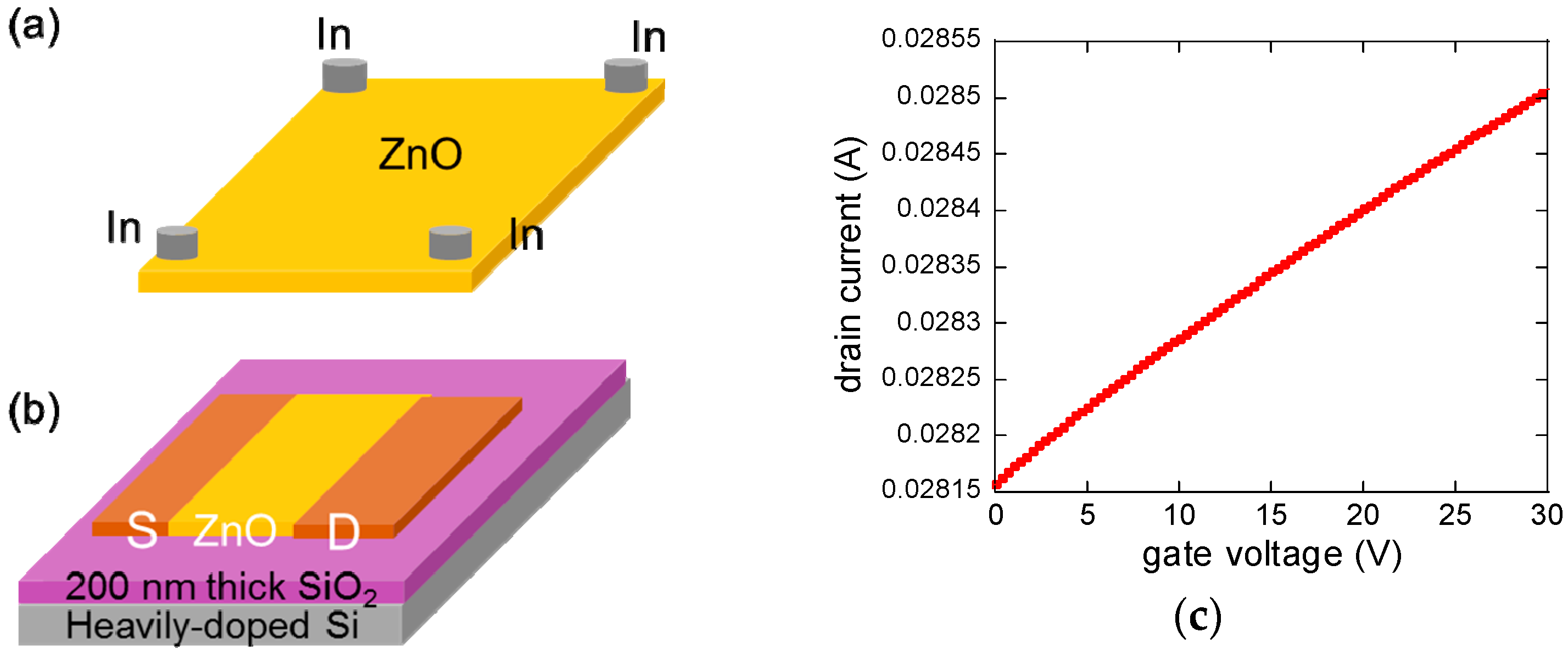
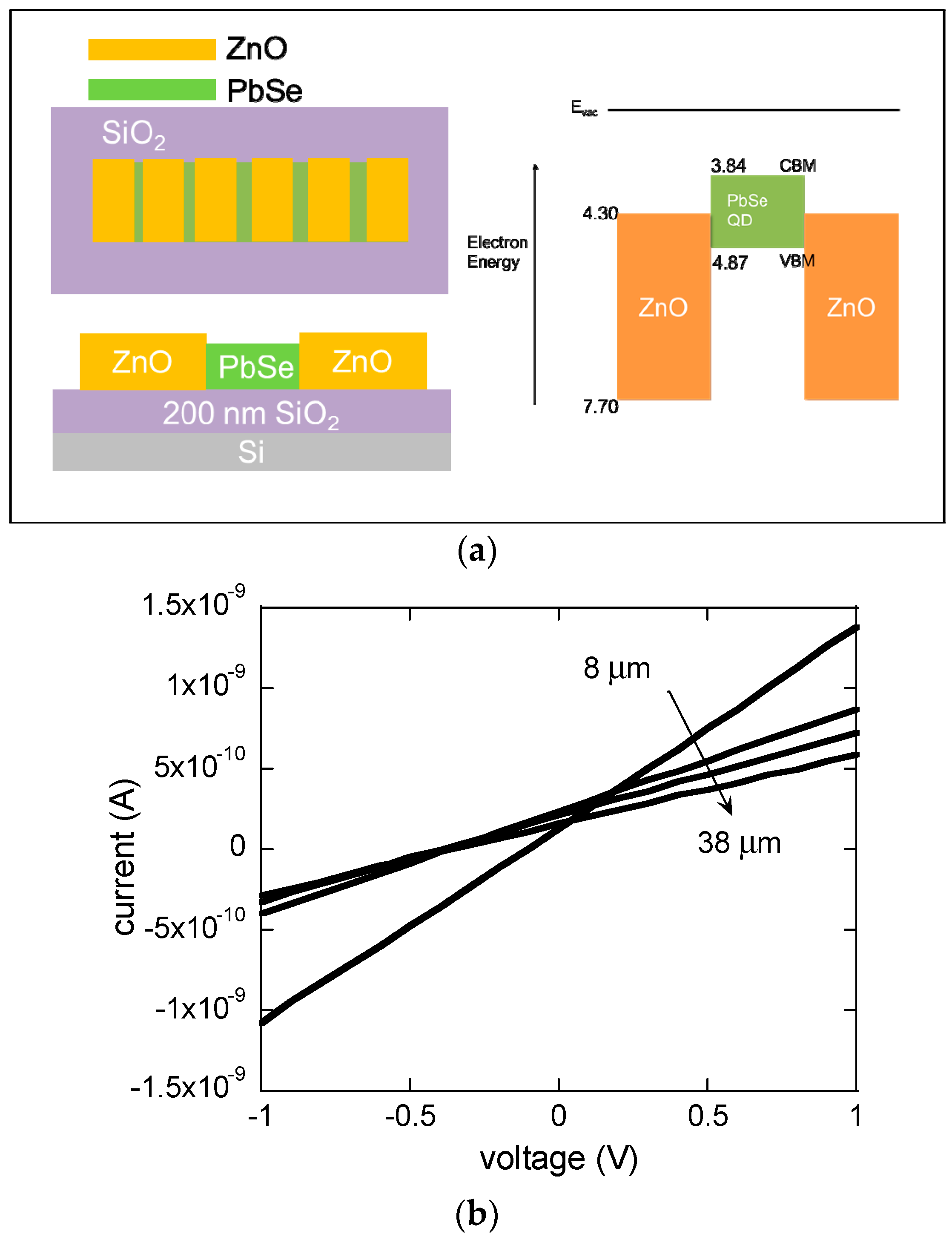
2. Materials and Methods
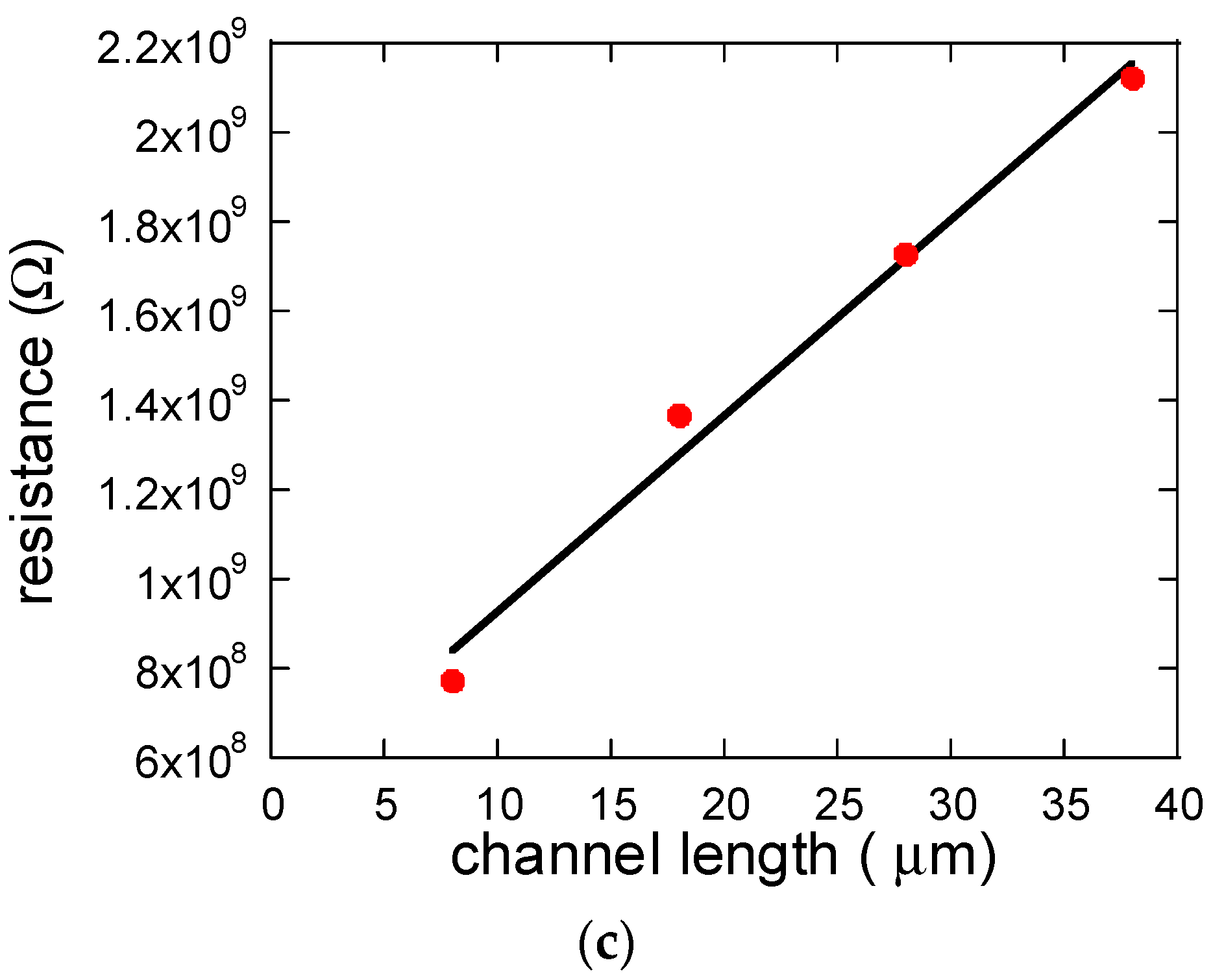
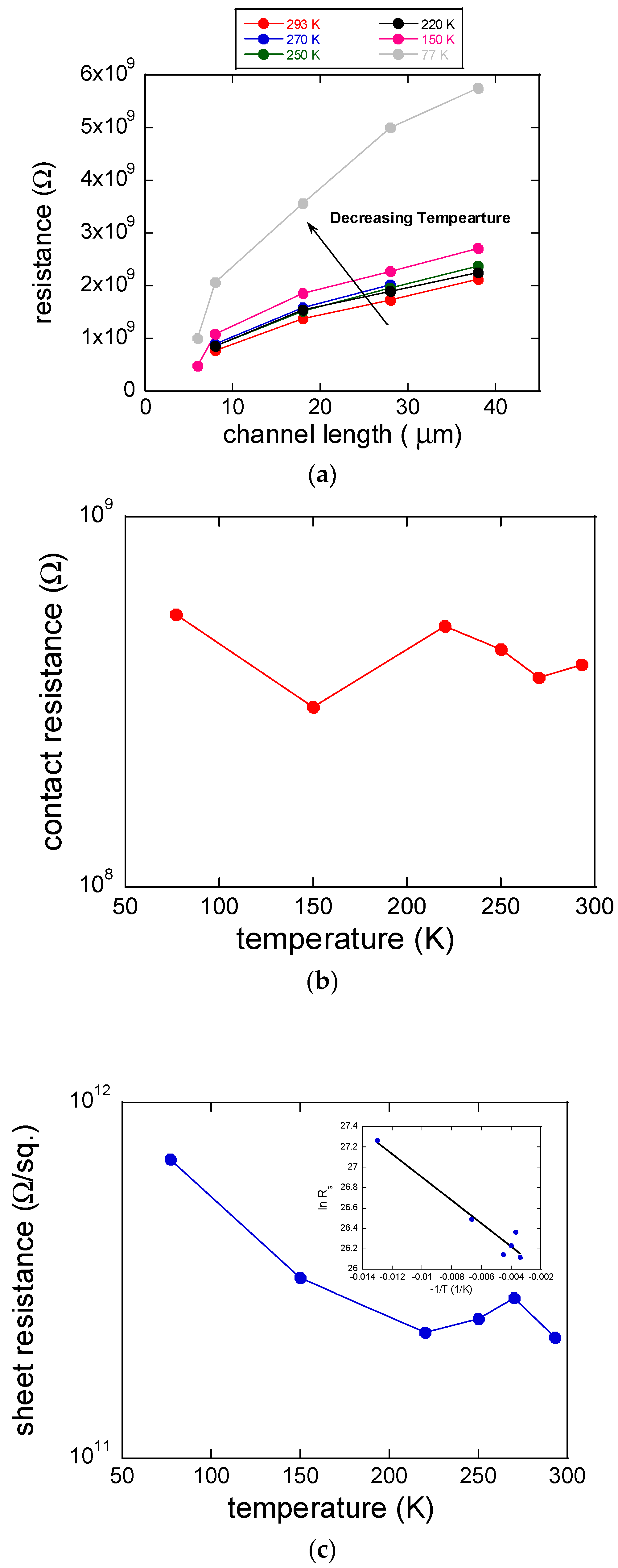
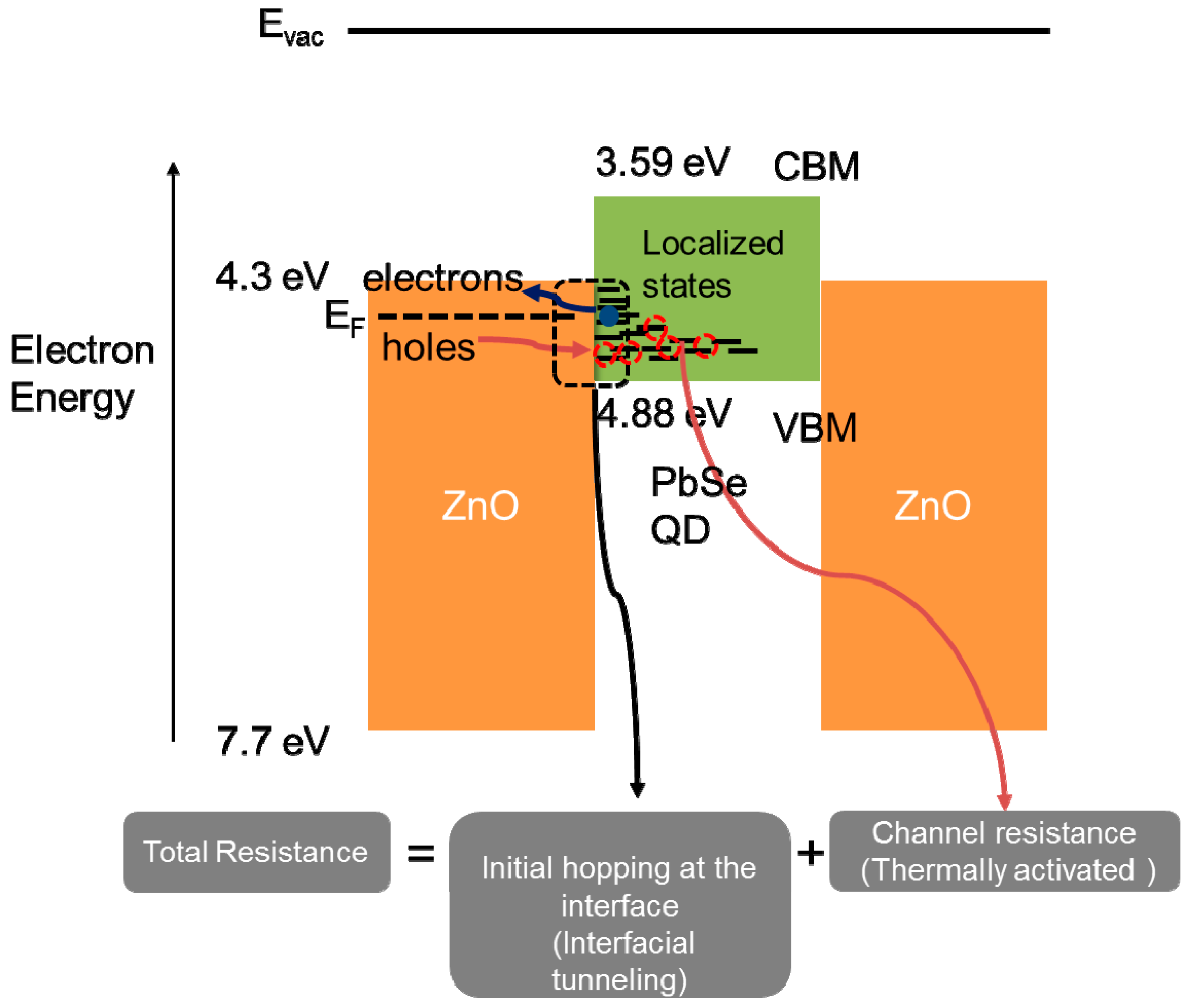
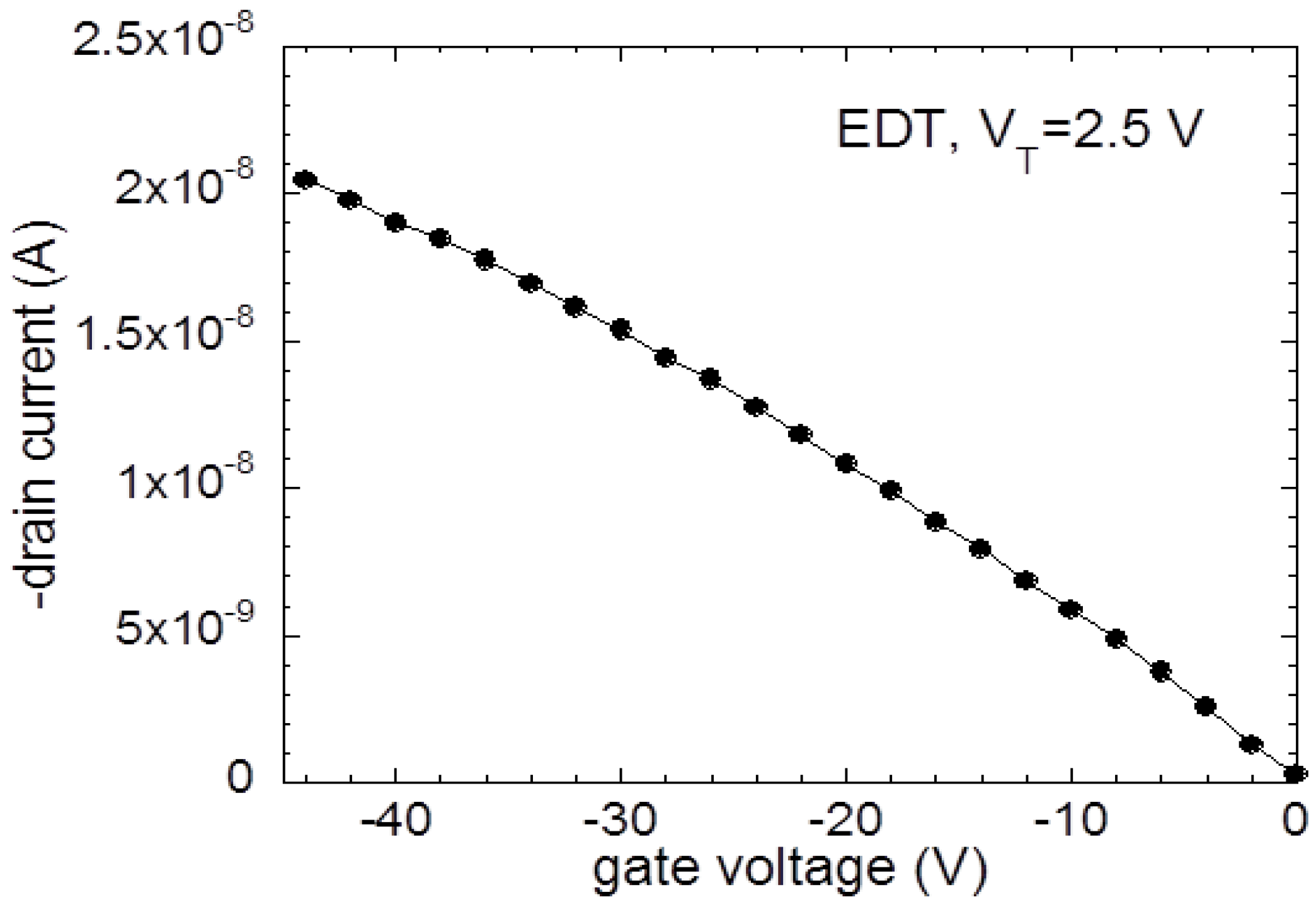
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ishii, H.; Sugiyama, K.; Ito, E.; Seki, K. Energy level alignment and interfacial electronic structures at organic/metal and organic/organic interfaces. Adv. Mater. 1999, 11, 605–625. [Google Scholar] [CrossRef]
- Scott, J.C.; Malliaras, G.G. Charge injection and recombination at the metal–organic interface. Chem. Phys. Lett. 1999, 299, 115–119. [Google Scholar] [CrossRef]
- Kim, Y.; Cho, S.; Jeong, S.; Ko, D.-H.; Ko, H.; You, N.; Chang, M.; Reichmanis, E.; Park, J.-Y.; Park, S.Y. Competition between charge transport and energy barrier in injection-limited metal/quantum dot nanocrystal contacts. Chem. Mater. 2014, 26, 6393–6400. [Google Scholar] [CrossRef]
- Hill, I.; Rajagopal, A.; Kahn, A.; Hu, Y. Molecular level alignment at organic semiconductor-metal interfaces. Appl. Phys. Lett. 1998, 73, 662–664. [Google Scholar] [CrossRef]
- Tvrdy, K.; Frantsuzov, P.A.; Kamat, P.V. Photoinduced electron transfer from semiconductor quantum dots to metal oxide nanoparticles. Proc. Natl. Acad. Sci. USA 2011, 108, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Mondal, R.; Ko, S.; Norton, J.E.; Miyaki, N.; Becerril, H.A.; Verploegen, E.; Toney, M.F.; Brédas, J.-L.; McGehee, M.D.; Bao, Z. Molecular design for improved photovoltaic efficiency: Band gap and absorption coefficient engineering. J. Mater. Chem. 2009, 19, 7195–7197. [Google Scholar] [CrossRef]
- D’Innocenzo, V.; Srimath Kandada, A.R.; De Bastiani, M.; Gandini, M.; Petrozza, A. Tuning the light emission properties by band gap engineering in hybrid lead halide perovskite. J. Am. Chem. Soc. 2014, 136, 17730–17733. [Google Scholar] [CrossRef] [PubMed]
- Vega, E.; Mollar, M.; Mari, B. Bandgap Tuning of MAPbI3—XBrx Thin Film Perovskites for Photovoltaic Applications. In Proceedings of the Renewable and Sustainable Energy Conference (IRSEC), Marrakech, Morocco, 10–13 December 2015. [Google Scholar]
- Kim, Y.; Ko, H.; Park, B. Tuning charge transfer at the electron donor/acceptor assembly through vibration-induced aggregation of P3HT chains in solution. Mater. Chem. Phys. 2019, 223, 576–582. [Google Scholar] [CrossRef]
- Jara, D.H.; Yoon, S.J.; Stamplecoskie, K.G.; Kamat, P.V. Size-dependent photovoltaic performance of CuInS2 quantum dot-sensitized solar cells. Chem. Mater. 2014, 26, 7221–7228. [Google Scholar] [CrossRef]
- Lee, K.-H.; Lee, J.-H.; Kang, H.-D.; Park, B.; Kwon, Y.; Ko, H.; Lee, C.; Lee, J.; Yang, H. Over 40 cd/A efficient green quantum dot electroluminescent device comprising uniquely large-sized quantum dots. ACS Nano 2014, 8, 4893–4901. [Google Scholar] [CrossRef]
- Liu, Y.; Gibbs, M.; Puthussery, J.; Gaik, S.; Ihly, R.; Hillhouse, H.W.; Law, M. Dependence of carrier mobility on nanocrystal size and ligand length in PbSe nanocrystal solids. Nano Lett. 2010, 10, 1960–1969. [Google Scholar] [CrossRef] [PubMed]
- Crisp, R.W.; Kroupa, D.M.; Marshall, A.R.; Miller, E.M.; Zhang, J.; Beard, M.C.; Luther, J.M. Metal halide solid-state surface treatment for high efficiency PbS and PbSe QD solar cells. Sci. Rep. 2015, 5, 9945. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Chang, M.; Park, B. Near infrared-induced optical gating at the lead-sulfide (PbS)/pentacene interface. Thin Solid Films 2018, 651, 85–90. [Google Scholar] [CrossRef]
- Weidman, M.C.; Nguyen, Q.; Smilgies, D.-M.; Tisdale, W.A. Impact of Size Dispersity, Ligand Coverage, and Ligand Length on the Structure of PbS Nanocrystal Superlattices. Chem. Mater. 2018, 30, 807–816. [Google Scholar] [CrossRef]
- Kamat, P.V. Quantum dot solar cells. Semiconductor nanocrystals as light harvesters. J. Phys. Chem. C 2008, 112, 18737–18753. [Google Scholar] [CrossRef]
- Wise, F.W. Lead salt quantum dots: The limit of strong quantum confinement. Acc. Chem. Res. 2000, 33, 773–780. [Google Scholar] [CrossRef]
- Ellingson, R.J.; Beard, M.C.; Johnson, J.C.; Yu, P.; Micic, O.I.; Nozik, A.J.; Shabaev, A.; Efros, A.L. Highly efficient multiple exciton generation in colloidal PbSe and PbS quantum dots. Nano Lett. 2005, 5, 865–871. [Google Scholar] [CrossRef]
- Ma, W.; Swisher, S.L.; Ewers, T.; Engel, J.; Ferry, V.E.; Atwater, H.A.; Alivisatos, A.P. Photovoltaic performance of ultrasmall PbSe quantum dots. ACS Nano 2011, 5, 8140–8147. [Google Scholar] [CrossRef]
- Kuo, C.Y.; Su, M.S.; Hsu, Y.C.; Lin, H.N.; Wei, K.H. An organic hole transport layer enhances the performance of colloidal PbSe quantum dot photovoltaic devices. Adv. Funct. Mater. 2010, 20, 3555–3560. [Google Scholar] [CrossRef]
- Caruge, J.; Halpert, J.E.; Wood, V.; Bulović, V.; Bawendi, M. Colloidal quantum-dot light-emitting diodes with metal-oxide charge transport layers. Nat. Photonics 2008, 2, 247–250. [Google Scholar] [CrossRef]
- Rekemeyer, P.H.; Chang, S.; Chuang, C.H.M.; Hwang, G.W.; Bawendi, M.G.; Gradečak, S. Enhanced photocurrent in pbs quantum dot photovoltaics via ZnO nanowires and band alignment engineering. Adv. Energy Mater. 2016, 6, 1600848. [Google Scholar] [CrossRef]
- Shirasaki, Y.; Supran, G.J.; Bawendi, M.G.; Bulović, V. Emergence of colloidal quantum-dot light-emitting technologies. Nat. Photonics 2013, 7, 13. [Google Scholar] [CrossRef]
- Kim, J.-H.; Han, C.-Y.; Lee, K.-H.; An, K.-S.; Song, W.; Kim, J.; Oh, M.S.; Do, Y.R.; Yang, H. Performance improvement of quantum dot-light-emitting diodes enabled by an alloyed ZnMgO nanoparticle electron transport layer. Chem. Mater. 2014, 27, 197–204. [Google Scholar] [CrossRef]
- Tu, Y.; Wang, S.; Zhang, Y.; Chen, L.; Fang, Y.; Du, Z. Balanced carrier injection of quantum dots light-emitting diodes: The case of interface barrier of bilayer ZnO electron transport layer. Nanotechnology 2018, 29, 485203. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Jiang, Y.; Peng, H.; Wei, J.; Zhang, S.; Chen, S. Efficient quantum dot light-emitting diodes with a Zn 0.85 Mg 0.15 O interfacial modification layer. Nanoscale 2017, 9, 8962–8969. [Google Scholar] [CrossRef] [PubMed]
- Hoye, R.L.; Ehrler, B.; Böhm, M.L.; Muñoz-Rojas, D.; Altamimi, R.M.; Alyamani, A.Y.; Vaynzof, Y.; Sadhanala, A.; Ercolano, G.; Greenham, N.C. Improved Open-Circuit Voltage in ZnO–PbSe Quantum Dot Solar Cells by Understanding and Reducing Losses Arising from the ZnO Conduction Band Tail. Adv. Energy Mater. 2014, 4, 1301544. [Google Scholar] [CrossRef] [PubMed]
- Timp, B.A.; Zhu, X.-Y. Electronic energy alignment at the PbSe quantum dots/ZnO (1010) interface. Surf. Sci. 2010, 604, 1335–1341. [Google Scholar] [CrossRef]
- Leschkies, K.S.; Beatty, T.J.; Kang, M.S.; Norris, D.J.; Aydil, E.S. Solar cells based on junctions between colloidal PbSe nanocrystals and thin ZnO films. ACS Nano 2009, 3, 3638–3648. [Google Scholar] [CrossRef]
- Park, B.; Ko, H.; Kim, M. Simultaneous probing of nanocrystal (NC)-ligand interaction-induced charge transfer/transport properties at the electron donor (lead selenide NC)/acceptor (zinc oxide) functional interface. Phys. B Condens. Matter 2019, 553, 40–46. [Google Scholar] [CrossRef]
- Jasieniak, J.; Califano, M.; Watkins, S.E. Size-dependent valence and conduction band-edge energies of semiconductor nanocrystals. ACS Nano 2011, 5, 5888–5902. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, M.; Han, C.-Y.; Yang, H.; Park, B. Band to Band Tunneling at the Zinc Oxide (ZnO) and Lead Selenide (PbSe) Quantum Dot Contact; Interfacial Charge Transfer at a ZnO/PbSe/ZnO Probe Device. Materials 2019, 12, 2289. https://doi.org/10.3390/ma12142289
Kim M, Han C-Y, Yang H, Park B. Band to Band Tunneling at the Zinc Oxide (ZnO) and Lead Selenide (PbSe) Quantum Dot Contact; Interfacial Charge Transfer at a ZnO/PbSe/ZnO Probe Device. Materials. 2019; 12(14):2289. https://doi.org/10.3390/ma12142289
Chicago/Turabian StyleKim, Minkyong, Chang-Yeol Han, Heesun Yang, and Byoungnam Park. 2019. "Band to Band Tunneling at the Zinc Oxide (ZnO) and Lead Selenide (PbSe) Quantum Dot Contact; Interfacial Charge Transfer at a ZnO/PbSe/ZnO Probe Device" Materials 12, no. 14: 2289. https://doi.org/10.3390/ma12142289
APA StyleKim, M., Han, C.-Y., Yang, H., & Park, B. (2019). Band to Band Tunneling at the Zinc Oxide (ZnO) and Lead Selenide (PbSe) Quantum Dot Contact; Interfacial Charge Transfer at a ZnO/PbSe/ZnO Probe Device. Materials, 12(14), 2289. https://doi.org/10.3390/ma12142289






