A New Broadband and Strong Absorption Performance FeCO3/RGO Microwave Absorption Nanocomposites
Abstract
:1. Introduction
2. Experimental
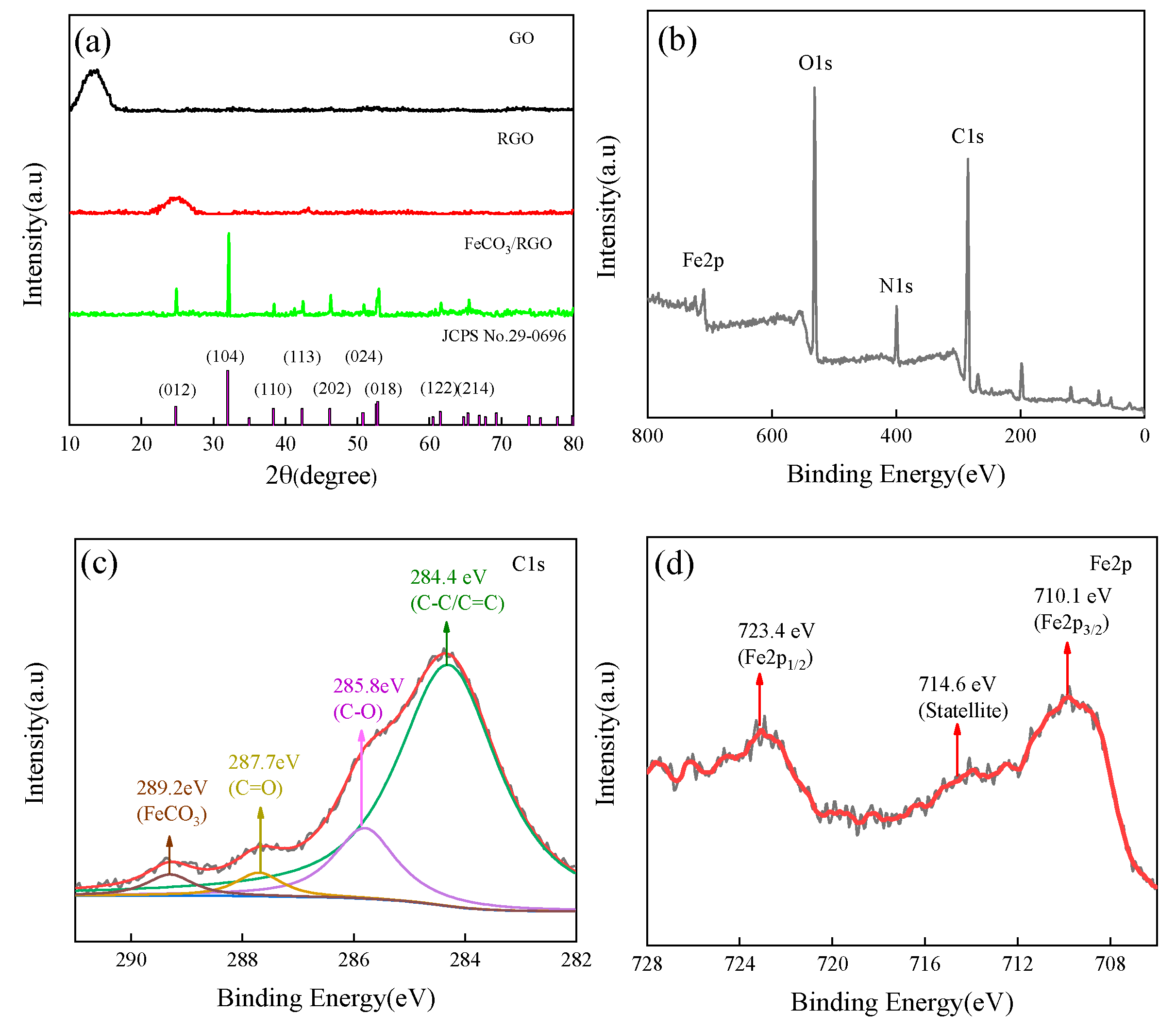
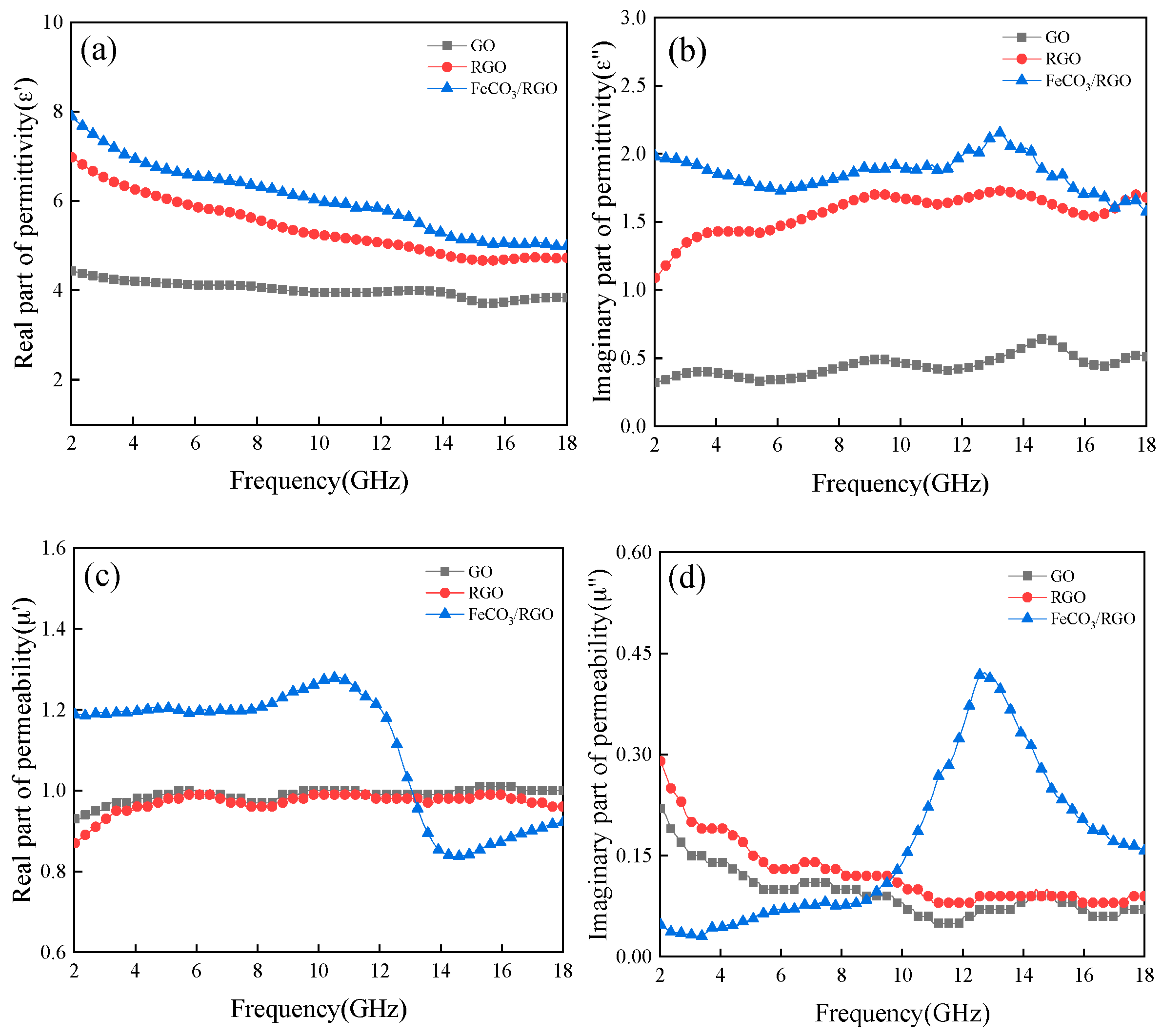
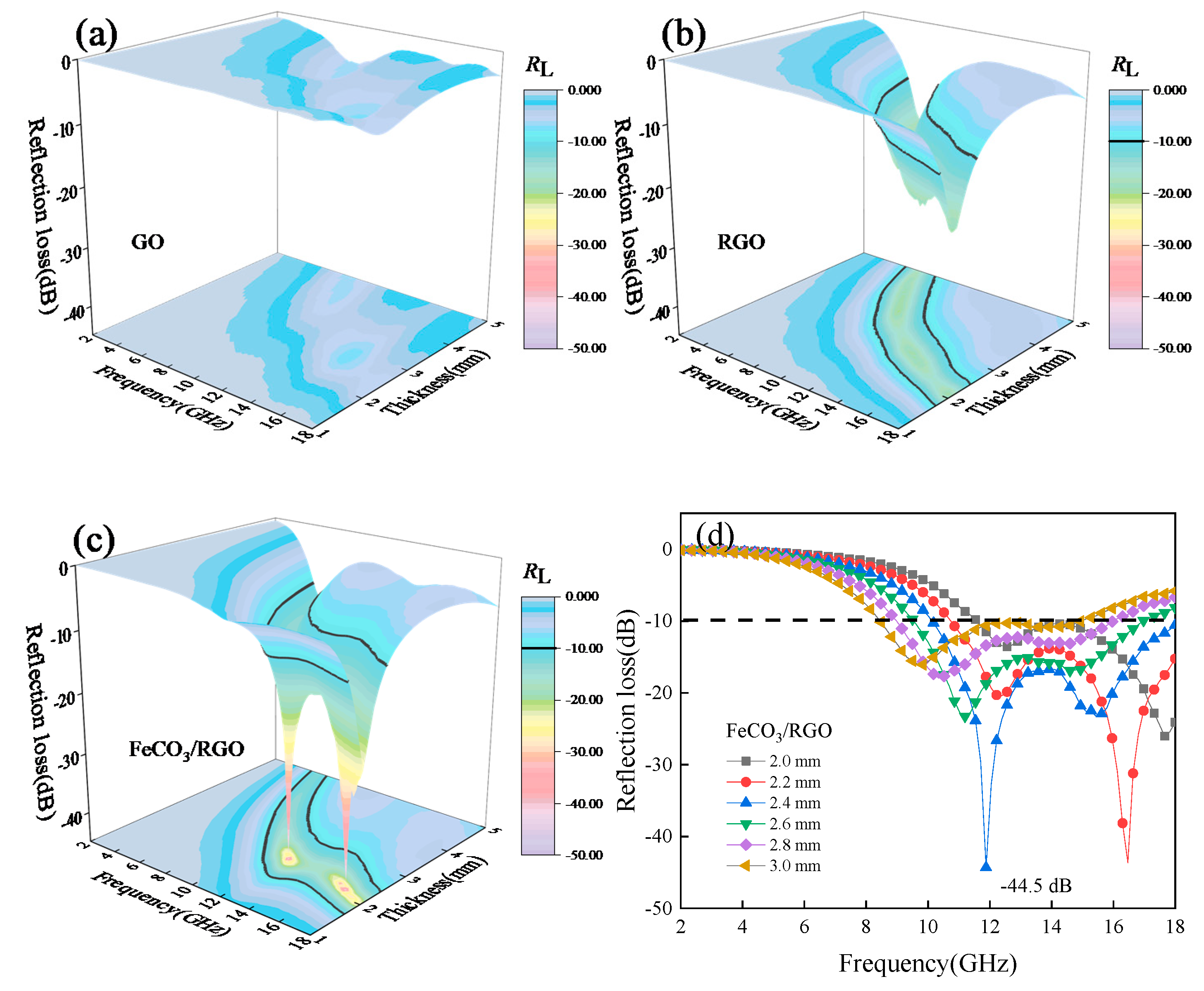
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zhang, X.J.; Wang, G.S.; Cao, W.Q.; Wei, Y.Z.; Liang, J.F.; Guo, L.; Cao, M.S. Enhanced microwave absorption property of reduced graphene oxide (RGO)-MnFe2O4 nanocomposites and polyvinylidene fluoride. ACS Appl. Mater. Interfaces 2014, 6, 7471. [Google Scholar] [CrossRef]
- Radoń, A.; Włodarczyk, P. Influence of water on the dielectric properties, electrical conductivity and microwave absorption properties of amorphous yellow dextrin. Cellulose 2019, 26, 2987–2998. [Google Scholar] [CrossRef] [Green Version]
- Mohammadkhani, F.; Montazer, M.; Latifi, M. Microwave absorption characterization and wettability of magnetic nano iron oxide/recycled PET nanofibers web. J. Text. Inst. 2019, 110, 989–999. [Google Scholar] [CrossRef]
- Plyushch, A.; Zhai, T.; Xia, H.; Santillo, C.; Verdolotti, L.; Lavorgna, M.; Kuzhir, P. Ultra-Light reduced graphene oxide based aerogel/foam absorber of microwave radiation. Materials 2019, 12, 213. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Ma, S.Q.; Li, X.; Zhao, X.; Cheng, X.W.; Liu, J. Preparation and microwave absorption properties of microsheets VO2 (M). J. Alloys Compd. 2019, 791, 307–315. [Google Scholar] [CrossRef]
- Li, Y.; Li, D.; Yang, J.; Luo, H.; Chen, F.; Wang, X.; Gong, R. Enhanced microwave absorption and surface wave attenuation properties of Co0.5Ni0.5Fe2O4 fibers/reduced graphene oxide composites. Materials 2018, 11, 508. [Google Scholar]
- Meng, F.B.; Wang, H.G.; Huang, F.; Guo, Y.F.; Wang, Z.Y.; Hui, D.; Zhou, Z.W. Graphene-based microwave absorbing composites: A review and prospective. Compos. Part B Eng. 2018, 137, 260–277. [Google Scholar] [CrossRef]
- Chen, C.; Xi, J.B.; Zhou, E.Z.; Peng, L.; Chen, Z.C.; Gao, C. Porous graphene microflowers for high-performance microwave absorption. Nano Micro Lett. 2018, 10, 26. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.W.; Wang, Y.Z.; Xiong, G.Y.; Li, D.; Li, Q.; Ma, C.; Guo, R.; Luo, H. Facile synthesis of ZnFe2O4/reduced graphene oxide nanohybrids for enhanced microwave absorption properties. Mater. Res. Bull. 2014, 61, 292–297. [Google Scholar] [CrossRef]
- Cui, G.Z.; Liu, Y.L.; Zhou, W.; Lv, X.L.; Hu, J.N.; Zhang, G.Y.; Gu, G.X. Excellent microwave absorption properties derived from the synthesis of hollow Fe3O4@reduced graphite oxide (RGO) nanocomposites. Nanomaterials 2019, 9, 141. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, X.M.; Zhang, W.Z.; Huang, S. One-pot synthesis of MnFe2O4 nanoparticles-decorated reduced graphene oxide for enhanced microwave absorption properties. Mater. Technol. 2016, 32, 32–37. [Google Scholar] [CrossRef]
- Feng, J.T.; Hou, Y.H.; Wang, Y.C.; Li, L.C. Synthesis of hierarchical ZnFe2O4@SiO2@RGO core-shell microspheres for enhanced electromagnetic wave absorption. ACS Appl. Mater. Interfaces 2017, 9, 14103–14111. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.C.; Cai, X.; Xu, D.H.; Chen, W.Y.; Fang, Y.P.; Yu, X.Y. Mn doped FeCO3/reduced graphene composite as anode material for high performance lithium-ion batteries. Appl. Surf. Sci. 2018, 428, 73–81. [Google Scholar] [CrossRef]
- Gu, X.; Yan, C.L.; Yan, L.T.; Cao, L.; Niu, F.E.; Liu, D.D.; Dai, P.C.; Li, L.J.; Yang, J.; Zhao, X.B. Carbonates (bicarbonates)/reduced graphene oxide as anode materials for sodium-ion batteries. J. Mater. Chem. A. 2017, 5, 24645–24650. [Google Scholar] [CrossRef]
- Xu, D.H.; Liu, W.J.; Zhang, C.C.; Cai, X.; Chen, W.Y.; Fang, Y.P.; Yu, X.Y. Monodispersed FeCO3 nanorods anchored on reduced graphene oxide as mesoporous composite anode for high-performance lithium-ion batteries. J. Power Sources 2017, 364, 359–366. [Google Scholar] [CrossRef]
- He, J.Z.; Zheng, Q.C.; Sun, X.; Jin, C.S.; Xi, W.X.; Mao, S.C. Axiolitic ZnO rods wrapped with reduced graphene oxide: Fabrication, microstructure and highly efficient microwave absorption. Mater. Lett. 2019, 241, 14–17. [Google Scholar] [CrossRef]
- Moussa, H.; Girot, E.; Mozet, K.; Alem, H.; Medjahdi, G.; Schneider, R. ZnO rods/reduced graphene oxide composites prepared via a solvothermal reaction for efficient sunlight-driven photocatalysis. Appl. Catal. B Environ. 2016, 185, 11–21. [Google Scholar] [CrossRef]
- Heuer, J.K.; Stubbins, J. An XPS characterization of FeCO3 films from CO2 corrosion. Corros. Sci. 1999, 41, 1231–1243. [Google Scholar] [CrossRef]
- Lu, M.M.; Cao, W.Q.; Shi, H.L.; Xiao, Y.F.; Jian, Y.; Zhi, L.H.; Hai, B.J.; Wen, Z.W.; Jie, Y.; Mao, S.C. Multi-wall carbon nanotubes decorated with ZnO nanocrystals: Mild solution-process synthesis and highly efficient microwave absorption properties at elevated temperature. J. Mater. Chem. A. 2014, 2, 10540–10547. [Google Scholar] [CrossRef]
- Acher, O.; Gourrierec, P.L.; Perrin, G.; Baclet, P.; Roblin, O. Demonstration of anisotropic composites with tuneable microwave permeability manufactured from ferromagnetic thin films. IEEE Trans. Microw. Theory Tech. 1996, 44, 674–684. [Google Scholar] [CrossRef]
- Wen, F.S.; Yi, H.B.; Qiao, L.; Zheng, H.; Zhou, D.; Li, F.S. Analyses on double resonance behavior in microwave magnetic permeability of multiwalled carbon nanotube composites containing Ni catalyst. Appl. Phys. Lett. 2008, 92, 04257. [Google Scholar] [CrossRef]
- Aharoni, A. Some Recent Developments in Micromagnetics at the Weizmann Institute of Science. J. Appl. Phys. 1959, 30, S70–S78. [Google Scholar] [CrossRef]
- Vleck, V. Concerning the theory of ferromagnetic resonance absorption. Phys. Rev. 1950, 78, 266–274. [Google Scholar] [CrossRef]
- Shu, R.W.; Zhang, G.Y.; Zhang, J.B.; Wang, X.; Wang, M.; Gan, Y.; Shi, J.J.; He, J. Synthesis and high-performance microwave absorption of reduced graphene oxide/zinc ferrite hybrid nanocomposite. Mater. Lett. 2018, 215, 229–232. [Google Scholar] [CrossRef]
- Wang, Y.P.; Peng, Z.; Jiang, W. Controlled synthesis of Fe3O4@SnO2/RGO nanocomposite for microwave absorption enhancement. Ceram. Int. 2016, 42, 10682–10689. [Google Scholar] [CrossRef]
- Zhang, Y.L.; Wang, X.X.; Cao, M.S. Confinedly implanted NiFe2O4-rGO: Cluster tailoring and highly tunable electromagnetic properties for selective-frequency microwave absorption. Nano Res. 2018, 11, 1426–1436. [Google Scholar] [CrossRef]
- Shu, R.W.; Li, W.J.; Zhou, X.; Tian, D.D.; Zhang, G.Y.; Gan, Y.; Shi, J.J.; He, J. Facile preparation and microwave absorption properties of RGO/MWCNTs/ZnFe2O4 hybrid nanocomposites. J. Alloys Compd. 2018, 743, 163–174. [Google Scholar] [CrossRef]
- Zhang, K.; Gao, X.; Zhang, Q.; Li, T.; Chen, H.; Chen, X. Preparation and microwave absorption properties of asphalt carbon coated reduced graphene oxide/magnetic CoFe2O4 hollow particles modified multi-wall carbon nanotube composites. J. Alloys Compd. 2017, 723, 912–921. [Google Scholar] [CrossRef]
- Zong, M.; Huang, Y.; Wu, H.W.; Zhao, Y.; Liu, P.B.; Wang, L. Facile preparation of RGO/Cu2O/Cu composite and its excellent microwave absorption properties. Mater. Lett. 2013, 109, 112–115. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, B.C.; Xiang, J.Y.; Su, C.; Mu, C.P.; Wen, F.S.; Liu, Z.Y. Microwave Absorption Properties of CoS2 Nanocrystals Embedded into Reduced Graphene Oxide. ACS Appl. Mater. Interfaces 2017, 9, 28868–28875. [Google Scholar] [CrossRef]

| Sample | RL (dB) | Effective Bandwidth (GHz) (RL < −10 dB) | Thickness (mm) | Wt. (%) | Reference |
|---|---|---|---|---|---|
| Fe3O4/RGO | −41.89 | 4.2 | 2.5 | 50 | [10] |
| Fe3O4@SnO2/RGO | −45.5 | 3 | 4.5 | 50 | [25] |
| MnFe2O4/RGO | −29 | 4.88 | 3 | 10 | [1] |
| NiFe2O4/RGO | −58 | 4.08 | 2.7 | 27 | [26] |
| RGO/MWCNTs/ZnFe2O4 | −23.8 | 2.6 | 1.5 | 50 | [27] |
| RGO/MWCNTs/CoFe2O4 | −46.8 | 3.4 | 1.6 | 50 | [28] |
| RGO/Cu2O/Cu | −51.8 | 4.1 | 1.3 | 50 | [29] |
| CoS2/RGO | −56.9 | 4.1 | 2.2 | 50 | [30] |
| FeCO3/RGO | −44.5 | 7.9 | 2.4 | 60 | This work |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, W.; Wei, S.; Wang, Y.; Wang, B.; Liang, Y.; Huang, Y.; Xu, B. A New Broadband and Strong Absorption Performance FeCO3/RGO Microwave Absorption Nanocomposites. Materials 2019, 12, 2206. https://doi.org/10.3390/ma12132206
Huang W, Wei S, Wang Y, Wang B, Liang Y, Huang Y, Xu B. A New Broadband and Strong Absorption Performance FeCO3/RGO Microwave Absorption Nanocomposites. Materials. 2019; 12(13):2206. https://doi.org/10.3390/ma12132206
Chicago/Turabian StyleHuang, Wei, Shicheng Wei, Yujiang Wang, Bo Wang, Yi Liang, Yuwei Huang, and Binshi Xu. 2019. "A New Broadband and Strong Absorption Performance FeCO3/RGO Microwave Absorption Nanocomposites" Materials 12, no. 13: 2206. https://doi.org/10.3390/ma12132206
APA StyleHuang, W., Wei, S., Wang, Y., Wang, B., Liang, Y., Huang, Y., & Xu, B. (2019). A New Broadband and Strong Absorption Performance FeCO3/RGO Microwave Absorption Nanocomposites. Materials, 12(13), 2206. https://doi.org/10.3390/ma12132206




