Structural and Superconducting Properties of Thermal Treatment-Synthesised Bulk YBa2Cu3O7−δ Superconductor: Effect of Addition of SnO2 Nanoparticles
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. X-ray Diffraction Analysis
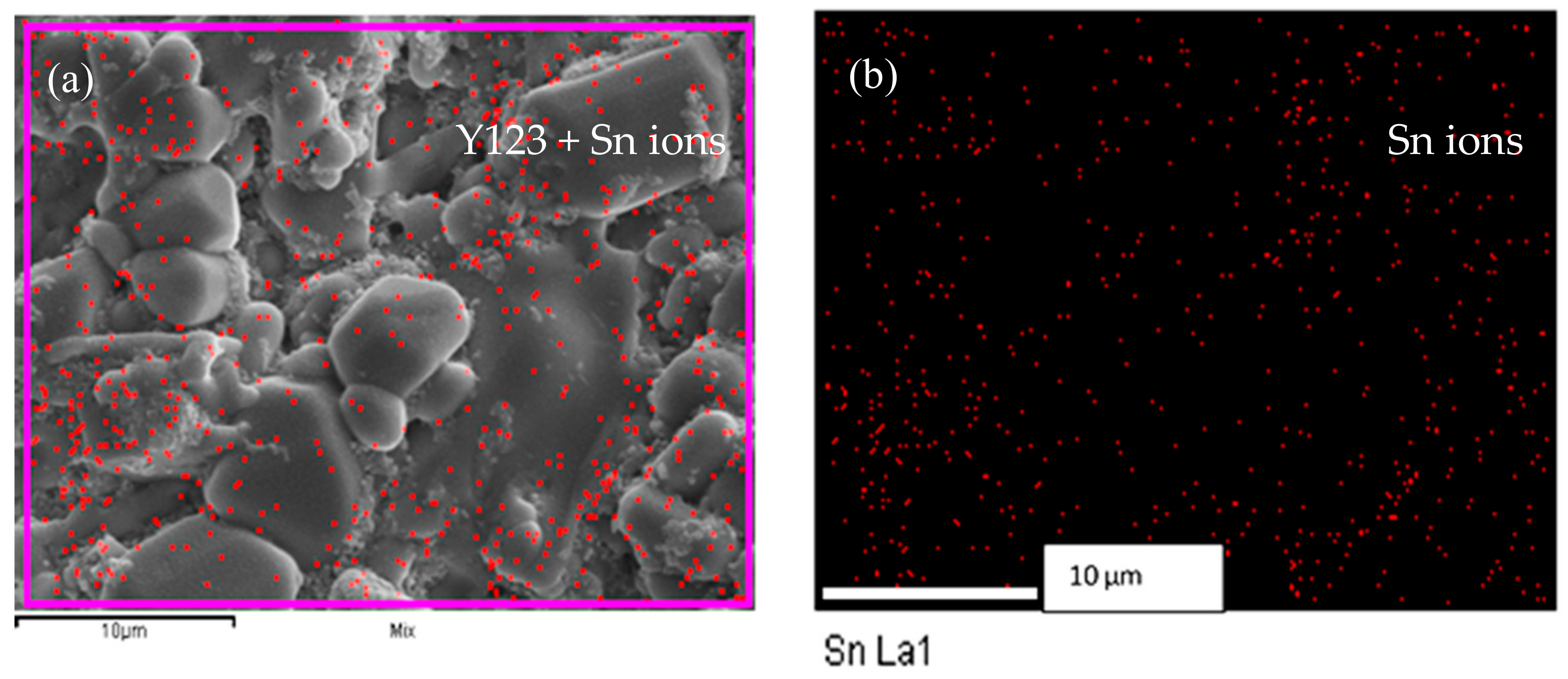
3.2. Scanning Electron Microscopy and Elemental Analysis
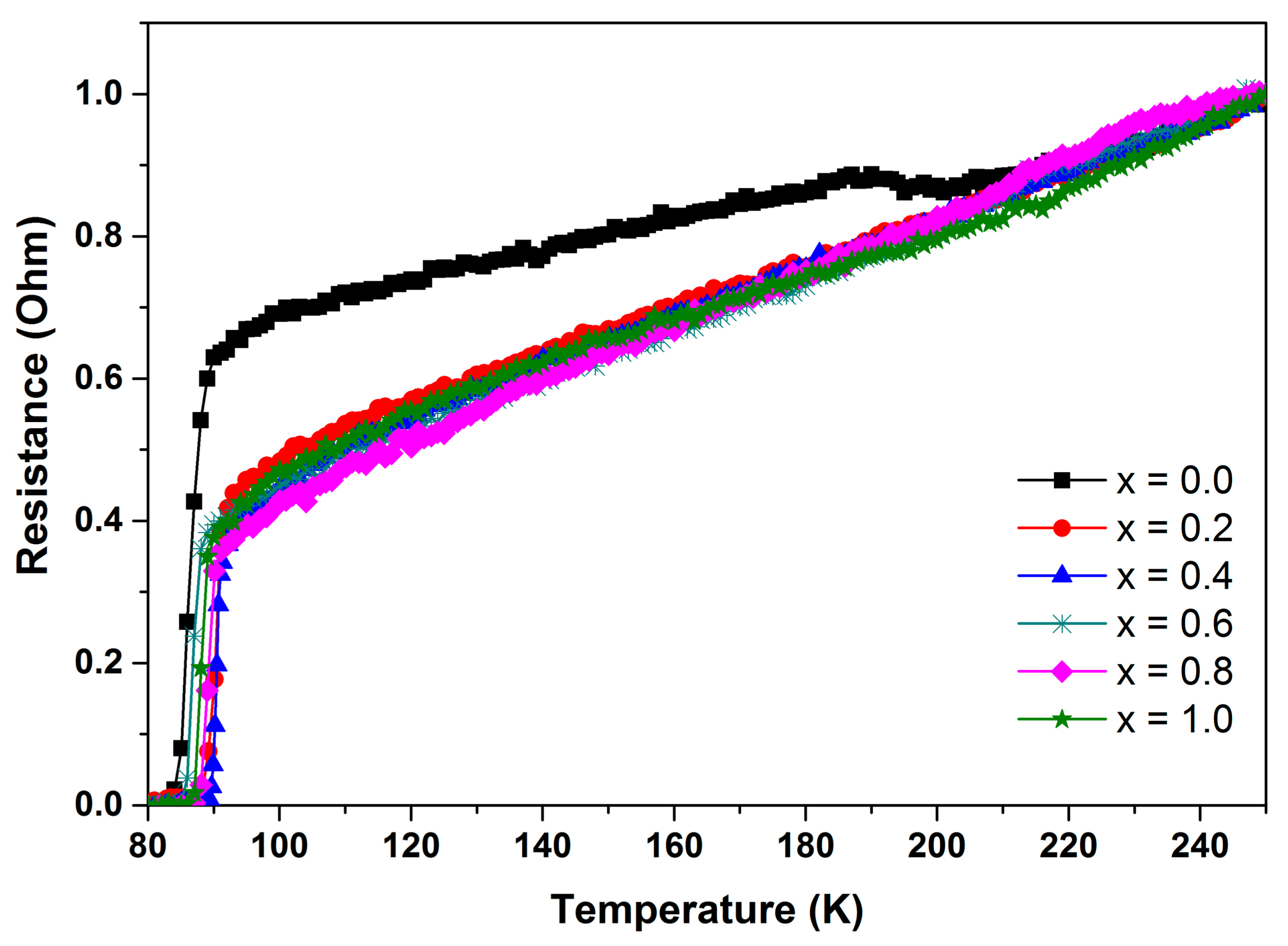
3.3. Electrical Resistance Measurement
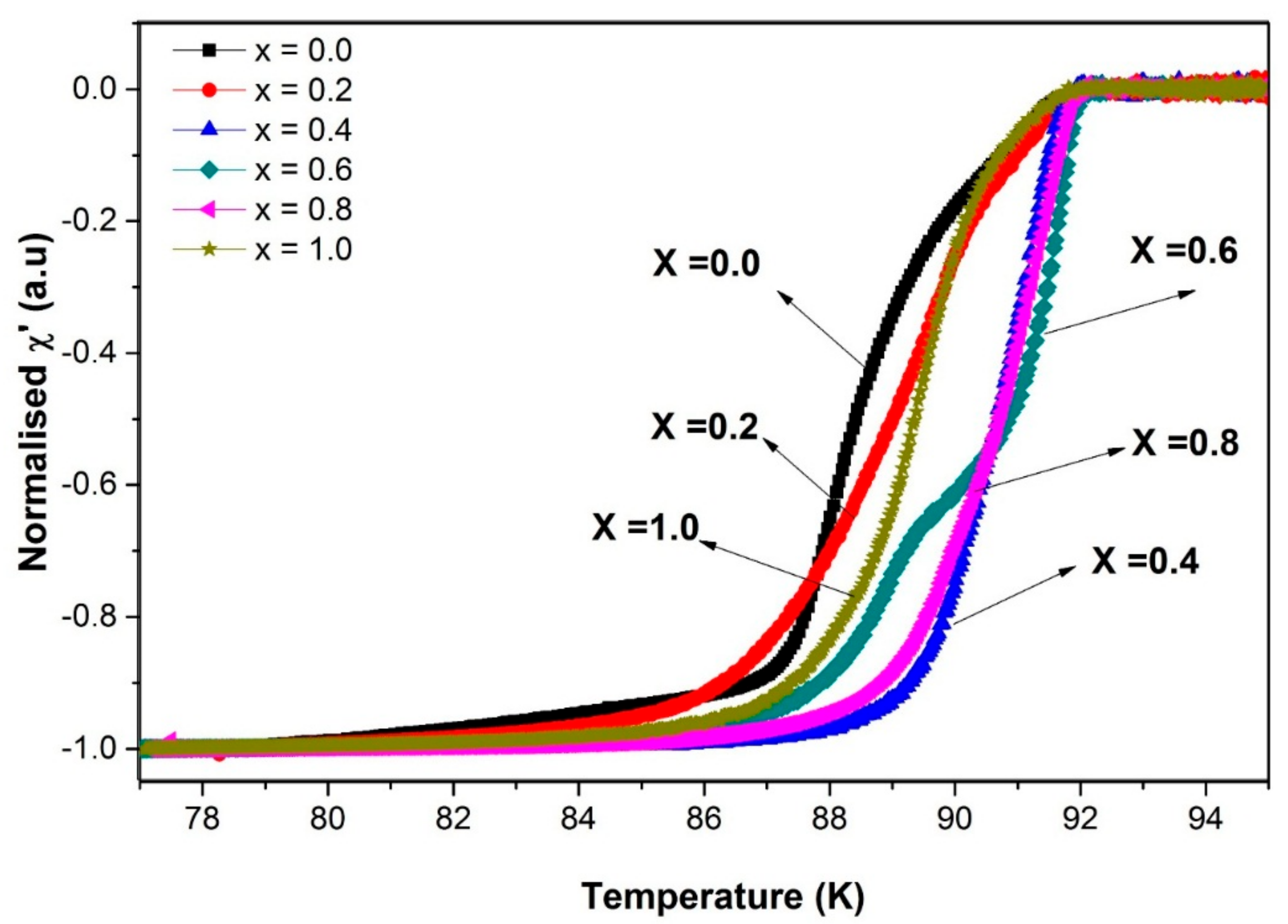
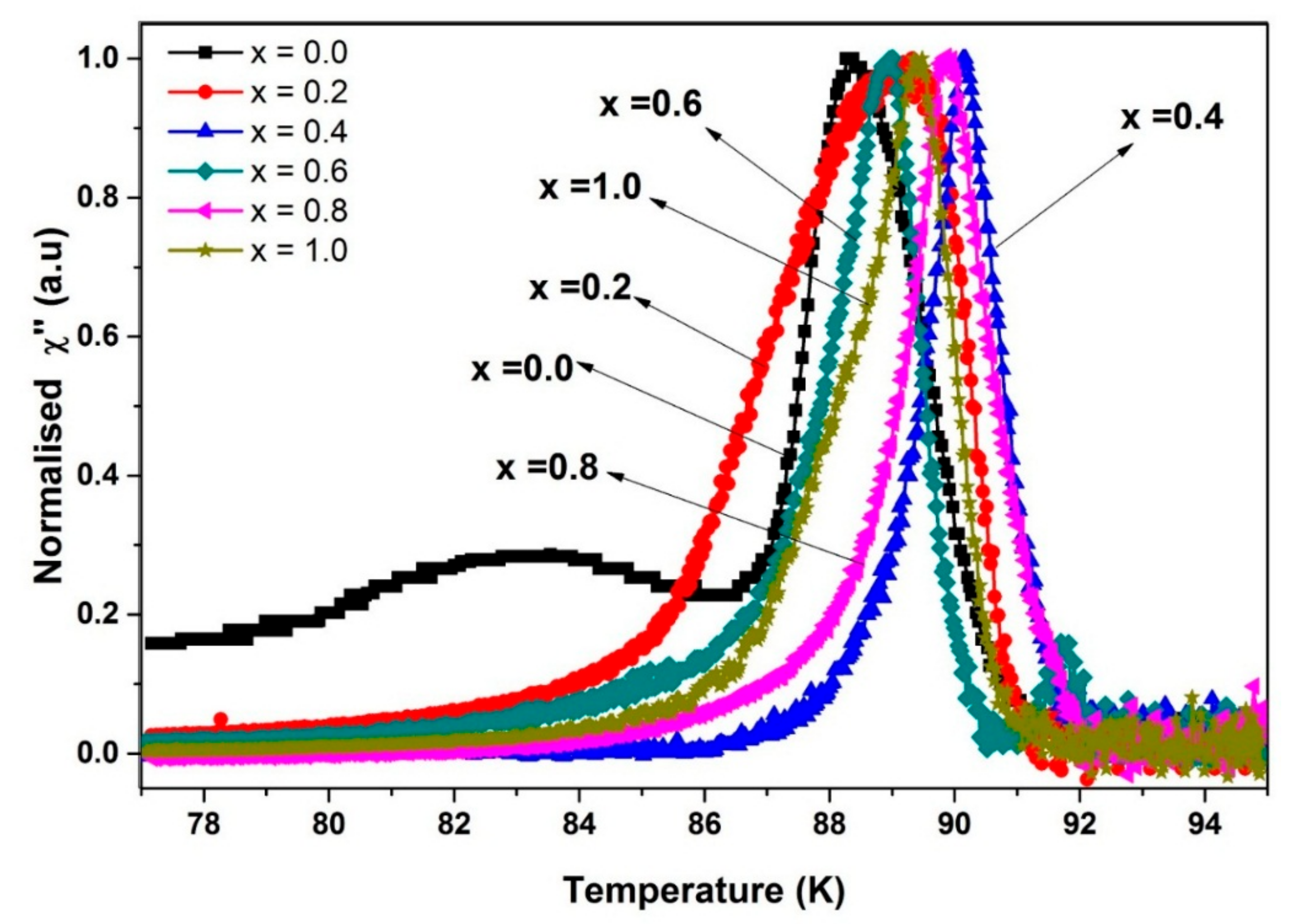
3.4. Alternating Current Susceptibility
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hott, R.; Kleiner, R.; Wolf, T.; Zwicknagl, G. Review on superconducting materials. In Digital Encyclopedia of Applied Physics; American Cancer Society: Atlanta, GA, USA, 2016; pp. 1–55. [Google Scholar]
- Haugan, T.; Barnes, P.N.; Wheeler, R.; Meisenkothen, F.; Sumption, M.; Melsenkothen, F.; Sumption, M. Addition of nanoparticle dispersions to enhance flux pinning of the YBa2Cu3O7−x superconductor. Nature 2004, 430, 867–870. [Google Scholar] [CrossRef] [PubMed]
- Varma, C. High-temperature superconductivity: Mind the pseudogap. Nature 2010, 468, 184–185. [Google Scholar] [CrossRef] [PubMed]
- Klemm, R.A. (Ed.) Layered Superconductors; Oxford University Press: Oxford, UK, 2012. [Google Scholar]
- Rejith, P.P.; Vidya, S.; Vipin, L.; Solomon, S.; Thomas, J.K. Flux-pinning properties of nanocrystalline HfO2 added YBa2Cu3O7−δ superconductor. Phys. Status Solidi B 2014, 251, 809–814. [Google Scholar] [CrossRef]
- Öztürk, A.; Düzgün, I.İ.; Çelebi, S. The effect of partial Lu doping on magnetic behaviour of YBCO (123) superconductors. J. Alloys Compd. 2010, 495, 104–107. [Google Scholar] [CrossRef]
- Dihom, M.M.; Shaari, A.H.; Baqiah, H.; Al-Hada, N.M.; Talib, Z.A.; Kien, C.S.; Azis, R.S.; Kechik, M.M.A.; Pah, L.K.; Abd-Shukor, R. Structural and superconducting properties of Y(Ba1−xKx)2Cu3O7−δ ceramics. Ceram. Int. 2017, 43, 11339–11344. [Google Scholar] [CrossRef]
- Naseri, M.G.; Saion, E.B.; Ahangar, H.A.; Shaari, A.H.; Hashim, M. Simple synthesis and characterization of cobalt ferrite nanoparticles by a thermal treatment method. J. Nanomater. 2010, 2010, 8. [Google Scholar] [CrossRef]
- Naseri, M.G.; Saion, E.B.; Ahangar, H.A.; Hashim, M.; Shaari, A.H. Synthesis and characterization of manganese ferrite nanoparticles by thermal treatment method. J. Magn. Magn. Mater. 2011, 323, 1745–1749. [Google Scholar] [CrossRef]
- Khalilzadeh, N.; Saion, E.B.; Mirabolghasemi, H.; Crouse, K.A.; Shaari, A.H.B.; Hashim, M.B. Preparation and characterization of ultrafine nanoparticles of Cu doped lithium tetraborate. Results Phys. 2015, 5, 324–330. [Google Scholar] [CrossRef]
- Guner, S.B.; Gorur, O.; Celik, S.; Dogruer, M.; Yildirim, G.; Varilci, A.; Terzioglu, C. Effect of zirconium diffusion on the microstructural and superconducting properties of YBa2Cu3O7−δ superconductors. J. Alloys Compd. 2012, 540, 260–266. [Google Scholar] [CrossRef]
- Gupta, S.; Yadav, R.S.; Das, B. Flux Pinning by Nano Particles Embedded in Polycrystalline Y-123 Superconductors. ISST J. Appl. Phys. 2011, 2, 1–5. [Google Scholar]
- Jin, L.H.; Zhang, S.N.; Yu, Z.M.; Li, C.S.; Feng, J.Q.; Sulpice, A.; Wang, Y.; Zhang, P.X. Influences of BaZrO3 particles on the microstructure and flux pinning of YBCO film prepared by using modified TFA-MOD approach. Mater. Chem. Phys. 2015, 149–150, 188–192. [Google Scholar] [CrossRef]
- Khalid, N.A.; Kechik, M.M.A.; Baharuddin, N.A.; Kien, C.S.; Baqiah, H.; Yusuf, N.N.M.; Shaari, A.H.; Hashim, A.; Talib, Z.A. Impact of carbon nanotubes addition on transport and superconducting properties of YBa2Cu3O7−δ ceramics. Ceram. Int. 2018, 44, 9568–9573. [Google Scholar] [CrossRef]
- Ramli, A.; Shaari, A.H.; Baqiah, H.; Kean, C.S.; Kechik, M.M.A.; Talib, Z.A. Role of Nd2O3 nanoparticles addition on microstructural and superconducting properties of YBa2Cu3O7−δ ceramics. J. Rare Earths 2016, 34, 895–900. [Google Scholar] [CrossRef]
- Rejith, P.P.; Vidya, S.; Thomas, J.K. Improvement of Critical Current Density in YBa2Cu3O7−δ Superconductor with Nano TiO2 Addition. Mater. Mater. Today Proc. 2015, 2, 997–1001. [Google Scholar] [CrossRef]
- Teranishi, R.; Miyanaga, Y.; Yamada, K.; Mori, N.; Mukaida, M.; Miura, M.; Yoshizumi, M.; Izumi, T.; Namba, M.; Awaji, S.; et al. Effects of tin-compounds addition on Jcand microstructure for YBCO films. Phys. C Supercond. Its Appl. 2010, 470, 1246–1248. [Google Scholar] [CrossRef]
- Choi, S.M.; Shin, G.M.; Yoo, S.I. Flux pinning characteristics of Sn-doped YBCO film by the MOD process. Phys. C Supercond. Its Appl. 2013, 485, 154–159. [Google Scholar] [CrossRef]
- Miyanaga, Y.; Teranishi, R.; Yamada, K.; Mori, N.; Mukaida, M.; Kiss, T.; Inoue, M.; Nakaoka, K.; Yoshizumi, M.; Izumi, T.; et al. Effects of Sn-doping on JC-B properties and crystalline structure for YBCO films by advanced TFA-MOD method. Phys. C Supercond. Its Appl. 2009, 469, 15–20. [Google Scholar] [CrossRef]
- He, Z.H.; Habisreuther, T.; Bruchlos, G.; Litzkendorf, D.; Gawalek, W. Investigation of microstructure of textured YBCO with addition of nanopowder SnO2. Phys. C Supercond. Its Appl. 2001, 356, 277–284. [Google Scholar] [CrossRef]
- Bhargava, A.; Mackinnon, I.D.R.; Yamashita, T.; Page, D. Bulk manufacture of YBCO powders by coprecipitation. Phys. C Supercond. 1995, 241, 53–62. [Google Scholar] [CrossRef]
- Teranishi, R.; Miyanaga, Y.; Yamada, K.; Mori, N.; Mukaida, M.; Inoue, M.; Kiss, T.; Miura, M.; Yoshizumi, M.; Izumi, T.; et al. Doping of Tin-oxides pinning centers into YBCO films by MOD method. J. Phys. Conf. Ser. 2010, 234, 22039. [Google Scholar] [CrossRef]
- Salama, A.H.; El-Hofy, M.; Rammah, Y.S.; Elkhatib, M. Effect of magnetic and nonmagnetic nano metal oxides doping on the critical temperature of a YBCO superconductor. Adv. Nat. Sci. Nanosci. Nanotechnol. 2015, 6. [Google Scholar] [CrossRef]
- Girgsdies, F. Peak Profile Analysis in X-ray Powder Diffraction; Fritz-Haber-Institut der MPG: Berlin, Germany, 2015. [Google Scholar]
- Dihom, M.M.; Shaari, A.H.; Baqiah, H.; Al-Hada, N.M.; Kien, C.S.; Azis, R.S.; Kechik, M.M.A.; Talib, Z.A.; Abd-Shukor, R. Microstructure and superconducting properties of Ca substituted Y(Ba1−xCax)2Cu3O7−δ ceramics prepared by thermal treatment method. Results Phys. 2017, 7, 407–412. [Google Scholar] [CrossRef]
- Vanderbemden, P.; Bradley, A.D.; Doyle, R.A.; Lo, W.; Astill, D.M.; Cardwell, D.A.; Campbell, A.M. Superconducting properties of natural and artificial grain boundaries in bulk melt-textured YBCO. Phys. C Supercond. 1998, 302, 257–270. [Google Scholar] [CrossRef]
- Hannachi, E.; Slimani, Y.; Ben Azzouz, F.; Ekicibil, A. Higher intra-granular and inter-granular performances of YBCO superconductor with TiO2 nano-sized particles addition. Ceram. Int. 2018, 44, 18836–18843. [Google Scholar] [CrossRef]
- Dew-Hughes, D. The critical current of superconductors: An historical review. Low Temp. Phys. 2001, 27, 967–979. [Google Scholar] [CrossRef]
- Salamati, H.; Kameli, P. Effect of deoxygenation on the weak-link behavior of YBa2Cu3O7−δ superconductors. Solid State Commun. 2003, 125, 407–411. [Google Scholar] [CrossRef]
- Sarmago, R.V.; Singidas, B.G. Low field AC susceptibility of YBCO: The frequency and field dependence of intra- and intergrain coupling losses in the absence of vortices. Supercond. Sci. Technol. 2004, 17, S578. [Google Scholar] [CrossRef]
- Clem, J.R. Granular and superconducting-glass properties of the high-temperature superconductors. Phys. C Supercond. 1988, 153–155, 50–55. [Google Scholar] [CrossRef]
- Kechik, A.; Mustafa, M. Improvement of Critical Current Density in YBa2Cu3O7−δ Films with Nano-Inclusions. Ph.D. Thesis, University of Birmingham, Birmingham, UK, 2011. [Google Scholar]
- Deguchi, K.; Tanatar, M.A.; Mao, Z.; Ishiguro, T.; Maeno, Y. Superconducting Double Transition and the Upper Critical Field Limit of Sr2RuO4 in Parallel Magnetic Fields. J. Phys. Soc. Jpn. 2002, 71, 2839–2842. [Google Scholar] [CrossRef]
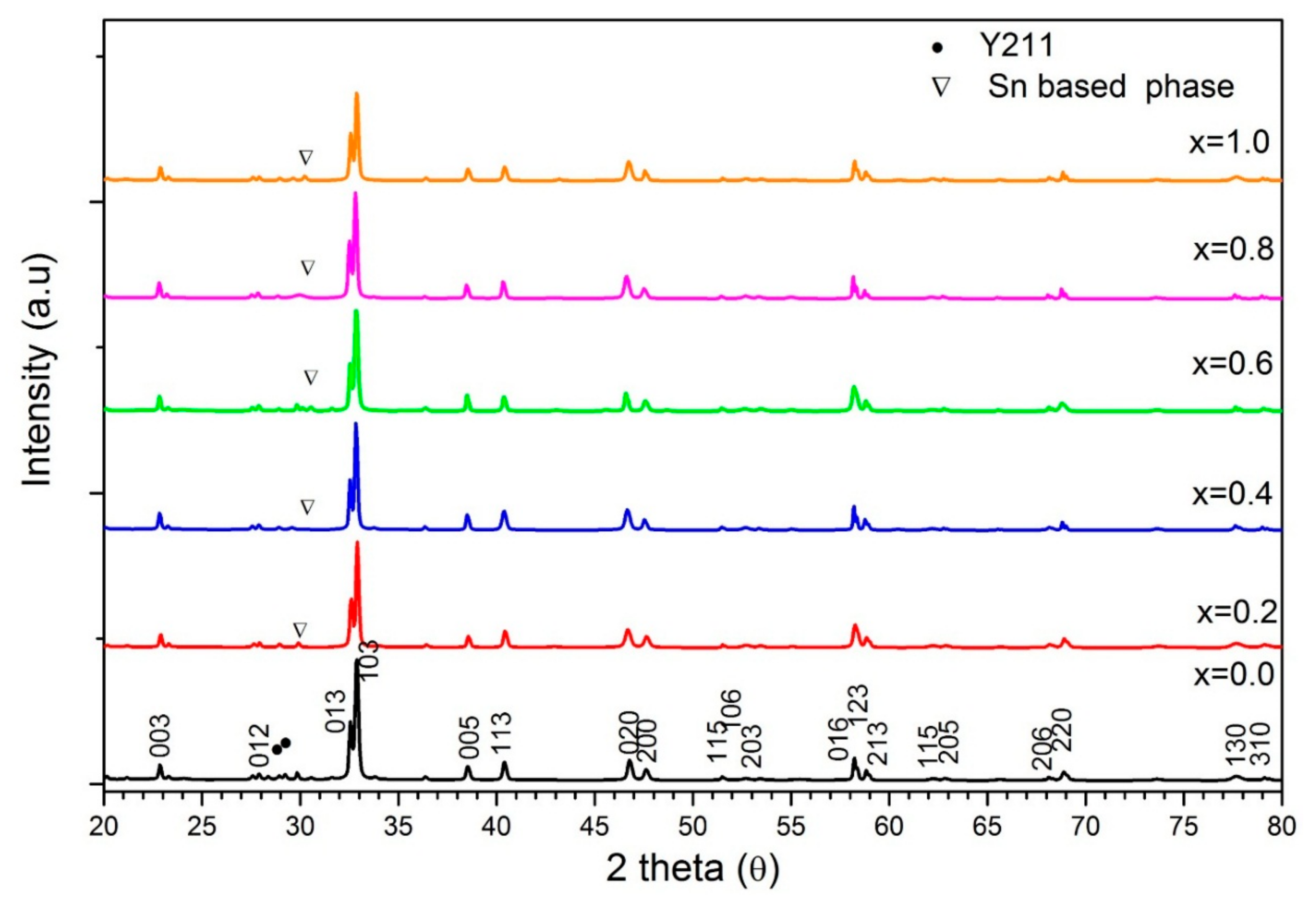


| SnO2 Addition (x = wt.%) | Y123% | Y211% | Sn Based Phase% | Orthorhombicity (10−3) | Crystallite Size (nm) | |||
|---|---|---|---|---|---|---|---|---|
| 0.0 | 91.5 | 8.5 | - | 3.8201(2) | 3.8846(3) | 11.676(1) | 8.372 | 91 ± 11 |
| 0.02 | 94.2 | 1.8 | 4.0 | 3.8211 | 3.8842 | 11.6760 | 8.189 | 100 ± 28 |
| 0.4 | 95.8 | 0.1 | 4.2 | 3.8232(1) | 3.8862(2) | 11.6793(7) | 8.185 | 98 ± 28 |
| 0.06 | 91.5 | - | 8.5 | 3.8239 | 3.8864 | 11.6840 | 8.067 | 170 ± 65 |
| 0.8 | 93.8 | - | 6.2 | 3.8218(1) | 3.8864(2) | 11.6740(8) | 8.381 | 110 ± 26 |
| 1.0 | 94.0 | - | 6.0 | 3.8863(2) | 11.6793(8) | 8.185 | 95 ± 20 |
| SnO2 Addition (x = wt.%) | Tc-onset (K) | Tc-offset (K) | ∆Tc (K) | Ref. |
|---|---|---|---|---|
| 0.0 | 90 | 83 | 7 | This work |
| 0.2 | 92 | 88 | 4 | This work |
| 0.4 | 91 | 88 | 3 | This work |
| 0.6 | 89 | 85 | 4 | This work |
| 0.8 | 90 | 86 | 4 | This work |
| 1.0 | 90 | 86 | 4 | This work |
| 0.0 | 87 | 92 | 5 | [23] |
| 0.2 | 67 | 79 | 12 | [23] |
| SnO2 Addition (x = wt.%) | Hac (Oe) | Tc-onset (K) | Tcj (K) | Tp (K) | I0 (μA) |
|---|---|---|---|---|---|
| 0.0 | 1 | 91.5 | 90.6 | 88.3 | 146 |
| 0.2 | 1 | 91.7 | 90.9 | 89.3 | 147 |
| 0.4 | 1 | 92.0 | 91.8 | 90.2 | 664 |
| 0.6 | 1 | 92.0 | 90.1 | 88.9 | 70 |
| 0.8 | 1 | 92.0 | 90.8 | 89.9 | 111 |
| 1.0 | 1 | 91.8 | 91.2 | 89.4 | 189 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mohd Yusuf, N.N.; Awang Kechik, M.M.; Baqiah, H.; Soo Kien, C.; Kean Pah, L.; Shaari, A.H.; Wan Jusoh, W.N.W.; Abd Sukor, S.I.; Mousa Dihom, M.; Talib, Z.A.; et al. Structural and Superconducting Properties of Thermal Treatment-Synthesised Bulk YBa2Cu3O7−δ Superconductor: Effect of Addition of SnO2 Nanoparticles. Materials 2019, 12, 92. https://doi.org/10.3390/ma12010092
Mohd Yusuf NN, Awang Kechik MM, Baqiah H, Soo Kien C, Kean Pah L, Shaari AH, Wan Jusoh WNW, Abd Sukor SI, Mousa Dihom M, Talib ZA, et al. Structural and Superconducting Properties of Thermal Treatment-Synthesised Bulk YBa2Cu3O7−δ Superconductor: Effect of Addition of SnO2 Nanoparticles. Materials. 2019; 12(1):92. https://doi.org/10.3390/ma12010092
Chicago/Turabian StyleMohd Yusuf, Nur Nabilah, Mohd Mustafa Awang Kechik, Hussein Baqiah, Chen Soo Kien, Lim Kean Pah, Abdul Halim Shaari, Wan Nur Wathiq Wan Jusoh, Safia Izzati Abd Sukor, Mustafa Mousa Dihom, Zainal Abidin Talib, and et al. 2019. "Structural and Superconducting Properties of Thermal Treatment-Synthesised Bulk YBa2Cu3O7−δ Superconductor: Effect of Addition of SnO2 Nanoparticles" Materials 12, no. 1: 92. https://doi.org/10.3390/ma12010092
APA StyleMohd Yusuf, N. N., Awang Kechik, M. M., Baqiah, H., Soo Kien, C., Kean Pah, L., Shaari, A. H., Wan Jusoh, W. N. W., Abd Sukor, S. I., Mousa Dihom, M., Talib, Z. A., & Abd-Shukor, R. (2019). Structural and Superconducting Properties of Thermal Treatment-Synthesised Bulk YBa2Cu3O7−δ Superconductor: Effect of Addition of SnO2 Nanoparticles. Materials, 12(1), 92. https://doi.org/10.3390/ma12010092





